Solitary Intra-Osseous Myofibroma of the Jaw: A Case Report and Review of Literature
Abstract
:1. Introduction
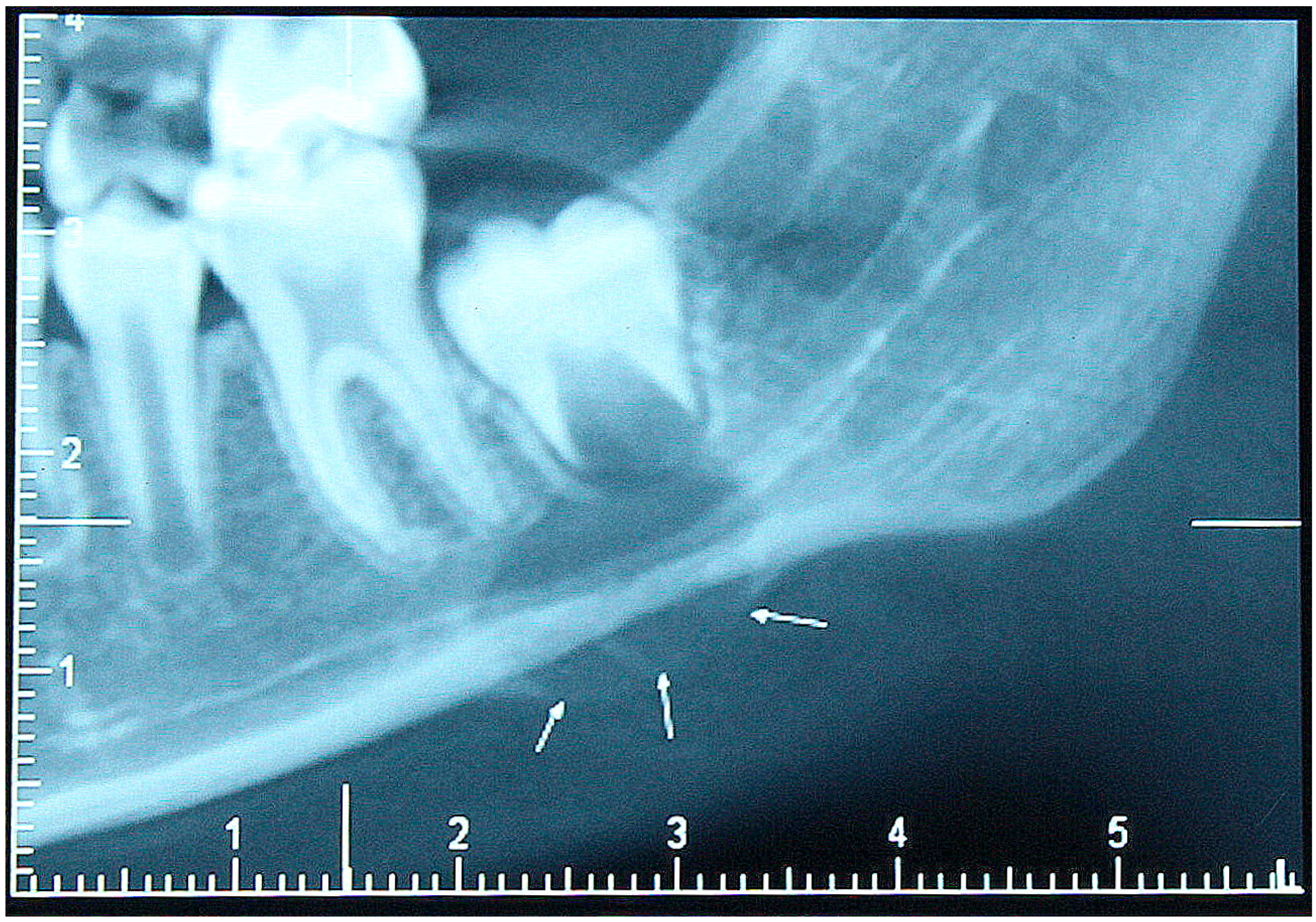
2. Patient Description
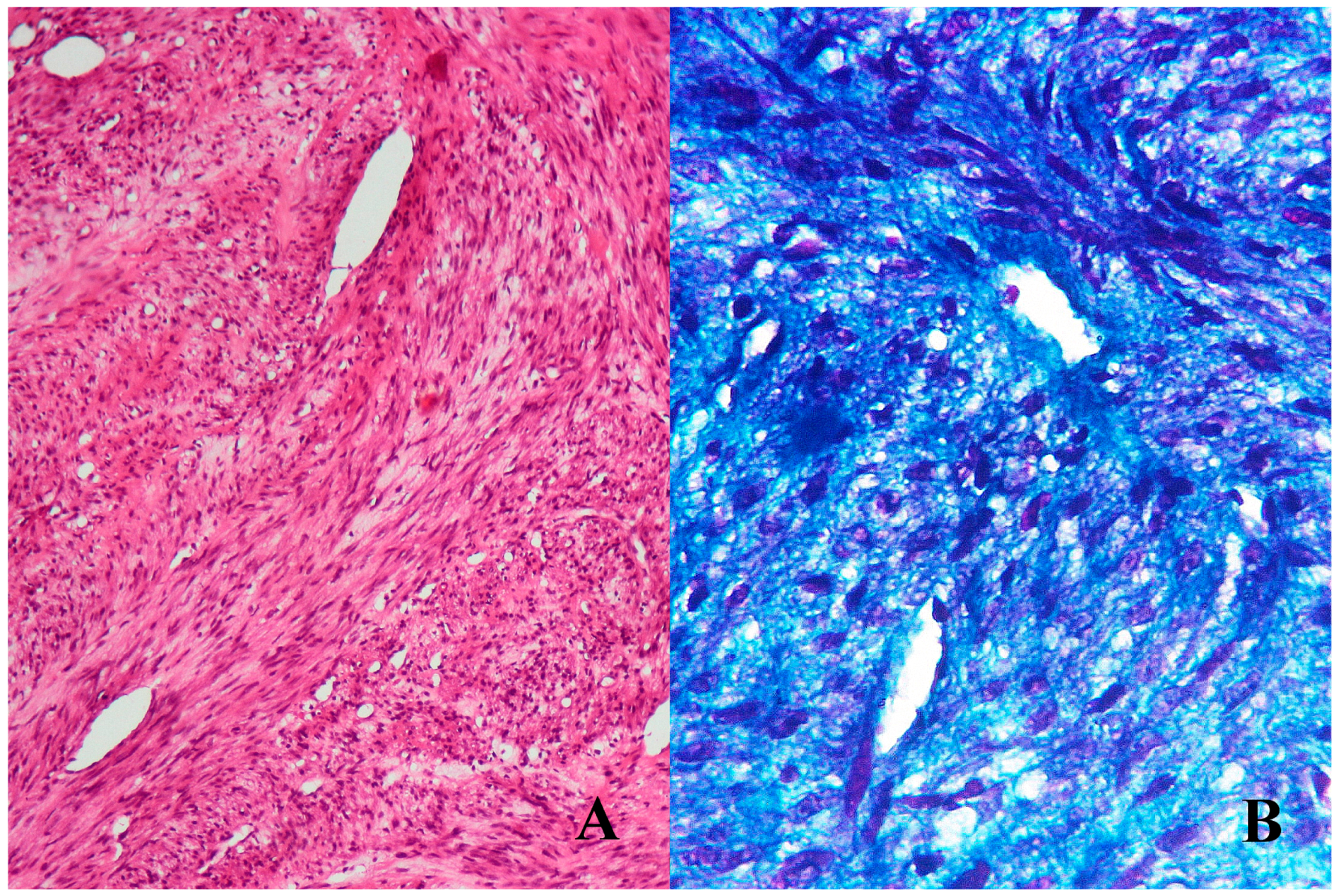
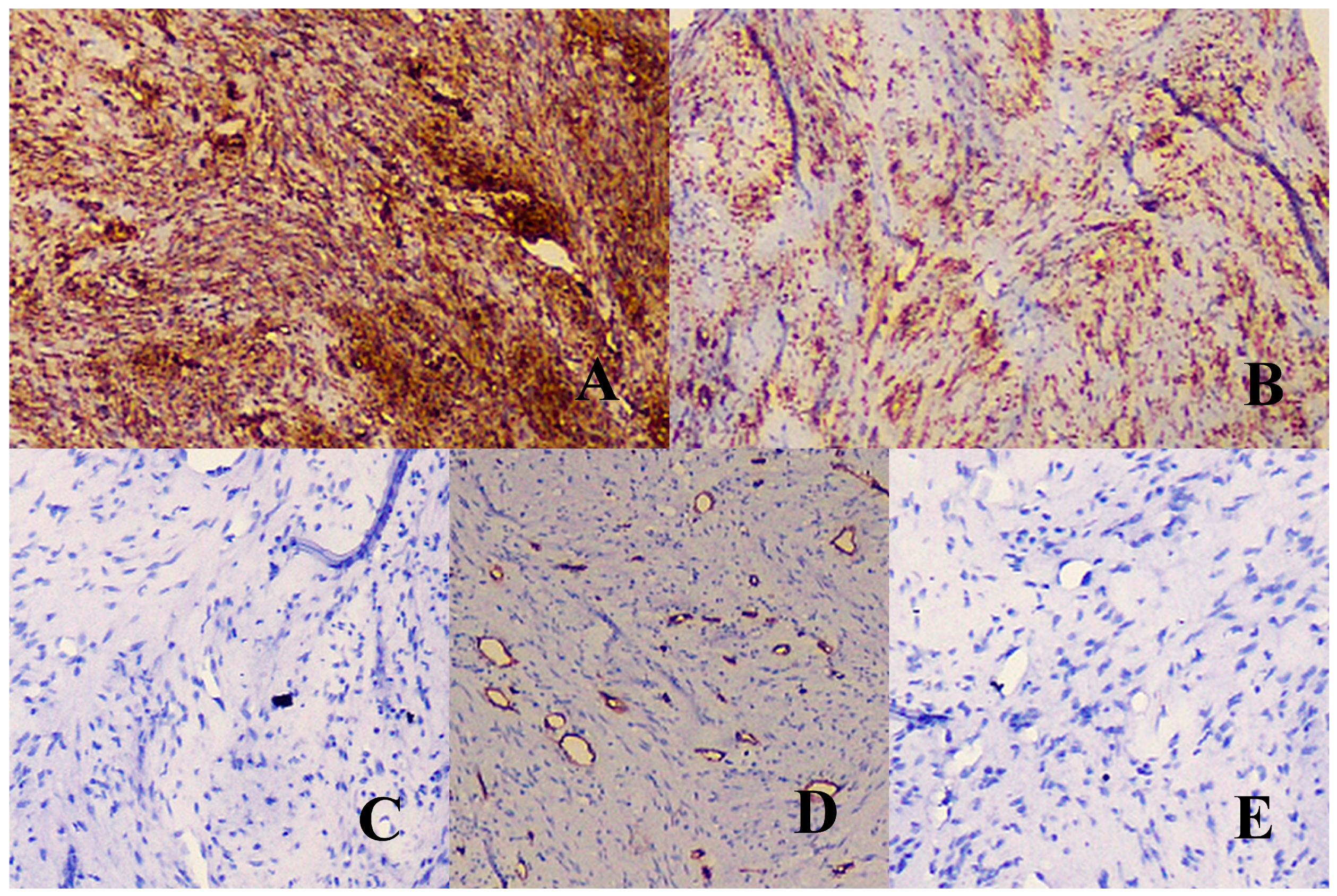
3. Discussion
Author Contributions
Conflicts of Interest
References
- Shemesh, S.; Kosashvili, Y.; Sidon, E.; Fichman, S.; Cohen, N.; Velkes, S. Solitary intraosseous myofibroma of the tibia in an adult patient: A case report. J. Bone Oncol. 2014, 3, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Zanella, T.A. Intraosseous myofibroma of the jaw—Systematic review. Investigação 2015, 14, 182–188. [Google Scholar]
- Gonzçlez, J.L.; Reyes, E.J.O.; Cuevas, G.J.C.; Rodrêguez, L.E.; Rodrêguez, C.A.A.; Garcêa, V.F.J.; Farfçn, M.J.E.; Vega-Memije, M.E. Intraosseous myofibroma of the mandible: A case report. Int. J. Odontostomat. 2013, 7, 339–342. [Google Scholar] [CrossRef]
- Ramadorai, A.; Rajsekaran, A.; Narayanan, V. A case report of solitary, intraosseous, adult-onset myofibroma of the mandible. J. Maxillofac. Oral Surg. 2010, 9, 280–283. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Son, S.M.; Kim, K.W.; Lee, O.J. Solitary myofibroma of the adult mandible: A case report and review of literature. Korean J. Pathol. 2014, 48, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Yamada, H.; Saito, T.; Nakaoka, K.; Kumagai, K.; Fujihara, H.; Mishima, K.; Hamada, Y. Solitary myofibroma of the mandible in an adult with magnetic resonance imaging and positron emission tomography findings: A case report. World J. Surg. Oncol. 2014, 12, 69. [Google Scholar] [CrossRef] [PubMed]
- Souza, D.P.; Loureiro, C.C.; Rejas, R.A.; Sousa, S.O.; Raitz, R. Intraosseousmyofibroma simulating an odontogenic lesion. J. Oral Sci. 2009, 51, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Chattaraj, M.; Gayen, S.; Chatterjee, R.P.; Shah, N.; Kundu, S. Solitary Myofibroma of the mandible in a six-year old-child: Diagnosis of a rare lesion. J. Clin. Diagn. Res. 2017, 11, 13–15. [Google Scholar] [CrossRef] [PubMed]
