Ultra-Processed Food, Reward System and Childhood Obesity
Abstract
1. Introduction
2. Methods
3. Childhood Obesity
4. Feeding Regulation and Brain Reward System
4.1. Feeding Regulation
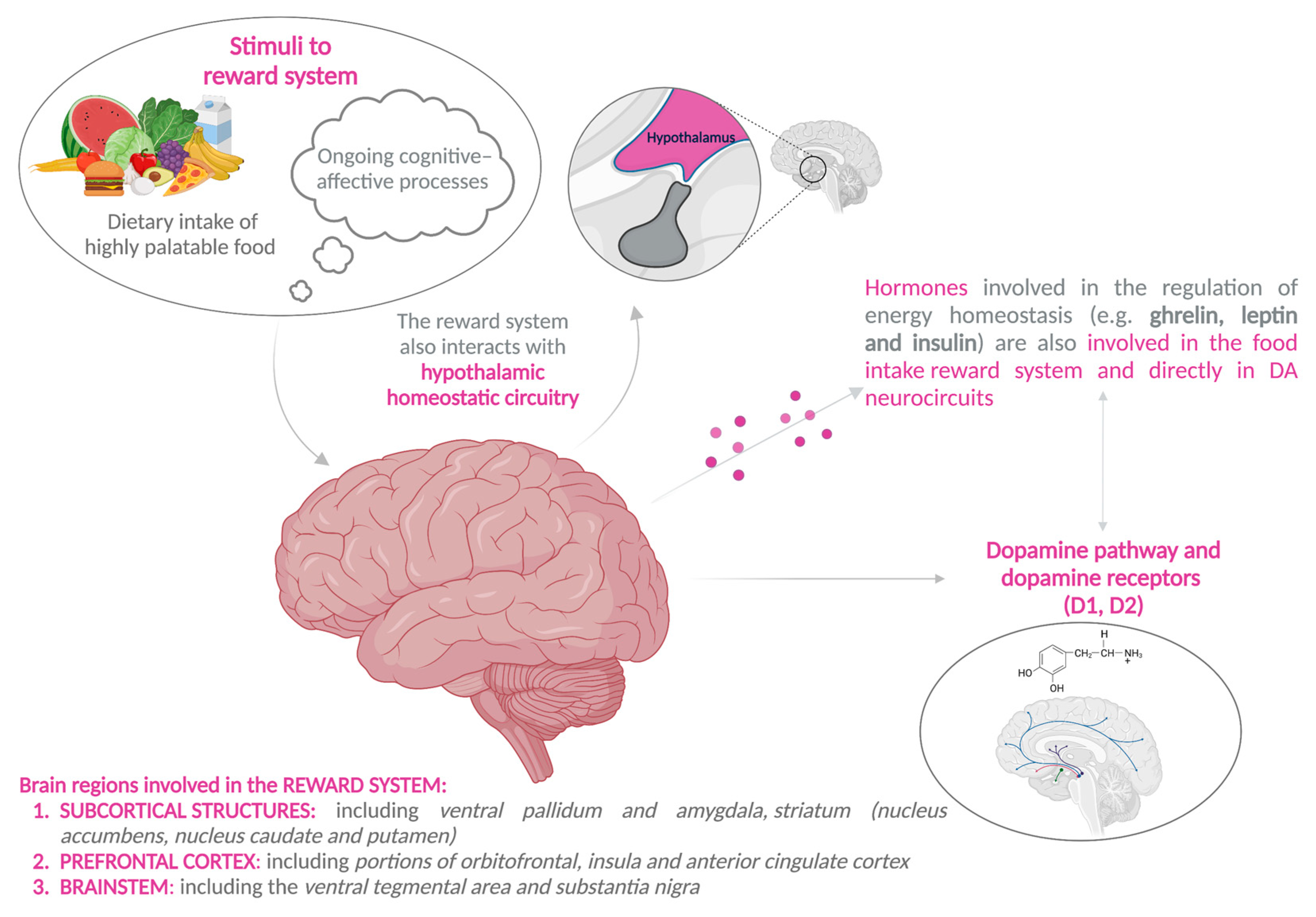
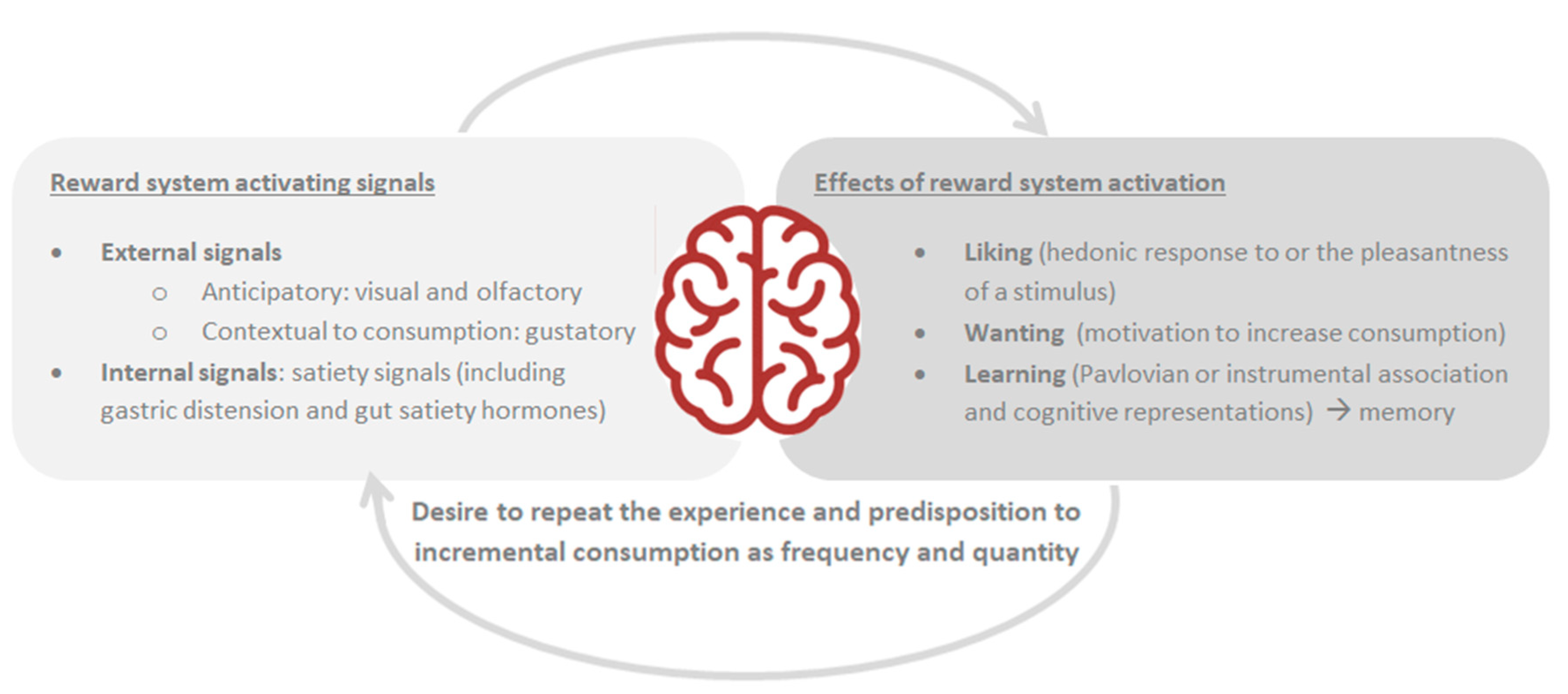
4.2. Brain Reward System
5. Ultra Processed Food in Childhood Obesity
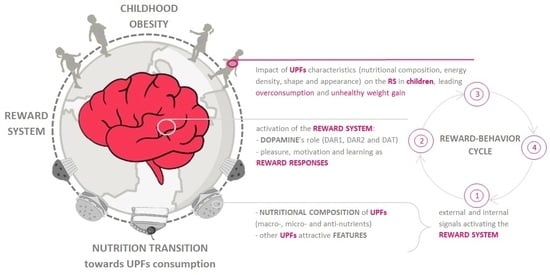
6. Ultra-Processed Foods and Reward System in Children
6.1. Nutritional Factors Characterizing UPFs with a Potential Impact on the Reward System and Predisposing toward Overconsumption
6.2. Effect of UPFs on the Reward System, Promoting Excessive Energy Consumption
6.3. “Addictive-like Behaviour” and Ultra-Processed Foods: The Debate in the Literature on Whether or Not It Is Possible to Talk about “Addiction to Ultra-Processed Foods”
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 28 December 2022).
- Sahoo, K.; Sahoo, B.; Choudhury, A.; Sofi, N.; Kumar, R.; Bhadoria, A. Childhood Obesity: Causes and Consequences. J. Fam. Med. Prim. Care 2015, 4, 187. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Levy, R.B.; Moubarac, J.-C.; Louzada, M.L.; Rauber, F.; Khandpur, N.; Cediel, G.; Neri, D.; Martinez-Steele, E.; et al. Ultra-Processed Foods: What They Are and How to Identify Them. Public Health Nutr. 2019, 22, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Huffman, S.L.; Piwoz, E.G.; Vosti, S.A.; Dewey, K.G. Babies, Soft Drinks and Snacks: A Concern in Low- and Middle-Income Countries? Matern. Child. Nutr. 2014, 10, 562–574. [Google Scholar] [CrossRef]
- Rousham, E.K.; Goudet, S.; Markey, O.; Griffiths, P.; Boxer, B.; Carroll, C.; Petherick, E.S.; Pradeilles, R. Unhealthy Food and Beverage Consumption in Children and Risk of Overweight and Obesity: A Systematic Review and Meta-Analysis. Adv. Nutr. 2022, 13, 1669–1696. [Google Scholar] [CrossRef] [PubMed]
- Pries, A.M.; Filteau, S.; Ferguson, E.L. Snack Food and Beverage Consumption and Young Child Nutrition in Low- and Middle-Income Countries: A Systematic Review. Matern. Child. Nutr. 2019, 15 (Suppl. 4), e12729. [Google Scholar] [CrossRef]
- GBD 2017 Diet Collaborators Health Effects of Dietary Risks in 195 Countries, 1990-2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [CrossRef]
- Costa, C.S.; Rauber, F.; Leffa, P.S.; Sangalli, C.N.; Campagnolo, P.D.B.; Vitolo, M.R. Ultra-Processed Food Consumption and Its Effects on Anthropometric and Glucose Profile: A Longitudinal Study during Childhood. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 177–184. [Google Scholar] [CrossRef]
- Blundell, J.; de Graaf, C.; Hulshof, T.; Jebb, S.; Livingstone, B.; Lluch, A.; Mela, D.; Salah, S.; Schuring, E.; van der Knaap, H.; et al. Appetite Control: Methodological Aspects of the Evaluation of Foods. Obes. Rev. 2010, 11, 251–270. [Google Scholar] [CrossRef]
- Amin, T.; Mercer, J.G. Hunger and Satiety Mechanisms and Their Potential Exploitation in the Regulation of Food Intake. Curr. Obes. Rep. 2016, 5, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Weltens, N.; Zhao, D.; Van Oudenhove, L. Where Is the Comfort in Comfort Foods? Mechanisms Linking Fat Signaling, Reward, and Emotion. Neurogastroenterol. Motil. 2014, 26, 303–315. [Google Scholar] [CrossRef]
