Neurological Complications and Associated Risk Factors in Children Affected with Chronic Kidney Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Recruitment
2.2. Data Collection
2.3. Statistical Analysis
3. Results
4. Discussion
4.1. Limitations
4.2. Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levey, A.S.; Eckardt, K.U.; Tsukamoto, Y.; Levin, A.; Coresh, J.; Rossert, J.; Zeeuw, D.D.; Hostetter, T.H.; Lameire, N.; Eknoyan, G. Definition and classification of chronic kidney disease: A position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2005, 67, 2089–2100. [Google Scholar] [CrossRef] [PubMed]
- Farag, Y.M.; Kari, J.A.; Singh, A.K. Chronic kidney disease in the Arab world: A call for action. Nephron Clin. Pract. 2012, 121, 120–123. [Google Scholar] [CrossRef]
- Al-Eisa, A.; Naseef, M.; Al-Hamad, N.; Pinto, R.; Al-Shimeri, N.; Tahmaz, M. Chronic renal failure in Kuwaiti children: An eight-year experience. Pediatr. Nephrol. 2005, 20, 1781–1785. [Google Scholar] [CrossRef] [PubMed]
- Hamed, R.M. The spectrum of chronic renal failure among Jordanian children. J. Nephrol. 2002, 15, 130–135. [Google Scholar] [PubMed]
- Al Harbi, N. Chronic renal failure in children in Asir region of Saudi Arabia. Saudi J. Kidney Dis. Transpl. 1997, 8, 294–297. [Google Scholar] [PubMed]
- Kari, J.A. Chronic renal failure in children in the Western area of Saudi Arabia. Saudi J. Kidney Dis. Transpl. 2006, 17, 19–24. [Google Scholar] [PubMed]
- Gheissari, A.; Hemmatzadeh, S.; Merrikhi, A.; Tehrani, S.F.; Madihi, Y. Chronic kidney disease in children: A report from a tertiary care center over 11 years. J. Nephropathol. 2012, 1, 177. [Google Scholar] [CrossRef] [PubMed]
- Harambat, J.; Van Stralen, K.J.; Kim, J.J.; Tizard, E.J. Epidemiology of chronic kidney disease in children. Pediatr. Nephrol. 2012, 27, 363–373. [Google Scholar] [CrossRef] [PubMed]
