Parapneumonic Effusion Versus Pulmonary Empyema in Children: Analysis of Risk Factors and Laboratory Predictors Through a Single Center Experience
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Collection
2.3. Statistical Analysis
3. Results
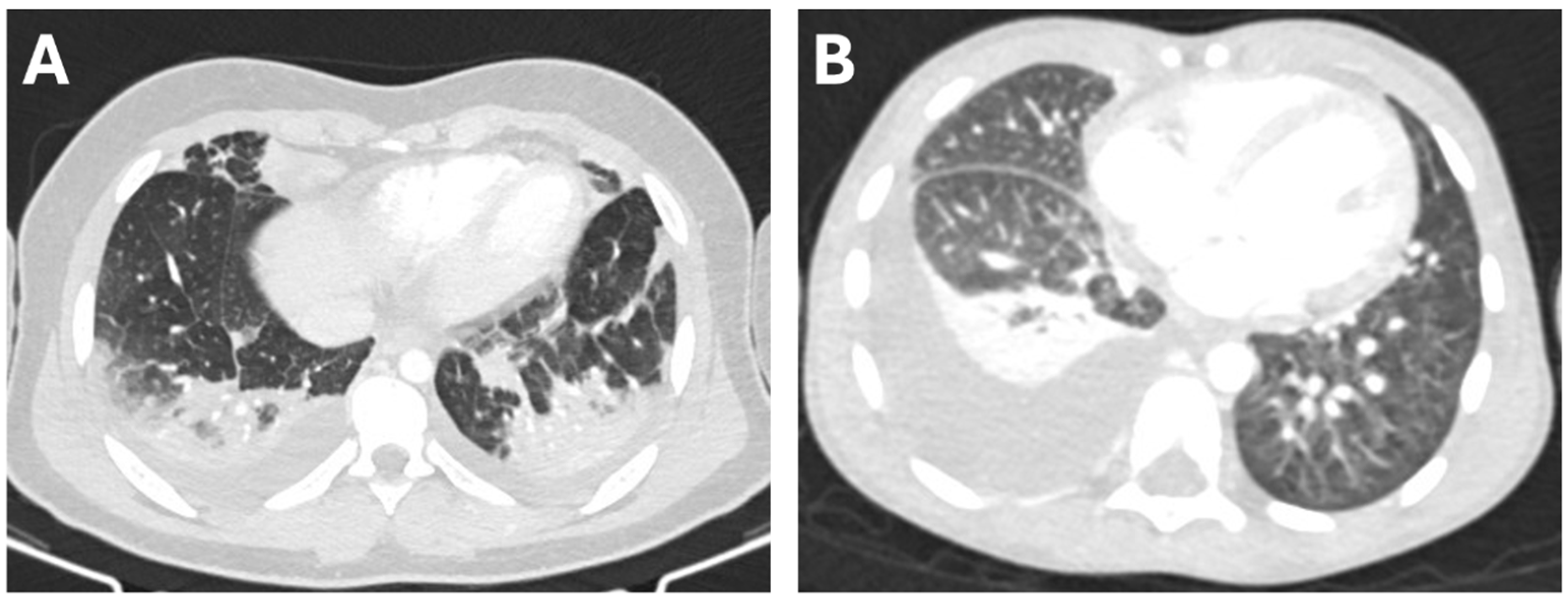
3.1. Patient Characteristics and Clinical Presentation
3.2. Laboratory Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAP | Community Acquired Pneumonia |
| PEf | Pleural Effusion |
| PEm | Pleural Empyema |
| CT | Computed Tomography |
| WBC | White Blood Cells |
| CRP | C-Reactive Protein |
| PCT | Procalcitonin |
| SpO2 | Peripheral Oxygen Saturation |
| LDH | Lactate Dehydrogenase |
| ROC | Receiver Operating Characteristic |
| AUC | Area Under the Curve |
References
- Buonsenso, D.; Cusenza, F.; Passadore, L.; Bonanno, F.; Calanca, C.; Mariani, F.; Di Martino, C.; Rasmi, S.; Esposito, S. Parapneumonic empyema in children: A scoping review of the literature. Ital. J. Pediatr. 2024, 50, 136. [Google Scholar] [CrossRef]
- Maffey, A.; Colom, A.; Venialgo, C.; Acastello, E.; Garrido, P.; Cozzani, H.; Eguiguren, C.; Teper, A. Clinical, functional, and radiological outcome in children with pleural empyema. Pediatr. Pulmonol. 2019, 54, 525–530. [Google Scholar] [CrossRef]
- Wiese, A.D.; Griffin, M.R.; Zhu, Y.; Mitchel, E.F., Jr.; Grijalva, C.G. Changes in empyema among U.S. children in the pneumococcal conjugate vaccine era. Vaccine 2016, 34, 6243–6249. [Google Scholar] [CrossRef] [PubMed]
- Azzari, C.; Serranti, D.; Nieddu, F.; Moriondo, M.; Casini, A.; Lodi, L.; de Benedictis, F.M.; De Vitis, E.; Cavone, F.; Cortimiglia, M.; et al. Significant impact of pneumococcal conjugate vaccination on pediatric parapneumonic effusion: Italy 2006–2018. Vaccine 2019, 37, 2704–2711. [Google Scholar] [CrossRef] [PubMed]
- Tąpolska-Jóźwiak, K.; Gowin, E.; Pasieka, A.; Wasiak, A.; Stasik, P.; Łoś, A.; Sobkowiak, P.; Kycler, Z.; Jończyk-Potoczna, K.; Wojsyk-Banaszak, I. Increasing incidence of severe complicated pneumonia in children caused by Streptococcus pyogenes. Pediatr. Pulmonol. 2024, 59, 1836–1839. [Google Scholar] [CrossRef] [PubMed]
- Kim, O.H.; Kim, W.S.; Kim, M.J.; Jung, J.Y.; Suh, J.H. US in the diagnosis of pediatric chest diseases. Radiographics 2000, 20, 653–671. [Google Scholar] [CrossRef]
- Safari, S.; Baratloo, A.; Elfil, M.; Negida, A. Evidence Based Emergency Medicine; Part 5 Receiver Operating Curve and Area under the Curve. Emergency 2016, 4, 111–113. [Google Scholar]
- Yin, J.; Tian, L. Joint inference about sensitivity and specificity at the optimal cut-off point associated with Youden index. Comput. Stat. Data Anal. 2014, 77, 1–13. [Google Scholar] [CrossRef]
- Masarweh, K.; Gur, M.; Toukan, Y.; Bar-Yoseph, R.; Kassis, I.; Gut, G.; Hakim, F.; Nir, V.; Bentur, L. Factors associated with complicated pneumonia in children. Pediatr. Pulmonol. 2021, 56, 2700–2706. [Google Scholar] [CrossRef]
