Perioperative Point-of-Care Ultrasound in Children
Abstract
1. Introduction
2. Lungs and Airway
- (1)
- Lung sliding is a visualization of pleural movement between adjacent ribs. With respiration, lung movement causes the parietal and visceral pleura to slide against each other, creating the appearance of a shimmering linear structure referred to as “lung-sliding”.
- (2)
- Lung pulse refers to the pleural movement concurrent with cardiac contraction. Pulsations of blood flow result in small changes in lung volume, creating pleural movement.
- (3)
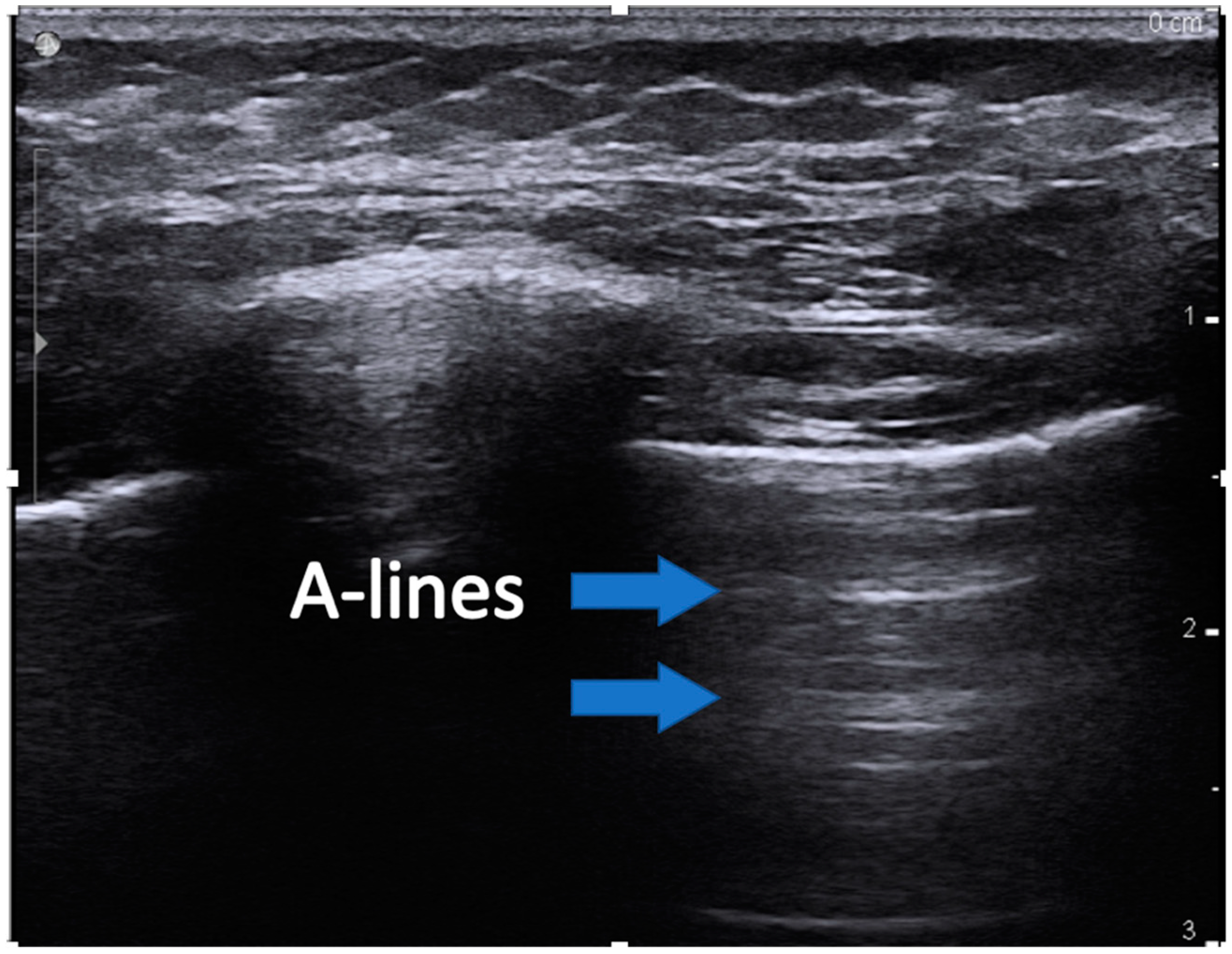
- A-lines are horizontal artifacts produced by air in the chest cavity. Air reflects ultrasound waves and prevents the visibility of structures beneath. Some of the reflected ultrasound waves, however, bounce back and forth between the muscle, and fascia of the chest wall and return to the transducer, generating reverberation artifact called a-lines (Figure 1).
