Secondary Prevention of Diabetes Type 1 with Oral Calcitriol and Analogs, the PRECAL Study
Abstract
1. Introduction
1.1. A Window of Opportunity to Prevent and Revert Seroconversion towards T1D Autoantibodies, and Intercept the Development of Multiple T1D Autoantibodies
1.2. The Role of Vitamin D
1.3. Primary Prevention of Type 1 Diabetes with Vitamin D
1.4. Secondary Prevention of Type 1 Diabetes with Calcitriol and Paricalcitol
1.5. Aim of the Study
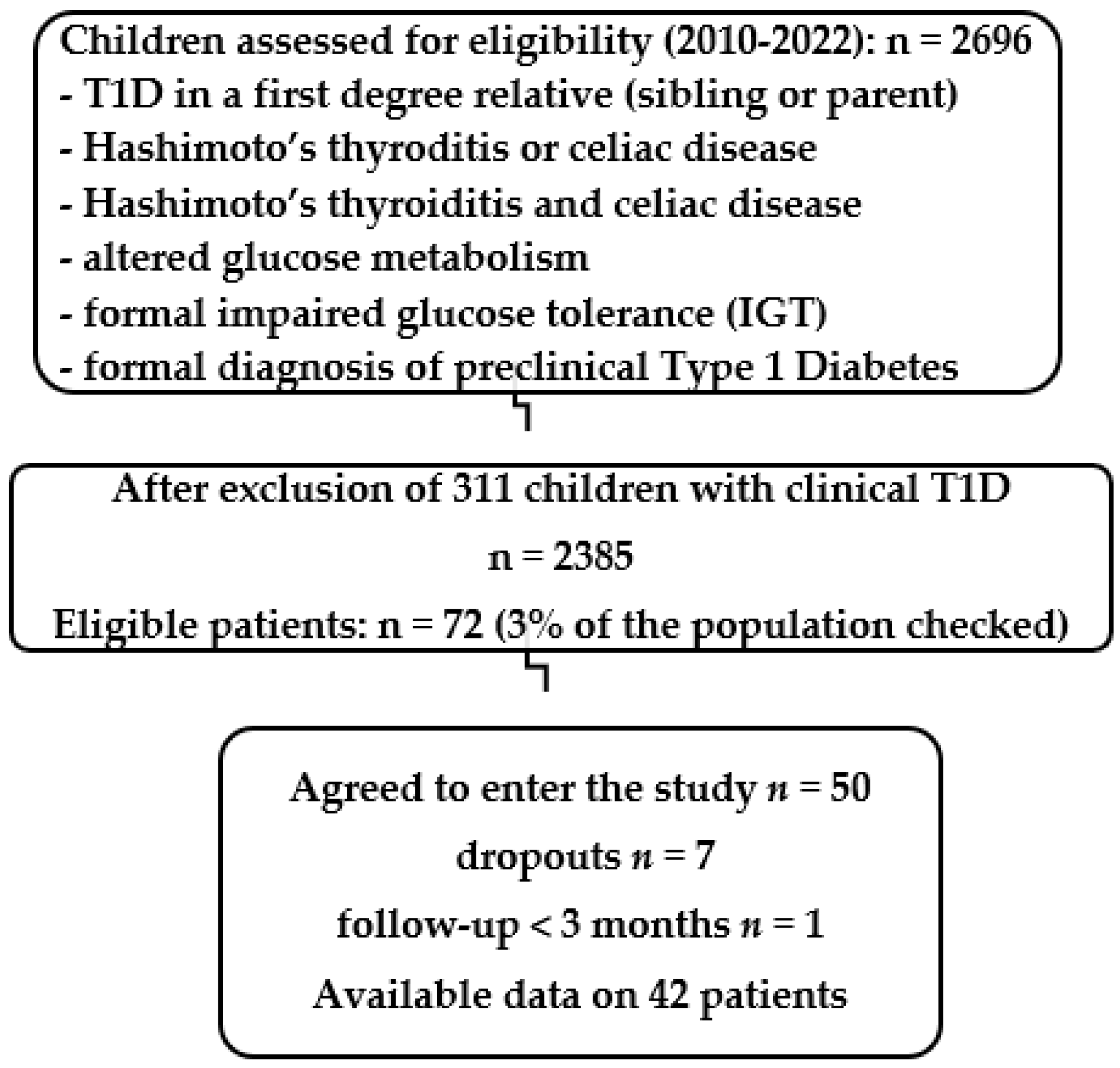
2. Materials and Methods
3. Results
3.1. Aggregated Data (Table 1)
| # | Gender | Screening Criteria | Age Years | 25(OH)D ng/ml | T1Ab(s) | Titer | HLA (PRE/PRO) Predisposing Protective | Treatment Dose (mcg) | Evolution: Insulin/ Years to Negativation/ Drops Out | Titer after Treatment | Follow-Up (Years) after Negativation | Evolution, Relapses, Adverse Events, Thyroid—Celiac Abs Negativation |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Single T1Ab-positive | Paricalcitol (P) Calcitriol (C) | |||||||||||
| 1 | Μ | T1D | 3.80 | 43.0 | IAA | 1.8 < 1.1 | PRE A24 | C 0.25 × 1 | Insulin immediately | |||
| 2 | M | T1D | 2.49 | 40.0 | IAA | 1.4 < 1.1 | N/A | P 2 × 3 | Insulin immediately | |||
| 3 | F | T1D | 9.28 | 13.1 | IA2 | 394 < 10 | N/A | C 0.5 × 1 | Insulin immediately | |||
| 4 | M | T1D | 7.80 | 29.0 | IAA | 250 < 10 | N/A | P 4 × 3 | drop out | |||
| 5 | M | Pre-T1D Hashimoto’s | 8.45 | 24.3 | GAD | 34.2 < 10 | N/A | P 2 × 3 | Insulin in 6 months | |||
| 6 | M | Pre-T1D | 11.91 | 33.1 | IAA | 1.3 < 1.1 | N/A | C 0.5 × 1 | 1.92 | 1.0 < 1.1 | 1.52 | |
| 7 | F | IGT | 10.59 | 20.0 | IAA | 10 < 10 | N/A | C 0.5 × 1 | 0.51 | 7.5 < 10 | 2.55 | Normalized glucose metabolism |
| 8 | F | IGT | 13.48 | 18.3 | IAA | 1.4 < 1.1 | PRE A24 | C 0.5 × 2 | 0.32 | 1.0 < 1.1 | 2.00 | Normalized glucose metabolism |
| 9 | M | IGT | 14.97 | 22.1 | ICA | 1.3 < 1.1 | N/A | C 0.5 × 3 | 0.90 | 0.9 < 1.1 | 4.02 | Normalized glucose metabolism |
| 10 | Μ | IGT | 12.25 | 27.0 | ICA | 1.1 < 1.1 | N/A | C 0.5 × 1 | 0.48 | 0.4 < 1.1 | 1.80 | Normalized glucose metabolism |
| 11 | M | IGT | 4.55 | 18.7 | IAA | 2.7 < 1.1 | PRE A02 | P 6 × 3 | 0.95 | 1.0 < 1.1 | 2.35 | Relapse, hypercalciuria |
| 12 | M | IGT | 8.94 | 21.3 | ΙAA | 1.3 < 1.1 | N/A | P 2 × 1 | 0.30 | 0.9 < 1.1 | 2.00 | Normalized glucose metabolism |
| 13 | F | IGT | 13.45 | 15.6 | IAA | 2.2 < 1.1 | N/A | C 0.5 × 1 | drop out | |||
| 14 | F | IGT | 4.92 | 44.8 | GAD | 47 < 10 | N/A | P 1 × 3 | 0.30 | 1.2 < 10 | 0.30 | Normalized glucose metabolism |
| 15 | M | IGT | 6.37 | 22.1 | ICA | 1.26 < 1.1 | N/A | P 2 × 3 | drop out | |||
| 16 | M | Mother T1D | 4.10 | 18.3 | IAA | 1.52 < 1.1 | PRE DR4 | C 0.5 × 2 | 0.57 | 0.34 < 1.1 | 0.60 | |
| 17 | F | Sibling T1D | 11.35 | 28.3 | IA2 | 16 < 10 | N/A | C 0.25 × 3 | 0.53 | 4.50 < 10 | 2.70 | |
| 18 | F | Sibling T1D | 1.31 | 22.0 | IAA | 1.39 < 1.1 | N/A | P 4 × 3 | 1.30 | 0.24 < 1.1 | 3.24 | Relapse, hypercalciuria, hypercalcemia |
| 19 | F | Father T1D | 4.91 | 20.0 | IA2 | 37.7 < 10 | PRE DR4 | P 2 × 3 | 0.50 | 2.0 < 10 | 2.50 | |
| 20 | M | Father T1D | 8.38 | 38.0 | ΙAA | 1.8 < 1.1 | PRE A02 | P 2 × 3 | Follow-up <3 m | |||
| 21 | Μ | Sibling T1D | 4.09 | 18.0 | ΙAA | 2.9 < 1.1 | N/A | C 0.5 × 1 | drop out | |||
| 22 | F | Sibling T1D | 10.67 | 33.8 | IAA | 1.8 < 1.1 | PRE A02/DR4 | C 0.5 × 1 | 1.9 | 1.0 < 1.1 | 9.24 | Relapse, hypercalciuria |
| 23 | F | Hashimoto’s Celiac disease | 9.28 | 27.0 | IAA | 1.3 < 1.1 | N/A | C 0.25 × 1 | 0.9 | 0.2 < 1.1 | 5.20 | |
| 24 | M | Hashimoto’s Celiac disease | 13.17 | 26.6 | GAD | 32 < 10 | PRE A02 | P 2 × 3 | 0.5 | 4 < 10 | 4.13 | |
| 25 | F | Hashimoto’s Celiac disease | 10.04 | 26.8 | ICA | 2.4 < 1.1 | PRO DQA10102 PRE DQA10501 | P 2 × 3 | 1.0 | 0.25 < 1.1 | 1.68 | |
| 26 | F | Hashimoto’s Celiac disease | 12.54 | 23.8 | IAA | 1.1 < 1.1 | N/A | C 0.5 × 1 | 0.46 | 0.5 < 1.1 | 5.00 | |
| 27 | F | Celiac disease | 4.13 | 27.8 | IAA | 1.8 < 1.1 | N/A | P 2 × 3 | 1.25 | 0.7 < 1.1 | 2.38 | Hypercalciuria, hypercalcemia |
| 28 | M | Celiac disease | 13.03 | 17.4 | ΙAA | 1.1 < 1.1 | N/A | C 0.5 × 1 | 0.60 | 0.9 < 1.1 | 5.86 | |