- Meganathan, P.; Awasthi, S. Predicting complicated parapneumonic effusion in community-acquired pneumonia: Hospital-based case-control study. Indian J. Pediatr. 2019, 86, 140–147. [Google Scholar] [CrossRef]
- Krenke, K.; Krawiec, M.; Kraj, G.; Peradzynska, J.; Krauze, A.; Kulus, M. Risk factors for local complications in children with community-acquired pneumonia. Clin. Respir. J. 2018, 12, 253–261. [Google Scholar] [CrossRef]
- Quaglietta, L.; Martinelli, M.; Staiano, A. Serious infectious events and ibuprofen administration in pediatrics: A narrative review in the era of COVID-19 pandemic. Ital. J. Pediatr. 2021, 47, 20. [Google Scholar] [CrossRef]
- Sodhi, M.; Khosrow-Khavar, F.; FitzGerald, J.M.; Etminan, M. Non-steroidal anti-inflammatory drugs and the risk of pneumonia complications: A systematic review. Pharmacotherapy 2020, 40, 970–977. [Google Scholar] [CrossRef]
- François, P.; Desrumaux, A.; Cans, C.; Pin, I.; Pavese, P.; Labarère, J. Prevalence and risk factors of suppurative complications in children with pneumonia. Acta Paediatr. 2010, 99, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Elemraid, M.A.; Thomas, M.F.; Blain, A.P.; Rushton, S.P.; Spencer, D.A.; Gennery, A.R.; Clark, J.E.; North East of England Pediatric Respiratory Infection Study Group. Risk factors for the development of pleural empyema in children. Pediatr. Pulmonol. 2015, 50, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Ooi, J.M.; Eg, K.P.; Chinna, K.; Nathan, A.M.; de Bruyne, J.A.; Thavagnanam, S. Predictive risk factors for complicated pneumonia in Malaysian children. J. Paediatr. Child Health 2019, 55, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Breuer, O.; Picard, E.; Benabu, N.; Erlichman, I.; Reiter, J.; Tsabari, R.; Shoseyov, D.; Kerem, E.; Cohen-Cymberknoh, M. Predictors of prolonged hospitalizations in pediatric complicated pneumonia. Chest 2018, 153, 172–180. [Google Scholar] [CrossRef]
- Balfour-Lynn, I.M.; Abrahamson, E.; Cohen, G.; Hartley, J.; King, S.; Parikh, D.; Spencer, D.; Thomson, A.H.; Urquhart, D.; Paediatric Pleural Diseases Subcommittee of the BTS Standards of Care Committee. BTS guidelines for the management of pleural infection in children. Thorax 2005, 60 (Suppl. S1), i1–i21. [Google Scholar] [CrossRef]
- Arrieta, A.C.; Osborne, S.; Grant, L.R.; Gessner, B.D.; Blaschke, A.J.; Hulten, K.G.; Nieves, D.J.; Miller, A.; Bender, C.; Isturiz, R.; et al. Parapneumonic empyema complicating community-acquired pneumonia: Etiology in the era of pneumococcal vaccination and role of molecular diagnosis. Pediatr. Infect. Dis. J. 2025, 44, 622–629. [Google Scholar] [CrossRef]
- Maglione, M.; Tipo, V.; Barbieri, E.; Ragucci, R.; Ciccarelli, A.S.; Esposito, C.; Carangelo, L.; Giannattasio, A. Changes in Respiratory Viruses’ Activity in Children During the COVID-19 Pandemic: A Systematic Review. J. Clin. Med. 2025, 14, 1387. [Google Scholar] [CrossRef]
