Microbial-Mediated Emissions of Greenhouse Gas from Farmland Soils: A Review
Abstract
1. Introduction
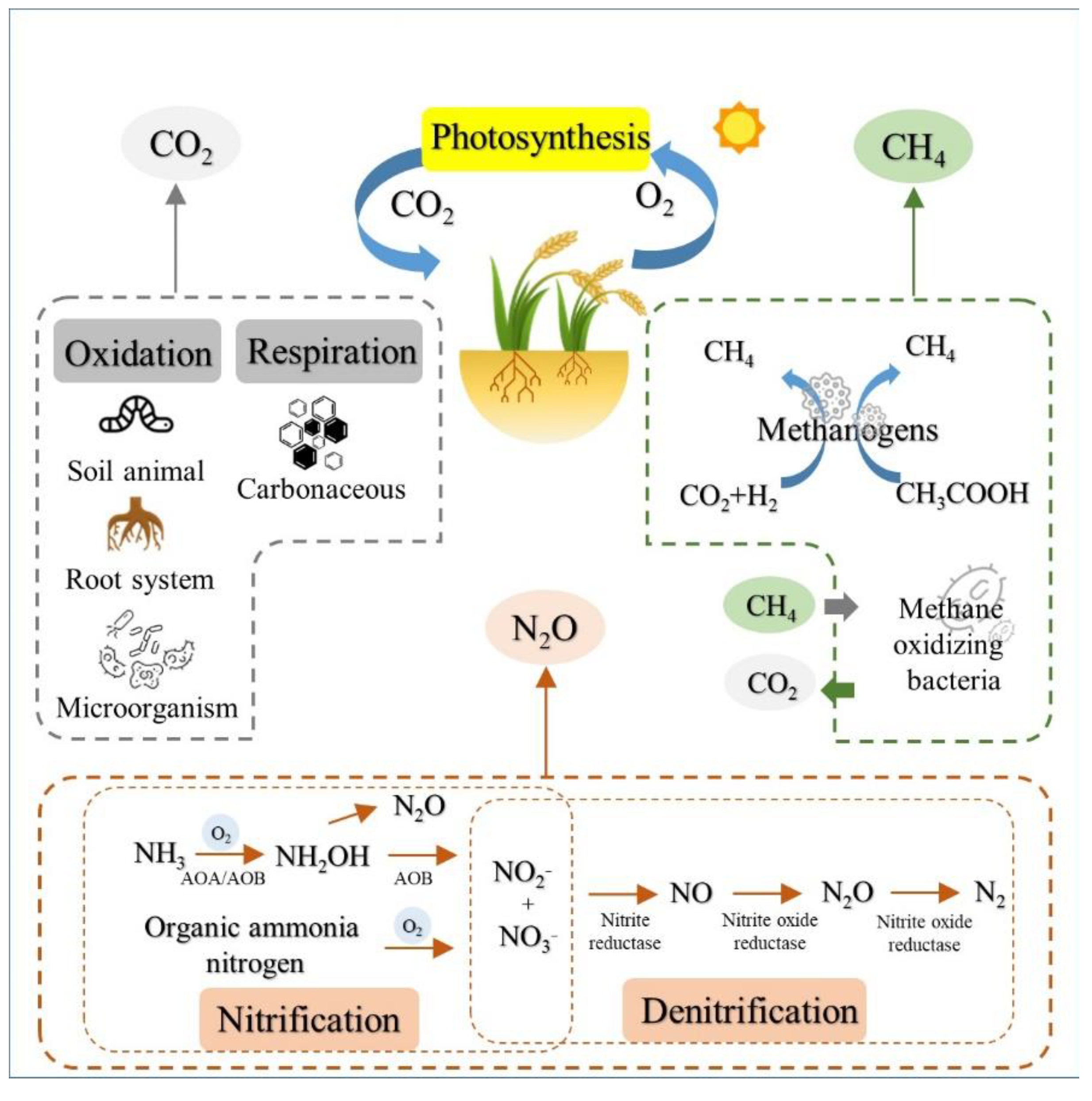
2. GHG Production in Agroecosystem
3. Environmental Factors Affecting GHG Emissions of Microorganisms in Farmland Soil
3.1. Soil Temperature
3.2. Soil Moisture
3.3. Soil Organic Matter
3.4. Soil pH
4. Microbial-Mediated Soil Emissions Reduction Mechanism under Different Soil Remediation Modes
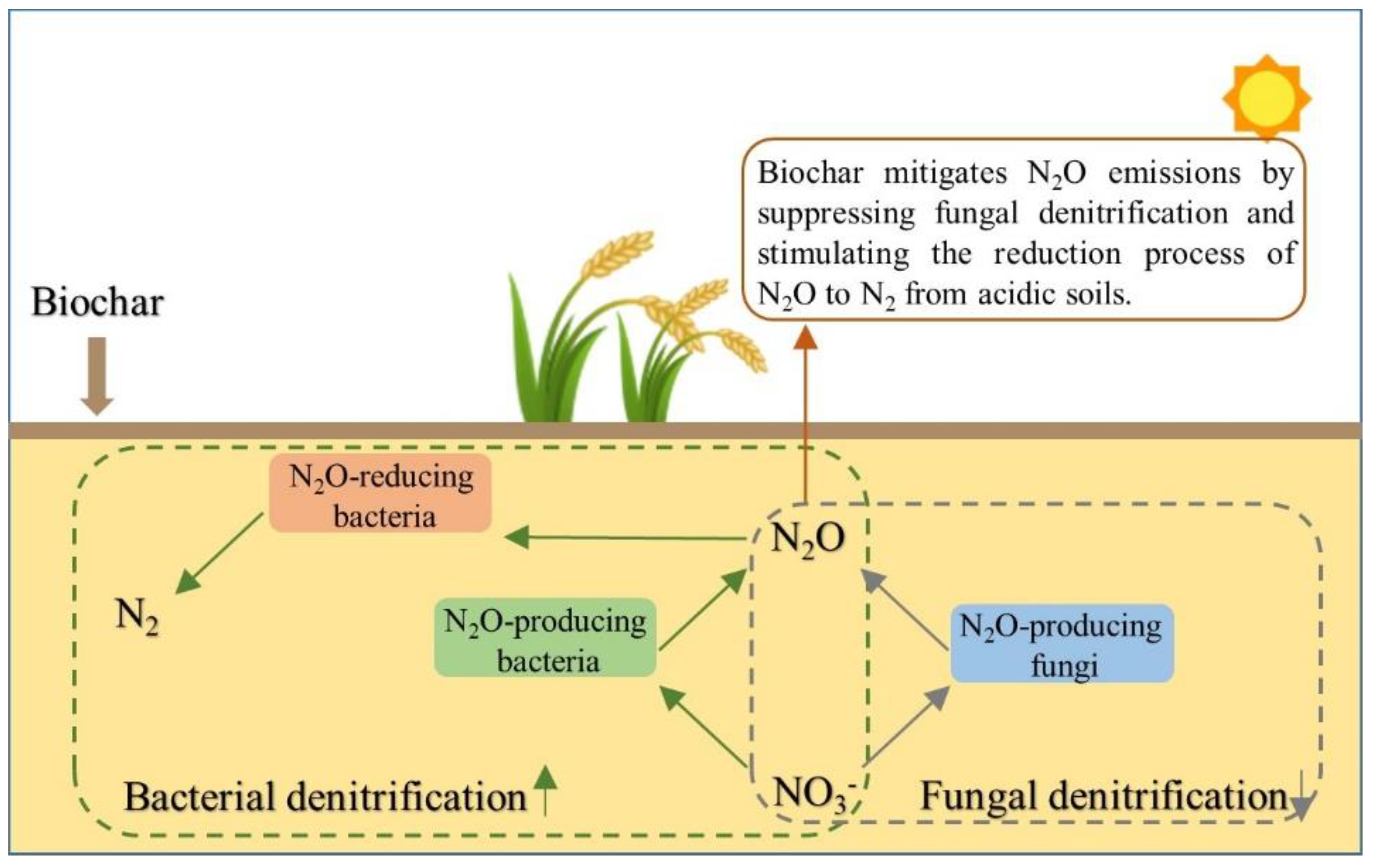
4.1. Biochar
4.2. Organic Fertilizer
4.3. Straw Returning
4.4. Microalgae Biofertilizer
5. Conclusions and Prospects
Author Contributions
Funding
Conflicts of Interest
References
- Hao, Z.; Zhao, Y.; Wang, X.; Wu, J.; Jiang, S.; Xiao, J.; Wang, K.; Zhou, X.; Liu, H.; Li, J.; et al. Thresholds in aridity and soil carbon-to-nitrogen ratio govern the accumulation of soil microbial residues. Commun. Earth Environ. 2021, 2, 236. [Google Scholar] [CrossRef]
- Oertel, C.; Matschullat, J.; Zurba, K.; Zimmermann, F.; Erasmi, S. Greenhouse gas emissions from soils A review. Geochemistry 2016, 76, 327–352. [Google Scholar] [CrossRef]
- Deng, C.X.; Li, R.R.; Xie, B.G.; Wan, Y.L.; Li, Z.W.; Liu, C.C. Impacts of the integrated pattern of water and land resources use on agricultural greenhouse gas emissions in China during 2006–2017: A water-land-energy-emissions nexus analysis. J. Clean. Prod. 2021, 308, 127221. [Google Scholar] [CrossRef]
- Ji, D.H.; Zhou, M.Q.; Wang, P.C.; Yang, Y.; Wang, T.; Sun, X.Y.; Hermans, C.; Yao, B.; Wang, G.C. Deriving Temporal and Vertical Distributions of Methane in Xianghe Using Ground-based Fourier Transform Infrared and Gas-analyzer Measurements. Adv. Atmos. Sci. 2020, 37, 597–607. [Google Scholar] [CrossRef]
- Tian, H.; Chen, G.; Lu, C.; Xu, X.; Ren, W.; Zhang, C.; Zhang, B.; Banger, K.; Tao, B.; Pan, S.; et al. Global methane and nitrous oxide emissions from terrestrial ecosystems due to multiple environmental changes. Ecosyst. Health Sustain. 2015, 1, 1–20. [Google Scholar] [CrossRef]
- Thompson, R.L.; Lassaletta, L.; Patra, P.K.; Wilson, C.; Wells, K.C.; Gressent, A.; Koffi, E.N.; Chipperfield, M.P.; Winiwarter, W.; Davidson, E.A.; et al. Acceleration of global N2O emissions seen from two decades of atmospheric inversion. Nat. Clim. Chang. 2019, 9, 993–998. [Google Scholar] [CrossRef]
- Cui, X.Q.; Zhou, F.; Ciais, P.; Davidson, E.A.; Tubiello, F.N.; Niu, X.Y.; Ju, X.T.; Canadell, J.G.; Bouwman, A.F.; Jackson, R.B.; et al. Global mapping of crop-specific emission factors highlights hotspots of nitrous oxide mitigation. Nat. Food 2021, 2, 886–893. [Google Scholar] [CrossRef]
- Tian, H.Q.; Xu, R.T.; Canadell, J.G.; Thompson, R.L.; Winiwarter, W.; Suntharalingam, P.; Davidson, E.A.; Ciais, P.; Jackson, R.B.; Janssens-Maenhout, G.; et al. A comprehensive quantification of global nitrous oxide sources and sinks. Nature 2020, 586, 248–256. [Google Scholar] [CrossRef]
- Syakila, A.; Kroeze, C. The global nitrous oxide budget revisited. Greenh. Gas. Meas. Manag. 2011, 1, 17–26. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, M.; Lin, H.; Ntacyabukura, T.; Wang, Y.; Zhu, B. Effects of different long-term crop straw management practices on ammonia volatilization from subtropical calcareous agricultural soil. Atmos. Ocean. Sci. Lett. 2020, 13, 232–239. [Google Scholar] [CrossRef]
- Zuo, S.S.; Wu, D.; Du, Z.L.; Xu, C.C.; Wu, W.L. Effects of white-rot fungal pretreatment of corn straw return on greenhouse gas emissions from the North China Plain soil. Sci. Total Environ. 2022, 807, 150837. [Google Scholar] [CrossRef]
- Sun, J.; Haiyun, P.; Jianmin, C.; Xinming, W.; Min, W.; Weijun, L.; Lingxiao, Y.; Qingzhu, Z.; Wenxing, W.; Abdelwahid, M. An estimation of CO2 emission via agricultural crop residue open field burning in China from 1996 to 2013. J. Clean. Prod. 2016, 112, 2625–2631. [Google Scholar] [CrossRef]
