Lentinan Impairs the Early Development of Zebrafish Embryos, Possibly by Disrupting Glucose and Lipid Metabolism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Research Compound
2.2. Zebrafish Maintenance and Embryo Collection
2.3. Embryo Acute Toxicity Test
2.4. Pathological Alteration Analyses
2.5. Biochemical Indexes Determination
2.6. Quantitative Real Time-Polymerase Chain Reaction (qRT-PCR) Analyses
2.7. Statistical Analysis
3. Results
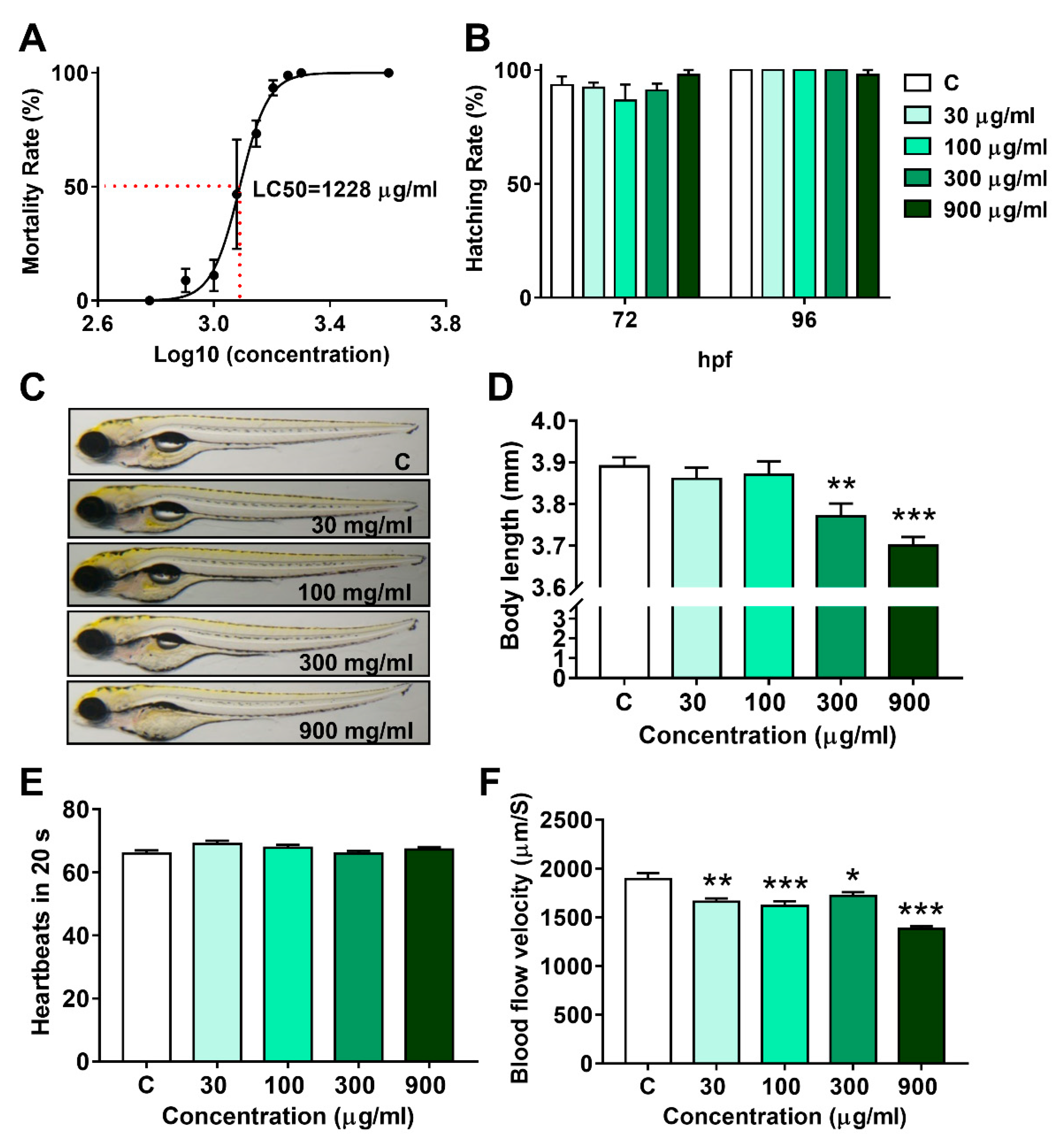
3.1. Lethal Effect and Developmental Toxicity of LNT in Larval Zebrafish
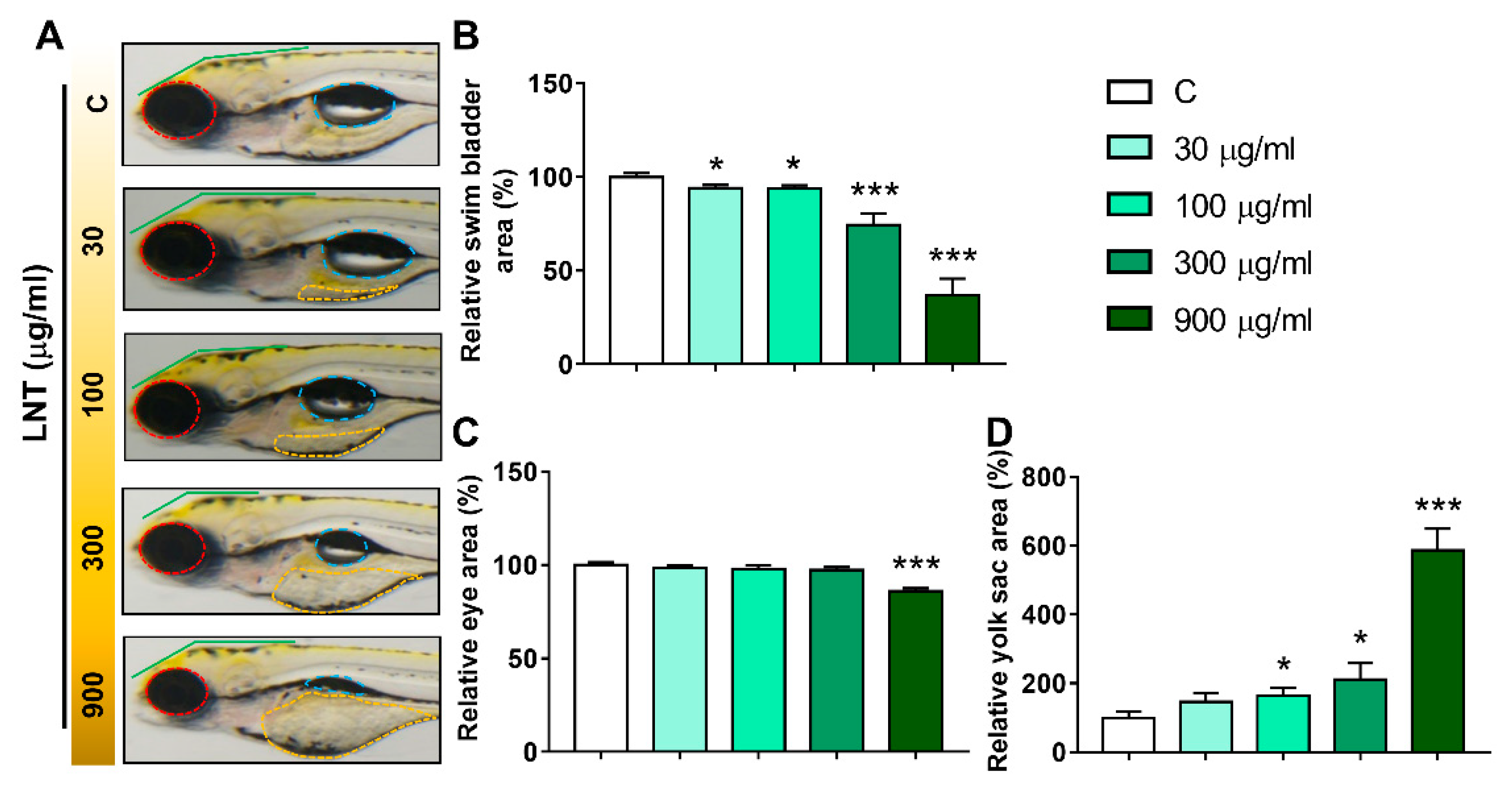
3.2. LNT Induced Pathological Alterations in Larval Zebrafish
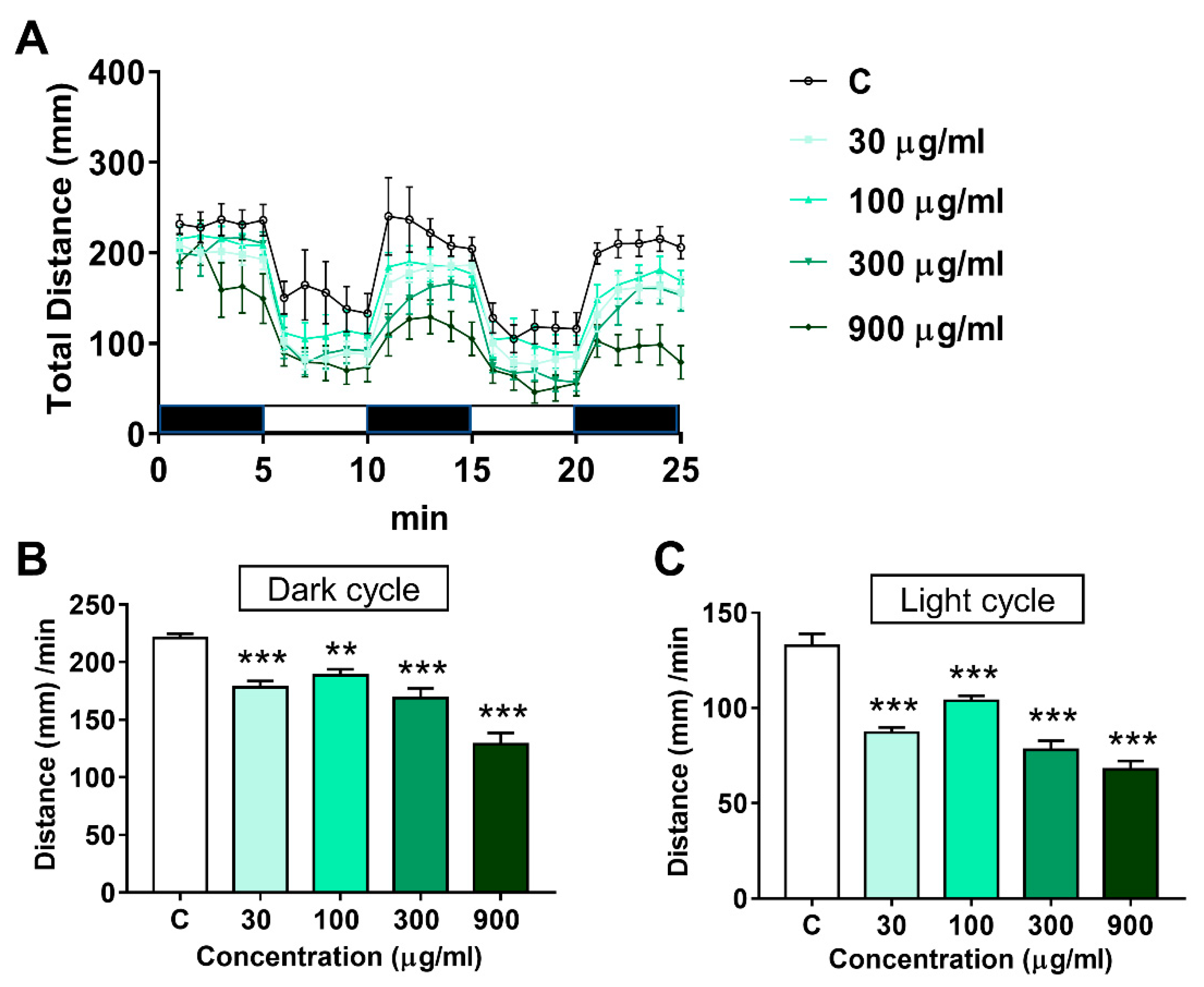
3.3. Effects of LNT Exposure on the Locomotor Activity in Larval Zebrafish
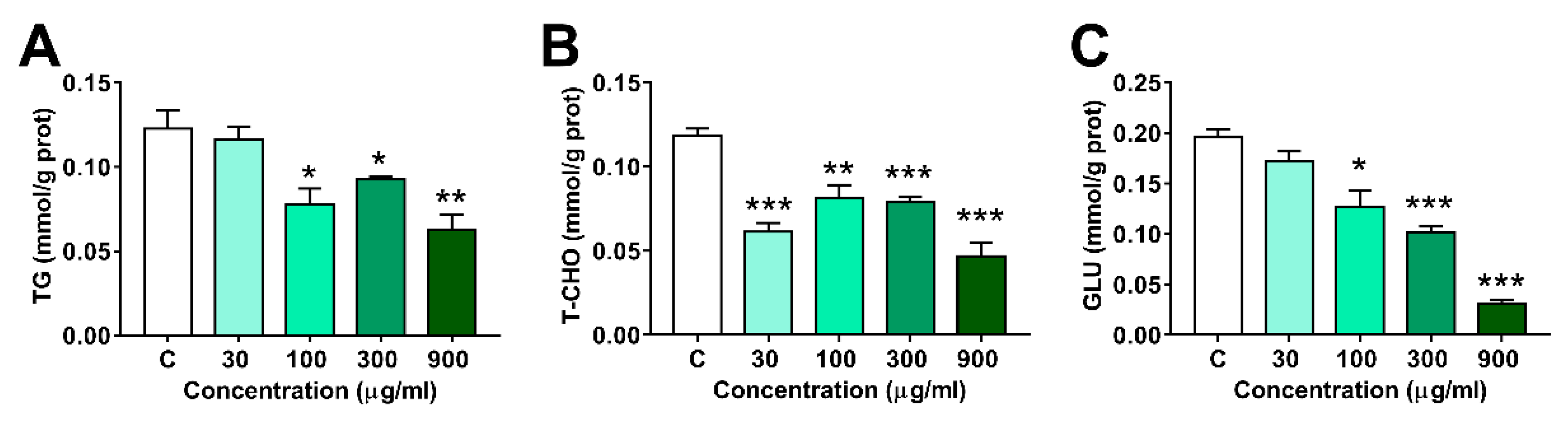
3.4. Effects of LNT Exposure on the Glu, TG and T-CHO Levels in Larval Zebrafish
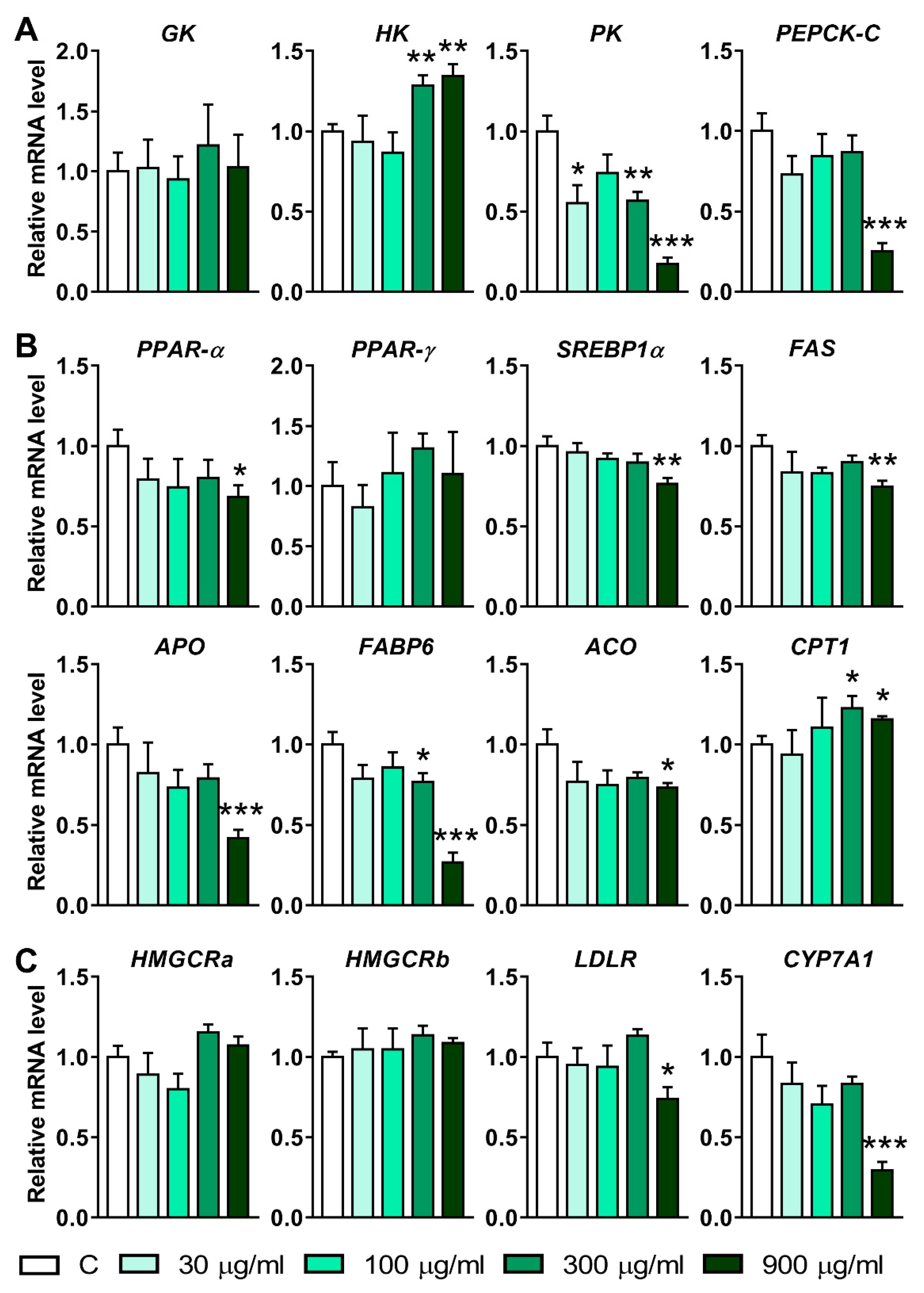
3.5. Effects of LNT Exposure on the Genes Involved in Glucose, Lipid and Cholesterol Metabolism in Larval Zebrafish
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, Y.; Jin, H.; Yu, J.; Qu, C.; Wang, Q.; Yang, S.; Ma, S.; Ni, J. Quality control and immunological activity of lentinan samples produced in China. Int. J. Biol. Macromol. 2020, 159, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Zhang, G.; Kuai, J.; Fan, P.; Wang, X.; Zhou, P.; Yang, D.; Zheng, X.; Liu, X.; Wu, Q.; et al. Lentinan inhibits tumor angiogenesis via interferon γ and in a T cell independent manner. J. Exp. Clin. Cancer Res. 2018, 37, 260. [Google Scholar] [CrossRef] [Green Version]
- Maeda, Y.Y.; Hamuro, J.; Chihara, G. The mechanisms of action of anti-tumour polysaccharides. I. The effects of antilymphocyte serum on the anti-tumour activity of lentinan. Int. J. Cancer 1971, 8, 41–46. [Google Scholar] [CrossRef]
- Sasaki, T.; Takasuka, N. Further study of the structure of lentinan, an anti-tumor polysaccharide from Lentinus edodes. Carbohydr. Res. 1976, 47, 99–104. [Google Scholar] [CrossRef]