| 29 | F | Celiac disease | 2.84 | 12.5 | ΙAA | 1.4 < 1.1 | PRE DQA10105 | C 0.5 × 1 | 1.60 | 0.9 < 1.1 | 2.70 | Relapse, hypercalciuria, negativized celiac disease Abs |
| 30 | F | Hashimoto’s | 3.28 | 19.5 | ICA | 1.5 < 1.1 | PRE A02 | C 0.5 × 1 | 0.37 | 0.1 < 1.1 | 0.37 | |
| 31 | Μ | Hashimoto’s | 2.18 | 19.0 | IA2 | 16.2 < 10 | N/A | C 0.5 × 1 | 1.20 | 8.5 < 10 | 6.80 | Negativized thyroid Abs |
| 32 | F | Hashimoto’s | 0.65 | 22.0 | IAA | 1.5 < 1.1 | N/A | P 1 × 2 | 0.30 | 0.9 < 1.1 | 2.60 | Hypercalciuria, hypercalcemia, negativized thyroid Abs |
| 33 | M | Hashimoto’s | 1.40 | 30.4 | IAA | 8.3 < 1.1 | N/A | P 1 × 3 | 0.50 | 0.52 < 1.1 | 0.90 | Hypercalcemia, negativized thyroid Abs |
| 34 | F | Hashimoto’s | 2.47 | 17.0 | ΙAA | 1.2 < 1.1 | N/A | C 0.25 × 1 | drop out | |||
| Multiple T1Ab positive | N/A | |||||||||||
| 35 | F | T1D | 4.37 | 18.7 | IAA GAD | 1.2 < 1.1 54 < 10 | N/A | P 2 × 3 | Insulin in 0.80 years | |||
| 36 | Μ | T1D | 16.37 | 18.3 | ΙA2 GAD | 37.5 < 10 188 < 10 | PRE DR4 | P 12 × 3 | Insulin in 0.85 years | |||
| 37 | F | Τ1D | 6.65 | 22.7 | IAA ICA GAD | 1.3 < 1.1 1.2 < 1.1 79 < 10 | PRE DRB10404 | C 0.25 × 1 | Insulin in 2 years | |||
| 38 | F | T1D | 5.09 | 19.6 | IA2 GAD | 400 < 10 125.2 < 10 | N/A | P 8 × 3 | Insulin in 0.95 years | Negativized Celiac disease Abs | ||
| 39 | F | T1D | 6.10 | 25.7 | ΙCA GAD | 4.75 < 1.1 280 < 10 | N/A | P 2 × 3 | Insulin in 1 week | |||
| 40 | M | Pre-T1D | 10.53 | 22.0 | IAA GAD | 1.3 < 1.1 93 < 10 | N/A | C 0.5 × 3 | Insulin in 3 years | |||
| 41 | M | Hashimoto’s | 1.85 | 15.8 | IAA GAD | 1.3 < 1.1 12 < 10 | N/A | C 0.25 × 1 | 0.41 | 0.5 < 1.1 6.0 < 10 | 8.05 | Negativized thyroid Abs |
| 42 | Μ | Sibling T1D | 1.90 | 21.0 | ICA GAD | 1.2 < 1.1 12 < 10 | N/A | P 1 × 3 | 0.58 | 0.44 < 1.1 4.0 < 10 | 1.00 | Relapse |
| 43 | M | Mother T1D | 11.32 | 20.7 | IAA ICA | 1.2 < 1.1 1.2 < 1.1 | N/A | C 0.5 × 1 | Drop out | |||
| 44 | Μ | Mother T1D | 4.90 | 25.5 | ΙAA GAD | 1.2 < 1.1 1.2 < 1 | N/A | P 2 × 1 | Drop out | |||
| HLA Predisposing | ||||||||||||
| 45 | F | Mother T1D | 8.30 | 13.0 | - | PRE A24 | C 0.5 × 1 | 4.5 | ||||
| 46 | F | Mother T1D | 10.29 | 15.0 | - | PRE A24 | C 0.5 × 1 | 4.5 | ||||
| 47 | M | Sibling T1D | 2.49 | 30.8 | - | PRE A24 | C 0.5 × 1 | 1.0 | ||||
| 48 | F | Father T1D | 11.78 | 19.5 | - | PRE A24, DR3 | C 0.5 × 3 | 1.0 | ||||
| 49 | M | Hashimoto’s Celiac disease | 12.25 | 16.0 | - | PRE A02 | C 0.5 × 1 | 1.0 | ||||
| 50 | M | Pre-T1D | 3.88 | 29.9 | - | PRE A02 | C 0.25 × 2 | 3.5 | Hypercalciuria | |||
3.2. Single T1Ab-Positive Children
Relapses in Single T1Ab-Positive Children Who Achieved Reversion of Seroconversion
3.3. Multiple T1Ab Positive Children
3.4. Children with Predisposing HLA
3.5. Observed Effects on Thyroid and Celiac Disease Related Abs
3.6. Vitamin D Status
3.7. Adverse Events: Hypercalcemia, Hypercalciuria and Nephrocalcinosis
3.8. Statistical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roep, B.O.; Thomaidou, S.; van Tienhoven, R.; Zaldumbide, A. Type 1 diabetes mellitus as a disease of the β-cell (do not blame the immune system?). Nat. Rev. Endocrinol. 2021, 17, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Pociot, F.; Lernmark, Å. Genetic risk factors for type 1 diabetes. Lancet 2016, 387, 2331–2339. [Google Scholar] [CrossRef]
- Pociot, F.; Akolkar, B.; Concannon, P.; Erlich, H.A.; Julier, C.; Morahan, G.; Nierras, C.R.; Todd, J.A.; Rich, S.S.; Nerup, J. Genetics of Type 1 Diabetes: What’s Next? Diabetes 2010, 59, 1561–1571. [Google Scholar] [CrossRef] [PubMed]
- Rewers, M.; Ludvigsson, J. Environmental risk factors for type 1 diabetes. Lancet 2016, 387, 2340–2348. [Google Scholar] [CrossRef] [PubMed]
- Katsarou, A.; Gudbjörnsdottir, S.; Rawshani, A.; Dabelea, D.; Bonifacio, E.; Anderson, B.J.; Jacobsen, L.M.; Schatz, D.A.; Lernmark, Å. Type 1 Diabetes mellitus. Nat. Rev. Dis. Prim. 2017, 3, 17016. [Google Scholar] [CrossRef]
