Abdominal Wall Defects—Current Treatments
Abstract
1. Gastroschisis
1.1. Introduction
1.2. Complex Gastroschisis
1.3. Diagnostic Approach and Prenatal Management
1.4. Timing of Delivery
1.5. Route of Delivery
1.6. Postnatal Care
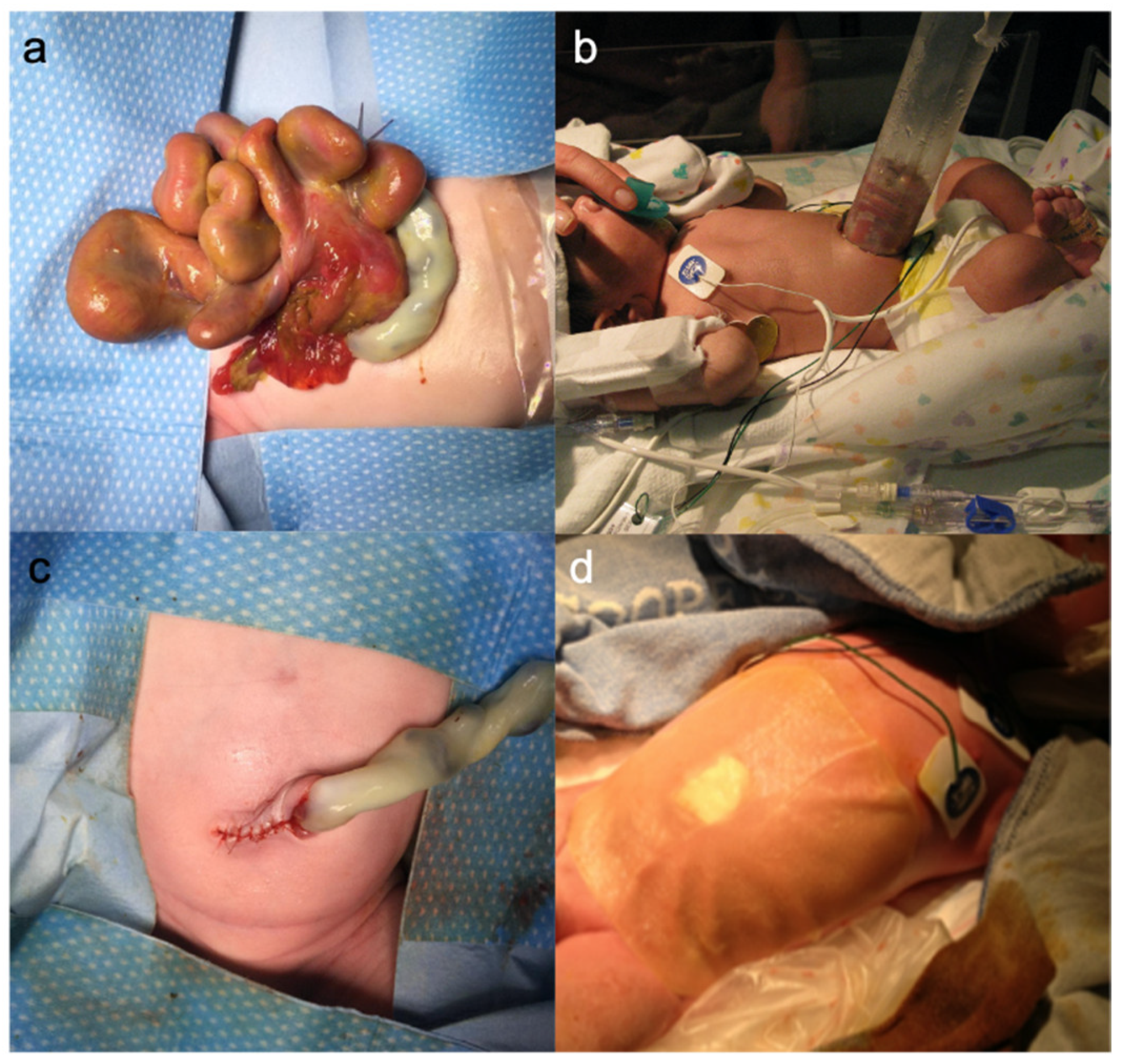
1.7. Surgical Treatment
1.8. Primary Versus Staged Reduction
1.9. Technique of Staged Reduction
1.10. Technique of Primary Reduction
1.11. Abdominal Wall Closure
1.11.1. Sutured Closure
1.11.2. Sutureless Closure of the Abdominal Wall
1.12. Treatment of Complicated Gastroschisis
1.13. Gastroschisis and Intestinal Atresia
1.14. Postoperative Course
1.15. Complications
2. Omphalocele
2.1. Introduction
2.2. Embryology and Pathogenesis
2.3. Prevalence and Epidemiology
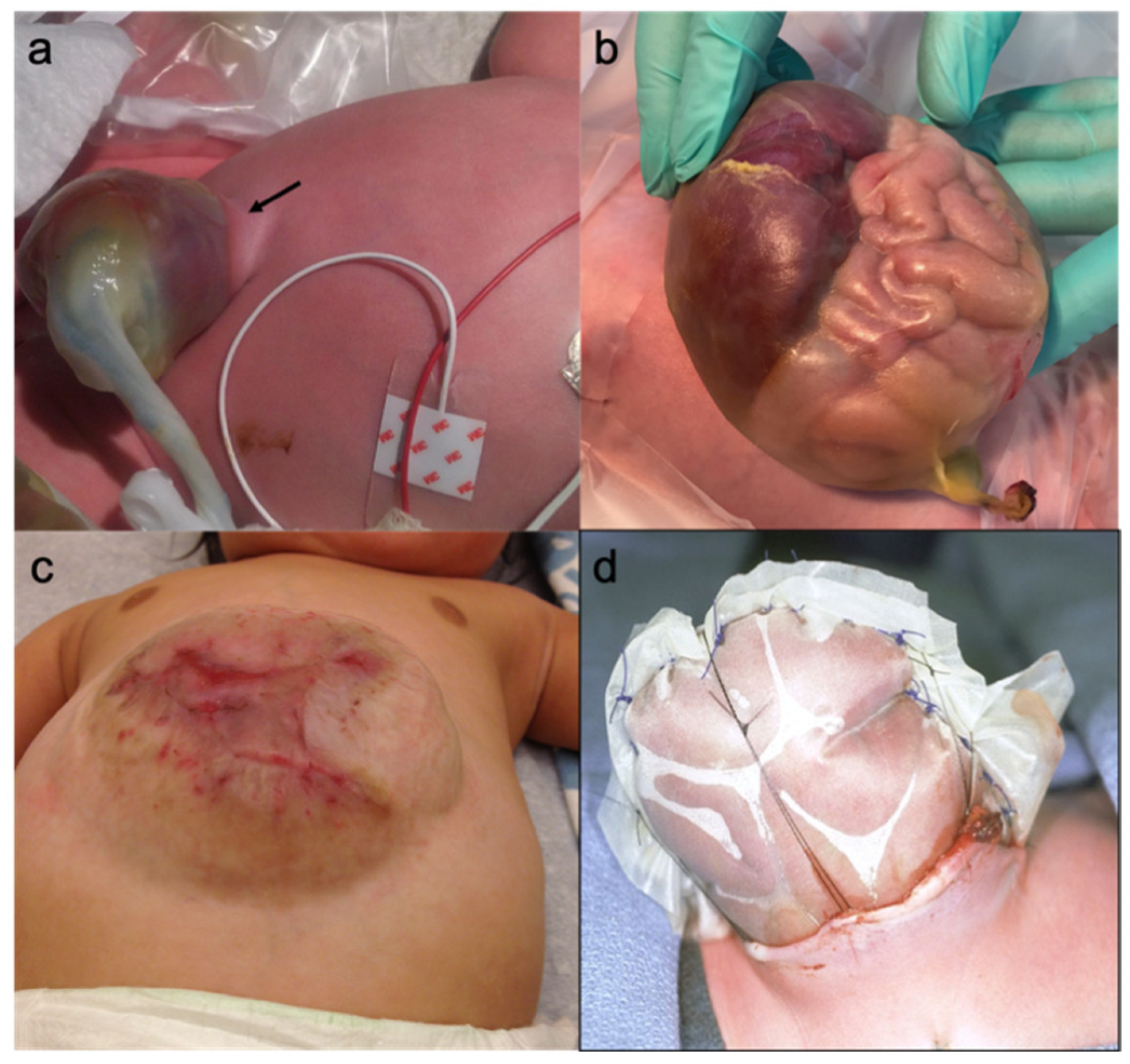
2.4. Giant Omphalocele
2.5. Hernia into the Cord
2.6. Prenatal Diagnosis
2.7. Monitoring of Fetal Grwoth
2.8. Delivery Timing and Route
2.9. Postnatal Resuscitation and Care
2.10. Postpartal Management
2.11. Treatment
2.12. Primary Repair
2.13. Staged Repair
2.14. Delayed Repair (Paint and Wait)
2.15. Long Term Complications and Outcome
Author Contributions
Funding
Conflicts of Interest
References
- Oakes, M.C.; Porto, M.; Chung, J.H. Advances in prenatal and perinatal diagnosis and management of gastroschisis. Semin. Pediatr. Surg. 2018, 27, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.M.; Isenburg, J.; Salemi, J.L.; Arnold, K.E.; Mai, C.T.; Aggarwal, D.; Arias, W.; Carrino, G.E.; Ferrell, E.; Folorunso, O.; et al. Increasing Prevalence of Gastroschisis—14 States, 1995–2012. Morb. Mortal. Wkly. Rep. 2016, 65, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Loane, M.; Dolk, H.; Bradbury, I.; EUROCAT Working Group. Increasing prevalence of gastroschisis in Europe 1980–2002: A phenomenon re-stricted to younger mothers? Paediatr. Perinat. Epidemiol. 2007, 21, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Kirby, R.S.; Marshall, J.; Tanner, J.P.; Salemi, J.L.; Feldkamp, M.L.; Marengo, L.; Meyer, R.E.; Druschel, C.M.; Rickard, R.; Kucik, J.E. Prevalence and Correlates of Gastroschisis in 15 States, 1995 to 2005. Obstet. Gynecol. 2013, 122, 275–281. [Google Scholar] [CrossRef]
- Kilby, M.D. The incidence of gastroschisis. BMJ 2006, 332, 250–251. [Google Scholar] [CrossRef] [PubMed]
- Stallings, E.B.; Isenburg, J.L.; Short, T.D.; Heinke, D.; Kirby, R.S.; Romitti, P.A.; Canfield, M.A.; O’Leary, L.A.; Liberman, R.F.; Forestieri, N.E.; et al. Population-based birth defects data in the United States, 2012–2016: A focus on abdominal wall defects. Birth Defects Res. 2019, 111, 1436–1447. [Google Scholar] [CrossRef]
- Clark, R.H.; Sousa, J.; Laughon, M.M.; Tolia, V.N. Gastroschisis prevalence substantially decreased from 2009 through 2018 after a 3-fold increase from 1997 to 2008. J. Pediatr. Surg. 2020, 55, 2640–2641. [Google Scholar] [CrossRef]
- Short, T.D.; Stallings, E.B.; Isenburg, J.; O’Leary, L.A.; Yazdy, M.M.; Bohm, M.K.; Ethen, M.; Chen, X.; Tran, T.; Fox, D.J.; et al. Gastroschisis Trends and Ecologic Link to Opioid Prescription Rates—United States, 2006–2015. MMWR. Morb. Mortal. Wkly. Rep. 2019, 68, 31–36. [Google Scholar] [CrossRef]
- Baldacci, S.; Santoro, M.; Coi, A.; Mezzasalma, L.; Bianchi, F.; Pierini, A. Lifestyle and sociodemographic risk factors for gastroschisis: A systematic review and meta-analysis. Arch. Dis. Child. 2020, 105, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Chen, Y.-L.; Li, J.; Li, L.-L.; Jiang, C.-Z.; Zhou, C.; Liu, C.-X.; Li, D.; Gong, T.-T.; Wu, Q.-J.; et al. Decreasing prevalence and time trend of gastroschisis in 14 cities of Liaoning Province: 2006–2015. Sci. Rep. 2016, 6, 33333. [Google Scholar] [CrossRef] [PubMed]
- Schmedding, A.; Wittekind, B.; Salzmann-Manrique, E.; Schloesser, R.; Rolle, U. Decentralized surgery of abdominal wall defects in Germany. Pediatr. Surg. Int. 2020, 36, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Spitz, L.; Coran, A.G. Operative Pediatric Surgery, 7th ed.; CRC Press, Taylor and Francis Group: London, UK, 2013; pp. 309–319. [Google Scholar]
- Ghionzoli, M.; James, C.P.; David, A.L.; Shah, D.; Tan, A.W.; Iskaros, J.; Drake, D.P.; Curry, J.I.; Kiely, E.M.; Cross, K.; et al. Gastroschisis with intestinal atresia—Predictive value of antenatal diagnosis and outcome of postnatal treatment. J. Pediatr. Surg. 2012, 47, 322–328. [Google Scholar] [CrossRef]
- Snyder, C.L.; Miller, K.A.; Sharp, R.J.; Murphy, J.P.; Andrews, W.A.; Holcomb 3rd, G.W.; Gittes, G.K.; Ashcraft, K.W. Management of intestinal atresia in patients with gastroschisis. J. Pediatr. Surg. 2001, 36, 1542–1545. [Google Scholar] [CrossRef]