- Haba, S.; Hamaoka, T.; Takatsu, K.; Kitagawa, M. Selective suppression of t-cell activity in tumor-bearing mice and its improvement by lentinan, a potent anti-tumor polysaccharide. Int. J. Cancer 1976, 18, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Kozarski, M.; Klaus, A.; Nikšić, M.; Vrvić, M.M.; Todorović, N.; Jakovljević, D.; Van Griensven, L.J.L.D. Antioxidative activities and chemical characterization of polysaccharide extracts from the widely used mushrooms Ganoderma applanatum, Ganoderma lucidum, Lentinus edodes and Trametes versicolor. J. Food Compos. Anal. 2012, 26, 144–153. [Google Scholar] [CrossRef]
- Morales, D.; Rutckeviski, R.; Villalva, M.; Abreu, H.; Soler-Rivas, C.; Santoyo, S.; Iacomini, M.; Smiderle, F.R. Isolation and comparison of α- and β-Dglucans from shiitake mushrooms (Lentinula edodes) with different biological activities. Carbohydr. Polym. 2020, 229, 115521. [Google Scholar] [CrossRef] [PubMed]
- Hatvani, N. Antibacterial effect of the culture fluid of Lentinus edodes mycelium grown in submerged liquid culture. Int. J. Antimicrob. Agents 2001, 17, 71–74. [Google Scholar] [CrossRef]
- Zhao, Y.M.; Yang, J.M.; Liu, Y.H.; Zhao, M.; Wang, J. Ultrasound assisted extraction of polysaccharides from Lentinus edodes and its anti-hepatitis B activity in vitro. Int. J. Biol. Macromol. 2018, 107, 2217–2223. [Google Scholar] [CrossRef]
- Han, D.; Lee, H.T.; Lee, J.B.; Kim, Y.; Lee, S.J.; Yoon, J.W. A Bioprocessed Polysaccharide from Lentinus edodes Mycelia Cultures with Turmeric Protects Chicks from a Lethal Challenge of Salmonella gallinarum. J. Food Prot. 2017, 80, 245–250. [Google Scholar] [CrossRef]
- Song, X.; Ren, Z.; Wang, X.; Jia, L.; Zhang, C. Antioxidant, anti-inflammatory and renoprotective effects of acidic-hydrolytic polysaccharides by spent mushroom compost (Lentinula edodes) on LPS-induced kidney injury. Int. J. Biol. Macromol. 2020, 151, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Li, J.; Kong, F.; Lin, J.; Gao, Y. Induction of immunomodulating cytokines by a new polysaccharide-peptide complex from culture mycelia of Lentinus edodes. Immunopharmacology 1998, 40, 187–198. [Google Scholar] [CrossRef]
- Jeff, I.B.; Fan, E.; Tian, M.; Song, C.; Yan, J.; Zhou, Y. In vivo anticancer and immunomodulating activities of mannogalactoglucan-type polysaccharides from Lentinus edodes (Berkeley) Singer. Cent. Eur. J. Immunol. 2016, 41, 47–53. [Google Scholar] [CrossRef]
- Wang, T.; He, H.; Liu, X.; Liu, C.; Liang, Y.; Mei, Y. Mycelial polysaccharides of Lentinus edodes (shiitake mushroom) in submerged culture exert immunoenhancing effect on macrophage cells via MAPK pathway. Int. J. Biol. Macromol. 2019, 130, 745–754. [Google Scholar] [CrossRef]