- Hoshino, T.; Nishima, D.; Enseki, M.; Umehara, N.; Fukasawa, C.; Ishiwada, N. Pediatric parapneumonic effusion/pleural empyema in Japan: A nationwide survey. Pediatr. Infect. Dis. J. 2022, 41, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Maglione, M.; Pascarella, A.; Botti, C.; Ricci, G.; Morelli, F.; Camelia, F.; Micillo, A.; Calì, C.; Savoia, F.; Tipo, V.; et al. Changing Epidemiology of Acute Viral Respiratory Infections in Hospitalized Children: The Post-Lockdown Effect. Children 2022, 9, 1242. [Google Scholar] [CrossRef] [PubMed]
- Nygaard, U.; Kirkby, N.S.; Bloch, J.; Sethi, N.J.; Nielsen, A.C.Y.; Poulsen, A.; Buchvald, F.; von Linstow, M.L.; Vissing, N.H. Parapneumonic effusion in children: Rapid pathogen detection in pleural fluid using multiplex bacterial PCR. Acta Paediatr. 2023, 112, 1555–1557. [Google Scholar] [CrossRef] [PubMed]
- Ho, E.C.; Olson, K.E.; Butler, M.; Birkholz, M.; Miller, K.; MacBrayne, C.E.; Jung, S.; Messacar, K.; Asturias, E.J.; Dominguez, S.R. Clinical Impact of Pleural Fluid Streptococcus pneumoniae Polymerase Chain Reaction Testing in Children with Complicated Pneumonia. Clin. Infect. Dis. 2024, 79, 1487–1494. [Google Scholar] [CrossRef]
- Haggie, S.; Selvadurai, H.; Gunasekera, H.; Fitzgerald, D.A.; Lord, D.; Chennapragada, M.S. Pediatric empyema: Are ultrasound characteristics at the time of intervention predictive of reintervention? Pediatr. Pulmonol. 2022, 57, 1643–1650. [Google Scholar] [CrossRef]
- Thakur, C.; Mathew, J.L.; Saxena, A.K.; Angrup, A.; Samujh, R. Six Versus Three Doses of Intrapleural Streptokinase in Childhood Empyema: A Randomized Controlled Trial. Pediatr. Pulmonol. 2025, 60, e27465. [Google Scholar] [CrossRef]
- Forster, J.; Piazza, G.; Goettler, D.; Tanzberger, E.L.; Kenntner, S.; Schoen, C.; Streng, A.; Liese, J.G. Empiric Antibiotic Therapy in 1402 Children with Parapneumonic Effusion/Pleural Empyema in Germany: A Long-term Surveillance Study. Pediatr. Infect. Dis. J. 2024, 43, 651–656. [Google Scholar] [CrossRef]
- Ratta, A.; Nascimben, F.; Angotti, R.; Todesco, C.; Carlini, V.; Fusi, G.; De Biagi, L.; Straziuso, S.; Italiano, F.; Domenichelli, V.; et al. Pleural drainage vs video-assisted thoracoscopic debridement in children affected by pleural empyema. Pediatr. Surg. Int. 2023, 39, 287. [Google Scholar] [CrossRef]
- Bell, A.C.J.; Baker, C.; Duret, A. Is chest drain insertion and fibrinolysis therapy equivalent to video-assisted thoracoscopic surgery to treat children with parapneumonic effusions? Arch. Dis. Child. 2023, 108, 940–942. [Google Scholar] [CrossRef]
- Gesellschaft für Pädiatrische Pneumologie/Deutsche Gesellschaft für Pädiatrische Infektiologie Management der Ambulant Erworbenen Pneumonie bei Kindern und Jugendlichen (Paediatric Community-Acquired Pneumonia, pCAP) Aktualisierte Version 2.0. Available online: https://register.awmf.org/de/leitlinien/detail/048-013 (accessed on 25 January 2024).