- VanDeVoorde, R.G.; Warady, B.A. Management of chronic kidney disease. In Pediatric Nephrology, 6th ed.; Updat Enlarg Ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1661–1692. [Google Scholar]
- Johnson, R.J.; Warady, B.A. Long-term neurocognitive outcomes of patients with end-stage renal disease during infancy. Pediatr. Nephrol. 2013, 28, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Gerson, A.C.; Butler, R.; Moxey-Mims, M.; Wentz, A.; Shinnar, S.; Lande, M.B.; Mendley, S.R.; Warady, B.A.; Furth, S.L.; Hooper, S.R. Neurocognitive outcomes in children with chronic kidney disease: Current findings and contemporary endeavors. Ment. Retard. Dev. Disabil. Res. Rev. 2006, 12, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Gipson, D.; Wetherington, C.; Duquette, P.; Hooper, S. The nervous system and chronic kidney disease in children. Pediatr. Nephrol. 2004, 19, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Warady, B.A.; Chadha, V. Chronic kidney disease in children: The global perspective. Pediatr. Nephrol. 2007, 22, 1999–2009. [Google Scholar] [CrossRef] [PubMed]
- Alfrey, A.C.; LeGendre, G.R.; Kaehny, W.D. The dialysis encephalopathy syndrome. Possible aluminum intoxication. N. Engl. J. Med. 1979, 294, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, S.P.; Bergstein, J.M.; Sherrard, D.J. Aluminum intoxication from aluminum-containing phosphate binders in children with azotemia not undergoing dialysis. N. Engl. J. Med. 1984, 310, 1079–1084. [Google Scholar] [CrossRef] [PubMed]
- Teschan, P.E.; Ginn, H.E.; Bourne, J.R.; Ward, J.W.; Hamel, B.; Nunnally, J.C.; Musso, M.; Vaughn, W.K. Quantitative in- dices of clinical uremia. Kidney Int. 1979, 15, 676–697. [Google Scholar] [CrossRef] [PubMed]
