Maternal Voice and Tactile Stimulation Modulate Oxytocin in Mothers of Hospitalized Preterm Infants: A Randomized Crossover Trial
Abstract
:1. Introduction
2. Methods
2.1. Patients and Their Characteristics
2.2. Study Design and Interventions
2.3. Sample Size Calculation
2.4. Primary and Secondary Outcomes
2.5. Statistical Analysis
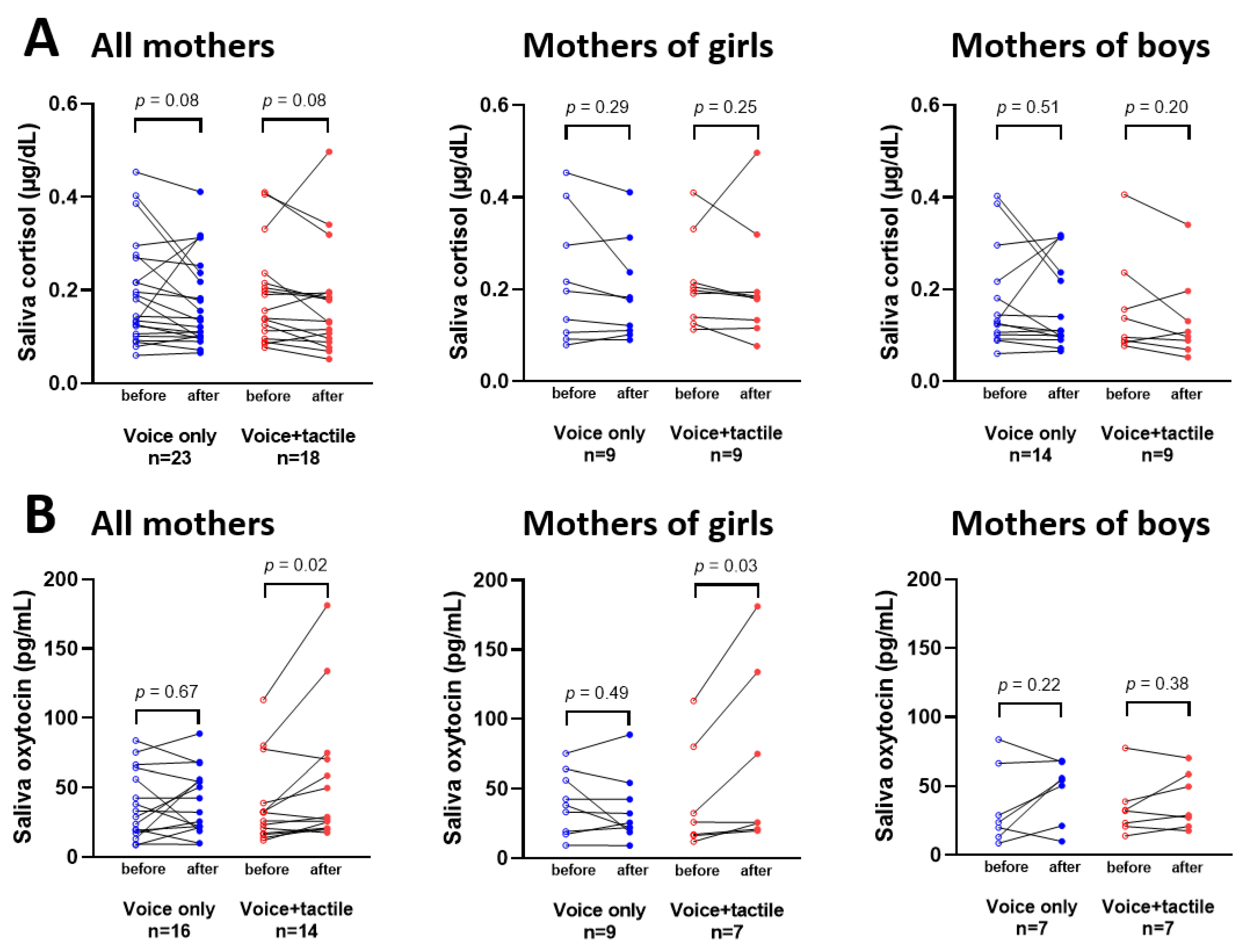
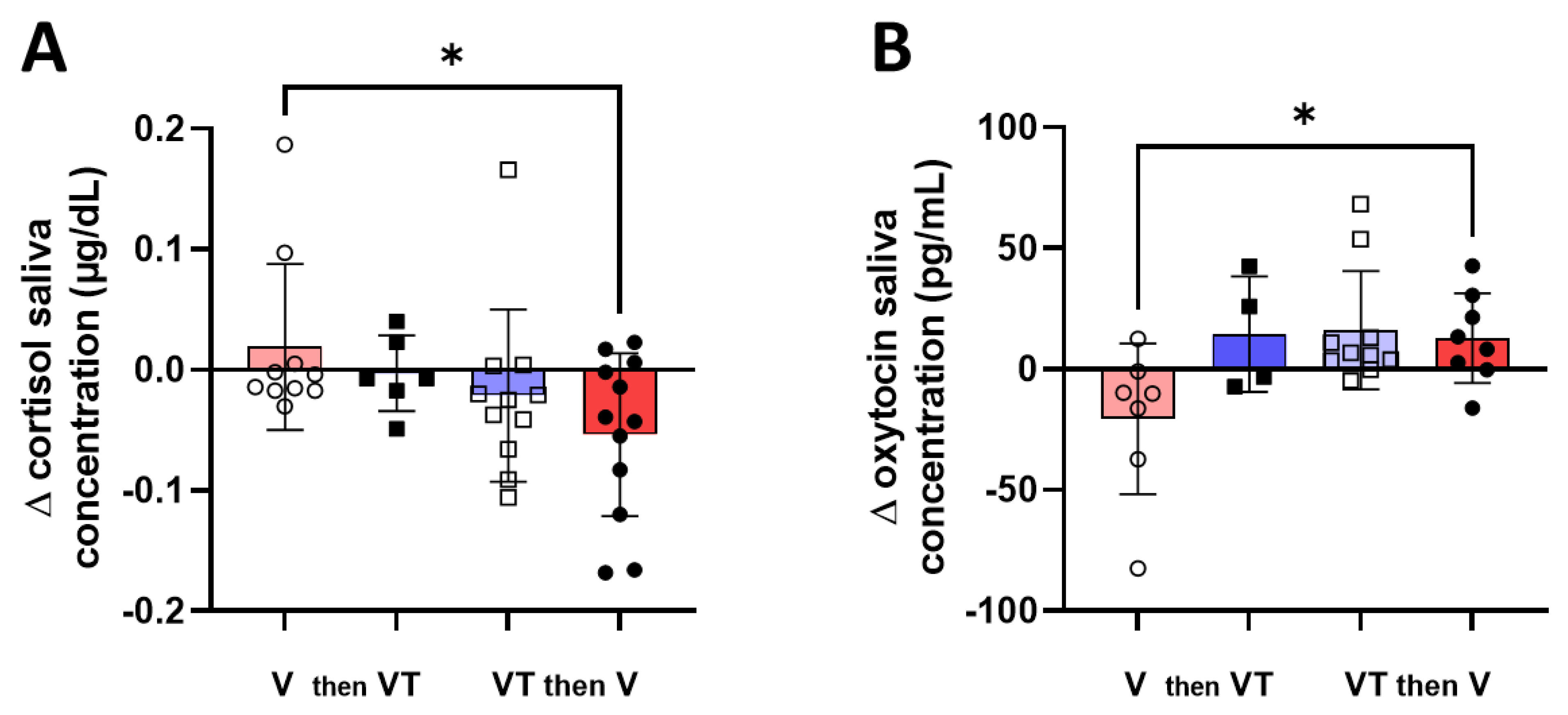
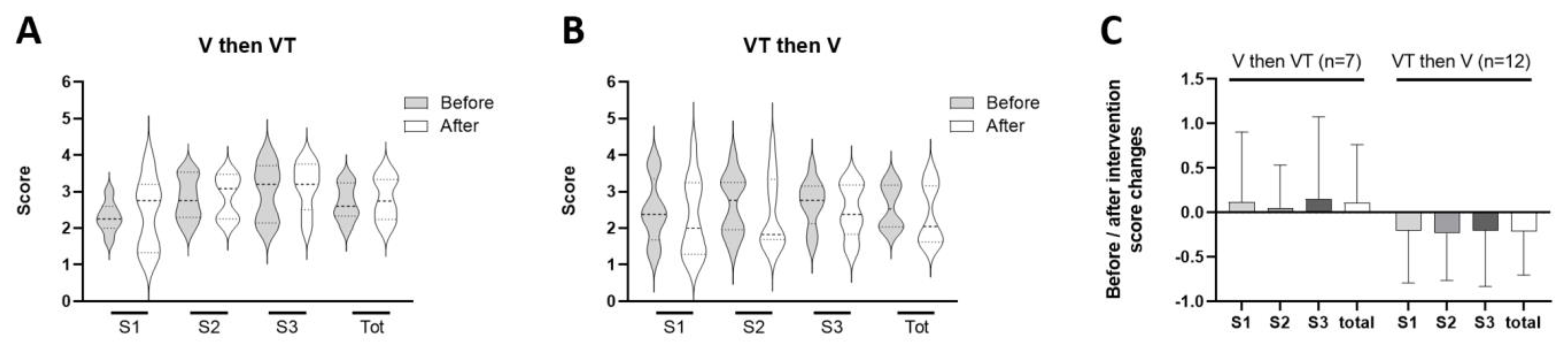
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Delobel-Ayoub, M.; Arnaud, C.; White-Koning, M.; Casper, C.; Pierrat, V.; Garel, M.; Burguet, A.; Roze, J.C.; Matis, J.; Picaud, J.C.; et al. Behavioral problems and cognitive performance at 5 years of age after very preterm birth: The EPIPAGE Study. Pediatrics 2009, 123, 1485–1492. [Google Scholar] [CrossRef] [PubMed]
- Volpe, J.J. Systemic inflammation, oligodendroglial maturation, and the encephalopathy of prematurity. Ann. Neurol. 2011, 70, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J.; Gluckman, P.D.; Godfrey, K.M.; Harding, J.E.; Owens, J.A.; Robinson, J.S. Fetal nutrition and cardiovascular disease in adult life. Lancet 1993, 341, 938–941. [Google Scholar] [CrossRef]
- Heim, C.; Nemeroff, C.B. The impact of early adverse experiences on brain systems involved in the pathophysiology of anxiety and affective disorders. Biol. Psychiatry 1999, 46, 1509–1522. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, A.; Belliveau, C.; Mechawar, N.; Walker, C.D. Effects of Early Life Stress on the Developing Basolateral Amygdala-Prefrontal Cortex Circuit: The Emerging Role of Local Inhibition and Perineuronal Nets. Front. Hum. Neurosci. 2021, 15, 669120. [Google Scholar] [CrossRef]
- Filippa, M.; Devouche, E.; Arioni, C.; Imberty, M.; Gratier, M. Live maternal speech and singing have beneficial effects on hospitalized preterm infants. Acta Paediatr. 2013, 102, 1017–1020. [Google Scholar] [CrossRef]
- Filippa, M.; Panza, C.; Ferrari, F.; Frassoldati, R.; Kuhn, P.; Balduzzi, S.; D’Amico, R. Systematic review of maternal voice interventions demonstrates increased stability in preterm infants. Acta Paediatr. 2017, 106, 1220–1229. [Google Scholar] [CrossRef]
- Filippa, M.; Monaci, M.G.; Spagnuolo, C.; Di Benedetto, M.; Serravalle, P.; Grandjean, D. Oxytocin Levels Increase and Anxiety Decreases in Mothers Who Sing and Talk to Their Premature Infants during a Painful Procedure. Children 2023, 10, 334. [Google Scholar] [CrossRef]
