Hypoglossal Nerve Stimulator Placement for Pediatric Trisomy 21 Patients with Refractory Obstructive Sleep Apnea: A Case Series
Abstract
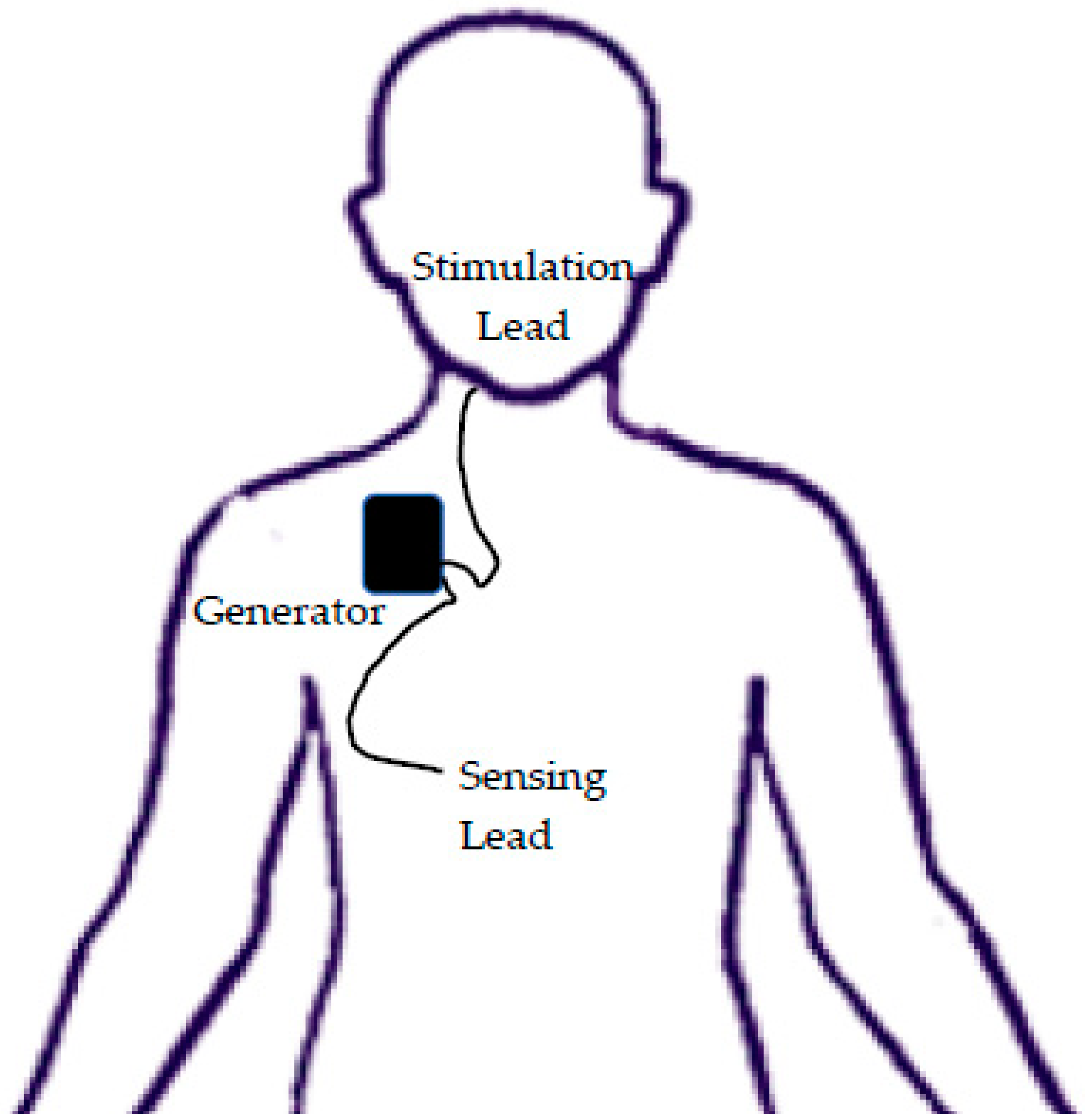
1. Introduction
2. Case Presentations
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ridore, S.; Debbarma, S.; Nazir, R.; Bennett, D.S.; Sedky, K. Obstructive Sleep Apnea in Individuals with Down Syndrome: A Meta-Analytic Literature Review. J. Sleep Sleep Disord. Res. 2017. [Google Scholar] [CrossRef]
- Farhood, Z.; Isley, J.W.; Ong, A.A.; Nguyen, S.A.; Camilon, T.J.; LaRosa, A.C.; White, D.R. Adenotonsillectomy outcomes in patients with Down syndrome and obstructive sleep apnea. Laryngoscope 2017, 127, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- De Miguel-Díez, J.R.; Villa-Asensi, J.; Alvarez-Sala, L. Prevalence of sleep-disordered breathing in children with Down syndrome: Polygraphic findings in 108 children. Sleep 2003, 26, 1006–1009. [Google Scholar] [CrossRef] [PubMed]
- Nation, J.; Brigger, M. The Efficacy of Adenotonsillectomy for Obstructive Sleep Apnea in Children with Down Syndrome: A Systematic Review. Otolaryngol. Head Neck Surg. 2017, 157, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.R.; Bennett, M.L.; Smith, P.L.; de Backer, W.; Hedner, J.; Boudewyns, A.; van de Heyning, P.; Ejnell, H.; Hochban, W.; Knaack, L.; et al. Therapeutic electrical stimulation of the hypoglossal nerve in obstructive sleep apnea. Arch. Otolaryngol. Head Neck Surg. 2001, 127, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Inspire Patient Manual. Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf13/P130008C.pdf (accessed on 4 January 2020).
- Eastwood, P.R.; Barnes, M.; Walsh, J.H.; Maddison, K.J.; Hee, G.; Schwartz, A.R.; Smith, P.L.; Malhotra, A.; Mcevoy, R.D.; Wheatley, J.R.; et al. Treating obstructive sleep apnea with hypoglossal nerve stimulation. Sleep 2011, 34, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