- Hulstijn-Dirkmaat, G.M.; Damhuis, I.H.; Jetten, M.L.; Koster, A.M.; Schröder, C.H. The cognitive development of pre-school children treated for chronic renal failure. Pediatr. Nephrol. 1995, 9, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Ledermann, S.E.; Scanes, M.E.; Fernando, O.N.; Duffy, P.G.; Madden, S.J.; Trompeter, R.S. Long-term outcome of peritoneal dialysis in infants. J. Pediatr. 2000, 136, 24–29. [Google Scholar] [CrossRef]
- Greenbaum, L.A.; Warady, B.A.; Furth, S.L. Current Advances in Chronic Kidney Disease in Children: Growth, Cardiovascular, and Neurocognitive Risk Factors. Semin. Nephrol. 2009, 29, 425–434. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | N | % |
|---|---|---|
Gender
| 276 149 | 64.9 35.1 |
Age
| 90 335 | 21.2 78.8 |
Stage of CKD
| 112 105 195 | 26.4 24.7 45.9 |
Renal diagnosis
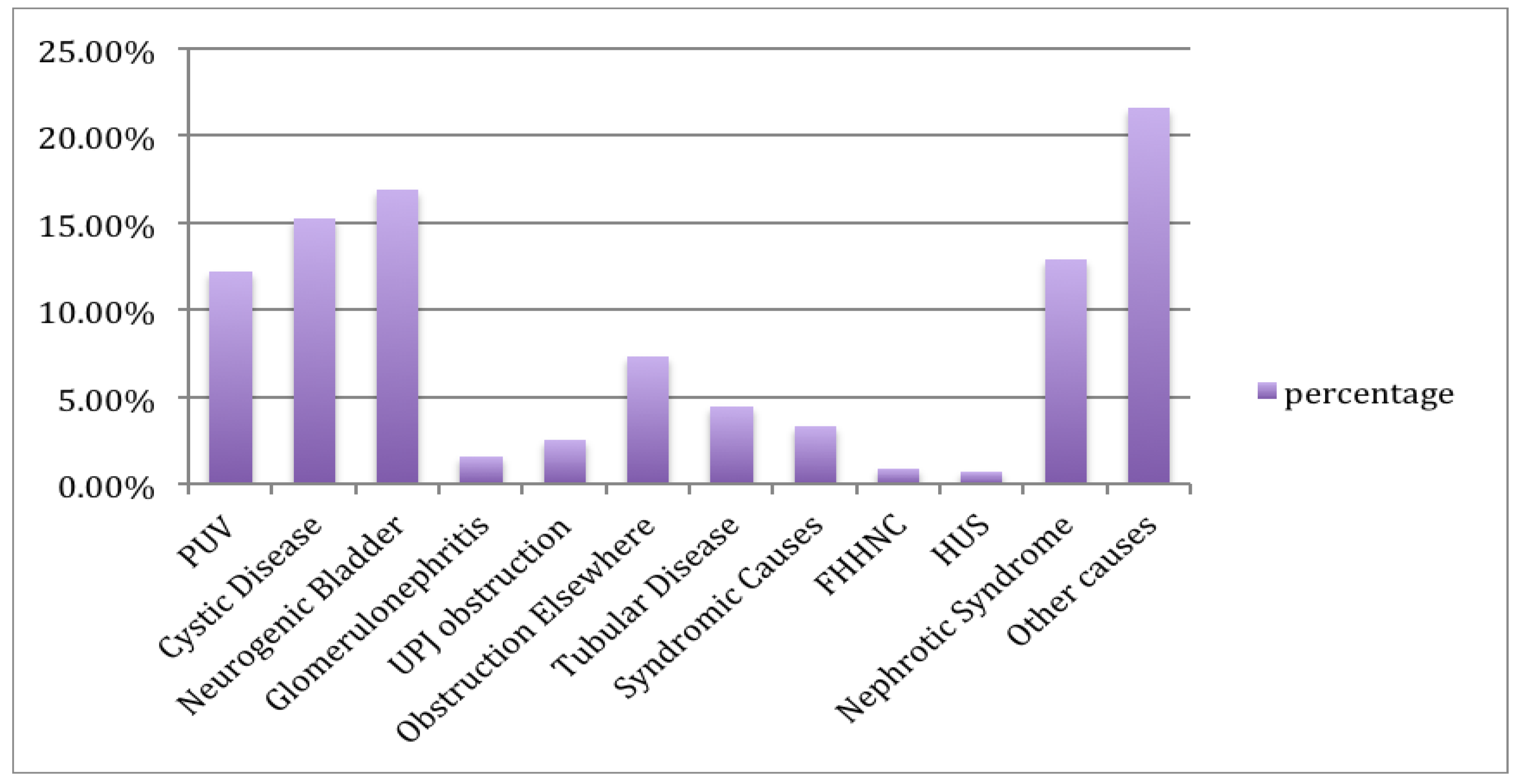
| 52 65 72 7 11 31 19 92 14 4 3 55 | 12.2 15.3 16.9 1.6 2.6 7.3 4.5 21.6 3.3 0.9 0.7 12.9 |
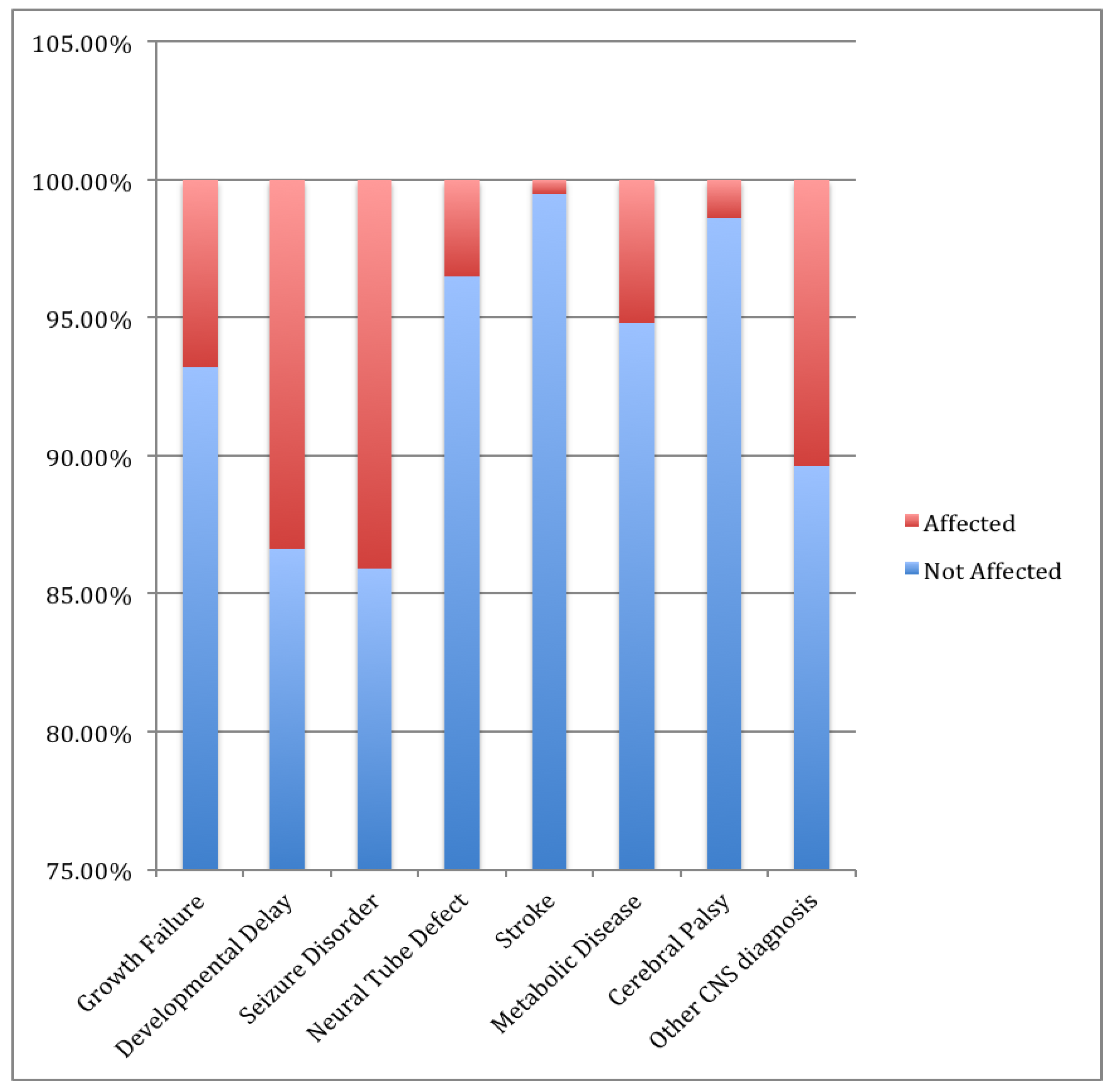
Growth Failure
| 29 396 | 6.8 93.2 |
Developmental Delay
| 57 368 | 13.4 86.6 |
Seizure Disorder
| 60 365 | 14.1 85.9 |
Neural Tube Defect
| 15 410 | 3.5 96.5 |
Stroke
| 2 423 | 0.5 99.5 |
Metabolic Disease
| 22 403 | 5.2 94.8 |
Cerebral Palsy
| 6 419 | 1.4 98.6 |
CNS diagnosis
| 381 5 19 7 6 6 1 | 89.6 1.2 4.5 1.6 1.4 1.4 0.2 |
Management
| 262 52 74 37 | 61.6 12.2 17.4 8.7 |
Antiepileptic Medication use
| 31 394 | 7.3 92.7 |
| Correlation Between | OR (Yes/No) | p-Values | |
|---|---|---|---|
| Variable 1 | Variable 2 | ||
| Age of diagnosis | CKD Stage | - | 0.453 |
| Growth failure | 0.762 | 0.591 | |
| Developmental delay | 0.569 | 0.156 | |
| Neurological disease | 1.359 | 0.240 | |
| Management | - | <0.001 | |