- Chen, H.H.; Li, X.C.; Hu, F.; Shi, W. Soil nitrous oxide emissions following crop residue addition: A meta-analysis. Glob. Chang. Biol. 2013, 19, 2956–2964. [Google Scholar] [CrossRef]
- Sun, D.Y.; Tang, X.F.; Li, J.; Liu, M.; Hou, L.J.; Yin, G.Y.; Chen, C.; Zhao, Q.; Klumper, U.; Han, P. Chlorate as a comammox Nitrospira specific inhibitor reveals nitrification and N2O production activity in coastal wetland. Soil Biol. Biochem. 2022, 173, 108782. [Google Scholar] [CrossRef]
- Terrer, C.; Phillips, R.P.; Hungate, B.A.; Rosende, J.; Pett, R.J.; Craig, M.E.; van Groenigen, K.J.; Keenan, T.F.; Sulman, B.N.; Stocker, B.D.; et al. A trade-off between plant and soil carbon storage under elevated CO2. Nature 2021, 591, 599–603. [Google Scholar] [CrossRef]
- Zheng, X.; Moses, A.A.; Hui, W.; Peng, H.; Shan, L.; Na, C.; Songyuan, W.; Hongling, Z.; Hui, D.; Kebin, L. Effect of lignin and plant growth-promoting bacteria (Staphylococcus pasteuri) on microbe-plant Co-remediation: A PAHs-DDTs Co-contaminated agricultural greenhouse study. Chemosphere 2020, 256, 127079. [Google Scholar] [CrossRef]
- Saunois, M.; Stavert, A.R.; Poulter, B.; Bousquet, P.; Canadell, J.G.; Jackson, R.B.; Raymond, P.A.; Dlugokencky, E.J.; Houweling, S.; Patra, P.K.; et al. The Global Methane Budget 2000–2017. Earth Syst. Sci. Data 2020, 12, 1561–1623. [Google Scholar] [CrossRef]
- Malyan, S.K.; Bhatia, A.; Kumar, A.; Gupta, D.K.; Singh, R.; Kumar, S.S.; Tomer, R.; Kumar, O.; Jain, N. Methane production, oxidation and mitigation: A mechanistic understanding and comprehensive evaluation of influencing factors. Sci. Total Environ. 2016, 572, 874–896. [Google Scholar] [CrossRef]
- Mitra, S.; Majumdar, D.; Wassmann, R. Methane production and emission in surface and subsurface rice soils and their blends. Agric. Ecosyst. Environ. 2012, 158, 94–102. [Google Scholar] [CrossRef]
- Zhong, L.; Bowatte, S.; Newton, P.C.D.; Hoogendoorn, C.J.; Luo, D. An increased ratio of fungi to bacteria indicates greater potential for N2O production in a grazed grassland exposed to elevated CO2. Agric. Ecosyst. Environ. 2018, 254, 111–116. [Google Scholar] [CrossRef]
- Wang, C.; Amon, B.; Schulz, K.; Mehdi, B. Factors That Influence Nitrous Oxide Emissions from Agricultural Soils as Well as Their Representation in Simulation Models: A Review. Agronomy 2021, 11, 770. [Google Scholar] [CrossRef]
- Muhammad, Z.M.; Edouard, M.; Alain, R.; Martial, B.; Alain, B. Xylophagous termites: A potential sink for atmospheric nitrous oxide. Eur. J. Soil Biol. 2012, 53, 121–125. [Google Scholar]
- Lu, S.B.; Xu, Y.; Fu, X.P.; Xiao, H.; Ding, W.; Zhang, Y.J. Patterns and Drivers of Soil Respiration and Vegetation at Different Altitudes in Southern China. Appl. Ecol. Environ. Res. 2019, 17, 3097–3106. [Google Scholar] [CrossRef]
- Kellman, L.; Myette, A.; Beltrami, H. Depth-Dependent Mineral Soil CO2 Production Processes: Sensitivity to Harvesting-Induced Changes in Soil Climate. PLoS ONE 2015, 10, e0134171. [Google Scholar] [CrossRef]
- Wang, Q.K.; Zhao, X.C.; Chen, L.C.; Yang, Q.P.; Chen, S.; Zhang, W.D. Global synthesis of temperature sensitivity of soil organic carbon decomposition: Latitudinal patterns and mechanisms. Funct. Ecol. 2019, 33, 514–523. [Google Scholar] [CrossRef]
- Carey, J.C.; Tang, J.W.; Templer, P.H.; Kroeger, K.D.; Crowther, T.W.; Burton, A.J.; Dukes, J.S.; Emmett, B.; Frey, S.D.; Heskel, M.A.; et al. Temperature response of soil respiration largely unaltered with experimental warming. Proc. Natl. Acad. Sci. USA 2016, 113, 13797–13802. [Google Scholar] [CrossRef]
- Voigt, C.; Lamprecht, R.E.; Marushchak, M.E.; Lind, S.E.; Novakovskiy, A.; Aurela, M.; Martikainen, P.J.; Biasi, C. Warming of subarctic tundra increases emissions of all three important greenhouse gases-carbon dioxide, methane, and nitrous oxide. Glob. Chang. Biol. 2017, 23, 3121–3138. [Google Scholar] [CrossRef]
