Psychometric Performance of HRQoL Measures: An Australian Paediatric Multi-Instrument Comparison Study Protocol (P-MIC)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Aims
2.2. Study Design
2.3. Timeline
2.4. Participants
- Sample one, recruited via hospital: 1000 parents/caregivers will be recruited via The RCH, Melbourne, Australia, allowing data to be collected from children with a wide range of health conditions and severities.
- Sample two, online panel population sample: 1500 parents/caregivers of children from a general population sample will be recruited via an online survey company, Pureprofile Australia, allowing data to be collected from a large population sample. This sample will be further divided into two groups:
- ○
- Sample 2a, online panel population sample, normal follow-up: 1300 parent/caregivers will be followed-up in line with the rest of the cohort, two to eight weeks after initial survey.
- ○
- Sample 2b, online panel population sample, short follow-up: to assess reliability, 200 parents/caregiver will be followed up at a shorter interval, two days after initial survey.
- Sample three, online panel disease-specific groups sample: 3600 parents/caregivers of children with one of nine health conditions will be recruited via an online survey company, Pureprofile Australia. Disease groups were chosen that had sufficient prevalence to allow for recruitment of groups and with a focus on conditions with larger expected HRQoL decrements [14]. Currently six of the disease groups have been confirmed and include: attention-deficit/hyperactivity Disorder (ADHD), anxiety and/or depression, autism spectrum disorder (ASD), asthma, dental decay, and sleep problems. An additional three disease groups will be selected from the following: diabetes, epilepsy, eating disorder, recurrent abdominal pain, frequent headaches. We aim to collect data on 400 unique children from each of the nine disease groups.
2.5. Recruitment
2.5.1. Sample One, Sample Recruited via Hospital (n = 1000)
2.5.2. Sample Two (Online Panel Population Sample, n = 1500) and Sample Three (Online Panel Disease Groups Sample, n = 3600)
2.6. Data Collection
2.6.1. Initial Survey
2.6.2. Follow-Up Survey
2.7. Participant Duration and Reimbursement
2.8. Sample Size
2.9. Statistical Analysis
2.9.1. Validity
2.9.2. Reliability
2.9.3. Responsiveness
2.9.4. Acceptability and Feasibility
2.9.5. Consistency
2.9.6. Factor Analysis
2.9.7. Item Response Theory
2.10. Data Security
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Overview of Instruments
| Instrument | Description | Application in This Study | Justification for Inclusion in P-MIC |
|---|---|---|---|
| Core non-HRQoL Instruments | |||
| EQ-HWB-S v1.2 | Part of the EuroQol family of instruments, the EQ-HWB-S is a new experimental instrument designed to assess the impact of being a care recipient or caregiver on health and wellbeing [20]. The short version currently has 9 items. | See Figure 1 and Table 1. | Included following advice from consumer advisory group who felt it was important to include a carer. QoL measure due to the strong relationship between child and carer. QoL in children who have chronic conditions. The EQ-HWB-S was chosen because it is a promising new instrument that requires further validation work and is an opportunity to add to the evidence base. |
| SDQ | The SDQ is a well-validated behavioral screening questionnaire used to assess a child’s emotional and behavioral wellbeing [21,22]. It has 25 items measuring 5 domains [23]. Cut points on each domain can be used to indicate borderline or clinically elevated symptoms. Versions of the SDQ exist for children aged 2–17 years, with self-report available for children aged 11–17 years. | See Figure 1 and Table 1. | Included to capture emotional wellbeing of children to allow the exploration of how HRQoL instruments perform for children with a mental health problem or mental health comorbidity. The SDQ was chosen because it is well validated across a wide age range with cut points to indicate clinically relevant symptoms. |
| Core HRQoL Instruments | |||
