Knowledge about Hand Hygiene and Related Infectious Disease Awareness among Primary School Children in Germany †
Abstract
1. Introduction
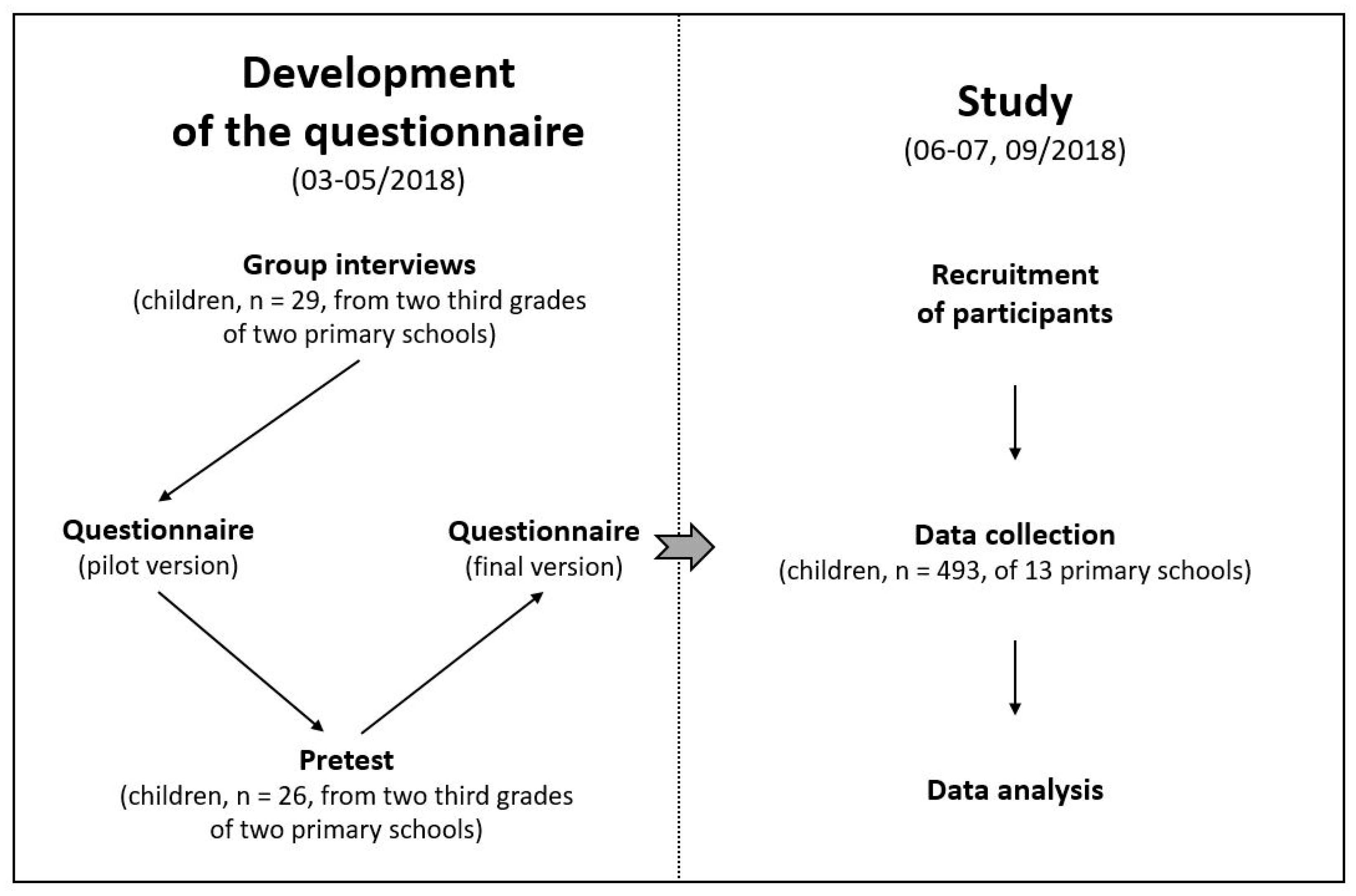
2. Materials and Methods
2.1. Participants
2.2. Development of the Questionnaire
2.3. Data Collection and Analyses
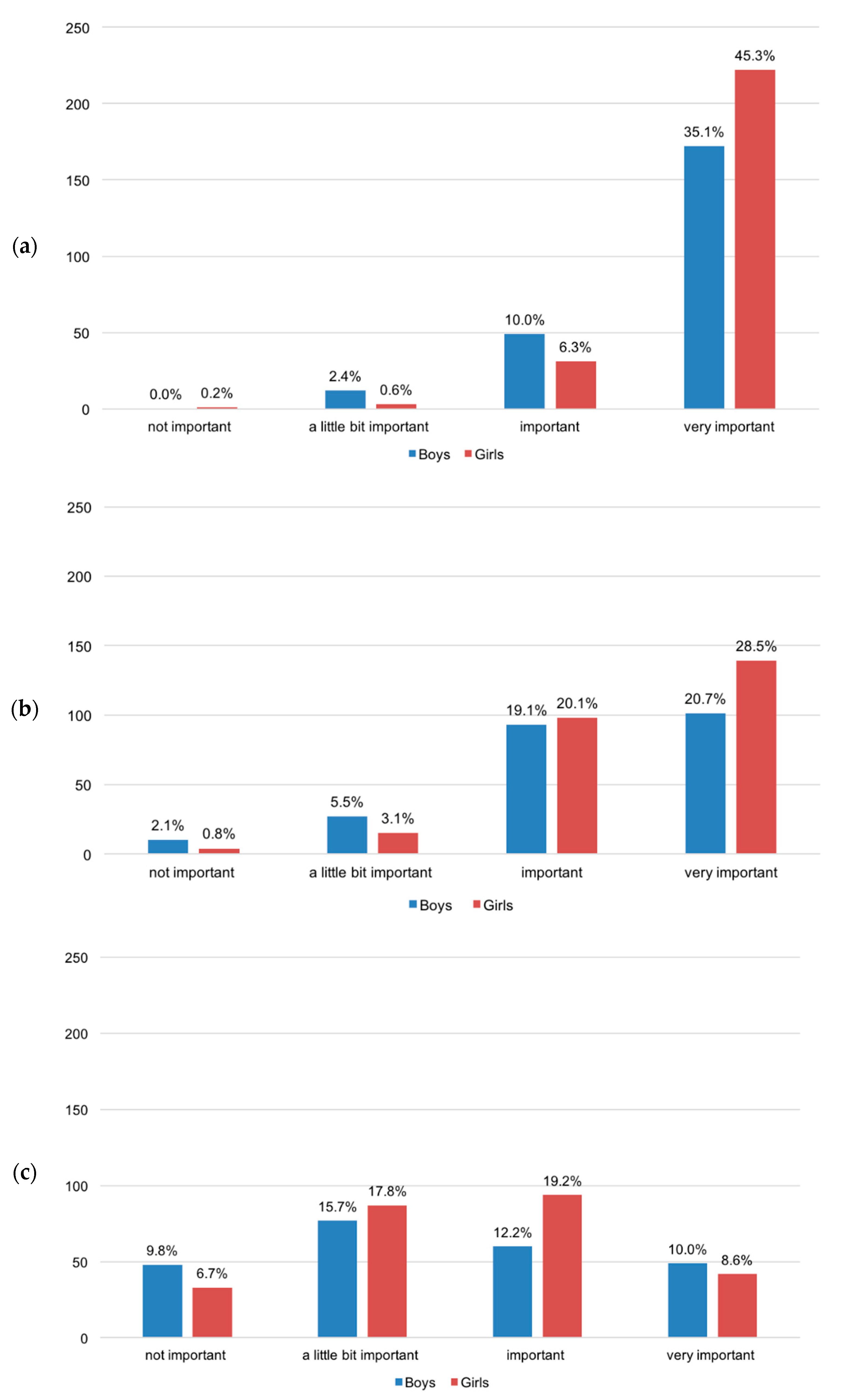
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Municipalities 1 | Number of Inhabitants 2 | City and Municipality Type 3 | Number of Primary Schools in the Municipalities 4 | Number of Participating Primary Schools from the Municipalities |
|---|---|---|---|---|
| Altenberge | 10,327 | Larger small town | 2 | 2 |
| Emsdetten | 36,029 | Little medium-sized town | 6 | 1 |
| Greven | 37,753 | Little medium-sized town | 5 | 2 |
| Hörstel | 20,344 | Little medium-sized town | 5 | 2 |
| Ladbergen | 6688 | Little small town | 1 | 1 |
| Laer | 6744 | Little small town | 1 | 1 |
| Lengerich | 22,660 | Little medium-sized town | 4 | 3 |
| Lienen | 8604 | Little small town | 3 | 1 |
| Lotte | 14,095 | Larger small town | 3 | 1 |
| Westerkappeln | 11,241 | Larger small town | 2 | 1 |
| Primary School 1 | Total Number of School Children 2 | Number of Classes | Number of Grade 3 Classes | Number of Sanitary Facilities for School Children (Girls/Boys) 3 | Total Number of Toilets Seats for School Children Girls/Boys/Total (%) 4 | Total Number of Urinals for School Children (Only Boys) |
|---|---|---|---|---|---|---|
| 01 | 268 | 12 | 3 | 4 (2/2) | 7/4/11 (4.1) | 10 |
| 02 | 261 | 11 | 2 | 8 (4/4) | 11/7/18 (6.9) | 14 |
| 03 | 157 | 8 | 2 | 6 (3/3) | 8/6/14 (8.9) | 12 |
| 04 | 340 | 14 | 3 | 5 5 | 13/8/21 (6.2) | 7 |
| 05 | 267 | 11 | 3 | 2 (1/1) | 7/5/12 (4.5) | 10 |
| 06 | 104 | 4 | 1 | 2 (1/1) | 3/4/7 (6.7) | 2 |
| 07 | 103 | 4 | 1 | 2 (1/1) | 4/2/6 (5.8) | 4 |
| 08 | 161 | 8 | 2 | 2 (1/1) | 4/5/9 (5.6) | 5 |
| 09 | 94 | 4 | 1 | 2 (1/1) | 4/3/7 (7.4) | 4 |
| 10 | 202 | 8 | 2 | 2 (1/1) | 4/3/7 (3.5) | 3 |
| 11 | 240 | 11 | 3 | 4 (2/2) | 10/7/17 (7.1) | 11 |
| 12 | 192 | 8 | 2 | 2 (1/1) | 6/6/12 (6.2) | 2 |
| 13 | 226 | 12 | 3 | 2 (1/1) | 14/7/21 (9.3) | 7 |
| Age in Years | Number (Percentage) 1 of School Children | ||
|---|---|---|---|
| Girls | Boys | Total | |
| 8 | 58 (11.8%) | 45 (9.1%) | 103 (20.9%) |
| 9 | 178 (36.2%) | 168 (34.1%) | 346 (70.3%) |
| 10 | 19 (3.9%) | 18 (3.7%) | 37 (7.5%) |
| 11 | 2 (0.4%) | 4 (0.8%) | 6 (1.2%) |
| Total | 257 (52.2%) | 235 (47.8%) | 492 (100.0%) |
References
