Examining the Relationship between Cost and Quality of Care in the Neonatal Intensive Care Unit and Beyond
Abstract
1. Introduction
2. Health Care Spending
3. Stewardship Campaigns: Reducing Waste
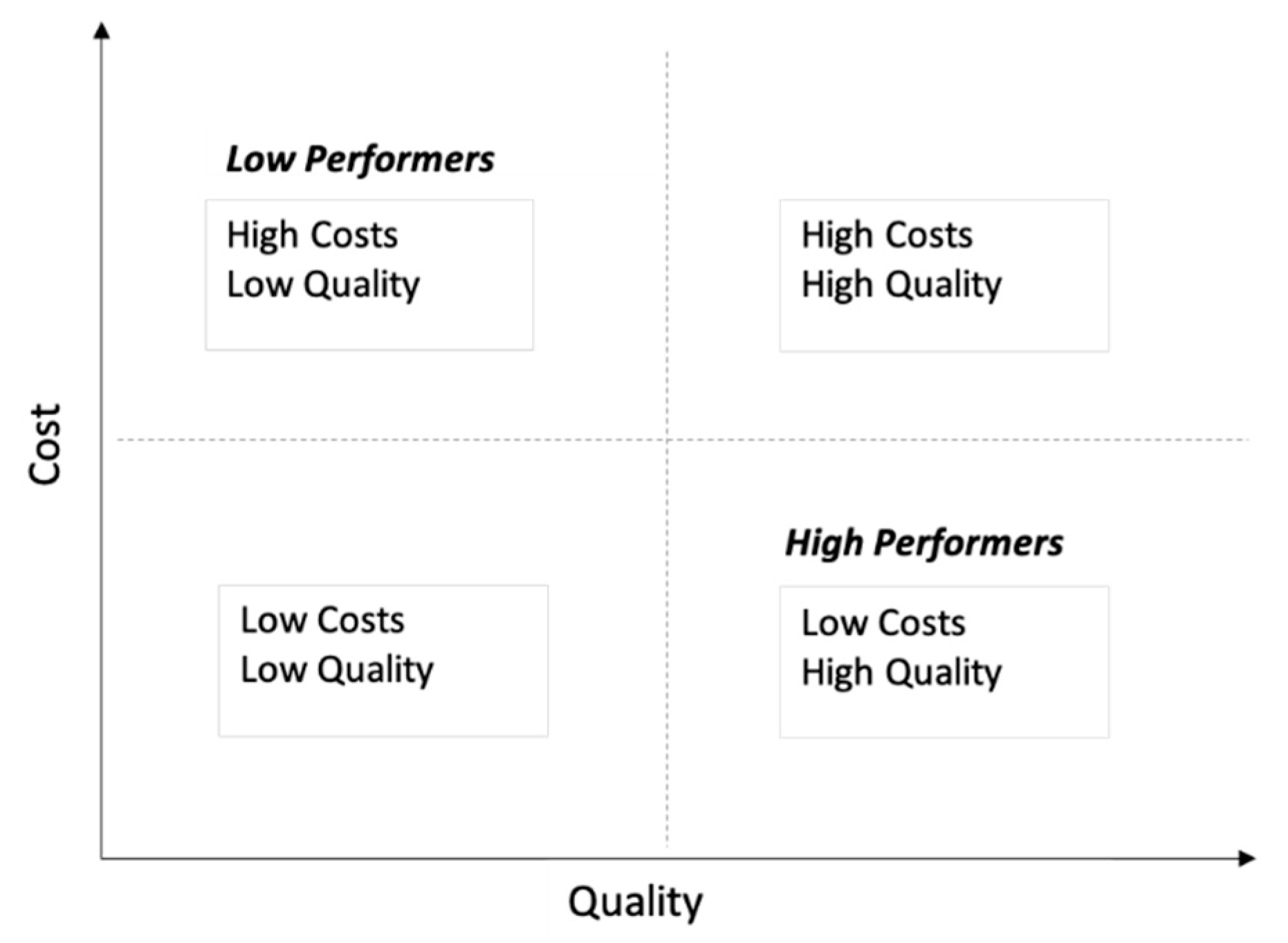
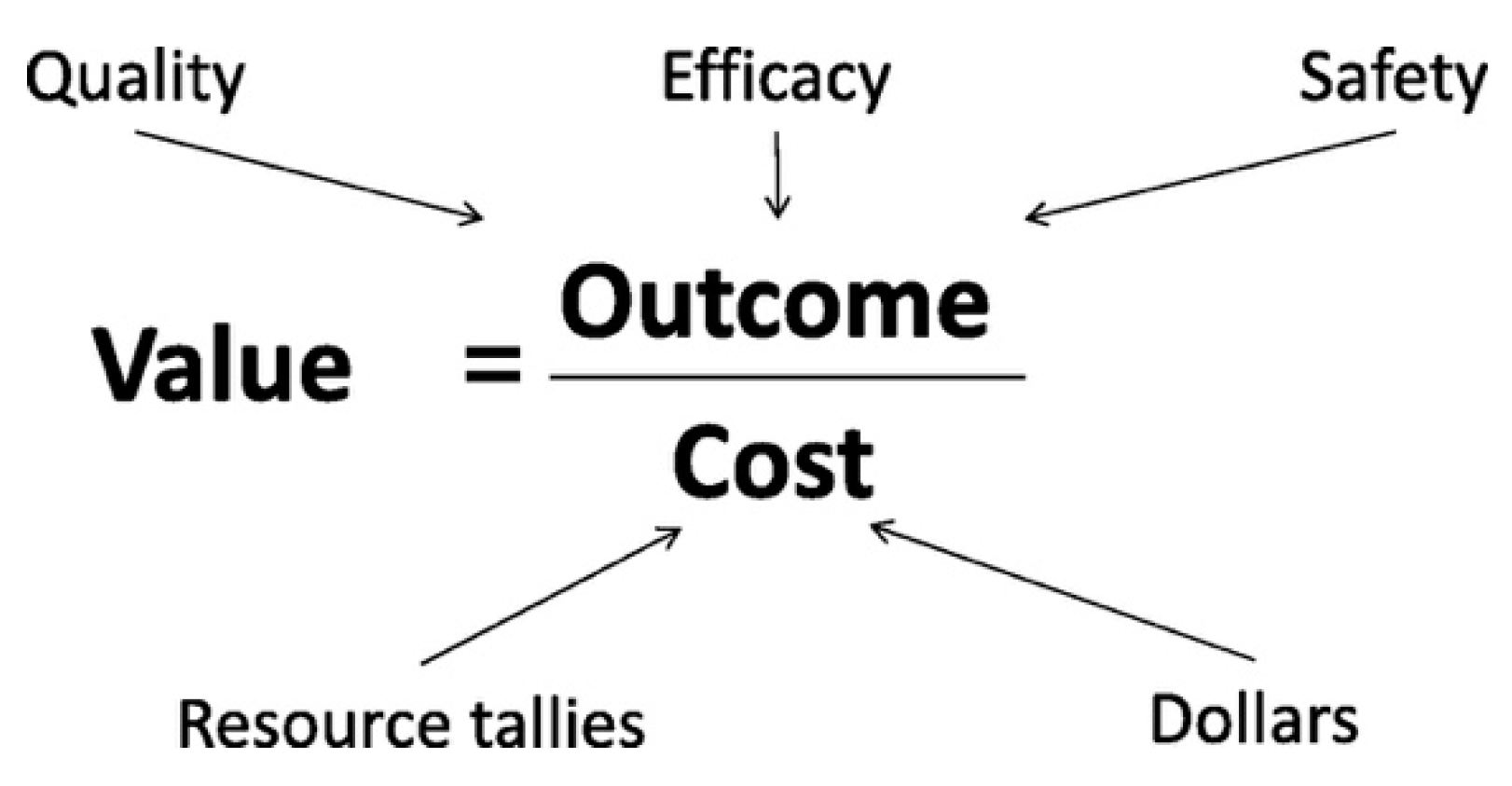
4. Defining Cost and Quality of Care
5. Cost of Pediatric Care
6. Cost of Neonatal Care
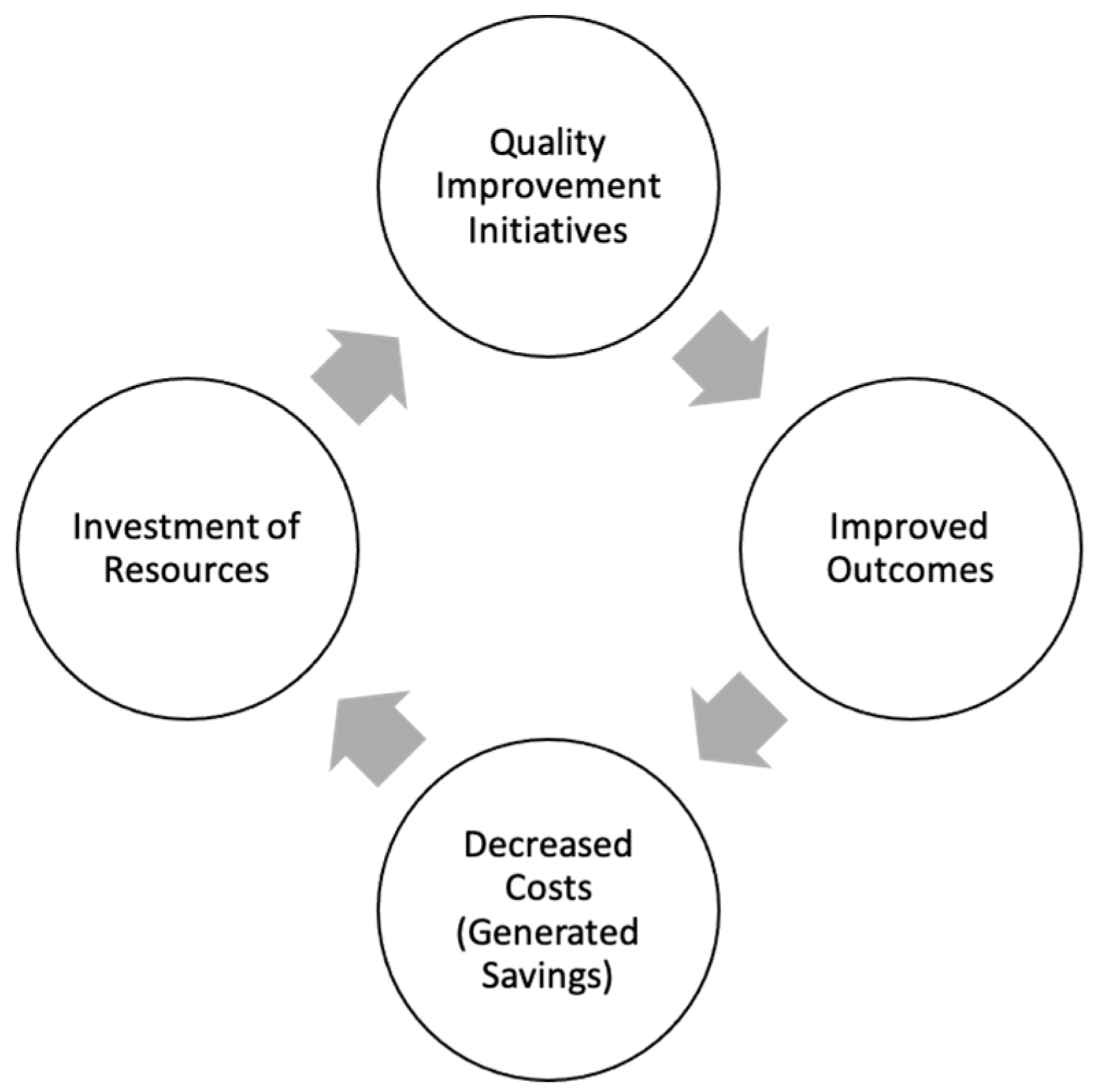
7. How Do We Evaluate Value in Neonatal Care?
8. Next Steps
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Berwick, D.M.; Nolan, T.W.; Whittington, J. The Triple Aim: Care, Health, And Cost. Health Aff. 2008, 27, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Bui, A.L.; Dieleman, J.L.; Hamavid, H.; Birger, M.; Chapin, A.; Duber, H.C.; Horst, C.; Reynolds, A.; Squires, E.; Chung, P.J.; et al. Spending on Children’s Personal Health Care in the United States, 1996–2013. JAMA Pediatr. 2017, 171, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Schneider, E.C.; Sarnak, D.O.; Squires, D.; Shah, A.; Doty, M.M. Mirror, Mirror 2017: International Comparison Reflects Flaws and Opportunities for Better, U.S. Health Care. Commonw. Fund 2017. [Google Scholar] [CrossRef]
- Fairbrother, G.; Guttmann, A.; Klein, J.D.; Simpson, L.A.; Thomas, P.; Kempe, A. Higher Cost, but Poorer Outcomes: The US Health Disadvantage and Implications for Pediatrics. Pediatrics 2015, 135, 961–964. [Google Scholar] [CrossRef]
- Choosing Wisley. Clinician Lists. Available online: www.choosingwisely.org/clinician-lists/ (accessed on 18 November 2020).
- Cassel, C.K.; Guest, J.A. Choosing wisely: Helping physicians and patients make smart decisions about their care. JAMA 2012, 307, 1801–1802. [Google Scholar] [CrossRef] [PubMed]
- Eaton, K.P.; Levy, K.; Soong, C.; Pahwa, A.K.; Petrilli, C.; Ziemba, J.B.; Cho, H.J.; Alban, R.; Blanck, J.F.; Parsons, A.S. Evidence-Based Guidelines to Eliminate Repetitive Laboratory Testing. JAMA Intern. Med. 2017, 177, 1833–1839. [Google Scholar] [CrossRef]