- Nandan, D.; Agarwal, S.; Bidhuri, N.; Shrivastava, K.; Nanda, P.; Lata, S. Role of Intrapleural Urokinase in Empyema Thoracis. Indian J. Pediatr. 2019, 86, 1099–1104. [Google Scholar] [CrossRef]

| PEf Group (n = 17) | PEm Group (n = 18) | p-Value | |
|---|---|---|---|
| Demographic data | |||
| Male sex, n (%) | 8 (47.1) | 8 (44.4) | 1.0 |
| Female sex, n (%) | 9 (52.9) | 10 (55.6) | 1.0 |
| Age (yrs) * | 7.1 (1.4–13.4) | 5.2 (0.2–13) | 0.34 |
| Comorbidity | |||
| Respiratory conditions, n (%) | 1 (5.8) | 0 (0) | 1.0 |
| Other, n (%) | 0 (0) | 1 (5.5) | 1.0 |
| Clinical Features | |||
| Fever, n (%) | 16 (94.1) | 17 (94.4) | 1.0 |
| Dyspnea, n (%) | 8 (47.1) | 12 (66.6) | 0.31 |
| Abdominal Pain, n (%) | 3 (17.6) | 8 (44.4) | 0.15 |
| Chest Pain, n (%) | 1 (5.9) | 3 (16.7) | 0.61 |
| Cough, n (%) | 13 (76.5) | 11 (61.1) | 0.47 |
| SpO2% 1, median (range) | 93.8 (85–100) | 93.2 (85–99) | 0.3 |
| RR 2 (acts/min), median (range) | 30 (20–70) | 30 (25–66) | 0.3 |
| Therapeutic Interventions | |||
| Invasive respiratory support, n (%) | 0 (0) | 2 (11.1) | 0.48 |
| Pleural Drainage, n (%) | 0 (0) | 13 (72) | <0.001 |
| Surgery, n (%) | 0 (0) | 3 (16.6) | 0.23 |
| Sub-Intensive Care admission, n (%) | 1 (5.9) | 12 (66.6) | <0.001 |
| Intensive Care Unit admission, n (%) | 0 (0) | 4 (22.2) | 0.1 |
| Hospital stay (days) * | 3 (2–7) | 23 (7–88) | <0.001 |
| Laboratory Data | PEf Group (n = 17) | PEm Group (n = 18) | p-Value |
|---|---|---|---|
| WBC 1 (cell/μL) | 11,050 (6810–28,810) | 22,355 (9420–38,300) | <0.001 |
| Neutrophils (cell/μL) | 6660 (4450–26,660) | 10,020 (290–30,870) | 0.05 |
| Lymphocytes (cell/μL) | 2530 (830–8410) | 1580 (870–7660) | 0.10 |
| CRP 2 (mg/L) | 30.34 (2.23–371.99) | 273.9 (31–578.79) | <0.001 |
| PCT 3 (ng/mL) | 0.25 (0.05–15.0) | 4.6 (0.07–175) | <0.01 |
| Ferritin (ng/mL) | 297.45 (26.5–514) | 691.7 (312–2375) | 0.001 |
| Albumin (g/dL) | 4 (2.9–4.8) | 3.1 (2.5–5.0) | 0.001 |
| Time to CRP normalization * (days) | 3 (0.0–6.0) | 15 (3–61) | <0.001 |
| Time to PCT normalization * (days) | 0 (0–6) | 10 (0–25) | <0.001 |
| Cut-Off Value | AUC 1 (95%CI) | p-Value | Sensitivity (%) | Specificity (%) | |
|---|---|---|---|---|---|
| WBC 2 | 20,560 | 0.624 (0.438–0.810) | 0.19 | 33.3 | 94.1 |
| Neutrophils | 7450 | 0.727 (0.559–0.895) | 0.008 | 83 | 71 |
| Lymphocytes | 1470 | 0.431 (0.239–0.623) | 0.48 | 61 | 47 |
| CRP 3 | 185 | 0.735 (0.568–0.902) | 0.006 | 61 | 88 |
| PCT 4 | 0.6 | 0.746 (0.577–0.915) | 0.004 | 76 | 81 |
| Ferritin | 472.8 | 0.835 (0.665–1) | <0.001 | 79 | 87 |
| Albumin | 3.7 | 0.822 (0.681–0.963) | <0.001 | 83 | 76 |