- Berridge, K.C.; Kringelbach, M.L. Pleasure Systems in the Brain. Neuron 2015, 86, 646–664. [Google Scholar] [CrossRef] [PubMed]
- Morales, I.; Berridge, K.C. “Liking” and “Wanting” in Eating and Food Reward: Brain Mechanisms and Clinical Implications. Physiol. Behav. 2020, 227, 113152. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.T.; Denniss, A.R. An Introduction to Writing Narrative and Systematic Reviews-Tasks, Tips and Traps for Aspiring Authors. Heart Lung Circ. 2018, 27, 893–898. [Google Scholar] [CrossRef] [PubMed]
- Baethge, C.; Goldbeck-Wood, S.; Mertens, S. SANRA—A Scale for the Quality Assessment of Narrative Review Articles. Res. Integr. Peer Rev. 2019, 4, 5. [Google Scholar] [CrossRef]
- Bradwisch, S.A.; Smith, E.M.; Mooney, C.; Scaccia, D. Obesity in Children and Adolescents: An Overview. Nursing 2020, 50, 60–66. [Google Scholar] [CrossRef]
- Taghizadeh, S.; Farhangi, M.A. The Effectiveness of Pediatric Obesity Prevention Policies: A Comprehensive Systematic Review and Dose–Response Meta-Analysis of Controlled Clinical Trials. J. Transl. Med. 2020, 18, 480. [Google Scholar] [CrossRef] [PubMed]
- Kansra, A.R.; Lakkunarajah, S.; Jay, M.S. Childhood and Adolescent Obesity: A Review. Front. Pediatr. 2021, 8, 581461. [Google Scholar] [CrossRef] [PubMed]
- Epicentro-ISS Istituto Superiore di Sanità Sistema Di Sorveglianza Okkio Alla SALUTE 2019. Available online: https://www.epicentro.iss.it/okkioallasalute/indagine-2019-dati (accessed on 2 January 2023).
- Blüher, M. Obesity: Global Epidemiology and Pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- NCD Risk Factor Collaboration (NCD-RisC) Worldwide Trends in Body-Mass Index, Underweight, Overweight, and Obesity from 1975 to 2016: A Pooled Analysis of 2416 Population-Based Measurement Studies in 128·9 Million Children, Adolescents, and Adults. Lancet 2017, 390, 2627–2642. [CrossRef]
- Geserick, M.; Vogel, M.; Gausche, R.; Lipek, T.; Spielau, U.; Keller, E.; Pfäffle, R.; Kiess, W.; Körner, A. Acceleration of BMI in Early Childhood and Risk of Sustained Obesity. N. Engl. J. Med. 2018, 379, 1303–1312. [Google Scholar] [CrossRef] [PubMed]
- Rokholm, B.; Baker, J.L.; Sørensen, T.I.A. The Levelling off of the Obesity Epidemic since the Year 1999--a Review of Evidence and Perspectives. Obes. Rev. 2010, 11, 835–846. [Google Scholar] [CrossRef]
- Qasim, A.; Turcotte, M.; de Souza, R.J.; Samaan, M.C.; Champredon, D.; Dushoff, J.; Speakman, J.R.; Meyre, D. On the Origin of Obesity: Identifying the Biological, Environmental and Cultural Drivers of Genetic Risk among Human Populations: Evolution and Genetic Risk of Obesity. Obes. Rev. 2018, 19, 121–149. [Google Scholar] [CrossRef] [PubMed]
- Jebeile, H.; Kelly, A.S.; O’Malley, G.; Baur, L.A. Obesity in Children and Adolescents: Epidemiology, Causes, Assessment, and Management. Lancet Diabetes Endocrinol. 2022, 10, 351–365. [Google Scholar] [CrossRef] [PubMed]
