Respiratory Diagnostic Tools in Neuromuscular Disease
Abstract
1. Introduction
2. Clinical Assessment
3. Evaluation of Lung Function
3.1. Spirometry
3.2. Lung Volumes
4. Peak Cough Flow
5. Respiratory Muscle Strength Testing
5.1. Maximal Expiratory Pressure
5.2. Maximal Inspiratory Pressure
5.3. Sniff Nasal Inspiratory Pressure
6. Evaluation of Breathing during Sleep
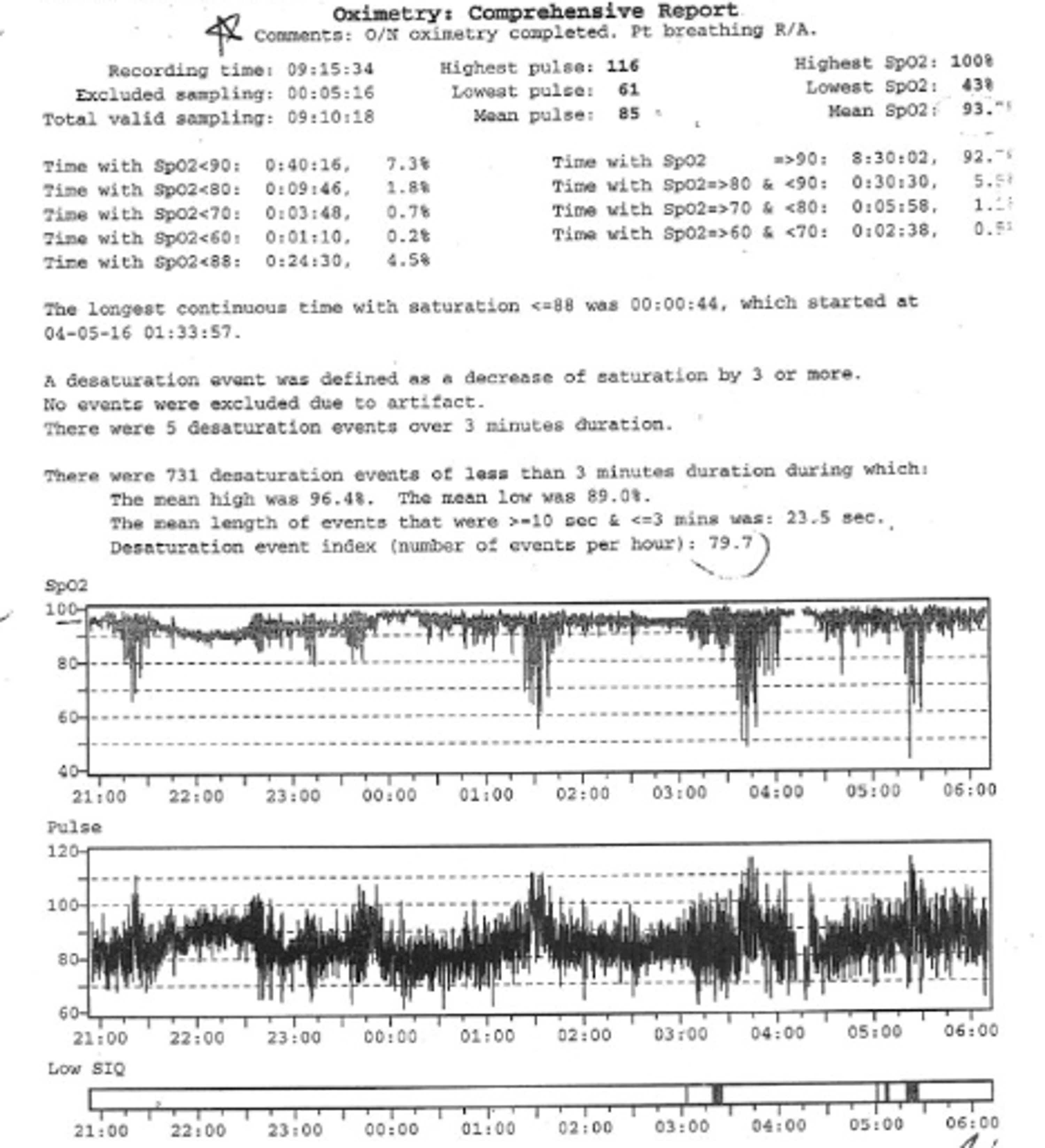
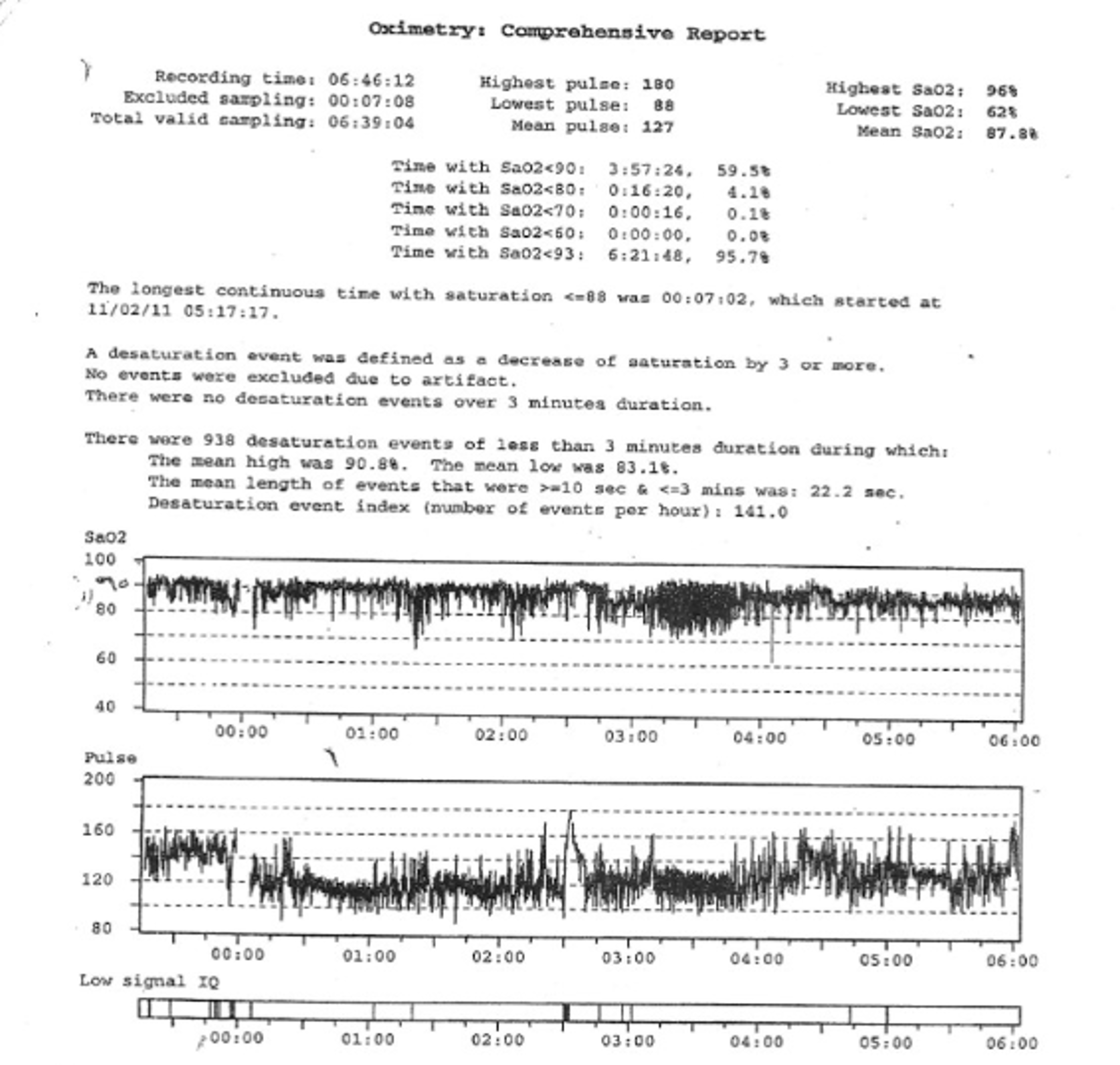
6.1. Oximetry
6.2. Capnography
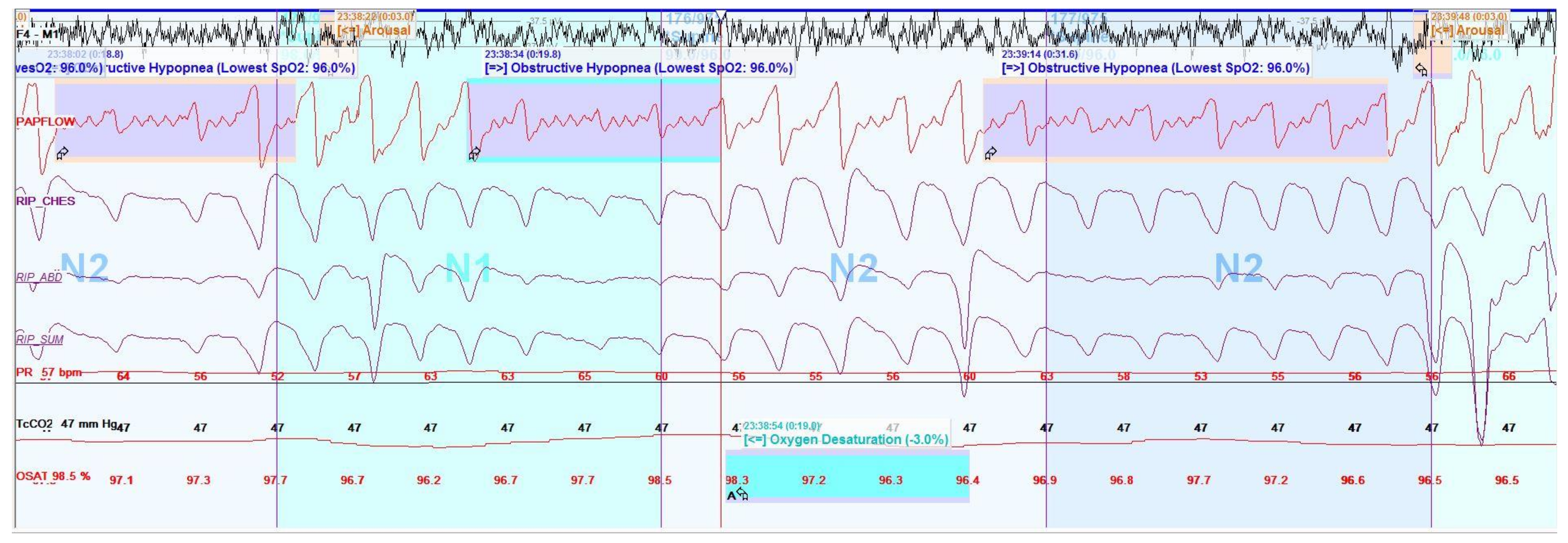
6.3. Polysomnography
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Panitch, H.B. The pathophysiology of respiratory impairment in pediatric neuromuscular diseases. Pediatrics 2009, 123 (Suppl. 4), S215–S218. [Google Scholar] [CrossRef] [PubMed]
- Panitch, H.B. Respiratory Implications of Pediatric Neuromuscular Disease. Respir. Care 2017, 62, 826–848. [Google Scholar] [CrossRef] [PubMed]
- Hull, J.; Aniapravan, R.; Chan, E.; Chatwin, M.; Forton, J.; Gallagher, J.; Gibson, N.; Gordon, J.; Hughes, I.; McCulloch, R.; et al. British Thoracic Society guideline for respiratory management of children with neuromuscular weakness. Thorax 2012, 67 (Suppl. 1), i1–i40. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.D. Pulmonary function testing in neuromuscular disorders. Pediatrics 2009, 123 (Suppl. 4), S219–S221. [Google Scholar] [CrossRef] [PubMed]
- Birnkrant, D.J.; Bushby, K.M.; Amin, R.S.; Bach, J.R.; Benditt, J.O.; Eagle, M.; Finder, J.D.; Kalra, M.S.; Kissel, J.T.; Koumbourlis, A.C.; et al. The respiratory management of patients with Duchenne muscular dystrophy: A DMD care considerations working group specialty article. Pediatr. Pulmonol. 