- World Health Organization. WHO Guidelines on Hand Hygiene in Health Care: A Summary, WHO/IER/PSP/2009.07 ed.; WHO Press: Geneva, Switzerland, 2009. [Google Scholar]
- Von Lengerke, T.; Lutze, B.; Krauth, C.; Lange, K.; Stahmeyer, J.T.; Chaberny, I.F. Promoting Hand Hygiene Compliance: PSYGIENE—A Cluster-Randomized Controlled Trial of Tailored Interventions. Dtsch. Arztebl. Int. 2017, 114, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Wetzker, W.; Bunte-Schönberger, K.; Walter, J.; Pilarski, G.; Gastmeier, P.; Reichardt, C. Compliance with hand hygiene: Reference data from the national hand hygiene campaign in Germany. J. Hosp. Infect. 2016, 92, 328–331. [Google Scholar] [CrossRef]
- Kingston, L.; O′Connell, N.H.; Dunne, C.P. Hand hygiene-related clinical trials reported since 2010: A systematic review. J. Hosp. Infect. 2016, 92, 309–320. [Google Scholar] [CrossRef]
- Harrison, A.G.; Lin, T.; Wang, P. Mechanisms of SARS-CoV-2 Transmission and Pathogenesis. Trends Immunol. 2020, 41, 1100–1115. [Google Scholar] [CrossRef]
- Köck, R.; Becker, K.; Cookson, B.; van Gemert-Pijnen, J.E.; Harbarth, S.; Kluytmans, J.; Mielke, M.; Peters, G.; Skov, R.L.; Struelens, M.J.; et al. Methicillin-resistant Staphylococcus aureus (MRSA): Burden of disease and control challenges in Europe. Eurosurveillance 2010, 15, 19688. [Google Scholar] [CrossRef]
- Martin, M.J.; Corey, B.W.; Sannio, F.; Hall, L.R.; MacDonald, U.; Jones, B.T.; Mills, E.G.; Harless, C.; Stam, J.; Maybank, R.; et al. Anatomy of an extensively drug-resistant Klebsiella pneumoniae outbreak in Tuscany, Italy. Proc. Natl. Acad. Sci. USA 2021, 118, 118. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, D.M.P.; Forde, B.M.; Kidd, T.J.; Harris, P.N.A.; Schembri, M.A.; Beatson, S.A.; Paterson, D.L.; Walker, M.J. Antimicrobial Resistance in ESKAPE Pathogens. Clin. Microbiol. Rev. 2020, 33, e00181-19. [Google Scholar] [CrossRef]
- Jurke, A.; Daniels-Haardt, I.; Silvis, W.; Berends, M.S.; Glasner, C.; Becker, K.; Köck, R.; Friedrich, A.W. Changing epidemiology of meticillin-resistant Staphylococcus aureus in 42 hospitals in the Dutch-German border region, 2012 to 2016: Results of the search-and-follow-policy. Eurosurveillance 2019, 24, 1800244. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.; Thomson, N.; Weill, F.X.; Holt, K.E. Genomic insights into the emergence and spread of antimicrobial-resistant bacterial pathogens. Science 2018, 360, 733–738. [Google Scholar] [CrossRef]