| PedsQL Generic Core 4.0 | The PedsQL Generic Core 4.0 is a 23 item HRQoL instrument covering 4 domains [24]. Validated version of the PedsQL exist for children aged 2–18 years with self-report available for children ≥8 years [24]. | See Figure 1 and Table 1. | Identified as priority for inclusion as it is a generic instrument widely used in pediatric populations with some evidence for performance, but requiring more. |
| TANDI v2 | The TANDI is a new proxy HRQoL instrument developed specifically for children ≤3 years based on the structure of the EQ-5D-Y-3L [25]. It consists of 6 items covering 6 domains [25]. | See Figure 1 and Table 1. | Identified as priority for inclusion because it is a promising new instrument for young children requiring further validation work. |
| EQ-5D-Y (3L and 5L) | The EQ-5D-Y-3L and recently developed EQ-5D-Y-5L, are youth versions adapted from the EQ-5D adult version [26,27]. Both the EQ-5D-Y-3L and EQ-5D-Y-5L are HRQoL instruments consisting of 5 items covering 5 domains. The EQ-5D-Y-3L has 3 severity levels and the 5L has 5. The EQ-5D-Y also includes a Visual Analogue Scale (VAS). Validated versions of the EQ-5D-Y exist for children aged 4–18 years, with self-report from ≥8 years. | See Figure 1 and Table 1. | The EQ-5D-Y-3L was identified as priority for inclusion as it is a generic instrument widely used in pediatric populations with good evidence for performance, but requiring more [13]. The EQ-5D-Y-5L was identified as priority for inclusion because it is a promising new instrument that requires further validation work. |
| CHU9D | The CHU9D was developed for the purpose of measuring HRQoL in children and young people [28]. It consists of 9 items covering 9 domains. It was initially developed for children aged 7–11 years; however, has been validated in children up to 17 years [29,30]. Additionally, a proxy version of the instrument is being trialed in younger children. | See Figure 1 and Table 1. | Identified as priority for inclusion as it is a generic instrument widely used in pediatric populations with good evidence for performance, but requiring more [13]. |
| Additional HRQoL Instruments | |||
| AQoL-6D Adolescent | The AQoL-6D Adolescent was adapted from the AQoL-6D adult version [31]. It is a HRQoL instrument consisting of 20 items covering 6 domains. It was developed with adolescents aged 12–18 years, however, has been used in children aged 11 years [32,33]. | See Figure 1 and Table 1. | Although not as widely used as the core instruments in pediatric populations [13], this instrument was identified for inclusion as an additional instrument because of the opportunity to add to the evidence base regarding how this ‘adolescent’ tool might perform in younger children. |
| HUI2/3 | The HUI3 was developed to address issues in the HUI2 and allow for the use of the HUI in both clinical and general populations. It consists of 15 items with between 4 and 6 severity levels. The HUI2 classification system has 7 domains [34] and the HUI3 has 8 [35]. The HUI is officially recommended for use in children ≥5 years, with self-report recommended for children ≥8; however, some studies have used the HUI in children as young as 1 year old [36]. | See Figure 1 and Table 1. | This instrument was identified for inclusion because it is a generic instrument widely used in pediatric populations [13]. It was chosen as an additional instrument because the evidence for the psychometric performance of this instrument is more mixed compared with that of core instruments [13]. |
| EQ-5D-5L | The EQ-5D-5L is an adult HRQoL instrument adapted from the EQ-5D-3L [37]. It consists of 5 items with 5 severity levels covering 5 domains. The EQ-5D-5L is recommended to be used in adults and children as young as 16 years. | See Figure 1 and Table 1. | Identified for inclusion as additional instrument because of the opportunity to add to the evidence base regarding how this ‘adult’ tool might perform in younger adolescents. |
