Microbiological and Clinical Characteristics of Pediatric Sepsis Patients with and without Septic Shock: A Retrospective Study at a Tertiary Pediatric Hospital in China
Abstract
Highlights
- Pediatric septic shock patients exhibited significantly higher mortality (17.4% vs. 4.7%, p < 0.001) and prolonged hospitalization (14.8 vs. 12.0 days, p = 0.003) compared to non-septic-shock sepsis patients.
- Elevated lactate (OR = 1.49, 95% CI = 1.29–1.75) and pulmonary infection (OR = 2.25, 95% CI = 1.35–3.73) were identified as independent risk factors for septic shock progression.
- Early monitoring of lactate levels and pulmonary infection status could improve risk stratification for septic shock in pediatric sepsis, enabling timely interventions.
- Antibiotic stewardship optimization is critical, as combination antibiotic therapy was frequently used in viral (79%) and fungal (86.5%) sepsis despite limited efficacy, prolonging hospitalization in fungal cases (20.74 vs. 12.97 days, p = 0.017).
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Microbiological Analysis
2.3. Statistical Analysis
3. Results
3.1. Study Population
3.2. Comparison of Baseline Characteristics in Patients with and without Septic Shock
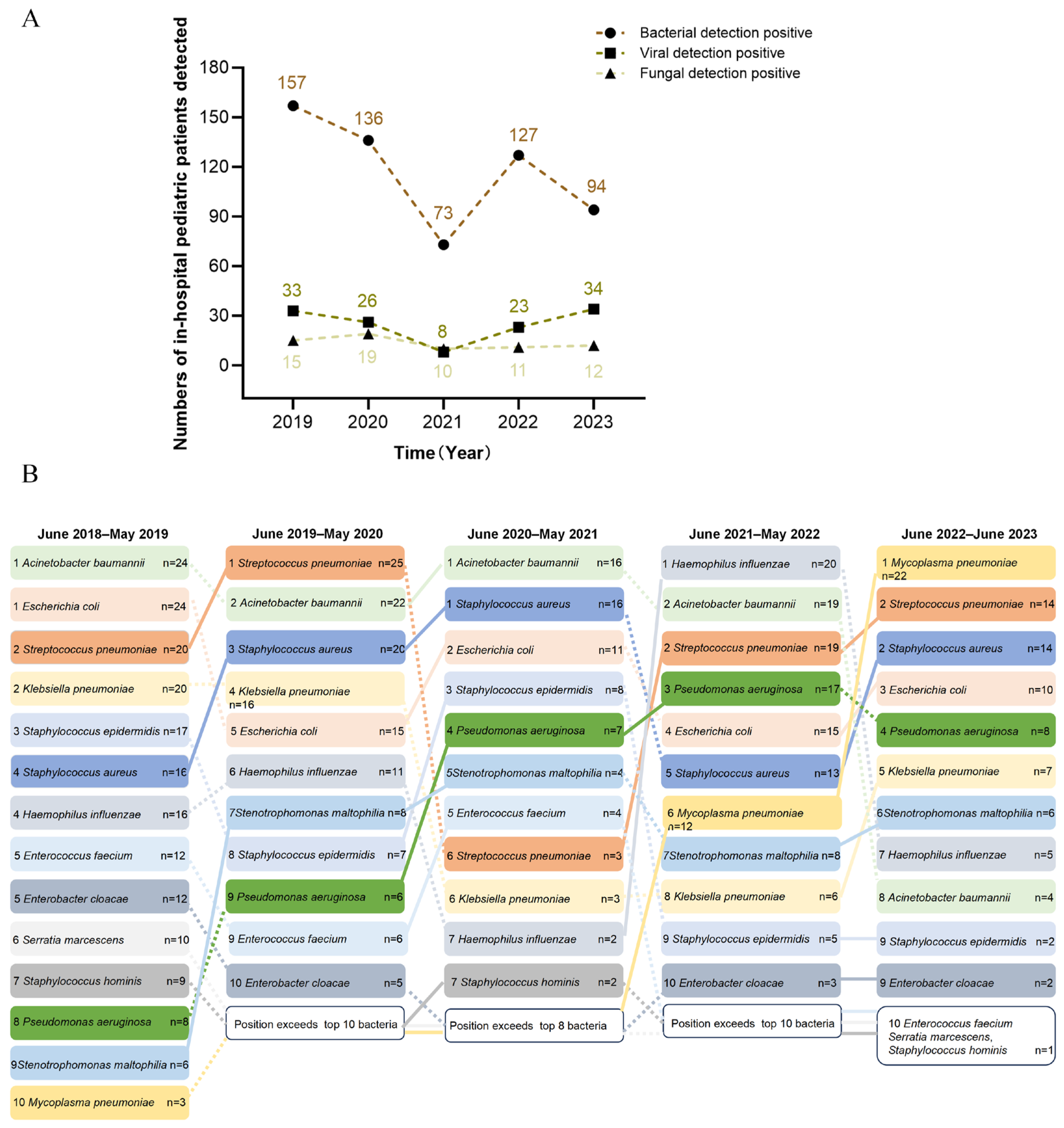
3.3. Epidemiological Trends of Pathogens in Pediatric Sepsis
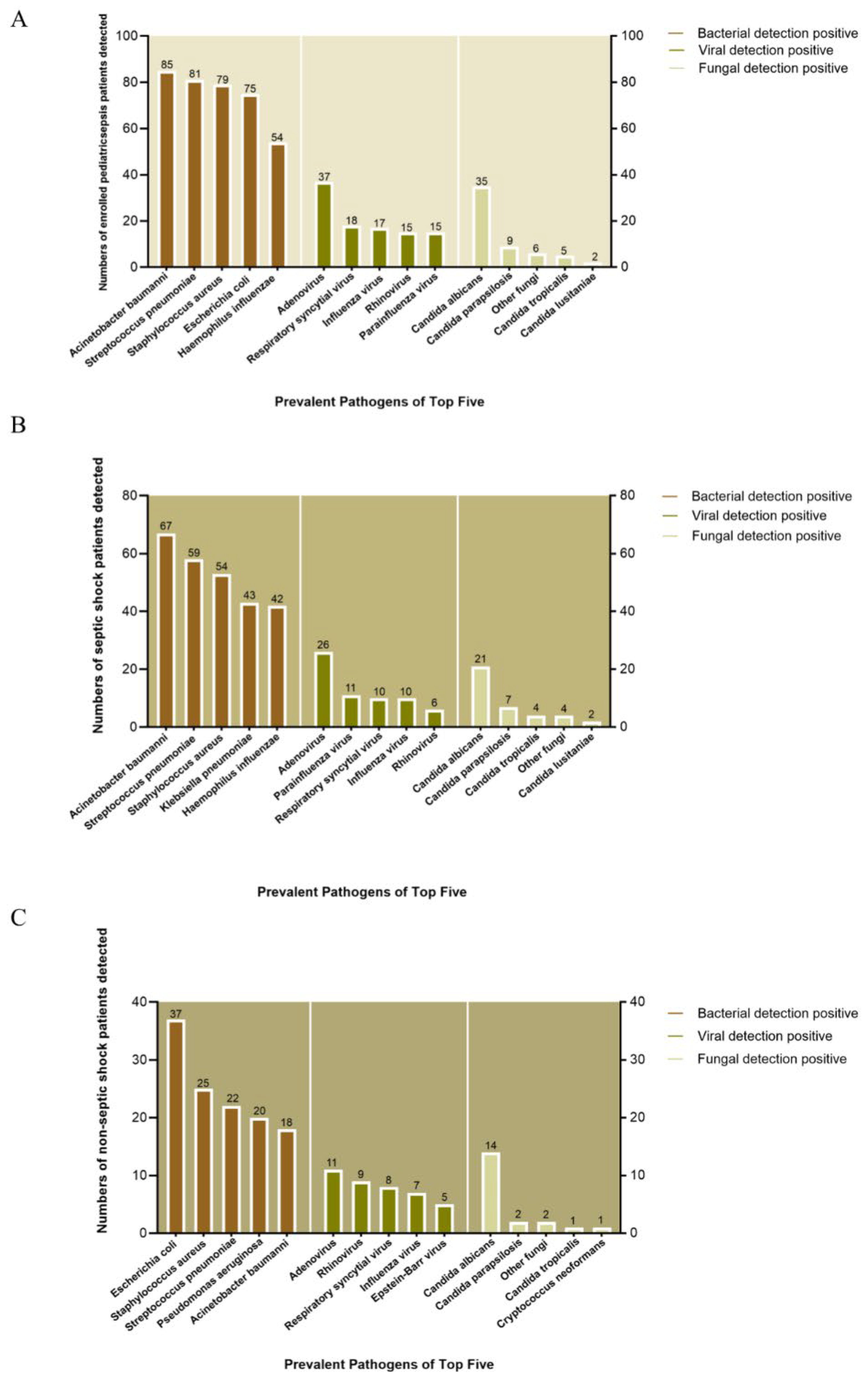
3.4. Prevalent Pathogens Between Children with and without Septic Shock
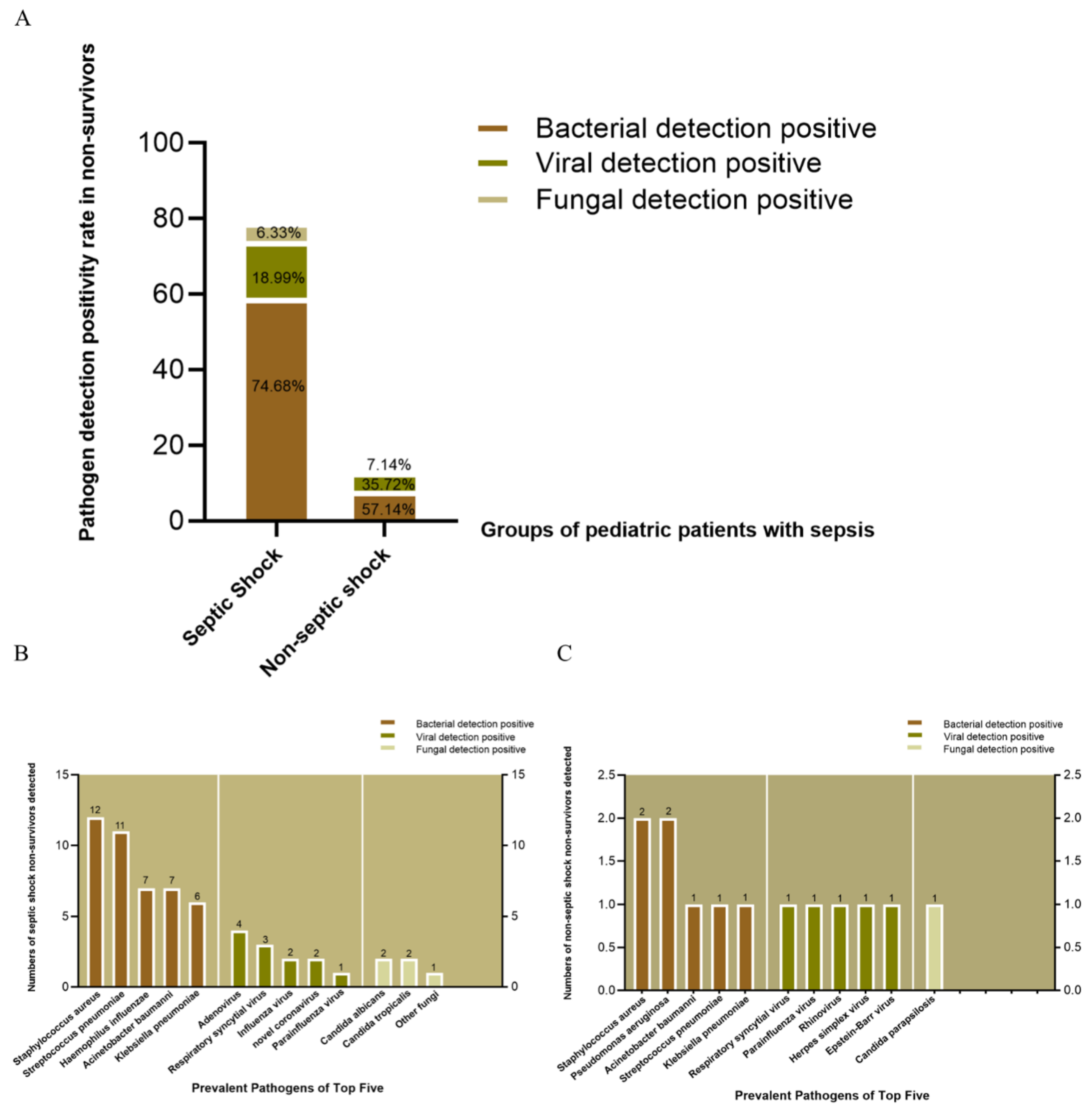
3.5. Prevalent Pathogens in Non-Survivors with and without Septic Shock
3.6. Antibiotic Utilization in Viral and Fungal Infection Between Patients in the Septic Shock and Non-Septic-Shock Groups
3.7. Univariate and Multivariate Analysis of Risk Factors Associated with Septic Shock
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| OR | Odds ratio |
| CI | Confidence interval |
| A. baumannii | Acinetobacter baumannii |
| E. coli | Escherichia coli |
| S. aureus | Staphylococcus aureus |
| S. pneumoniae | Streptococcus pneumoniae |
| P. aeruginosa | Pseudomonas aeruginosa |
| M. pneumoniae | Mycoplasma pneumoniae |
| K. pneumoniae | Klebsiella pneumoniae |
| H. influenzae | Haemophilus influenzae |
| NLR | Neutrophil-to-lymphocyte ratio |
| RSV | Respiratory syncytial virus |
| PLT | Platelet count |
| PLT/MPV | Platelet-to-mean platelet volume ratio |