- Strollo, P.J.; Soose, R.J.; Maurer, J.T.; de Vries, N.; Cornelius, J.; Froymovich, O.; Hanson, R.D.; Padhya, T.A.; Steward, D.L.; Gillespie, M.B.; et al. Upper-airway stimulation for obstructive sleep apnea. N. Engl. J. Med. 2014, 370, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Strollo, P.J.; Gillespie, M.B.; Soose, R.J.; Maurer, J.T.; de Vries, N.; Cornelius, J.R.; Hanson, R.D.; Padhya, T.A.; Steward, D.L.; Woodson, B.T.; et al. Upper Airway Stimulation for Obstructive Sleep Apnea: Durability of the Treatment Effect at 18 Months. Sleep 2015, 38, 1593–1598. [Google Scholar] [CrossRef] [PubMed]
- Diercks, G.R.; Wentland, C.; Keamy, D.G.; Kinane, T.B.; Skotko, B.G.; de Guzman, V.; Grealish, E.; Dobrowski, J.M.; Soose, R.J.; Hartnicke, C.J. Hypoglossal Nerve Stimulation in Adolescents with Down Syndrome and Obstructive Sleep Apnea. JAMA Otolaryngol. Head Neck Surg. 2018, 144, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Gan, T.J.; Diemunsch, P.; Habib, A.S.; Kovac, A.; Kranke, P.; Meyer, T.A.; Watcha, M.; Chung, F.; Angus, S.; Apfel, C.C.; et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth. Analg. 2014, 118, 85–113. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, F.W.; Stricker, P.A.; Gurnaney, H.; Mcclung, H.; Meador, M.R.; Sussman, E.; Burgess, B.J.; Ciampa, B.; Mendelsohn, J.; Rehman, M.A.; et al. Bradycardia during induction of anesthesia with sevoflurane in children with Down syndrome. Anesth. Analg. 2010, 111, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- Caloway, C.L.; Diercks, G.R.; Keamy, D.G.; de Guzman, V.; Soose, R.J.; Raol, N.; Shott, S.R.; Ishman, S.L.; Hartnick, C.J. Update on hypoglossal nerve stimulation in children with down syndrome and obstructive sleep apnea. Laryngoscope 2019. [Google Scholar] [CrossRef] [PubMed]
| Patient | Demographics | ||||
| Age (year) | Weight (kg) | Trisomy 21 Body Mass Index Percentile a | Sex | American Society of Anesthesia Status | |
| 1 | 10 | 36.7 | 50–75% | F | 2 |
| 2 | 19 | 62.1 | 50–75% | F | 3 |
| 3 | 13 | 69 | 90–95% | M | 3 |
| Patient | Baseline PSG | ||||
| Total Apnea–Hypopnea Index (AHI) | Obstructive AHI | Central AHI | Total Pulse Oximetry Nadir | End Tidal Carbon Dioxide Maximum | |