- Chen, H.; Zhu, T.; Li, B.; Fang, C.; Nie, M. The thermal response of soil microbial methanogenesis decreases in magnitude with changing temperature. Nat. Commun. 2020, 11, 5733. [Google Scholar] [CrossRef]
- Walker, T.W.N.; Kaiser, C.; Strasser, F.; Herbold, C.W.; Leblans, N.I.W.; Woebken, D.; Janssens, I.A.; Sigurdsson, B.D.; Richter, A. Microbial temperature sensitivity and biomass change explain soil carbon loss with warming. Nat. Clim. Chang. 2018, 8, 885–889. [Google Scholar] [CrossRef]
- Nottingham, A.T.; Whitaker, J.; Ostle, N.J.; Bardgett, R.D.; McNamara, N.P.; Fierer, N.; Salinas, N.; Ccahuana, A.J.Q.; Turner, B.L.; Meir, P. Microbial responses to warming enhance soil carbon loss following translocation across a tropical forest elevation gradient. Ecol. Lett. 2019, 22, 1889–1899. [Google Scholar] [CrossRef]
- Cui, P.Y.; Fan, F.L.; Yin, C.; Song, A.L.; Huang, P.R.; Tang, Y.J.; Zhu, P.; Peng, C.; Li, T.Q.; Wakelin, S.A.; et al. Long-term organic and inorganic fertilization alters temperature sensitivity of potential N2O emissions and associated microbes. Soil Biol. Biochem. 2016, 93, 131–141. [Google Scholar] [CrossRef]
- Jannis, H.; Shurong, L.; Harry, V.; Nicolas, B. Abiotic nitrous oxide production from hydroxylamine in soils and their dependence on soil properties. Soil Biol. Biochem. 2015, 84, 107–115. [Google Scholar]
- Li, J.; Junmin, P.; Elise, P.; Changming, F.; Ming, N. Spatial heterogeneity of temperature sensitivity of soil respiration: A global analysis of field observations. Soil Biol. Biochem. 2020, 141, 107675. [Google Scholar] [CrossRef]
- Hu, H.; Chen, D.; He, J.-Z. Microbial regulation of terrestrial nitrous oxide formation: Understanding the biological pathways for prediction of emission rates. FEMS Microbiol. Rev. 2015, 39, 729–749. [Google Scholar] [CrossRef]
- Hu, H.; Xu, Z.; He, J. Ammonia-Oxidizing Archaea Play a Predominant Role in Acid Soil Nitrification. Adv. Agron. 2014, 125, 261–302. [Google Scholar]
- Liao, B.; Wu, X.; Yu, Y.F.; Luo, S.Y.; Hu, R.G.; Lu, G.A. Effects of mild alternate wetting and drying irrigation and mid-season drainage on CH4 and N2O emissions in rice cultivation. Sci. Total Environ. 2020, 698, 134212. [Google Scholar] [CrossRef]
- Liang, K.; Zhong, X.; Huang, N.; Lampayan, R.M.; Liu, Y.; Pan, J.; Peng, B.; Hu, X.; Fu, Y. Nitrogen losses and greenhouse gas emissions under different N and water management in a subtropical double-season rice cropping system. Sci. Total Environ. 2017, 609, 46–57. [Google Scholar] [CrossRef]
- Tirol-Padre, A.; Minamikawa, K.; Tokida, T.; Wassmann, R.; Yagi, K. Site-specific feasibility of alternate wetting and drying as a greenhouse gas mitigation option in irrigated rice fields in Southeast Asia: A synthesis. Soil Sci. Plant Nutr. 2018, 64, 2–13. [Google Scholar] [CrossRef]
- Humphrey, V.; Berg, A.; Ciais, P.; Gentine, P.; Jung, M.; Reichstein, M.; Seneviratne, S.I.; Frankenberg, C. Soil moisture-atmosphere feedback dominates land carbon uptake variability. Nature 2021, 592, 65–69. [Google Scholar] [CrossRef]
- Sylvie, D.; Katell, Q. Analytical pyrolysis as a tool to probe soil organic matter. J. Anal. Appl. Pyrolysis 2015, 111, 108–120. [Google Scholar]
- Li, Y.; Dong, S.; Liu, S.; Zhou, H.; Gao, Q.; Cao, G.; Wang, X.; Su, X.; Zhang, Y.; Tang, L.; et al. Seasonal changes of CO2, CH4 and N2O fluxes in different types of alpine grassland in the Qinghai-Tibetan Plateau of China. Soil Biol. Biochem. 2015, 80, 306–314. [Google Scholar] [CrossRef]
- Chen, H.; Yang, Z.; Chu, R.K.; Tolic, N.; Liang, L.; Graham, D.E.; Wullschleger, S.D.; Gu, B. Molecular Insights into Arctic Soil Organic Matter Degradation under Warming. Environ. Sci. Technol. 2018, 52, 4555–4564. [Google Scholar] [CrossRef]