- Arnold, M.A.; Chang, D.C.; Nabaweesi, R.; Colombani, P.M.; Bathurst, M.A.; Mon, K.S.; Hosmane, S.; Abdullah, F. Risk stratification of 4344 patients with gastroschisis into simple and complex categories. J. Pediatr. Surg. 2007, 42, 1520–1525. [Google Scholar] [CrossRef] [PubMed]
- Melov, S.J.; Tsang, I.; Cohen, R.; Badawi, N.; Walker, K.; Soundappan, S.S.V.; Alahakoon, T.I. Complexity of gastroschisis predicts outcome: Epidemiology and experience in an Australian tertiary center. BMC Pregnancy Childb. 2018, 18, 222. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, R.; Polak, M.; Kumar, T. A Proposed Classification for the Spectrum of Vanishing Gastroschisis. Eur. J. Pediatr. Surg. 2012, 23, 72–75. [Google Scholar] [CrossRef][Green Version]
- D’Antonio, F.; Virgone, C.; Rizzo, G.; Khalil, A.; Baud, D.; Cohen-Overbeek, T.E.; Kuleva, M.; Salomon, L.J.; Flacco, M.E.; Manzoli, L.; et al. Prenatal Risk Factors and Outcomes in Gastroschisis: A Meta-Analysis. Pediatrics 2015, 136, e159–e169. [Google Scholar] [CrossRef]
- Cyr, D.R.; A Mack, L.; A Schoenecker, S.; Patten, R.M.; Shepard, T.H.; Shuman, W.P.; A Moss, A. Bowel migration in the normal fetus: US detection. Radiology 1986, 161, 119–121. [Google Scholar] [CrossRef]
- Shalaby, A.; Davenport, M. Closed gastroschisis. Pediatr. Surg. Int. 2010, 27, 335. [Google Scholar] [CrossRef]
- Davenport, M.; Haugen, S.; Greenough, A.; Nicolaides, K. Closed gastroschisis: Antenatal and postnatal features. J. Pediatr. Surg. 2001, 36, 1834–1837. [Google Scholar] [CrossRef]
- Choudhury, R.A.; Yoeli, D.; Hoeltzel, G.; Moore, H.B.; Prins, K.; Kovler, M.; Goldstein, S.D.; Holland-Cunz, S.G.; Adams, M.; Roach, J.; et al. STEP improves long-term survival for pediatric short bowel syndrome patients: A Markov decision analysis. J. Pediatr. Surg. 2020, 55, 1802–1808. [Google Scholar] [CrossRef]
- Meier, C.F.; Camargo, S.M.R.; Hunziker, S.; Leu, S.; Holland-Cunz, S.G.; Verrey, F.; Vuille-Dit-Bille, R.N. Mucosal Monosaccharide Transporter Expression in Newborns with Jejunoileal Atresia and Along the Adult Intestine. J. Pediatr. Gastroenterol. Nutr. 2019, 69, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Meier, C.F.; Camargo, S.M.R.; Hunziker, S.; Moehrlen, U.; Gros, S.J.; Bode, P.K.; Leu, S.; Meuli, M.; Holland-Cunz, S.; Verrey, F.; et al. Intestinal IMINO transporter SIT1 is not expressed in human newborns. Am. J. Physiol. Liver Physiol. 2018, 315, G887–G895. [Google Scholar] [CrossRef] [PubMed]
- Landisch, R.M.; Yin, Z.; Christensen, M.; Szabo, A.; Wagner, A.J. Outcomes of gastroschisis early delivery: A systematic review and meta-analysis. J. Pediatr. Surg. 2017, 52, 1962–1971. [Google Scholar] [CrossRef] [PubMed]
- Baud, D.; Lausman, A.; Alfaraj, M.A.; Seaward, G.; Kingdom, J.; Windrim, R.; Langer, J.C.; Kelly, E.N.; Ryan, G. Expectant Management Compared with Elective Delivery at 37 Weeks for Gastroschisis. Obstet. Gynecol. 2013, 121, 990–998. [Google Scholar] [CrossRef] [PubMed]
- Sparks, T.N.; Shaffer, B.L.; Page, J.; Caughey, A.B. Gastroschisis: Mortality risks with each additional week of expectant man-agement. Am. J. Obstet. Gynecol. 2017, 216, 66.e61–66.e67. [Google Scholar] [CrossRef]
- Amin, R.; Domack, A.; Bartoletti, J.; Peterson, E.; Rink, B.; Bruggink, J.; Christensen, M.; Johnson, A.; Polzin, W.; Wagner, A.J. National Practice Patterns for Prenatal Monitoring in Gastroschisis: Gastroschisis Outcomes of Delivery (GOOD) Provider Sur-vey. Fetal Diagn. Therap. 2019, 45, 125–130. [Google Scholar] [CrossRef]
- Schib, K.; Schumacher, M.; Meuli, M.; Tharakan, S.; Subotic, U. Prenatal and Postnatal Management of Gastroschisis in Ger-man-Speaking Countries: Is There a Standardized Management? Eur. J. Pediatr. Surg. 2018, 28, 183–193. [Google Scholar] [CrossRef]
- Abdel-Latif, M.E.; Bolisetty, S.; Abeywardana, S.; Lui, K. Mode of delivery and neonatal survival of infants with gastroschisis in Australia and New Zealand. J. Pediatr. Surg. 2008, 43, 1685–1690. [Google Scholar] [CrossRef]
- Kirollos, D.W.; Abdel-Latif, M.E. Mode of delivery and outcomes of infants with gastroschisis: A meta-analysis of observational studies. Arch. Dis. Child Fetal Neonatal Ed. 2018, 103, F355–F363. [Google Scholar] [CrossRef]
- Segal, I.; Kang, C.; Albersheim, S.G.; Skarsgard, E.D.; Lavoie, P.M. Surgical site infections in infants admitted to the neonatal in-tensive care unit. J. Pediatr. Surg. 2014, 49, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Friedman, A.M.; Ananth, C.V.; Siddiq, Z.; D’Alton, M.E.; Wright, J.D. Gastroschisis: Epidemiology and mode of delivery, 2005–2013. Am. J. Obstet. Gynecol. 2016, 215, 348.e1–348.e9. [Google Scholar] [CrossRef] [PubMed]