- Roberto, C.A.; Swinburn, B.; Hawkes, C.; Huang, T.T.-K.; Costa, S.A.; Ashe, M.; Zwicker, L.; Cawley, J.H.; Brownell, K.D. Patchy Progress on Obesity Prevention: Emerging Examples, Entrenched Barriers, and New Thinking. Lancet 2015, 385, 2400–2409. [Google Scholar] [CrossRef]
- Brown, C.L.; Halvorson, E.E.; Cohen, G.M.; Lazorick, S.; Skelton, J.A. Addressing Childhood Obesity: Opportunities for Prevention. Pediatr. Clin. N. Am. 2015, 62, 1241–1261. [Google Scholar] [CrossRef] [PubMed]
- Mahumud, R.A.; Sahle, B.W.; Owusu-Addo, E.; Chen, W.; Morton, R.L.; Renzaho, A.M.N. Association of Dietary Intake, Physical Activity, and Sedentary Behaviours with Overweight and Obesity among 282,213 Adolescents in 89 Low and Middle Income to High-Income Countries. Int. J. Obes. 2021, 45, 2404–2418. [Google Scholar] [CrossRef] [PubMed]
- Fang, K.; Mu, M.; Liu, K.; He, Y. Screen Time and Childhood Overweight/Obesity: A Systematic Review and Meta-Analysis. Child. Care Health Dev. 2019, 45, 744–753. [Google Scholar] [CrossRef]
- Sadeghirad, B.; Duhaney, T.; Motaghipisheh, S.; Campbell, N.R.C.; Johnston, B.C. Influence of Unhealthy Food and Beverage Marketing on Children’s Dietary Intake and Preference: A Systematic Review and Meta-Analysis of Randomized Trials: Meta-Analysis of Unhealthy Food and Beverage Marketing. Obes. Rev. 2016, 17, 945–959. [Google Scholar] [CrossRef]
- Poorolajal, J.; Sahraei, F.; Mohamdadi, Y.; Doosti-Irani, A.; Moradi, L. Behavioral Factors Influencing Childhood Obesity: A Systematic Review and Meta-Analysis. Obes. Res. Clin. Pract. 2020, 14, 109–118. [Google Scholar] [CrossRef]
- Kavey, R.-E.W. How Sweet It Is: Sugar-Sweetened Beverage Consumption, Obesity, and Cardiovascular Risk in Childhood. J. Am. Diet. Assoc. 2010, 110, 1456–1460. [Google Scholar] [CrossRef]
- Dereń, K.; Weghuber, D.; Caroli, M.; Koletzko, B.; Thivel, D.; Frelut, M.-L.; Socha, P.; Grossman, Z.; Hadjipanayis, A.; Wyszyńska, J.; et al. Consumption of Sugar-Sweetened Beverages in Paediatric Age: A Position Paper of the European Academy of Paediatrics and the European Childhood Obesity Group. Ann. Nutr. Metab. 2019, 74, 296–302. [Google Scholar] [CrossRef]
- Abbasalizad Farhangi, M.; Mohammadi Tofigh, A.; Jahangiri, L.; Nikniaz, Z.; Nikniaz, L. Sugar-Sweetened Beverages Intake and the Risk of Obesity in Children: An Updated Systematic Review and Dose-Response Meta-Analysis. Pediatr. Obes. 2022, 17, e12914. [Google Scholar] [CrossRef] [PubMed]
- Calcaterra, V.; Cena, H.; Magenes, V.C.; Vincenti, A.; Comola, G.; Beretta, A.; Di Napoli, I.; Zuccotti, G. Sugar-Sweetened Beverages and Metabolic Risk in Children and Adolescents with Obesity: A Narrative Review. Nutrients 2023, 15, 702. [Google Scholar] [CrossRef] [PubMed]
- Ogden, C.L.; Carroll, M.D.; Fakhouri, T.H.; Hales, C.M.; Fryar, C.D.; Li, X.; Freedman, D.S. Prevalence of Obesity Among Youths by Household Income and Education Level of Head of Household—United States 2011–2014. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.S.; Barlow, S.E.; Rao, G.; Inge, T.H.; Hayman, L.L.; Steinberger, J.; Urbina, E.M.; Ewing, L.J.; Daniels, S.R. Severe Obesity in Children and Adolescents: Identification, Associated Health Risks, and Treatment Approaches: A Scientific Statement From the American Heart Association. Circulation 2013, 128, 1689–1712. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.M.; Butcher, K.F. (Kristin F. Childhood Obesity: Trends and Potential Causes. Future Child. 2006, 16, 19–45. [Google Scholar] [CrossRef]
