Cytotoxicity and Thermal Characterization Assessment of Excipients for the Development of Innovative Lyophilized Formulations for Oncological Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Formulations to Be Lyophilized
2.2. Thermal Characterization
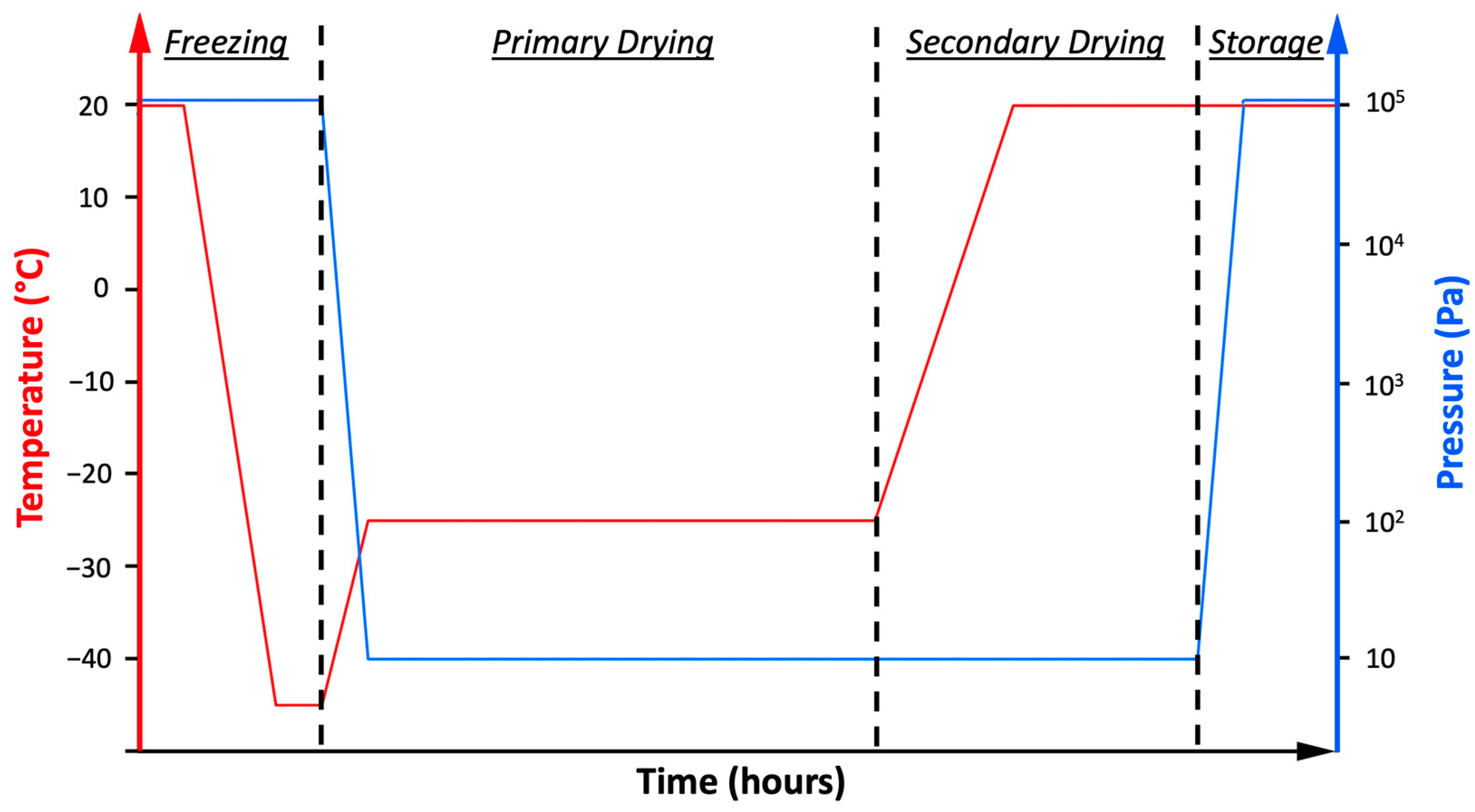
2.3. Freeze-Drying Process
2.4. Freeze-Dried Samples Evaluation
2.5. Cell Cultures
2.6. Cytotoxicity Assay
2.7. Statistical Analysis
3. Results
3.1. Thermal Characterization
3.2. Freeze-Dried Samples Evaluation
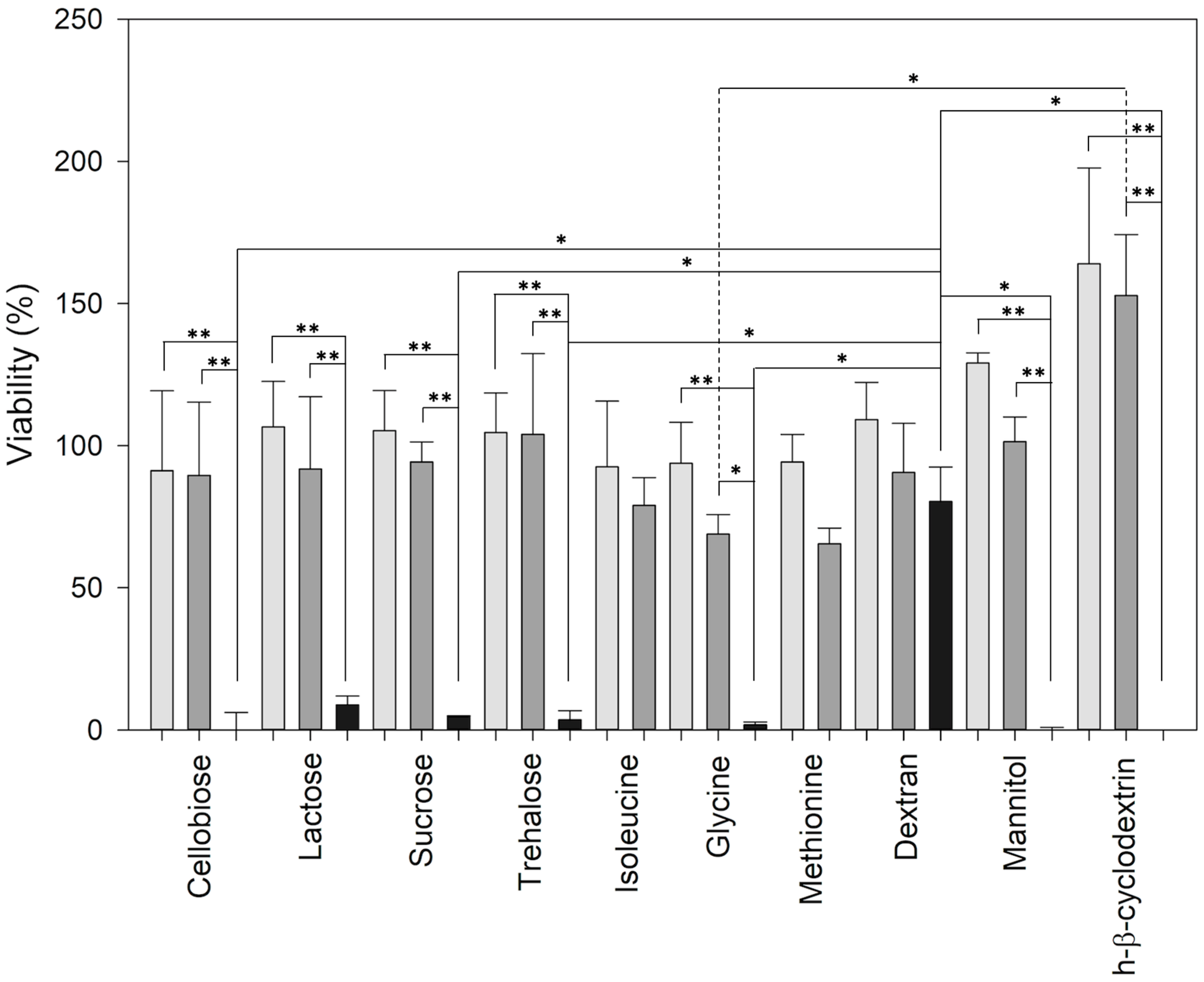
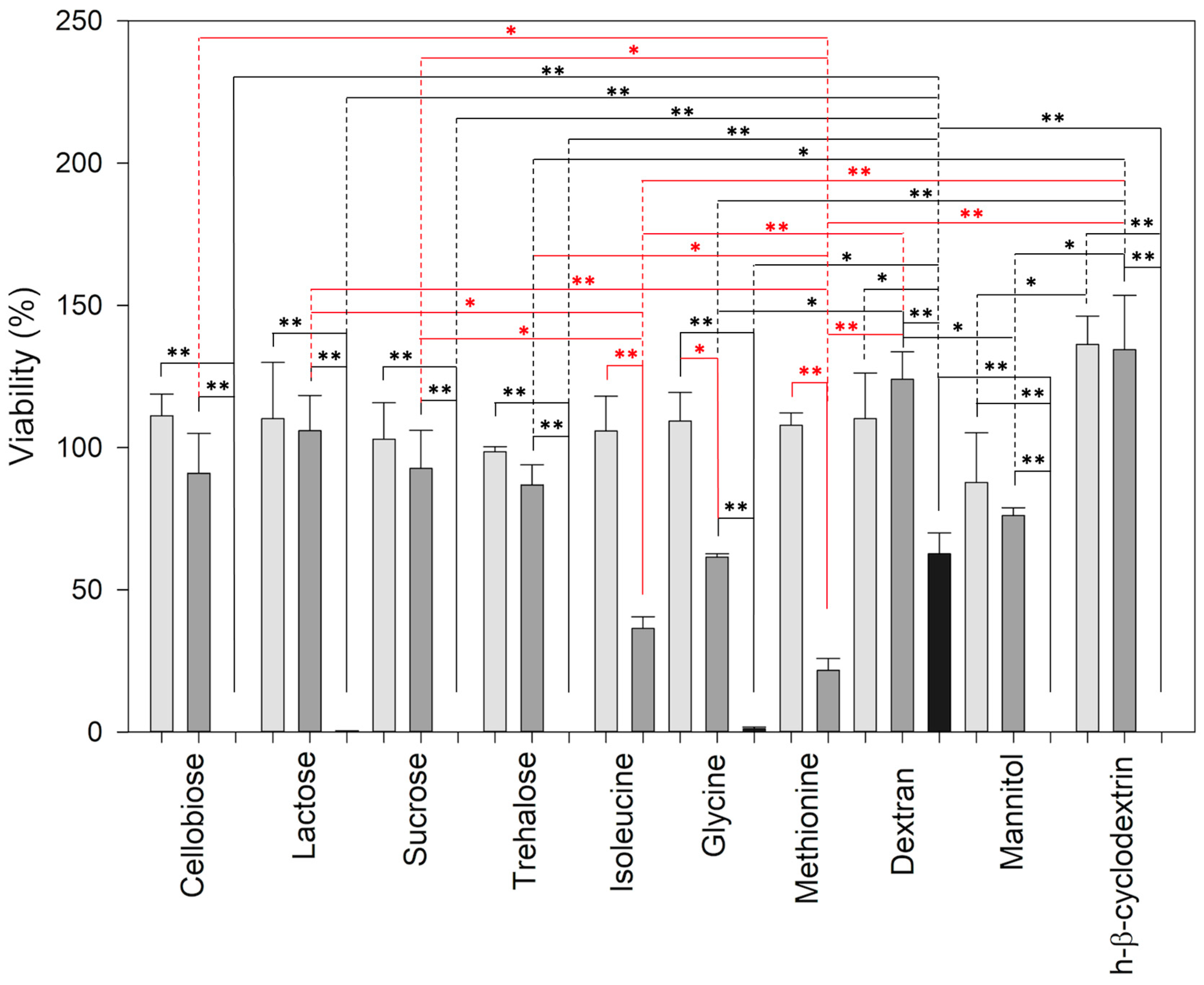
3.3. Cytotoxicity Assay Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kesik-Brodacka, M. Progress in biopharmaceutical development. Biotechnol. Appl. Biochem. 2018, 65, 306–322. [Google Scholar] [CrossRef] [PubMed]
- Zeb, A.; Rana, I.; Choi, H.I.; Lee, C.H.; Baek, S.W.; Lim, C.W.; Khan, N.; Arif, S.T.; Sahar, N.U.; Alvi, A.M.; et al. Potential and Applications of Nanocarriers for Efficient Delivery of Biopharmaceuticals. Pharmaceutics 2020, 12, 1184. [Google Scholar] [CrossRef] [PubMed]
- Bjelošević, M.; Zvonar Pobirk, A.; Planinšek, O.; Ahlin Grabnar, P. Excipients in freeze-dried biopharmaceuticals: Contributions toward formulation stability and lyophilisation cycle optimisation. Int. J. Pharm. 2020, 576, 119029. [Google Scholar] [CrossRef] [PubMed]
- Emami, F.; Vatanara, A.; Park, E.J.; Na, D.H. Drying Technologies for the Stability and Bioavailability of Biopharmaceuticals. Pharmaceutics 2018, 10, 131. [Google Scholar] [CrossRef]
- Arsiccio, A.; Paladini, A.; Pattarino, F.; Pisano, R. Designing the Optimal Formulation for Biopharmaceuticals: A New Approach Combining Molecular Dynamics and Experiments. J. Pharm. Sci. 2019, 108, 431–438. [Google Scholar] [CrossRef]