| RDW | Red cell distribution width |
| L/A | Lactate-to-albumin ratio |
References
- Carlton, E.F.; Barbaro, R.P.; Iwashyna, T.; Prescott, H.C. Cost of Pediatric Severe Sepsis Hospitalizations. JAMA Pediatr. 2019, 173, 986–987. [Google Scholar] [CrossRef]
- Tan, B.; Wong, J.J.-M.; Sultana, R.; Koh, J.C.J.W.; Jit, M.; Mok, Y.H.; Lee, J.H. Global Case-Fatality Rates in Pediatric Severe Sepsis and Septic Shock: A Systematic Review and Meta-analysis. JAMA Pediatr. 2019, 173, 352–362. [Google Scholar] [CrossRef]
- Kyösti, E.; Ala-Kokko, T.I.; Ohtonen, P.; Peltoniemi, O.; Rautiainen, P.; Kataja, J.; Ebeling, H.; Liisanantti, J.H. Factors associated with health-related quality of life 6 years after ICU discharge in a Finnish paediatric population: A cohort study. Intensive Care Med. 2018, 44, 1378–1387. [Google Scholar] [CrossRef]
- Zimmerman, J.J.; Banks, R.; Berg, R.A.; Zuppa, A.; Newth, C.J.; Wessel, D.; Pollack, M.M.; Meert, K.L.; Hall, M.W.; Quasney, M.; et al. Critical Illness Factors Associated with Long-Term Mortality and Health-Related Quality of Life Morbidity Following Community-Acquired Pediatric Septic Shock. Crit. Care Med. 2020, 48, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Carlton, E.F.; Gebremariam, A.; Maddux, A.B.; McNamara, N.; Barbaro, R.P.; Cornell, T.T.; Iwashyna, T.J.; Prosser, L.A.; Zimmerman, J.; Weiss, S.; et al. New and Progressive Medical Conditions After Pediatric Sepsis Hospitalization Requiring Critical Care. JAMA Pediatr. 2022, 176, e223554. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.; Giroir, B.; Randolph, A. International pediatric sepsis consensus conference: Definitions for sepsis and organ dysfunction in pediatrics. Pediatr. Crit. Care Med. 2005, 6, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Gebara, B.M. Values for systolic blood pressure. Pediatr. Crit. Care Med. 2005, 6, 500. [Google Scholar] [CrossRef]
- Levy, M.M.; Fink, M.P.; Marshall, J.C.; Abraham, E.; Angus, D.; Cook, D.; Cohen, J.; Opal, S.M.; Vincent, J.-L.; Ramsay, G. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit. Care Med. 2003, 31, 1250–1256. [Google Scholar] [CrossRef]