- Von Eiff, C.; Jansen, B.; Kohnen, W.; Becker, K. Infections associated with medical devices: Pathogenesis, management and prophylaxis. Drugs 2005, 65, 179–214. [Google Scholar] [CrossRef]
- Becker, K.; Both, A.; Weisselberg, S.; Heilmann, C.; Rohde, H. Emergence of coagulase-negative staphylococci. Expert Rev. Anti. Infect. Ther. 2020, 18, 349–366. [Google Scholar] [CrossRef] [PubMed]
- Chesshyre, E.; Goff, Z.; Bowen, A.; Carapetis, J. The prevention, diagnosis and management of central venous line infections in children. J. Infect. 2015, 71 (Suppl. 1), S59–S75. [Google Scholar] [CrossRef] [PubMed]
- Marcos, M.; Soriano, A.; Iñurrieta, A.; Martínez, J.A.; Romero, A.; Cobos, N.; Hernández, C.; Almela, M.; Marco, F.; Mensa, J. Changing epidemiology of central venous catheter-related bloodstream infections: Increasing prevalence of Gram-negative pathogens. J. Antimicrob. Chemother. 2011, 66, 2119–2125. [Google Scholar] [CrossRef] [PubMed]
- Dumford, D., 3rd; Skalweit, M.J. Antibiotic-Resistant Infections and Treatment Challenges in the Immunocompromised Host: An Update. Infect. Dis. Clin. N. Am. 2020, 34, 821–847. [Google Scholar] [CrossRef] [PubMed]
- Rhee, Y.; Heung, M.; Chen, B.; Chenoweth, C.E. Central line-associated bloodstream infections in non-ICU inpatient wards: A 2-year analysis. Infect. Control Hosp. Epidemiol. 2015, 36, 424–430. [Google Scholar] [CrossRef]
- Bloomfield, S.F.; Aiello, A.E.; Cookson, B.; O’Boyle, C.; Larson, E.L. The effectiveness of hand hygiene procedures in reducing the risks of infections in home and community settings including handwashing and alcohol-based hand sanitizers. Am. J. Infect. Control 2007, 35, S27–S64. [Google Scholar] [CrossRef]
- Amin, R.; Hatakeyama, Y.; Kitazawa, T.; Matsumoto, K.; Fujita, S.; Seto, K.; Hasegawa, T. Capturing the trends in hospital standardized mortality ratios for pneumonia: A retrospective observational study in Japan (2010 to 2018). Environ. Health Prev. Med. 2020, 25, 2. [Google Scholar] [CrossRef]
- Winocour, P.H. Diabetes and chronic kidney disease: An increasingly common multi-morbid disease in need of a paradigm shift in care. Diabet. Med. 2018, 35, 300–305. [Google Scholar] [CrossRef]
- Schaumburg, F.; Sertic, S.M.; Correa-Martinez, C.; Mellmann, A.; Köck, R.; Becker, K. Acquisition and colonization dynamics of antimicrobial-resistant bacteria during international travel: A prospective cohort study. Clin. Microbiol. Infect. 2019, 25, 1287.e1281–1287.e1287. [Google Scholar] [CrossRef]
