Palliative Sedation Therapy in Pediatrics: An Algorithm and Clinical Practice Update
Abstract
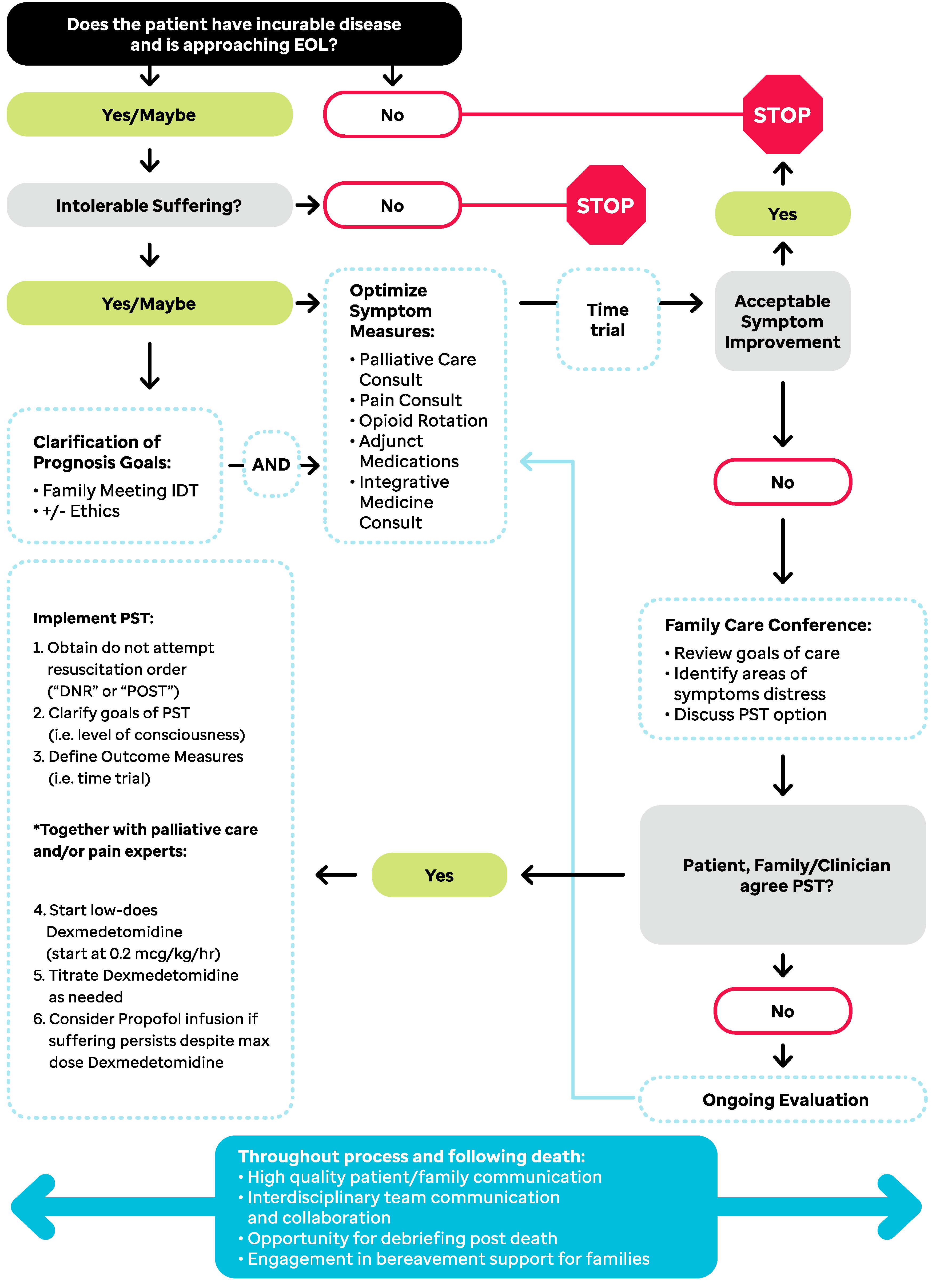
1. Introduction
1.1. Historical PST Practices
1.2. New Directions in the Practice of PST
2. Case-Based Application of PST Algorithm
2.1. Case 1: “When PST Is NOT Indicated”
Take Home Points
2.2. Case 2: “Thinking Outside the Box; An Example of Proportional Sedation Instead of PST to the Point of Unconsciousness”
Take Home Points
2.3. Case 3: “Dexmedetomidine as a Bridge to PST to the Point of Unconsciousness”
Take Home Points
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Behrman, R.E.; Field, M.J. (Eds.) When Children Die: Improving Palliative and End-of-Life Care for Children and Their Families; National Academies Press: Washington, DC, USA, 2003. [Google Scholar]
- Wolfe, J.; Klar, N.; Grier, H.E.; Duncan, J.; Salem-Schatz, S.; Emanuel, E.J.; Weeks, J.C. Understanding of Prognosis Among Parents of Children Who Died of Cancer. JAMA 2000, 284, 2469–2475. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, J.; Orellana, L.; Ullrich, C.; Cook, E.F.; Kang, T.I.; Rosenberg, A.; Geyer, R.; Feudtner, C.; Dussel, V. Symptoms and Distress in Children with Advanced Cancer: Prospective Patient-Reported Outcomes from the PediQUEST Study. J. Clin. Oncol. 2015, 33, 1928–1935. [Google Scholar] [CrossRef] [PubMed]
- Morita, T.; Tsuneto, S.; Shima, Y. Definition of Sedation for Symptom Relief: A Systematic Literature Review and a Proposal of Operational Criteria. J. Pain Symptom Manag. 2002, 24, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Manzini, J.L. Palliative sedation: Ethical perspectives from Latin America in comparison with European recommendations. Curr. Opin. Support. Palliat. Care 2011, 5, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Pousset, G.; Bilsen, J.; Cohen, J.; Mortier, F.; Deliens, L. Continuous Deep Sedation at the End of Life of Children in Flanders, Belgium. J. Pain Symptom Manag. 2011, 41, 449–455. [Google Scholar] [CrossRef]