- Silvestri, M.T.; Bongiovanni, T.R.; Glover, J.G.; Gross, C.P. Impact of price display on provider ordering: A systematic review. J. Hosp. Med. 2015, 11, 65–76. [Google Scholar] [CrossRef]
- Heekin, A.M.; Kontor, J.; Sax, H.C.; Keller, M.S.; Wellington, A.; Weingarten, S. Choosing Wisely clinical decision support adherence and associated inpatient outcomes. Am. J. Manag. Care 2018, 24, 361–366. [Google Scholar]
- Ho, T.; Dukhovny, D.; Zupancic, J.A.; Goldmann, D.A.; Horbar, J.D.; Pursley, D.M. Choosing Wisely in Newborn Medicine: Five Opportunities to Increase Value. Pediatrics 2015, 136, e482–e489. [Google Scholar] [CrossRef]
- Ho, T.; Buus-Frank, M.E.; Edwards, E.M.; Morrow, K.A.; Ferrelli, K.; Srinivasan, A.; Pollock, D.A.; Dukhovny, D.; Zupancic, J.A.; Pursley, D.M.; et al. Adherence of Newborn-Specific Antibiotic Stewardship Programs to CDC Recommendations. Pediatrics 2018, 142, e20174322. [Google Scholar] [CrossRef]
- Dukhovny, D.; Buus-Frank, M.E.; Edwards, E.M.; Ho, T.; Morrow, K.A.; Srinivasan, A.; Pollock, D.A.; Zupancic, J.A.; Pursley, D.M.; Goldmann, D.; et al. A Collaborative Multicenter QI Initiative to Improve Antibiotic Stewardship in Newborns. Pediatrics 2019, 144, e20190589. [Google Scholar] [CrossRef] [PubMed]
- Pursley, D.M.; Zupancic, J.A.F. Using Neonatal Intensive Care Units More Wisely for At-Risk Newborns and Their Families. JAMA Netw. Open 2020, 3, e205693. [Google Scholar] [CrossRef] [PubMed]
- Goodman, D.C.; Little, G.A.; Harrison, W.N.; Moen, A.; Mowitz, M.E.; Ganduglia-Cazaban, C. The Dartmouth Atlas of Neonatal Intensive Care: A Report of the Dartmouth Atlas Project; The Dartmouth Institute of Health Policy & Clinical Practice, Geisel School of Medicine at Dartmouth: Lebanon, NH, USA, 2019. [Google Scholar]