| PROMIS-25 Paediatric Profile | The PROMIS-25 Paediatric Profile is a relatively new HRQoL instrument. It consists of 25 items covering 6 domains [38]. It has child-self report and proxy-report versions available, with child self-report recommended for ≥8 years age and proxy report from ≥5 years. | See Figure 1 and Table 1. | Identified for inclusion as additional instrument because it is a promising new instrument requiring further validation work. |
| Disease Specific Instruments | |||
| PedsQL Asthma Module (Asthma) | The PedsQL Asthma Module is a validated asthma specific pediatric HRQoL instrument [39]. It consists of 28 items (26 items for children aged 2–4 years) covering 4 domains [39]. There are self and proxy report versions for children ≥2 years. | See Figure 1 and Table 1. | The PedsQL Asthma Module was chosen as the disease-specific instrument for the asthma group as it is an asthma specific HRQoL instrument that has previously been validated in children. |
| KIDSCREEN-27 (ASD) | The KIDSCREEN-27 is a validated generic HRQoL instrument developed for use in children and adolescents and was adapted from the KIDSCREEN-52 [40,41]. It consists of 27 items covering 5 domains. A self-report and proxy version of the KIDSCREEN-27 is available. | See Figure 1 and Table 1. | The KIDSCREEN-27 was chosen as the disease-specific instrument for the ASD group as, although this is a generic HRQoL instrument, it has previously been recommended as a robust HRQoL instrument in children with ASD and no appropriate ASD-specific HRQoL instrument or symptom scale was available [42]. Additionally, a recent literature review found the KIDSCREEN-27 was one of two instruments that aligned with the Convention on the Rights of Persons with a Disability [43]. |
| SDSC (Sleep problems) | The Sleep Disturbance Scale for Children (SDSC) is a validated questionnaire designed to assess sleep-related behaviors in children and adolescents [44]. It consists of 26 items covering 6 domains. Only a proxy report version of the SDSC is available and it was initially validated in children aged 6–16 years; however, another study has successfully utilized the SDSC in children as young as 3 years [45]. | As only a proxy report version of the instrument is available, this study will only collect the SDSC via proxy report. | The SDSC was chosen as the disease-specific instrument for the sleep problems group as it is a validated symptom measure in children covering a range of domains and no appropriate sleep-specific HRQoL instrument was available. |
| CPQ 11-14 (Dental decay) | The Child Perceptions Questionnaire (CPQ) 11-14 is a validated child self-reported oral HRQOL instrument, and is the most commonly used oral HRQoL instrument in children [46,47]. The CPQ 11-14 consists of 16 core items and 2 global items. It was initially designed for children aged 11–14 years; however, recent evidence indicates it may be used in children as young as 5 [48]. | As only a self-report version of the 16-item instrument is available, this study will generate a proxy report version to ensure the ability to collect this instrument via proxy report in cases where the child is not able to self-report. | The CPQ 11-14 was chosen as the disease-specific instrument for the dental decay group as it is an oral health-specific HRQoL instrument that has previously been validated in children. |
| RCADS-25 (Mental health- anxiety and/or depression) | The Revised Children’s Anxiety and Depression Scale (RCADS-25) is a symptom-based scale measuring anxiety and depression in children [49]. It consists of 25 items, 15 related to anxiety and 10 related to depression, covering 6 domains (generalized anxiety disorder, major depressive disorder, obsessive compulsive disorder, panic disorder, separation anxiety disorder and social phobia). The RCADS-25 has been validated in children aged 7–18 years and has both child self-report and proxy report versions available [49]. | See Figure 1 and Table 1. | The RCADS-25 was chosen as the disease-specific instrument for the anxiety and/or depression group as it is a validated symptom measure in children covering a range of domains and no appropriate depression- or anxiety-specific HRQoL instrument was available. |