- Papavasiliou, E.; Payne, S.; Brearley, S.; Brown, J.; Seymour, J. Continuous Sedation (CS) Until Death: Mapping the Literature by Bibliometric Analysis. J. Pain Symptom Manag. 2013, 45, 1073–1082.e10. [Google Scholar] [CrossRef]
- Medicine AAoHaP. Statement on Palliative Sedation. 2014. Available online: http://aahpm.org/positions/palliative-sedation (accessed on 10 October 2022).
- Anghelescu, D.L.; Knapp, E.; Johnson, L.M.; Baker, J.N. The role of the pediatric anesthesiologist in relieving suffering at the end of life: When is palliative sedation appropriate in pediatrics? Paediatr. Anaesth. 2017, 27, 443–444. [Google Scholar] [CrossRef]
- Lo, B.; Rubenfeld, G. Palliative sedation in dying patients: “We turn to it when everything else hasn’t worked”. JAMA 2005, 294, 1810–1816. [Google Scholar] [CrossRef]
- De Graeff, A.; Dean, M. Palliative sedation therapy in the last weeks of life: A literature review and recommendations for standards. J. Palliat. Med. 2007, 10, 67–85. [Google Scholar] [CrossRef]
- Cherny, N.I.; Portenoy, R.K. Sedation in the Management of Refractory Symptoms: Guidelines for Evaluation and Treatment. J. Palliat. Care 1994, 10, 31–38. [Google Scholar] [CrossRef]
- Sterckx, S.; Raus, K.; Mortier, F. Continuous Sedation at the End of Life: Ethical, Clinical and Legal Perspectives; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Janssens, R.; van Delden, J.J.; Widdershoven, G.A. Palliative sedation: Not just normal medical practice. Ethical reflections on the Royal Dutch Medical Association’s guideline on palliative sedation. J. Med. Ethics 2012, 38, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Cona, N. Physician Assisted Suicide Legalization in the United States. 2022. Available online: https://digitalcommons.sacredheart.edu/acadfest/2022/all/111/ (accessed on 10 October 2022).
