Targeted Therapy for Pulmonary Hypertension in Premature Infants
Abstract
1. Introduction
2. Materials and Methods
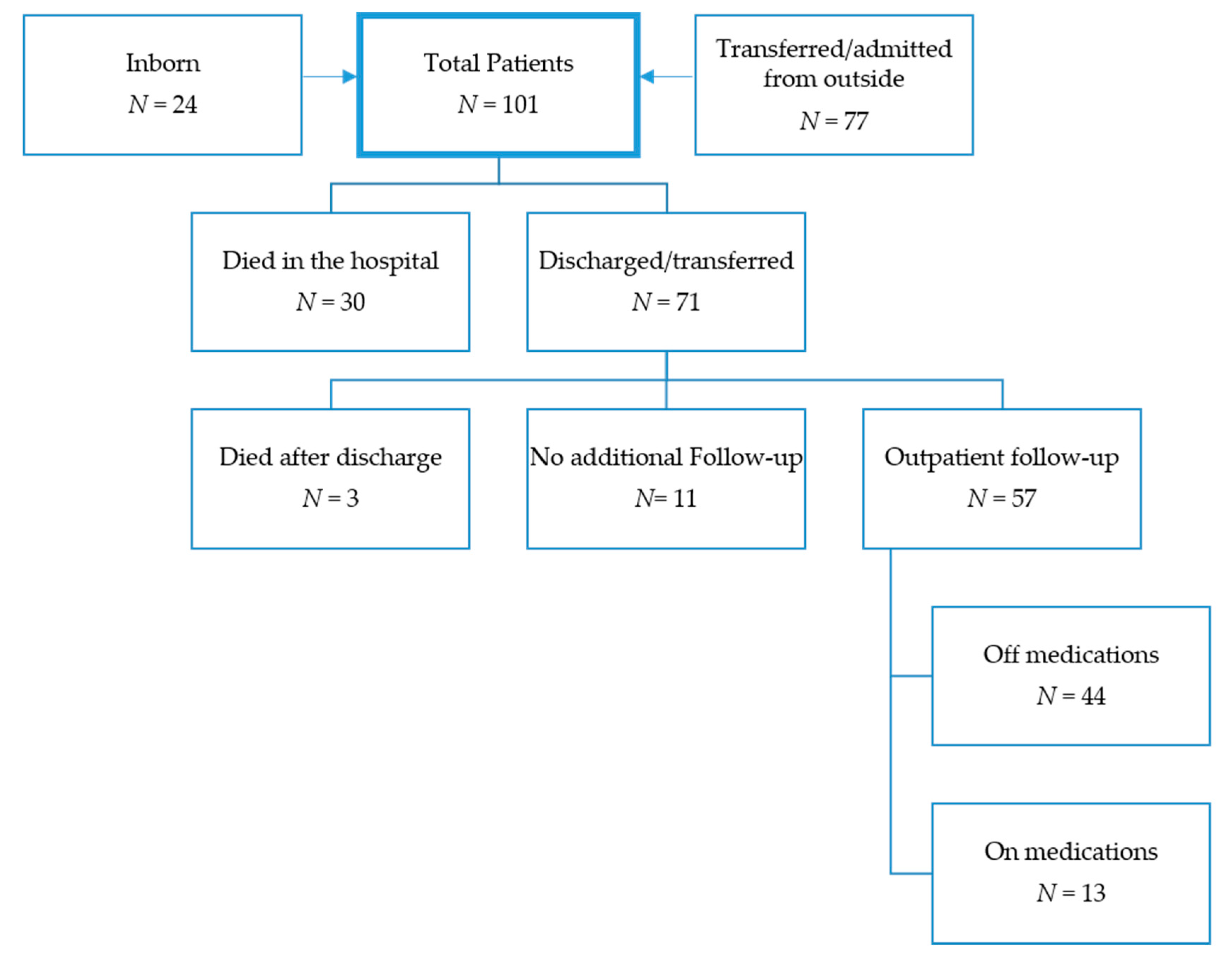
3. Results
3.1. Patient Population
3.1.1. Birth and Pregnancy
3.1.2. Pulmonary Hypertension
3.1.3. Associated Lesions and NICU Course
3.2. Pharmacologic Treatment with Targeted PH Medications
3.2.1. Sildenafil
3.2.2. Bosentan
3.2.3. Inhaled Iloprost
3.2.4. Intravenous Epoprostenol and Subcutaneous Treprostenil
3.3. Morbidity and Mortality
3.4. Discharge and Follow-Up
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abman, S.H. Monitoring cardiovascular function in infants with chronic lung disease of prematurity. Arch. Dis. Child. Fetal Neonatal Ed. 2002, 87. [Google Scholar] [CrossRef]
- Mourani, P.M.; Abman, S.H. Pulmonary vascular disease in bronchopulmonary dysplasia: Pulmonary hypertension and beyond. Curr. Opin. Pediatr. 2013, 25, 329–337. [Google Scholar] [CrossRef]
- Kinsella, J.P.; Greenough, A.; Abman, S.H. Bronchopulmonary dysplasia. Lancet 2006, 367, 1421–1431. [Google Scholar] [CrossRef]
- Jobe, A.H.; Bancalari, E. Bronchopulmonary Dysplasia. Am. J. Respir. Crit. Care Med. 2001, 163, 1723–1729. [Google Scholar] [CrossRef] [PubMed]
- An, H.S.; Bae, E.J.; Kim, G.B.; Kwon, B.S.; Beak, J.S.; Kim, E.K.; Kim, H.S.; Choi, J.H.; Noh, C.I.; Yun, Y.S. Pulmonary hypertension in preterm infants with bronchopulmonary dysplasia. Korean Circ. J. 2010, 40, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Bhat, R.; Salas, A.A.; Foster, C.; Carlo, W.A.; Ambalavanan, N. Prospective analysis of pulmonary hypertension in extremely low birth weight infants. Pediatrics 2012, 129, e682–e689. [Google Scholar] [CrossRef] [PubMed]
- Lagatta, J.M.; Hysinger, E.B.; Zaniletti, I.; Wymore, E.M.; Vyas-Read, S.; Yallapragada, S.; Nelin, L.D.; Truog, W.E.; Padula, M.A.; Porta, N.F.M.; et al. The Impact of Pulmonary Hypertension in Preterm Infants with Severe Bronchopulmonary Dysplasia through 1 Year. J. Pediatr. 2018, 203, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Weismann, C.G.; Asnes, J.D.; Bazzy-Asaad, A.; Tolomeo, C.; Ehrenkranz, R.A.; Bizzarro, M.J. Pulmonary hypertension in preterm infants: Results of a prospective screening program. J. Perinatol. 2017, 37, 572–577. [Google Scholar] [CrossRef]
- Ali, Z.; Schmidt, P.; Dodd, J.; Jeppesen, D.L. Predictors of bronchopulmonary dysplasia and pulmonary hypertension in newborn children. Dan. Med. J. 2013, 60, A4688. [Google Scholar]