- Chen, S.; Liu, C.; Huang, X.; Hu, L.; Huang, Y.; Chen, H.; Fang, Q.; Dong, N.; Li, M.; Tang, W.; et al. Comparison of immunomodulatory effects of three polysaccharide fractions from Lentinula edodes water extracts. J. Funct. Foods 2020, 66, 103791. [Google Scholar] [CrossRef]
- Murphy, E.A.; Davis, J.M.; Carmichael, M.D. Immune modulating effects of beta-glucan. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Shao, Y.; Wang, Z.; Guo, Y. Effects of dietary yeast β-glucans supplementation on growth performance, gut morphology, intestinal Clostridium perfringens population and immune response of broiler chickens challenged with necrotic enteritis. Anim. Feed Sci. Technol. 2016, 215, 144–155. [Google Scholar] [CrossRef]
- Vetvicka, V.; Oliveira, C. beta(1-3)(1-6)-d-Glucans modulate immune status in pigs: Potential importance for efficiency of commercial farming. Ann. Transl. Med. 2014, 2, 16. [Google Scholar] [PubMed]
- Pilarski, F.; Ferreira de Oliveira, C.A.; Darpossolo de Souza, F.P.B.; Zanuzzo, F.S. Different beta-glucans improve the growth performance and bacterial resistance in Nile tilapia. Fish Shellfish Immunol. 2017, 70, 25–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.S.; Liau, S.Y.; Huang, C.T.; Nan, F.H. Beta 1,3/1,6-glucan and vitamin C immunostimulate the non-specific immune response of white shrimp (Litopenaeus vannamei). Fish Shellfish Immunol. 2016, 57, 269–277. [Google Scholar] [CrossRef]
- Kim, Y.S.; Ryu, J.H.; Han, S.J.; Choi, K.H.; Nam, K.B.; Jang, I.H.; Lemaitre, B.; Brey, P.T.; Lee, W.J. Gram-negative bacteria-binding protein, a pattern recognition receptor for lipopolysaccharide and beta-1,3-glucan that mediates the signaling for the induction of innate immune genes in Drosophila melanogaster cells. J. Biol. Chem. 2000, 275, 32721–32727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ina, K.; Kataoka, T.; Ando, T. The Use of Lentinan for Treating Gastric Cancer. Anticancer Agents Med. Chem. 2013, 13, 681–688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, L.J.; Qi, M.; Li, N.; Lei, Y.H.; Zhang, D.M.; Chen, J.X. Natural products and their derivatives: Promising modulators of tumor immunotherapy. J. Leukoc. Biol. 2020, 108, 493–508. [Google Scholar] [CrossRef]