- Khairnar, S.; Kini, R.; Harwalkar, M.; Salunkhe, K.; Chaudhari, S. A Review on Freeze Drying Process of Pharmaceuticals. Int. J. Res. Pharm. Sci. 2012, 2013, 76–94. [Google Scholar]
- Bahr, M.M.; Amer, M.S.; Abo-El-Sooud, K.; Abdallah, A.N.; El-Tookhy, O.S. Preservation techniques of stem cells extracellular vesicles: A gate for manufacturing of clinical grade therapeutic extracellular vesicles and long-term clinical trials. Int. J. Vet. Sci. Med. 2020, 8, 1–8. [Google Scholar] [CrossRef]
- Chang, L.; Shepherd, D.; Sun, J.; Ouellette, D.; Grant, K.L.; Tang, X.C.; Pikal, M.J. Mechanism of protein stabilization by sugars during freeze-drying and storage: Native structure preservation, specific interaction, and/or immobilization in a glassy matrix? J. Pharm. Sci. 2005, 94, 1427–1444. [Google Scholar] [CrossRef]
- Tang, X.; Pikal, M.J. Design of freeze-drying processes for pharmaceuticals: Practical advice. Pharm. Res. 2004, 21, 191–200. [Google Scholar] [CrossRef]
- Rayaprolu, B.M.; Strawser, J.J.; Anyarambhatla, G. Excipients in parenteral formulations: Selection considerations and effective utilization with small molecules and biologics. Drug Dev. Ind. Pharm. 2018, 44, 1565–1571. [Google Scholar] [CrossRef]
- Medi, M.B.; Chintala, R.; Bhambhani, A. Excipient selection in biologics and vaccines formulation development. Eur. Pharm. Rev. 2014, 19, 16–20. [Google Scholar]
- Chaudhari, S.P.; Patil, P.S. Pharmaceutical excipients: A review. Int. J. Adv. Pharm. Biol. Chem. 2012, 1, 21–34. [Google Scholar]
