Temporal and Atemporal Provider Network Analysis in a Breast Cancer Cohort from an Academic Medical Center (USA)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Network Representation
2.3. Network Analysis
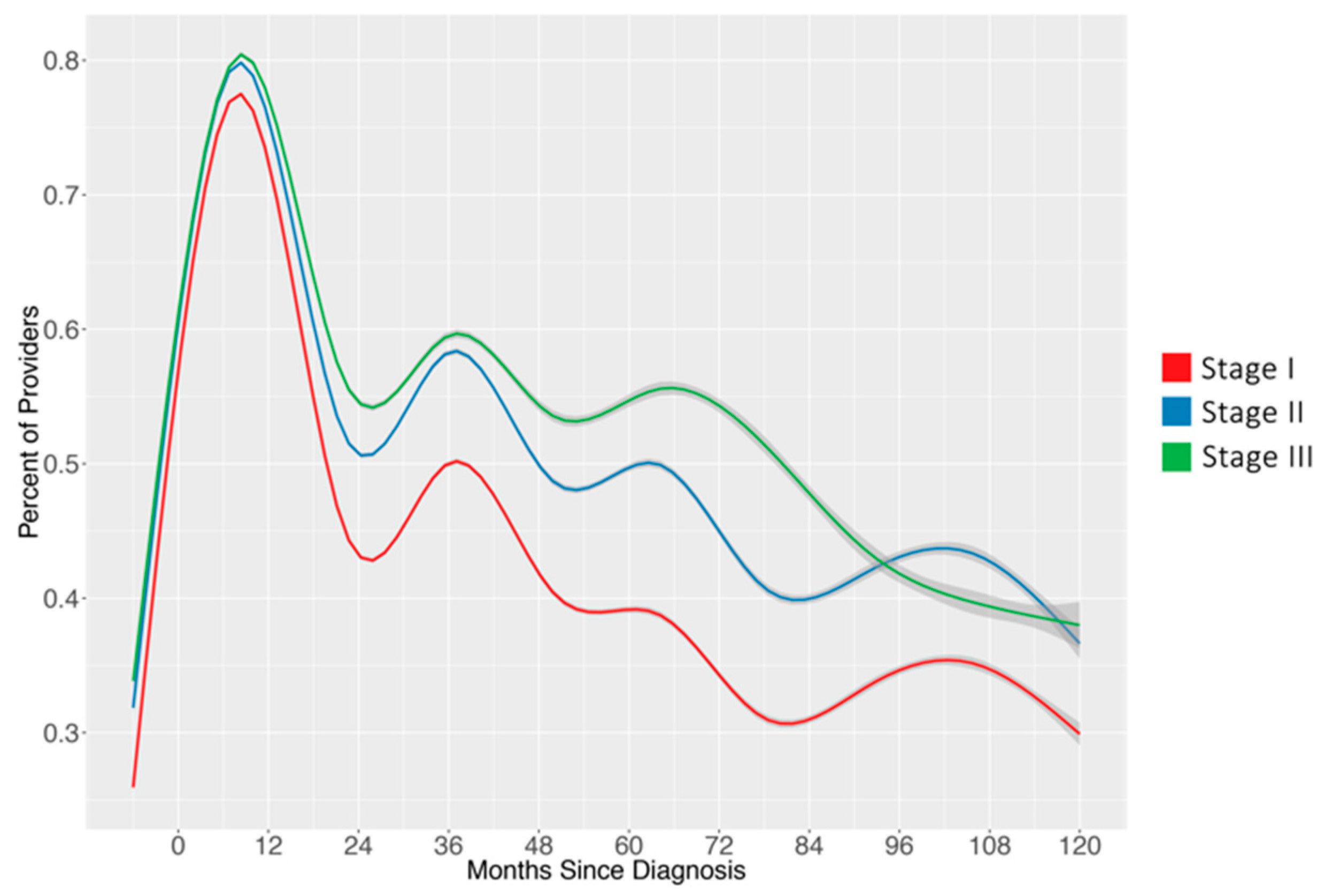
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- van Beek, A.P. Communication, advice exchange and job satisfaction of nursing staff: A social network analyses of 35 long-term care units. BMC Health Serv. Res. 2011, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Creswick, N.; Westbrook, J.I.; Braithwaite, J. Understanding communication networks in the emergency department. BMC Health Serv. Res. 2009, 9, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Alexander, G.L.; Steege, L.M.; Pasupathy, K.S.; Wise, K. Case studies of IT sophistication in nursing homes: A mixed method approach to examine communication strategies about pressure ulcer prevention practices. Int. J. Ind. Ergon. 2015, 49, 156–166. [Google Scholar] [CrossRef]
- Alexander, G.L.; Pasupathy, K.S.; Steege, L.M.; Strecker, E.B.; Carley, K.M. Multi-disciplinary communication networks for skin risk assessment in nursing homes with high IT sophistication. Int. J. Ind. Ergon. 2014, 83, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Steitz, B.D.; Levy, M.A. A social network analysis of cancer provider collaboration. AMIA Annu. Symp. Proc. 2016, 2016, 1987–1996. [Google Scholar] [PubMed]
- Barnett, M.L.; Landon, B.E.; O’Malley, A.J.; Keating, N.L.; Christakis, N.A. Mapping physician networks with self-reported and administrative data. Health Serv. Res. 2011, 46, 1592–1609. [Google Scholar] [CrossRef] [PubMed]
- Boyer, L.; Belzeaux, R.; Maurel, O.; Baumstarck Barrau, K.; Samuelian, J.C. A social network analysis of healthcare professional relationships in a French hospital. Int. J. Health Care Qual. Assur. 2010, 23, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Rangachari, P. Knowledge sharing networks related to hospital quality measurement and reporting. Health Care Manag. Rev. 2008, 33, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Wiemken, T.L.W.P.M.; Ramirez, J.A.R.M.; Polgreen, P.P.M.; Peyrani, P.P.; Carrico, R.M.C.P.R.F. Evaluation of the knowledge-sharing social network of hospital-based infection preventionists in Kentucky. Am. J. Infect. Control 2012, 40, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Menchik, D.A.; Meltzer, D.O. The cultivation of esteem and retrieval of scientific knowledge in physician networks. J. Health Soc. Behav. 2010, 51, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Creswick, N.; Westbrook, J.I. Social network analysis of medication advice-seeking interactions among staff in an Australian hospital. Int. J. Med. Inform. 2010, 79, e116–e125. [Google Scholar] [CrossRef] [PubMed]
- Sabot, K.; Wickremasinghe, D.; Blanchet, K.; Avan, B.; Schellenberg, J. Use of social network analysis methods to study professional advice and performance among healthcare providers: A systematic review. Syst. Rev. 2017, 6, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Barnett, M.L.; Christakis, N.A.; O’Malley, J.; Onnela, J.-P.; Keating, N.L.; Landon, B.E. Physician patient-sharing networks and the cost and intensity of care in US hospitals. Med. Care 2012, 50, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Bridewell, W.; Das, A.K. Social network analysis of physician interactions: The effect of institutional boundaries on breast cancer care. AMIA Annu. Symp. Proc. 2011, 2011, 152–160. [Google Scholar]