- (4)
- B-lines are created when air is replaced in the alveoli by fluid, septal thickening, or other interstitial lung disease and which transmit the sound waves. B-lines originate at the pleural line and are long, vertical hyperechoic lines traversing the entire image depth (Figure 2). Up to two B-lines per rib space are normal, especially in dependent areas of the lung.
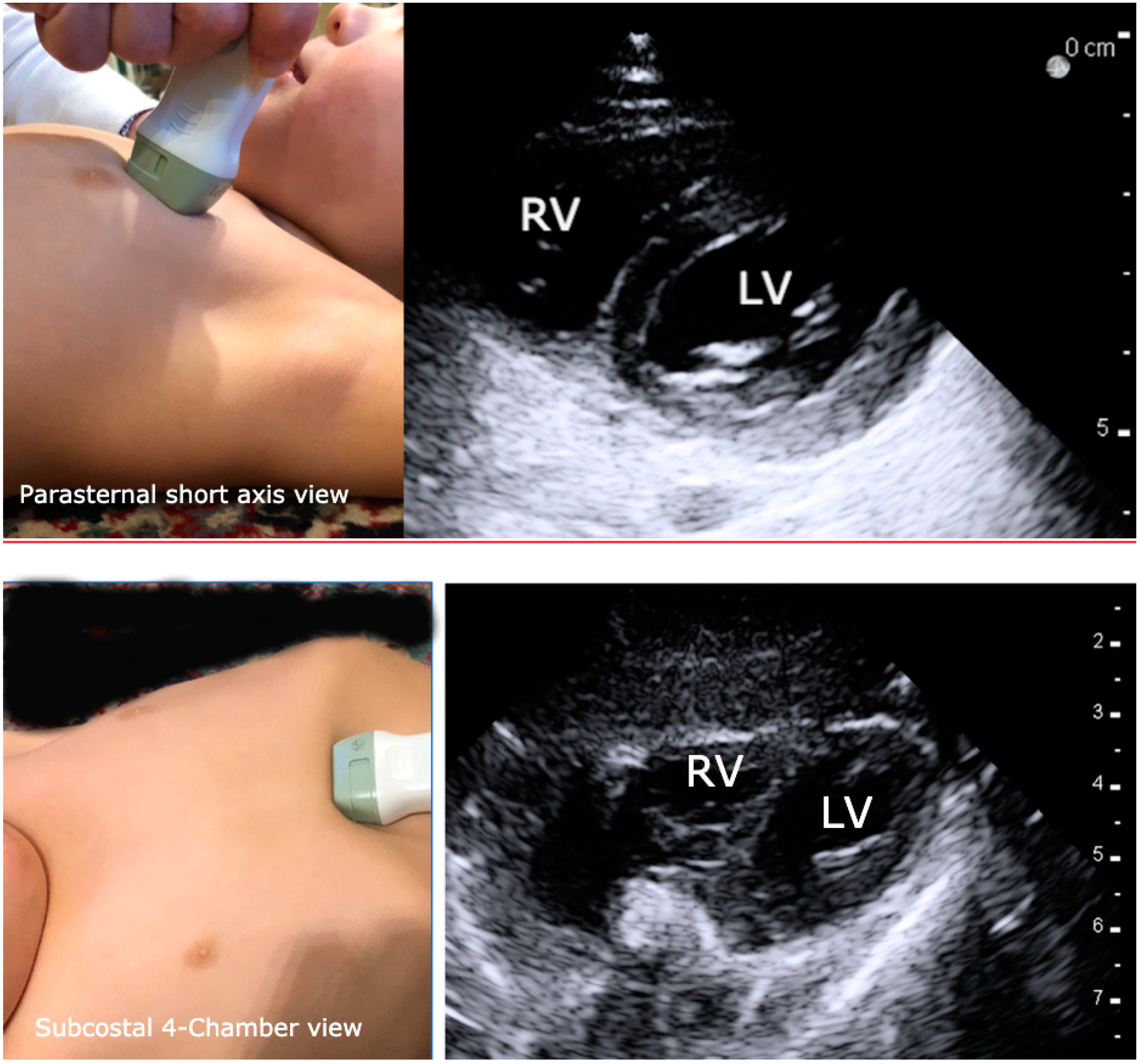
3. Focused Cardiac Ultrasound (FOCUS)
4. Gastric
5. Conclusions
Funding
Conflicts of Interest
References
- Legler, D.; Nugent, M. Doppler Localization of the Internal Jugular Vein Facilitates Central Venous Cannulation. Anesthesiology 1984, 60, 481. [Google Scholar] [CrossRef] [PubMed]
- Saugel, B.; Scheeren, T.W.L.; Teboul, J.-L. Ultrasound-guided central venous catheter placement: A structured review and recommendations for clinical practice. Crit. Care 2017, 21, 225. [Google Scholar] [CrossRef] [PubMed]
- Fraga, M.V.; Stoller, J.Z.; Glau, C.L.; De Luca, D.; Rempell, R.G.; Wenger, J.L.; Yek Kee, C.; Muhly, W.T.; Boretsky, K.; Conlon, T.W. Seeing Is Believing: Ultrasound in Pediatric Procedural Performance. Pediatrics 2019, 144, e20191401. [Google Scholar]
- Shekelle, P.G.; Wachter, R.M.; Pronovost, P.J.; Schoelles, K.; McDonald, K.M.; Dy, S.M.; Shojania, K.; Reston, J.; Berger, Z.; Johnsen, B.; et al. Making health care safer II: An updated critical analysis of the evidence for patient safety practices. Évid. Rep. Assess. 2013, 1–945. [Google Scholar]
- Lam, D.K.; Corry, G.N.; Tsui, B.C. Evidence for the Use of Ultrasound Imaging in Pediatric Regional Anesthesia. Reg. Anesthesia Pain Med. 2016, 41, 229–241. [Google Scholar] [CrossRef]
- Oberndorfer, U.; Marhofer, P.; Bösenberg, A.; Willschke, H.; Felfernig, M.; Weintraud, M.; Kapral, S.; Kettner, S.C. Ultrasonographic guidance for sciatic and femoral nerve blocks in children. Br. J. Anaesth. 2007, 98, 797–801. [Google Scholar] [CrossRef]
- Adler, A.C.; Belon, C.A.; Guffey, D.M.; Minard, C.G.; Patel, N.V.; Chandrakantan, A. Real-Time Ultrasound Improves Accuracy of Caudal Block in Children. Anesthesia Analg. 2020, 130, 1002–1007. [Google Scholar] [CrossRef]
- Boretsky, K.R.; Camelo, C.; Waisel, D.B.; Falciola, V.; Sullivan, C.; Brusseau, E.; Eastburn, E.; Gomez-Moraz, A.; Luckanavanich, W. Confirmation of success rate of landmark-based caudal blockade in children using ultrasound: A prospective analysis. Pediatr. Anesth. 2020, 30, 671–675. [Google Scholar] [CrossRef]
- Sinskey, J.L.; Vecchione, T.M.; Ekstrom, B.G.; Boretsky, K. Benefits of Ultrasound Imaging for Placement of Caudal Epidural Blockade in 3 Pediatric Patients. A&A Pract. 2018, 10, 307–309. [Google Scholar] [CrossRef]