- Pifferi, G.; Restani, P. The safety of pharmaceutical excipients. Il Farm. 2003, 58, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Osterberg, R.E.; See, N.A. Toxicity of excipients—A Food and Drug Administration perspective. Int. J. Toxicol. 2003, 22, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Kiss, L.; Walter, F.R.; Bocsik, A.; Veszelka, S.; Ozsvári, B.; Puskás, L.G.; Szabó-Révész, P.; Deli, M.A. Kinetic analysis of the toxicity of pharmaceutical excipients Cremophor EL and RH40 on endothelial and epithelial cells. J. Pharm. Sci. 2013, 102, 1173–1181. [Google Scholar] [CrossRef]
- Dai, Q.; Liu, X.; He, T.; Yang, C.; Jiang, J.; Fang, Y.; Fu, Z.; Yuan, Y.; Bai, S.; Qiu, T.; et al. Excipient of paclitaxel induces metabolic dysregulation and unfolded protein response. iScience 2021, 24, 103170. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, R.; Chong, L.B.; Zou, L.; Tsakalozou, E.; Ni, Z.; Giacomini, K.M.; Kroetz, D.L. Interaction of Commonly Used Oral Molecular Excipients with P-glycoprotein. AAPS J. 2021, 23, 106. [Google Scholar] [CrossRef]
- Belayneh, A.; Tadese, E.; Molla, F. Safety and Biopharmaceutical Challenges of Excipients in Off-Label Pediatric Formulations. Int. J. Gen. Med. 2020, 13, 1051–1066. [Google Scholar] [CrossRef]
- Rouaz, K.; Chiclana-Rodríguez, B.; Nardi-Ricart, A.; Suñé-Pou, M.; Mercadé-Frutos, D.; Suñé-Negre, J.M.; Pérez-Lozano, P.; García-Montoya, E. Excipients in the Paediatric Population: A Review. Pharmaceutics 2021, 13, 387. [Google Scholar] [CrossRef]
- Schmitt, G. Safety of Excipients in Pediatric Formulations—A Call for Toxicity Studies in Juvenile Animals? Children 2015, 2, 191–197. [Google Scholar] [CrossRef]