- Liu, Z.; He, J.; Zhu, T.; Hu, C.; Bo, R.; Wusiman, A.; Hu, Y.; Wang, D. Lentinan-Functionalized Graphene Oxide Is an Effective Antigen Delivery System That Modulates Innate Immunity and Improves Adaptive Immunity. ACS Appl. Mater. Interfaces 2020, 12, 39014–39023. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Zhou, Y.; Zheng, Z.; Tang, W.; Song, M.; Wang, J.; Wang, K. Lentinan inhibited colon cancer growth by inducing endoplasmic reticulum stress-mediated autophagic cell death and apoptosis. Carbohydr. Polym. 2021, 267, 118154. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the safety of “Lentinus edodes extract” as a Novel Food ingredient. EFSA J. 2010, 8, 1685. [Google Scholar] [CrossRef]
- Moriyuki, H.; Ichimura, M. Acute toxicity of lentinan in mice and rats. J. Toxicol. Sci. 1980, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Shimazu, H.; Takeda, K.; Onodera, C.; Makita, I.; Hashi, T.; Yamazoe, T.; Kokuba, Y.; Tanigawa, H.; Ohkuma, S.; Shinpo, K.; et al. Intravenous chronic toxicity of lentinan in rats: 6-month treatment and 3-month recovery. J. Toxicol. Sci. 1980, 5, 33–57. [Google Scholar] [CrossRef]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Universities Federation for Animal Welfare: Wheathamsted, UK, 1959. [Google Scholar]
- Sewell, F.; Edwards, J.; Prior, H.; Robinson, S. Opportunities to Apply the 3Rs in Safety Assessment Programs. ILAR J. 2016, 57, 234–245. [Google Scholar] [CrossRef] [Green Version]
- Geisler, R.; Köhler, A.; Dickmeis, T.; Strähle, U. Archiving of zebrafish lines can reduce animal experiments in biomedical research. EMBO Rep. 2017, 18, 1–2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Wei, L.; Wang, Y.; He, B.; Kong, B.; Zhu, J.; Jin, Y.; Fu, Z. Evaluation of development, locomotor behavior, oxidative stress, immune responses and apoptosis in developing zebrafish (Danio rerio) exposed to TBECH (tetrabromoethylcyclohexane). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2018, 217, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, R.; Jin, Y. Differential responses of larval zebrafish to the fungicide propamocarb: Endpoints at development, locomotor behavior and oxidative stress. Sci. Total Environ. 2020, 731, 139136. [Google Scholar] [CrossRef] [PubMed]