- Brantberg, A.; Blaas, H.G.; Haugen, S.E.; Eik-Nes, S.H. Characteristics and outcome of 90 cases of fetal omphalocele. Ultrasound Obstet. Gynecol. 2005, 26, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Petrosyan, M.; Sandler, A.D. Closure methods in gastroschisis. Semin. Pediatr. Surg. 2018, 27, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Driver, C.; Bruce, J.; Bianchi, A.; Doig, C.; Dickson, A.; Bowen, J. The contemporary outcome of gastroschisis. J. Pediatr. Surg. 2000, 35, 1719–1723. [Google Scholar] [CrossRef]
- Henrich, K.; Huemmer, H.P.; Reingruber, B.; Weber, P.G. Gastroschisis and omphalocele: Treatments and long-term outcomes. Pediatr. Surg. Int. 2007, 24, 167–173. [Google Scholar] [CrossRef]
- Kunz, S.N.; Tieder, J.S.; Whitlock, K.; Jackson, J.C.; Avansino, J.R. Primary fascial closure versus staged closure with silo in patients with gastroschisis: A meta-analysis. J. Pediatr. Surg. 2013, 48, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.D.; Chun, K.; Moores, D.C.; Andrews, H. Gastroschisis: A simple technique for staged silo closure. J. Pediatr. Surg. 1995, 30, 1169–1171. [Google Scholar] [CrossRef]
- Wu, Y.; Vogel, A.M.; A Sailhamer, E.; Somme, S.; Santore, M.J.; Chwals, W.J.; Statter, M.B.; Liu, D.C. Primary insertion of a silastic spring-loaded silo for gastroschisis. Am. Surg. 2003, 69, 1083–1086. [Google Scholar]
- Jensen, A.R.; Waldhausen, J.H.; Kim, S.S. The use of a spring-loaded silo for gastroschisis: Impact on practice patterns and outcomes. Arch. Surg. 2009, 144, 516–519. [Google Scholar] [CrossRef]
- Emil, S. Surgical strategies in complex gastroschisis. Semin. Pediatr. Surg. 2018, 27, 309–315. [Google Scholar] [CrossRef]
- Yaster, M.; Scherer, T.L.; Stone, M.M.; Maxwell, L.G.; Schleien, C.L.; Wetzel, R.C.; Buck, J.R.; Nichols, D.G.; Colombani, P.M.; Dudgeon, D.L.; et al. Prediction of successful primary closure of congenital abdominal wall defects using intraoperative measurements. J. Pediatr. Surg. 1989, 24, 1217–1220. [Google Scholar] [CrossRef]
- Stringel, G. Large gastroschisis: Primary repair with gore-tex patch. J. Pediatr. Surg. 1993, 28, 653–655. [Google Scholar] [CrossRef]
- Krasna, I.H. Is early fascial closure necessary for omphalocele and gastroschisis? J. Pediatr. Surg. 1995, 30, 23–28. [Google Scholar] [CrossRef]
- Bianchi, A.; Dickson, A.P.; Alizai, N.K. Elective delayed midgut reduction-No anesthesia for gastroschisis: Selection and con-version criteria. J. Pediatr. Surg. 2002, 37, 1334–1336. [Google Scholar] [CrossRef] [PubMed]
- Witt, R.G.; Zobel, M.; Padilla, B.; Lee, H.; MacKenzie, T.C.; Vu, L. Evaluation of Clinical Outcomes of Sutureless vs. Sutured Closure Techniques in Gastroschisis Repair. JAMA Surg. 2019, 154, 33–39. [Google Scholar] [CrossRef] [PubMed]
- De Francisci, G.; Papasidero, A.E.; Spinazzola, G.; Galante, D.; Caruselli, M.; Pedrotti, D.; Caso, A.; Lambo, M.; Melchionda, M.; Faticato, M.G. Update on complications in pediatric anesthesia. Pediatr. Rep. 2013, 5, e2. [Google Scholar] [CrossRef]
- Wenk, K.; Sick, B.; Sasse, T.; Moehrlen, U.; Meuli, M.; Vuille-Dit-Bille, R.N. Incidence of metachronous contralateral inguinal hernias in children following unilateral repair—A meta-analysis of prospective studies. J. Pediatr. Surg. 2015, 50, 2147–2154. [Google Scholar] [CrossRef]
- Sandler, A.; Lawrence, J.; Meehan, J.; Phearman, L.; Soper, R. A “plastic” sutureless abdominal wall closure in gastroschisis. J. Pediatr. Surg. 2004, 39, 738–741. [Google Scholar] [CrossRef]
- Harris, E.L.; Minutillo, C.; Hart, S.; Warner, T.M.; Ravikumara, M.; Nathan, E.A.; Dickinson, J.E. The long term physical consequences of gastroschisis. J. Pediatr. Surg. 2014, 49, 1466–1470. [Google Scholar] [CrossRef]
- Tullie, L.G.C.; Bough, G.M.; Shalaby, A.; Kiely, E.M.; Curry, J.I.; Pierro, A.; De Coppi, P.; Cross, K.M.K. Umbilical hernia following gastroschisis closure: A common event? Pediatr. Surg. Int. 2016, 32, 811–814. [Google Scholar] [CrossRef]
- Youssef, F.; Gorgy, A.; Arbash, G.; Puligandla, P.S.; Baird, R.J. Flap versus fascial closure for gastroschisis: A systematic review and meta-analysis. J. Pediatr. Surg. 2016, 51, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Dewberry, L.C.; Hilton, S.A.; Vuille-Dit-Bille, R.N.; Liechty, K.W. Is Tapering Enteroplasty an Alternative to Resection of Dilated Bowel in Small Intestinal Atresia? J. Surg. Res. 2020, 246, 1–5. [Google Scholar] [CrossRef]
- Wallace, B.; Schuepbach, F.; Gaukel, S.; Marwan, A.I.; Staerkle, R.F.; Vuille-dit-Bille, R.N. Evidence according to Cochrane Systematic Reviews on Alterable Risk Factors for Anastomotic Leakage in Colorectal Surgery. Gastroenterol. Res. Pract. 2020, 2020, 9057963. [Google Scholar] [CrossRef]
- Balgi, S.; Singhal, S.; Mueller, G.; Batton, B. Feeding Intolerance and Poor Growth in Infants with Gastroschisis: Longitudinal Experience with Consecutive Patients over Thirteen Years. J. Neonatal Surg. 2015, 4, 42. [Google Scholar] [CrossRef]