- Valeur, K.S.; Holst, H.; Allegaert, K. Excipients in Neonatal Medicinal Products: Never Prescribed, Commonly Administered. Pharmaceut. Med. 2018, 32, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Pottel, J.; Armstrong, D.; Zou, L.; Fekete, A.; Huang, X.P.; Torosyan, H.; Bednarczyk, D.; Whitebread, S.; Bhhatarai, B.; Liang, G.; et al. The activities of drug inactive ingredients on biological targets. Science 2020, 369, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Horváth, T.; Bartos, C.; Bocsik, A.; Kiss, L.; Veszelka, S.; Deli, M.A.; Újhelyi, G.; Szabó-Révész, P.; Ambrus, R. Cytotoxicity of Different Excipients on RPMI 2650 Human Nasal Epithelial Cells. Molecules 2016, 21, 658. [Google Scholar] [CrossRef] [PubMed]
- Nemes, D.; Kovács, R.; Nagy, F.; Mező, M.; Poczok, N.; Ujhelyi, Z.; Pető, Á.; Fehér, P.; Fenyvesi, F.; Váradi, J.; et al. Interaction between Different Pharmaceutical Excipients in Liquid Dosage Forms—Assessment of Cytotoxicity and Antimicrobial Activity. Molecules 2018, 23, 1827. [Google Scholar] [CrossRef] [PubMed]
- Bieberich, A.A.; Rajwa, B.; Irvine, A.; Fatig, R.O., 3rd; Fekete, A.; Jin, H.; Kutlina, E.; Urban, L. Acute cell stress screen with supervised machine learning predicts cytotoxicity of excipients. J. Pharmacol. Toxicol. Methods 2021, 111, 107088. [Google Scholar] [CrossRef]
- Mi, X.; Shukla, D. Predicting the Activities of Drug Excipients on Biological Targets using One-Shot Learning. J. Phys. Chem. B 2022, 126, 1492–1503. [Google Scholar] [CrossRef]
- Arsiccio, A.; Pisano, R. Clarifying the role of cryo- and lyo-protectants in the biopreservation of proteins. Phys. Chem. Chem. Phys. 2018, 20, 8267–8277. [Google Scholar] [CrossRef]
- Ohtake, S.; Kita, Y.; Arakawa, T. Interactions of formulation excipients with proteins in solution and in the dried state. Adv. Drug Deliv. Rev. 2011, 63, 1053–1073. [Google Scholar] [CrossRef]