- Paré, M.C.; Bedard-Haughn, A. Soil organic matter quality influences mineralization and GHG emissions in cryosols: A field-based study of sub- to high Arctic. Glob. Chang. Biol. 2013, 19, 1126–1140. [Google Scholar] [CrossRef]
- Wang, H.H.; Shen, M.X.; Hui, D.F.; Chen, J.; Sun, G.F.; Wang, X.; Lu, C.Y.; Sheng, J.; Chen, L.G.; Luo, Y.Q.; et al. Straw incorporation influences soil organic carbon sequestration, greenhouse gas emission, and crop yields in a Chinese rice (Oryza sativa L.)-wheat (Triticum aestivum L.) cropping system. Soil Tillage Res. 2019, 195, 104377. [Google Scholar] [CrossRef]
- Pascual, J.; Wust, P.K.; Geppert, A.; Foesel, B.U.; Huber, K.J.; Overmann, J. Novel isolates double the number of chemotrophic species and allow the first description of higher taxa in Acidobacteria subdivision 4. Syst. Appl. Microbiol. 2015, 38, 534–544. [Google Scholar] [CrossRef]
- Xu, P.; Zhou, W.; Jiang, M.D.; Khan, I.; Wu, T.T.; Zhou, M.H.; Zhu, B.; Hu, R.G. Methane emission from rice cultivation regulated by soil hydrothermal condition and available carbon and nitrogen under a rice-wheat rotation system. Plant Soil 2022, 1–12. [Google Scholar] [CrossRef]
- Wu, Y.P.; Liu, T.; Peng, Q.; Shaaban, M.; Hu, R.G. Effect of straw returning in winter fallow in Chinese rice fields on greenhouse gas emissions: Evidence from an incubation study. Soil Res. 2015, 53, 298–305. [Google Scholar] [CrossRef]
- Cui, Y.; Meng, J.; Wang, Q.; Weiming, Z.; Weiming, Z.; Wenfu, C. Effects of straw and biochar addition on soil nitrogen, carbon, and super rice yield in cold waterlogged paddy soils of North China. J. Integr. Agric. 2017, 16, 1064–1074. [Google Scholar] [CrossRef]
- Zhao, S.X.; Schmidt, S.; Qin, W.; Li, J.; Li, G.X.; Zhang, W.F. Towards the circular nitrogen economy—A global meta-analysis of composting technologies reveals much potential for mitigating nitrogen losses. Sci. Total Environ. 2020, 704, 135401. [Google Scholar] [CrossRef]
- Awais, S.; Saba, S.; Abdul, R.; Fatima, A.; Muhammad, A.; Sher, M.S.; Taimoor, H.F.; Muhammad, A.; Muhammad, A.M.; Muhammad, M.A.; et al. Effect of animal manure, crop type, climate zone, and soil attributes on greenhouse gas emissions from agricultural soils-A global meta-analysis. J. Clean. Prod. 2021, 278, 124019. [Google Scholar]
- Qu, Z.; Wang, J.; Almoy, T.; Bakken, L.R. Excessive use of nitrogen in Chinese agriculture results in high N2O/(N2O+N2) product ratio of denitrification, primarily due to acidification of the soils. Glob. Chang. Biol. 2014, 20, 1685–1698. [Google Scholar] [CrossRef] [PubMed]
- Shaaban, M.; Wu, Y.P.; Peng, Q.A.; Lin, S.; Mo, Y.L.; Wu, L.; Hu, R.G.; Zhou, W. Effects of dicyandiamide and dolomite application on N2O emission from an acidic soil. Environ. Sci. Pollut. Res. 2016, 23, 6334–6342. [Google Scholar] [CrossRef] [PubMed]
- Galbally, I.E.; Meyer, C.P.; Wang, Y.P.; Smith, C.J.; Weeks, I.A. Nitrous oxide emissions from a legume pasture and the influences of liming and urine addition. Agric. Ecosyst. Environ. 2016, 136, 262–272. [Google Scholar] [CrossRef]
- Shaaban, M.; Peng, Q.-A.; Hu, R.; Wu, Y.; Lin, S.; Zhao, J. Dolomite application to acidic soils: A promising option for mitigating N2O emissions. Environ. Sci. Pollut. Res. 2015, 22, 19961–19970. [Google Scholar] [CrossRef]
- Wu, H.; Hao, X.; Xu, P.; Hu, J.; Jiang, M.; Shaaban, M.; Zhao, J.; Wu, Y.; Hu, R. CO2 and N2O emissions in response to dolomite application are moisture dependent in an acidic paddy soil. J. Soils Sediments 2020, 20, 3136–3147. [Google Scholar] [CrossRef]
- Shaaban, M.; Wu, Y.P.; Wu, L.; Hu, R.G.; Younas, A.; Nunez-Delgado, A.; Xu, P.; Sun, Z.; Lin, S.; Xu, X.Y.; et al. The Effects of pH Change through Liming on Soil N2O Emissions. Processes 2020, 8, 702. [Google Scholar] [CrossRef]