- Weng, Y.; Huang, Z.; Wu, A.; Yu, Q.; Lu, H.; Lou, Z.; Lu, L.; Bao, Z.; Jin, Y. Embryonic toxicity of epoxiconazole exposure to the early life stage of zebrafish. Sci. Total Environ. 2021, 778, 146407. [Google Scholar] [CrossRef]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- OECD. OECD Guidelines for the Testing of Chemicals. Fish Embryo Acute Toxicity (FET) Test; OECD: Paris, France, 2013. [Google Scholar]
- Mu, X.; Wang, K.; Chai, T.; Zhu, L.; Yang, Y.; Zhang, J.; Pang, S.; Wang, C.; Li, X. Sex specific response in cholesterol level in zebrafish (Danio rerio) after long-term exposure of difenoconazole. Environ. Pollut. 2015, 197, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Qi, S.; Wang, D.; Wang, K.; Zhu, L.; Chai, T.; Wang, C. Toxic effects of thifluzamide on zebrafish (Danio rerio). J. Hazard. Mater. 2016, 307, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Cocco, A.; Ronnberg, A.M.; Jin, Z.; Andre, G.I.; Vossen, L.E.; Bhandage, A.K.; Thörnqvist, P.-E.; Birnir, B.; Winberg, S. Characterization of the gamma-aminobutyric acid signaling system in the zebrafish (Danio rerio Hamilton) central nervous system by reverse transcription-quantitative polymerase chain reaction. Neuroscience 2017, 343, 300–321. [Google Scholar] [CrossRef] [PubMed]
- Al-Habsi, A.A.; Massarsky, A.; Moon, T.W. Exposure to gemfibrozil and atorvastatin affects cholesterol metabolism and steroid production in zebrafish (Danio rerio). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2016, 199, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Bao, Z.; Wan, Z.; Fu, Z.; Jin, Y. Polystyrene microplastic exposure disturbs hepatic glycolipid metabolism at the physiological, biochemical, and transcriptomic levels in adult zebrafish. Sci. Total Environ. 2020, 710, 136279. [Google Scholar] [CrossRef] [PubMed]
- Markman, M. Safety issues in using complementary and alternative medicine. J. Clin. Oncol. 2002, 20, 39–41. [Google Scholar]
- Lahlou, M. Screening of natural products for drug discovery. Expert Opin. Drug Discov. 2007, 2, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Willett, C.; Fremgen, T. Zebrafish: An Animal Model for Toxicological Studies. Curr. Protoc. Toxicol. 2003, 17, 1.7.1–1.7.18. [Google Scholar] [CrossRef] [PubMed]