2010, 45, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Stanojevic, S.; Wade, A.; Stocks, J.; Hankinson, J.; Coates, A.L.; Pan, H.; Rosenthal, M.; Corey, M.; Lebecque, P.; Cole, T.J. Reference ranges for spirometry across all ages: A new approach. Am. J. Respir. Crit. Care Med. 2008, 177, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, R.; Viegi, G.; Brusasco, V.; Crapo, R.O.; Burgos, F.; Casaburi, R.; Coates, A.; van der Grinten, C.P.; Gustafsson, P.; Hankinson, J.; et al. Interpretative strategies for lung function tests. Eur. Respir. J. 2005, 26, 948–968. [Google Scholar] [CrossRef] [PubMed]
- Dohna-Schwake, C.; Ragette, R.; Teschler, H.; Voit, T.; Mellies, U. Predictors of severe chest infections in pediatric neuromuscular disorders. Neuromuscul. Disord. 2006, 16, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Finder, J.D.; Birnkrant, D.; Carl, J.; Farber, H.J.; Gozal, D.; Iannaccone, S.T.; Kovesi, T.; Kravitz, R.M.; Panitch, H.; Schramm, C.; et al. Respiratory care of the patient with Duchenne muscular dystrophy: ATS consensus statement. Am. J. Respir. Crit. Care Med. 2004, 170, 456–465. [Google Scholar] [PubMed]
- Rideau, Y.; Jankowski, L.W.; Grellet, J. Respiratory function in the muscular dystrophies. Muscle Nerve 1981, 4, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Biggar, W.D.; Harris, V.A.; Eliasoph, L.; Alman, B. Long-term benefits of deflazacort treatment for boys with Duchenne muscular dystrophy in their second decade. Neuromuscul. Disord. 2006, 16, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Wanger, J.; Clausen, J.L.; Coates, A.; Pedersen, O.F.; Brusasco, V.; Burgos, F.; Casaburi, R.; Crapo, R.; Enright, P.; van der Grinten, C.P.; et al. Standardisation of the measurement of lung volumes. Eur. Respir. J. 2005, 26, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Fauroux, B.; Khirani, S. Neuromuscular disease and respiratory physiology in children: Putting lung function into perspective. Respirology 2014, 19, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Chatwin, M.; Ross, E.; Hart, N.; Nickol, A.H.; Polkey, M.I.; Simonds, A.K. Cough augmentation with mechanical insufflation/exsufflation in patients with neuromuscular weakness. Eur. Respir. J. 2003, 21, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.R.; Ishikawa, Y.; Kim, H. Prevention of pulmonary morbidity for patients with Duchenne muscular dystrophy. Chest 1997, 112, 1024–1028. [Google Scholar] [CrossRef] [PubMed]
- Stafler, P.; Carr, S.B. Non-cystic fibrosis bronchiectasis: Its diagnosis and management. Arch. Dis. Child. Educ. Pract. Ed. 2010, 95, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.R.; Saporito, L.R. Criteria for extubation and tracheostomy tube removal for patients with ventilatory failure. A different approach to weaning. Chest 1996, 110, 1566–1571. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.; Baiardi, P. Cough peak flows: Standard values for children and adolescents. Am. J. Phys. Med. Rehabil. 2008, 87, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Chiang, J.; Amin, R. Respiratory Care Considerations for Children with Medical Complexity. Children 2017, 4, 41. [Google Scholar] [CrossRef] [PubMed]
- Fauroux, B.; Quijano-Roy, S.; Desguerre, I.; Khirani, S. The value of respiratory muscle testing in children with neuromuscular disease. Chest 2015, 147, 552–559. [Google Scholar] [CrossRef] [PubMed]
- American Thoracic Society/European Respiratory Society. ATS/ERS Statement on respiratory muscle testing. Am. J. Respir. Crit. Care Med. 2002, 166, 518–624. [Google Scholar]
- Fauroux, B. Respiratory muscle testing in children. Paediatr. Respir. Rev. 2003, 4, 243–249. [Google Scholar] [CrossRef]
- Hahn, A.; Bach, J.R.; Delaubier, A.; Renardel-Irani, A.; Guillou, C.; Rideau, Y. Clinical implications of maximal respiratory pressure determinations for individuals with Duchenne muscular dystrophy. Arch. Phys. Med. Rehabil. 1997, 78, 1–6. [Google Scholar] [CrossRef]