- Seymour, C.W.; Liu, V.X.; Iwashyna, T.J.; Brunkhorst, F.M.; Rea, T.D.; Scherag, A.; Rubenfeld, G.; Kahn, J.M.; Shankar-Hari, M.; Singer, M.; et al. Assessment of Clinical Criteria for Sepsis: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 762–774. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Shankar-Hari, M.; Phillips, G.S.; Levy, M.L.; Seymour, C.W.; Liu, V.X.; Deutschman, C.S.; Angus, D.C.; Rubenfeld, G.D.; Singer, M.; for the Sepsis Definitions Task Force. Developing a New Definition and Assessing New Clinical Criteria for Septic Shock: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 775–787. [Google Scholar] [CrossRef]
- Schlapbach, L.J.; Watson, R.S.; Sorce, L.R.; Argent, A.C.; Menon, K.; Hall, M.W.; Akech, S.; Albers, D.J.; Alpern, E.R.; Balamuth, F.; et al. International Consensus Criteria for Pediatric Sepsis and Septic Shock. JAMA 2024, 331, 665–674. [Google Scholar] [CrossRef]
- Lu, Q.; Zhou, M.; Tu, Y.; Yao, Y.; Yu, J.; Cheng, S. Pathogen and antimicrobial resistance profiles of culture-proven neonatal sepsis in Southwest China, 1990–2014. J. Paediatr. Child Health 2016, 52, 939–943. [Google Scholar] [CrossRef]
- Schlapbach, L.J.; MacLaren, G.; Festa, M.; Alexander, J.; Erickson, S.; Beca, J.; Slater, A.; Schibler, A.; Pilcher, D.; Millar, J.; et al. Prediction of pediatric sepsis mortality within 1 h of intensive care admission. Intensive Care Med. 2017, 43, 1085–1096. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.L.; Fitzgerald, J.C.; Pappachan, J.; Wheeler, D.; Jaramillo-Bustamante, J.C.; Salloo, A.; Singhi, S.C.; Erickson, S.; Roy, J.A.; Bush, J.L.; et al. Global epidemiology of pediatric severe sepsis: The sepsis prevalence, outcomes, and therapies study. Am. J. Respir. Crit. Care Med. 2015, 191, 1147–1157. [Google Scholar] [CrossRef] [PubMed]
- Halasa, N.B.; Spieker, A.J.; Young, C.C.; Olson, S.M.; Newhams, M.M.; Amarin, J.Z.; Moffitt, K.L.; Nakamura, M.M.; Levy, E.R.; Soma, V.L.; et al. Life-Threatening Complications of Influenza vs Coronavirus Disease 2019 (COVID-19) in US Children. Clin. Infect. Dis. 2023, 76, e280–e290. [Google Scholar] [CrossRef] [PubMed]
- Rice, T.W.M.; Rubinson, L.; Uyeki, T.M.M.; Vaughn, F.L.; John, B.B.P.; Miller, R.R.I.; Higgs, E.M.; Randolph, A.G.M.; Smoot, B.E.; Thompson, B.T.M. Critical illness from 2009 pandemic influenza A virus and bacterial coinfection in the United States. Crit. Care Med. 2012, 40, 1487–1498. [Google Scholar] [CrossRef]