- Insel, R.A.; Dunne, J.L.; Atkinson, M.A.; Chiang, J.L.; Dabelea, D.; Gottlieb, P.A.; Greenbaum, C.J.; Herold, K.C.; Krischer, J.P.; Lernmark, Å.; et al. Staging Presymptomatic Type 1 Diabetes: A Scientific Statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 2015, 38, 1964–1974. [Google Scholar] [CrossRef]
- Oram, R.A.; Sims, E.K.; Evans-Molina, C. Beta cells in type 1 diabetes: Mass and function; sleeping or dead? Diabetologia 2019, 62, 567–577. [Google Scholar] [CrossRef]
- Yu, L.; Dong, F.; Miao, D.; Fouts, A.R.; Wenzlau, J.M.; Steck, A.K. Proinsulin/Insulin Autoantibodies Measured With Electrochemiluminescent Assay Are the Earliest Indicator of Prediabetic Islet Autoimmunity. Diabetes Care 2013, 36, 2266–2270. [Google Scholar] [CrossRef]
- Krischer, J.P.; Lynch, K.F.; Schatz, D.A.; Ilonen, J.; Lernmark, Å.; Hagopian, W.A.; Rewers, M.J.; She, J.-X.; Simell, O.G.; Toppari, J.; et al. The 6 year incidence of diabetes-associated autoantibodies in genetically at-risk children: The TEDDY study. Diabetologia 2015, 58, 980–987. [Google Scholar] [CrossRef]
- Ziegler, A.G.; Rewers, M.; Simell, O.; Simell, T.; Lempainen, J.; Steck, A.; Winkler, C.; Ilonen, J.; Veijola, R.; Knip, M.; et al. Seroconversion to Multiple Islet Autoantibodies and Risk of Progression to Diabetes in Children. JAMA 2013, 309, 2473–2479. [Google Scholar] [CrossRef]
- Steck, A.K.; Johnson, K.; Barriga, K.J.; Miao, D.; Yu, L.; Hutton, J.C.; Eisenbarth, G.S.; Rewers, M.J. Age of islet autoantibody appearance and mean levels of insulin, but not GAD or IA-2 autoantibodies, predict age of diagnosis of type 1 diabetes: Diabetes autoimmunity study in the young. Diabetes Care 2011, 34, 1397–1399. [Google Scholar] [CrossRef]
- Steck, A.K.; Vehik, K.; Bonifacio, E.; Lernmark, A.; Ziegler, A.-G.; Hagopian, W.A.; She, J.; Simell, O.; Akolkar, B.; Krischer, J.; et al. Predictors of Progression From the Appearance of Islet Autoantibodies to Early Childhood Diabetes: The Environmental Determinants of Diabetes in the Young (TEDDY). Diabetes Care 2015, 38, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Vehik, K.; Lynch, K.F.; Schatz, D.A.; Akolkar, B.; Hagopian, W.; Rewers, M.; She, J.-X.; Simell, O.; Toppari, J.; Ziegler, A.-G.; et al. Reversion of β-Cell Autoimmunity Changes Risk of Type 1 Diabetes: TEDDY Study. Diabetes Care 2016, 39, 1535–1542. [Google Scholar] [CrossRef]
- Knip, M.; Korhonen, S.; Kulmala, P.; Veijola, R.; Reunanen, A.; Raitakari, O.T.; Viikari, J.; Åkerblom, H.K. Prediction of Type 1 Diabetes in the General Population. Diabetes Care 2010, 33, 1206–1212. [Google Scholar] [CrossRef]
- Noble, J.A.; Valdes, A.M. Genetics of the HLA Region in the Prediction of Type 1 Diabetes. Curr. Diabetes Rep. 2011, 11, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Pöllänen, P.M.; Ryhänen, S.J.; Toppari, J.; Ilonen, J.; Vähäsalo, P.; Veijola, R.; Siljander, H.; Knip, M. Dynamics of Islet Autoantibodies During Prospective Follow-Up From Birth to Age 15 Years. J. Clin. Endocrinol. Metab. 2020, 105, e4638–e4651. [Google Scholar] [CrossRef]
- Chakhtoura, M.; Azar, S.T. The Role of Vitamin D Deficiency in the Incidence, Progression, and Complications of Type 1 Diabetes Mellitus. Int. J. Endocrinol. 2013, 2013, 148673. [Google Scholar] [CrossRef] [PubMed]
- Zabeen, B.; Nahar, J.; Ahmed, B.; Tayyeb, S.; Islam, N.; Azad, K. Vitamin D status in children and adolescents with type 1 diabetes in a specialized diabetes care centre in Bangladesh. Endocrinol. Diabetes Metab. 2021, 5, e00312. [Google Scholar] [CrossRef]
- Norris, J.M.; Lee, H.-S.; Frederiksen, B.; Erlund, I.; Uusitalo, U.; Yang, J.; Lernmark, Å.; Simell, O.; Toppari, J.; Rewers, M.; et al. Plasma 25-Hydroxyvitamin D Concentration and Risk of Islet Autoimmunity. Diabetes 2018, 67, 146–154. [Google Scholar] [CrossRef]
- Kirac, D.; Yazan, C.D.; Gezmis, H.; Yaman, A.; Haklar, G.; Sirikci, O.; Altunok, E.C.; Deyneli, O. VDBP, VDR Mutations and Other Factors Related With Vitamin D Metabolism May Be Associated With Type 1 Diabetes Mellitus. Cell. Mol. Biol. 2018, 64, 11–16. [Google Scholar]