- Bergholz, R.; Boettcher, M.; Reinshagen, K.; Wenke, K. Complex gastroschisis is a different entity to simple gastroschisis af-fecting morbidity and mortality—A systematic review and meta-analysis. J. Pediatr. Surg. 2014, 49, 1527–1532. [Google Scholar] [CrossRef]
- Derderian, S.C.; Good, R.; Vuille-Dit-Bille, R.N.; Carpenter, T.; Bensard, D.D. Central venous lines in critically ill children: Thrombosis but not infection is site dependent. J. Pediatr. Surg. 2019, 54, 1740–1743. [Google Scholar] [CrossRef] [PubMed]
- Giuffrè, M.; Piro, E.; Ortolano, R.; Siracusa, F.; Pinello, G.; La Placa, S.; Corsello, G.; Schierz, I. Predictive Factors of Abdominal Compartment Syndrome in Neonatal Age. Am. J. Perinatol. 2013, 31, 049–054. [Google Scholar] [CrossRef]
- Oldham, K.T.; Coran, A.G.; Drongowski, R.A.; Baker, P.J.; Wesley, J.R.; Polley Jr, T.Z. The development of necrotizing enterocolitis following repair of gastroschisis: A surprisingly high incidence. J. Pediatr. Surg. 1988, 23, 945–949. [Google Scholar] [CrossRef]
- Jayanthi, S.; Seymour, P.; Puntis, J.W.; Stringer, M.D. Necrotizing enterocolitis after gastroschisis repair: A preventable compli-cation? J. Pediatr. Surg. 1998, 33, 705–707. [Google Scholar] [CrossRef]
- Schlueter, R.K.; Abdessalam, S.F.; Raynor, S.C.; Cusick, R.A. Necrotizing Enterocolitis following Gastroschisis Repair: An Update. GMERJ 2019, 1, 5. [Google Scholar]
- Van Eijck, F.C.; Wijnen, R.M.; van Goor, H. The incidence and morbidity of adhesions after treatment of neonates with gas-troschisis and omphalocele: A 30-year review. J. Pediatr. Surg. 2008, 43, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Risby, K.; Husby, S.; Qvist, N.; Jakobsen, M.S. High mortality among children with gastroschisis after the neonatal period: A long-term follow-up study. J. Pediatr. Surg. 2017, 52, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Corey, K.M.; Hornik, C.P.; Laughon, M.M.; McHutchison, K.; Clark, R.H.; Smith, P.B. Frequency of anomalies and hospital outcomes in infants with gastroschisis and omphalocele. Early Hum. Dev. 2014, 90, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.; Salemi, J.L.; Tanner, J.P.; Ramakrishnan, R.; Feldkamp, M.L.; Marengo, L.K.; Meyer, R.E.; Druschel, C.M.; Rickard, R.; Kirby, R.S. Prevalence, Correlates, and Outcomes of Omphalocele in the United States, 1995–2005. Obstet. Gynecol. 2015, 126, 284–293. [Google Scholar] [CrossRef]
- Gross, R.E. Omphalocele (umbilical hernia). Am. J. Dis. Child. 1947, 74, 371. [Google Scholar]
- Gray, D.L.; Martin, C.M.; Crane, J.P. Differential diagnosis of first trimester ventral wall defect. J. Ultrasound Med. 1989, 8, 255–258. [Google Scholar] [CrossRef]
- Margulies, L. Omphalocele (Amniocele)—Its Anatomy and Etiology in Relation to Hernias of Umbilicus and the Umbilical Cord. Am. J. Obstet. Gynecol. 1945, 49, 695–699. [Google Scholar] [CrossRef]
- Hartwig, N.G.; Steffelaar, J.W.; Van de Kaa, C.; Schueler, J.A.; Vermeij-Keers, C. Abdominal wall defect associated with persistent cloaca. The embryologic clues in autopsy. Am. J. Clin. Pathol. 1991, 96, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Calzolari, E.; Volpato, S.; Bianchi, F.; Cianciulli, D.; Tenconi, R.; Clementi, M.; Calabrò, A.; Lungarotti, S.; Mastroiacovo, P.P.; Botto, L.; et al. Omphalocele and gastroschisis: A collaborative study of five Italian congenital malformation registries. Teratology 1993, 47, 47–55. [Google Scholar] [CrossRef]
- Springett, A.; Draper, E.S.; Rankin, J.; Rounding, C.; Tucker, D.; Stoianova, S.; Wellesley, D.; Morris, J.K. Birth prevalence and survival of exomphalos in England and Wales: 2005 to 2011. Birth Defects Res. Part A Clin. Mol. Teratol. 2014, 100, 721–725. [Google Scholar] [CrossRef] [PubMed]
- EUROCAT: Prevalence Charts and Tables—Prevalence per 10,000 Births. Omphalocele—2011 to 2018. Available online: https://eu-rd-platform.jrc.ec.europa.eu/eurocat/eurocat-data/prevalence_en (accessed on 13 December 2020).
- Raymond, S.L.; Downard, C.D.; Peter, S.D.S.; Baerg, J.; Qureshi, F.G.; Bruch, S.W.; Danielson, P.D.; Renaud, E.; Islam, S. Outcomes in omphalocele correlate with size of defect. J. Pediatr. Surg. 2019, 54, 1546–1550. [Google Scholar] [CrossRef] [PubMed]
- Forrester, M.B.; Merz, R.D. Impact of Demographic Factors on Prenatal Diagnosis and Elective Pregnancy Termination because of Abdominal Wall Defects, Hawaii, 1986–1997. Fetal Diagn. Ther. 1999, 14, 206–211. [Google Scholar] [CrossRef]
- Axt, R.; Quijano, F.; Boos, R.; Hendrik, H.J.; Jessberger, H.J.; Schwaiger, C.; Schmidt, W. Omphalocele and gastroschisis: Prenatal diagnosis and peripartal management. A case analysis of the years 1989–1997 at the Department of Obstetrics and Gynecology, University of Homburg/Saar. Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 87, 47–54. [Google Scholar] [CrossRef]