- Hussey, P.S.; Wertheimer, S.; Mehrotra, A. The Association Between Health Care Quality and Cost. Ann. Intern. Med. 2013, 158, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Nuti, S.V.; Li, S.-X.; Xu, X.; Ott, L.S.; Lagu, T.; Desai, N.R.; Murugiah, K.; Duan, M.; Martin, J.; Kim, N.; et al. Association of in-hospital resource utilization with post-acute spending in Medicare beneficiaries hospitalized for acute myocardial infarction: A cross-sectional study. BMC Health Serv. Res. 2019, 19, 190. [Google Scholar] [CrossRef]
- Häkkinen, U.; Rosenqvist, G.; Peltola, M.; Kapiainen, S.; Rättö, H.; Cots, F.; Geissler, A.; Or, Z.; Serdén, L.; Sund, R. Quality, cost, and their trade-off in treating AMI and stroke patients in European hospitals. Health Policy 2014, 117, 15–27. [Google Scholar] [CrossRef]
- Kang, H.-C.; Hong, J.-S. Association between costs and quality of acute myocardial infarction care hospitals under the Korea National Health Insurance program. Medicine 2017, 96, e7622. [Google Scholar] [CrossRef]
- Gupta, P.; Rettiganti, M. Relationship of Hospital Costs With Mortality in Pediatric Critical Care. Pediatr. Crit. Care Med. 2017, 18, 541–549. [Google Scholar] [CrossRef]
- Pasquali, S.K.; Jacobs, J.P.; Bove, E.L.; Gaynor, J.W.; He, X.; Gaies, M.G.; Hirsch-Romano, J.C.; Mayer, J.E.; Peterson, E.D.; Pinto, N.M.; et al. Quality-Cost Relationship in Congenital Heart Surgery. Ann. Thorac. Surg. 2015, 100, 1416–1421. [Google Scholar] [CrossRef]
- Romley, J.A.; Chen, A.Y.; Goldman, D.P.; Williams, R. Hospital Costs and Inpatient Mortality among Children Undergoing Surgery for Congenital Heart Disease. Health Serv. Res. 2014, 49, 588–608. [Google Scholar] [CrossRef]
- Lion, K.C.; Wright, D.R.; Spencer, S.; Zhou, C.; Del Beccaro, M.; Mangione-Smith, R. Standardized Clinical Pathways for Hospitalized Children and Outcomes. Pediatrics 2016, 137, e20151202. [Google Scholar] [CrossRef]