- Wan-Mohtar, W.A.A.Q.I.; Ilham, Z.; Jamaludin, A.A.; Rowan, N. Use of Zebrafish Embryo Assay to Evaluate Toxicity and Safety of Bioreactor-Grown Exopolysaccharides and Endopolysaccharides from European Ganoderma applanatum Mycelium for Future Aquaculture Applications. Int. J. Mol. Sci. 2021, 22, 1675. [Google Scholar] [CrossRef] [PubMed]
- Taufek, N.M.; Harith, H.H.; Abd Rahim, M.H.; Ilham, Z.; Rowan, N.; Wan, W.A.A.Q.I. Performance of mycelial biomass and exopolysaccharide from Malaysian Ganoderma lucidum for the fungivore red hybrid Tilapia (Oreochromis sp.) in Zebrafish embryo. Aquac. Rep. 2020, 17, 100322. [Google Scholar] [CrossRef]
- Park, H.; Lee, J.Y.; Park, S.; Song, G.; Lim, W. Developmental toxicity of fipronil in early development of zebrafish (Danio rerio) larvae: Disrupted vascular formation with angiogenic failure and inhibited neurogenesis. J. Hazard Mater. 2020, 385, 121531. [Google Scholar] [CrossRef]
- Zhu, J.J.; Thompson, C.B. Metabolic regulation of cell growth and proliferation. Nat. Rev. Mol. Cell Biol. 2019, 20, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-Y.; Li, N.-J.; Cheng, F.-Y.; Hsueh, J.-F.; Huang, C.-C.; Lu, F.-I.; Fu, T.-F.; Yan, S.-J.; Lee, Y.-H.; Wang, Y.-J. The Effect of the Chorion on Size-Dependent Acute Toxicity and Underlying Mechanisms of Amine-Modified Silver Nanoparticles in Zebrafish Embryos. Int. J. Mol. Sci. 2020, 21, 2864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brodin, T.; Piovano, S.; Fick, J.; Klaminder, J.; Heynen, M.; Jonsson, M. Ecological effects of pharmaceuticals in aquatic systems—impacts through behavioural alterations. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130580. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Liang, X.; Zhao, Y.; Gu, X.; Mao, Z.; Zeng, Q.; Chen, H.; Martyniuk, C.J. Molecular and behavioral responses of zebrafish embryos/larvae after sertraline exposure. Ecotoxicol. Environ. Saf. 2021, 208, 111700. [Google Scholar] [CrossRef]
- Selderslaghs, I.W.; Hooyberghs, J.; De Coen, W.; Witters, H.E. Locomotor activity in zebrafish embryos: A new method to assess developmental neurotoxicity. Neurotoxicol. Teratol. 2010, 32, 460–471. [Google Scholar] [CrossRef]
- Nafikov, R.; Beitz, D.C. Carbohydrate and Lipid Metabolism in Farm Animals. J. Nutr. 2007, 137, 702–705. [Google Scholar] [CrossRef] [Green Version]
- Behall, K.M.; Scholfield, D.J.; Hallfrisch, J.G.; Liljeberg-Elmståhl, H.G. Consumption of Both Resistant Starch and -Glucan Improves Postprandial Plasma Glucose and Insulin in Women. Diabetes Care 2006, 29, 976–981. [Google Scholar] [CrossRef]