- Grasmeijer, N.; Stankovic, M.; de Waard, H.; Frijlink, H.W.; Hinrichs, W.L. Unraveling protein stabilization mechanisms: Vitrification and water replacement in a glass transition temperature controlled system. Biochim. Biophys Acta 2013, 1834, 763–769. [Google Scholar] [CrossRef]
- Toniolo, S.P.; Afkhami, S.; Mahmood, A.; Fradin, C.; Lichty, B.D.; Miller, M.S.; Xing, Z.; Cranston, E.D.; Thompson, M.R. Excipient selection for thermally stable enveloped and non-enveloped viral vaccine platforms in dry powders. Int. J. Pharm. 2019, 561, 66–73. [Google Scholar] [CrossRef]
- Susa, F.; Bucca, G.; Limongi, T.; Cauda, V.; Pisano, R. Enhancing the preservation of liposomes: The role of cryoprotectants, lipid formulations and freezing approaches. Cryobiology 2021, 98, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Arsiccio, A.; Pisano, R. Water entrapment and structure ordering as protection mechanisms for protein structural preservation. J. Chem. Phys. 2018, 148, 055102. [Google Scholar] [CrossRef] [PubMed]
- Horn, J.; Tolardo, E.; Fissore, D.; Friess, W. Crystallizing amino acids as bulking agents in freeze-drying. Eur. J. Pharm. Biopharm. 2018, 132, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Larsen, B.S.; Skytte, J.; Svagan, A.J.; Meng-Lund, H.; Grohganz, H.; Löbmann, K. Using dextran of different molecular weights to achieve faster freeze-drying and improved storage stability of lactate dehydrogenase. Pharm. Dev. Technol. 2019, 24, 323–328. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration: Inactive Ingredient Search for Approved Drug Products U.S. Federal Government Regulatory Agency. Available online: https://www.accessdata.fda.gov/scripts/cder/iig/ (accessed on 7 July 2022).