- Check, J.; Gotteiner, N.; Liu, X.; Su, E.; Porta, N.; Steinhorn, R.; Mestan, K.K. Fetal growth restriction and pulmonary hypertension in premature infants with bronchopulmonary dysplasia. J. Perinatol. 2013, 33, 553–557. [Google Scholar] [CrossRef]
- Arjaans, S.; Zwart, E.A.H.; Ploegstra, M.-J.; Bos, A.F.; Kooi, E.M.W.; Hillege, H.L.; Berger, R.M.F. Identification of gaps in the current knowledge on pulmonary hypertension in extremely preterm infants: A systematic review and meta-analysis. Paediatr. Perinat. Epidemiol. 2018, 32, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Kim, H.S.; Choi, C.W.; Kim, E.K.; Kim, B.I.; Choi, J.H. Risk factors for pulmonary artery hypertension in preterm infants with moderate or severe bronchopulmonary dysplasia. Neonatology 2011, 101, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Devries, L.B.; Heyne, R.J.; Ramaciotti, C.; Brown, L.S.; Jaleel, M.A.; Kapadia, V.S.; Burchfield, P.J.; Brion, L.P. Mortality among infants with evolving bronchopulmonary dysplasia increases with major surgery and with pulmonary hypertension. J. Perinatol. 2017, 37, 1043–1046. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, U.; Feinstein, J.A.; Adatia, I.; Austin, E.D.; Mullen, M.P.; Hopper, R.K.; Hanna, B.; Romer, L.; Keller, R.L.; Fineman, J.; et al. Evaluation and Management of Pulmonary Hypertension in Children with Bronchopulmonary Dysplasia. J. Pediatr. 2017, 188, 24–34.e1. [Google Scholar] [CrossRef]
- Abman, S.H.; Hansmann, G.; Archer, S.L.; Ivy, D.D.; Adatia, I.; Chung, W.K.; Hanna, B.D.; Rosenzweig, E.B.; Raj, J.U.; Cornfield, D.; et al. Pediatric Pulmonary Hypertension: Guidelines from the American Heart Association and American Thoracic Society. Circulation 2015, 132, 2037–2099. [Google Scholar] [CrossRef]
- Krishnan, U. Management of pulmonary arterial hypertension in the neonatal unit. Cardiol. Rev. 2010, 18, 73–75. [Google Scholar] [CrossRef]
- Mourani, P.M.; Sontag, M.K.; Ivy, D.D.; Abman, S.H. Effects of long-term sildenafil treatment for pulmonary hypertension in infants with chronic lung disease. J Pediatr 2009, 154, 379–384. [Google Scholar] [CrossRef]
- Southard, A.E.; Edelmann, L.J.; Gelb, B.D.; Redon, R.; Ishikawa, S.; Fitch, K.; Conrad, D.; Pinto, D.; Redon, R.; Park, H.; et al. Role of copy number variants in structural birth defects. Pediatrics 2012, 129, 755–763. [Google Scholar] [CrossRef]