- Knoop, M.; Possovre, M.L.; Jacquens, A.; Charlet, A.; Baud, O.; Darbon, P. The Role of Oxytocin in Abnormal Brain Development: Effect on Glial Cells and Neuroinflammation. Cells 2022, 11, 3899. [Google Scholar] [CrossRef]
- Filippa, M.; Poisbeau, P.; Mairesse, J.; Monaci, M.G.; Baud, O.; Hüppi, P.; Grandjean, D.; Kuhn, P. Pain, parental involvement, and oxytocin in the neonatal intensive care unit. Front. Psychol. 2019, 10, 715. [Google Scholar] [CrossRef]
- Weber, A.; Harrison, T.M.; Sinnott, L.; Shoben, A.; Steward, D. Plasma and Urinary Oxytocin Trajectories in Extremely Premature Infants During NICU Hospitalization. Biol. Res. Nurs. 2017, 19, 549–558. [Google Scholar] [CrossRef]
- Vittner, D.; McGrath, J.; Robinson, J.; Lawhon, G.; Cusson, R.; Eisenfeld, L.; Walsh, S.; Young, E.; Cong, X. Increase in Oxytocin From Skin-to-Skin Contact Enhances Development of Parent-Infant Relationship. Biol. Res. Nurs. 2018, 20, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Cong, X.; Ludington-Hoe, S.M.; Hussain, N.; Cusson, R.M.; Walsh, S.; Vazquez, V.; Briere, C.E.; Vittner, D. Parental oxytocin responses during skin-to-skin contact in pre-term infants. Early Hum. Dev. 2015, 91, 401–406. [Google Scholar] [CrossRef]
- Vittner, D.; Butler, S.; Smith, K.; Makris, N.; Brownell, E.; Samra, H.; McGrath, J. Parent Engagement Correlates With Parent and Preterm Infant Oxytocin Release During Skin-to-Skin Contact. Adv. Neonatal Care 2019, 19, 73–79. [Google Scholar] [CrossRef]
- Weber, A.; Harrison, T.M.; Steward, D.; Sinnott, L.; Shoben, A. Oxytocin trajectories and social engagement in extremely premature infants during NICU hospitalization. Infant Behav. Dev. 2017, 48, 78–87. [Google Scholar] [CrossRef]
- Zinni, M.; Colella, M.; Batista Novais, A.R.; Baud, O.; Mairesse, J. Modulating the Oxytocin System During the Perinatal Period: A New Strategy for Neuroprotection of the Immature Brain? Front. Neurol. 2018, 9, 229. [Google Scholar] [CrossRef]
- Windle, R.J.; Kershaw, Y.M.; Shanks, N.; Wood, S.A.; Lightman, S.L.; Ingram, C.D. Oxytocin attenuates stress-induced c-fos mRNA expression in specific forebrain regions associated with modulation of hypothalamo-pituitary-adrenal activity. J. Neurosci. 2004, 24, 2974–2982. [Google Scholar] [CrossRef] [PubMed]