- Cena, H.; Fiechtner, L.; Vincenti, A.; Magenes, V.C.; De Giuseppe, R.; Manuelli, M.; Zuccotti, G.V.; Calcaterra, V. COVID-19 Pandemic as Risk Factors for Excessive Weight Gain in Pediatrics: The Role of Changes in Nutrition Behavior. A Narrative Review. Nutrients 2021, 13, 4255. [Google Scholar] [CrossRef]
- Galler, A.; Röbl, M.; Prinz, N.; Dannemann, A.; Gellhaus, I.; Kapellen, T.; Linke, S.; Schauerte, G.; Stein, R.; Weghuber, D.; et al. Weight Development in Children and Adolescents with Obesity during the COVID-19 Pandemic. Dtsch. Ärzteblatt Int. 2022, 119, 302–303. [Google Scholar] [CrossRef]
- Marcovecchio, M.L.; Gorman, S.; Watson, L.P.E.; Dunger, D.B.; Beardsall, K. Catch-Up Growth in Children Born Small for Gestational Age Related to Body Composition and Metabolic Risk at Six Years of Age in the UK. Horm. Res. Paediatr. 2020, 93, 119–127. [Google Scholar] [CrossRef]
- Valerio, G.; Maffeis, C.; Saggese, G.; Ambruzzi, M.A.; Balsamo, A.; Bellone, S.; Bergamini, M.; Bernasconi, S.; Bona, G.; Calcaterra, V.; et al. Diagnosis, Treatment and Prevention of Pediatric Obesity: Consensus Position Statement of the Italian Society for Pediatric Endocrinology and Diabetology and the Italian Society of Pediatrics. Ital. J. Pediatr. 2018, 44, 88. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.; Gasbarrini, A.; Mele, M. What Is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef]
- Indrio, F.; Martini, S.; Francavilla, R.; Corvaglia, L.; Cristofori, F.; Mastrolia, S.A.; Neu, J.; Rautava, S.; Russo Spena, G.; Raimondi, F.; et al. Epigenetic Matters: The Link between Early Nutrition, Microbiome, and Long-Term Health Development. Front. Pediatr. 2017, 5, 178. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, M.; Llewellyn, A.; Owen, C.G.; Woolacott, N. Simple Tests for the Diagnosis of Childhood Obesity: A Systematic Review and Meta-Analysis. Obes. Rev. 2016, 17, 1301–1315. [Google Scholar] [CrossRef]
- Calcaterra, V.; Rossi, V.; Mari, A.; Casini, F.; Bergamaschi, F.; Zuccotti, G.V.; Fabiano, V. Medical Treatment of Weight Loss in Children and Adolescents with Obesity. Pharmacol. Res. 2022, 185, 106471. [Google Scholar] [CrossRef] [PubMed]
- Jebeile, H.; Cardel, M.I.; Kyle, T.K.; Jastreboff, A.M. Addressing Psychosocial Health in the Treatment and Care of Adolescents with Obesity. Obesity 2021, 29, 1413–1422. [Google Scholar] [CrossRef]
- Cole, T.J.; Lobstein, T. Extended International (IOTF) Body Mass Index Cut-Offs for Thinness, Overweight and Obesity: Extended International BMI Cut-Offs. Pediatr. Obes. 2012, 7, 284–294. [Google Scholar] [CrossRef]
- WHO Child Growth Standards. Available online: https://www.who.int/tools/child-growth-standards (accessed on 2 January 2023).