- Mellies, U.; Ragette, R.; Schwake, C.; Boehm, H.; Voit, T.; Teschler, H. Daytime predictors of sleep disordered breathing in children and adolescents with neuromuscular disorders. Neuromuscul. Disord. 2003, 13, 123–128. [Google Scholar] [CrossRef]
- Stefanutti, D.; Fitting, J.W. Sniff nasal inspiratory pressure. Reference values in Caucasian children. Am. J. Respir. Crit. Care Med. 1999, 159, 107–111. [Google Scholar] [PubMed]
- Rafferty, G.F.; Leech, S.; Knight, L.; Moxham, J.; Greenough, A. Sniff nasal inspiratory pressure in children. Pediatr. Pulmonol. 2000, 29, 468–475. [Google Scholar] [CrossRef]
- Smyth, R.J.; Chapman, K.R.; Rebuck, A.S. Maximal inspiratory and expiratory pressures in adolescents. Normal values. Chest 1984, 86, 568–572. [Google Scholar] [CrossRef] [PubMed]
- Nicot, F.; Hart, N.; Forin, V.; Boulé, M.; Clément, A.; Polkey, M.I.; Lofaso, F.; Fauroux, B. Respiratory muscle testing: A valuable tool for children with neuromuscular disorders. Am. J. Respir. Crit. Care Med. 2006, 174, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Kirk, V.G.; Morielli, A.; Gozal, D.; Marcus, C.L.; Waters, K.A.; D’Andrea, L.A.; Rosen, C.L.; Deray, M.J.; Brouillette, R.T. Treatment of sleep-disordered breathing in children with myelomeningocele. Pediatr. Pulmonol. 2000, 30, 445–452. [Google Scholar] [CrossRef]
- Iber, C.A.; Chesson, A.; Quan, S.F. The AASM Manual for the Scoring of Sleep and Associated Events; American Academy of Sleep Medicine: Winchester, IL, USA, 2016. [Google Scholar]