- Iwashyna, T.J.; Christie, J.D.; Moody, J.; Kahn, J.M.; Asch, D.A. The structure of critical care transfer networks. Med. Care 2009, 47, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Chambers, D.; Wilson, P.; Thompson, C.; Harden, M. Social network analysis in healthcare settings: A systematic scoping review. PLoS ONE 2012, 7, e41911. [Google Scholar] [CrossRef] [PubMed]
- Wasserman, S.; Faust, K. Social Network Analysis: Methods and Applications; Cambridge University Press: New York, NY, USA, 1994. [Google Scholar]
- O’Malley, A.J.; Marsden, P.V. The analysis of social networks. Health Serv. Outcomes Res. Method 2008, 8, 222–269. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Rosen, D.W.; Wang, L.; Schaefer, D. Cloud-based design and manufacturing: A new paradigm in digital manufacturing and design innovation. Comput. Aided Des. 2015, 59, 1–14. [Google Scholar] [CrossRef]
- Casper, S. How do technology clusters emerge and become sustainable? Res. Policy 2007, 36, 438–455. [Google Scholar] [CrossRef]
- Cross, R.; Borgatti, S.P.; Parker, A. Making invisible work visible: Using social network analysis to support strategic collaboration. Calif. Manag. Rev. 2002, 44, 25–46. [Google Scholar] [CrossRef]
- Bonchi, F.; Castillo, C.; Gionis, A.; Jaimes, A. Social network analysis and mining for business applications. ACM Trans. Intell. Syst. Technol. 2011, 2, 1–37. [Google Scholar] [CrossRef]
- Larruscain, J.; Río-Belver, R.; Arraibi, J.R.; Garechana, G. Efficiency in knowledge transmission in R&D project networks: European renewable energy sector. J. Renew. Sustain. Energy 2017, 9, 065908. [Google Scholar]
- Latora, V.; Marchiori, M. Is the Boston subway a small-world network? Phys. A Stat. Mech. Appl. 2002, 314, 109–113. [Google Scholar] [CrossRef]
- Tichy, N.M.; Tushman, M.L.; Fombrun, C. Social network analysis for organizations. Acad. Manag. Rev. 1979, 4, 507–519. [Google Scholar] [CrossRef]
- Oborn, E.; Dawson, S. Knowledge and practice in multidisciplinary teams: Struggle, accommodation and privilege. Hum. Relat. 2010, 63, 1835–1857. [Google Scholar] [CrossRef]
- Hartgerink, J.M.; Cramm, J.M.; Bakker, T.J.E.M.; van Eijsden, A.M.; Mackenbach, J.P.; Nieboer, A.P. The importance of multidisciplinary teamwork and team climate for relational coordination among teams delivering care to older patients. J. Adv. Nurs. 2013, 70, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Schepman, S.; Hansen, J.; de Putter, I.D.; Batenburg, R.S.; de Bakker, D.H. The common characteristics and outcomes of multidisciplinary collaboration in primary health care: A systematic literature review. Int. J. Integr. Care 2015, 15, e027. [Google Scholar] [CrossRef] [PubMed]
- Committee on Quality of Health Care in America, Institute of Medicine (US). Crossing the Quality Chasm: A New Health System for the 21st Century; The National Academies Press: Washington, DC, USA, 2001.