| SWAN-adapted (ADHD) | The Strengths and Weaknesses of Attention-Deficit/Hyperactivity Disorder Symptoms and Normal Behavior Scale (SWAN) is an ADHD symptom scale that was developed as an adaptation of the Swanson, Nolan and Pelham (SNAP) rating scale [50]. The SWAN is an 18-item proxy-reported measure with 7 severity levels covering 3 symptom areas. The instrument is validated in children aged 6–18 years, but has been used in children as young as 4 [51]. | With approval from developer, instrument was adapted slightly to increase readability for lay user. As only a proxy report version of the instrument is available, this study will only collect the SWAN via proxy report. | The SWAN was chosen as the disease-specific instrument for the ADHD group as it is a validated symptom measure in children covering a range of domains and no appropriate ADHD-specific HRQoL instrument was available. |
Appendix B. Additional Screening Questions for Sample Three, Online Disease Group Panel
| Disease Group | Screening Question | Inclusion Criteria |
|---|---|---|
| Asthma | Has your child been diagnosed with Asthma by a doctor? Yes/No | If ‘Yes’ to screening question. |
| ASD | Has your child been diagnosed with autism spectrum disorder (ASD) by a doctor? Yes/No | If ‘Yes’ to screening question. |
| Sleep problems | How much is (study child)’s sleeping pattern or habits a problem for you? Not a problem at all/A small problem/A moderate problem/A large problem | If ‘A moderate problem’ or ‘A large problem’ to screening question. |
| Dental decay | Has your child had tooth decay in the last 2 years (excluding preventative care)? Yes/No | If ‘Yes’ to screening question. |
| Anxiety and/or depression | Assessed via the SDQ. | As per SDQ clinical cut off for internalizing (anxiety and depression) problems. |
| ADHD | Has your child been diagnosed with Attention-Deficit/Hyperactivity Disorder (ADHD) by a doctor? Yes/No | If ‘Yes’ to screening question. |
Appendix C. Items Used to Assist in the Measurement of Change in Child’s Health Status between Initial and Follow-Up Survey
| Item Description | Purpose | Sample 1, Sample Recruited via Hospital | Sample 2, Online Panel Population Sample | Sample 3, Online Disease Group Sample | |||
|---|---|---|---|---|---|---|---|
| Initial | Follow-Up | Initial | Follow-Up | Initial | Follow-Up | ||
| Hospital department the child is currently receiving care from (reported by parent/caregiver). | Help identify groups of children expected to change at follow-up. | X | |||||
| Two item Children with Special Health Care Needs (CSHCN) Screener [52]. | Help identify groups of children expected to change at follow-up. | X | X | X | |||
| Child’s common ongoing health conditions (selected from list), reported by parent/caregiver. Adapted from from the Longitudinal Study of Australian Children (LSAC), Australia’s first nationally representative longitudinal study of child development. | Help identify groups of children expected to change at follow-up. | X | X | X | |||
| Single item global health measure, reported by parent/caregiver and child self-report if able. | Help identify change in health status between surveys. | X | X | X | X | X | X |
| PedsQL | Help identify minimal clinically important changes in health status between surveys. | X | X | X | X | X | X |
| Parent/caregiver report of child’s change in general health between initial and follow-up survey. Adapted from similar items within a previously validated instrument, SF-36 [53]. | Help identify change in health status between surveys. | X | X | X | |||
| Parent/caregiver report of child’s change in main health condition (if reported in initial survey that child has online health condition) between initial and follow-up survey. Adapted from similar items within a previously validated instrument, SF-36 [53]. | Help identify change in health status between surveys. | X | X | X | |||