- Byron-Scott, R.; Haan, E.; Chan, A.; Bower, C.; Scott, H.; Clark, K. A population-based study of abdominal wall defects in South Australia and Western Australia. Paediatr. Périnat. Epidemiol. 1998, 12, 136–151. [Google Scholar] [CrossRef]
- Salihu, H.M.; Pierre-Louis, B.J.; Druschel, C.M.; Kirby, R.S. Omphalocele and gastroschisis in the state of New York, 1992–1999. Birth Defects Res. Part A Clin. Mol. Teratol. 2003, 67, 630–636. [Google Scholar] [CrossRef]
- Kirby, R.S. The prevalence of selected major birth defects in the United States. Semin. Perinatol. 2017, 41, 338–344. [Google Scholar] [CrossRef]
- Waller, D.K.; Shaw, G.M.; Rasmussen, S.A.; Hobbs, C.A.; Canfield, M.A.; Siega-Riz, A.M.; Gallaway, M.S.; Correa, A. National Birth Defects Prevention S: Prepregnancy obesity as a risk factor for structural birth defects. Arch. Pediat. Adol. Med. 2007, 161, 745–750. [Google Scholar] [CrossRef]
- Watkins, M.L.; Rasmussen, S.A.; Honein, M.A.; Botto, L.D.; Moore, C.A. Maternal obesity and risk for birth defects. Pediatrics 2003, 111, 1152–1158. [Google Scholar] [PubMed]
- Waller, D.K.; Keddie, A.M.; Canfield, M.A.; Scheuerle, A.E. Do infants with major congenital anomalies have an excess of mac-rosomia? Teratology 2001, 64, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Frolov, P.; Alali, J.; Klein, M.D. Clinical risk factors for gastroschisis and omphalocele in humans: A review of the literature. Pediatr. Surg. Int. 2010, 26, 1135–1148. [Google Scholar] [CrossRef] [PubMed]
- De Veciana, M.; Major, C.A.; Porto, M. Prediction of an abnormal karyotype in fetuses with omphalocele. Prenat. Diagn. 1994, 14, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Weksberg, R.; Shuman, C.; Beckwith, J.B. Beckwith-Wiedemann syndrome. Eur. J. Hum. Genet. 2010, 18, 8–14. [Google Scholar] [CrossRef]
- Achiron, R.; Yagel, S.; Weissman, A.; Lipitz, S.; Mashiach, S.; Goldman, B. Fetal lateral neck cysts: Early second-trimester trans-vaginal diagnosis, natural history and clinical significance. Ultrasound Obstet. Gynecol. 1995, 6, 396–399. [Google Scholar] [CrossRef] [PubMed]
- Campos, B.A.; Tatsuo, E.S.; Miranda, M.E. Omphalocele: How big does it have to be a giant one? J. Pediatr. Surg. 2009, 44, 1474–1475. [Google Scholar] [CrossRef]
- Hershenson, M.B.; Brouillette, R.T.; Klemka, L.; Raffensperger, J.D.; Poznanski, A.K.; Hunt, C.E. Respiratory insufficiency in newborns with abdominal wall defects. J. Pediatr. Surg. 1985, 20, 348–353. [Google Scholar] [CrossRef]
- Argyle, J.C. Pulmonary Hypoplasia in Infants with Giant Abdominal Wall Defects. Pediatr. Pathol. 1989, 9, 43–55. [Google Scholar] [CrossRef]
- Lipitz, S.; Ravia, J.; Zolti, M.; Achiron, R.; Wolf, Y.; Kazanstein, A.; Goldenberg, M.; Seidman, D. Sequential genetic events leading to conjoined twins in a monozygotic triplet pregnancy. Hum. Reprod. 1995, 10, 3130–3132. [Google Scholar] [CrossRef] [PubMed]
- Achiron, R.; Soriano, D.; Lipitz, S.; Mashiach, S.; Goldman, B.; Seidman, D.S. Fetal midgut herniation into the umbilical cord: Im-proved definition of ventral abdominal anomaly with the use of transvaginal sonography. Ultrasound Obstet. Gynecol. 1995, 6, 256–260. [Google Scholar] [CrossRef]
- Ceccanti, S.; Falconi, I.; Frediani, S.; Boscarelli, A.; Musleh, L.; Cozzi, D.A. Umbilical cord sparing technique for repair of congenital hernia into the cord and small omphalocele. J. Pediatr. Surg. 2017, 52, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Pal, K. Congenital hernia of the umbilical cord associated with extracelomic colonic atresia and perforation of gut in a newborn. Afr. J. Paediatr. Surg. 2014, 11, 74–76. [Google Scholar] [CrossRef]
- Klein, M.D.; Kosloske, A.M.; Hertzler, J.H. Congenital defects of the abdominal wall. A review of the experience in New Mexico. JAMA 1981, 245, 1643–1646. [Google Scholar] [CrossRef]
- EUROCAT: Prenatal Detection Rates Charts and Tables-Overall Proportion of Cases Prenatally Diagnosed, 2014–2018. Available online: https://eu-rd-platform.jrc.ec.europa.eu/eurocat/eurocat-data/prenatal-screening-and-diagnosis_en (accessed on 13 December 2020).
- Curtis, J.A.; Watson, L. Sonographic diagnosis of omphalocele in the first trimester of fetal gestation. J. Ultrasound Med. 1988, 7, 97–100. [Google Scholar] [CrossRef][Green Version]
- Brown, D.; Emerson, D.; Shulman, L.; Carson, S. Sonographic diagnosis of omphalocele during 10th week of gestation. Am. J. Roentgenol. 1989, 153, 825–826. [Google Scholar] [CrossRef]
- Pagliano, M.; Mossetti, M.; Ragno, P. Echographic Diagnosis of Omphalocele in the First Trimester of Pregnancy. J. Clin. Ultrasound 1990, 18, 658–660. [Google Scholar] [CrossRef] [PubMed]