- Sundaravel, S.; Anuthama, K.; Prasad, H.; Sherlin, H.J.; Ilayaraja, V. Intraosseous myofibroma of mandible: A rarity of jaws: With clinical, radiological, histopathological and immunohistochemical features. J. Oral Maxillofac. Pathol. 2013, 17, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.J.; Skelton, H.G.; Barret, T.L.; Lupton, G.P.; Graham, J.H. Cutaneous myofibroma. Mod. Pathol. 1989, 2, 603–609. [Google Scholar] [PubMed]
- McMenamin, M.E.; Fletcher, C.D. Malignant myopericytoma: Expanding the spectrum of tumours with myopericytic differentiation. Histopathology 2002, 41, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, C.D.; Bridge, J.A.; Hogendoorn, P.; Mertens, F. WHO Classification of Tumours of Soft Tissue and Bone, 4th ed.; IARC Press: Lyon, France, 2013; pp. 119–120. [Google Scholar]
- Darby, I.; Skalli, O.; Gabbiani, G. Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound healing. Lab. Investig. 1990, 63, 21–29. [Google Scholar] [PubMed]
- Nadel, E.M. Histopathology of estrogen-induced tumors in guinea pigs. J. Natl. Cancer Inst. 1950, 10, 1043–1065. [Google Scholar] [PubMed]
- Kauffman, S.L.; Stout, A.P. Congenital mesenchymal tumors. Cancer 1965, 18, 460–476. [Google Scholar] [CrossRef]
- Narchi, H. Four half-siblings with infantile myrofibromatosis: A case for autosomal-recessive inheritance. Clin. Genet. 2001, 59, 134–135. [Google Scholar] [CrossRef] [PubMed]
- Zand, D.J.; Huff, D.; Everman, D.; Russell, K.; Saitta, S.; McDonald-McGinn, D.; Zackai, E.H. Autosomal dominant inheritance of infantile myofibromatosis. Am. J. Med. Genet. A 2004, 126, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, L.; Kindblom, L.G.; Meiskindblom, J.M. Adult myofibromatosis-reactive or neoplastic. Lab. Investig. 1995, 72, A8. [Google Scholar]
- Poon, C.K.; Kwan, P.C. Myofibroma of the mandible: A case report. Chin. J. Oral Maxillofac. Surg. 2005, 16, 156–165. [Google Scholar]
- Rai, B.; Ludusan, E.; McGovern, B.; Sharif, F. Mandibular swelling in a 5-year-old child–mandibular myofibroma. BMJ Case Rep. 2014. [Google Scholar] [CrossRef] [PubMed]