| Parent/caregiver report of major health event between initial and follow-up survey (new treatment/therapy, new medication, accident or injury, new diagnosis, new illness, unplanned doctor/hospital visit). If any major health event selected, parent/caregiver asked to report if this event made the child’s health better worse or it had no change. | Help identify direction of change in health status between surveys. | X | X | X | |||
Appendix D. Detailed Sample Size Calculations
Appendix D.1. Responsiveness Sample Size Calculation
Appendix D.2. Known-Group Validity Sample Size Calculation
References
- Kromm, S.K.; Bethell, J.; Kraglund, F.; Edwards, S.A.; Laporte, A.; Coyte, P.C.; Ungar, W.J. Characteristics and quality of pediatric cost-utility analyses. Qual. Life Res. 2012, 21, 1315–1325. [Google Scholar] [CrossRef]
- Germain, N.; Aballéa, S.; Toumi, M. Measuring the health-related quality of life in young children: How far have we come? J. Mark. Access Health Policy 2019, 7, 1618661. [Google Scholar] [CrossRef]
- Dawson, J.; Doll, H.; Fitzpatrick, R.; Jenkinson, C.; Carr, A.J. The routine use of patient reported outcome measures in healthcare settings. BMJ 2010, 340, c186. [Google Scholar] [CrossRef]
- Bulfone, L.; Younie, S.; Carter, R. Health Technology Assessment: Reflections from the Antipodes. Value Health 2009, 12, S28–S38. [Google Scholar] [CrossRef][Green Version]
- Services Australia, Commonwealth of Australia. Annual Report 2019–2020. 2020. Available online: https://www.servicesaustralia.gov.au/sites/default/files/annual-report-2019-20v2.pdf (accessed on 1 June 2021).
- Services Australia, Commonwealth of Australia. PBS Expenditure and Precriptions Report 1 July 2019 to 30 June 2020. 2020. Available online: https://www.pbs.gov.au/statistics/expenditure-prescriptions/2019-2020/PBS_Expenditure_and_Prescriptions_Report_1-July-2019_to_30-June-2020.pdf (accessed on 1 June 2021).
- Higginson, I.J.; Carr, A.J. Using quality of life measures in the clinical setting. BMJ 2001, 322, 1297. [Google Scholar] [CrossRef]
- Bele, S.; Chugh, A.; Mohamed, B.; Teela, L.; Haverman, L.; Santana, M.J. Patient-Reported Outcome Measures in Routine Pediatric Clinical Care: A Systematic Review. Front. Pediatrics 2020, 8, 364. [Google Scholar] [CrossRef]
- Janssens, A.; Thompson Coon, J.; Rogers, M.; Allen, K.; Green, C.; Jenkinson, C.; Tennant, A.; Logan, S.; Morris, C. A systematic review of generic multidimensional patient-reported outcome measures for children, part I: Descriptive characteristics. Value Health 2015, 18, 315–333. [Google Scholar] [CrossRef]
- Finch, A.P.; Brazier, J.E.; Mukuria, C. What is the evidence for the performance of generic preference-based measures? A systematic overview of reviews. Eur. J. Health Econ. 2018, 19, 557–570. [Google Scholar] [CrossRef]
- Ungar, W. Economic Evaluation in Child Health; Oxford University Press: Oxford, UK, 2009. [Google Scholar]
- Chen, G.; Ratcliffe, J. A Review of the Development and Application of Generic Multi-Attribute Utility Instruments for Paediatric Populations. Pharmacoeconomics 2015, 33, 1013–1028. [Google Scholar] [CrossRef]
- Rowen, D.; Keetharuth, A.D.; Poku, E.; Wong, R.; Pennington, B.; Wailoo, A. A Review of the Psychometric Performance of Selected Child and Adolescent Preference-Based Measures Used to Produce Utilities for Child and Adolescent Health. Value Health 2021, 24, 443–460. [Google Scholar] [CrossRef]
- Liu, T.; Lingam, R.; Lycett, K.; Mensah, F.K.; Muller, J.; Hiscock, H.; Huque, M.H.; Wake, M. Parent-reported prevalence and persistence of 19 common child health conditions. Arch. Dis. Child. 2018, 103, 548–556. [Google Scholar] [CrossRef]
- Anthoine, E.; Moret, L.; Regnault, A.; Sébille, V.; Hardouin, J.-B. Sample size used to validate a scale: A review of publications on newly-developed patient reported outcomes measures. Health Qual. Life Outcomes 2014, 12, 1–10. [Google Scholar] [CrossRef]
- Varni, J.W.; Burwinkle, T.M.; Seid, M.; Skarr, D. The PedsQL 4.0 as a pediatric population health measure: Feasibility, reliability, and validity. Ambul. Pediatr. 2003, 3, 329–341. [Google Scholar] [CrossRef]
- Devlin, N.J.; Shah, K.K.; Feng, Y.; Mulhern, B.; van Hout, B. Valuing health-related quality of life: An EQ-5D-5L value set for England. Health Econ. 2018, 27, 7–22. [Google Scholar] [CrossRef] [PubMed]