- Phibbs, C.S.; Schmitt, S.K.; Cooper, M.; Gould, J.B.; Lee, H.C.; Profit, J.; Lorch, S.A. Birth Hospitalization Costs and Days of Care for Mothers and Neonates in California, 2009–2011. J. Pediatr. 2019, 204, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.B.; Green, N.S.; Steiner, C.A.; Meikle, S.; Howse, J.L.; Poschman, K.; Dias, T.; Potetz, L.; Davidoff, M.J.; Damus, K.; et al. Cost of Hospitalization for Preterm and Low Birth Weight Infants in the United States. Pediatrics 2007, 120, e1–e9. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.J.; Patel, A.L.; Jegier, B.J.; Engstrom, J.L.; Meier, P.P. Cost of Morbidities in Very Low Birth Weight Infants. J. Pediatr. 2013, 162, 243–249.e1. [Google Scholar] [CrossRef] [PubMed]
- Massaro, A.N.; Murthy, K.; Zaniletti, I.; Cook, N.; Di Geronimo, R.; Dizon, M.L.; Hamrick, S.E.; McKay, V.J.; Natarajan, G.; Rao, R.; et al. Intercenter Cost Variation for Perinatal Hypoxic-Ischemic Encephalopathy in the Era of Therapeutic Hypothermia. J. Pediatr. 2016, 173, 76–83.e1. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Liu, J.; Profit, J.; Hintz, S.R.; Gould, J.B. Survival Without Major Morbidity Among Very Low Birth Weight Infants in California. Pediatrics 2020, 146, e20193865. [Google Scholar] [CrossRef]
- Horbar, J.D.; Edwards, E.M.; Greenberg, L.T.; Morrow, K.A.; Soll, R.F.; Buus-Frank, M.E.; Buzas, J.S. Variation in Performance of Neonatal Intensive Care Units in the United States. JAMA Pediatr. 2017, 171, e164396. [Google Scholar] [CrossRef]
- Schulman, J.; Dimand, R.J.; Lee, H.C.; Duenas, G.V.; Bennett, M.V.; Gould, J.B. Neonatal Intensive Care Unit Antibiotic Use. Pediatrics 2015, 135, 826–833. [Google Scholar] [CrossRef]
- Effect of corticosteroids for fetal maturation on perinatal outcomes: NIH Consensus Development Panel on the Effect of Corticosteroids for Fetal Maturation on Perinatal Outcomes. JAMA 1995, 273, 413–418. [CrossRef]