- Trottier-Boucher, M.N.; Lapointe, A.; Malo, J.; Fournier, A.; Raboisson, M.J.; Martin, B.; Moussa, A. Sildenafil for the Treatment of Pulmonary Arterial Hypertension in Infants with Bronchopulmonary Dysplasia. Pediatr. Cardiol. 2015, 36, 1255–1260. [Google Scholar] [CrossRef]
- Slaughter, J.L.; Pakrashi, T.; Jones, D.E.; South, A.P.; Shah, T.A. Echocardiographic detection of pulmonary hypertension in extremely low birth weight infants with bronchopulmonary dysplasia requiring prolonged positive pressure ventilation. J. Perinatol. 2011, 31, 1–6. [Google Scholar] [CrossRef]
- Khemani, E.; McElhinney, D.B.; Rhein, L.; Andrade, O.; Lacro, R.V.; Thomas, K.C.; Mullen, M.P. Pulmonary Artery Hypertension in Formerly Premature Infants With Bronchopulmonary Dysplasia: Clinical Features and Outcomes in the Surfactant Era. Pediatrics 2007, 120, 1260–1269. [Google Scholar] [CrossRef] [PubMed]
- Mahgoub, L.; Kaddoura, T.; Kameny, A.R.; Lopez Ortego, P.; Vanderlaan, R.D.; Kakadekar, A.; Dicke, F.; Rebeyka, I.; Calderone, C.A.; Redington, A.; et al. Pulmonary vein stenosis of ex-premature infants with pulmonary hypertension and bronchopulmonary dysplasia, epidemiology, and survival from a multicenter cohort. Pediatr. Pulmonol. 2017, 52, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Del Cerro, M.J.; Sabaté Rotés, A.; Cartõn, A.; Deiros, L.; Bret, M.; Cordeiro, M.; Verdú, C.; Barrios, M.I.; Albajara, L.; Gutierrez-Larraya, F. Pulmonary hypertension in bronchopulmonary dysplasia: Clinical findings, cardiovascular anomalies and outcomes. Pediatr. Pulmonol. 2014, 49, 49–59. [Google Scholar] [CrossRef] [PubMed]
- DM, D.; DW, K.; KO, M.; WT, M. Pulmonary vein stenosis: Prematurity and associated conditions. Pediatrics 2008, 122, e656–e661. [Google Scholar]
- Mourani, P.M.; Ivy, D.D.; Gao, D.; Abman, S.H. Pulmonary vascular effects of inhaled nitric oxide and oxygen tension in bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 2004, 170, 1006–1013. [Google Scholar] [CrossRef]
- Barst, R.J.; Beghetti, M.; Pulido, T.; Layton, G.; Konourina, I.; Zhang, M.; Ivy, D.D. STARTS-2: Long-term survival with oral sildenafil monotherapy in treatment-naive pediatric pulmonary arterial hypertension. Circulation 2014, 129, 1914–1923. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Center for Drug Evaluation and Research FDA Drug Safety Communication: FDA Recommends against Use of Revatio (Sildenafil) in Children with Pulmonary Hypertension; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2012.