| OR 1 | 95% CI | p-Value | ||
|---|---|---|---|---|
| WBC 2 | 0.99 | 0.99 | 1.00 | 0.52 |
| Neutrophils | 1.01 | 0.99 | 1.00 | 0.54 |
| Lymphocytes | 1.01 | 0.99 | 1.00 | 0.51 |
| CRP 3 | 0.99 | 0.97 | 1.02 | 0.74 |
| PCT 4 | 0.98 | 0.83 | 1.15 | 0.79 |
| Ferritin | 0.99 | 0.99 | 1.00 | 0.17 |
| Albumin | 2.39 | 0.19 | 30.23 | 0.50 |
| PEf Group (n = 17) | PEm Group (n = 18) | p-Value | |
|---|---|---|---|
| Microbiological Data | |||
| Urinary Pneumococcal Antigen (n/tested, %) | 0/11 (0) | 4/16 (25) | 0.10 |
| Positive Mycoplasma Serology (n/tested, %) | 8/14 (57.1) | 7/13 (53.8) | 1.0 |
| Positive Blood Culture (n/tested, %) | 1/12 (8.3) | 2/16 (12.5) | 1 |
| Isolated pathogen on respiratory sample, n (%) * | |||
| Mycoplasma pneumoniae | 7 (41.2) | 1 (5.5) | |
| Chlamydia pneumoniae | 0 (0) | 1 (5.5) | |
| Metapneumovirus | 2 (11.8) | 1 (5.5) | |
| Adenovirus | 2 (11.8) | 3 (16.7) | |
| Respiratory syncytial virus | 2 (11.8) | 1 (5.5) | |
| Rhino/Enterovirus | 4 (23.5) | 6 (33.3) | |
| Parainfluenza virus | 4 (23.5) | 2 (11.1) | |
| Influenza virus | 0 (0) | 1 (5.5) | |
| Coronavirus | 2 (11.8) | 1 (5.5) | |
| None | 4 (23.5) | 2 (11.1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Improta, M.; Morlino, F.; Ragucci, R.; D’Anna, C.; Muzzica, S.; Tipo, V.; Giannattasio, A.; Maglione, M. Parapneumonic Effusion Versus Pulmonary Empyema in Children: Analysis of Risk Factors and Laboratory Predictors Through a Single Center Experience. Children 2025, 12, 1103. https://doi.org/10.3390/children12081103
Improta M, Morlino F, Ragucci R, D’Anna C, Muzzica S, Tipo V, Giannattasio A, Maglione M. Parapneumonic Effusion Versus Pulmonary Empyema in Children: Analysis of Risk Factors and Laboratory Predictors Through a Single Center Experience. Children. 2025; 12(8):1103. https://doi.org/10.3390/children12081103
Chicago/Turabian StyleImprota, Marta, Francesca Morlino, Roberta Ragucci, Carolina D’Anna, Stefania Muzzica, Vincenzo Tipo, Antonietta Giannattasio, and Marco Maglione. 2025. "Parapneumonic Effusion Versus Pulmonary Empyema in Children: Analysis of Risk Factors and Laboratory Predictors Through a Single Center Experience" Children 12, no. 8: 1103. https://doi.org/10.3390/children12081103
APA StyleImprota, M., Morlino, F., Ragucci, R., D’Anna, C., Muzzica, S., Tipo, V., Giannattasio, A., & Maglione, M. (2025). Parapneumonic Effusion Versus Pulmonary Empyema in Children: Analysis of Risk Factors and Laboratory Predictors Through a Single Center Experience. Children, 12(8), 1103. https://doi.org/10.3390/children12081103