- Schwartz, K.L.; Morris, S.K. Travel and the Spread of Drug-Resistant Bacteria. Curr. Infect. Dis. Rep. 2018, 20, 29. [Google Scholar] [CrossRef]
- Hassing, R.J.; Alsma, J.; Arcilla, M.S.; van Genderen, P.J.; Stricker, B.H.; Verbon, A. International travel and acquisition of multidrug-resistant Enterobacteriaceae: A systematic review. Eurosurveillance 2015, 20, 30074. [Google Scholar] [CrossRef] [PubMed]
- Van Alen, S.; Ballhausen, B.; Peters, G.; Friedrich, A.W.; Mellmann, A.; Köck, R.; Becker, K. In the centre of an epidemic: Fifteen years of LA-MRSA CC398 at the University Hospital Münster. Vet. Microbiol. 2017, 200, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Cooke, F.J.; Brown, N.M. Community-associated methicillin-resistant Staphylococcus aureus infections. Br. Med. Bull. 2010, 94, 215–227. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heiden, S.E.; Hübner, N.O.; Bohnert, J.A.; Heidecke, C.D.; Kramer, A.; Balau, V.; Gierer, W.; Schaefer, S.; Eckmanns, T.; Gatermann, S.; et al. A Klebsiella pneumoniae ST307 outbreak clone from Germany demonstrates features of extensive drug resistance, hypermucoviscosity, and enhanced iron acquisition. Genome Med. 2020, 12, 113. [Google Scholar] [CrossRef]
- Arato, V.; Raso, M.M.; Gasperini, G.; Berlanda Scorza, F.; Micoli, F. Prophylaxis and Treatment against Klebsiella pneumoniae: Current Insights on This Emerging Anti-Microbial Resistant Global Threat. Int. J. Mol. Sci. 2021, 22, 4042. [Google Scholar] [CrossRef]
- Köck, R.; Becker, K.; Idelevich, E.A.; Jurke, A.; Glasner, C.; Hendrix, R.; Friedrich, A.W. Prevention and control of multidrug-resistant bacteria in The Netherlands and Germany—The impact of healthcare structures. Int. J. Environ. Res. Public Health 2020, 17, 2337. [Google Scholar] [CrossRef]
- Lee, R.L.; Loke, A.Y.; Wu, C.S.; Ho, A.P. The lifestyle behaviours and psychosocial well-being of primary school students in Hong Kong. J. Clin. Nurs. 2010, 19, 1462–1472. [Google Scholar] [CrossRef]
- Khan, K.M.; Chakraborty, R.; Brown, S.; Sultana, R.; Colon, A.; Toor, D.; Upreti, P.; Sen, B. Association between Handwashing Behavior and Infectious Diseases among Low-Income Community Children in Urban New Delhi, India: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2021, 18, 12535. [Google Scholar] [CrossRef]
- United Nations Children’s Fund (UNICEF); World Health Organization (WHO). Drinking Water, Sanitation and Hygiene in Schools: Global Baseline Report 2018; United Nations Children’s Fund (UNICEF) and World Health Organization: New York, NY, USA, 2018.
