Treatment of Complex Regional Pain Syndrome in Children and Adolescents: A Structured Literature Scoping Review
Abstract
1. Introduction
- Type 1, previously known as reflex sympathetic dystrophy (RSD), whose cause is not always known.
- Type 2, which results from nerve damage.
- Type 3, or not otherwise specified CRPS, which partly shares clinical and diagnostic aspects with the previous types [2].
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Selection Criteria
2.3. Data Extraction and Criteria Appraisal
2.4. Risk of Bias Assessment
3. Results
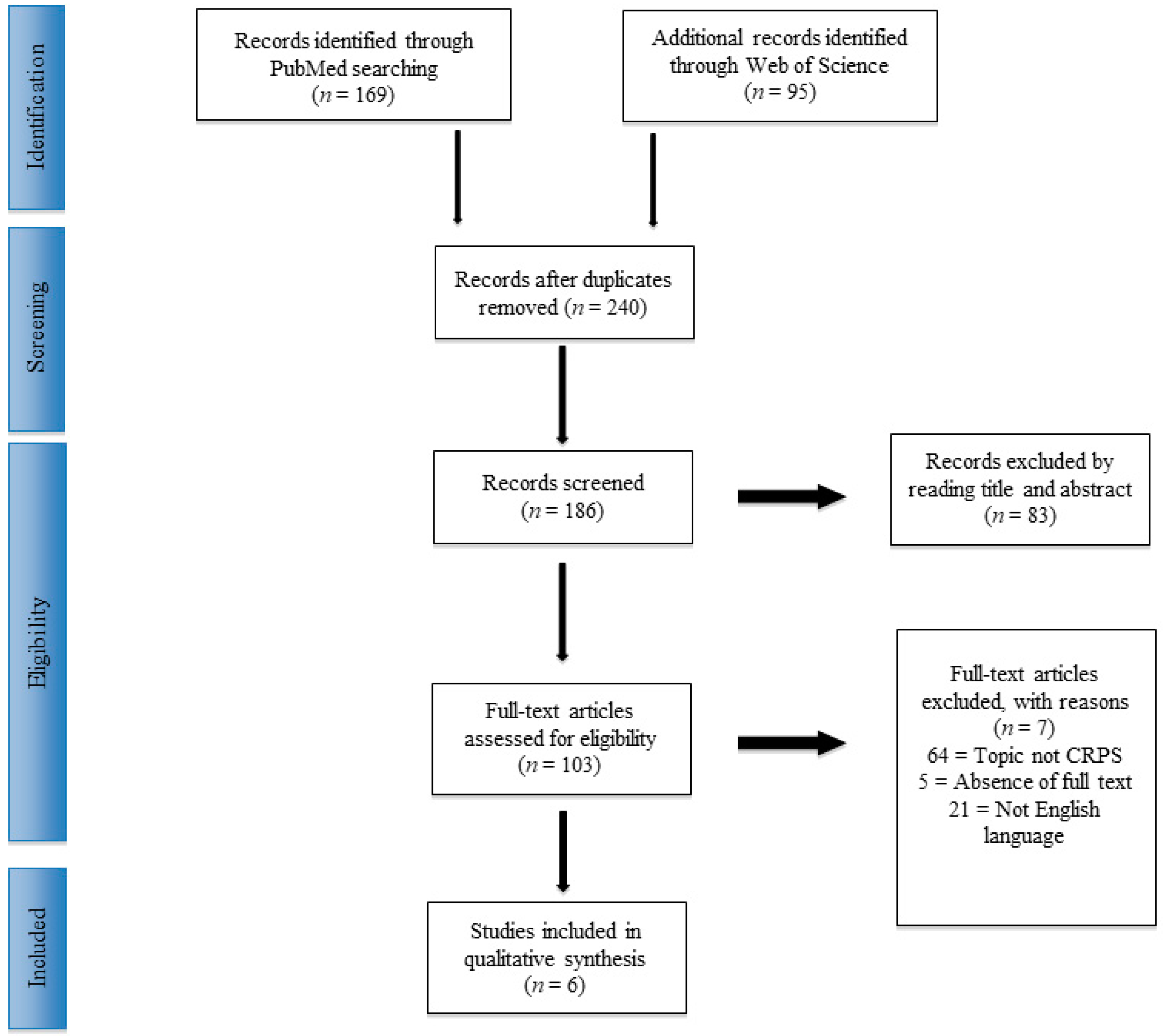
3.1. Study Selection
3.2. Physical Therapy and Cognitive Behavioral Treatment
3.3. Pharmacological Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
| Title | Authors | Year of Publication | New Castle-Ottawa Criteria | |||
|---|---|---|---|---|---|---|
| Selection | Comparability | Outcome | Final Evaluation | |||
| Complex Regional Pain Syndrome in Children: a Multidisciplinary Approach and Invasive Techniques for the Management of Nonresponders | Manuel J. Rodriguez-Lopez et al. | 2015 |  |  |  | Poor quality |
| A randomized controlled trial of amitriptyline versus gabapentin for complex regional pain syndrome type I and neuropathic pain in children | S.C. Brown et al., | 2016 |  |  |  | Good quality |
| Continuous Regional Anesthesia and Inpatient Rehabilitation for Pediatric Complex Regional Pain Syndrome | C. Donado et al. | 2017 |  |  |  | Good quality |
| Continuous Peripheral Nerve Blocks at Home for Treatment of Recurrent Complex Regional Pain Syndrome I in Children | C. Dadure et al. | 2005 |  |  |  | Fair quality |
| A Day-Hospital Approach to Treatment of Pediatric Complex Regional Pain Syndrome: Initial Functional Outcomes | D. E. Logan et al. | 2012 |  |  |  | Good quality |
| Short- and Long-term Outcomes. of Children with Complex Regional Pain Syndrome Type I Treated with Exercise Therapy | D. D. Sherry et al. | 1999 |  |  |  | Good quality |
| Spinal cord stimulation in adolescents with complex regional pain syndrome type I (CRPS-I) | G. L. Olsson et al. | 2008 |  |  |  | Fair quality |
| Treatment of Reflex Sympathetic Dystrophy in Children Using a Prostacyclin Analog | G. Petje et al. | 2005 |  |  |  | Fair quality |
| Treatment of Reflex Dystrophy in Children Using a Prostacyclin Analog | G. Petje et al, | 2003 |  |  |  | Good quality |
| Short- and long- term results of an inpatient programme to manage complex regional pain Syndrome in children and adolescents | G. Cucchiaro et al, | 2017 |  |  |  | Fair quality |
| Subanesthetic Ketamina infusions for the treatment of children and adolescents with chronic pain: a longitudinal study | Kathy A. Sheehy et al | 2015 |  |  |  | Fair quality |
| Complex regional pain syndrome in children and adolescents | Kachko et al. | 2008 |  |  |  | Fair quality |