- Benacerraf, B.R.; Saltzman, D.H.; Estroff, J.A.; Frigoletto, F.D. Abnormal karyotype of fetuses with omphalocele: Prediction based on omphalocele contents. Obstet. Gynecol. 1990, 75, 317–319. [Google Scholar]
- Van Zalen-Sprock, R.M.; Vugt, J.M.; van Geijn, H.P. First-trimester sonography of physiological midgut herniation and early diagnosis of omphalocele. Prenat. Diagn. 1997, 17, 511–518. [Google Scholar] [CrossRef]
- Fleurke-Rozema, H.; Van De Kamp, K.; Bakker, M.; Pajkrt, E.; Bilardo, C.; Snijders, R. Prevalence, timing of diagnosis and pregnancy outcome of abdominal wall defects after the introduction of a national prenatal screening program. Prenat. Diagn. 2017, 37, 383–388. [Google Scholar] [CrossRef]
- Calvert, N.; Damiani, S.; Sunario, J.; Bower, C.; Dickinson, J.E. The outcomes of pregnancies following a prenatal diagnosis of fetal exomphalos in Western Australia. Aust. N. Z. J. Obstet. Gynaecol. 2009, 49, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Salomon, L.J.; Sotiriadis, A.; Wulff, C.B.; Odibo, A.; Akolekar, R. Risk of miscarriage following amniocentesis or chorionic villus sampling: Systematic review of literature and updated meta-analysis. Ultrasound Obstet. Gynecol. 2019, 54, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Pescia, G.; Guex, N.; Iseli, C.; Brennan, L.; Osteras, M.; Xenarios, I.; Farinelli, L.; Conrad, B. Cell-free DNA testing of an extended range of chromosomal anomalies: Clinical experience with 6,388 consecutive cases. Genet. Med. 2016, 19, 169–175. [Google Scholar] [CrossRef]
- Xu, L.-L.; Zhen, L.; Lou, J.-W.; Tang, H.-S.; Pan, M.; Han, J.; Yang, X.; Li, D.-Z. Can cell-free DNA testing be used in pregnancies with isolated fetal omphalocele? Preliminary evidence from cytogenetic results of prenatal cases. J. Matern. Neonatal Med. 2021, 34, 624–628. [Google Scholar] [CrossRef]
- Heider, A.L.; A Strauss, R.; A Kuller, J. Omphalocele: Clinical outcomes in cases with normal karyotypes. Am. J. Obstet. Gynecol. 2004, 190, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.D.; Nyberg, D.A.; Mack, L.A.; Pretorius, D.H. Fetal omphalocele: Prenatal US detection of concurrent anomalies and other predictors of outcome. Radiology 1989, 173, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Poznanski, A.K. Fetal omphalocele: Prenatal US detection of concurrent anomalies and other predictors of outcome. Radiology 1990, 177, 883–884. [Google Scholar] [CrossRef]
- Nicholas, S.S.; Stamilio, D.M.; Dicke, J.M.; Gray, D.L.; Macones, G.A.; Odibo, A.O. Predicting adverse neonatal outcomes in fetuses with abdominal wall defects using prenatal risk factors. Am. J. Obstet. Gynecol. 2009, 201, 383.e1–383.e6. [Google Scholar] [CrossRef] [PubMed]
- Hidaka, N.; Tsukimori, K.; Hojo, S.; Fujita, Y.; Yumoto, Y.; Masumoto, K.; Taguchi, T.; Wake, N. Correlation between the presence of liver herniation and perinatal outcome in prenatally diagnosed fetal omphalocele. J. Périnat. Med. 2009, 37, 66–71. [Google Scholar] [CrossRef]
- Hidaka, N.; Murata, M.; Yumoto, Y.; Hojo, S.; Fujita, Y.; Masumoto, K.; Taguchi, T.; Tsukimori, K.; Wake, N. Characteristics and perinatal course of prenatally diagnosed fetal abdominal wall defects managed in a tertiary center in Japan. J. Obstet. Gynaecol. Res. 2009, 35, 40–47. [Google Scholar] [CrossRef]
- Siemer, J.; Hilbert, A.; Hart, N.; Hoopmann, M.; Schneider, U.; Girschick, G.; Müller, A.; Schild, R.L. Specific weight formula for fetuses with abdominal wall defects. Ultrasound Obstet. Gynecol. 2008, 31, 397–400. [Google Scholar] [CrossRef]
- Deng, K.; Qiu, J.; Dai, L.; Yi, L.; Deng, C.; Mu, Y.; Zhu, J. Perinatal mortality in pregnancies with omphalocele: Data from the Chinese national birth defects monitoring network, 1996–2006. BMC Pediatr. 2014, 14, 160. [Google Scholar] [CrossRef]
- Heinke, D.; Nestoridi, E.; Hernandez-Diaz, S.; Williams, P.L.; Rich-Edwards, J.W.; Lin, A.E.; Van Bennekom, C.M.; Mitchell, A.A.; Nembhard, W.N.; Fretts, R.C.; et al. Risk of Stillbirth for Fetuses With Specific Birth Defects. Obstet. Gynecol. 2020, 135, 133–140. [Google Scholar] [CrossRef]
- Gamba, P.; Midrio, P. Abdominal wall defects: Prenatal diagnosis, newborn management, and long-term outcomes. Semin. Pediatr. Surg. 2014, 23, 283–290. [Google Scholar] [CrossRef]
- Lurie, S.; Sherman, D.; Bukovsky, I. Omphalocele delivery enigma: The best mode of delivery still remains dubious. Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 82, 19–22. [Google Scholar] [CrossRef]
- Segel, S.Y.; Marder, S.J.; Parry, S.; Macones, G.A. Fetal abdominal wall defects and mode of delivery: A systematic review. Obstet. Gynecol. 2001, 98, 867–873. [Google Scholar] [CrossRef]
- Skarsgard, E.D. Immediate versus staged repair of omphaloceles. Semin. Pediatr. Surg. 2019, 28, 89–94. [Google Scholar] [CrossRef]