- Shannon, D. Effective physician-to-physician communication: An essential ingredient for care coordination. Phys. Exec. 2012, 38, 16–21. [Google Scholar]
- Kesson, E.M.; Allardice, G.M.; George, W.D.; Burns, H.J.G.; Morrison, D.S. Effects of multidisciplinary team working on breast cancer survival: Retrospective, comparative, interventional cohort study of 13,722 women. BMJ 2012, 344, e2718. [Google Scholar] [CrossRef] [PubMed]
- McDonald, K.M.; Sundaram, V.; Bravata, D.M.; Lewis, R.; Lin, N.; Kraft, S.A.; McKinnon, M.; Paguntalan, H.; Owens, D.K. Closing the Quality Gap: A Critical Analysis of Quality Improvement Strategies; Agency for Healthcare Research and Quality (AHRQ): Rockville, MD, USA, 2007; Volume 7.
- Peikes, D.; Chen, A.; Schore, J.; Brown, R. Effects of care coordination on hospitalization, quality of care, and health care expenditures among medicare beneficiaries. JAMA 2009, 301, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Forster, A.J. The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann. Intern. Med. 2003, 138, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Casteigts, A.; Flocchini, P.; Quattrociocchi, W.; Santoro, N. Time-varying graphs and dynamic networks. Int. J. Parallel Emerg. Distrib. Syst. 2012, 27, 387–408. [Google Scholar] [CrossRef]
- Cancer Registry. Available online: https://seer.cancer.gov/registries/cancer_registry/cancer_registry.html (accessed on 30 July 2018).
- Tennessee Cancer Registry. Available online: https://www.tn.gov/health/health-program-areas/tcr.html (accessed on 30 July 2018).
- Pollack, C.E.; Weissman, G.E.; Lemke, K.W.; Hussey, P.S.; Weiner, J.P. Patient sharing among physicians and costs of care: A network analytic approach to care coordination using claims data. J. Gen. Intern. Med. 2012, 28, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Csardi, G.; Nepusz, T. The igraph software package for complex network research. InterJournal Complex Syst. 2006, 1695, 1–9. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing, version 3.0; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Samarth, C.N.; Gloor, P.A. Process efficiency. Redesigning social networks to improve surgery patient flow. J. Healthc. Inf. Manag. 2009, 23, 20–26. [Google Scholar] [PubMed]
- Merrill, J.A.; Sheehan, B.M.; Carley, K.M.; Stetson, P.D. Transition networks in a cohort of patients with congestive heart failure. Appl. Clin. Inform. 2015, 6, 548–564. [Google Scholar] [PubMed]
- Chen, Y.; Kho, A.N.; Liebovitz, D.; Ivory, C.; Osmundson, S.; Bian, J.; Malin, B.A. Learning bundled care opportunities from electronic medical records. J. Biomed. Inform. 2018, 77, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xie, W.; Gunter, C.A.; Liebovitz, D.; Mehrotra, S.; Zhang, H.; Malin, B. Inferring clinical workflow efficiency via electronic medical record utilization. AMIA Annu. Symp. Proc. 2015, 2015, 416–425. [Google Scholar] [PubMed]
- Fowler, J.H.; Christakis, N.A. Dynamic spread of happiness in a large social network: Longitudinal analysis over 20 years in the Framingham Heart Study. BMJ 2008, 337, a2338. [Google Scholar] [CrossRef] [PubMed]
- Christakis, N.A.; Fowler, J.H. The collective dynamics of smoking in a large social network. N. Engl. J. Med. 2008, 358, 2249–2258. [Google Scholar] [CrossRef] [PubMed]