- Houck, P.M.; Bratzler, D.W.; Nsa, W.; Ma, A.; Bartlett, J.G. Timing of antibiotic administration and outcomes for Medicare patients hospitalized with community-acquired pneumonia. Arch. Intern. Med. 2004, 164, 637–644. [Google Scholar] [CrossRef]
- Kumar, A.; Roberts, D.; Wood, K.E.; Light, B.; Parrillo, J.E.; Sharma, S.; Suppes, R.; Feinstein, D.; Zanotti, S.; Taiberg, L.; et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit. Care Med. 2006, 34, 1589–1596. [Google Scholar] [CrossRef]
- Ferreira, F.L.; Bota, D.P.; Bross, A.; Mélot, C.; Vincent, J.-L. Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 2001, 286, 1754–1758. [Google Scholar] [CrossRef]
- Cullen, D.J.; Chernow, B. Predicting outcome in critically ill patients. Crit. Care Med. 1994, 22, 1345–1348. [Google Scholar] [CrossRef]
- Shortell, S.M.; Zimmerman, J.E.; Rousseau, D.M.; Gillies, R.R.; Wagner, D.P.; Draper, E.A.; Kanus, W.A.; Duffy, J. The performance of intensive care units: Does good management make a difference? Med. Care 1994, 32, 508–525. [Google Scholar] [CrossRef]
- Burgunder, L.; Heyrend, C.; Olson, J.; Stidham, C.; Lane, R.D.; Workman, J.K.; Larsen, G.Y. Medication and Fluid Management of Pediatric Sepsis and Septic Shock. Pediatr. Drugs 2022, 24, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Fritz, C.Q.; Edwards, K.M.; Self, W.H.; Grijalva, C.G.; Zhu, Y.; Arnold, S.R.; McCullers, J.A.; Ampofo, K.; Pavia, A.T.; Wunderink, R.G.; et al. Prevalence, Risk Factors, and Outcomes of Bacteremic Pneumonia in Children. Pediatrics 2019, 144, e20183090. [Google Scholar] [CrossRef] [PubMed]
- Bard, J.D.; TeKippe, E.M.; Kraft, C.S. Diagnosis of Bloodstream Infections in Children. J. Clin. Microbiol. 2016, 54, 31418–31424. [Google Scholar] [CrossRef] [PubMed]
- Salud, D.; Reeder, R.W.; Banks, R.K.; Meert, K.L.; Berg, R.A.; Zuppa, A.; Newth, C.J.; Hall, M.W.; Quasney, M.; Sapru, A.; et al. Association of Pathogen Type with Outcomes of Children Encountering Community-Acquired Pediatric Septic Shock. Pediatr. Crit. Care Med. 2022, 23, 635–645. [Google Scholar] [CrossRef]
- Liu, R.M.; Yu, Z.; Xiao, C.; Xu, F.; Xiao, S.; He, J.B.; Shi, Y.; Hua, Y.; Zhou, J.B.; Zhang, G.; et al. Epidemiology and Clinical Characteristics of Pediatric Sepsis in PICUs in Southwest China: A Prospective Multicenter Study. Pediatr. Crit. Care Med. 2024, 25, 425–433. [Google Scholar] [CrossRef]