- Tapia, G.; Mårild, K.; Dahl, S.R.; Lund-Blix, N.A.; Viken, M.K.; Lie, B.A.; Njølstad, P.R.; Joner, G.; Skrivarhaug, T.; Cohen, A.S.; et al. Maternal and Newborn Vitamin D–Binding Protein, Vitamin D Levels, Vitamin D Receptor Genotype, and Childhood Type 1 Diabetes. Diabetes Care 2019, 42, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.A.; Rahimzadeh, M.; Brierley, C.; Gration, B.; Doree, C.; Kimber, C.E.; Cajide, A.P.; Lamikanra, A.A.; Roberts, D.J. The role of vitamin D in increasing circulating T regulatory cell numbers and modulating T regulatory cell phenotypes in patients with inflammatory disease or in healthy volunteers: A systematic review. PLoS ONE 2019, 14, e0222313. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Sharma, P.; Girgis, C.M.; Gunton, J.E. Vitamin D and Beta Cells in Type 1 Diabetes: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 14434. [Google Scholar] [CrossRef]
- Rak, K.; Bronkowska, M. Immunomodulatory Effect of Vitamin D and Its Potential Role in the Prevention and Treatment of Type 1 Diabetes Mellitus—A Narrative Review. Molecules 2018, 24, 53. [Google Scholar] [CrossRef] [PubMed]
- Di Rosa, M.; Malaguarnera, M.; Nicoletti, F.; Malaguarnera, L. Vitamin D3: A helpful immuno-modulator. Immunology 2011, 134, 123–139. [Google Scholar] [CrossRef]
- Kittaka, A.; Saito, N.; Honzawa, S.; Takenouchi, K.; Ishizuka, S.; Chen, T.C.; Peleg, S.; Kato, S.; Arai, M.A. Creative synthesis of novel vitamin D analogs for health and disease. J. Steroid Biochem. Mol. Biol. 2007, 103, 269–276. [Google Scholar] [CrossRef]
- Maestro, M.A.; Seoane, S. The Centennial Collection of VDR Ligands: Metabolites, Analogs, Hybrids and Non-Secosteroidal Ligands. Nutrients 2022, 14, 4927. [Google Scholar] [CrossRef]
- Leyssens, C.; Verlinden, L.; Verstuyf, A. The future of vitamin D analogs. Front. Physiol. 2014, 5, 122. [Google Scholar] [CrossRef]
- Bothou, C.; Alexopoulos, A.; Dermitzaki, E.; Kleanthous, K.; Papadimitriou, A.; Mastorakos, G.; Papadimitriou, D.T. Successful Treatment of Severe Atopic Dermatitis with Calcitriol and Paricalcitol in an 8-Year-Old Girl. Case Rep. Pediatr. 2018, 2018, 9643543. [Google Scholar] [CrossRef]
- Papadimitriou, D.T.; Bothou, C.; Dermitzaki, E.; Alexopoulos, A.; Mastorakos, G. Treatment of alopecia totalis/universalis/focalis with vitamin D and analogs: Three case reports and a literature review. World J. Clin. Pediatr. 2021, 10, 192–199. [Google Scholar] [CrossRef]
- Zaveri, N.T.; Murphy, B.J. 2.25—Nuclear Hormone Receptors. In Comprehensive Medicinal Chemistry II; Taylor, J.B., Triggle, D.J., Eds.; Elsevier: Oxford, UK, 2007; pp. 993–1036. [Google Scholar]
- Mathieu, C.; Waer, M.; Laureys, J.; Rutgeerts, O.; Bouillon, R. Prevention of autoimmune diabetes in NOD mice by 1,25 dihydroxyvitamin D3. Diabetologia 1994, 37, 552–558. [Google Scholar] [CrossRef]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Hyppönen, E.; Läärä, E.; Reunanen, A.; Järvelin, M.-R.; Virtanen, S.M. Intake of vitamin D and risk of type 1 diabetes: A birth-cohort study. Lancet 2001, 358, 1500–1503. [Google Scholar] [CrossRef] [PubMed]
- Karvonen, M.; Pitkäniemi, J.; Tuomilehto, J. The onset age of type 1 diabetes in Finnish children has become younger. The Finnish Childhood Diabetes Registry Group. Diabetes Care 1999, 22, 1066–1070. [Google Scholar] [CrossRef] [PubMed]
- Harjutsalo, V.; Sund, R.; Knip, M.; Groop, P.-H. Incidence of Type 1 Diabetes in Finland. Jama 2013, 310, 427–428. [Google Scholar] [CrossRef]
- Ghalwash, M.; Dunne, J.L.; Lundgren, M.; Rewers, M.; Ziegler, A.-G.; Anand, V.; Toppari, J.; Veijola, R.; Hagopian, W. Two-age islet-autoantibody screening for childhood type 1 diabetes: A prospective cohort study. Lancet Diabetes Endocrinol. 2022, 10, 589–596. [Google Scholar] [CrossRef]
- Beik, P.; Ciesielska, M.; Kucza, M.; Kurczewska, A.; Kuźmińska, J.; Maćkowiak, B.; Niechciał, E. Prevention of Type 1 Diabetes: Past Experiences and Future Opportunities. J. Clin. Med. 2020, 9, 2805. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, D.; Marakaki, C.; Fretzayas, A.; Nicolaidou, P.; Papadimitriou, A. Negativation of type 1 diabetes-associated autoantibodies to glutamic acid decarboxylase and insulin in children treated with oral calcitriol. J. Diabetes 2013, 5, 344–348. [Google Scholar] [CrossRef]