- Granfeldt, Y.; Nyberg, L.; Björck, I. Muesli with 4 g oat β-glucans lowers glucose and insulin responses after a bread meal in healthy subjects. Eur. J. Clin. Nutr. 2007, 62, 600–607. [Google Scholar] [CrossRef] [Green Version]
- El Khoury, D.; Cuda, C.; Luhovyy, B.L.; Anderson, G.H. Beta Glucan: Health Benefits in Obesity and Metabolic Syndrome. J. Nutr. Metab. 2012, 2012, 851362. [Google Scholar] [CrossRef] [Green Version]
- Aoe, S.; Mio, K.; Yamanaka, C.; Kuge, T. Low Molecular Weight Barley β-Glucan Affects Glucose and Lipid Metabolism by Prebiotic Effects. Nutrients 2021, 13, 130. [Google Scholar] [CrossRef]
- Tan, V.P.; Miyamoto, S. HK2/hexokinase-II integrates glycolysis and autophagy to confer cellular protection. Autophagy 2015, 11, 963–964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méndez-Lucas, A.; Duarte, J.A.; Sunny, N.E.; Satapati, S.; He, T.; Fu, X.; Bermúdez, J.; Burgess, S.C.; Perales, J.C. Share PEPCK-M expression in mouse liver potentiates, not replaces, PEPCK-C mediated gluconeogenesis. J. Hepatol. 2013, 59, 105–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schormann, N.; Hayden, K.L.; Lee, P.; Banerjee, S.; Chattopadhyay, D. An overview of structure, function, and regulation of pyruvate kinases. Protein Sci. 2019, 28, 1771–1784. [Google Scholar] [CrossRef]
- Wolever, T.M.; Tosh, S.M.; Gibbs, A.L.; Brand-Miller, J.; Duncan, A.M.; Hart, V.; Lamarche, B.; Thomson, B.A.; Duss, R.; Wood, P.J. Physicochemical properties of oat beta-glucan influence its ability to reduce serum LDL cholesterol in humans: A randomized clinical trial. Am. J. Clin. Nutr. 2010, 92, 723–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Harding, S.V.; Eck, P.; Thandapilly, S.J.; Gamel, T.H.; Abdel-Aal, E.S.M.; Crow, G.H.; Tosh, S.M.; Jones, P.J.H.; Ames, N.P. High-molecular-weight beta-glucan decreases serum cholesterol differentially based on the CYP7A1 rs3808607 polymorphism in mildly hypercholesterolemic adults. J. Nutr. 2016, 146, 720–727. [Google Scholar] [CrossRef]
- Wang, Y.; Harding, S.V.; Thandapilly, S.J.; Tosh, S.M.; Jones, P.J.; Ames, N.P. Barley β-glucan reduces blood cholesterol levels via interrupting bile acid metabolism. Br. J. Nutr. 2017, 118, 822–829. [Google Scholar] [CrossRef] [Green Version]