- Schlapbach, L.J.; Straney, L.; Alexander, J.; MacLaren, G.; Festa, M.; Schibler, A.; Slater, A. Mortality related to invasive infections, sepsis, and septic shock in critically ill children in Australia and New Zealand, 2002-13: A multicentre retrospective cohort study. Lancet Infect. Dis. 2015, 15, 46–54. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Evidence Review for Managing and Treating Suspected Sepsis in Acute Hospital Settings; Antibiotic Treatment in People with Suspected Sepsis; Suspected Sepsis: Recognition, Diagnosis and Early Management; National Institute for Health and Care Excellence (NICE): London, UK, 2024. [Google Scholar]
- Shadvar, K.; Nader-Djalal, N.; Vahed, N.; Sanaie, S.; Iranpour, A.; Mahmoodpoor, A.; Vahedian-Azimi, A.; Samim, A.; Rahimi-Bashar, F. Comparison of lactate/albumin ratio to lactate and lactate clearance for predicting outcomes in patients with septic shock admitted to intensive care unit: An observational study. Sci. Rep. 2022, 12, 13047. [Google Scholar] [CrossRef]
- Park, H.; Lee, J.; Oh, D.K.; Park, M.H.; Lim, C.-M.; Lee, S.-M.; Lee, H.Y. Serial evaluation of the serum lactate level with the SOFA score to predict mortality in patients with sepsis. Sci. Rep. 2023, 13, 6351. [Google Scholar] [CrossRef]
- He, X.-L.; Liao, X.-L.; Xie, Z.-C.; Han, L.; Yang, X.-L.; Kang, Y. Pulmonary Infection Is an Independent Risk Factor for Long-Term Mortality and Quality of Life for Sepsis Patients. BioMed Res. Int. 2016, 2016, 4213712. [Google Scholar] [CrossRef]
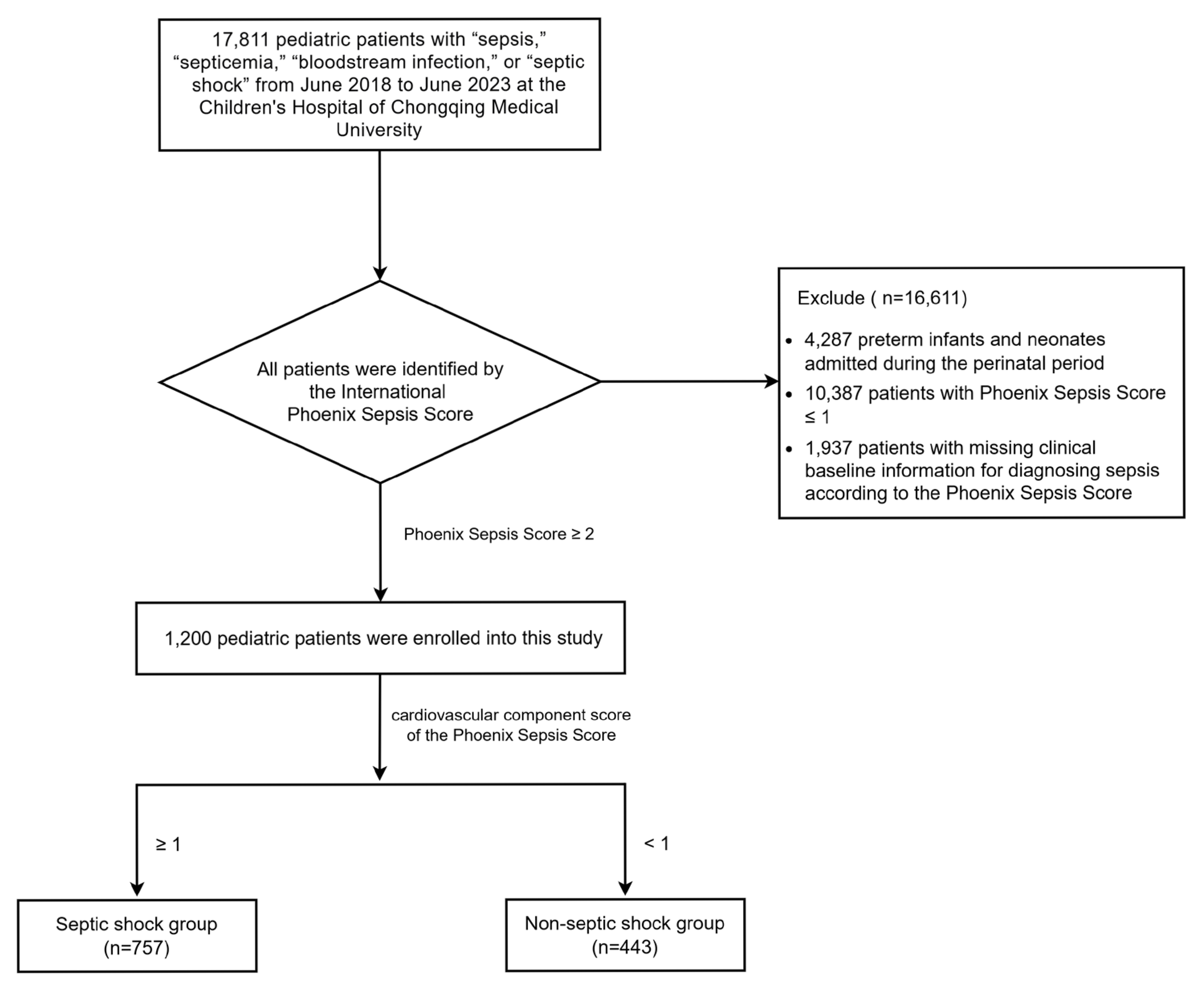
| Characteristics | Enrolled Children with Sepsis | Septic Shock Group (n = 757) | Non-Septic-Shock Group (n = 443) | Z/(X2) | p Value | |
|---|---|---|---|---|---|---|
| Gender (n,%) | (1.972) | 0.160 | ||||
| Male | 670(55.8%) | 411(54.3%) | 259(58.5%) | |||
| Female | 530(44.2%) | 346(45.7%) | 184(41.5%) | |||
| Age (n,%) | (18.792) | 0.002 | ||||
| <1 month | 146(12.2%) | 113(14.9%) | 33(7.4%) | (14.624) | <0.001 | |
| 1–11 months | 305(25.4%) | 182(24.0%) | 123(27.8%) | |||
| 1–2 years | 152(12.7%) | 90(11.9%) | 62(14.0%) | |||
| 2–5 years | 244(20.3%) | 147(19.4%) | 97(21.9%) | |||
| 5–12 years | 255(21.2%) | 169(22.4%) | 86(19.4%) | |||
| 12–18 years | 98(8.2%) | 56(7.4%) | 42(9.5%) | |||
| Site of pathogen detection (n,%) | (39.685) | <0.001 | ||||