| Pediatric complex regional pain syndrome | Low et al. | 2007 |  |  |  | Fair quality |
| Physical therapy and cognitive behavioural treatment | Lee et al. | 2002 |  |  |  | Good quality |
| Reflex sympathetic dystrophy in children: treatment with trans-cutaneous electric nerve stimulation | Kesler et al. | 1988 |  |  |  | Fair quality |
References
- Chang, C.; McDonnell, P.; Gershwin, M.E. Complex regional pain syndrome—False hopes and miscommunications. Autoimmun. Rev. 2019, 18, 270–278. [Google Scholar] [CrossRef]
- Lascombes, P.; Mamie, C. Complex regional pain syndrome type I in children: What is new? Orthop. Traumatol. Surg. Res. 2017, 103, S135–S142. [Google Scholar] [CrossRef]
- Barrett, M.J.; Barnett, P.L.J. Complex regional pain type 1. Pediat. Emerg. Care 2016, 32, 185–189. [Google Scholar] [CrossRef]
- Rabin, J.; Brown, M.; Alexander, S. Update in the Treatment of Chronic Pain within Pediatric Patients. Curr. Probl. Pediatr. Adolesc. Health Care 2017, 47, 167–172. [Google Scholar] [CrossRef]
- Weissmann, R.; Uziel, Y. Pediatric complex regional pain syndrome: A review. Pediatr. Rheumatol. Online J. 2016, 14, 29. [Google Scholar] [CrossRef]
- Williams, G.; Howard, R. The Pharmacological Management of Complex Regional Pain Syndrome in Pediatric Patients. Paediatr. Drugs 2016, 18, 243–250. [Google Scholar] [CrossRef]
- Xu, J.; Yang, J.; Lin, P.; Rosenquist, E.; Cheng, J. Intravenous Therapies for Complex Regional Pain Syndrome: A Systematic Review. Anesth. Analg. 2016, 122, 843–856. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef]
- Sherry, D.D.; Wallace, C.A.; Kelley, C.; Kidder, M.; Sapp, L. Short- and long-term outcomes of children with complex regional pain syndrome type I treated with exercise therapy. Clin. J. Pain 1999, 15, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Scharff, L.; Sethna, N.F.; McCarthy, C.F.; Scott-Sutherland, J.; Shea, A.M.; Sullivan, P.; Meier, P.; Zurakowski, D.; Masek, B.J.; et al. Physical therapy and cognitive-behavioral treatment for complex regional pain syndromes. J. Pediatr. 2002, 141, 135–140. [Google Scholar] [CrossRef]
- Logan, D.E.; Carpino, E.A.; Chiang, G.; Condon, M.; Firn, E.; Gaughan, V.J. A day-hospital approach to treatment of pediatric complex regional pain syndrome: Initial functional outcomes. Clin. J. Pain 2012, 28, 766–774. [Google Scholar] [CrossRef]
- Petje, G.; Radler, C.; Aigner, N.; Walik, N.; Kriegs, A.G.; Grill, F. Treatment of reflex sympathetic dystrophy in children using a prostacyclin analog: Preliminary results. Clin. Orthop. Relat. Res. 2005, 433, 178–182. [Google Scholar] [CrossRef]