- Willschke, H.; Bosenberg, A.; Marhofer, P.; Schwindt, J.; Weintraud, M.; Kapral, S.; Kettner, S. Epidural Catheter Placement in Neonates: Sonoanatomy and Feasibility of Ultrasonographic Guidance in Term and Preterm Neonates. Reg. Anesth. Pain Med. 2007, 32, 34–40. [Google Scholar] [CrossRef]
- Ponde, V.C.; Bedekar, V.V.; Desai, A.P.; Puranik, K.A. Does ultrasound guidance add accuracy to continuous caudal-epidural catheter placements in neonates and infants? Pediatr. Anesth. 2017, 27, 1010–1014. [Google Scholar] [CrossRef]
- Vecchione, T.M.; Boretsky, K.R. Ultrasound Images of the Epidural Space through the Acoustic Window of the Infant. Anesthesiology 2017, 126, 562. [Google Scholar] [CrossRef]
- Bosenberg, A. Innovative peripheral nerve blocks facilitated by ultrasound guidance. Pediatr. Anesth. 2018, 28, 684–685. [Google Scholar] [CrossRef]
- Eastburn, E.; Hernandez, M.A.; Boretsky, K. Technical success of the ultrasound-guided supra-inguinal fascia iliaca compartment block in older children and adolescents for hip arthroscopy. Pediatr. Anesth. 2017, 27, 1120–1124. [Google Scholar] [CrossRef]
- Vecchione, T.; Zurakowski, D.; Boretsky, K. Thoracic Paravertebral Nerve Blocks in Pediatric Patients. Anesth. Analg. 2016, 123, 1588–1590. [Google Scholar] [CrossRef]
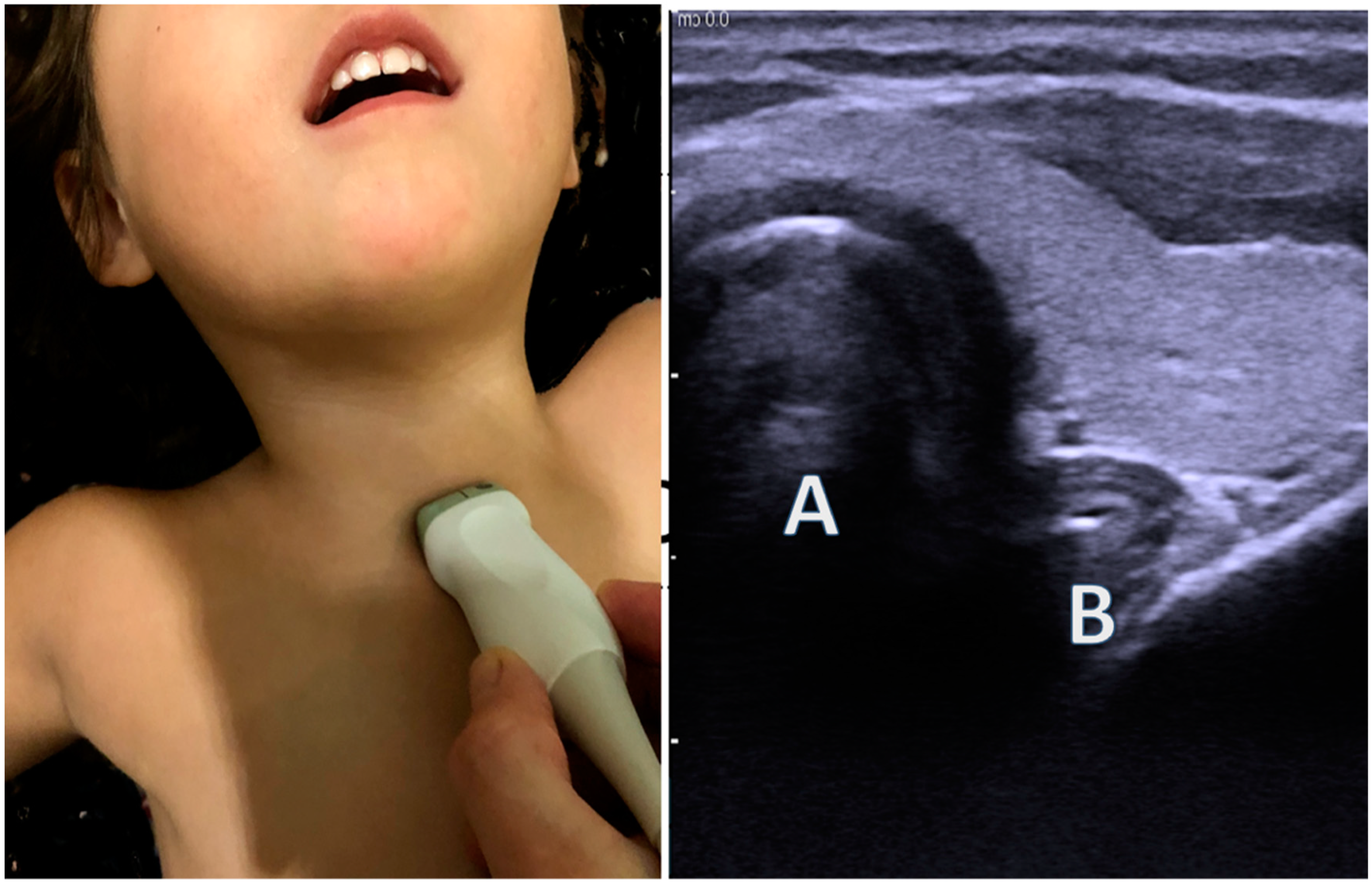
- Chen, R. Synopsis of the point-of-care ultrasound assessment for perioperative emergencies. Can. J. Anesth./J. Can. D’anesthésie 2019, 66, 448–460. [Google Scholar] [CrossRef]
- Kars, M.S.; Morad, A.G.; Haskins, S.C.; Boublik, J.; Boretsky, K. Point-of-care ultrasound for the pediatric regional anesthesiologist and pain specialist: A technique review. Reg. Anesth. Pain Med. 2020. [Google Scholar] [CrossRef]
- Su, E.; Dalesio, N.M.; Pustavoitau, A. Point-of-care ultrasound in pediatric anesthesiology and critical care medicine. Can. J. Anesth./J. Can. D’anesthésie 2018, 65, 485–498. [Google Scholar] [CrossRef]
- Moore, C.L.; Copel, J.A. Point-of-Care Ultrasonography. N. Engl. J. Med. 2011, 364, 749–757. [Google Scholar] [CrossRef]
- Volpicelli, G.; Elbarbary, M.; Blaivas, M.; Lichtenstein, D.A.; Mathis, G.; Kirkpatrick, A.W.; Melniker, L.; Gargani, L.; Noble, V.E.; Via, G.; et al. International Liaison Committee on Lung Ultrasound (ILC-LUS) for International Consensus Conference on Lung Ultrasound (ICC-LUS). International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med. 2012, 38, 577–591. [Google Scholar]
- Quintela, P.A.; Erroz, I.O.; Matilla, M.M.; Blanco, S.R.; Zubillaga, D.M.; Santos, L.R. Usefulness of bedside ultrasound compared to capnography and X-ray for tracheal intubation. An. Pediatría 2014, 81, 283–288. [Google Scholar] [CrossRef]