- Jatrana, S.; Hasan, M.M.; Mamun, A.A.; Fatima, Y. Global Variation in Hand Hygiene Practices Among Adolescents: The Role of Family and School-Level Factors. Int. J. Environ. Res. Public Health 2021, 18, 4984. [Google Scholar] [CrossRef]
- Mbakaya, B.C.; Kalembo, F.W.; Zgambo, M. Use, adoption, and effectiveness of tippy-tap handwashing station in promoting hand hygiene practices in resource-limited settings: A systematic review. BMC Public Health 2020, 20, 1005. [Google Scholar] [CrossRef]
- Gizaw, Z.; Addisu, A. Evidence of Households’ Water, Sanitation, and Hygiene (WASH) Performance Improvement Following a WASH Education Program in Rural Dembiya, Northwest Ethiopia. Environ. Health Insights 2020, 14, 1178630220903100. [Google Scholar] [CrossRef] [PubMed]
- Kamtsiuris, P.; Atzpodien, K.; Ellert, U.; Schlack, R.; Schlaud, M. Prävalenz von somatischen Erkrankungen bei Kindern und Jugendlichen in Deutschland. Ergebnisse des Kinder- und Jugendgesundheitssurveys (KiGGS) [Prevalence of somatic diseases in German children and adolescents. Results of the German Health Interview and Examination Survey for Children and Adolescents (KiGGS)]. Bundesgesundheitsblatt Gesundh. Gesundh. 2007, 50, 686–700. [Google Scholar] [CrossRef]
- Reeves, L.M.; Priest, P.C.; Poore, M.R. School toilets: Facilitating hand hygiene? A review of primary school hygiene facilities in a developed country. J. Public Health 2012, 34, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Neuzil, K.M.; Hohlbein, C.; Zhu, Y. Illness among schoolchildren during influenza season: Effect on school absenteeism, parental absenteeism from work, and secondary illness in families. Arch. Pediatr. Adolesc. Med. 2002, 156, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Amadi, C. Impact of water, sanitation, and hygiene interventions on improving health outcomes among school children. J. Environ. Public Health 2013, 2013, 984626. [Google Scholar] [CrossRef]
- Azor-Martinez, E.; Cobos-Carrascosa, E.; Seijas-Vazquez, M.L.; Fernández-Sánchez, C.; Strizzi, J.M.; Torres-Alegre, P.; Santisteban-Martínez, J.; Gimenez-Sanchez, F. Hand Hygiene Program Decreases School Absenteeism Due to Upper Respiratory Infections. J. Sch. Health 2016, 86, 873–881. [Google Scholar] [CrossRef]
- Chittleborough, C.R.; Nicholson, A.L.; Young, E.; Bell, S.; Campbell, R. Implementation of an educational intervention to improve hand washing in primary schools: Process evaluation within a randomised controlled trial. BMC Public Health 2013, 13, 757. [Google Scholar] [CrossRef]
- Greene, L.E.; Freeman, M.C.; Akoko, D.; Saboori, S.; Moe, C.; Rheingans, R. Impact of a school-based hygiene promotion and sanitation intervention on pupil hand contamination in Western Kenya: A cluster randomized trial. Am. J. Trop. Med. Hyg. 2012, 87, 385–393. [Google Scholar] [CrossRef]
- Lang, M.C. Implementation of an evidence-based hand hygiene program in elementary schools in Ghana, as part of a City-to-City partnership between Ottawa public health and KEEA health directorate. Fam. Community Health 2012, 35, 203–211. [Google Scholar] [CrossRef]
- Nandrup-Bus, I. Mandatory handwashing in elementary schools reduces absenteeism due to infectious illness among pupils: A pilot intervention study. Am. J. Infect. Control 2009, 37, 820–826. [Google Scholar] [CrossRef]
- Schmidt, W.P.; Wloch, C.; Biran, A.; Curtis, V.; Mangtani, P. Formative research on the feasibility of hygiene interventions for influenza control in UK primary schools. BMC Public Health 2009, 9, 390. [Google Scholar] [CrossRef]
- Celik, L.A.; Pancoe, D.L. Healthy school environment: Effectiveness of hand washing instruction in an elementary school setting. NASN Sch. Nurse 2012, 27, 194–196. [Google Scholar] [CrossRef] [PubMed]
- Lecky, D.M.; McNulty, C.A.; Adriaenssens, N.; Koprivová Herotová, T.; Holt, J.; Touboul, P.; Merakou, K.; Koncan, R.; Olczak-Pienkowska, A.; Avô, A.B.; et al. What are school children in Europe being taught about hygiene and antibiotic use? J. Antimicrob. Chemother. 2011, 66 (Suppl. 5), v13–v21. [Google Scholar] [CrossRef] [PubMed]