- Yang, Y.; Sun, K.; Liu, J.; Chen, Y.L.; Han, L.F. Changes in soil properties and CO2 emissions after biochar addition: Role of pyrolysis temperature and aging. Sci. Total Environ. 2022, 839, 156333. [Google Scholar] [CrossRef]
- Gasco, G.; Paz-Ferreiro, J.; Cely, P.; Plaza, C.; Mendez, A. Influence of pig manure and its biochar on soil CO2 emissions and soil enzymes. Ecol. Eng. 2016, 95, 19–24. [Google Scholar] [CrossRef]
- Benavente, I.; Gasco, G.; Plaza, C.; Paz-Ferreiro, J.; Mendez, A. Choice of pyrolysis parameters for urban wastes affects soil enzymes and plant germination in a Mediterranean soil. Sci. Total Environ. 2018, 634, 1308–1314. [Google Scholar] [CrossRef]
- Yu, Z.; Chen, L.; Pan, S.; Li, Y.; Kuzyakov, Y.; Xu, J.; Brookes, P.C.; Luo, Y. Feedstock determines biochar-induced soil priming effects by stimulating the activity of specific microorganisms. Eur. J. Soil Sci. 2018, 69, 521–534. [Google Scholar] [CrossRef]
- Yang, F.; Xu, Z.B.; Huang, Y.D.; Tsang, D.C.W.; Ok, Y.S.; Zhao, L.; Qiu, H.; Xu, X.Y.; Cao, X.D. Stabilization of dissolvable biochar by soil minerals: Release reduction and organo-mineral complexes formation. J. Hazard. Mater. 2021, 412, 125213. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.J.; Su, W.Q.; Parikh, S.J.; Li, Y.; Tang, C.X.; Xu, J.M. Intact and washed biochar caused different patterns of nitrogen transformation and distribution in a flooded paddy soil. J. Clean. Prod. 2021, 293, 126259. [Google Scholar] [CrossRef]
- Wang, L.; Gao, C.C.; Yang, K.; Sheng, Y.Q.; Xu, J.; Zhao, Y.X.; Lou, J.; Sun, R.; Zhu, L.Z. Effects of biochar aging in the soil on its mechanical property and performance for soil CO2 and N2O emissions. Sci. Total Environ. 2021, 782, 146824. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wang, C.; Shen, J.L.; Li, Y.; Wu, J.S. Response of CH4 emissions to straw and biochar applications in double-rice cropping systems: Insights from observations and modeling. Environ. Pollut. 2018, 235, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shen, J.L.; Liu, J.Y.; Qin, H.L.; Yuan, Q.; Fan, F.L.; Hu, Y.J.; Wang, J.; Wei, W.X.; Li, Y.; et al. Microbial mechanisms in the reduction of CH4 emission from double rice cropping system amended by biochar: A four-year study. Soil Biol. Biochem. 2019, 135, 251–263. [Google Scholar] [CrossRef]
- He, L.L.; Shan, J.; Zhao, X.; Wang, S.Q.; Yan, X.Y. Variable responses of nitrification and denitrification in a paddy soil to long-term biochar amendment and short-term biochar addition. Chemosphere 2019, 234, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Lourenco, K.S.; Dimitrov, M.R.; Pijl, A.; Soares, J.R.; Do Carmo, J.B.; van Veen, J.A.; Cantarella, H.; Kuramae, E.E. Dominance of bacterial ammonium oxidizers and fungal denitrifiers in the complex nitrogen cycle pathways related to nitrous oxide emission. GCB Bioenergy 2018, 10, 645–660. [Google Scholar] [CrossRef]
- Mothapo, N.V.; Chen, H.H.; Cubeta, M.A.; Shi, W. Nitrous oxide producing activity of diverse fungi from distinct agroecosystems. Soil Biol. Biochem. 2013, 66, 94–101. [Google Scholar] [CrossRef]
- Ji, C.; Han, Z.Q.; Zheng, F.W.; Wu, S.; Wang, J.Y.; Wang, J.D.; Zhang, H.; Zhang, Y.C.; Liu, S.W.; Li, S.Q.; et al. Biochar reduced soil nitrous oxide emissions through suppressing fungal denitrification and affecting fungal community assembly in a subtropical tea plantation. Agric. Ecosyst. Environ. 2022, 326, 107784. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, Y.; Mao, J.; Chen, B. Effects of biochar nanoparticles on seed germination and seedling growth. Environ. Pollut. 2020, 256, 113409. [Google Scholar] [CrossRef]
- Jan, M.; Josephine, G.; Munoo, P.; Ulf, L.; Juergen, K.; Ondrej, M.; Wolfram, B. Toxicity screening of biochar-mineral composites using germination tests. Chemosphere 2018, 207, 91–100. [Google Scholar]