- Ionova, Y.; Wilson, L. Biologic excipients: Importance of clinical awareness of inactive ingredients. PLoS ONE 2020, 15, e0235076. [Google Scholar] [CrossRef] [PubMed]
- Rao, V.A.; Kim, J.J.; Patel, D.S.; Rains, K.; Estoll, C.R. A Comprehensive Scientific Survey of Excipients Used in Currently Marketed, Therapeutic Biological Drug Products. Pharm. Res. 2020, 37, 200. [Google Scholar] [CrossRef]
- Rycerz, L. Practical remarks concerning phase diagrams determination on the basis of differential scanning calorimetry measurements. J. Therm. Anal. Calorim. 2013, 113, 231–238. [Google Scholar] [CrossRef]
- Horn, J.; Friess, W. Detection of Collapse and Crystallization of Saccharide, Protein, and Mannitol Formulations by Optical Fibers in Lyophilization. Front. Chem. 2018, 6, 4. [Google Scholar] [CrossRef]
- Ray, P.; Rielly, C.D.; Stapley, A.G.F. A freeze-drying microscopy study of the kinetics of sublimation in a model lactose system. Chem. Eng. Sci. 2017, 172, 731–743. [Google Scholar] [CrossRef]
- Pisano, R. Automatic control of a freeze-drying process: Detection of the end point of primary drying. Dry. Technol. 2022, 40, 140–157. [Google Scholar] [CrossRef]
- Patel, S.M.; Nail, S.L.; Pikal, M.J.; Geidobler, R.; Winter, G.; Hawe, A.; Davagnino, J.; Rambhatla Gupta, S. Lyophilized Drug Product Cake Appearance: What Is Acceptable? J. Pharm. Sci. 2017, 106, 1706–1721. [Google Scholar] [CrossRef] [PubMed]
- Bjelošević, M.; Seljak, K.B.; Trstenjak, U.; Logar, M.; Brus, B.; Ahlin Grabnar, P. Aggressive conditions during primary drying as a contemporary approach to optimise freeze-drying cycles of biopharmaceuticals. Eur. J. Pharm. Sci. 2018, 122, 292–302. [Google Scholar] [CrossRef]
- Ward, K.R.; Matejtschuk, P. Characterization of Formulations for Freeze-Drying. In Lyophilization of Pharmaceuticals and Biologicals: New Technologies and Approaches; Ward, K.R., Matejtschuk, P., Eds.; Springer: New York, NY, USA, 2019; pp. 1–32. [Google Scholar]
- Kim, A.I.; Akers, M.J.; Nail, S.L. The Physical State of Mannitol after Freeze-Drying: Effects of Mannitol Concentration, Freezing Rate, and a Noncrystallizing Cosolute. J. Pharm. Sci. 1998, 87, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Gil, A.; Barreneche, C.; Moreno, P.; Solé, C.; Inés Fernández, A.; Cabeza, L.F. Thermal behaviour of d-mannitol when used as PCM: Comparison of results obtained by DSC and in a thermal energy storage unit at pilot plant scale. Appl. Energy 2013, 111, 1107–1113. [Google Scholar] [CrossRef]
- De Beer, T.R.; Wiggenhorn, M.; Hawe, A.; Kasper, J.C.; Almeida, A.; Quinten, T.; Friess, W.; Winter, G.; Vervaet, C.; Remon, J.P. Optimization of a pharmaceutical freeze-dried product and its process using an experimental design approach and innovative process analyzers. Talanta 2011, 83, 1623–1633. [Google Scholar] [CrossRef]
- Jaroch, K.; Jaroch, A.; Bojko, B. Cell cultures in drug discovery and development: The need of reliable in vitro-in vivo extrapolation for pharmacodynamics and pharmacokinetics assessment. J. Pharm. Biomed. Anal. 2018, 147, 297–312. [Google Scholar] [CrossRef]
- Hamon, J.; Renner, M.; Jamei, M.; Lukas, A.; Kopp-Schneider, A.; Bois, F.Y. Quantitative in vitro to in vivo extrapolation of tissues toxicity. Toxicol. Vitr. 2015, 30, 203–216. [Google Scholar] [CrossRef]