- Jurek, B.; Slattery, D.A.; Hiraoka, Y.; Liu, Y.; Nishimori, K.; Aguilera, G.; Neumann, I.D.; van den Burg, E.H. Oxytocin Regulates Stress-Induced Crf Gene Transcription through CREB-Regulated Transcription Coactivator 3. J. Neurosci. 2015, 35, 12248–12260. [Google Scholar] [CrossRef] [PubMed]
- Windle, R.J.; Shanks, N.; Lightman, S.L.; Ingram, C.D. Central oxytocin administration reduces stress-induced corticosterone release and anxiety behavior in rats. Endocrinology 1997, 138, 2829–2834. [Google Scholar] [CrossRef] [PubMed]
- Neumann, I.D.; Wigger, A.; Torner, L.; Holsboer, F.; Landgraf, R. Brain oxytocin inhibits basal and stress-induced activity of the hypothalamo-pituitary-adrenal axis in male and female rats: Partial action within the paraventricular nucleus. J. Neuroendocrinol. 2000, 12, 235–243. [Google Scholar] [CrossRef]
- Pineda, R.; Guth, R.; Herring, A.; Reynolds, L.; Oberle, S.; Smith, J. Enhancing sensory experiences for very preterm infants in the NICU: An integrative review. J. Perinatol. 2017, 37, 323–332. [Google Scholar] [CrossRef]
- Maitre, N.L.; Key, A.P.; Slaughter, J.C.; Yoder, P.J.; Neel, M.L.; Richard, C.; Wallace, M.T.; Murray, M.M. Neonatal multisensory processing in preterm and term infants predicts sensory reactivity and internalizing tendencies in early childhood. Brain Topogr. 2020, 33, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Lejeune, F.; Brand, L.A.; Palama, A.; Parra, J.; Marcus, L.; Barisnikov, K.; Debillon, T.; Gentaz, E.; Berne-Audéoud, F. Preterm infant showed better object handling skills in a neonatal intensive care unit during silence than with a recorded female voice. Acta Paediatr. 2019, 108, 460–467. [Google Scholar] [CrossRef]
- Northrup, J.B. Contingency detection in a complex world: A developmental model and implications for atypical development. Int. J. Behav. Dev. 2017, 41, 723–734. [Google Scholar] [CrossRef]
- Lejeune, F.; Lordier, L.; Pittet, M.P.; Schoenhals, L.; Grandjean, D.; Huppi, P.S.; Filippa, M.; Borradori Tolsa, C. Effects of an Early Postnatal Music Intervention on Cognitive and Emotional Development in Preterm Children at 12 and 24 Months: Preliminary Findings. Front. Psychol. 2019, 10, 494. [Google Scholar] [CrossRef] [PubMed]
- Miles, M.S.; Funk, S.G.; Carlson, J. Parental Stressor Scale: Neonatal intensive care unit. Nurs. Res. 1993, 42, 148–152. [Google Scholar] [CrossRef]
- Benzies, K.M.; Magill-Evans, J.E.; Hayden, K.A.; Ballantyne, M. Key components of early intervention programs for preterm infants and their parents: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2013, 13 (Suppl. S1), S10. [Google Scholar] [CrossRef]