- Brown, S.; Johnston, B.; Amaria, K.; Watkins, J.; Campbell, F.; Pehora, C.; McGrath, P. A randomized controlled trial of amitriptyline versus gabapentin for complex regional pain syndrome type I and neuropathic pain in children. Scand. J. Pain 2016, 13, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Donado, C.; Lobo, K.; Velarde-Álvarez, M.F.; Kim, J.; Kenney, A.; Logan, D.; Berde, C.B. Continuous Regional Anesthesia and Inpatient Rehabilitation for Pediatric Complex Regional Pain Syndrome. Reg. Anesthesia Pain Med. 2017, 42 (Suppl. 4), 527–534. [Google Scholar] [CrossRef]
- Berde, C.B.; Lebel, A. Complex regional pain syndromes in children and adolescents. Anesthesiology 2005, 102, 252–255. [Google Scholar] [CrossRef]
- Dickson, S.K. Including Parents in the Treatment of Pediatric Complex Regional Pain Syndrome. Pediatr. Nurs. 2017, 43, 16–21. [Google Scholar]
- Frot, M.; Faillenot, I.; Mauguière, F. Processing of nociceptive input from posterior to anterior insula in humans. Hum. Brain Mapp. 2014, 35, 5486–5499. [Google Scholar] [CrossRef]
- Lebel, A.; Becerra, L.; Wallin, D.; Moulton, E.A.; Morris, S.; Pendse, G.; Jasciewicz, J.; Stein, M.; Aiello-Lammens, M.; Grant, E.; et al. fMRI reveals distinct CNS processing during symptomatic and recovered complex regional pain syndrome in children. Brain 2008, 131, 1854–1879. [Google Scholar] [CrossRef]
- Linnman, C.; Becerra, L.; Lebel, A.; Berde, C.; Grant, P.E.; Borsook, D. Transient and persistent pain induced connectivity alterations in pediatric complex regional pain syndrome. PLoS ONE 2013, 8, e57205. [Google Scholar] [CrossRef]
- Diers, M.; Yilmaz, P.; Rance, M.; Thieme, K.; Gracely, R.H.; Rolko, C.; Schley, M.T.; Kiessling, U.; Wang, H.; Flor, H. Treatment-related changes in brain activation in patients with fibromyalgia syndrome. Exp. Brain Res. 2012, 218, 619–628. [Google Scholar] [CrossRef]
- Kregel, J.; Coppieters, I.; DePauw, R.; Malfliet, A.; Danneels, L.; Nijs, J.; Cagnie, B.; Meeus, M. Does Conservative Treatment Change the Brain in Patients with Chronic Musculoskeletal Pain? A Systematic Review. Pain Physician 2017, 20, 139–154. [Google Scholar]
- Dworkin, R.H.; O’Connor, A.B.; Audette, J.; Baron, R.; Gourlay, G.K.; Haanpää, M.L.; Kent, J.L.; Kranem, E.J.; Lebel, A.A.; Levy, R.M.; et al. Recommendations for the pharmacological management of neuropathic pain: An overview and literature update. Mayo Clin. Proc. 2010, 85, 3–14. [Google Scholar] [CrossRef]
- Kremer, M.; Salvat, E.; Muller, A.; Yalcin, I.; Barrot, M. Antidepressants and gabapentinoids in neuropathic pain: Mechanistic insights. Neuroscience 2016, 338, 183–206. [Google Scholar] [CrossRef]
| Author | Subjects | Dignosis Criteria | Assessment | Treatment | Results | Limits |
|---|---|---|---|---|---|---|
| Brown et al. 2016 | Amitriptyine Group: n = 14; Garbapentin Group: n = 15. | Modified International Association for the Study of Pain (IASP) clinical and research criteria. | Coloured Analogue Scale (CAS) Pain 6-weeks post-trial start; Sleep disability as measured on an internally developed 5-point Likert scale; Adverse events. | Amitriptylin 10 mg (at bedtime). Gabapentin at 900 mg/d (300 mg three times per day. | CAS p = 0.77. Sleep p = 0.26. Adverse events p = 0.75. | Small sample size. No randomization. No placebo group. No medium- and long-term follow-up. |