- European Medicines Agency. Assessment Report for Revatio; European Medicines Agency: Amsterdam, The Netherlands, 2011; Volume 44. [Google Scholar]
- Barst, R.J.; Ivy, D.D.; Gaitan, G.; Szatmari, A.; Rudzinski, A.; Garcia, A.E.; Sastry, B.K.S.; Pulido, T.; Layton, G.R.; Serdarevic-Pehar, M.; et al. A randomized, double-blind, placebo-controlled, dose-ranging study of oral sildenafil citrate in treatment-naive children with pulmonary arterial hypertension. Circulation 2012, 125, 324–334. [Google Scholar] [CrossRef]
- Abman, S.H.; Kinsella, J.P.; Rosenzweig, E.B.; Krishnan, U.; Kulik, T.; Mullen, M.; Wessel, D.L.; Steinhorn, R.; Adatia, I.; Hanna, B.; et al. Implications of the U.S. Food and Drug Administration warning against the use of sildenafil for the treatment of pediatric pulmonary hypertension. Am. J. Respir. Crit. Care Med. 2013, 187, 572–575. [Google Scholar] [CrossRef]
- Backes, C.H.; Reagan, P.B.; Smith, C.V.; Jadcherla, S.R.; Slaughter, J.L. Sildenafil Treatment of Infants with Bronchopulmonary Dysplasia-Associated Pulmonary Hypertension. Hosp. Pediatr. 2016, 6, 27–33. [Google Scholar] [CrossRef]
- Nyp, M.; Sandritter, T.; Poppinga, N.; Simon, C.; Truog, W.E. Sildenafil citrate, bronchopulmonary dysplasia and disordered pulmonary gas exchange: Any benefits? J Perinatol 2012, 32, 64–69. [Google Scholar] [CrossRef]
- Kadmon, G.; Schiller, O.; Dagan, T.; Bruckheimer, E.; Birk, E.; Schonfeld, T. Pulmonary hypertension specific treatment in infants with bronchopulmonary dysplasia. Pediatr. Pulmonol. 2017, 52, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; Krishnamurthy, M.B.; O’Heney, J.L.; Paul, E.; Sehgal, A. Sildenafil therapy in bronchopulmonary dysplasia-associated pulmonary hypertension: A retrospective study of efficacy and safety. Eur. J. Pediatr. 2015, 174, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Wardle, A.J.; Wardle, R.; Luyt, K.; Tulloh, R. The utility of sildenafil in pulmonary hypertension: A focus on bronchopulmonary dysplasia. Arch. Dis. Child. 2013, 98, 613–617. [Google Scholar] [CrossRef] [PubMed]
- Van der Graaf, M.; Rojer, L.A.; Helbing, W.A.; Reiss, I.K.M.; Etnel, J.R.G.; Bartelds, B. Sildenafil for bronchopulmonary dysplasia and pulmonary hypertension: A meta-analysis. Pulm. Circ. 2019, 9. [Google Scholar] [CrossRef]
- Cohen, J.L.; Nees, S.N.; Valencia, G.A.; Rosenzweig, E.B.; Krishnan, U.S. Sildenafil Use in Children with Pulmonary Hypertension. J. Pediatr. 2019, 205, 29–34. [Google Scholar] [CrossRef]
- Siehr, S.L.; McCarthy, E.K.; Ogawa, M.T.; Feinstein, J.A. Reported Sildenafil Side Effects in Pediatric Pulmonary Hypertension Patients. Front. Pediatr. 2015, 3. [Google Scholar] [CrossRef]
- Krishnan, U.; Krishnan, S.; Gewitz, M. Treatment of pulmonary hypertension in children with chronic lung disease with newer oral therapies. Pediatr. Cardiol. 2008, 29, 1082–1086. [Google Scholar] [CrossRef]
- Ivy, D.D.; Doran, A.K.; Smith, K.J.; Mallory, G.B.; Beghetti, M.; Barst, R.J.; Brady, D.; Law, Y.; Parker, D.; Claussen, L.; et al. Short- and Long-Term Effects of Inhaled Iloprost Therapy in Children With Pulmonary Arterial Hypertension. J. Am. Coll. Cardiol. 2008, 51, 161–169. [Google Scholar] [CrossRef]