- Korzeniewska, A.; Wang, Y.; Benz, H.L.; Fifer, M.S.; Collard, M.; Milsap, G.; Cervenka, M.C.; Martin, A.; Gotts, S.J.; Crone, N.E. Changes in human brain dynamics during behavioral priming and repetition suppression. Prog. Neurobiol. 2020, 189, 101788. [Google Scholar] [CrossRef]
- Tang, Y.; Benusiglio, D.; Lefevre, A.; Hilfiger, L.; Althammer, F.; Bludau, A.; Hagiwara, D.; Baudon, A.; Darbon, P.; Schimmer, J.; et al. Social touch promotes interfemale communication via activation of parvocellular oxytocin neurons. Nat. Neurosci. 2020, 23, 1125–1137. [Google Scholar] [CrossRef]
- Kozhemiako, N.; Nunes, A.S.; Vakorin, V.A.; Chau, C.M.; Moiseev, A.; Ribary, U.; Grunau, R.E.; Doesburg, S.M. Sex differences in brain connectivity and male vulnerability in very preterm children. Hum. Brain Mapp. 2020, 41, 388–400. [Google Scholar] [CrossRef]
- Ditzen, B.; Schaer, M.; Gabriel, B.; Bodenmann, G.; Ehlert, U.; Heinrichs, M. Intranasal oxytocin increases positive communication and reduces cortisol levels during couple conflict. Biol. Psychiatry 2009, 65, 728–731. [Google Scholar] [CrossRef]
- Amini-Khoei, H.; Mohammadi-Asl, A.; Amiri, S.; Hosseini, M.J.; Momeny, M.; Hassanipour, M.; Rastegar, M.; Haj-Mirzaian, A.; Mirzaian, A.H.; Sanjarimoghaddam, H.; et al. Oxytocin mitigated the depressive-like behaviors of maternal separation stress through modulating mitochondrial function and neuroinflammation. Prog. Neuropsychopharmacol. Biol. Psychiatry 2017, 76, 169–178. [Google Scholar] [CrossRef]
- Karelina, K.; Stuller, K.A.; Jarrett, B.; Zhang, N.; Wells, J.; Norman, G.J.; DeVries, A.C. Oxytocin mediates social neuroprotection after cerebral ischemia. Stroke 2011, 42, 3606–3611. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Liu, S.; Bai, X.; Gao, Y.; Liu, G.; Wang, X.; Liu, D.; Li, T.; Hao, A.; Wang, Z. Oxytocin inhibits lipopolysaccharide-induced inflammation in microglial cells and attenuates microglial activation in lipopolysaccharide-treated mice. J. Neuroinflamm. 2016, 13, 77. [Google Scholar] [CrossRef] [PubMed]
- Rajamani, K.T.; Wagner, S.; Grinevich, V.; Harony-Nicolas, H. Oxytocin as a Modulator of Synaptic Plasticity: Implications for Neurodevelopmental Disorders. Front. Synaptic Neurosci. 2018, 10, 17. [Google Scholar] [CrossRef]