- Gyamfi-Bannerman, C.; Zupancic, J.A.F.; Sandoval, G.; Grobman, W.A.; Blackwell, S.C.; Tita, A.T.N.; Reddy, U.M.; Jain, L.; Saade, G.R.; Rouse, D.J.; et al. Cost-effectiveness of Antenatal Corticosteroid Therapy vs No Therapy in Women at Risk of Late Preterm Delivery. JAMA Pediatr. 2019, 173, 462–468. [Google Scholar] [CrossRef]
- Phibbs, C.S.; Phibbs, R.H.; Wakeley, A.; Schlueter, M.A.; Sniderman, S.; Tooley, W.H. Cost effects of surfactant therapy for neonatal respiratory distress syndrome. J. Pediatr. 1993, 123, 953–962. [Google Scholar] [CrossRef]
- Johnson, T.J.; Patel, A.L.; Bigger, H.R.; Engstrom, J.L.; Meier, P.P. Cost Savings of Human Milk as a Strategy to Reduce the Incidence of Necrotizing Enterocolitis in Very Low Birth Weight Infants. Neonatology 2015, 107, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Trang, S.; Zupancic, J.A.; Unger, S.; Kiss, A.; Bando, N.; Wong, S.; Gibbins, S.; O’Connor, D.L. Cost-Effectiveness of Supplemental Donor Milk Versus Formula for Very Low Birth Weight Infants. Pediatrics 2018, 141, e20170737. [Google Scholar] [CrossRef] [PubMed]
- Ganapathy, V.; Hay, J.W.; Kim, J.H. Costs of Necrotizing Enterocolitis and Cost-Effectiveness of Exclusively Human Milk-Based Products in Feeding Extremely Premature Infants. Breastfeed Med. 2012, 7, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Hampson, G.; Roberts, S.L.E.; Lucas, A.; Parkin, D. An economic analysis of human milk supplementation for very low birth weight babies in the USA. BMC Pediatr. 2019, 19, 337. [Google Scholar] [CrossRef]
- Johnson, T.J.; Patel, A.L.; Bigger, H.R.; Engstrom, J.L.; Meier, P.P. Economic Benefits and Costs of Human Milk Feedings: A Strategy to Reduce the Risk of Prematurity-Related Morbidities in Very-Low-Birth-Weight Infants. Adv. Nutr. 2014, 5, 207–212. [Google Scholar] [CrossRef]