- Ramsingh, D.; Frank, E.; Haughton, R.; Schilling, J.; Gimenez, K.M.; Banh, E.; Rinehart, J.; Cannesson, M. Auscultation versus Point-of-care Ultrasound to Determine Endotracheal versus Bronchial Intubation. Anesthesiology 2016, 124, 1012–1020. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, J.; Takki, S.; Tammisto, T. Effect of the Trendelenburg Tilt and Other Procedures on the Position of Endotracheal Tubes. Lancet 1969, 293, 850–853. [Google Scholar] [CrossRef]
- Ezri, T.; Khazin, V.; Szmuk, P.; Medalion, B.; Shechter, P.; Priel, I.; Loberboim, M.; Weinbroum, A.A. Use of the Rapiscope vs chest auscultation for detection of accidental bronchial intubation in non-obese patients undergoing laparoscopic cholecystectomy. J. Clin. Anesth. 2006, 18, 118–123. [Google Scholar]
- Sooragonda, S.G.; Arora, S.; Jain, D.; Yaddanapudi, S. Lung sliding sign to detect endobronchial intubation in children: An observational feasibility trial. Eur. J. Anaesthesiol. 2020, 37, 143–145. [Google Scholar] [PubMed]
- Vezzani, A.; Manca, T.; Brusasco, C.; Santori, G.; Valentino, M.; Nicolini, F.; Molardi, A.; Gherli, T.; Corradi, F. Diagnostic value of chest ultrasound after cardiac surgery: A comparison with chest X-ray and auscultation. J. Cardiothorac. Vasc. Anesth. 2014, 28, 1527–1532. [Google Scholar]
- Sim, S.S.; Lien, W.C.; Chou, H.C.; Chong, K.M.; Liu, S.H.; Wang, C.H.; Chen, S.Y.; Hsu, C.Y.; Yen, Z.S.; Chang, W.T.; et al. Ultrasonographic lung sliding sign in confirming proper endotracheal intubation during emergency intubation. Resuscitation 2012, 8, 307–312. [Google Scholar]
- Shah, V.P.; Tunik, M.G.; Tsung, J.W. Prospective Evaluation of Point-of-Care Ultrasonography for the Diagnosis of Pneumonia in Children and Young Adults. JAMA Pediatr. 2013, 167, 119–125. [Google Scholar] [CrossRef]
- Pereda, M.A.; Chavez, M.A.; Hooper-Miele, C.C.; Gilman, R.H.; Steinhoff, M.C.; Ellington, L.E.; Gross, M.; Price, C.; Tielsch, J.M.; Checkley, W. Lung Ultrasound for the Diagnosis of Pneumonia in Children: A Meta-analysis. Pediatrics 2015, 135, 714–722. [Google Scholar] [CrossRef]
- Jones, B.P.; Tay, E.T.; Elikashvili, I.; Sanders, J.E.; Paul, A.Z.; Nelson, B.P.; Spina, L.A.; Tsung, J.W. Feasibility and Safety of Substituting Lung Ultrasonography for Chest Radiography When Diagnosing Pneumonia in Children. Chest 2016, 150, 131–138. [Google Scholar] [CrossRef]
- Silva, F.; Copetti, R. Pulmonary Ultrasound. In Pediatric Emergency and Critical Care Ultrasound; Doniger, S., Ed.; Cambridge University Press: Cambridge, UK, 2013; Volume 11. [Google Scholar]
- Hatch, L.D.; Grubb, P.H.; Lea, A.S.; Walsh, W.F.; Markham, M.H.; Whitney, G.M.; Slaughter, J.C.; Stark, A.R.; Ely, E.W. Endotracheal Intubation in Neonates: A Prospective Study of Adverse Safety Events in 162 Infants. J. Pediatr. 2016, 168, 62–66.e6. [Google Scholar] [CrossRef]