- Mokkink, L.B.; Terwee, C.B.; Patrick, D.L.; Alonso, J.; Stratford, P.W.; Knol, D.L.; Bouter, L.M.; de Vet, H.C. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J. Clin. Epidemiol. 2010, 63, 737–745. [Google Scholar] [CrossRef]
- Lamping, D.L.; Schroter, S.; Marquis, P.; Marrel, A.; Duprat-Lomon, I.; Sagnier, P.-P. The Community-Acquired Pneumonia Symptom Questionnaire: A New, Patient-Based Outcome Measure to Evaluate Symptoms in Patients with Community-Acquired Pneumonia. Chest 2002, 122, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Peasgood, T.; Mukuria, C.; Carlton, J.; Connell, J.; Devlin, N.; Jones, K.; Lovett, R.; Naidoo, B.; Rand, S.; Rejon-Parrilla, J.C.; et al. What is the best approach to adopt for identifying the domains for a new measure of health, social care and carer-related quality of life to measure quality-adjusted life years? Application to the development of the EQ-HWB? Eur. J. Health Econ. 2021, 22, 1067–1081. [Google Scholar] [CrossRef]
- Goodman, R.; Ford, T.; Simmons, H.; Gatward, R.; Meltzer, H. Using the Strengths and Difficulties Questionnaire (SDQ) to screen for child psychiatric disorders in a community sample. Br. J. Psychiatry 2000, 177, 534–539. [Google Scholar] [CrossRef]
- Stone, L.L.; Otten, R.; Engels, R.C.; Vermulst, A.A.; Janssens, J.M. Psychometric properties of the parent and teacher versions of the strengths and difficulties questionnaire for 4- to 12-year-olds: A review. Clin. Child Fam. Psychol. Rev. 2010, 13, 254–274. [Google Scholar] [CrossRef]
- Goodman, R. The Strengths and Difficulties Questionnaire: A research note. J. Child Psychol. Psychiatry 1997, 38, 581–586. [Google Scholar] [CrossRef]
- Varni, J.W.; Seid, M.; Kurtin, P.S. PedsQL 4.0: Reliability and validity of the Pediatric Quality of Life Inventory version 4.0 generic core scales in healthy and patient populations. Med. Care 2001, 39, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Verstraete, J.; Ramma, L.; Jelsma, J. Validity and reliability testing of the Toddler and Infant (TANDI) Health Related Quality of Life instrument for very young children. J. Patient-Rep. Outcomes 2020, 4, 94. [Google Scholar] [CrossRef] [PubMed]
- Wille, N.; Badia, X.; Bonsel, G.; Burström, K.; Cavrini, G.; Devlin, N.; Egmar, A.C.; Greiner, W.; Gusi, N.; Herdman, M.; et al. Development of the EQ-5D-Y: A child-friendly version of the EQ-5D. Qual. Life Res. 2010, 19, 875–886. [Google Scholar] [CrossRef]
- Kreimeier, S.; Åström, M.; Burström, K.; Egmar, A.C.; Gusi, N.; Herdman, M.; Kind, P.; Perez-Sousa, M.A.; Greiner, W. EQ-5D-Y-5L: Developing a revised EQ-5D-Y with increased response categories. Qual. Life Res. 2019, 28, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
- Stevens, K. Developing a descriptive system for a new preference-based measure of health-related quality of life for children. Qual. Life Res. 2009, 18, 1105–1113. [Google Scholar] [CrossRef]
- Ratcliffe, J.; Stevens, K.; Flynn, T.; Brazier, J.; Sawyer, M. An assessment of the construct validity of the CHU9D in the Australian adolescent general population. Qual. Life Res. 2012, 21, 717–725. [Google Scholar] [CrossRef]
- Stevens, K.; Ratcliffe, J. Measuring and valuing health benefits for economic evaluation in adolescence: An assessment of the practicality and validity of the child health utility 9D in the Australian adolescent population. Value Health 2012, 15, 1092–1099. [Google Scholar] [CrossRef]
- Moodie, M.; Richardson, J.; Rankin, B.; Iezzi, A.; Sinha, K. Predicting time trade-off health state valuations of adolescents in four Pacific countries using the Assessment of Quality-of-Life (AQoL-6D) instrument. Value Health 2010, 13, 1014–1027. [Google Scholar] [CrossRef]
- Ratcliffe, J.; Stevens, K.; Flynn, T.; Brazier, J.; Sawyer, M.G. Whose Values in Health? An Empirical Comparison of the Application of Adolescent and Adult Values for the CHU-9D and AQOL-6D in the Australian Adolescent General Population. Value Health 2012, 15, 730–736. [Google Scholar] [CrossRef] [PubMed]