- Huang, C.; Weiyue, W.; Shizhong, Y.; Muhammad, A.; Yuhui, Q. Role of biochar and Eisenia fetida on metal bioavailability and biochar effects on earthworm fitness. Environ. Pollut. 2020, 263, 114586. [Google Scholar] [CrossRef] [PubMed]
- Malev, O.; Contin, M.; Licen, S.; Barbieri, P.; De Nobili, M. Bioaccumulation of polycyclic aromatic hydrocarbons and survival of earthworms (Eisenia andrei) exposed to biochar amended soils. Environ. Sci. Pollut. Res. 2016, 23, 3491–3502. [Google Scholar] [CrossRef] [PubMed]
- Li, L.J.; You, M.Y.; Shi, H.A.; Ding, X.L.; Qiao, Y.F.; Han, X.Z. Soil CO2 emissions from a cultivated Mollisol: Effects of organic amendments, soil temperature, and moisture. Eur. J. Soil Biol. 2013, 55, 83–90. [Google Scholar] [CrossRef]
- Wang, C.; Ma, X.F.; Shen, J.L.; Chen, D.; Zheng, L.; Ge, T.D.; Li, Y.; Wu, J.S. Reduction in net greenhouse gas emissions through a combination of pig manure and reduced inorganic fertilizer application in a double-rice cropping system: Three-year results. Agric. Ecosyst. Environ. 2022, 326, 107799. [Google Scholar] [CrossRef]
- Li, B.Z.; Song, H.; Cao, W.C.; Wang, Y.J.; Chen, J.S.; Guo, J.H. Responses of soil organic carbon stock to animal manure application: A new global synthesis integrating the impacts of agricultural managements and environmental conditions. Glob. Chang. Biol. 2021, 27, 5356–5367. [Google Scholar] [CrossRef]
- Tian, H.Q.; Lu, C.Q.; Ciais, P.; Michalak, A.M.; Canadell, J.G.; Saikawa, E.; Huntzinger, D.N.; Gurney, K.R.; Sitch, S.; Zhang, B.W.; et al. The terrestrial biosphere as a net source of greenhouse gases to the atmosphere. Nature 2016, 531, 225–228. [Google Scholar] [CrossRef]
- Yuan, J.; Yuan, Y.; Zhu, Y.; Cao, L. Effects of different fertilizers on methane emissions and methanogenic community structures in paddy rhizosphere soil. Sci. Total Environ. 2018, 627, 770–781. [Google Scholar] [CrossRef]
- Li, Y.J.; Zheng, Q.; Yang, R.; Zhuang, S.; Lin, W.; Li, Y.Z. Evaluating microbial role in reducing N2O emission by dual isotopocule mapping following substitution of inorganic fertilizer for organic fertilizer. J. Clean. Prod. 2021, 326, 129442. [Google Scholar] [CrossRef]
- Guo, L.J.; Zhang, L.; Liu, L.; Sheng, F.; Cao, C.G.; Li, C.F. Effects of long-term no tillage and straw return on greenhouse gas emissions and crop yields from a rice-wheat system in central China. Agric. Ecosyst. Environ. 2021, 322, 107650. [Google Scholar] [CrossRef]
- Wu, X.H.; Wang, W.; Xie, K.J.; Yin, C.M.; Hou, H.J.; Xie, X.L. Combined effects of straw and water management on CH4 emissions from rice fields. J. Environ. Manag. 2019, 231, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.D.; He, C.; Cheng, H.Y.; Liu, B.Y.; Li, S.S.; Wang, Q.; Liu, Y.; Zhao, X.; Zhang, H.L. Responses of greenhouse gas emissions to residue returning in China’s croplands and influential factors: A meta-analysis. J. Environ. Manag. 2021, 289, 112486. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Dai, M.W.; Dai, S.L.; Dong, X.J. Current status and environment impact of direct straw return in China’s cropland—A review. Ecotoxicol. Environ. Saf. 2018, 159, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Y.; Wang, K.; Meng, S.X.; Zheng, X.H.; Zhou, Z.X.; Han, S.H.; Chen, D.L.; Yang, Z.P. Effects of irrigation, fertilization and crop straw management on nitrous oxide and nitric oxide emissions from a wheat-maize rotation field in northern China. Agric. Ecosyst. Environ. 2011, 140, 226–233. [Google Scholar] [CrossRef]
- Xu, C.; Han, X.; Ru, S.H.; Cardenas, L.; Rees, R.M.; Wu, D.; Wu, W.L.; Meng, F.Q. Crop straw incorporation interacts with N fertilizer on N2O emissions in an intensively cropped farmland. Geoderma 2019, 341, 129–137. [Google Scholar] [CrossRef]