| Lower respiratory tract | 371(47.3%) | 278(52.2%) | 93(36.9%) | (15.971) | <0.001 | |
| Blood | 204(26.0%) | 141(26.5%) | 63(25.0%) | (0.188) | 0.665 | |
| Skin/wound discharge | 104(13.2%) | 53(9.9%) | 51(20.2%) | (15.777) | <0.001 | |
| Catheter | 37(4.7%) | 27(5.1%) | 10(4.0%) | (0.459) | 0.498 | |
| Pleural/abdominal effusion | 22(2.8%) | 14(2.6%) | 8(3.2%) | (0.189) | 0.664 | |
| Cerebrospinal fluid | 10(1.3%) | 2(0.4%) | 8(3.2%) | (10.661) | 0.001 | |
| Other | 37(4.7%) | 18(3.3%) | 19(7.5%) | (6.601) | 0.010 | |
| Types of bacterial detection | (9.578) | 0.023 | ||||
| No bacteria detection | 613(51.1%) | 362(47.8%) | 251(56.7%) | (8.737) | 0.003 | |
| Single bacterial detection positive | 442(36.8%) | 294(38.8%) | 148(33.4%) | (3.540) | 0.060 | |
| Dual bacterial detection positive | 100(8.3%) | 68(9.0%) | 32(7.2%) | (1.132) | 0.287 | |
| Polybacterial detection positive | 45(3.8%) | 33(4.4%) | 12(2.7%) | (2.109) | 0.146 | |
| Types of viral detection | (2.250) | 0.522 | ||||
| No virus detected | 1076(89.7%) | 683(90.2%) | 393(88.7%) | (0.689) | 0.407 | |
| Single viral detection positive | 111(9.2%) | 67(8.9%) | 44(10.0%) | (0.389) | 0.533 | |
| Dual viral detection positive | 12(1.0%) | 7(0.9%) | 5(1.1%) | (0.117) | 0.732 | |
| Polyviral detection positive | 1(0.1%) | 0 | 1(0.2%) | (1.710) | 0.191 | |
| Types of fungal detection | (1.521) | 0.217 | ||||
| No fungus detected | 1133(94.4%) | 710(93.8%) | 423(95.5%) | |||
| Fungal detection positive | 67(5.6%) | 47(6.2%) | 20(4.5%) | |||
| Types of pathogen co-detection | (5.600) | 0.133 | ||||
| Bacterial plus viral detection positive | 20(40.0%) | 16(47.1%) | 4(25.0%) | (2.206) | 0.137 | |
| Bacterial plus fungal detection positive | 26(52.0%) | 14(41.2%) | 12(75.0%) | (4.987) | 0.026 | |
| Viral plus fungal detection positive | 1(2.0%) | 1(2.9%) | 0 | (0.480) | 0.488 | |
| Multi-pathogen detection positive | 3(6.0%) | 3(8.8%) | 0 | (1.502) | 0.220 | |
| Pulmonary infection (n,%) | (85.243) | <0.001 | ||||
| Yes | 815(67.9%) | 586(77.4%) | 229(51.7%) | |||
| No | 189(15.8%) | 87(11.5%) | 102(23.0%) | |||
| Unknown | 196(16.3%) | 84(11.1%) | 112(25.3%) | |||
| Ventilator support (n,%) | (68.149) | <0.001 | ||||
| Yes | 507(42.3%) | 388(51.3%) | 119(26.9%) | |||
| No | 693(57.7%) | 369(48.7%) | 324(73.1%) | |||