- Nirvikalpa, N.; Narayanan, V. Intraosseous infantile myofibroma of the mandible. Ann. Maxillofac. Surg. 2011, 1, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Sedghizadeh, P.P.; Allen, C.M.; Kalmar, J.R.; Miloro, M.; Suster, S. Solitary central myofibroma presenting in the gnathic region. Ann. Diagn. Pathol. 2004, 8, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Allon, I.; Vered, M.; Buchner, A.; Dayanm, D. Central (intraosseous)myofibroma of the mandible: Clinical, radiologic and histopathologic features of a rare lesion. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2007, 103, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Siegal, G.P.; Wei, S. Solitary, adult-onset, intraosseous myofibroma of the finger: Report of a case and review of literature. Hand (N. Y.) 2015, 10, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Narang, B.R.; Palaskar, S.J.; Bartake, A.R.; Pawar, R.B.; Rongte, S. Intraosseous neurofibroma of the mandible: A case report and review of literature. J. Clin. Diagn. Res. 2017, 11, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Gosau, M.; Draenert, F.G.; Winter, W.A.; Mueller-Hoecker, J.; Driemel, O. Fibrosarcoma of the childhood mandible. Head Face Med. 2008, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Divyambika, C.V.; Sathasivasubramanian, S.; Krithika, C.L.; Malathi, N.; Prathiba, D. Pediatric oral leiomyosarcoma: Rare case report. J. Can. Res. Ther. 2012, 8, 282–285. [Google Scholar] [CrossRef] [PubMed]
- Brunschweiler, B.; Guedj, N.; Lenoir, T.; Faillot, T.; Rillardon, L.; Guigui, P. Oncogenousosteomalacia and myopericytoma of the thoracic spine: A case report. J. Spine 2009, 4, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Heim-Hall, J.; Yohe, S.L. Application of Immunohistochemistry to Soft Tissue Neoplasms. Arch. Pathol. Lab. Med. 2008, 132, 476–489. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C. Immunohistochemistry in diagnosis of soft tissue tumours. Histopathology 2011, 58, 1001–1012. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, F.J.; Folpe, A.L.; Giannini, C.; Perry, A. Pathology of peripheral nerve sheath tumors: Diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012, 123, 295–319. [Google Scholar] [CrossRef] [PubMed]