- Gunness, P.; Gidley, M.J. Mechanisms underlying the cholesterol-lowering properties of soluble dietary fibre polysaccharides. Food Funct. 2010, 1, 149–155. [Google Scholar] [CrossRef]
- Ina, K.; Furuta, R.; Kataoka, T.; Kayukawa, S.; Yoshida, T.; Miwa, T.; Yamamura, Y.; Takeuchi, Y. Lentinan prolonged survival in patients with gastric cancer receiving S-1-based chemotherapy. World J. Clin. Oncol. 2011, 2, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Oba, K.; Kobayashi, M.; Matsui, T.; Kodera, Y.; Sakamoto, J. Individual patient based meta-analysis of lentinan for unresectable/recurrent gastric cancer. Anticancer. Res. 2009, 29, 2739–2745. [Google Scholar]
- Sima, P.; Vannucci, L.; Vetvicka, V. β-glucans and cholesterol (Review). Int. J. Mol. Med. 2018, 41, 1799–1808. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wouk, J.; Dekker, R.F.H.; Queiroz, E.A.I.F.; Barbosa-Dekker, A.M. β-Glucans as a panacea for a healthy heart? Their roles in preventing and treating cardiovascular diseases. Int. J. Biol. Macromol. 2021, 177, 176–203. [Google Scholar] [CrossRef] [PubMed]
- Morales, D.; Shetty, S.A.; López-Plaza, B.; Gómez-Candela, C.; Smidt, H.; Marín, F.R.; Soler-Rivas, C. Modulation of human intestinal microbiota in a clinical trial by consumption of a β-d-glucan-enriched extract obtained from Lentinula edodes. Eur. J. Nutr. 2021, 60, 3249–3265. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miao, W.; He, L.; Zhang, T.; Li, C. Lentinan Impairs the Early Development of Zebrafish Embryos, Possibly by Disrupting Glucose and Lipid Metabolism. Processes 2022, 10, 120. https://doi.org/10.3390/pr10010120
Miao W, He L, Zhang T, Li C. Lentinan Impairs the Early Development of Zebrafish Embryos, Possibly by Disrupting Glucose and Lipid Metabolism. Processes. 2022; 10(1):120. https://doi.org/10.3390/pr10010120
Chicago/Turabian StyleMiao, Wenyu, Lingling He, Tao Zhang, and Chunqi Li. 2022. "Lentinan Impairs the Early Development of Zebrafish Embryos, Possibly by Disrupting Glucose and Lipid Metabolism" Processes 10, no. 1: 120. https://doi.org/10.3390/pr10010120
APA StyleMiao, W., He, L., Zhang, T., & Li, C. (2022). Lentinan Impairs the Early Development of Zebrafish Embryos, Possibly by Disrupting Glucose and Lipid Metabolism. Processes, 10(1), 120. https://doi.org/10.3390/pr10010120