- Badarau, D.O.; De Clercq, E.; Elger, B.S. Continuous Deep Sedation and Euthanasia in Pediatrics: Does One Really Exclude the Other for Terminally Ill Patients? J. Med. Philos. 2019, 44, 50–70. [Google Scholar] [CrossRef] [PubMed]
- Maltoni, M.; Pittureri, C.; Scarpi, E.; Piccinini, L.; Martini, F.; Turci, P.; Montanari, L.; Nanni, O.; Amadori, D. Palliative sedation therapy does not hasten death: Results from a prospective multicenter study. Ann. Oncol. 2009, 20, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Morita, T.; Chinone, Y.; Ikenaga, M.; Miyoshi, M.; Nakaho, T.; Nishitateno, K.; Sakonji, M.; Shima, Y.; Suenaga, K.; Takigawa, C.; et al. Efficacy and safety of palliative sedation therapy: A multicenter, prospective, observational study conducted on specialized palliative care units in Japan. J. Pain Symptom Manag. 2005, 30, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, J.; Grier, H.E.; Klar, N.; Levin, S.B.; Ellenbogen, J.M.; Salem-Schatz, S.; Emanuel, E.J.; Weeks, J.C. Symptoms and Suffering at the End of Life in Children with Cancer. N. Engl. J. Med. 2000, 342, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Hinds, P.S.; Oakes, L.L.; Hicks, J.; Anghelescu, D.L. End-of-life care for children and adolescents. Semin. Oncol. Nurs. 2005, 21, 53–62. [Google Scholar] [CrossRef]
- Dolinak, D. Opioid toxicity. Acad. Forensic Pathol. 2017, 7, 19–35. [Google Scholar] [CrossRef]
- Potter, J.; Shields, S.; Breen, R. Palliative Sedation, Compassionate Extubation, and the Principle of Double Effect: An Ethical Analysis. Am. J. Hosp. Palliat. Med. 2021, 38, 1536–1540. [Google Scholar] [CrossRef]
- Burns, J.; Jackson, K.; Sheehy, K.A.; Finkel, J.C.; Quezado, Z.M. The Use of Dexmedetomidine in Pediatric Palliative Care: A Preliminary Study. J. Palliat. Med. 2017, 20, 779–783. [Google Scholar] [CrossRef]
- Blais, S.; Cohen-Gogo, S.; Gouache, E.; Guerrini-Rousseau, L.; Brethon, B.; Rahal, I.; Petit, A.; Raimondo, G.; Pellegrino, B.; Orbach, D. End-of-life care in children and adolescents with cancer: Perspectives from a French pediatric oncology care network. Tumori J. 2021, 108, 223–229. [Google Scholar] [CrossRef]
- De Noriega, I.; Andrés, M.R.; Alba, R.M. Análisis Descriptivo de la Sedación Paliativa en una Unidad de Cuidados Paliativos Pediátricos. In Anales de Pediatría; Elsevier: Amsterdam, The Netherlands, 2022; Volume 96, pp. 385–393. [Google Scholar]
- Cuviello, A.A.N.; Morgan, K.J.; Bhatia, S.; Baker, J.N.; Anghelescu, D.L. Palliative Sedation Therapy Practice Comparison—A Survey of Pediatric Palliative Care and Pain Management Specialists. Am. J. Hosp. Palliat. Med. 2022, in press. [Google Scholar]
- Cuviello, A.; Anghelescu, D.; Johnson, L.-M.; Baker, J. Dexmedetomidine and Propofol Use for Palliative Sedation Therapy (PST) in Pediatric Oncology: A Ten-Year Review (GP750). J. Pain Symptom Manag. 2022, 63, 1139–1140. [Google Scholar] [CrossRef]
- Johnson, L.M.; Frader, J.; Wolfe, J.; Baker, J.N.; Anghelescu, D.L.; Lantos, J.D. Palliative Sedation With Propofol for an Adolescent With a DNR Order. Pediatrics 2017, 140, e20170487. [Google Scholar] [CrossRef]
- Anghelescu, D.L.; Hamilton, H.; Faughnan, L.G.; Johnson, L.-M.; Baker, J.N. Pediatric Palliative Sedation Therapy with Propofol: Recommendations Based on Experience in Children with Terminal Cancer. J. Palliat. Med. 2012, 15, 1082–1090. [Google Scholar] [CrossRef]
- Anghelescu, D.L.; Faughnan, L.; Baker, J.N.; Yang, J.; Kane, J.R.; Bs, L.G.F. Use of epidural and peripheral nerve blocks at the end of life in children and young adults with cancer: The collaboration between a pain service and a palliative care service. Pediatr. Anesthesia 2010, 20, 1070–1077. [Google Scholar] [CrossRef]
- Collins, J.J.; Grier, H.E.; Sethna, N.F.; Wilder, R.T.; Berde, C.B. Regional anesthesia for pain associated with terminal pediatric malignancy. Pain 1996, 65, 63–69. [Google Scholar] [CrossRef]