- Pekmezci, M.; Reuss, D.E.; Hirbe, A.C.; Dahiya, S.; Gutmann, D.H.; Von Deimling, A.; Horvai, A.E.; Perry, A. Morphologic and immunohistochemical features of malignant peripheral nerve sheath tumors and cellular schwannomas. Mod. Pathol. 2015, 28, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Geramizadeh, B.; Marzban, M.; Churg, A. Role of Immunohistochemistry in the Diagnosis of Solitary Fibrous Tumor: A Review. Iran J. Pathol. 2016, 11, 195–203. [Google Scholar] [PubMed]
- Surbhi Metgud, R.; Naik, S.; Patel, S. Spindle cell lesions: A review on immunohistochemical markers. J. Can. Res. Ther. 2017, 13, 412–418. [Google Scholar] [CrossRef]
- Woods, T.R.; Cohen, D.M.; Islam, M.N.; Rawal, Y.; Bhattacharyya, I. Desmoplastic Fibroma of the Mandible: A Series of Three Cases and Review of Literature. Head Neck Pathol. 2015, 9, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Coffin, C.M.; Watterson, J.; Priest, J.R.; Dehner, L.P. Extrapulmonary inflammatory myofibroblastic tumor (inflammatory pseudotumor). A clinicopathologic and immunohistochemical study of 84 cases. Am. J. Surg. Pathol. 1995, 19, 859–872. [Google Scholar] [CrossRef] [PubMed]
- Price, E.B.; Silliphant, W.M.; Shuman, R. Nodular fasciitis: A clinicopathologic analysis of 65 cases. Am. J. Clin. Pathol. 1961, 35, 122–136. [Google Scholar] [CrossRef] [PubMed]
- Honeyman, J.N.; La Quaglia, M.P. Desmoid Tumors in the Pediatric Population. Cancers 2012, 4, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Datta, V.; Rawal, Y.B.; Mincer, H.H.; Anderson, M.K. Myopericytoma of the oral cavity. Head Neck 2007, 29, 605–608. [Google Scholar] [CrossRef] [PubMed]
- Mathew, N.K.; Zhang, K.Y.; Batstone, M.D. Myopericytoma of the coronoid process: A case report and review of the literature. Oral Maxillofac. Surg. Cases 2015, 1, 25–28. [Google Scholar] [CrossRef]