- Papadimitriou, D.T. High Doses of Oral Calcitriol (up to 6 μg/day) and Paricalcitol (up to 72 μg/day) Have Successfully Intercepted Progression to Clinical Type 1 Diabetes for over 3 Years in a 10-Year-Old Boy. Endocr Rev. 2017. [CrossRef]
- Holick, M.F. The One-Hundred-Year Anniversary of the Discovery of the Sunshine Vitamin D3: Historical, Personal Experience and Evidence-Based Perspectives. Nutrients 2023, 15, 593. [Google Scholar] [CrossRef]
- Hoppe, B.; Kemper, M.J. Diagnostic examination of the child with urolithiasis or nephrocalcinosis. Pediatr. Nephrol. 2008, 25, 403–413. [Google Scholar] [CrossRef]
- Bingley, P.J.; Bonifacio, E.; Ziegler, A.-G.; Schatz, D.A.; Atkinson, M.A.; Eisenbarth, G.S.; Society, T.I.O.D. Proposed Guidelines on Screening for Risk of Type 1 Diabetes. Diabetes Care 2001, 24, 398. [Google Scholar] [CrossRef]
- Herold, K.C.; Bundy, B.N.; Long, S.A.; Bluestone, J.A.; DiMeglio, L.A.; Dufort, M.; Gitelman, S.E.; Gottlieb, P.A.; Krischer, J.P.; Linsley, P.S.; et al. An Anti-CD3 Antibody, Teplizumab, in Relatives at Risk for Type 1 Diabetes. N. Engl. J. Med. 2019, 381, 603–613. [Google Scholar] [CrossRef]
- Abela, A.G.; Fava, S. Why is the Incidence of Type 1 Diabetes Increasing? Curr. Diabetes Rev. 2021, 17, e030521193110. [Google Scholar] [CrossRef]
- Passanisi, S.; Salzano, G.; Aloe, M.; Bombaci, B.; Citriniti, F.; De Berardinis, F.; De Marco, R.; Lazzaro, N.; Lia, M.C.; Lia, R.; et al. Increasing trend of type 1 diabetes incidence in the pediatric population of the Calabria region in 2019–2021. Ital. J. Pediatr. 2022, 48, 66. [Google Scholar] [CrossRef]
- Papadimitriou, D.T.; Vassaras, A.K.; Holick, M.F. Association between population vitamin D status and SARS-CoV-2 related serious-critical illness and deaths: An ecological integrative approach. World J. Virol. 2021, 10, 111–129. [Google Scholar] [CrossRef] [PubMed]
- Salah, N.Y.; Madkour, S.S.; Soliman, K.S. Pancreatic shear wave elastography in children with type 1 diabetes: Relation to diabetes duration, glycemic indices, fasting C-peptide and diabetic complications. Pediatr. Radiol. 2022, 52, 2348–2358. [Google Scholar] [CrossRef]
- Wenzlau, J.M.; Juhl, K.; Yu, L.; Moua, O.; Sarkar, S.A.; Gottlieb, P.; Rewers, M.; Eisenbarth, G.S.; Jensen, J.; Davidson, H.W.; et al. The cation efflux transporter ZnT8 (Slc30A8) is a major autoantigen in human type 1 diabetes. Proc. Natl. Acad. Sci. USA 2007, 104, 17040–17045. [Google Scholar] [CrossRef] [PubMed]
- Schlosser, M.; Strebelow, M.; Rjasanowski, I.; Kerner, W.; Wassmuth, R.; Ziegler, M. Prevalence of Diabetes-Associated Autoantibodies in Schoolchildren: The Karlsburg Type 1 Diabetes Risk Study. Ann. N. Y. Acad. Sci. 2004, 1037, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Christie, M.R.; Roll, U.; Payton, M.A.; Hatfield, E.C.; Ziegler, A.-G. Validity of Screening for Individuals at Risk for Type I Diabetes by Combined Analysis of Antibodies to Recombinant Proteins. Diabetes Care 1997, 20, 965–970. [Google Scholar] [CrossRef]
- Redondo, M.J. On the road to universal screening for risk of type 1 diabetes. Lancet Diabetes Endocrinol. 2022, 10, 554–555. [Google Scholar] [CrossRef]
- Greer, R.M.; Portelli, S.L.; Hung, B.S.-M.; Cleghorn, G.J.; McMahon, S.K.; Batch, J.A.; Conwell, L.S. Serum vitamin D levels are lower in Australian children and adolescents with type 1 diabetes than in children without diabetes. Pediatr. Diabetes 2013, 14, 31–41. [Google Scholar] [CrossRef]
- Bener, A.; Alsaied, A.; Al-Ali, M.; Al-Kubaisi, A.; Basha, B.; Abraham, A.; Guiter, G.; Mian, M. High prevalence of vitamin D deficiency in type 1 diabetes mellitus and healthy children. Acta Diabetol. 2008, 46, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, M.E.; Niinistö, S.; Erlund, I.; Cuthbertson, D.; Nucci, A.M.; Honkanen, J.; Vaarala, O.; Hyöty, H.; Krischer, J.P.; Knip, M.; et al. Serum 25-hydroxyvitamin D concentration in childhood and risk of islet autoimmunity and type 1 diabetes: The TRIGR nested case–control ancillary study. Diabetologia 2020, 63, 780–787. [Google Scholar] [CrossRef]