- Keating, C.L.; Moodie, M.L.; Richardson, J.; Swinburn, B.A. Utility-Based Quality of Life of Overweight and Obese Adolescents. Value Health 2011, 14, 752–758. [Google Scholar] [CrossRef]
- Torrance, G.W.; Feeny, D.H.; Furlong, W.J.; Barr, R.D.; Zhang, Y.; Wang, Q. Multiattribute Utility Function for a Comprehensive Health Status Classification System: Health Utilities Index Mark 2. Med. Care 1996, 34, 702–722. [Google Scholar] [CrossRef]
- Feeny, D.; Furlong, W.; Torrance, G.W.; Goldsmith, C.H.; Zhu, Z.; Depauw, S.; Denton, M.; Boyle, M. Multiattribute and Single-Attribute Utility Functions for the Health Utilities Index Mark 3 System. Med. Care 2002, 40, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Gemke, R.J.; Bonsel, G.J. Reliability and validity of a comprehensive health status measure in a heterogeneous population of children admitted to intensive care. J. Clin. Epidemiol. 1996, 49, 327–333. [Google Scholar] [CrossRef]
- Herdman, M.; Gudex, C.; Lloyd, A.; Janssen, M.F.; Kind, P.; Parkin, D.; Bonsel, G.; Badia, X. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual. Life Res. 2011, 20, 1727–1736. [Google Scholar] [CrossRef]
- Northwestern University. Intro to PROMIS®. 2021. Available online: https://www.healthmeasures.net/explore-measurement-systems/promis/intro-to-promis (accessed on 25 July 2021).
- Varni, J.W.; Burwinkle, T.M.; Rapoff, M.A.; Kamps, J.L.; Olson, N. The PedsQL in pediatric asthma: Reliability and validity of the Pediatric Quality of Life Inventory generic core scales and asthma module. J. Behav. Med. 2004, 27, 297–318. [Google Scholar] [CrossRef]
- Ravens-Sieberer, U.; Herdman, M.; Devine, J.; Otto, C.; Bullinger, M.; Rose, M.; Klasen, F. The European KIDSCREEN approach to measure quality of life and well-being in children: Development, current application, and future advances. Qual. Life Res. 2014, 23, 791–803. [Google Scholar] [CrossRef]
- Ravens-Sieberer, U.; Auquier, P.; Erhart, M.; Gosch, A.; Rajmil, L.; Bruil, J.; Power, M.; Duer, W.; Cloetta, B.; Czemy, L.; et al. The KIDSCREEN-27 quality of life measure for children and adolescents: Psychometric results from a cross-cultural survey in 13 European countries. Qual. Life Res. 2007, 16, 1347–1356. [Google Scholar] [CrossRef]
- Tavernor, L.; Barron, E.; Rodgers, J.; McConachie, H. Finding out what matters: Validity of quality of life measurement in young people with ASD. Child Care Health Dev. 2013, 39, 592–601. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.; Young, D.; Gilson, K.-M.; Swift, E.; Chan, J.; Gibbs, L.; Tonmukayakul, U.; Reddihough, D.; Williams, K. A Rights-Based Approach for Service Providers to Measure the Quality of Life of Children with a Disability. Value Health 2018, 21, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Bruni, O.; Ottaviano, S.; Guidetti, V.; Romoli, M.; Innocenzi, M.; Cortesi, F.; Giannotti, F. The Sleep Disturbance Scale for Children (SDSC). Construction and validation of an instrument to evaluate sleep disturbances in childhood and adolescence. J. Sleep Res. 1996, 5, 251–261. [Google Scholar] [CrossRef]
- Romeo, D.M.; Bruni, O.; Brogna, C.; Ferri, R.; Galluccio, C.; De Clemente, V.; Di Jorio, M.; Quintiliani, M.; Ricci, D.; Mercuri, E. Application of the Sleep Disturbance Scale for Children (SDSC) in preschool age. Eur. J. Paediatr. Neurol. 2013, 17, 374–382. [Google Scholar] [CrossRef]
- Thomson, W.M.; Foster Page, L.A.; Robinson, P.G.; Do, L.G.; Traebert, J.; Mohamed, A.R.; Turton, B.J.; McGrath, C.; Bekes, K.; Hirsch, C.; et al. Psychometric assessment of the short-form Child Perceptions Questionnaire: An international collaborative study. Community Dent. Oral Epidemiol. 2016, 44, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, F.; Rodd, H.; Deery, C.; Marshman, Z. Assessment of the quality of measures of child oral health-related quality of life. BMC Oral Health 2014, 14, 40. [Google Scholar] [CrossRef]
- Foster Page, L.A.; Boyd, D.; Thomson, W.M. Do we need more than one Child Perceptions Questionnaire for children and adolescents? BMC Oral Health 2013, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Ebesutani, C.; Korathu-Larson, P.; Nakamura, B.J.; Higa-McMillan, C.; Chorpita, B. The Revised Child Anxiety and Depression Scale 25-Parent Version: Scale Development and Validation in a School-Based and Clinical Sample. Assessment 2017, 24, 712–728. [Google Scholar] [CrossRef] [PubMed]