- Horesh, A.; Tsur, A.M.; Bardugo, A.; Twig, G. Adolescent and Childhood Obesity and Excess Morbidity and Mortality in Young Adulthood-a Systematic Review. Curr. Obes. Rep. 2021, 10, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Pulgarón, E.R. Childhood Obesity: A Review of Increased Risk for Physical and Psychological Comorbidities. Clin. Ther. 2013, 35, A18–A32. [Google Scholar] [CrossRef]
- Ferrante, A.W. Obesity-Induced Inflammation: A Metabolic Dialogue in the Language of Inflammation. J. Intern. Med. 2007, 262, 408–414. [Google Scholar] [CrossRef]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O.; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of Inflammation and Cardiovascular Disease: Application to Clinical and Public Health Practice: A Statement for Healthcare Professionals From the Centers for Disease Control and Prevention and the American Heart Association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef]
- Calcaterra, V.; Klersy, C.; Muratori, T.; Telli, S.; Caramagna, C.; Scaglia, F.; Cisternino, M.; Larizza, D. Prevalence of Metabolic Syndrome (MS) in Children and Adolescents with Varying Degrees of Obesity. Clin. Endocrinol. 2008, 68, 868–872. [Google Scholar] [CrossRef] [PubMed]
- De Leonibus, C.; Marcovecchio, M.L.; Chiarelli, F. Update on Statural Growth and Pubertal Development in Obese Children. Pediatr. Rep. 2012, 4, e35. [Google Scholar] [CrossRef] [PubMed]
- Witchel, S.F.; Burghard, A.C.; Tao, R.H.; Oberfield, S.E. The Diagnosis and Treatment of PCOS in Adolescents: An Update. Curr. Opin. Pediatr. 2019, 31, 562–569. [Google Scholar] [CrossRef]
- Marcus, C.L.; Brooks, L.J.; Ward, S.D.; Draper, K.A.; Gozal, D.; Halbower, A.C.; Jones, J.; Lehmann, C.; Schechter, M.S.; Sheldon, S.; et al. Diagnosis and Management of Childhood Obstructive Sleep Apnea Syndrome. Pediatrics 2012, 130, e714–e755. [Google Scholar] [CrossRef] [PubMed]
- Rankin, J.; Matthews, L.; Cobley, S.; Han, A.; Sanders, R.; Wiltshire, H.D.; Baker, J.S. Psychological Consequences of Childhood Obesity: Psychiatric Comorbidity and Prevention. Adolesc. Health Med. Ther. 2016, 7, 125–146. [Google Scholar] [CrossRef]
- Topçu, S.; Orhon, F.Ş.; Tayfun, M.; Uçaktürk, S.A.; Demirel, F. Anxiety, Depression and Self-Esteem Levels in Obese Children: A Case-Control Study. J. Pediatr. Endocrinol. Metab. 2016, 29, 254. [Google Scholar] [CrossRef]
- Katzmarzyk, P.T.; Barlow, S.; Bouchard, C.; Catalano, P.M.; Hsia, D.S.; Inge, T.H.; Lovelady, C.; Raynor, H.; Redman, L.M.; Staiano, A.E.; et al. An Evolving Scientific Basis for the Prevention and Treatment of Pediatric Obesity. Int. J. Obes. 2014, 38, 887–905. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.; Moore, T.H.; Hooper, L.; Gao, Y.; Zayegh, A.; Ijaz, S.; Elwenspoek, M.; Foxen, S.C.; Magee, L.; O’Malley, C.; et al. Interventions for Preventing Obesity in Children. Cochrane Database Syst. Rev. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Scarborough, P.; Rayner, M.; Briggs, A.D.M. Should We Tax Unhealthy Food and Drink? Proc. Nutr. Soc. 2018, 77, 314–320. [Google Scholar] [CrossRef]
- Figlewicz, D.P.; Sipols, A.J. Energy Regulatory Signals and Food Reward. Pharmacol. Biochem. Behav. 2010, 97, 15–24. [Google Scholar] [CrossRef]
- Lutter, M.; Nestler, E.J. Homeostatic and Hedonic Signals Interact in the Regulation of Food Intake. J. Nutr. 2009, 139, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.A.; Stuber, G.D. Overlapping Brain Circuits for Homeostatic and Hedonic Feeding. Cell. Metabolism 2018, 27, 42–56. [Google Scholar] [CrossRef]
- Kenny, P.J. Common Cellular and Molecular Mechanisms in Obesity and Drug Addiction. Nat. Rev. Neurosci. 2011, 12, 638–651. [Google Scholar] [CrossRef]
- Berridge, K.C. Food Reward: Brain Substrates of Wanting and Liking. Neurosci. Biobehav. Rev. 1996, 20, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Hoebel, B.G.; Teitelbaum, P. Hypothalamic Control of Feeding and Self-Stimulation. Science 1962, 135, 375–377. [Google Scholar] [CrossRef]
- Margules, D.L.; Olds, J. Identical “Feeding” and “Rewarding” Systems in the Lateral Hypothalamus of Rats. Science 1962, 135, 374–375. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A. Dopamine, Learning and Motivation. Nat. Rev. Neurosci. 2004, 5, 483–494. [Google Scholar] [CrossRef]