- Fronczak, C.M.; Barón, A.E.; Chase, H.P.; Ross, C.; Brady, H.L.; Hoffman, M.; Eisenbarth, G.S.; Rewers, M.; Norris, J.M. In Utero Dietary Exposures and Risk of Islet Autoimmunity in Children. Diabetes Care 2003, 26, 3237–3242. [Google Scholar] [CrossRef]
- Chiu, K.C.; Chu, A.; Go, V.L.W.; Saad, M.F. Hypovitaminosis D is associated with insulin resistance and β cell dysfunction. Am. J. Clin. Nutr. 2004, 79, 820–825. [Google Scholar] [CrossRef]
- Stene, L.C.; Joner, G. Use of cod liver oil during the first year of life is associated with lower risk of childhood-onset type 1 diabetes: A large, population-based, case-control study. Am. J. Clin. Nutr. 2003, 78, 1128–1134. [Google Scholar] [CrossRef]
- Zipitis, C.S.; Akobeng, A.K. Vitamin D supplementation in early childhood and risk of type 1 diabetes: A systematic review and meta-analysis. Arch. Dis. Child. 2008, 93, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.-Y.; Zhang, W.; Chen, J.J.; Zhang, Z.-L.; Han, S.-F.; Qin, L.-Q. Vitamin D Intake and Risk of Type 1 Diabetes: A Meta-Analysis of Observational Studies. Nutrients 2013, 5, 3551–3562. [Google Scholar] [CrossRef]
- Jacobsen, R.; Moldovan, M.; Vaag, A.A.; Hypponen, E.; Heitmann, B.L. Vitamin D fortification and seasonality of birth in type 1 diabetic cases: D-tect study. J. Dev. Orig. Health Dis. 2015, 7, 114–119. [Google Scholar] [CrossRef] [PubMed]
- García, D.; Angel, B.; Carrasco, E.; Albala, C.; Santos, J.L.; Pérez-Bravo, F. VDR polymorphisms influence the immune response in type 1 diabetic children from Santiago, Chile. Diabetes Res. Clin. Pr. 2007, 77, 134–140. [Google Scholar] [CrossRef]
- Pani, M.A.; Knapp, M.; Donner, H.; Braun, J.; Baur, M.P.; Usadel, K.H.; Badenhoop, K. Vitamin D receptor allele combinations influence genetic susceptibility to type 1 diabetes in Germans. Diabetes 2000, 49, 504–507. [Google Scholar] [CrossRef]
- Kamel, M.M.; Fouad, S.A.; Salaheldin, O.; El-Razek, A.E.-R.A.A.; El-Fatah, A.I.A. Impact of vitamin D receptor gene polymorphisms in pathogenesis of Type-1 diabetes mellitus. Int. J. Clin. Exp. Med. 2014, 7, 5505–5510. [Google Scholar] [PubMed]
- Ongagna, J.; Pinget, M.; Belcourt, A. Vitamin D-binding protein gene polymorphism association with IA-2 autoantibodies in type 1 diabetes. Clin. Biochem. 2005, 38, 415–419. [Google Scholar] [CrossRef]
- Ongagna, J.; Kaltenbacher, M.; Sapin, R.; Pinget, M.; Belcourt, A. The HLA-DQB alleles and amino acid variants of the vitamin D-binding protein in diabetic patients in Alsace. Clin. Biochem. 2001, 34, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Blanton, D.; Han, Z.; Bierschenk, L.; Linga-Reddy, M.P.; Wang, H.; Clare-Salzler, M.; Haller, M.; Schatz, D.; Myhr, C.; She, J.-X.; et al. Reduced Serum Vitamin D–Binding Protein Levels Are Associated With Type 1 Diabetes. Diabetes 2011, 60, 2566–2570. [Google Scholar] [CrossRef]
- Sørensen, I.M.; Joner, G.; Jenum, P.A.; Eskild, A.; Brunborg, C.; Torjesen, P.A.; Stene, L.C. Vitamin D-binding protein and 25-hydroxyvitamin D during pregnancy in mothers whose children later developed type 1 diabetes. Diabetes/Metab. Res. Rev. 2016, 32, 883–890. [Google Scholar] [CrossRef]
- Felício, K.M.; De Souza, A.C.C.B.; Neto, J.F.A.; De Melo, F.T.C.; Carvalho, C.T.; Arbage, T.P.; Brito, H.A.D.R.; Peixoto, A.S.; De Oliveira, A.F.; Resende, F.D.S.; et al. Glycemic Variability and Insulin Needs in Patients with Type 1 Diabetes Mellitus Supplemented with Vitamin D: A Pilot Study Using Continuous Glucose Monitoring System. Curr. Diabetes Rev. 2018, 14, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Bogdanou, D.; Penna-Martinez, M.; Filmann, N.; Chung, T.; Moran-Auth, Y.; Wehrle, J.; Cappel, C.; Huenecke, S.; Herrmann, E.; Koehl, U.; et al. T-lymphocyte and glycemic status after vitamin D treatment in type 1 diabetes: A randomized controlled trial with sequential crossover. Diabetes/Metab. Res. Rev. 2017, 33, e2865. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liao, L.; Yan, X.; Huang, G.; Lin, J.; Lei, M.; Wang, X.; Zhou, Z. Protective effects of 1-α-hydroxyvitamin D3 on residual β-cell function in patients with adult-onset latent autoimmune diabetes (LADA). Diabetes/Metab. Res. Rev. 2009, 25, 411–416. [Google Scholar] [CrossRef]