- Cai, Z.G.; Pan, C.C.; Yu, D.H.; Feng, Z.Z.; Ma, L.; Zhao, Y.; Xu, H.B. Myofibroblastic sarcomas: A clinicopathologic analysis of 15 cases and review of literature. Int. J. Clin. Exp. Pathol. 2016, 9, 1568–1577. [Google Scholar]
- Tandon, A.; Sethi, K.; Pratap Singh, A. Oral Rhabdomyosarcoma:A review. J. Clin. Exp. Dent. 2012, 4, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Troulis, M.J.; Williams, W.B.; Kaban, L.B. Staged protocol for resection, skeletal reconstruction, and oral rehabilitation of children with jaw tumors. J. Oral Maxillofac. Surg. 2004, 62, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Sugatani, T.; Inui, M.; Tagawa, T.; Seki, Y.; Mori, A.; Yoneda, J. Myofibroma of the mandible. Clinicopathological study and review of the literature. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1995, 80, 303–309. [Google Scholar] [CrossRef]
| Pathology | Microscopic Features | Immunohistochemistry |
|---|---|---|
| Benign Tumors | ||
| Intra-osseous myofibroma | Alternating growth pattern show fascicular and cellular areas characterized by polygonal cells at the center and elongated cells at the periphery. Absence of pleomorphism, nuclear atypia and mitosis. | α-SMA and vimentin positive. |
| Leiomyoma | Homogenous fascicular pattern made up of spindle cells with cigar shaped nucleus and bright eosinophilic cytoplasm. | Desmin, α-SMA, muscle specific actin (HHF-35), calponin positive. |
| Schwannoma | Cellular areas showing palisading growth pattern (Antoni A) intermixed with fibrillar unorganized cellular areas (Antoni B). | Diffuse S-100 podoplanin, calretinin, neurofibromin, CD34, glial fibrillary acidic protein (GFAP), collagen IV positive. Occasionally positive for cytokeratin (CK). |
| Neurofibroma | Spindle shaped cells with wavy nuclei showing fascicular or storiform growth pattern, at times myxoid areas are seen. | Expression of markers in descending order of immuno-reactivity S-100, CD34, SRY (sex determining region Y)-box 10 (Sox 10), collagen IV, calretinin, podoplanin, epithelial membrane antigen (EMA) positive. |