- Louisse, J.; Beekmann, K.; Rietjens, I.M. Use of physiologically based kinetic modeling-based reverse dosimetry to predict in vivo toxicity from in vitro data. Chem. Res. Toxicol. 2017, 30, 114–125. [Google Scholar] [CrossRef]
- Algharably, E.A.H.; Kreutz, R.; Gundert-Remy, U. Importance of in vitro conditions for modeling the in vivo dose in humans by in vitro–in vivo extrapolation (IVIVE). Arch. Toxicol. 2019, 93, 615–621. [Google Scholar] [CrossRef]
- Uchida, T.; Furukawa, M.; Kikawada, T.; Yamazaki, K.; Gohara, K. Trehalose uptake and dehydration effects on the cryoprotection of CHO–K1 cells expressing TRET1. Cryobiology 2019, 90, 30–40. [Google Scholar] [CrossRef]
- Brocker, C.; Thompson, D.C.; Vasiliou, V. The role of hyperosmotic stress in inflammation and disease. BioMol. Concepts 2012, 3, 345–364. [Google Scholar] [CrossRef]
- Cvetkovic, L.; Perisic, S.; Titze, J.; Jäck, H.-M.; Schuh, W. The impact of hyperosmolality on activation and differentiation of B lymphoid cells. Front. Immunol. 2019, 10, 828. [Google Scholar] [CrossRef]
- Go, W.Y.; Liu, X.; Roti, M.A.; Liu, F.; Ho, S.N. NFAT5/TonEBP mutant mice define osmotic stress as a critical feature of the lymphoid microenvironment. Proc. Natl. Acad. Sci. USA 2004, 101, 10673–10678. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.L.; Li, Y.; Xu, S.; Nie, Y.; Zhang, J. NFAT5 Regulated by STUB1, Facilitates Malignant Cell Survival and p38 MAPK Activation by Upregulating AQP5 in Chronic Lymphocytic Leukemia. Biochem. Genet. 2021, 59, 870–883. [Google Scholar] [CrossRef] [PubMed]
- Sana, I.; Mantione, M.E.; Angelillo, P.; Muzio, M. Role of NFAT in Chronic Lymphocytic Leukemia and Other B-Cell Malignancies. Front. Oncol. 2021, 11, 651057. [Google Scholar] [CrossRef] [PubMed]
- Drews-Elger, K.; Ortells, M.C.; Rao, A.; López-Rodriguez, C.; Aramburu, J. The Transcription Factor NFAT5 Is Required for Cyclin Expression and Cell Cycle Progression in Cells Exposed to Hypertonic Stress. PLoS ONE 2009, 4, e5245. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Morita, H.; Matsuda, M.; Katakura, Y.; Hirata, M.; Hashimoto, S. NFAT5 promotes oral squamous cell carcinoma progression in a hyperosmotic environment. Lab. Investig. 2021, 101, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Hunger, J.; Bernecker, A.; Bakker, H.J.; Bonn, M.; Richter, R.P. Hydration dynamics of hyaluronan and dextran. Biophys. J. 2012, 103, L10–L12. [Google Scholar] [CrossRef]
- Reich-Slotky, R.; Bachegowda, L.S.; Ancharski, M.; Mendeleyeva, L.; Rubinstein, P.; Rennert, H.; Shore, T.; van Besien, K.; Cushing, M. How we handled the dextran shortage: An alternative washing or dilution solution for cord blood infusions. Transfusion 2015, 55, 1147–1153. [Google Scholar] [CrossRef]
- Soeiro, V.C.; Melo, K.R.T.; Alves, M.G.C.F.; Medeiros, M.J.C.; Grilo, M.L.P.M.; Almeida-Lima, J.; Pontes, D.L.; Costa, L.S.; Rocha, H.A.O. Dextran: Influence of Molecular Weight in Antioxidant Properties and Immunomodulatory Potential. Int. J. Mol. Sci. 2016, 17, 1340. [Google Scholar] [CrossRef]
- Thakral, S.; Sonje, J.; Munjal, B.; Bhatnagar, B.; Suryanarayanan, R. Mannitol as an Excipient for Lyophilized Injectable Formulations. J. Pharm. Sci. 2022, in press. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute: Drugs Approved for Dfferent Types of Cancer. Available online: https://www.cancer.gov/about-cancer/treatment/drugs/cancer-type (accessed on 7 July 2022).