- Chen, Z.X.; Tu, X.S.; Meng, H.; Chen, C.; Chen, Y.J.; Elrys, A.S.; Cheng, Y.; Zhang, J.B.; Cai, Z.C. Microbial process-oriented understanding of stimulation of soil N2O emission following the input of organic materials. Environ. Pollut. 2021, 284, 117176. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Guo, L.J.; Liu, T.Q.; Li, C.F.; Cao, C.G. Effects of tillage practices and straw returning methods on greenhouse gas emissions and net ecosystem economic budget in rice wheat cropping systems in central China. Atmos. Environ. 2015, 122, 636–644. [Google Scholar] [CrossRef]
- Zhu, H.J.; Tao, J.; Yan, X.M.; Zhou, B.J.; Mwangi, J.K. Short-Term Effects of Straw Application on Carbon Recycle in a Rice-Rapeseed Rotation System. Aerosol Air Qual. Res. 2016, 16, 3358–3363. [Google Scholar] [CrossRef]
- Jiang, C.M.; Yu, W.T.; Ma, Q.; Xu, Y.G.; Zou, H. Alleviating global warming potential by soil carbon sequestration: A multi-level straw incorporation experiment from a maize cropping system in Northeast China. Soil Tillage Res. 2017, 170, 77–84. [Google Scholar] [CrossRef]
- Liang, S.; Zhang, H. Different Responses of Greenhouse Gas Emissions to Straw Application at Different Seasons in Northeast China. Sains Malays. 2019, 48, 1347–1355. [Google Scholar] [CrossRef]
- Zhang, H.; Liang, S.; Wang, Y.H.; Liu, S.W.; Sun, H.D. Greenhouse gas emissions of rice straw return varies with return depth and soil type in paddy systems of Northeast China. Arch. Agron. Soil Sci. 2021, 67, 1591–1602. [Google Scholar] [CrossRef]
- Li, S.H.; Guo, L.J.; Cao, C.G.; Li, C.F. Effects of straw returning levels on carbon footprint and net ecosystem economic benefits from rice-wheat rotation in central China. Environ. Sci. Pollut. Res. 2021, 28, 5742–5754. [Google Scholar] [CrossRef]
- Wu, G.; Ling, J.; Xu, Y.P.; Zhao, D.Q.; Liu, Z.X.; Wen, Y.; Zhou, S.L. Effects of soil warming and straw return on soil organic matter and greenhouse gas fluxes in winter wheat seasons in the North China Plain. J. Clean. Prod. 2022, 356, 131810. [Google Scholar] [CrossRef]
- Alvarez, A.L.; Weyers, S.L.; Goemann, H.M.; Peyton, B.M.; Gardner, R.D. Microalgae, soil and plants: A critical review of microalgae as renewable resources for agriculture. Algal Res. 2021, 54, 102200. [Google Scholar] [CrossRef]
- de Siqueira, C.J.; Lucia, C.M.; Peixoto, A.P.; Roberto, C.P.; Rodrigues, D.A.I.; Jose, R.V. Microalgae biofilm in soil: Greenhouse gas emissions, ammonia volatilization and plant growth. Sci. Total Environ. 2017, 574, 1640–1648. [Google Scholar]
- Bharti, A.; Velmourougane, K.; Prasanna, R. Phototrophic biofilms: Diversity, ecology and applications. J. Appl. Phycol. 2017, 29, 2729–2744. [Google Scholar] [CrossRef]
- Marks, E.A.N.; Minon, J.; Pascual, A.; Montero, O.; Navas, L.M.; Rad, C. Application of a microalgal slurry to soil stimulates heterotrophic activity and promotes bacterial growth. Sci. Total Environ. 2017, 605, 610–617. [Google Scholar] [CrossRef]
- Knoche, K.L.; Aoyama, E.; Hasan, K.; Minteer, S.D. Role of Nitrogenase and Ferredoxin in the Mechanism of Bioelectrocatalytic Nitrogen Fixation by the Cyanobacteria Anabaena variabilis SA-1 Mutant Immobilized on Indium Tin Oxide (ITO) Electrodes. Electrochim. Acta 2017, 232, 396–403. [Google Scholar] [CrossRef]
- Mallappa, M.; Amrita, K.; Kunal, R.; Siddarthan, V.; Radha, P.; Balasubramanian, R.; Firoz, H.; Lata, N.; Yashbir, S.S.; Awadhesh, B.R.; et al. Beneficial cyanobacteria and eubacteria synergistically enhance bioavailability of soil nutrients and yield of okra. Heliyon 2016, 2, e00066. [Google Scholar]
- Renuka, N.; Prasanna, R.; Sood, A.; Ahluwalia, A.S.; Bansal, R.; Babu, S.; Singh, R.; Shivay, Y.S.; Nain, L. Exploring the efficacy of wastewater-grown microalgal biomass as a biofertilizer for wheat. Environ. Sci. Pollut. Res. 2016, 23, 6608–6620. [Google Scholar] [CrossRef]