- Swanson, J.M.; Schuck, S.; Porter, M.M.; Carlson, C.; Hartman, C.A.; Sergeant, J.A.; Clevenger, W.; Wasdell, M.; McCleary, R.; Lakes, K.; et al. Categorical and Dimensional Definitions and Evaluations of Symptoms of ADHD: History of the SNAP and the SWAN Rating Scales. Int. J. Educ. Psychol. Assess 2012, 10, 51–70. [Google Scholar]
- Serrien, D.J.; Sovijärvi-Spapé, M.M.; Rana, G. Developmental changes in motor control: Insights from bimanual coordination. Dev. Psychol. 2014, 50, 316–323. [Google Scholar] [CrossRef]
- Bethell, C.D.; Read, D.; Stein, R.E.K.; Blumberg, S.J.; Wells, N.; Newacheck, P.W. Identifying Children with Special Health Care Needs: Development and Evaluation of a Short Screening Instrument. Ambul. Pediatrics 2002, 2, 38–48. [Google Scholar] [CrossRef]
- Stansfeld, S.A.; Roberts, R.; Foot, S.P. Assessing the validity of the SF-36 General Health Survey. Qual. Life Res. 1997, 6, 217–224. [Google Scholar] [CrossRef]
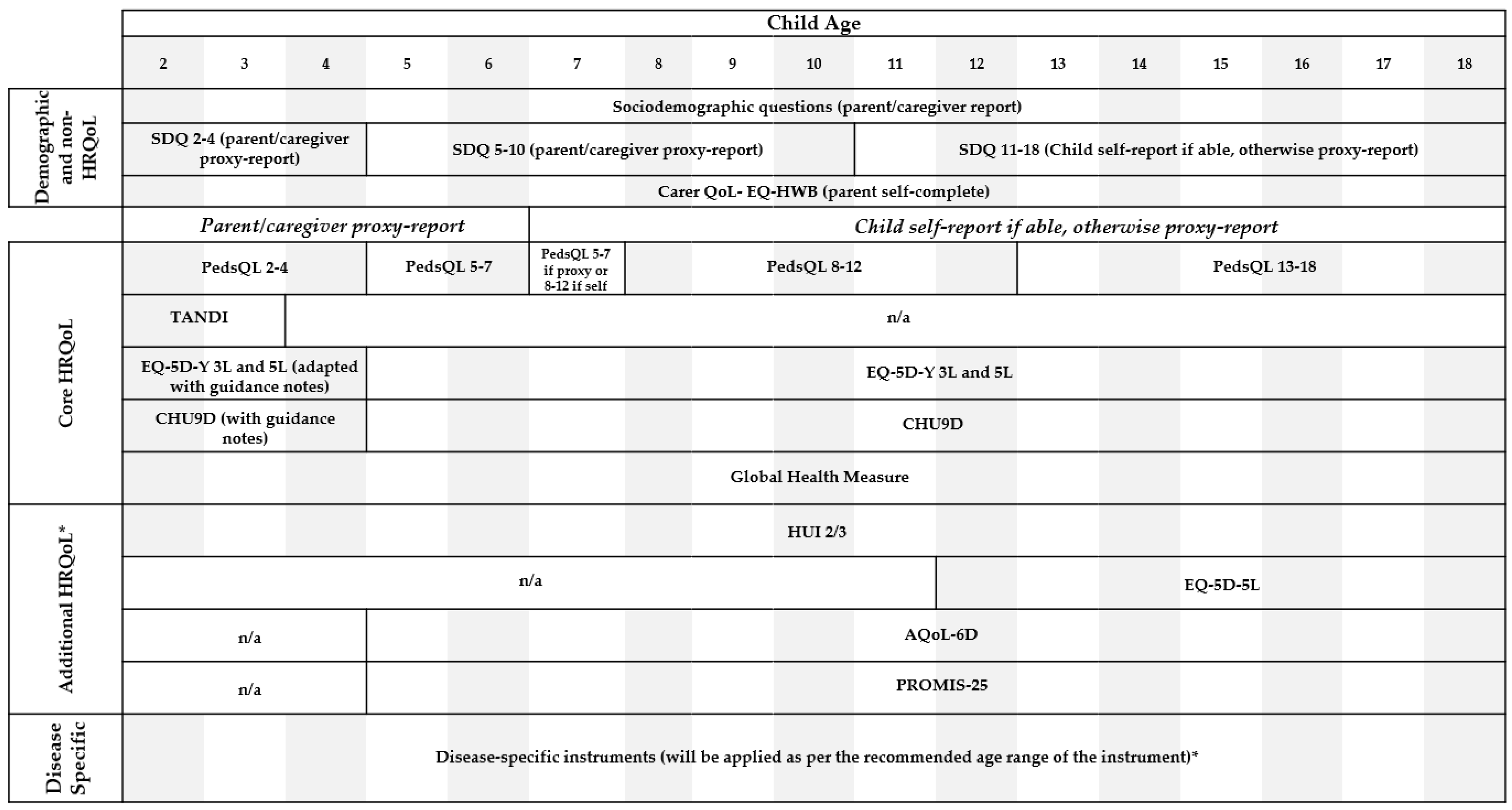
| Instrument | Sample 1, Sample Recruited via Hospital | Sample 2, Online Panel Population Sample | Sample 3, Online Disease Group Sample | |||
|---|---|---|---|---|---|---|
| Initial | Follow-Up | Initial | Follow-Up | Initial | Follow-Up | |
| Demographic and non-HRQoL instruments | ||||||
| Sociodemographic questions | X | X | X | |||
| SDQ | X | X | X | |||
| EQ-HWB | X | X | X | |||
| Core HRQoL instruments | ||||||
| PedsQL | X | X | X | X | X | X |
| TANDI (if <=3 years) | X | X | X | X | X | X |
| EQ-5D-Y 3L & 5L original (if >=5 years) | X | X | X | X | X | X |
| EQ-5D-Y 3L & 5L adapted (if <=4 years) | X | X | ||||
| EQ-5D-Y 3L original and adapted or EQ-5D-Y 5L original and adapted (if <=4 years) | X * | X * | X * | X * | ||
| CHU9D | X | X | X | X | X | X |
| Global Health Measure (single item) | X | X | X | X | X | X |
| Additional HRQoL instrument blocks | ||||||
| HUI 2/3 and EQ-5D-5L (>11 years) | X * | X * | X * | X * | ||
| AQoL-6D (>4 years) | X * | X * | X * | X * | ||
| PROMIS-25 (>4 years) | X * | X * | X * | X * | ||
| Disease specific instruments | ||||||
| Disease specific instruments (as per the recommended age range of the instrument) | X * | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, R.; Mulhern, B.; McGregor, K.; Yip, S.; O'Loughlin, R.; Devlin, N.; Hiscock, H.; Dalziel, K.; on behalf of the Quality Of Life in Kids: Key Evidence to Strengthen Decisions in Australia (QUOKKA) Project Team. Psychometric Performance of HRQoL Measures: An Australian Paediatric Multi-Instrument Comparison Study Protocol (P-MIC). Children 2021, 8, 714. https://doi.org/10.3390/children8080714
Jones R, Mulhern B, McGregor K, Yip S, O'Loughlin R, Devlin N, Hiscock H, Dalziel K, on behalf of the Quality Of Life in Kids: Key Evidence to Strengthen Decisions in Australia (QUOKKA) Project Team. Psychometric Performance of HRQoL Measures: An Australian Paediatric Multi-Instrument Comparison Study Protocol (P-MIC). Children. 2021; 8(8):714. https://doi.org/10.3390/children8080714
Chicago/Turabian StyleJones, Renee, Brendan Mulhern, Kristy McGregor, Shilana Yip, Rachel O'Loughlin, Nancy Devlin, Harriet Hiscock, Kim Dalziel, and on behalf of the Quality Of Life in Kids: Key Evidence to Strengthen Decisions in Australia (QUOKKA) Project Team. 2021. "Psychometric Performance of HRQoL Measures: An Australian Paediatric Multi-Instrument Comparison Study Protocol (P-MIC)" Children 8, no. 8: 714. https://doi.org/10.3390/children8080714
APA StyleJones, R., Mulhern, B., McGregor, K., Yip, S., O'Loughlin, R., Devlin, N., Hiscock, H., Dalziel, K., & on behalf of the Quality Of Life in Kids: Key Evidence to Strengthen Decisions in Australia (QUOKKA) Project Team. (2021). Psychometric Performance of HRQoL Measures: An Australian Paediatric Multi-Instrument Comparison Study Protocol (P-MIC). Children, 8(8), 714. https://doi.org/10.3390/children8080714