- Piastra, M.; De Luca, D.; De Carolis, M.P.; Tempera, A.; Stival, E.; Caliandro, F.; Pietrini, D.; Conti, G.; De Rosa, G. Nebulized iloprost and noninvasive respiratory support for impending hypoxaemic respiratory failure in formerly preterm infants: A case series. Pediatr. Pulmonol. 2012, 47, 757–762. [Google Scholar] [CrossRef]
- Zaidi, A.N.; Dettorre, M.D.; Ceneviva, G.D.; Thomas, N.J. Epoprostenol and home mechanical ventilation for pulmonary hypertension associated with chronic lung disease. Pediatr. Pulmonol. 2005, 40, 265–269. [Google Scholar] [CrossRef]
- Rugolotto, S.; Errico, G.; Beghini, R.; Ilic, S.; Richelli, C.; Padovani, E.M. Weaning of epoprostenol in a small infant receiving concomitant bosentan for severe pulmonary arterial hypertension secondary to bronchopulmonary dysplasia. Minerva Pediatr. 2006, 58, 491–494. [Google Scholar] [PubMed]
- Ferdman, D.J.; Rosenzweig, E.B.; Zuckerman, W.A.; Krishnan, U. Subcutaneous treprostinil for pulmonary hypertension in chronic lung disease of infancy. Pediatrics 2014, 134, e274–e278. [Google Scholar] [CrossRef] [PubMed]
- Adatia, I.; Haworth, S.G.; Wegner, M.; Barst, R.J.; Ivy, D.; Stenmark, K.R.; Karkowsky, A.; Rosenzweig, E.; Aguilar, C. Clinical trials in neonates and children: Report of the pulmonary hypertension academic research consortium pediatric advisory committee. Pulm. Circ. 2013, 3, 252–266. [Google Scholar] [CrossRef] [PubMed]
| All Patients n = 101 | Survived n = 68 | Died n = 33 | p | |
|---|---|---|---|---|
| Sex | 0.976 | |||
| Male | 61 (60.4%) | 41 (60.3%) | 20 (60.6%) | |
| Female | 40 (39.6%) | 27 (39.7%) | 13 (39.4%) | |
| Multiple Gestation | 25 (24.8%) | 18 (26.5%) | 7 (21.2%) | 0.566 |
| Type of Delivery | 0.803 | |||
| Vaginal | 21 (20.8%) | 14 (20.6%) | 7 (21.2%) | |
| C-section | 61 (60.4%) | 40 (58.8%) | 21 (63.6%) | |
| Unknown | 19 (18.8%) | 14 (20.6%) | 5 (15.2%) | |
| Born outside hospital | 79 (78.2%) | 49 (72.1%) | 28 (84.8%) | 0.157 |
| Gestational age (weeks) | 25 (IQR 24–27, range 23–32) | 26 (IQR 24–27, range 23–32) | 25 (IQR 24–26, range 23–30) | 0.428 |
| Birth weight (grams) n = 78 | 646 (IQR 540–790, range 270–1790) | 663 (IQR 550–840, range 378–1790) | 587 (IQR 485–728, range 270–1335) | 0.065 |
| Prenatal steroids n = 54 | 50/54 (92.6%) | 32/35 (91.4%) | 18/19 (94.7%) | 1.00 * |
| Oligohydramnios n = 62 | 6/62 (9.7%) | 4/43 (9.3%) | 2/19 (10.5%) | 1.00 * |
| Chorioamnionitis n = 65 | 9/65 (13.9%) | 5/43 (11.6%) | 4/22 (18.2%) | 0.473 * |
| Maternal hypertension n = 67 | 14/67 (20.9%) | 10/46 (21.7%) | 4/21 (19.1%) | 1.00 * |
| Surfactant n = 62 | 50/62 (80.7%) | 31/40 (77.5%) | 19/22 (86.4%) | 0.512 * |
| Total n = 101 | Survived n = 68 | Died n = 33 | p-Value | |
|---|---|---|---|---|
| Atrial communication | 43 (42.6%) | 25 (36.8%) | 18 (54.5%) | 0.090 |
| Small ventricular septal defect | 2 (2.00%) | 1 (1.5%) | 1 (3.0%) | 0.549 * |
| Pulmonary vein stenosis | 14 (13.9%) | 9 (13.2%) | 5 (55.2%) | 0.768 * |
| Patent ductus arteriosus | 75 (74.3%) | 50 (73.5%) | 25 (75.8%) | 0.810 |
| Ligated/occluded | 48 (64%) | 34 (50.0%) | 14 (42.4%) | 0.307 |
| Retinopathy of prematurity n = 91 | 73/91 (80.2%) | 45/61 (73.8%) | 28/30 (93.3%) | 0.028 |