| WBC[×109/L,M(P25,P75)] | 11.88(6.64,18.02) | 11.8(6.68,17.82) | 12.22(6.46,18.24) | –0.010 | 0.992 | |
| PLT[×109/L,M(P25,P75)] | 226(108.5,371) | 254(143,392.5) | 163(68.75,311.25) | 7.066 | <0.001 | |
| PLT/MPV[×109/(L×fL),M(P25,P75)] | 25.09(14.17,38.72) | 26.57(16.05,40.02) | 22.79(9.34,34.93) | 4.053 | <0.001 | |
| NLR/M(P25,P75) | 2.81(1.36,6.16) | 2.59(1.35,5.45) | 3.38(1.39,7.56) | –2.839 | 0.005 | |
| PDW/M(P25,P75) | 12.1(10.5,14.6) | 12(10.5,14.4) | 12.2(10.6,15.1) | –1.213 | 0.225 | |
| RDW/M(P25,P75) | 14.9(13.5,19.8) | 15.1(13.6,26.5) | 14.5(13.4,17.38) | 3.186 | 0.001 | |
| PT/M(P25,P75) | 1.17(1.06,1.43) | 1.16(1.04,1.38) | 1.33(1.12,1.47) | –6.041 | <0.001 | |
| D-dimer/M(P25,P75) | 4.03(2.17,9.70) | 3.61(1.82,9.21) | 4.78(2.62,11.08) | –4.963 | <0.001 | |
| Lactic Acid/[mmol/L,M(P25,P75)] | 1.8(1.1,3.58) | 2(1.2,4.4) | 1.41(0.9,2.4) | 6.797 | <0.001 | |
| In-hospital stay/[d,M(P25,P75)] | 13.65(7.59,23.76) | 14.8(7.6,27.3) | 12.0(7.6,19.4) | 2.982 | 0.003 | |
| In-hospital mortality (n,%) | –40.498 | <0.001 | ||||
| Yes | 153(12.8%) | 132(17.4%) | 21(4.7%) | |||
| No | 1047(87.2%) | 625(82.6%) | 422(95.3%) | |||
| Characteristics | Bacterial Sepsis Patients (n = 552) | Viral Sepsis Patients (n = 100) | Fungal Sepsis Patients (n = 37) | p Value |
|---|---|---|---|---|
| Types of antibiotic medication (n,%) | 0.321 | |||
| Monotherapy | 38(6.8%) | 21(21%) | 5(13.5%) | |
| Combination therapy | 514(93.2%) | 79(79%) | 32(86.5%) | |
| Antibiotic medication regimen (n,%) | 0.023 | |||
| β-lactam antibiotics | 265(48%) | 67(67%) | 16(43.2%) | |
| β-lactam antibiotics plus macrolide antibiotics | 47(8.5%) | 4(4%) | 3(8.1%) | |
| β-lactam antibiotics plus quinolone antibiotics | 41(7.4%) | 1(1%) | 1(2.7%) | |
| β-lactam antibiotics plus vancomycin | 152(27.5%) | 20(20%) | 7(18.9%) | |
| β-lactam antibiotics plus macrolide antibiotics plus other antibiotics | 1(0.2%) | 1(1%) | 4(10.8%) | |
| β-lactam antibiotics plus vancomycin plus other antibiotics | 46(8.4%) | 6(6%) | 6(16.3%) | |
| Only vancomycin | 0 | 1(1%) | 0 | |
| Ventilator support (n,%) | 0.341 | |||
| Yes | 274(49.6%) | 37(37%) | 17(45.9%) | |
| No | 278(50.4%) | 63(63%) | 20(54.1%) | |
| In-hospital stay/[d,M(P25,P75)] | 16.07(9.40,29.94) | 12.97(6.38,19.52) | 20.74(11.37,29.46) | 0.017 |
| In-hospital mortality (n,%) | 0.235 | |||
| Yes | 64(11.6%) | 16(16%) | 3(8.1%) | |