- Ataie-Jafari, A.; Loke, S.-C.; Rahmat, A.B.; Larijani, B.; Abbasi, F.; Leow, M.K.; Yassin, Z. A randomized placebo-controlled trial of alphacalcidol on the preservation of beta cell function in children with recent onset type 1 diabetes. Clin. Nutr. 2013, 32, 911–917. [Google Scholar] [CrossRef]
- Pitocco, D.; Crino, A.; Di Stasio, E.; Manfrini, S.; Guglielmi, C.; Spera, S.; Anguissola, G.B.; Visalli, N.; Suraci, C.; Matteoli, M.C.; et al. The effects of calcitriol and nicotinamide on residual pancreatic β-cell function in patients with recent-onset Type 1 diabetes (IMDIAB XI). Diabet. Med. 2006, 23, 920–923. [Google Scholar] [CrossRef] [PubMed]
- Giri, D.; Pintus, D.; Burnside, G.; Ghatak, A.; Mehta, F.; Paul, P.; Senniappan, S. Treating vitamin D deficiency in children with type I diabetes could improve their glycaemic control. BMC Res. Notes 2017, 10, 465. [Google Scholar] [CrossRef]
- Chen, S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.; Lipsky, P.E. Modulatory Effects of 1,25-Dihydroxyvitamin D3 on Human B Cell Differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef]
- Pincikova, T.; Nilsson, K.; Moen, I.E.; Karpati, F.; Fluge, G.; Hollsing, A.; Knudsen, P.K.; Lindblad, A.; Mared, L.; Pressler, T.; et al. Inverse relation between vitamin D and serum total immunoglobulin G in the Scandinavian Cystic Fibrosis Nutritional Study. Eur. J. Clin. Nutr. 2010, 65, 102–109. [Google Scholar] [CrossRef]
- Peelen, E.; Rijkers, G.; Meerveld-Eggink, A.; Meijvis, S.; Vogt, M.; Tervaert, J.W.C.; Hupperts, R.; Damoiseaux, J. Relatively high serum vitamin D levels do not impair the antibody response to encapsulated bacteria. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 61–69. [Google Scholar] [CrossRef]
- McGrath, J. Does ‘imprinting’ with low prenatal vitamin D contribute to the risk of various adult disorders? Med. Hypotheses 2001, 56, 367–371. [Google Scholar] [CrossRef]
- Bizzarri, C.; Pitocco, D.; Napoli, N.; Di Stasio, E.; Maggi, D.; Manfrini, S.; Suraci, C.; Cavallo, M.G.; Cappa, M.; Ghirlanda, G.; et al. No protective effect of calcitriol on beta-cell function in recent-onset type 1 diabetes: The IMDIAB XIII trial. Diabetes Care 2010, 33, 1962–1963. [Google Scholar] [CrossRef]
- Hmami, F.; Oulmaati, A.; Amarti, A.; Kottler, M.-L.; Bouharrou, A. Overdose or hypersensitivity to vitamin D? Arch. Pediatr. Organe Off. Soc. Fr. Pediatr. 2014, 21, 1115–1119. [Google Scholar] [CrossRef] [PubMed]
- Garland, C.F.; Kim, J.J.; Mohr, S.B.; Gorham, E.D.; Grant, W.B.; Giovannucci, E.L.; Baggerly, L.; Hofflich, H.; Ramsdell, J.W.; Zeng, K.; et al. Meta-analysis of All-Cause Mortality According to Serum 25-Hydroxyvitamin D. Am. J. Public Health 2014, 104, e43–e50. [Google Scholar] [CrossRef] [PubMed]
- Marcinowska-Suchowierska, E.; Kupisz-Urbańska, M.; Łukaszkiewicz, J.; Płudowski, P.; Jones, G. Vitamin D Toxicity—A Clinical Perspective. Front. Endocrinol. 2018, 9, 550. [Google Scholar] [CrossRef] [PubMed]
- Vieth, R. Vitamin D Toxicity, Policy, and Science. J. Bone Miner. Res. 2007, 22, V64–V68. [Google Scholar] [CrossRef]
- Shroff, R.; Knott, C.; Rees, L. The virtues of vitamin D—But how much is too much? Pediatr. Nephrol. 2010, 25, 1607–1620. [Google Scholar] [CrossRef]
- Lifshitz, F. Pediatric Endocrinology; Marcel Dekker, Inc.: New York, NY, USA, 2003. [Google Scholar]
- Jones, G. Pharmacokinetics of vitamin D toxicity. Am. J. Clin. Nutr. 2008, 88, 582S–586S. [Google Scholar] [CrossRef]
- Slatopolsky, E.; Finch, J.; Ritter, C.; Denda, M.; Morrissey, J.; Brown, A.; DeLuca, H. A new analog of calcitriol, 19-nor-1,25-(OH)2D2, suppresses parathyroid hormone secretion in uremic rats in the absence of hypercalcemia. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 1995, 26, 852–860. [Google Scholar] [CrossRef]