- Perkey, C. Pertuzumab/Trastuzumab/Hyaluronidase-Zzxf (Phesgo™). Oncol. Times 2021, 43, 6–17. [Google Scholar] [CrossRef]
- Abrantes, C.G.; Duarte, D.; Reis, C.P. An Overview of Pharmaceutical Excipients: Safe or Not Safe? J. Pharm. Sci. 2016, 105, 2019–2026. [Google Scholar] [CrossRef] [PubMed]
- Dirain, C.O.; Karnani, D.N.; Antonelli, P.J. Cytotoxicity of Ear Drop Excipients in Human and Mouse Tympanic Membrane Fibroblasts. Otolaryngol. Head Neck Surg. 2019, 162, 204–210. [Google Scholar] [CrossRef]
- Gurjar, R.; Chan, C.Y.S.; Curley, P.; Sharp, J.; Chiong, J.; Rannard, S.; Siccardi, M.; Owen, A. Inhibitory Effects of Commonly Used Excipients on P-Glycoprotein In Vitro. Mol. Pharm. 2018, 15, 4835–4842. [Google Scholar] [CrossRef] [PubMed]


| 1 mg/mL | 10 mg/mL | 100 mg/mL | |
|---|---|---|---|
| Cellobiose | √ | √ | √ |
| Lactose | √ | √ | √ |
| Sucrose | √ | √ | √ |
| Trehalose | √ | √ | √ |
| Isoleucine | √ | √ | |
| Glycine | √ | √ | √ |
| Methionine | √ | √ | |
| Dextran | √ | √ | √ |
| Mannitol | √ | √ | √ |
| Hp-β-cyclodextrin | √ | √ | √ |
| 100 mg/mL | 10 mg/mL | |||||||
|---|---|---|---|---|---|---|---|---|
| Tg′ | Teu | Tc | Tm | Tg′ | Teu | Tc | Tm | |
| Cellobiose | −32.5 | −26.3 | −33.3 | −28.2 | ||||
| Lactose | −31.6 | −26.2 | −31.7 | −27.2 | ||||
| Sucrose | −35.2 | −31.2 | −32.2 | −32.2 | ||||
| Trehalose | −31.8 | −27.2 | −31.1 | −29.2 | ||||
| Isoleucine | ND | ND | ||||||
| Glycine | −5.6; −4 | −3.1 | −5.4 | ND | ||||
| Methionine | ND | ND | ||||||
| Dextran | −13.4 | −6.5 | −13.8 | −10.3 | ||||
| Mannitol | −1.9 | −1.4 | −30.8; −3.2 | −20.8 | ||||
| Hp-β-cyclodextrin | −15.8 | −11.4 | −16.6 | −12.2 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Susa, F.; Limongi, T.; Millone, M.; Cauda, V.; Pisano, R. Cytotoxicity and Thermal Characterization Assessment of Excipients for the Development of Innovative Lyophilized Formulations for Oncological Applications. Processes 2022, 10, 2641. https://doi.org/10.3390/pr10122641
Susa F, Limongi T, Millone M, Cauda V, Pisano R. Cytotoxicity and Thermal Characterization Assessment of Excipients for the Development of Innovative Lyophilized Formulations for Oncological Applications. Processes. 2022; 10(12):2641. https://doi.org/10.3390/pr10122641
Chicago/Turabian StyleSusa, Francesca, Tania Limongi, Michela Millone, Valentina Cauda, and Roberto Pisano. 2022. "Cytotoxicity and Thermal Characterization Assessment of Excipients for the Development of Innovative Lyophilized Formulations for Oncological Applications" Processes 10, no. 12: 2641. https://doi.org/10.3390/pr10122641
APA StyleSusa, F., Limongi, T., Millone, M., Cauda, V., & Pisano, R. (2022). Cytotoxicity and Thermal Characterization Assessment of Excipients for the Development of Innovative Lyophilized Formulations for Oncological Applications. Processes, 10(12), 2641. https://doi.org/10.3390/pr10122641