| Solitary fibrous tumor | No distinct cellular pattern of growth, however staghorn branching of blood vessels and perivascular hyalinization is seen. | CD-34, signal transducer and activator of transcription-6 (STAT-6), CD-99, B-cell CLL/lymphoma 2 (Bcl-2) (>85%) positive. Nuclear reactivity for β-catenin (22–67%). |
| Desmoplastic fibroma | Monomorphic fascicles of spindle cells admixed with abundant wavy collagen fibers. Absence of branching vasculature. | Vimentin (92%), β-catenin (50%) and occasional positivity for α-SMA. |
| Inflammatory myofibroblastic tumor | Three patterns of plump spindle cell arrangements have been described, along with an infiltrate of chronic inflammatory cells: spindle cells within a myxoid stroma, fascicular or storiform arrangement in a collagenous stroma and hypocellular elongated spindle cell component in a dense collagenous stroma. | Positive markers include: α-SMA, HHF-35, desmin (~50%), anaplastic lymphoma kinase protein (ALK-1) (30–60%), both anti-pan cytokeratin (AE1/AE3) and anticytokeratin for cytokeratin peptide 8 (CAM 5.2) (<35%) respectively. |
| Nodular fasciitis | Three patterns have been described: Type 1: Myxomatous vascular and acellular central stroma with plump fibroblasts at the periphery. Type 2: Cellular stroma with slit like vascular spaces. Type 3: Fibromatous stroma which contains interlacing fiber bundles, spindle shaped fibroblasts and capillaries. | Vimentin, HHF-35, α-SMA positive. |
| Benign fibrous histiocytoma | Fibroblast and histiocyte proliferation in a storiform pattern, with occasional multinucleated giant cells and foam cells. | Vimentin, CD-68, 1-antitrypsin, 1-antichymotrypsin positive. |
| Desmoid tumor | Mature spindle shaped cell proliferation separated by bundles of fibrous tissue. | Focal positivity for α-SMA, desmin and nuclear reactivity for β-catenin. |
| Myopericytoma | Oval or spindle shaped cells with a concentric peri-vascular arrangement. | α-SMA, smooth muscle myosin heavy chain, h-caldesmon, and calponin positive. |
| Malignant Tumors | ||
