Placebo by Proxy in Neonatal Randomized Controlled Trials: Does It Matter?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Statistics
3. Results
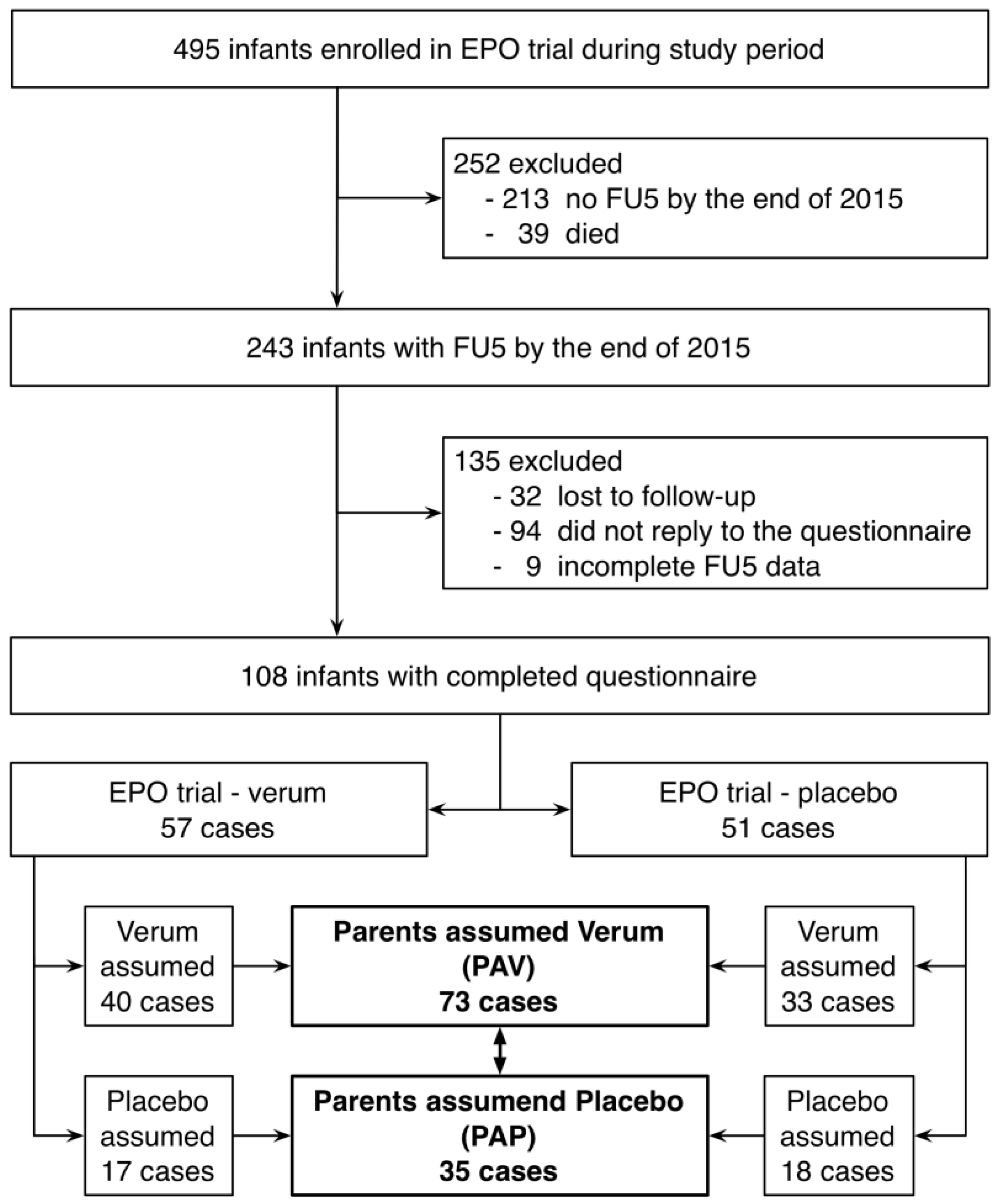
3.1. Study Population
3.2. Comparison Between Groups
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Finniss, D.G.; Kaptchuk, T.J.; Miller, F.; Benedetti, F. Biological, clinical, and ethical advances of placebo effects. Lancet 2010, 375, 686–695. [Google Scholar] [CrossRef]
- Kaptchuk, T.J.; Miller, F.G. Placebo effects in medicine. N. Engl. J. Med. 2015, 373, 8–9. [Google Scholar] [CrossRef] [PubMed]
- LeWitt, P.A.; Kim, S. The pharmacodynamics of placebo: Expectation effects of price as a proxy for efficacy. Neurology 2015, 84, 766–767. [Google Scholar] [CrossRef] [PubMed]
- Grelotti, D.J.; Kaptchuk, T.J. Placebo by proxy. BMJ 2011, 343, d4345. [Google Scholar] [CrossRef] [PubMed]
- Conzemius, M.G.; Evans, R.B. Caregiver placebo effect for dogs with lameness from osteoarthritis. J. Am. Vet. Med. Assoc. 2012, 241, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Whalley, B.; Hyland, M.E. Placebo by proxy: The effect of parents’ beliefs on therapy for children’s temper tantrums. J. Behav. Med. 2013, 36, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Fauchere, J.C.; Dame, C.; Vonthein, R.; Koller, B.; Arri, S.; Wolf, M.; Bucher, H.U. An approach to using recombinant erythropoietin for neuroprotection in very preterm infants. Pediatrics 2008, 122, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Melchers, P.; Preuss, U. Kaufman assessment battery for children (german version). Frankf. Am. Main Pearson Assess. 2009. [Google Scholar]
- Largo, R.H.; Graf, S.; Kundu, S.; Hunziker, U.; Molinari, L. Predicting developmental outcome at school age from infant tests of normal, at-risk and retarded infants. Dev. Med. Child Neurol. 1990, 32, 30–45. [Google Scholar] [CrossRef] [PubMed]
- Jones, B.; Jarvis, P.; Lewis, J.A.; Ebbutt, A.F. Trials to assess equivalence: The importance of rigorous methods. BMJ 1996, 313, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Piaggio, G.; Elbourne, D.R.; Pocock, S.J.; Evans, S.J.; Altman, D.G.; Group, C. Reporting of noninferiority and equivalence randomized trials: Extension of the consort 2010 statement. JAMA 2012, 308, 2594–2604. [Google Scholar] [CrossRef] [PubMed]
- Mangione-Smith, R.; McGlynn, E.A.; Elliott, M.N.; Krogstad, P.; Brook, R.H. The relationship between perceived parental expectations and pediatrician antimicrobial prescribing behavior. Pediatrics 1999, 103, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Bauchner, H.; Pelton, S.I.; Klein, J.O. Parents, physicians, and antibiotic use. Pediatrics 1999, 103, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.Y.; Warre, R.; Qiu, X.M.; O’Brien, K.; Lee, S.K. Parents as practitioners in preterm care. Early Hum. Dev. 2014, 90, 781–785. [Google Scholar] [CrossRef] [PubMed]