| Petje et al. 2003 | n = 7 | Skin examination; burning, dysesthesia, paresthesia and hypalgesia to cold. Skin cyanosis, mottling, hyperhidrosis, edema and coldness of the extremity and muscles, joint affliction duo to muscle hypotrophy or atrophy and range of motion (ROM) of the joints in the involved extremity. Bonica classification. | Visual analog scale VAS (0–10 points). | Intermittent intravenous infusion of Iloprost at 2 ng/kg/minute for approximately 6 h per day on 3 consecutive days + physiotherapy and psychologic. | VAS = p < 0.05. All patients had headache at the first day of infusion. 3 patients had flushing. 2 patients had vomiting. 86% of the sample had a decrease in systolic blood pressure with an average of 7 mm Hg (5–15 mm Hg) in the first 30 min after administration of Iloprost. | No control group Retrospective series. Small number of cases. |
| Donado et al. 2017 | n = 102 | Modified International Association for the Study of Pain (IASP) clinical and research criteria. | Preadmission, discharge and 4-month follow-up Pain Score (PS), Pain-Related Functional Disability (PFD) and sleep disturbances (SD) | Continuous regional anesthesia (epidural or peripheral catheter). | PS preadmission median = 7.0; IQR, 5.8–8.2. PS discharge = 3.1; IQR, 1.5–5.4; (p < 0.0001). PS 4-month follow-up = 4.3; IQR, 2.0–6.0; (p < 0.0001). PFD at admission had a moderate positive correlation with PDF at discharge (r, 0.5; p < 0.0001) SD = Yes 48.04%. | Retrospective design. Completed a full course of cognitive behavioral therapy |
| Logan et al., 2012 | n = 56 | Modified IASP clinical and research criteria. | At admission and at discharge. Numeric rating Scale (NRS). Functional Disability Inventory (FDI). Lower extremity functional scale: (LEFS). Canadian Occupational Performance Measure: (COPM). Multidimensional Anxiety Scale for Children (MASC). Children’s Depression Inventory (CDI). Bruininks-Oseretsky Test of Motor Proficiency, 2nd edition (BOT-2). | Patients participated in daily physical therapy, occupational therapy and psychological treatment and received nursing and medical care as necessary. | NRS = p < 0.001 FDI = p < 0.001 LEFS = p< 0.001 COPM = < 0.001 MASC = p< 0.001 CDI p = 0.003 All BOT-2 domains = p < 0.001. Patients underwent any procedures (e.g., nerve blocks) during or immediately prior to participation in the rehabilitation program. | No randomization. No control group. No isolated treatment effects. Uncontrolled prior treatment history in analyses. |