| Low-grade fibrosarcoma | Malignant fibroblasts show typical herring bone pattern, combined with high mitotic activity and nuclear atypia. | Vimentin positive. Variable positive expression of: α-SMA, HHF35, neuron-specific enolase, desmin, S-100, CD34, and CK. |
| Leiomyosarcoma | Palisaded pattern with densely packed spindle cells with fibrillar eosinophilic cytoplasm showing indistinct cell borders. Pleomorphism and high mitotic index is evident. | Diagnosis is based expression of any two of the following markers: α-SMA, desmin, HHF35, calponin. EMA and CK are occasionally positive (10–30%). |
| Myofibrosarcoma | Show varied patterns which include fibrosarcoma like areas. Nuclear pleomorphism is ubiquitous. | Vimentin (100%), Fibronectin (100%), α-SMA (~90%), HHF35 (~78%), calponin (67%), and desmin (~20%) positive. |
| Rhabdomyosarcoma | Ovoid or elongated rhabdomyoblasts with eosinophilic granular cytoplasm. Few blast cells show cross striations. Multinucleated tumor giant cells and abnormal mitotic figures are occasionally seen in a myxoid stroma. | Nuclear reactivity for myogenin and myogenic differentiation 1 (MyoD1). Diffuse desmin and rare α-SMA positive (~10%). |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhupar, A.; Carvalho, K.; Sawant, P.; Spadigam, A.; Syed, S. Solitary Intra-Osseous Myofibroma of the Jaw: A Case Report and Review of Literature. Children 2017, 4, 91. https://doi.org/10.3390/children4100091
Dhupar A, Carvalho K, Sawant P, Spadigam A, Syed S. Solitary Intra-Osseous Myofibroma of the Jaw: A Case Report and Review of Literature. Children. 2017; 4(10):91. https://doi.org/10.3390/children4100091
Chicago/Turabian StyleDhupar, Anita, Karla Carvalho, Poonam Sawant, Anita Spadigam, and Shaheen Syed. 2017. "Solitary Intra-Osseous Myofibroma of the Jaw: A Case Report and Review of Literature" Children 4, no. 10: 91. https://doi.org/10.3390/children4100091
APA StyleDhupar, A., Carvalho, K., Sawant, P., Spadigam, A., & Syed, S. (2017). Solitary Intra-Osseous Myofibroma of the Jaw: A Case Report and Review of Literature. Children, 4(10), 91. https://doi.org/10.3390/children4100091