- Yazbeck, S.; Ndoye, M.; Khan, A.H. Omphalocele: A 25-year experience. J. Pediatr. Surg. 1986, 21, 761–763. [Google Scholar] [CrossRef]
- Akakpo-Numado, G.K.; Gnassingbe, K.; Boume, M.A.; Sakiye, K.A.; Mihluedo-Agbolan, K.; Attipou, K.; Tekou, H. Emergency treatment of a ruptured huge omphalocele by simple suture of its membrane. Ann. Surg. Innov. Res. 2012, 6. [Google Scholar] [CrossRef]
- Krummel, T.M.; Sieber, W.K. Closure of congenital abdominal wall defects with umbilicoplasty. Surg. Gynecol. Obstet. 1987, 165, 168–169. [Google Scholar]
- Lee, S.L.; DuBois, J.J.; Greenholz, S.K.; Huffman, S.G. Advancement flap umbilicoplasty after abdominal wall closure: Postoper-ative results compared with normal umbilical anatomy. J. Pediatr. Surg. 2001, 36, 1168–1170. [Google Scholar] [CrossRef] [PubMed]
- Michel, J.-L.; Kassir, R.; Harper, L.; Gavage, L.; Frade, F.; Clermidi, P.; Sauvat, F.; Ramful, D.; Jean-Luc, M.; Rani, K.; et al. ZORRO: Z Omphaloplasty Repair for Omphalocele. J. Pediatr. Surg. 2018, 53, 1424–1427. [Google Scholar] [CrossRef]
- Pacilli, M.; Spitz, L.; Kiely, E.M.; Curry, J.; Pierro, A. Staged repair of giant omphalocele in the neonatal period. J. Pediatr. Surg. 2005, 40, 785–788. [Google Scholar] [CrossRef]
- Gonzalez, K.W.; Chandler, N.M. Ruptured omphalocele: Diagnosis and management. Semin. Pediat. Surg. 2019, 28, 101–105. [Google Scholar] [CrossRef]
- Gross, R.E. A new method for surgical treatment of large omphaloceles. Surgery 1948, 24, 277–292. [Google Scholar]
- Schuster, S.R. A new method for the staged repair of large omphaloceles. Surg. Gynecol. Obstet. 1967, 125, 837–850. [Google Scholar]
- Yokomori, K.; Ohkura, M.; Kitano, Y.; Hori, T.; Nakajo, T. Advantages and pitfalls of amnion inversion repair for the treatment of large unruptured omphalocele: Results of 22 cases. J. Pediatr. Surg. 1992, 27, 882–884. [Google Scholar] [CrossRef]
- Hong, A.; Sigalet, D.; Guttman, F.; Laberge, J.; Croitoru, D. Sequential sac ligation for giant omphalocele. J. Pediatr. Surg. 1994, 29, 413–415. [Google Scholar] [CrossRef]
- Martin, A.E.; Khan, A.; Kim, D.S.; Muratore, C.S.; Luks, F.I. The use of intraabdominal tissue expanders as a primary strategy for closure of giant omphaloceles. J. Pediatr. Surg. 2009, 44, 178–182. [Google Scholar] [CrossRef]
- Brown, M.F.; Wright, L. Delayed external compression reduction of an omphalocele (DECRO): An alternative method of treatment for moderate and large omphaloceles. J. Pediatr. Surg. 1998, 33, 1113–1116. [Google Scholar] [CrossRef]
- De Lorimier, A.A.; Adzick, N.S.; Harrison, M.R. Amnion inversion in the treatment of giant omphalocele. J. Pediatr. Surg. 1991, 26, 804–807. [Google Scholar] [CrossRef]
- Levy, S.; Tsao, K.; Cox, C.S.; Phatak, U.R.; Lally, K.P.; Andrassy, R.J. Component separation for complex congenital abdominal wall defects: Not just for adults anymore. J. Pediatr. Surg. 2013, 48, 2525–2529. [Google Scholar] [CrossRef] [PubMed]
- Heller, L.; McNichols, C.H.; Ramirez, O.M. Component Separations. Semin. Plast. Surg. 2012, 26, 25–28. [Google Scholar] [CrossRef]
- Mortellaro, V.E.; Peter, S.D.S.; Fike, F.B.; Islam, S. Review of the evidence on the closure of abdominal wall defects. Pediatr. Surg. Int. 2010, 27, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Moazam, F.; Rodgers, B.M.; Talbert, J.L. Use of teflon mesh for repair of abdominal wall defects in neonates. J. Pediatr. Surg. 1979, 14, 347–351. [Google Scholar] [CrossRef]
- Clifton, M.S.; Heiss, K.F.; Keating, J.J.; Mackay, G.; Ricketts, R.R. Use of tissue expanders in the repair of complex abdominal wall defects. J. Pediatr. Surg. 2011, 46, 372–377. [Google Scholar] [CrossRef]
- Wagner, J.P.; Cusick, R.A. Paint and wait management of giant omphaloceles. Semin. Pediatr. Surg. 2019, 28, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Ein, S.H.; Langer, J.C. Delayed management of giant omphalocele using silver sulfadiazine cream: An 18-year experience. J. Pediatr. Surg. 2012, 47, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Whitehouse, J.S.; Gourlay, D.M.; Masonbrink, A.R.; Aiken, J.J.; Calkins, C.M.; Sato, T.T.; Arca, M.J. Conservative management of giant omphalocele with topical povidone-iodine and its effect on thyroid function. J. Pediatr. Surg. 2010, 45, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Lunzer, H.; Menardi, G.; Brezinka, C. Long-term follow-up of children with prenatally diagnosed omphalocele and gas-troschisis. J. Matern. Fetal Med. 2001, 10, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Partridge, E.A.; Peranteau, W.H.; Flake, A.W.; Adzick, N.S.; Hedrick, H.L. Frequency and complications of inguinal hernia repair in giant omphalocele. J. Pediatr. Surg. 2015, 50, 1673–1675. [Google Scholar] [CrossRef]