- Corbett, D.; Wise, R.A. Intracranial Self-Stimulation in Relation to the Ascending Dopaminergic Systems of the Midbrain: A Moveable Electrode Mapping Study. Brain Res. 1980, 185, 1–15. [Google Scholar] [CrossRef]
- Schultz, W. Getting Formal with Dopamine and Reward. Neuron 2002, 36, 241–263. [Google Scholar] [CrossRef]
- Morris, G.; Nevet, A.; Arkadir, D.; Vaadia, E.; Bergman, H. Midbrain Dopamine Neurons Encode Decisions for Future Action. Nat. Neurosci. 2006, 9, 1057–1063. [Google Scholar] [CrossRef]
- Castro, D.C.; Cole, S.L.; Berridge, K.C. Lateral Hypothalamus, Nucleus Accumbens, and Ventral Pallidum Roles in Eating and Hunger: Interactions between Homeostatic and Reward Circuitry. Front. Syst. Neurosci. 2015, 9, 90. [Google Scholar] [CrossRef] [PubMed]
- Saper, C.B.; Chou, T.C.; Elmquist, J.K. The Need to Feed. Neuron 2002, 36, 199–211. [Google Scholar] [CrossRef]
- Blundell, J.E.; Lawton, C.L.; Hill, A.J. Mechanisms of Appetite Control and Their Abnormalities in Obese Patients. Horm. Res. 1993, 39, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Halford, J.C.G.; Harrold, J.A. Satiety-Enhancing Products for Appetite Control: Science and Regulation of Functional Foods for Weight Management. Proc. Nutr. Soc. 2012, 71, 350–362. [Google Scholar] [CrossRef]
- Davis, J. Hunger, Ghrelin and the Gut. Brain Res. 2018, 1693, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Van Kleef, E.; Van Trijp, J.C.M.; Van Den Borne, J.J.G.C.; Zondervan, C. Successful Development of Satiety Enhancing Food Products: Towards a Multidisciplinary Agenda of Research Challenges. Crit. Rev. Food Sci. Nutr. 2012, 52, 611–628. [Google Scholar] [CrossRef] [PubMed]
- Demori, I.; Grasselli, E. Stress-Related Weight Gain: Mechanisms Involving Feeding Behavior, Metabolism, Gut Microbiota and Inflammation. J. Nutr. Food Sci. 2016, 6, 457. [Google Scholar] [CrossRef]
- Mietus-Snyder, M.L.; Lustig, R.H. Childhood Obesity: Adrift in the “Limbic Triangle”. Annu. Rev. Med. 2008, 59, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Oliver, G.; Wardle, J. Perceived Effects of Stress on Food Choice. Physiol. Behav. 1999, 66, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.G.; Cohen, P.; Kasen, S.; Brook, J.S. Childhood Adversities Associated with Risk for Eating Disorders or Weight Problems during Adolescence or Early Adulthood. Am. J. Psychiatry 2002, 159, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Roemmich, J.N.; Wright, S.M.; Epstein, L.H. Dietary Restraint and Stress-Induced Snacking in Youth. Obes. Res. 2002, 10, 1120–1126. [Google Scholar] [CrossRef]
- Espel-Huynh, H.M.; Muratore, A.F.; Lowe, M.R. A Narrative Review of the Construct of Hedonic Hunger and Its Measurement by the Power of Food Scale: Hedonic Hunger Review. Obes. Sci. Pract. 2018, 4, 238–249. [Google Scholar] [CrossRef]
- Alonso-Alonso, M.; Woods, S.C.; Pelchat, M.; Grigson, P.S.; Stice, E.; Farooqi, S.; Khoo, C.S.; Mattes, R.D.; Beauchamp, G.K. Food Reward System: Current Perspectives and Future Research Needs. Nutr. Rev. 2015, 73, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Leng, G.; Adan, R.A.H.; Belot, M.; Brunstrom, J.M.; de Graaf, K.; Dickson, S.L.; Hare, T.; Maier, S.; Menzies, J.; Preissl, H.; et al. The Determinants of Food Choice. Proc. Nutr. Soc. 2017, 76, 316–327. [Google Scholar] [CrossRef]
- Palmiter, R.D. Is Dopamine a Physiologically Relevant Mediator of Feeding Behavior? Trends Neurosci. 2007, 30, 375–381. [Google Scholar] [CrossRef]
- Beaulieu, J.-M.; Gainetdinov, R.R. The Physiology, Signaling, and Pharmacology of Dopamine Receptors. Pharmacol. Rev. 2011, 63, 182–217. [Google Scholar] [CrossRef] [PubMed]
- Tritsch, N.X.; Sabatini, B.L. Dopaminergic Modulation of Synaptic Transmission in Cortex and Striatum. Neuron 2012, 76, 33–50. [Google Scholar] [CrossRef]
- Pothos, E.N.; Davila, V.; Sulzer, D. Presynaptic Recording of Quanta from Midbrain Dopamine Neurons and Modulation of the Quantal Size. J. Neurosci. 1998, 18, 4106–4118. [Google Scholar] [CrossRef]