- Katz, E.S.; D’Ambrosio, C.M. Pediatric obstructive sleep apnea syndrome. Clin. Chest Med. 2010, 31, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Wakai, M.; Nishikage, H.; Goshima, K.; Yamamoto, J. Polysomnographic Features of Idiopathic Central Sleep Apnea. Psychiatry Clin. Neurosci. 2002, 56, 323–324. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, C.D.; Walsh, B.K. Noninvasive Monitoring of Oxygen and Ventilation. Respir. Care 2017, 62, 751–764. [Google Scholar] [CrossRef] [PubMed]
- Bauman, K.A.; Kurili, A.; Schmidt, S.L.; Rodriguez, G.M.; Chiodo, A.E.; Sitrin, R.G. Home-based overnight transcutaneous capnography/pulse oximetry for diagnosing nocturnal hypoventilation associated with neuromuscular disorders. Arch. Phys. Med. Rehabil. 2012, 94, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Randerath, W.J.; Treml, M.; Priegnitz, C.; Stieglitz, S.; Hagmeyer, L.; Morgenstern, C. Evaluation of a Noninvasive Algorithm for Differentiation of Obstructive and Central Hypopneas. Sleep 2013, 36, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.L.; Witmans, M.; Barrowman, N.; Hoey, L.; Su, S.; Reddy, D.; Narang, I. Paediatric sleep resources in Canada: The scope of the problem. Paediatr. Child Health 2014, 19, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Hukins, C.A.; Hillman, D.R. Daytime predictors of sleep hypoventilation in Duchenne muscular dystrophy. Am. J. Respir. Crit. Care Med. 2000, 161, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Finkel, R.S.; Mercuri, E.; Meyer, O.H.; Simonds, A.K.; Schroth, M.K.; Graham, R.J.; Kirschner, J.; Iannaccone, S.T.; Crawford, T.O.; Woods, S. Diagnosis and management of spinal muscular atrophy: Part 2: Pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul. Disord. 2017, 28, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.L.; Gaboury, I.; Keilty, K.; Banwell, B.; Vajsar, J.; Anderson, P.; Ni, A.; Maclusky, I. Nocturnal Hypoventilation: Predictors and Outcomes in Childhood Progressive Neuromuscular Disease. Arch. Dis. Child. 2010, 95, 998–1003. [Google Scholar] [CrossRef] [PubMed]