- Boretsky, K.R. Images in Anesthesiology: Point-of-care Ultrasound to Diagnose Esophageal Intubation. Anesthesiology 2018, 129, 190. [Google Scholar] [CrossRef]
- Hoffmann, B.; Gullett, J.P.; Hill, H.F.; Fuller, D.; Westergaard, M.C.; Hosek, W.T.; Smith, J.A. Bedside Ultrasound of the Neck Confirms Endotracheal Tube Position in Emergency Intubations. Ultraschall Med. Eur. J. Ultrasound 2014, 35, 451–458. [Google Scholar] [CrossRef]
- Lahham, S.; Baydoun, J.; Bailey, J.; Sandoval, S.; Wilson, S.P.; Fox, J.C.; Slattery, D.E. A Prospective Evaluation of Transverse Tracheal Sonography during Emergent Intubation by Emergency Medicine Resident Physicians. J. Ultrasound Med. 2017, 36, 2079–2085. [Google Scholar] [CrossRef][Green Version]
- Boretsky, K.R.; Kantor, D.B.; Dinardo, J.A.; Oren-Grinberg, A. Focused Cardiac Ultrasound in the Pediatric Perioperative Setting. Anesth. Analg. 2019, 129, 925–932. [Google Scholar] [CrossRef]
- Adler, A.C.; Brown, K.A.; Conlin, F.T.; Thammasitboon, S.; Chandrakantan, A. Cardiac and lung point-of-care ultrasound in pediatric anesthesia and critical care medicine: Uses, pitfalls, and future directions to optimize pediatric care. Pediatr. Anesth. 2019, 29, 790–798. [Google Scholar] [CrossRef]
- Adler, A.C. Perioperative Point-of-Care Ultrasound in Pediatric Anesthesiology: A Case Series Highlighting Intraoperative Diagnosis of Hemodynamic Instability and Alteration of Management. J. Cardiothorac. Vasc. Anesth. 2018, 32, 1411–1414. [Google Scholar] [CrossRef]
- Via, G.; Hussain, A.; Wells, M.; Reardon, R.; Elbarbary, M.; Noble, V.E.; Tsung, J.W.; Neskovic, A.N.; Price, S.; Oren-Grinberg, A.; et al. International Evidence-Based Recommendations for Focused Cardiac Ultrasound. J. Am. Soc. Echocardiogr. 2014, 27, 683.e1–683.e33. [Google Scholar] [CrossRef]
- Marin, J.R.; Lewiss, R.E. American Academy of Pediatrics, Committee on Pediatric Emergency Medicine. Point-Of-Care ultrasonography by pediatric emergency medicine physicians. Pediatrics 2015, 135, 1113–1122. [Google Scholar]
- Sicari, R.; Galderisi, M.; Voigt, J.-U.; Habib, G.; Zamorano, J.L.; Lancellotti, P.; Badano, L.P. The use of pocket-size imaging devices: A position statement of the European Association of Echocardiography. Eur. J. Echocardiogr. 2011, 12, 85–87. [Google Scholar] [CrossRef]
- Zimmerman, J.M.; Coker, B.J. The Nuts and Bolts of Performing Focused Cardiovascular Ultrasound (FoCUS). Anesth. Analg. 2017, 124, 753–760. [Google Scholar] [CrossRef]
- Longjohn, M.; Wan, J.; Joshi, V.; Pershad, J. Point-Of-Care echocardiography by pediatric emergency physicians. Pediatr. Emerg. Care 2011, 27, 693–696. [Google Scholar]