- Abdelhafeez, A.H.; Schultz, J.A.; Ertl, A.; Cassidy, L.D.; Wagner, A.J. The risk of volvulus in abdominal wall defects. J. Pediatr. Surg. 2015, 50, 570–572. [Google Scholar] [CrossRef] [PubMed]
- van Eijck, F.C.; de Blaauw, I.; Bleichrodt, R.P.; Rieu, P.N.; van der Staak, F.H.; Wijnen, M.H.; Wijnen, R.M. Closure of giant omphaloceles by the abdominal wall component separation technique in infants. J. Pediatr. Surg. 2008, 43, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Danzer, E.; Gerdes, M.; D’Agostino, J.A.; Bernbaum, J.; Siegle, J.; Hoffman, C.; Rintoul, N.E.; Liechty, K.W.; Flake, A.W.; Adzick, N.S.; et al. Prospective, interdisciplinary follow-up of children with prenatally diagnosed giant omphalocele: Short-term neurode-velopmental outcome. J. Pediatr. Surg. 2010, 45, 718–723. [Google Scholar] [CrossRef] [PubMed]
| Gastroschisis | Omphalocele | |
|---|---|---|
| Location | Mostly right of umbilical cord | Midline |
| Etiology | Involution of right umbilical vein | Failed omphalomesenteric duct involution vs. failed lateral abdominal wall closure |
| Associated disorders/anomalies | Intestinal Atresia (10–15%), perforation, necrosis | Cardiac (32%), chromosomal (17%), CNS (8%), other (GU, GI, musculoskeletal, eyes, ears, face, neck (21%)). Isolated omphalocele without anomalies (22%) |
| Special forms | Complex gastroschisis: Gastroschisis associated with intestinal disorders (atresia, perforation, etc.), 11–28% Closing/closed gastroschisis: abdominal wall defect getting smaller, strangulated eviscerated bowel, towards end of pregnancy Vanishing gastroschisis: Involution of the eviscerated bowel (due to closed gastroschisis) | Giant omphalocele: abdomino-visceral disproportion, no clear definition (defect ≥ 5 cm and/or herniated liver of >50–75%) Hernia into the cord: Cuff of skin at base of sack, normal abdominal wall muscles, rarely associated disorders, primary closure often possible. |
| Prenatal ultrasound | Free floating herniated bowel, umbilical cord insertion in abdominal wall, seldomly liver herniation | Membranous sac covering viscera (prenatal sac rupture possible), umbilical cord insertion into sac, liver herniation |
| Mode of delivery | vaginal or caesarean | Small omphaloceles: vaginal. Large omphaloceles/herniated liver: caesarean |
| Timing of delivery | Elective late preterm or early term (35–38 wGA) | At term |
| Postnatal resuscitation and care | Place saline soaked gauzes around defect Plastic bag around lower ½ of patient Get IV access (avoid umbilical vein) Avoid hypothermia Oro- (or naso-) gastric (+ rectal) decompression Avoid mask ventilation (no intubation by default) | |
| Reduction of simple gastroschisis | Staged- seems favorable over primary reduction |
| Closure of simple gastroschisis | Sutured (needs general anesthesia, more infections) versus sutureless (more hernias) closure |
| Reduction of complicated gastroschisis | Staged reduction (spring-loaded silo) |
| Closure of complicated gastroschisis | Sutured versus sutureless |
| Gastroschisis and atresia | Primary resection/anastomosis vs. ostomy formation vs. delayed primary closure (after silo treatment) |
| Postoperative course | Time to return of bowel function: up to 4 weeks |
| Complications | Abdominal compartment syndrome, NEC (mostly benign), midgut volvulus (seldom), adhesive Small bowel obstruction (first year of life) |
| Repair of hernias into the cord/small omphaloceles | Primary repair |
| Repair of larger (giant) omphaloceles without abdomino-visceral disproportion/pulmonary hypertension | Staged repair (e.g., Schuster) |
| Repair of giant omphaloceles with abdomino-visceral disproportion/pulmonary hypertension | Paint and wait |
| Complications | Gastroesophageal reflux, feeding difficulties, Failure to thrive, chronic lung disease, inguinal hernias, abdominal compartment syndrome, midgut volvulus (seldom), adhesive small bowel obstruction |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bielicki, I.N.; Somme, S.; Frongia, G.; Holland-Cunz, S.G.; Vuille-dit-Bille, R.N. Abdominal Wall Defects—Current Treatments. Children 2021, 8, 170. https://doi.org/10.3390/children8020170
Bielicki IN, Somme S, Frongia G, Holland-Cunz SG, Vuille-dit-Bille RN. Abdominal Wall Defects—Current Treatments. Children. 2021; 8(2):170. https://doi.org/10.3390/children8020170
Chicago/Turabian StyleBielicki, Isabella N., Stig Somme, Giovanni Frongia, Stefan G. Holland-Cunz, and Raphael N. Vuille-dit-Bille. 2021. "Abdominal Wall Defects—Current Treatments" Children 8, no. 2: 170. https://doi.org/10.3390/children8020170
APA StyleBielicki, I. N., Somme, S., Frongia, G., Holland-Cunz, S. G., & Vuille-dit-Bille, R. N. (2021). Abdominal Wall Defects—Current Treatments. Children, 8(2), 170. https://doi.org/10.3390/children8020170