- Pedersen, C.A.; Ascher, J.A.; Monroe, Y.L.; Prange, A.J., Jr. Oxytocin induces maternal behavior in virgin female rats. Science 1982, 216, 648–650. [Google Scholar] [CrossRef] [PubMed]
- Francis, D.D.; Champagne, F.C.; Meaney, M.J. Variations in maternal behaviour are associated with differences in oxytocin receptor levels in the rat. J. Neuroendocrinol. 2000, 12, 1145–1148. [Google Scholar] [CrossRef]
- Forcada-Guex, M.; Borghini, A.; Pierrehumbert, B.; Ansermet, F.; Muller-Nix, C. Prematurity, maternal posttraumatic stress and consequences on the mother-infant relationship. Early Hum. Dev. 2011, 87, 21–26. [Google Scholar] [CrossRef]
- Forcada-Guex, M.; Pierrehumbert, B.; Borghini, A.; Moessinger, A.; Muller-Nix, C. Early dyadic patterns of mother-infant interactions and outcomes of prematurity at 18 months. Pediatrics 2006, 118, e107–e114. [Google Scholar] [CrossRef]
- Feldman, R.; Gordon, I.; Schneiderman, I.; Weisman, O.; Zagoory-Sharon, O. Natural variations in maternal and paternal care are associated with systematic changes in oxytocin following parent-infant contact. Psychoneuroendocrinology 2010, 35, 1133–1141. [Google Scholar] [CrossRef]
- Moriceau, S.; Shionoya, K.; Jakubs, K.; Sullivan, R.M. Early-life stress disrupts attachment learning: The role of amygdala corticosterone, locus ceruleus corticotropin releasing hormone, and olfactory bulb norepinephrine. J. Neurosci. 2009, 29, 15745–15755. [Google Scholar] [CrossRef] [PubMed]
- Alves, E.; Fielder, A.; Ghabriel, N.; Sawyer, M.; Buisman-Pijlman, F.T. Early social environment affects the endogenous oxytocin system: A review and future directions. Front. Endocrinol. 2015, 6, 32. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Raine, A.; Chan, F.; Venables, P.H.; Mednick, S.A. Early maternal and paternal bonding, childhood physical abuse and adult psychopathic personality. Psychol. Med. 2010, 40, 1007–1016. [Google Scholar] [CrossRef]
- Hammock, E.A.; Young, L.J. Oxytocin, vasopressin and pair bonding: Implications for autism. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2006, 361, 2187–2198. [Google Scholar] [CrossRef] [PubMed]
- Shishido, E.; Shuo, T.; Takahata, K.; Horiuchi, S. Changes in salivary oxytocin levels and bonding disorder in women from late pregnancy to early postpartum: A pilot study. PLoS ONE 2019, 14, e0221821. [Google Scholar] [CrossRef]
- Weiss, G. Endocrinology of parturition. J. Clin. Endocrinol. Metab. 2000, 85, 4421–4425. [Google Scholar] [CrossRef]