- Lundblad, B.; Hellström, A.L. Perceptions of school toilets as a cause for irregular toilet habits among schoolchildren aged 6 to 16 years. J. Sch. Health 2005, 75, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Laffolie, J.; Ibrahimi, G.; Zimmer, K.P. Poor Perception of School Toilets and Increase of Functional Constipation. Klin. Padiatr. 2021, 233, 5–9. [Google Scholar] [CrossRef]
- Lundblad, B.; Hellström, A.L.; Berg, M. Children’s experiences of attitudes and rules for going to the toilet in school. Scand. J. Caring Sci. 2010, 24, 219–223. [Google Scholar] [CrossRef]
- Chittleborough, C.R.; Nicholson, A.L.; Basker, E.; Bell, S.; Campbell, R. Factors influencing hand washing behaviour in primary schools: Process evaluation within a randomized controlled trial. Health Educ. Res. 2012, 27, 1055–1068. [Google Scholar] [CrossRef]
- Senior, E. We love our school toilets: Involving primary school students in improving their school toilets. Glob. Health Promot. 2014, 21, 23–28. [Google Scholar] [CrossRef]
- Vernon, S.; Lundblad, B.; Hellstrom, A.L. Children’s experiences of school toilets present a risk to their physical and psychological health. Child. Care Health Dev. 2003, 29, 47–53. [Google Scholar] [CrossRef]
- Heudorf, U.; Voigt, K.; Eikmann, T.; Exner, M. Hygiene in Schulen—Auch eine wichtige Aufgabe des offentlichen Gesundheitsdienstes [Hygiene in schools—An important issue for the public health services]. Gesundheitswesen 2011, 73, 730–736. [Google Scholar] [CrossRef]
- Loosveldt, G. Face-To-Face Interviews. In International Handbook of Survey Methodology; de Leeuw, E.D., Hox, J.J., Dillman, D.A., Eds.; Psychology Press (Taylor & Francis): London, UK, 2008; pp. 201–220. [Google Scholar]
| School Grade | Number (Percentage) 1 of School Children 2 | ||
|---|---|---|---|
| Girls (%) | Boys (%) | Total (%) | |
| 1 (Very Good) | 19 (3.9) | 21 (4.3) | 40 (8.1) |
| 2 (Good) | 55 (11.2) | 32 (6.5) | 87 (17.7) |
| 3 (Satisfactory) | 92 (18.7) | 57 (11.6) | 149 (30.3) |
| 4 (Sufficient) | 57 (11.6) | 64 (13.0) | 121 (24.6) |
| 5 (Poor) | 22 (4.5) | 31 (6.3) | 53 (10.8) |
| 6 (Deficient) | 11 (2.2) | 31 (6.3) | 42 (8.5) |
| Total | 256 (52.0) | 236 (48.0) | 492 (100.0) |
| Frequency to Visit the Toilet at School per Day | Number (Percentage) 1 of School Children | ||
|---|---|---|---|
| Girls (%) | Boys (%) | Total (%) | |
| Mostly not at all | 112 (22.7) | 106 (21.5) | 218 (44.2) |
| 1 time | 44 (8.9) | 44 (8.9) | 88 (17.8) |
| 2 times | 67 (13.6) | 55 (11.2) | 122 (24.7) |
| 3 times | 22 (4.5) | 14 (2.8) | 36 (7.3) |
| More than 3 times | 9 (1.8) | 14 (2.8) | 23 (4.7) |
| Total | 254 (51.5) | 233 (47.3) | 487 (98.8) 2 |
| Category 1 | Quantity of Answers 2 | Exemplary Answer |
|---|---|---|
| Uncleanliness of the sanitary facilities | 243 | “… the toilets are always dirty.” (boy, 9 years old) “…and there is not much cleaning.” (girl, 9 years old) “…there are mostly spiders.” (girl, 9 years old) “…the toilet is a bit disgusting.” (girl, 8 years old) |
| (Mis-)behavior of classmates | 141 | “…sometimes there is urine on the toilet seat.” (girl, 9 years old) “…no one washes their hands and then touches the door handle!” (girl, 8 years old) “the toilet seat is sometimes dirty and some forget to flush.” (girl, 9 years old) “…they always look from underneath, when you are sitting on the toilet.” (boy, 10 years old) “…everyone from school goes to the toilet.” (girl, 10 years old) “…some children put themselves on the toilet with street shoes and then the toilet is dirty.” (girl, 9 years old) |
| General condition of the sanitary facilities (e.g., aeration, temperature, operability of the equipment and lack of hygiene articles) | 82 | “…it smells in the toilet.” (girl, 10 years old) “…the toilet room is often wet.” (boy, 9 years old) “…it is always cold.” (boy, 9 years old) “…and there is usually no toilet paper.” (girl, 8 years old) |
| Conflict of interests | 18 | “…I won’t be able to learn.” (boy, 9 years old) “…If it takes too long time (to go to the toilet), I miss the lesson or don’t get to know some nice things.” (girl, 9 years old) |