| Gender | CKD Stage | - | 0.946 |
| Growth failure | 0.875 | 0.737 | |
| Developmental delay | 1.092 | 0.086 | |
| Neurological disease | 0.749 | 0.188 | |
| Neurological disease | CKD stage | - | 0.015 |
| Management | - | 0.009 | |
| Use of antiepileptic medication | 0.175 | <0.001 | |
Renal diagnosis
| Neurological disease | 0.646 0.621 1.274 1.538 1.722 0.821 1.510 1.416 3.841 0.667 0.668 0.661 | 0.193 0.121 0.366 0.317 0.371 0.631 0.385 0.154 0.011 0.159 0.223 0.205 |
Each renal diagnosis
| Growth failure | 0.862 1.609 1.296 0.931 0.725 1.067 0.606 1.351 0.112 0.931 0.931 1.310 | 0.791 0.443 0.640 0.470 0.763 0.932 0.032 0.551 <0.001 0.587 0.683 0.666 |
Renal diagnosis
| Developmental delay | 2.765 1.623 0.952 0.928 1.564 1.049 0.818 0.667 0.189 0.865 0.865 1.071 | 0.084 0.282 0.896 0.945 0.670 0.931 0.756 0.206 0.001 0.429 0.494 0.873 |
Renal diagnosis
| Seizure disorder | 1.298 3.888 0.621 0.986 0.426 1.579 0.439 0.802 0.394 0.857 0.858 1.396 | 0.569 0.017 0.154 0.990 0.062 0.036 0.076 0.496 0.077 0.415 0.034 0.464 |
| Different Groups of Interest | Neurological Disease | |
|---|---|---|
| N | % | |
CKD stage
| 26 35 77 | 23.2 33.3 39.5 |
Management
| 75 21 35 9 | 28.6 40.4 47.3 24.3 |
Use of antiepileptic medications
| 22 118 | 71.0 29.9 |
Renal diagnosis
| 13 16 27 3 5 9 8 36 9 0 0 14 | 25.0 24.6 37.5 42.9 45.5 29.0 42.1 39.1 64.3 0.0 0.0 25.5 |
Gender
| 97 43 | 35.1 28.9 |
Age group
| 25 115 | 27.8 34.3 |
| Renal Diagnosis | Developmental Delay N (%) | Seizure Disorder N (%) | NTD N (%) | CNS Diagnosis N (%) |
|---|---|---|---|---|