- Craighead, A.F.; Caughey, A.B.; Chaudhuri, A.; Yieh, L.; Hersh, A.R.; Dukhovny, D. Cost-effectiveness of probiotics for necrotizing enterocolitis prevention in very low birth weight infants. J. Perinatol. 2020, 1–10. [Google Scholar] [CrossRef]
- Lee, S.K.; Normand, C.; McMillan, D.; Ohlsson, A.; Vincer, M.; Lyons, C. Evidence for changing guidelines for routine screening for retinopathy of prematurity. Arch. Pediatr. Adolesc. Med. 2001, 155, 387–395. [Google Scholar] [CrossRef]
- Yanovitch, T.L.; Siatkowski, R.M.; McCaffree, M.; Corff, K.E. Retinopathy of prematurity in infants with birth weight>or=1250 grams-incidence, severity, and screening guideline cost-analysis. J. AAPOS 2006, 10, 128–134. [Google Scholar] [CrossRef]
- Porter, M.E. What Is Value in Health Care? N. Engl. J. Med. 2010, 363, 2477–2481. [Google Scholar] [CrossRef]
- Dukhovny, D.; Pursley, D.M.; Kirpalani, H.M.; Horbar, J.H.; Zupancic, J.A.F. Evidence, Quality, and Waste: Solving the Value Equation in Neonatology. Pediatrics 2016, 137, e20150312. [Google Scholar] [CrossRef]
- Ho, T.; Zupancic, J.A.; Pursley, D.M.; Dukhovny, D. Improving Value in Neonatal Intensive Care. Clin. Perinatol. 2017, 44, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Kaempf, J.W.; Zupancic, J.A.F.; Wang, L.; Grunkemeier, G.L. A Risk-Adjusted, Composite Outcomes Score and Resource Utilization Metrics for Very Low-Birth-Weight Infants. JAMA Pediatr. 2015, 169, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Profit, J.; Kowalkowski, M.A.; Zupancic, J.A.F.; Pietz, K.; Richardson, P.; Draper, D.; Hysong, S.J.; Thomas, E.J.; Petersen, L.A.; Gould, J.B. Baby-MONITOR: A Composite Indicator of NICU Quality. Pediatrics 2014, 134, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, P.; Tigue, P.M.; Perrin, J. The Value Proposition for Pediatric Care. JAMA Pediatr. 2019, 173, 1125–1126. [Google Scholar] [CrossRef]