| No | 488(88.4%) | 84(84%) | 34(91.9%) |
| Variables | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| OR | 95%CI | p Value | OR | 95%CI | p Value | |
| Bacterial detection positive | 1.426 | 1.127–1.807 | 0.003 | 1.152 | 0.771–1.724 | 0.489 |
| Viral detection positive | 0.852 | 0.584–1.250 | 0.407 | |||
| Fungal detection positive | 1.400 | 0.830–2.445 | 0.219 | |||
| Age (<1month, ≥1month) | 2.180 | 1.467–3.321 | <0.001 | 2.062 | 1.001–4.498 | 0.058 |
| PLT | 1.002 | 1.001–1.003 | <0.001 | 0.999 | 0.992–1.006 | 0.824 |
| PLT/MPV | 1.015 | 1.007–1.023 | <0.001 | 1.019 | 0.953–1.089 | 0.588 |
| PT | 0.936 | 0.802–1.095 | 0.399 | |||
| D-dimer | 0.998 | 0.989–1.007 | 0.594 | |||
| NLR | 0.984 | 0.968–1.001 | 0.062 | |||
| RDW | 1.016 | 1.007–1.026 | <0.001 | 1.01 | 0.993–1.029 | 0.249 |
| Lactic acid | 1.408 | 1.286–1.557 | <0.001 | 1.487 | 1.294–1.751 | <0.001 |
| Pulmonary infection | 3.000 | 2.171–4.156 | <0.001 | 2.246 | 1.351–3.734 | 0.002 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, K.-C.; Li, Q.-Y.; Chen, L.; Zhao, Y.; Liu, H.; Kong, Z.-X.; Luo, Z.-X. Microbiological and Clinical Characteristics of Pediatric Sepsis Patients with and without Septic Shock: A Retrospective Study at a Tertiary Pediatric Hospital in China. Children 2025, 12, 1146. https://doi.org/10.3390/children12091146
Peng K-C, Li Q-Y, Chen L, Zhao Y, Liu H, Kong Z-X, Luo Z-X. Microbiological and Clinical Characteristics of Pediatric Sepsis Patients with and without Septic Shock: A Retrospective Study at a Tertiary Pediatric Hospital in China. Children. 2025; 12(9):1146. https://doi.org/10.3390/children12091146
Chicago/Turabian StylePeng, Kai-Cheng, Qin-Yuan Li, Lin Chen, Yan Zhao, Hui Liu, Zhen-Xuan Kong, and Zheng-Xiu Luo. 2025. "Microbiological and Clinical Characteristics of Pediatric Sepsis Patients with and without Septic Shock: A Retrospective Study at a Tertiary Pediatric Hospital in China" Children 12, no. 9: 1146. https://doi.org/10.3390/children12091146
APA StylePeng, K.-C., Li, Q.-Y., Chen, L., Zhao, Y., Liu, H., Kong, Z.-X., & Luo, Z.-X. (2025). Microbiological and Clinical Characteristics of Pediatric Sepsis Patients with and without Septic Shock: A Retrospective Study at a Tertiary Pediatric Hospital in China. Children, 12(9), 1146. https://doi.org/10.3390/children12091146