| Sherry et al., 1999 | n = 103 | IASP clinical and research criteria. | Visual analog scale (VAS) and Brief Symptom Inventory (BSI) at admission and remission. | An intensive exercise program (most received a daily program of 4 h of aerobic, functionally directed exercises, 1–2 h of hydrotherapy and desensitization). No medications or modalities were used. All had a screening psychological evaluation. | VAS = p = 0.021. BSI depression p = 0.037. BSI paranoid ideation p = 0.048. 1 child (2%) was dysfunctional with CRPS pain, and 5 (10%) had persistent mild pain but were fully functional. Median time between remission of the first episode of CRPS and the start of the second episode = 2 months (range = 2 weeks to 4 years). Predictors of recurrent episodes: previous suicide attempts (p = 0.026,), history of an eating disorder (p = 0.028). | No long-term follow-up. No control group. |
| Lee et al., 2002 | n = 28. Group A = PT once per week for 6 weeks Group B = PT 3 times per week for 6 weeks. | Wilder et al. criteria. | Pretreatment, at completion of the treatment program and (3) long-term follow-up at 6 to 12 months. Visual analog scale (VAS), Standardized gait impairment score (SGIS), Child Health Questionnaire (CHQ-CF87), Child Depression Inventory (CDI), Revised Children’s Manifest Anxiety Scale (CMAS), Compliance. | Individualized physical therapy. Individualized 6 weekly sessions cognitive behavioral therapy. Standard educational program. | At the short-term follow-up, both groups showed improvement in all five outcome measures related to pain and physical functioning (p < 0.001 for all measures with a change in median values). There were no between-group differences in any of these measures at baseline or at either follow-up assessment. 79% compliance good. | Small sample. Not standardized after the 6-week protocol. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vescio, A.; Testa, G.; Culmone, A.; Sapienza, M.; Valenti, F.; Di Maria, F.; Pavone, V. Treatment of Complex Regional Pain Syndrome in Children and Adolescents: A Structured Literature Scoping Review. Children 2020, 7, 245. https://doi.org/10.3390/children7110245
Vescio A, Testa G, Culmone A, Sapienza M, Valenti F, Di Maria F, Pavone V. Treatment of Complex Regional Pain Syndrome in Children and Adolescents: A Structured Literature Scoping Review. Children. 2020; 7(11):245. https://doi.org/10.3390/children7110245
Chicago/Turabian StyleVescio, Andrea, Gianluca Testa, Annalisa Culmone, Marco Sapienza, Fabiana Valenti, Fabrizio Di Maria, and Vito Pavone. 2020. "Treatment of Complex Regional Pain Syndrome in Children and Adolescents: A Structured Literature Scoping Review" Children 7, no. 11: 245. https://doi.org/10.3390/children7110245
APA StyleVescio, A., Testa, G., Culmone, A., Sapienza, M., Valenti, F., Di Maria, F., & Pavone, V. (2020). Treatment of Complex Regional Pain Syndrome in Children and Adolescents: A Structured Literature Scoping Review. Children, 7(11), 245. https://doi.org/10.3390/children7110245