| PUV (n = 52) Cystic disease (n = 65) Neurogenic Bladder (n = 72) Glomerulonephritis (n = 7) UPJ obstruction (n = 11) Obstruction otherwise (n = 31) Tubular disease (n = 19) Other causes (n = 92) Syndromic causes (n = 14) FHHNC (n = 4) HUS (n = 3) Nephrotic syndrome (n = 55) | 3 (5.8) 6 (9.2) 10 (13.9) 1 (14.3) 1 (9.1) 4 (12.9) 3 (15.8) 16 (17.4) 6 (42.9) 0 (0.0) 0 (0.0) 7 (12.7) | 6 (11.5) 3 (4.6) 14 (19.4) 1 (14.3) 3 (27.3) 3 (9.7) 5 (26.3) 15 (16.3) 4 (28.6) 0 (0.0) 0 (0.0) 6 (10.9) | 2 (3.8) 3 (4.6) 1 (1.4) 0 (0.0) 1 (9.1) 0 (0.0) 0 (0.0) 7 (7.6) 1 (7.1) 0 (0.0) 0 (0.0) 0 (0.0) | 3 (5.8) 2 (3.0) 13 (18.1) 2 (28.6) 2 (18.2) 4 (13.0) 1 (5.3) 10 (11.0) 4 (28.5) 0 (0.0) 0 (0.0) 3 (5.4) |
| Age Group | Conservative N (%) | Peritoneal Dialysis N (%) | Hemodialysis N (%) | Surgical N (%) |
|---|---|---|---|---|
| Younger than 2 years (n = 90) | 73 (81.1) | 7 (7.8) | 3 (3.3) | 7 (7.8) |
| Older than 2 years (n = 335) | 189 (56.4) | 45 (13.4) | 71 (21.2) | 30 (9.0) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Safdar, O.; Sindi, S.; Nazer, N.; Milyani, A.; Makki, A. Neurological Complications and Associated Risk Factors in Children Affected with Chronic Kidney Disease. Children 2020, 7, 59. https://doi.org/10.3390/children7060059
Safdar O, Sindi S, Nazer N, Milyani A, Makki A. Neurological Complications and Associated Risk Factors in Children Affected with Chronic Kidney Disease. Children. 2020; 7(6):59. https://doi.org/10.3390/children7060059
Chicago/Turabian StyleSafdar, Osama, Sulafa Sindi, Njood Nazer, Asmaa Milyani, and Abdulrahman Makki. 2020. "Neurological Complications and Associated Risk Factors in Children Affected with Chronic Kidney Disease" Children 7, no. 6: 59. https://doi.org/10.3390/children7060059
APA StyleSafdar, O., Sindi, S., Nazer, N., Milyani, A., & Makki, A. (2020). Neurological Complications and Associated Risk Factors in Children Affected with Chronic Kidney Disease. Children, 7(6), 59. https://doi.org/10.3390/children7060059