- Kutty, S.; Attebery, J.E.; Yeager, E.M.; Natarajan, S.; Li, L.; Peng, Q.; Truemper, E.; Hammel, J.M.; Danford, D.A. Transthoracic Echocardiography in Pediatric Intensive Care. Pediatr. Crit. Care Med. 2014, 15, 329–335. [Google Scholar] [CrossRef]
- Arnal, L.E.; Stein, F. Pediatric septic shock: Why has mortality decreased?—The utility of goal-directed therapy. Semin. Pediatr. Infect. Dis. 2003, 14, 165–172. [Google Scholar] [CrossRef]
- Conlon, T.W.; Falkensammer, C.B.; Hammond, R.S.; Nadkarni, V.M.; Berg, R.A.; Topjian, A.A. Association of Left Ventricular Systolic Function and Vasopressor Support With Survival Following Pediatric Out-of-Hospital Cardiac Arrest*. Pediatr. Crit. Care Med. 2015, 16, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Conlon, T.W.; Ishizuka, M.; Himebauch, A.S.; Cohen, M.S.; Berg, R.A.; Nishisaki, A. Hemodynamic Bedside Ultrasound Image Quality and Interpretation After Implementation of a Training Curriculum for Pediatric Critical Care Medicine Providers*. Pediatr. Crit. Care Med. 2016, 17, 598–604. [Google Scholar] [CrossRef]
- Sivitz, A.; Nagdev, A. Heart Failure Secondary to Dilated Cardiomyopathy. Pediatr. Emerg. Care 2012, 28, 163–166. [Google Scholar] [CrossRef]
- Hernandez, C.; Shuler, K.; Hannan, H.; Sonyika, C.; Likourezos, A.; Marshall, J. CAUSE: Cardiac arrest ultra-sound exam—A better approach to managing patients in primary non-arrhythmogenic cardiac arrest. Resuscitation 2008, 76, 198–206. [Google Scholar] [CrossRef]
- Varriale, P.; Maldonado, J.M. Echocardiographic observations during in hospital cardiopulmonary resuscitation. Crit. Care Med. 1997, 25, 1717–1720. [Google Scholar]
- Niendorff, D.F.; Rassias, A.J.; Palac, R.; Beach, M.L.; Costa, S.; Greenberg, M. Rapid cardiac ultrasound of inpatients suffering PEA arrest performed by nonexpert sonographers. Resuscitation 2005, 67, 81–87. [Google Scholar] [CrossRef]
- Rich, S.; Sheikh, A.; Gallastegui, J.; Kondos, G.T.; Mason, T.; Lam, W. Determination of left ventricular ejection fraction by visual estimation during real-time two-dimensional echocardiography. Am. Heart J. 1982, 104, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Hope, M.D.; De La Pena, E.; Yang, P.C.; Liang, D.H.; McConnell, M.V.; Rosenthal, D.N. A visual approach for the accurate determination of echocardiographic left ventricular ejection fraction by medical students. J. Am. Soc. Echocardiogr. 2003, 16, 824–831. [Google Scholar] [CrossRef]
- Walker, B.J.; Long, J.B.; Sathyamoorthy, M.; Birstler, J.; Wolf, C.; Bosenberg, A.T.; Flack, S.H.; Krane, E.J.; Sethna, N.F.; Suresh, S.; et al. Complications in pediatric regional anesthesia: An analysis of more than 100,000 blocks from the pediatric regional anesthesia network. Anesthesiology 2018, 129, 721–732. [Google Scholar]