- Cooper, M.G.; Keneally, J.P.; Kinchington, D. Continuous brachial plexus neural blockade in a child with intractable cancer pain. J. Pain Symptom Manag. 1994, 9, 277–281. [Google Scholar] [CrossRef]
- Anghelescu, D.L.; Guo, A.; Morgan, K.J.; Frett, M.; Prajapati, H.; Gold, R.; Federico, S.M. Pain Outcomes After Celiac Plexus Block in Children and Young Adults with Cancer. J. Adolesc. Young Adult Oncol. 2018, 7, 666–672. [Google Scholar] [CrossRef]
- Aram, L.; Krane, E.J.; Kozloski, L.J.; Yaster, M. Tunneled Epidural Catheters for Prolonged Analgesia in Pediatric Patients. Anesthesia Analg. 2001, 92, 1432–1438. [Google Scholar] [CrossRef]
- Berde, C.B.; Fischel, N.; Filardi, P.J.; Coe, C.S.; Grier, H.E.; Bernstein, S.C. Caudal epidural morphine analgesia for an infant with advanced neuroblastoma: Report of a case. Pain 1989, 36, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Plancarte, R.; Patt, R. Intractable upper body pain in a pediatric patient relieved with cervical epidural opioid administration. J. Pain Symptom Manag. 1991, 6, 98–99. [Google Scholar] [CrossRef] [PubMed]
- Coyne, P.J.; Smith, T.; Laird, J.; Hansen, L.A.; Drake, D. Effectively starting and titrating intrathecal analgesic therapy in patients with refractory cancer pain. Clin. J. Oncol. Nurs. 2005, 9, 581. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chambers, W. Nerve blocks in palliative care. Br. J. Anaesth. 2008, 101, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Baker, L.; Lee, M.; Regnard, C.; Crack, L.; Callin, S. Evolving spinal analgesia practice in palliative care. Palliat. Med. 2004, 18, 507–515. [Google Scholar] [CrossRef]
- Gertler, R.; Brown, H.C.; Mitchell, D.H.; Silvius, E.N. Dexmedetomidine: A Novel Sedative-Analgesic Agent. Bayl. Univ. Med. Cent. Proc. 2001, 14, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.-Q.; Chen, S.-Q.; Yao, X.; Xie, C.-B.; Wen, S.; Liu, K.-X. Clinical benefits of dexmedetomidine versus propofol in adult intensive care unit patients: A meta-analysis of randomized clinical trials. J. Surg. Res. 2013, 185, 833–843. [Google Scholar] [CrossRef]
- Jaap Vuyk, M. Pharmacokinetic and pharmacodynamic interactions between opioids and propofol. J. Clin. Anesth. 1997, 9, 23S–26S. [Google Scholar] [CrossRef]
- Wiener, L.; Kazak, A.E.; Noll, R.B.; Patenaude, A.F.; Kupst, M.J. Standards for the Psychosocial Care of Children with Cancer and Their Families: An Introduction to the Special Issue. Pediatric Blood Cancer 2015, 62, S419–S424. [Google Scholar] [CrossRef]
- Weaver, M.S.; Heinze, K.E.; Kelly, K.P.; Wiener, L.; Casey, R.L.; Bell, C.J.; Wolfe, J.; Garee, A.M.; Watson, A.; Hinds, P.S. Palliative Care as a Standard of Care in Pediatric Oncology. Pediatr. Blood Cancer 2015, 62, S829–S833. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuviello, A.; Johnson, L.-M.; Morgan, K.J.; Anghelescu, D.L.; Baker, J.N. Palliative Sedation Therapy in Pediatrics: An Algorithm and Clinical Practice Update. Children 2022, 9, 1887. https://doi.org/10.3390/children9121887
Cuviello A, Johnson L-M, Morgan KJ, Anghelescu DL, Baker JN. Palliative Sedation Therapy in Pediatrics: An Algorithm and Clinical Practice Update. Children. 2022; 9(12):1887. https://doi.org/10.3390/children9121887
Chicago/Turabian StyleCuviello, Andrea, Liza-Marie Johnson, Kyle J. Morgan, Doralina L. Anghelescu, and Justin N. Baker. 2022. "Palliative Sedation Therapy in Pediatrics: An Algorithm and Clinical Practice Update" Children 9, no. 12: 1887. https://doi.org/10.3390/children9121887
APA StyleCuviello, A., Johnson, L.-M., Morgan, K. J., Anghelescu, D. L., & Baker, J. N. (2022). Palliative Sedation Therapy in Pediatrics: An Algorithm and Clinical Practice Update. Children, 9(12), 1887. https://doi.org/10.3390/children9121887