| 1 | 35 | 35 | 0.14 | 78 | N/A |
| 2 | 36 | 34.3 | 1.19 | 83 | 53 |
| 3 | 19.7 b | 19.7 | 0 | 85 | 55 |
| Midazolam Premedication | Inhalation Induction | Sevoflurane for Induction | Nitrous Oxide for Induction | Paralytic Used for Intubation | Sevoflurane for Maintenance | Paralytic Used for Maintenance | |
| Patient 1 | 0.41 | yes | yes | no | no | yes | no |
| Patient 2 | 0 | yes | yes | yes | no | yes | no |
| Patient 3 | 0.22 | yes | yes | no | no | yes | no |
| Fentanyl mcg/kg | Hydromorphone mg/kg | Ketorolac mg/kg | Dexmedetomidine mcg/kg | Glycopyrrolate mg/kg | Decadron mg/kg | Zofran mg/kg | |
| Patient 1 | 1.82 | 0.004 | 0 | 0.65 | 0.0027 | 0 | 0.11 |
| Patient 2 | 3.22 | 0 | 0 | 0.84 | 0 | 0.13 | 0.06 |
| Patient 3 | 1.45 | 0 | 0.434 | 0.29 | 0.0029 | 0.09 | 0.08 |
| Initial Obstructive AHI | Post-HNS Obstructive AHI | Percentage Change | |
|---|---|---|---|
| Patient 1 | 35.0 | 2.8 | 92.0 |
| Patient 2 | 34.3 | 5.9 | 82.8 |
| Patient 3 | 19.7 a | 2.5 b | 87.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karlik, J.B.; Raol, N.; Gilbertson, L. Hypoglossal Nerve Stimulator Placement for Pediatric Trisomy 21 Patients with Refractory Obstructive Sleep Apnea: A Case Series. Children 2020, 7, 81. https://doi.org/10.3390/children7080081
Karlik JB, Raol N, Gilbertson L. Hypoglossal Nerve Stimulator Placement for Pediatric Trisomy 21 Patients with Refractory Obstructive Sleep Apnea: A Case Series. Children. 2020; 7(8):81. https://doi.org/10.3390/children7080081
Chicago/Turabian StyleKarlik, Joelle B., Nikhila Raol, and Laura Gilbertson. 2020. "Hypoglossal Nerve Stimulator Placement for Pediatric Trisomy 21 Patients with Refractory Obstructive Sleep Apnea: A Case Series" Children 7, no. 8: 81. https://doi.org/10.3390/children7080081
APA StyleKarlik, J. B., Raol, N., & Gilbertson, L. (2020). Hypoglossal Nerve Stimulator Placement for Pediatric Trisomy 21 Patients with Refractory Obstructive Sleep Apnea: A Case Series. Children, 7(8), 81. https://doi.org/10.3390/children7080081