- Amin, R.; MacLusky, I.; Zielinski, D.; Adderley, R.; Carnevale, F.; Chiang, J.; Côté, A.; Daniels, C.; Daigneault, P.; Harrison, C.; et al. Pediatric home mechanical ventilation: A Canadian Thoracic Society clinical practice guideline executive summary. Can. J. Respir. Crit. Care Sleep Med. 2017, 1, 7–36. [Google Scholar] [CrossRef]



| Test | PFT Findings |
|---|---|
| FEV1 | ↓ |
| FEV1/FVC | Normal |
| FVC or VC | ↓ |
| TLC | ↓ |
| RV | ↑ |
| FRC | Normal |
| MIP | ↓ |
| MEP | ↓ |
| PCF | ↓ |
| Recommendation | Diagnostic Test Results |
|---|---|
| Initiate cough augmentation device | PCF < 270 L/min for children ≥12 years of age |
| MEP < 60 cmH2O | |
| Perform overnight sleep monitoring | FVC < 60% predicted |
| MIP < 40 cmH2O | |
| Clinical symptoms suggestive of SDB | |
| Loss of ambulation | |
| Children with NMD that will never have the ability to ambulate | |
| Infants with NMD | |
| Initiate nocturnal non-invasive ventilation (DMD specifically) | Baseline awake SpO2 < 95% |
| Baseline awake pCO2 > 45 mmHg | |
| FVC < 30–50% if undergoing a surgical procedure | |
| Evidence of SDB on PSG |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiang, J.; Mehta, K.; Amin, R. Respiratory Diagnostic Tools in Neuromuscular Disease. Children 2018, 5, 78. https://doi.org/10.3390/children5060078
Chiang J, Mehta K, Amin R. Respiratory Diagnostic Tools in Neuromuscular Disease. Children. 2018; 5(6):78. https://doi.org/10.3390/children5060078
Chicago/Turabian StyleChiang, Jackie, Kevan Mehta, and Reshma Amin. 2018. "Respiratory Diagnostic Tools in Neuromuscular Disease" Children 5, no. 6: 78. https://doi.org/10.3390/children5060078
APA StyleChiang, J., Mehta, K., & Amin, R. (2018). Respiratory Diagnostic Tools in Neuromuscular Disease. Children, 5(6), 78. https://doi.org/10.3390/children5060078