- Goldenberg, M.M. Paricalcitol, a new agent for the management of secondary hyperparathyroidism in patients undergoing chronic renal dialysis. Clin. Ther. 1999, 21, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Carroll, M.F.; Schade, D.S. A practical approach to hypercalcemia. Am. Fam. Physician 2003, 67, 1959–1966. [Google Scholar]
- Pak, C.Y.; Sakhaee, K.; Moe, O.W.; Poindexter, J.; Adams-Huet, B.; Pearle, M.S.; Zerwekh, J.E.; Preminger, G.M.; Wills, M.R.; Breslau, N.A.; et al. Defining hypercalciuria in nephrolithiasis. Kidney Int. 2011, 80, 777–782. [Google Scholar] [CrossRef]
- Sims, E.K.; Besser, R.E.; Dayan, C.; Rasmussen, C.G.; Greenbaum, C.; Griffin, K.J.; Hagopian, W.; Knip, M.; Long, A.E.; Martin, F.; et al. Screening for Type 1 Diabetes in the General Population: A Status Report and Perspective. Diabetes 2022, 71, 610–623. [Google Scholar] [CrossRef] [PubMed]
- Oram, R.A.; Sharp, S.A.; Pihoker, C.; Ferrat, L.; Imperatore, G.; Williams, A.; Redondo, M.J.; Wagenknecht, L.; Dolan, L.M.; Lawrence, J.M.; et al. Utility of Diabetes Type–Specific Genetic Risk Scores for the Classification of Diabetes Type among Multiethnic Youth. Diabetes Care 2022, 45, 1124–1131. [Google Scholar] [CrossRef]
- Chmiel, R.; Giannopoulou, E.Z.; Winkler, C.; Achenbach, P.; Ziegler, A.-G.; Bonifacio, E. Progression from single to multiple islet autoantibodies often occurs soon after seroconversion: Implications for early screening. Diabetologia 2014, 58, 411–413. [Google Scholar] [CrossRef]
- Jonsdottir, B.; Larsson, C.; Lundgren, M.; Ramelius, A.; Jönsson, I.; Larsson, H.E. Childhood thyroid autoimmunity and relation to islet autoantibodies in children at risk for type 1 diabetes in the diabetes prediction in skåne (DiPiS) study. Autoimmunity 2018, 51, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Simell, S.; Hoppu, S.; Simell, T.; Ståhlberg, M.-R.; Viander, M.; Routi, T.; Simell, V.; Veijola, R.; Ilonen, J.; Hyöty, H.; et al. Age at Development of Type 1 Diabetes– and Celiac Disease–Associated Antibodies and Clinical Disease in Genetically Susceptible Children Observed From Birth. Diabetes Care 2010, 33, 774–779. [Google Scholar] [CrossRef]
- Ferrat, L.A.; Vehik, K.; Sharp, S.A.; Lernmark, Å.; Rewers, M.J.; She, J.-X.; Ziegler, A.-G.; Toppari, J.; Akolkar, B.; Krischer, J.P.; et al. A combined risk score enhances prediction of type 1 diabetes among susceptible children. Nat. Med. 2020, 26, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Fabre, J. Modern medicine and the Hippocratic doctors of ancient Greece. J. R. Soc. Med. 1998, 91, 161–163. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Coke, R. Hippocratic philosophy. Rev. Med. Chile 1999, 127, 611–614. [Google Scholar] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papadimitriou, D.T.; Dermitzaki, E.; Christopoulos, P.; Papagianni, M.; Kleanthous, K.; Marakaki, C.; Papadimitriou, A.; Mastorakos, G. Secondary Prevention of Diabetes Type 1 with Oral Calcitriol and Analogs, the PRECAL Study. Children 2023, 10, 862. https://doi.org/10.3390/children10050862
Papadimitriou DT, Dermitzaki E, Christopoulos P, Papagianni M, Kleanthous K, Marakaki C, Papadimitriou A, Mastorakos G. Secondary Prevention of Diabetes Type 1 with Oral Calcitriol and Analogs, the PRECAL Study. Children. 2023; 10(5):862. https://doi.org/10.3390/children10050862
Chicago/Turabian StylePapadimitriou, Dimitrios T., Eleni Dermitzaki, Panagiotis Christopoulos, Maria Papagianni, Kleanthis Kleanthous, Chrysanthi Marakaki, Anastasios Papadimitriou, and George Mastorakos. 2023. "Secondary Prevention of Diabetes Type 1 with Oral Calcitriol and Analogs, the PRECAL Study" Children 10, no. 5: 862. https://doi.org/10.3390/children10050862
APA StylePapadimitriou, D. T., Dermitzaki, E., Christopoulos, P., Papagianni, M., Kleanthous, K., Marakaki, C., Papadimitriou, A., & Mastorakos, G. (2023). Secondary Prevention of Diabetes Type 1 with Oral Calcitriol and Analogs, the PRECAL Study. Children, 10(5), 862. https://doi.org/10.3390/children10050862