- Cass, W.A.; Zahniser, N.R. Potassium Channel Blockers Inhibit D2 Dopamine, but Not A1 Adenosine, Receptor-Mediated Inhibition of Striatal Dopamine Release. J. Neurochem. 1991, 57, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Congar, P.; Bergevin, A.; Trudeau, L.-E. D2 Receptors Inhibit the Secretory Process Downstream From Calcium Influx in Dopaminergic Neurons: Implication of K + Channels. J. Neurophysiol. 2002, 87, 1046–1056. [Google Scholar] [CrossRef]
- Johnson, P.M.; Kenny, P.J. Dopamine D2 Receptors in Addiction-like Reward Dysfunction and Compulsive Eating in Obese Rats. Nat. Neurosci. 2010, 13, 635–641. [Google Scholar] [CrossRef]
- Wang, C.; Buck, D.C.; Yang, R.; Macey, T.A.; Neve, K.A. Dopamine D2 Receptor Stimulation of Mitogen-Activated Protein Kinases Mediated by Cell Type-Dependent Transactivation of Receptor Tyrosine Kinases. J. Neurochem. 2005, 93, 899–909. [Google Scholar] [CrossRef]
- Stice, E.; Yokum, S.; Zald, D.; Dagher, A. Dopamine-Based Reward Circuitry Responsivity, Genetics, and Overeating. In Behavioral Neurobiology of Eating Disorders; Adan, R.A.H., Kaye, W.H., Eds.; Current Topics in Behavioral Neurosciences; Springer: Berlin/Heidelberg, Germany, 2010; Volume 6, pp. 81–93. ISBN 978-3-642-15130-9. [Google Scholar]
- Baik, J.-H. Dopamine Signaling in Reward-Related Behaviors. Front. Neural Circuits 2013, 7, 152. [Google Scholar] [CrossRef] [PubMed]
- Kozuka, C.; Kaname, T.; Shimizu-Okabe, C.; Takayama, C.; Tsutsui, M.; Matsushita, M.; Abe, K.; Masuzaki, H. Impact of Brown Rice-Specific γ-Oryzanol on Epigenetic Modulation of Dopamine D2 Receptors in Brain Striatum in High-Fat-Diet-Induced Obesity in Mice. Diabetologia 2017, 60, 1502–1511. [Google Scholar] [CrossRef]
- Hommel, J.D.; Trinko, R.; Sears, R.M.; Georgescu, D.; Liu, Z.-W.; Gao, X.-B.; Thurmon, J.J.; Marinelli, M.; DiLeone, R.J. Leptin Receptor Signaling in Midbrain Dopamine Neurons Regulates Feeding. Neuron 2006, 51, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, I.S.; Bullmore, E.; Keogh, J.; Gillard, J.; O’Rahilly, S.; Fletcher, P.C. Leptin Regulates Striatal Regions and Human Eating Behavior. Science 2007, 317, 1355. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Baler, R.D. Reward, Dopamine and the Control of Food Intake: Implications for Obesity. Trends Cogn. Sci. 2011, 15, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Abizaid, A.; Liu, Z.-W.; Andrews, Z.B.; Shanabrough, M.; Borok, E.; Elsworth, J.D.; Roth, R.H.; Sleeman, M.W.; Picciotto, M.R.; Tschöp, M.H.; et al. Ghrelin Modulates the Activity and Synaptic Input Organization of Midbrain Dopamine Neurons While Promoting Appetite. J. Clin. Investig. 2006, 116, 3229–3239. [Google Scholar] [CrossRef]
- Khanh, D.V.; Choi, Y.-H.; Moh, S.H.; Kinyua, A.W.; Kim, K.W. Leptin and Insulin Signaling in Dopaminergic Neurons: Relationship between Energy Balance and Reward System. Front. Psychol. 2014, 5, 846. [Google Scholar] [CrossRef] [PubMed]
- Bruijnzeel, A.W.; Corrie, L.W.; Rogers, J.A.; Yamada, H. Effects of Insulin and Leptin in the Ventral Tegmental Area and Arcuate Hypothalamic Nucleus on Food Intake and Brain Reward Function in Female Rats. Behav. Brain Res. 2011, 219, 254–264. [Google Scholar] [CrossRef]
- Mebel, D.M.; Wong, J.C.; Dong, Y.J.; Borgland, S.L. Insulin in the Ventral Tegmental Area Reduces Hedonic Feeding and Suppresses Dopamine Concentration via Increased Reuptake: Insulin Attenuates Somatodendritic Dopamine. Eur. J. Neurosci. 2012, 36, 2336–2346. [Google Scholar] [CrossRef] [PubMed]
- Könner, A.C.; Hess, S.; Tovar, S.; Mesaros, A.; Sánchez-Lasheras, C.; Evers, N.; Verhagen, L.A.W.; Brönneke, H.S.; Kleinridders, A.; Hampel, B.; et al. Role for Insulin Signaling in Catecholaminergic Neurons in Control of Energy Homeostasis. Cell. Metab. 2011, 13, 720–728. [Google Scholar] [CrossRef]
- Meyer, M.R.; Haas, E.; Barton, M. Gender Differences of Cardiovascular Disease: New Perspectives for Estrogen Receptor Signaling. Hypertension 2006, 47, 1019–1026. [Google Scholar] [CrossRef]
- Sienkiewicz-Jarosz, H.; Scinska, A.; Swiecicki, L.; Lipczynska-Lojkowska, W.; Kuran, W.; Ryglewicz, D.; Kolaczkowski, M.; Samochowiec, J.; Bienkowski, P. Sweet Liking in Patients with Parkinson’s Disease. J. Neurol. Sci. 2013, 329, 17–22. [Google Scholar] [CrossRef]