- Christakis, N.A.; Fowler, J.H. Social contagion theory: Examining dynamic social networks and human behavior. Stat. Med. 2012, 32, 556–577. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, K.; James, P. How to do (or not to do) … a social network analysis in health systems research. Health Policy Plan. 2012, 27, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Srinivasan, U.; Uddin, S.; Chawla, S. Application of network analysis on healthcare. In Proceedings of the IEEE/ACM International Conference on Advances in Social Network Analysis and Mining, Beijing, China, 17–20 August 2014; pp. 596–603. [Google Scholar]
- Steitz, B.D.; Weinberg, S.T.; Danciu, I.; Unertl, K.M. Managing and communicating operational workflow. Appl. Clin. Inform. 2016, 7, 59–68. [Google Scholar] [PubMed]


| Calculation | Definition | Interpretation | |
|---|---|---|---|
| Network Density | The percentage of potential connections in a network that are actual connections. | A measure to quantify the relative degree of connectivity within a network. | |
| Network Care Density | The average number of patients shared per provider connection. | A measure to quantify the amount of patient sharing between providers in a network. | |
| Degree Centrality | The sum of unique connections adjacent to a single node. | The total number of connections associated with a single node. | The number of providers who share a patient with a single provider of interest. |
| Temporal Edge | Pair of providers associated with overlapping treatment of a single patient. | Connection between nodes relative to time at which each node was present in the network. | Provider–provider connections that represent instances in which care was likely coordinated. |
| Atemporal Edge | Pairwise combination of providers associated with treatment of a single patient. | Connection between nodes, irrespective of time when node was present in the network. | Provider–provider connections that represent potential connections based on caring for a shared patient. |
| Stage I | Stage II | Stage III | Stage I–III | |
|---|---|---|---|---|
| Number of Patients | 2116 | 1452 | 514 | 4082 |
| Number of Providers | 1090 | 948 | 503 | 2190 |
| Unique Temporal Edges | 35,402 | 23,265 | 9789 | 54,695 |
| Unique Atemporal Edges | 167,318 | 107,018 | 41,686 | 249,075 |
| Node Size | ||||
| Mean (range) | 16.3 (1, 1084) | 10.7 (1, 675) | 5.6 (1, 164) | 31.4 (1, 2351) |
| Median | 4 | 3 | 2 | 179 |
| Temporal Edge Size | ||||
| Mean (range) | 3.4 (1, 371) | 3.5 (1, 400) | 3.1 (1, 164) | 4.2 (1, 838) |
| Median | 2 | 2 | 2 | 2 |
| Atemporal Edge Size | ||||
| Mean (range) | 1.8 (1, 467) | 1.7 (1, 306) | 1.5 (1, 157) | 2.2 (1, 908) |
| Median | 1 | 1 | 1 | 1 |
| Providers per Patient | ||||
| Mean (range) | 15.3 (1, 414) | 15.1 (1, 64) | 15.7 (1, 44) | 15.3 (1, 74) |
| Median | 12 | 12 | 13 | 12 |
| Appointments per Patient | ||||
| Mean (range) | 70.5 (1, 414) | 72.8 (1, 498) | 80.4 (1, 363) | 72.7 (1, 498) |
| Median | 50 | 56 | 65 | 54 |
| Number of Diagnoses | Number of Patients | Number of Providers | Number of Temporal Edges | Number of Atemporal Edges | Sum of Temporal Edge Weights | Sum of Atemporal Edge Weights | Temporal Network Density | Atemporal Network Density | |