| Lack of need | 14 | “… I do not need to (use the toilet).” (girl, 9 years old) |
| Discomfort using the toilet | 14 | “…it is scary down there.” (boy, 9 years old) “…it’s uncomfortable for me there.” (girl, 9 years old) “…I feel more at ease at home.” (girl, 9 years old) |
| Awareness of microbial pathogens | 8 | “…there are a lot of bacteria.” (girl, 9 years old) “…because there can also always be bacteria.” (girl, 8 years old) |
| Other | 7 | “…because otherwise we are always get trouble.” (boy, 8 years old)“…it is impolite.” (boy, 9 years old) “…that is no fun.” (boy, 9 years old) |
| Statements (Number of Valid Answers) “I Wash My Hands at School…” | Number (Percentage) 1 of Answers | |||
|---|---|---|---|---|
| Never (%) | Seldom (%) | Often (%) | Always (%) | |
| 1. “…when the hands are dirty.” (n = 492) | 11 (2.2) | 51 (10.4) | 157 (31.9) | 273 (55.5) |
| 2. “…before eating, e.g., bread during the break or lunch.” (n = 489) | 120 (24.5) | 134 (27.4) | 119 (24.3) | 116 (23.7) |
| 3. “…before I go to the toilet.” (n = 485) | 366 (75.5) | 75 (15.5) | 24 (4.9) | 20 (4.1) |
| 4. “…after I went to the toilet.” (n = 485) | 6 (1.2) | 21 (4.3) | 86 (17.7) | 372 (76.7) |
| 5. “…after physical education.” (n = 490) | 294 (60.0) | 139 (28.4) | 40 (8.2) | 17 (3.5) |
| 6. “…after the break.” (n = 485) | 173 (35.7) | 182 (37.5) | 92 (19.0) | 38 (7.8) |
| 7. “…when I worked with paint, e.g., watercolor while art lesson.” (n = 491) | 7 (1.4) | 32 (6.5) | 96 (19.6) | 356 (72.5) |
| 8. “…after sniffing or blowing the nose.” (n = 489) | 121 (24.7) | 140 (28.6) | 111 (22.7) | 117 (23.9) |
| 9. “…after sneezing.” (n = 488) | 150 (30.7) | 145 (29.7) | 114 (23.4) | 79 (16.2) |
| Statements (Number of Valid Answers) “I Wash My Hands at Home…” | Number (Percentage) 1 of Answers | |||
|---|---|---|---|---|
| Never (%) | Seldom (%) | Often (%) | Always (%) | |
| 1. “…before touching food, e.g., when I help to cook at home.” (n = 492) | 17 (3.5) | 31 (6.3) | 70 (14.2) | 374 (76.0) |
| 2. “…when the hands are dirty.” (n = 491) | 6 (1.2) | 23 (4.7) | 100 (20.4) | 362 (73.7) |
| 3. “…before lunch.” (n = 490) | 31 (6.3) | 80 (16.3) | 119 (24.3) | 260 (53.1) |
| 4. “…before I go to the toilet.” (n = 488) | 342 (70.1) | 98 (20.1) | 28 (5.7) | 20 (4.1) |
| 5. “…after I went to the toilet.” (n = 490) | 5 (1.0) | 26 (5.3) | 88 (18.0) | 371 (75.7) |
| 6. “…after I played outside.” (n = 489) | 54 (11.0) | 124 (25.4) | 165 (33.7) | 146 (29.9) |
| 7. “…after sniffing or blowing the nose.” (n = 486) | 139 (28.6) | 153 (31.5) | 90 (18.5) | 104 (21.4) |
| 8. “…after sneezing.” (n = 488) | 171 (35.0) | 131 (26.8) | 102 (20.9) | 84 (17.2) |
| 9. “…after I touched an animal, e.g., a dog or a cat.” (n = 486) | 90 (18.5) | 88 (18.1) | 112 (23.0) | 196 (40.3) |
| 10. “…when I come home.” (n = 490) | 94 (19.2) | 114 (23.3) | 115 (23.5) | 167 (34.1) |
| 11. “…before going to bed.” (n = 491) | 130 (26.5) | 111 (22.6) | 116 (23.6) | 134 (27.3) |
| 12. “…after getting up.” (n = 488) | 137 (28.1) | 117 (24.0) | 98 (20.1) | 136 (27.9) |
| 13. “…after contact with sick people who are ill with cough or sniffles for example.” (n = 489) | 18 (3.7) | 34 (7.0) | 95 (19.4) | 342 (69.9) |
| 14. “…when I was at the doctor.” (n = 493) | 87 (17.6) | 97 (19.7) | 119 (24.1) | 190 (38.5) |
| 15. “…when I visited someone in the hospital.” (n = 488) | 38 (7.8) | 54 (11.1) | 97 (19.9) | 299 (61.3) |
| Statements (Number of Valid Answers) | Options | Number (Percentage) of Answers | Significance 1 p-Value (Chi-Squared Test 2) | ||
|---|---|---|---|---|---|
| Girls (%) | Boys (%) | Total (%) | |||
| 1. “I can be vaccinated against specific diseases.” (n = 493) | False | 11 (4.3) | 14 (5.9) | 25 (5.1) | p = 0.404 (0.698) |
| Right | 246 (95.7) | 222 (94.1) | 468 (94.9) | ||
| 2. “I touch my face with unwashed hands after going to the toilet.” (n = 493) | False | 253 (98.4) | 227 (96.2) | 480 (97.4) | p= 0.118 (2.441) |
| Right | 4 (1.6) | 9 (3.8) | 13 (2.6) | ||
| 3. “I cover an open wound on my body with a plaster or bandage.” (n = 490) | False | 16 (6.2) | 24 (10.3) | 40 (8.2) | p= 0.100 (2.707) |