- Ruegger, C.M.; Kraus, A.; Koller, B.; Natalucci, G.; Latal, B.; Waldesbuhl, E.; Fauchere, J.C.; Held, L.; Bucher, H.U. Randomized controlled trials in very preterm infants: Does inclusion in the study result in any long-term benefit? Neonatology 2014, 106, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Rheims, S.; Cucherat, M.; Arzimanoglou, A.; Ryvlin, P. Greater response to placebo in children than in adults: A systematic review and meta-analysis in drug-resistant partial epilepsy. PLoS Med. 2008, 5, e166. [Google Scholar] [CrossRef] [PubMed]
- Janiaud, P.; Cornu, C.; Lajoinie, A.; Djemli, A.; Cucherat, M.; Kassai, B. Is the perceived placebo effect comparable between adults and children? A meta-regression analysis. Pediatr. Res. 2017, 81, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, F. Placebo and the new physiology of the doctor-patient relationship. Physiol. Rev. 2013, 93, 1207–1246. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.T.; Loscalzo, J.; Kaptchuk, T.J. Genetics and the placebo effect: The placebome. Trends Mol. Med. 2015, 21, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Price, D.D.; Finniss, D.G.; Benedetti, F. A comprehensive review of the placebo effect: Recent advances and current thought. Annu. Rev. Psychol. 2008, 59, 565–590. [Google Scholar] [CrossRef] [PubMed]
- Colloca, L.; Benedetti, F. Placebo analgesia induced by social observational learning. Pain 2009, 144, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Singer, L.T.; Fulton, S.; Kirchner, H.L.; Eisengart, S.; Lewis, B.; Short, E.; Min, M.O.; Kercsmar, C.; Baley, J.E. Parenting very low birth weight children at school age: Maternal stress and coping. J. Pediatrics 2007, 151, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Muller-Nix, C.; Forcada-Guex, M.; Pierrehumbert, B.; Jaunin, L.; Borghini, A.; Ansermet, F. Prematurity, maternal stress and mother-child interactions. Early Hum. Dev. 2004, 79, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Forcada-Guex, M.; Borghini, A.; Pierrehumbert, B.; Ansermet, F.; Muller-Nix, C. Prematurity, maternal posttraumatic stress and consequences on the mother-infant relationship. Early Hum. Dev. 2011, 87, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Shah, E.; Pimentel, M. Placebo effect in clinical trial design for irritable bowel syndrome. J. Neurogastroenterol. Motil. 2014, 20, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Curie, A.; Yang, K.; Kirsch, I.; Gollub, R.L.; des Portes, V.; Kaptchuk, T.J.; Jensen, K.B. Placebo responses in genetically determined intellectual disability: A meta-analysis. PLoS ONE 2015, 10, e0133316. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.P.; Wislar, J.S. Response rates and nonresponse errors in surveys. JAMA 2012, 307, 1805–1806. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.P. Indicator and stratification methods for missing explanatory variables in multiple linear regression. J. Am. Stat. Assoc. 1996, 91, 222–230. [Google Scholar] [CrossRef]

| Characteristics | Included in the Present Study (n = 108) | Not Included in the Present Study (n = 135) | p-value b |
|---|---|---|---|