- Olza, I.; Uvnas-Moberg, K.; Ekstrom-Bergstrom, A.; Leahy-Warren, P.; Karlsdottir, S.I.; Nieuwenhuijze, M.; Villarmea, S.; Hadjigeorgiou, E.; Kazmierczak, M.; Spyridou, A.; et al. Birth as a neuro-psycho-social event: An integrative model of maternal experiences and their relation to neurohormonal events during childbirth. PLoS ONE 2020, 15, e0230992. [Google Scholar] [CrossRef]
- Walter, M.H.; Abele, H.; Plappert, C.F. The Role of Oxytocin and the Effect of Stress During Childbirth: Neurobiological Basics and Implications for Mother and Child. Front. Endocrinol. 2021, 12, 742236. [Google Scholar] [CrossRef]
- Kutahyalioglu, N.S.; Scafide, K.N. Effects of family-centered care on bonding: A systematic review. J. Child Health Care 2022. [Google Scholar] [CrossRef]
- Theofanopoulou, C.; Boeckx, C.; Jarvis, E.D. A hypothesis on a role of oxytocin in the social mechanisms of speech and vocal learning. Proc. Biol. Sci. 2017, 284, 20170988. [Google Scholar] [CrossRef] [PubMed]
- Tops, M. Oxytocin: Envy or engagement in others? Biol. Psychiatry 2010, 67, e5–e6, author reply e7. [Google Scholar] [CrossRef]
- Ooishi, Y.; Mukai, H.; Watanabe, K.; Kawato, S.; Kashino, M. Increase in salivary oxytocin and decrease in salivary cortisol after listening to relaxing slow-tempo and exciting fast-tempo music. PLoS ONE 2017, 12, e0189075. [Google Scholar] [CrossRef]
- Martins, D.; Gabay, A.S.; Mehta, M.; Paloyelis, Y. Salivary and plasmatic oxytocin are not reliable trait markers of the physiology of the oxytocin system in humans. eLife 2020, 9, e62456. [Google Scholar] [CrossRef] [PubMed]
- Leng, G.; Sabatier, N. Measuring Oxytocin and Vasopressin: Bioassays, Immunoassays and Random Numbers. J. Neuroendocrinol. 2016, 28, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Kagerbauer, S.M.; Gempt, J.; Podtschaske, A.; Hapfelmeier, A.; Schneider, G. Oxytocin levels in saliva correlate better than plasma levels with concentrations in the cerebrospinal fluid of patients in neurocritical care. J. Neuroendocrinol. 2018, 30, e12596. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, A.R.; Romero, R.; Keefe, D.; Parra, M.; Oyarzun, E.; Behnke, E. Oxytocin secretion and human parturition: Pulse frequency and duration increase during spontaneous labor in women. Am. J. Obstet. Gynecol. 1991, 165, 1515–1523. [Google Scholar] [CrossRef]
- Grewen, K.M.; Davenport, R.E.; Light, K.C. An investigation of plasma and salivary oxytocin responses in breast- and formula-feeding mothers of infants. Psychophysiology 2010, 47, 625–632. [Google Scholar] [CrossRef] [PubMed]