| Right | 241 (93.8) | 209 (89.7) | 450 (91.8) | ||
| 4. “I keep distance from ill people who are suffer e.g., from cough or sniffles.” (n = 491) | False | 25 (9.8) | 27 (11.4) | 52 (10.6) | p= 0.556 (0.347) |
| Right | 230 (90.2) | 209 (88.6) | 439 (89.4) | ||
| 5. “I wash my hands with soap.” (n = 492) | False | 0 (0.0) | 3 (1.3) | 3 (0.6) | p= 0.108 2 / |
| Right | 257 (100.0) | 232 (98.7) | 489 (99.4) | ||
| 6. “I play a lot on the computer, game console or watch a lot of television.” (n = 485) | False | 239 (94.1) | 173 (74.9) | 412 (84.9) | p < 0.001 (34.889) |
| Right | 15 (5.9) | 58 (25.1) | 73 (15.1) | ||
| 7. “I drink from my classmates’ water bottle.” (n = 489) | False | 247 (97.2) | 229 (97.4) | 476 (97.3) | p= 0.889 (0.019) |
| Right | 7 (2.8) | 6 (2.6) | 13 (2.7) | ||
| Organ (System) Manifestation | Specified Infections | Number of Answers |
|---|---|---|
| Respiratory tract infections | Influenza Cold Tonsillitis Bronchitis Pneumonia | 77 18 4 3 2 |
| Gastrointestinal tract infections | Gastrointestinal diseases Hepatitis | 84 1 |
| Childhood infections | Scarlet Measles Chickenpox Rubella Mumps | 37 15 15 2 1 |
| Skin infections | Herpes Hand-foot-and-mouth disease Shingles Warts Scabies Eczema | 8 4 4 2 1 1 |
| Central nervous system infections | Meningitis Rabies | 1 1 |
| Eye infections | Conjunctivitis | 4 |
| Urinary tract infections | Cystitis | 1 |
| Other | Ebola Pest | 1 1 |
| Statements (Number of Valid Answers) | Options | Number (Percentage) of Answers | Significance p-Value (Chi-Squared Test 1) | ||
|---|---|---|---|---|---|
| Girls (%) | Boys (%) | Total (%) | |||
| 1. “I will stay at home if possible.” (n = 492) | False | 7 (2.7) | 12 (5.1) | 19 (3.9) | p = 0.176 |
| Right | 249 (97.3) | 224 (94.9) | 473 (96.1) | (1.827) | |
| 2. “I don’t shake hands with other people.” (n = 492) | False | 24 (9.3) | 22 (9.4) | 46 (9.3) | p = 0.993 |
| Right | 233 (90.7) | 213 (90.6) | 446(90.7) | (0.000) | |
| 3. “I cough or sneeze in the crook of my arm and not into my hand.” (n = 492) | False | 9 (3.5) | 10 (4.2) | 19 (3.9) | p = 0.678 |
| Right | 247 (96.5) | 226 (95.8) | 473 (96.1) | (0.172) | |
| 4. “I cough or sneeze into the hand and not in the crook of my arm.” (n = 491) | False | 241 (94.1) | 216 (91.9) | 457 (93.1) | p = 0.332 |
| Right | 15 (5.9) | 19 (8.1) | 34 (6.9) | (0.942) | |
| 5. “I don’t spit on the floor.” (n = 489) | False | 33 (12.9) | 40 (17.1) | 73 (14.9) | p = 0.198 |
| Right | 222 (87.1) | 194 (82.9) | 416 (85.1) | (1.657) | |
| 6. “I meet with my friends.” (n = 491) | False | 235 (92.2) | 207 (87.7) | 442 (90.0) | p = 0.101 |
| Right | 20 (7.8) | 29 (12.3) | 49 (10.0) | (2.696) | |
| 7. “I wash my hands especially thoroughly after using the toilet.” (n = 493) | False | 3 (1.2) | 7 (3.0) | 10 (2.0) | p = 0.206 1 |
| Right | 254 (98.8) | 229 (97.0) | 483 (98.0) | / | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klar, K.; Knaack, D.; Kampmeier, S.; Hein, A.K.; Görlich, D.; Steltenkamp, S.; Weyland, U.; Becker, K. Knowledge about Hand Hygiene and Related Infectious Disease Awareness among Primary School Children in Germany. Children 2022, 9, 190. https://doi.org/10.3390/children9020190
Klar K, Knaack D, Kampmeier S, Hein AK, Görlich D, Steltenkamp S, Weyland U, Becker K. Knowledge about Hand Hygiene and Related Infectious Disease Awareness among Primary School Children in Germany. Children. 2022; 9(2):190. https://doi.org/10.3390/children9020190
Chicago/Turabian StyleKlar, Kristin, Dennis Knaack, Stefanie Kampmeier, Anna Katharina Hein, Dennis Görlich, Siegfried Steltenkamp, Ulrike Weyland, and Karsten Becker. 2022. "Knowledge about Hand Hygiene and Related Infectious Disease Awareness among Primary School Children in Germany" Children 9, no. 2: 190. https://doi.org/10.3390/children9020190
APA StyleKlar, K., Knaack, D., Kampmeier, S., Hein, A. K., Görlich, D., Steltenkamp, S., Weyland, U., & Becker, K. (2022). Knowledge about Hand Hygiene and Related Infectious Disease Awareness among Primary School Children in Germany. Children, 9(2), 190. https://doi.org/10.3390/children9020190