| Pregnancy complications 1 | 48/108 (44.4) | 89/135 (65.9) | <0.01 |
| Prenatal steroids | 103/108 (95.4) | 129/135 (95.6) | 1.00 |
| Male sex | 73/108 (67.6) | 67/135 (49.6) | <0.01 |
| Gestational age (weeks) | 28.9 ± 1.8 | 29.2 ± 1.7 | 0.14 |
| Birth weight (g) | 1177 ± 322 | 1183 ± 351 | 0.95 |
| Head circumference at birth (cm) | 26.7 ± 2.2 | 26.6 ± 2.1 | 0.58 |
| Intraventricular hemorrhage III–IV | 6/108 (5.6) | 3/134 (2.2) | 0.19 |
| Sepsis | 9/108 (8.3) | 16/135 (11.8) | 0.40 |
| Retinopathy of prematurity (any stage) | 9/107 (8.4) | 14/134 (10.4) | 0.66 |
| Necrotizing enterocolitis | 4/108 (3.7) | 3/135 (2.2) | 0.70 |
| Bronchopulmonary dysplasia | 35/108 (32.4) | 45/135 (33.3) | 0.89 |
| Maternal age, years | 32.1 ± 5.5 | 31.9 ± 5.9 | 0.70 |
| SES | 5.6 ± 2.3 | 6.2 ± 2.4 | 0.02 |
| Characteristics | Parents Assumed Verum (PAV) (n = 73) | Parents Assumed Placebo (PAP) (n = 35) | p-value b |
|---|---|---|---|
| Pregnancy complications 1 | 34/73 (46.6) | 14/35 (40.0) | 0.54 |
| Prenatal steroids | 69/73 (94.5) | 34/35 (97.2) | 1.00 |
| Male sex | 49/73 (67.1) | 24/35 (68.6) | 1.00 |
| Gestational age (weeks) | 28.7 ± 1.8 | 29.2 ± 1.6 | 0.24 |
| Birth weight (g) | 1166 ± 336 | 1198 ± 296 | 0.69 |
| Head circumference at birth (cm) | 26.5 ± 2.3 | 27.0 ± 1.9 | 0.23 |
| Intraventricular hemorrhage III–IV | 6/67 (8.2) | 0/35 (0.0) | 0.17 |
| Sepsis | 9/73 (12.3) | 0/35 (0.0) | 0.03 |
| Retinopathy of prematurity (any stage) | 7/73 (9.6) | 2/34 (5.9) | 0.72 |
| Necrotizing enterocolitis | 4/73 (5.5) | 0/35 (0.0) | 0.30 |
| Bronchopulmonary dysplasia | 25/73 (34.2) | 10/35 (28.6) | 0.66 |
| SES | 5.6 ± 2.2 | 5.5 ± 2.4 | 0.60 |
| Maternal age (years) | 32.3 ± 5.6 | 31.6 ± 5.4 | 0.40 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burkart, T.L.; Kraus, A.; Koller, B.; Natalucci, G.; Latal, B.; Fauchère, J.-C.; Bucher, H.U.; Rüegger, C.M.; For the Swiss EPO Neuroprotection Trial Group. Placebo by Proxy in Neonatal Randomized Controlled Trials: Does It Matter? Children 2017, 4, 43. https://doi.org/10.3390/children4060043
Burkart TL, Kraus A, Koller B, Natalucci G, Latal B, Fauchère J-C, Bucher HU, Rüegger CM, For the Swiss EPO Neuroprotection Trial Group. Placebo by Proxy in Neonatal Randomized Controlled Trials: Does It Matter? Children. 2017; 4(6):43. https://doi.org/10.3390/children4060043
Chicago/Turabian StyleBurkart, Tiziana L., Andrea Kraus, Brigitte Koller, Giancarlo Natalucci, Beatrice Latal, Jean-Claude Fauchère, Hans Ulrich Bucher, Christoph M. Rüegger, and For the Swiss EPO Neuroprotection Trial Group. 2017. "Placebo by Proxy in Neonatal Randomized Controlled Trials: Does It Matter?" Children 4, no. 6: 43. https://doi.org/10.3390/children4060043
APA StyleBurkart, T. L., Kraus, A., Koller, B., Natalucci, G., Latal, B., Fauchère, J.-C., Bucher, H. U., Rüegger, C. M., & For the Swiss EPO Neuroprotection Trial Group. (2017). Placebo by Proxy in Neonatal Randomized Controlled Trials: Does It Matter? Children, 4(6), 43. https://doi.org/10.3390/children4060043