- Ali, M.A.; Sattar, M.A.; Islam, M.N.; Inubushi, K. Integrated effects of organic, inorganic and biological amendments on methane emission, soil quality and rice productivity in irrigated paddy ecosystem of Bangladesh: Field study of two consecutive rice growing seasons. Plant Soil 2014, 378, 239–252. [Google Scholar] [CrossRef]
- Prasanna, R.; Adak, A.; Verma, S.; Bidyarani, N.; Babu, S.; Pal, M.; Shivay, Y.S.; Nain, L. Cyanobacterial inoculation in rice grown under flooded and SRI modes of cultivation elicits differential effects on plant growth and nutrient dynamics. Ecol. Eng. 2015, 84, 532–541. [Google Scholar] [CrossRef]
- Shrestha, R.C.; Ghazaryan, L.; Poodiack, B.; Zorin, B.; Gross, A.; Gillor, O.; Khozin-Goldberg, I.; Gelfand, I. The effects of microalgae-based fertilization of wheat on yield, soil microbiome and nitrogen oxides emissions. Sci. Total Environ. 2022, 806, 151320. [Google Scholar] [CrossRef]
- Zhang, S.P.; Wang, L.; Wei, W.; Hu, J.J.; Mei, S.H.; Zhao, Q.Y.; Tsang, Y.F. Enhanced roles of biochar and organic fertilizer in microalgae for soil carbon sink. Biodegradation 2018, 29, 313–321. [Google Scholar] [CrossRef]
- Hu, J.J.; Guo, H.C.; Xue, Y.Y.; Gao, M.T.; Zhang, S.P.; Tsang, Y.F.; Li, J.X.; Wang, Y.N.; Wang, L. Using a mixture of microalgae, biochar, and organic manure to increase the capacity of soil to act as carbon sink. J. Soils Sediments 2019, 19, 3718–3727. [Google Scholar] [CrossRef]
- Bhardwaj, D.; Ansari, M.W.; Sahoo, R.K.; Tuteja, N. Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microb. Cell Fact. 2014, 13, 66. [Google Scholar] [CrossRef]
- Yen, H.W.; Ho, S.H.; Chen, C.Y.; Chang, J.S. CO2, NOx and SOx removal from flue gas via microalgae cultivation: A critical review. Biotechnol. J. 2015, 10, 829–839. [Google Scholar] [CrossRef]
- Zeraatkar, A.K.; Ahmadzadeh, H.; Talebi, A.F.; Moheimani, N.R.; McHenry, M.P. Potential use of algae for heavy metal bioremediation, a critical review. J. Environ. Manag. 2016, 181, 817–831. [Google Scholar] [CrossRef]

| Soil Type | Straw Type | GHG | Compared with No Straw Addition | Year | Ref. |
|---|---|---|---|---|---|
| Rice-wheat | Rice-wheat | CH4 | +35.0% | 2015 | [87] |
| Rice-rapeseed | Rapeseed | CO2 | +6.3% | 2016 | [88] |
| CH4 | +32.9% | ||||
| Maize-crop | Maize straw | N2O | −11.0~27.0% | 2017 | [89] |
| Rice paddy | Rice straw | CH4 | +39.1% | 2017 | [48] |
| N2O | −77.8% | ||||
| Rice paddy | Rice straw | CO2 | +14.8~27.5% | 2019 | [90] |
| CH4 | +36.9~182.1% | ||||
| N2O | −23.5~40.6% | ||||
| Rice-wheat | Wheat | CH4 | +36.6~80.1% | 2021 | [91] |
| Rice-wheat | Rice-wheat | CH4 | +41.20% | 2021 | [80] |
| N2O | +47.50% | ||||
| Rice-wheat | Rice-wheat | CH4 | +5.4~72.2% | 2021 | [92] |
| N2O | −3.3~31.4% | ||||
| Wheat | Wheat | CO2 | +11.5~28.3% | 2022 | [93] |
| N2O | +37.1~48.4% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Zhao, R.; Zhao, D.; Liu, S.; Fu, J.; Zhang, Y.; Dai, N.; Song, D.; Ding, H. Microbial-Mediated Emissions of Greenhouse Gas from Farmland Soils: A Review. Processes 2022, 10, 2361. https://doi.org/10.3390/pr10112361
Wang H, Zhao R, Zhao D, Liu S, Fu J, Zhang Y, Dai N, Song D, Ding H. Microbial-Mediated Emissions of Greenhouse Gas from Farmland Soils: A Review. Processes. 2022; 10(11):2361. https://doi.org/10.3390/pr10112361
Chicago/Turabian StyleWang, Han, Rui Zhao, Dan Zhao, Shejiang Liu, Jianfeng Fu, Yuxin Zhang, Nan Dai, Dan Song, and Hui Ding. 2022. "Microbial-Mediated Emissions of Greenhouse Gas from Farmland Soils: A Review" Processes 10, no. 11: 2361. https://doi.org/10.3390/pr10112361
APA StyleWang, H., Zhao, R., Zhao, D., Liu, S., Fu, J., Zhang, Y., Dai, N., Song, D., & Ding, H. (2022). Microbial-Mediated Emissions of Greenhouse Gas from Farmland Soils: A Review. Processes, 10(11), 2361. https://doi.org/10.3390/pr10112361