| Variable | V, Then VT (n = 12) | VT, Then V (n = 14) | p-Value * |
|---|---|---|---|
| Mothers | |||
| Maternal age at delivery, years | 0.35 | ||
| median (IQR) | 32 (30–38) | 35 (33–37) | |
| mean (SD) | 33 (8.5) | 35 (4.5) | |
| Ethnicity | 0.65 | ||
| Caucasian | 10 (83.3%) | 10 (71.4%) | |
| Hispanic | 1 (8.3%) | 1 (7.1%) | |
| Asian | 0 | 1 (7.1%) | |
| African | 0 | 1 (7.1%) | |
| Not reported | 1 (8.3%) | 1 (7.1%) | |
| Educational level | 0.31 ** | ||
| Tertiary | 6 (50.0%) | 4 (28.6%) | |
| Upper secondary | 1 (8.3%) | 4 (28.6%) | |
| Lower secondary | 5 (41.7%) | 2 (14.3%) | |
| Primary | 0 | 2 (14.3%) | |
| Not reported | 0 | 1 (7.1%) | |
| Family structure | 0.43 | ||
| Married couple | 5 (41.7%) | 9 (64.3%) | |
| Not married couple | 6 (50.0%) | 3 (21.4%) | |
| Single | 1 (8.3%) | 1 (7.1%) | |
| unknown | 0 | 1 (7.1) | |
| Singleton pregnancy | 7 (58.3%) | 9 (64.3%) | >0.99 |
| Preterm rupture of membranes > 24 h | 1 (8.3%) | 1 (7.1%) | >0.99 |
| Choriomamnionitis | 0 | 0 | - |
| IUGR < 10 perc. | 1 (8.3%) | 2 (14.3%) | >0.99 |
| Preeclampsia | 3 (25.0%) | 2 (14.3%) | 0.63 |
| Gestational diabetes | 1 (8.3%) | 0 | 0.46 |
| Vaginal delivery | 6 (50.0%) | 5 (35.7%) | 0.69 |
| Neonates | |||
| Male sex | 7 (58.3%) | 7 (50%) | 0.71 |
| Gestational age at birth, weeks | 0.13 | ||
| median (IQR) | 34.2 (34.0–34.3) | 33.2 (33.0–34.2) | |
| mean (SD) | 34.1 (0.4) | 33.3 (1.4) | |
| Birthweight, g | 0.59 | ||
| median (IQR) | 2183 (1754–2370) | 1900 (1850–2163) | |
| mean (SD) | 2080 (625) | 1974 (313) | |
| Head circumference, cm | 0.33 | ||
| median (IQR) | 30.0 (29.5–32.0) | 31.5 (30.3–32.8) | |
| mean (SD) | 30.6 (2.5) | 31.3 (2.5) | |
| Apgar score at 5 min | 0.89 | ||
| median (IQR) | 9.5 (7.8–10.0) | 9.0 (8.0–10.0) | |
| mean (SD) | 8.8 (2.3) | 8.8 (2.0) | |
| Early onset sepsis | 0 | 0 | - |
| Hypoglycemia | 2 (16.7%) | 1 (7.1%) | 0.58 |
| Transient tachypnea of the newborn | 8 (66.7%) | 6 (42.9%) | 0.27 |
| Exogenous surfactant | 0 | 0 | - |
| Phototherapy | 5 (41.7%) | 5 (35.7%) | >0.99 |
| Exclusive mother milk | 3 (25.0%) | 1 (7.1%) | 0.31 |
| Age at feeding autonomy, days | 0.36 | ||
| median (IQR) | 13.0 (4.5–19.5) | 19.0 (11.0–25.0) | |
| mean (SD) | 13.6 (15) | 18.0 (14) | |
| Abnormal cranial ultrasound | 0 | 0 | - |
| Age at hospital discharge, days | 0.03 | ||