| Intracranial pathology n = 96 | 53/96 (55.2%) | 30/64 (46.9%) | 23/32 (71.9%) | 0.020 |
| Necrotizing enterocolitis n = 97 | 31/97 (32.0%) | 19/65 (29.2%) | 12/32 (37.5%) | 0.412 |
| Sepsis n = 93 | 77/93 (82.8%) | 47/61 (77.1%) | 30/32 (93.8%) | 0.043 |
| Respiratory Support at Pulmonary Hypertension Diagnosis | Number of Patients (%) |
|---|---|
| Room air | 1 (1.0%) |
| Nasal cannula | 11 (10.9%) |
| Continuous positive airway pressure (CPAP) | 34 (33.7%) |
| Mechanical ventilator | 38 (37.6%) |
| High frequency oscillatory ventilator | 2 (2.0%) |
| Unknown | 15 (14.9%) |
| Respiratory Support at 36 weeks corrected | |
| Nasal Cannula | 3 (3.0%) |
| Continuous positive airway pressure (CPAP) | 36 (35.6%) |
| Mechanical ventilator | 37 (36.6%) |
| Unknown (but at least on supplemental oxygen) * | 25 (24.8%) |
| Respiratory support at discharge or transfer (N = 71) ** | |
| Room air | 6 (8.5%) |
| Nasal Cannula | 21 (29.6%) |
| CPAP or Bilevel positive airway pressure (BiPAP) | 21 (29.6%) |
| Mechanical ventilator | 3 (4.2%) |
| Tracheostomy | 20 (28.2%) |
| Medication (Method of Administration) | Patients Treated N(%) | Median Starting Age in Days (IQR, Range) | Typical Dosing | Died N (% of Total pts Receiving Med) | Patients on Medication at Discharge/Transfer N(% of Total Patients Receiving Med) | Stopped Medication for Side Effect N (%) |
|---|---|---|---|---|---|---|
| Sildenafil (PO/IV) | 99 (98.0%) | 146.5 (IQR 101–175, range 39–637) | Starting: 0.25–0.5 mg/kg/dose PO q8hrs (1/2 that dose if IV) Goal: 1 mg/kg/dose PO q8hrs(0.5 mg/kg/dose IV q8hrs) | 29 (29.3%) | 62 (62.3%) | 7 (7.1%) |
| Bosentan (PO) | 13 (12.9%) | 198.5 (IQR 172–168, range 122–406) | Starting: 1 mg/kg/dose q12hrs Goal: 2 mg/kg/dose q12hrs | 1 (7.7%) | 10 (76.9%) | 0 |
| Iloprost (inhaled) | 35 (34.7%) | 188 (IQR 148–261; range 16–426) | Starting: 1–2.5 mcg/dose q1–4 hrs (or continuous) Max: 2.5–7.5 mcg/dose | 16 (45.7%) | 1 (2.9%) | 6 (17.1%) |
| Epoprostenol (continuous intravenous) | 12 (11.9%) | 234 (IQR 197.5–315.5, range 97–533) | Starting: 2 ng/kg/min Max: 8–46 ng/kg/min | 6 (50%) | 4 (33.3%) | 0 |
| Treprostinil (continuous subcutaneous) | 9 (8.9%) | 196 (IQR 165–216, range 135–368) | Starting: 1.25 ng/kg/min Max: 14–74.5 ng/kg/min | 4 (44.4%) | 5 (55.6%) | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nees, S.N.; Rosenzweig, E.B.; Cohen, J.L.; Valencia Villeda, G.A.; Krishnan, U.S. Targeted Therapy for Pulmonary Hypertension in Premature Infants. Children 2020, 7, 97. https://doi.org/10.3390/children7080097
Nees SN, Rosenzweig EB, Cohen JL, Valencia Villeda GA, Krishnan US. Targeted Therapy for Pulmonary Hypertension in Premature Infants. Children. 2020; 7(8):97. https://doi.org/10.3390/children7080097
Chicago/Turabian StyleNees, Shannon N., Erika B. Rosenzweig, Jennifer L. Cohen, Gerson A. Valencia Villeda, and Usha S. Krishnan. 2020. "Targeted Therapy for Pulmonary Hypertension in Premature Infants" Children 7, no. 8: 97. https://doi.org/10.3390/children7080097
APA StyleNees, S. N., Rosenzweig, E. B., Cohen, J. L., Valencia Villeda, G. A., & Krishnan, U. S. (2020). Targeted Therapy for Pulmonary Hypertension in Premature Infants. Children, 7(8), 97. https://doi.org/10.3390/children7080097