- Habre, W.; Disma, N.; Virag, K.; Becke, K.; Hansen, T.G.; Jöhr, M.; Leva, B.; Morton, N.S.; Vermeulen, P.M.; Zielinska, M.; et al. Incidence of severe critical events in paediatric anaesthesia (APRICOT): A prospective multicentre observational study in 261 hospitals in Europe. Lancet Respir. Med. 2017, 5, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Pershad, J.; Chin, T. Early detection of cardiac disease masquerading as acute bronchospasm: The role of bedside limited echocardiography by the emergency physician. Pediatr. Emerg. Care 2003, 19, e1–e3. [Google Scholar] [CrossRef] [PubMed]
- Bramante, R.M.; Cirilli, A.; Raio, C.C. Point-of-Care Sonography in the Emergency Department Diagnosis of Acute H1N1 Influenza Myocarditis. J. Ultrasound Med. 2010, 29, 1361–1364. [Google Scholar] [CrossRef] [PubMed]
- Tsung, J.W.; Blaivas, M. Feasibility of correlating the pulse check with focused point-of-care echocardiography during pediatric cardiac arrest: A case series. Resuscitation 2008, 77, 264–269. [Google Scholar] [CrossRef]
- Tibballs, J.; Weeranatna, C. The influence of time on the accuracy of healthcare personnel to diagnose paediatric cardiac arrest by pulse palpation. Resuscitation 2010, 81, 671–675. [Google Scholar] [CrossRef]
- Tibballs, J.; Russell, P. Reliability of pulse palpation by healthcare personnel to diagnose paediatric cardiac arrest. Resuscitation 2009, 80, 61–64. [Google Scholar] [CrossRef]
- Breitkreutz, R.; Price, S.; Steiger, H.V.; Seeger, F.H.; Ilper, H.; Ackermann, H.; Rudolph, M.; Uddin, S.; Weigand, M.A.; Müller, E.; et al. Focused echocardiographic evaluation in life support and peri-resuscitation of emergency patients: A prospective trial. Resuscitation 2010, 81, 1527–1533. [Google Scholar] [CrossRef]
- Lasa, J.J.; Rogers, R.S.; Localio, R.; Shults, J.; Raymond, T.T.; Gaies, M.G.; Thiagarajan, R.R.; Laussen, P.C.; Kilbaugh, T.J.; Berg, R.A.; et al. Extracorporeal Cardiopulmonary Resuscitation (E-CPR) During Pediatric In-Hospital Cardiopulmonary Arrest Is Associated With Improved Survival to Discharge. Circulation 2016, 133, 165–176. [Google Scholar] [CrossRef]
- Nasr, V.G.; Gottlieb, E.A.; Adler, A.C.; Evans, M.A.; Sawardekar, A.; Dinardo, J.A.; Mossad, E.B.; Mittnacht, A.J. Selected 2018 Highlights in Congenital Cardiac Anesthesia. J. Cardiothorac. Vasc. Anesth. 2019, 33, 2833–2842. [Google Scholar] [CrossRef]
- Borland, L.M.; Sereika, S.M.; Woelfel, S.K.; Saitz, E.W.; Carrillo, P.A.; Lupin, J.L.; Motoyama, E.K. Pulmonary Aspiration in Pediatric Patients During General Anesthesia: Incidence and Outcome. J. Clin. Anesth. 1998, 10, 95–102. [Google Scholar] [CrossRef]