| median (IQR) | 21.0 (14.5–26.0) | 31.0 (26.0–36.0) | |
| mean (SD) | 21.5 (11.5) | 41.1 (10.0) |
| Intervention Order | Cortisol (µg/dL) | Oxytocin (pg/mL) | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| V first | n = 11 | n = 11 | n = 8 | n = 8 |
| Median (IQR) | 0.132 (0.107–0.217) | 0.111 (0.097–0.253) | 60.0 (23.3–79.4) | 26.8 (12.2–64.1) |
| Mean (SD) | 0.197 (0.183) | 0.158 (0.091) | 53.0 (29.7) | 35.2 (24.8) |
| VT second | n = 6 | n = 6 | n = 4 | n = 4 |
| Median (IQR) | 0.133 (0.094–0.168) | 0.121 (0.086–0.186) | 32.6 (23.7–66.4) | 64.5 (27.8–73.8) |
| Mean (SD) | 0.134 (0.041) | 0.131 (0.049) | 40.9 (25.1) | 55.4 (26.1) |
| VT first | n = 11 | n = 11 | n = 10 | n = 10 |
| Median (IQR) | 0.206 (0.113–0.332) | 0.179 (0.097–0.320) | 24.5 (15.6–49.2) | 26.5 (20.9–70.8) |
| Mean (SD) | 0.220 (0.119) | 0.199 (0.136) | 37.2 (33.3) | 53.3 (56.7) |
| V second | n = 12 | n = 12 | n = 8 | n = 8 |
| Median (IQR) | 0.207 (0.122–0.364) | 0.167 (0.101–0.233) | 26.3 (17.8–41.4) | 46.4 (22.9–55.5) |
| Mean (SD) | 0.237 (0.129) | 0.183 (0.100) | 32.3 (20.1) | 45.2 (22.7) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hirschel, J.; Carlhan-Ledermann, A.; Ferraz, C.; Brand, L.-A.; Filippa, M.; Gentaz, E.; Lejeune, F.; Baud, O. Maternal Voice and Tactile Stimulation Modulate Oxytocin in Mothers of Hospitalized Preterm Infants: A Randomized Crossover Trial. Children 2023, 10, 1469. https://doi.org/10.3390/children10091469
Hirschel J, Carlhan-Ledermann A, Ferraz C, Brand L-A, Filippa M, Gentaz E, Lejeune F, Baud O. Maternal Voice and Tactile Stimulation Modulate Oxytocin in Mothers of Hospitalized Preterm Infants: A Randomized Crossover Trial. Children. 2023; 10(9):1469. https://doi.org/10.3390/children10091469
Chicago/Turabian StyleHirschel, Jessica, Audrey Carlhan-Ledermann, Céline Ferraz, Laure-Anne Brand, Manuela Filippa, Edouard Gentaz, Fleur Lejeune, and Olivier Baud. 2023. "Maternal Voice and Tactile Stimulation Modulate Oxytocin in Mothers of Hospitalized Preterm Infants: A Randomized Crossover Trial" Children 10, no. 9: 1469. https://doi.org/10.3390/children10091469
APA StyleHirschel, J., Carlhan-Ledermann, A., Ferraz, C., Brand, L.-A., Filippa, M., Gentaz, E., Lejeune, F., & Baud, O. (2023). Maternal Voice and Tactile Stimulation Modulate Oxytocin in Mothers of Hospitalized Preterm Infants: A Randomized Crossover Trial. Children, 10(9), 1469. https://doi.org/10.3390/children10091469