|---|---|---|---|---|---|---|---|---|---|
| 2002 | 155 | 1424 | 458 | 596 | 2033 | 1814 | 2831 | 1.56 | 1.57 |
| 2003 | 156 | 1678 | 533 | 1309 | 3355 | 4425 | 4843 | 1.52 | 1.62 |
| 2004 | 174 | 1840 | 569 | 1919 | 4273 | 6575 | 6239 | 1.7 | 1.76 |
| 2005 | 173 | 2023 | 631 | 2748 | 5095 | 9714 | 7550 | 1.73 | 1.7 |
| 2006 | 202 | 2249 | 682 | 3378 | 5340 | 12,082 | 8078 | 1.69 | 1.53 |
| 2007 | 205 | 2461 | 753 | 4372 | 6625 | 15,408 | 9993 | 1.63 | 1.6 |
| 2008 | 256 | 2625 | 786 | 5455 | 7962 | 18,805 | 12,177 | 1.66 | 1.63 |
| 2009 | 276 | 2799 | 790 | 7055 | 9453 | 24,153 | 14,800 | 1.85 | 1.79 |
| 2010 | 271 | 2989 | 863 | 9658 | 11,841 | 33,066 | 19,188 | 1.88 | 1.94 |
| 2011 | 303 | 3127 | 945 | 11,581 | 13,614 | 40,552 | 22,146 | 1.82 | 1.84 |
| 2012 | 331 | 3366 | 995 | 13,016 | 14,601 | 44,778 | 23,394 | 1.8 | 1.69 |
| 2013 | 406 | 3593 | 1038 | 14,663 | 16,387 | 51,366 | 27,015 | 1.84 | 1.74 |
| 2014 | 356 | 3711 | 1034 | 14,729 | 16,212 | 52,382 | 26,794 | 1.83 | 1.83 |
| 2015 | 418 | 3775 | 1074 | 15,366 | 17,493 | 52,505 | 28,240 | 1.66 | 1.76 |
| 2016 | 400 | 3826 | 1076 | 14,142 | 17,025 | 49,263 | 29,263 | 1.5 | 1.74 |
| Full-Time | Part-Time | |||||
|---|---|---|---|---|---|---|
| Medical Oncologist 1 | Medical Oncologist 2 | Medical Oncologist 3 | Medical Oncologist 4 | Medical Oncologist 5 | Medical Oncologist 6 | |
| Overall Degree Centrality | ||||||
| Temporal | 1159 | 1034 | 950 | 423 | 517 | 342 |
| Atemporal | 1963 | 1979 | 1864 | 994 | 1493 | 836 |
| Overall Care Density | ||||||
| Temporal | 12.3 | 12.3 | 14.2 | 6.9 | 7.8 | 5.4 |
| Atemporal | 7.2 | 6.4 | 7.2 | 2.9 | 2.7 | 2.2 |
| Yearly Temporal Care Density | ||||||
| Year 1 | 4.1 | 4.86 | 6.4 | 3.77 | 2 | 3.74 |
| Year 2 | 6.64 | 6.69 | 7.07 | 4.49 | 4 | 4.11 |
| Year 3 | 5.95 | 7.72 | 6.96 | 4.47 | 5.04 | 4.34 |
| Year 4 | 6.94 | 7.69 | 7.6 | 5.11 | 5.16 | 4.2 |
| Year 5 | 6.76 | 7.51 | 8.79 | 4.97 | 5.84 | 4.22 |
| Year 6 | 6.69 | 7.46 | 9.34 | 5.03 | 6.19 | |
| Year 7 | 6.76 | 6.86 | 9.33 | 5.49 | ||
| Year 8 | 7.38 | 7.23 | 9.36 | |||
| Year 9 | 7.64 | 7.24 | ||||
| Year 10 | 7.17 | 7.28 | ||||
| Year 11 | 7.23 | 6.74 | ||||
| Year 12 | 7.25 | |||||
| Year 13 | 6.68 | |||||
| Year 14 | 6.66 | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Steitz, B.D.; Levy, M.A. Temporal and Atemporal Provider Network Analysis in a Breast Cancer Cohort from an Academic Medical Center (USA). Informatics 2018, 5, 34. https://doi.org/10.3390/informatics5030034
Steitz BD, Levy MA. Temporal and Atemporal Provider Network Analysis in a Breast Cancer Cohort from an Academic Medical Center (USA). Informatics. 2018; 5(3):34. https://doi.org/10.3390/informatics5030034
Chicago/Turabian StyleSteitz, Bryan D., and Mia A. Levy. 2018. "Temporal and Atemporal Provider Network Analysis in a Breast Cancer Cohort from an Academic Medical Center (USA)" Informatics 5, no. 3: 34. https://doi.org/10.3390/informatics5030034
APA StyleSteitz, B. D., & Levy, M. A. (2018). Temporal and Atemporal Provider Network Analysis in a Breast Cancer Cohort from an Academic Medical Center (USA). Informatics, 5(3), 34. https://doi.org/10.3390/informatics5030034