- Tan, Z.; Lee, S.Y. Pulmonary aspiration under GA: A 13-year audit in a tertiary pediatric unit. Pediatr. Anesth. 2016, 26, 547–552. [Google Scholar] [CrossRef]
- Sakai, T.; Planinsic, R.M.; Quinlan, J.J.; Handley, L.J.; Kim, T.-Y.; Hilmi, I.A. The Incidence and Outcome of Perioperative Pulmonary Aspiration in a University Hospital: A 4-Year Retrospective Analysis. Anesth. Analg. 2006, 103, 941–947. [Google Scholar] [CrossRef]
- Gagey, A.-C.; Siqueira, M.D.Q.; Desgranges, F.-P.; Combet, S.; Naulin, C.; Chassard, D.; Bouvet, L. Ultrasound assessment of the gastric contents for the guidance of the anaesthetic strategy in infants with hypertrophic pyloric stenosis: A prospective cohort study. Br. J. Anaesth. 2016, 116, 649–654. [Google Scholar] [CrossRef]
- Boretsky, K.R.; Perlas, A. Gastric Ultrasound Imaging to Direct Perioperative Care in Pediatric Patients. A&A Pract. 2019, 13, 443–445. [Google Scholar] [CrossRef]
- Perlas, A.; Mitsakakis, N.; Liu, L.; Cino, M.; Haldipur, N.; Davis, L.; Cubillos, J.; Chan, V. Validation of a mathematical model for ultrasound. Anesth. Analg. 2013, 116, 357–363. [Google Scholar] [PubMed]
- Cubillos, J.; Tse, C.; Chan, V.W.S.; Perlas, A. Bedside ultrasound assessment of gastric content: An observational study. Can. J. Anesth./J. Can. D’anesthésie 2012, 59, 416–423. [Google Scholar] [CrossRef]
- Spencer, A.O.; Walker, A.M.; Yeung, A.K.; Lardner, D.R.; Yee, K.; Mulvey, J.M.; Perlas, A. Ultrasound assessment of gastric volume in the fasted pediatric patient undergoing upper gastrointestinal endoscopy: Development of a predictive model using endoscopically suctioned volumes. Pediatr. Anesth. 2015, 25, 301–308. [Google Scholar]
- Bouvet, L.; Miquel, A.; Chassard, D.; Boselli, E.; Allaouchiche, B.; Benhamou, D. Could a single standardized ultrasonographic measurement of antral area be of interest for assessing gastric contents? A preliminary report. Eur. J. Anaesthesiol. 2009, 26, 1015–1019. [Google Scholar] [CrossRef]
- Kruisselbrink, R.; Gharapetian, A.; Chaparro, L.E.; Ami, N.; Richler, D.; Chan, V.W.S.; Perlas, A. Diagnostic Accuracy of Point-of-Care Gastric Ultrasound. Anesth. Analg. 2019, 128, 89–95. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boretsky, K. Perioperative Point-of-Care Ultrasound in Children. Children 2020, 7, 213. https://doi.org/10.3390/children7110213
Boretsky K. Perioperative Point-of-Care Ultrasound in Children. Children. 2020; 7(11):213. https://doi.org/10.3390/children7110213
Chicago/Turabian StyleBoretsky, Karen. 2020. "Perioperative Point-of-Care Ultrasound in Children" Children 7, no. 11: 213. https://doi.org/10.3390/children7110213
APA StyleBoretsky, K. (2020). Perioperative Point-of-Care Ultrasound in Children. Children, 7(11), 213. https://doi.org/10.3390/children7110213