- Lapcharoensap, W.; Lee, H.C.; Nyberg, A.; Dukhovny, D. Health Care and Societal Costs of Bronchopulmonary Dysplasia. NeoReviews 2018, 19, e211–e223. [Google Scholar] [CrossRef]
- American Academy of Pediatrics Committee on Fetus and Newborn. Hospital Discharge of the High-Risk Neonate. Pediatrics 2008, 122, 1119–1126. [Google Scholar] [CrossRef]
- Profit, J.; Gould, J.B.; Bennett, M.; Goldstein, B.A.; Draper, D.; Phibbs, C.S.; Lee, H.C. Racial/Ethnic Disparity in NICU Quality of Care Delivery. Pediatrics 2017, 140, e20170918. [Google Scholar] [CrossRef]
| Cost | Quality |
|---|---|
| Expenditure (hospital, staffing, pharmacy, etc.) Charges *: - facility and professional fees Charges * (with or without cost adjustment using Cost to Charge Ratios) Payments collected * Length of Stay Resource tallies with applied costs from literature | Morbidity Mortality Patient Safety Outcomes Complications Composite Measures Post-Acute Care Length of Stay |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Culbertson, L.; Dukhovny, D.; Lapcharoensap, W. Examining the Relationship between Cost and Quality of Care in the Neonatal Intensive Care Unit and Beyond. Children 2020, 7, 238. https://doi.org/10.3390/children7110238
Culbertson L, Dukhovny D, Lapcharoensap W. Examining the Relationship between Cost and Quality of Care in the Neonatal Intensive Care Unit and Beyond. Children. 2020; 7(11):238. https://doi.org/10.3390/children7110238
Chicago/Turabian StyleCulbertson, Lauren, Dmitry Dukhovny, and Wannasiri Lapcharoensap. 2020. "Examining the Relationship between Cost and Quality of Care in the Neonatal Intensive Care Unit and Beyond" Children 7, no. 11: 238. https://doi.org/10.3390/children7110238
APA StyleCulbertson, L., Dukhovny, D., & Lapcharoensap, W. (2020). Examining the Relationship between Cost and Quality of Care in the Neonatal Intensive Care Unit and Beyond. Children, 7(11), 238. https://doi.org/10.3390/children7110238