- Ireland, J.D.; Møller, A. LanguaL Food Description: A Learning Process. Eur. J. Clin. Nutr. 2010, 64 (Suppl. 3), S44–S48. [Google Scholar] [CrossRef]
- Becker, W.; Møller, A.; Ireland, J.; Roe, M.; Unwin, I.; Pakkala, H. Proposal for Structure and Detail of a EuroFIR Standard on Food Composition Data. II: Technical Annex. In Danish Food Information; European Food Information Resource Network: Brussels, Belgium, 2008. [Google Scholar]
- Lauria, F.; Dello Russo, M.; Formisano, A.; De Henauw, S.; Hebestreit, A.; Hunsberger, M.; Krogh, V.; Intemann, T.; Lissner, L.; Molnar, D.; et al. Ultra-Processed Foods Consumption and Diet Quality of European Children, Adolescents and Adults: Results from the I.Family Study. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 3031–3043. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A.; Levy, R.B.; Claro, R.M.; de Castro, I.R.R.; Cannon, G. Increasing Consumption of Ultra-Processed Foods and Likely Impact on Human Health: Evidence from Brazil. Public Health Nutr. 2011, 14, 5–13. [Google Scholar] [CrossRef]
- Parra, D.C.; da Costa-Louzada, M.L.; Moubarac, J.-C.; Bertazzi-Levy, R.; Khandpur, N.; Cediel, G.; Monteiro, C.A. Association between Ultra-Processed Food Consumption and the Nutrient Profile of the Colombian Diet in 2005. Salud Publica Mex. 2019, 61, 147–154. [Google Scholar] [CrossRef]
- Petrus, R.R.; do Amaral Sobral, P.J.; Tadini, C.C.; Gonçalves, C.B. The NOVA Classification System: A Critical Perspective in Food Science. Trends Food Sci. Technol. 2021, 116, 603–608. [Google Scholar] [CrossRef]
- Marino, M.; Puppo, F.; Del Bo’, C.; Vinelli, V.; Riso, P.; Porrini, M.; Martini, D. A Systematic Review of Worldwide Consumption of Ultra-Processed Foods: Findings and Criticisms. Nutrients 2021, 13, 2778. [Google Scholar] [CrossRef] [PubMed]
- Slimani, N.; Deharveng, G.; Southgate, D.A.T.; Biessy, C.; Chajès, V.; van Bakel, M.M.E.; Boutron-Ruault, M.C.; McTaggart, A.; Grioni, S.; Verkaik-Kloosterman, J.; et al. Contribution of Highly Industrially Processed Foods to the Nutrient Intakes and Patterns of Middle-Aged Populations in the European Prospective Investigation into Cancer and Nutrition Study. Eur. J. Clin. Nutr. 2009, 63 (Suppl. 4), S206–S225. [Google Scholar] [CrossRef] [PubMed]
- Davidou, S.; Christodoulou, A.; Fardet, A.; Frank, K. The Holistico-Reductionist Siga Classification According to the Degree of Food Processing: An Evaluation of Ultra-Processed Foods in French Supermarkets. Food Funct. 2020, 11, 2026–2039. [Google Scholar] [CrossRef]
- Moodie, R.; Stuckler, D.; Monteiro, C.; Sheron, N.; Neal, B.; Thamarangsi, T.; Lincoln, P.; Casswell, S. Lancet NCD Action Group Profits and Pandemics: Prevention of Harmful Effects of Tobacco, Alcohol, and Ultra-Processed Food and Drink Industries. Lancet 2013, 381, 670–679. [Google Scholar] [CrossRef]
- Ronto, R.; Wu, J.H.; Singh, G.M. The Global Nutrition Transition: Trends, Disease Burdens and Policy Interventions. Public Health Nutr. 2018, 21, 2267–2270. [Google Scholar] [CrossRef]
- Popkin, B.M. Relationship between Shifts in Food System Dynamics and Acceleration of the Global Nutrition Transition. Nutr. Rev. 2017, 75, 73–82. [Google Scholar] [CrossRef]
- Kelly, A.L.; Baugh, M.E.; Oster, M.E.; DiFeliceantonio, A.G. The Impact of Caloric Availability on Eating Behavior and Ultra-Processed Food Reward. Appetite 2022, 178, 106274. [Google Scholar] [CrossRef]
- Baraldi, L.G.; Martinez Steele, E.; Canella, D.S.; Monteiro, C.A. Consumption of Ultra-Processed Foods and Associated Sociodemographic Factors in the USA between 2007 and 2012: Evidence from a Nationally Representative Cross-Sectional Study. BMJ Open. 2018, 8, e020574. [Google Scholar] [CrossRef] [PubMed]
- Moubarac, J.-C.; Batal, M.; Martins, A.P.B.; Claro, R.; Levy, R.B.; Cannon, G.; Monteiro, C. Processed and Ultra-Processed Food Products: Consumption Trends in Canada from 1938 to 2011. Can. J. Diet. Pract. Res. 2014, 75, 15–21. [Google Scholar] [CrossRef]
- Neri, D.; Steele, E.M.; Khandpur, N.; Cediel, G.; Zapata, M.E.; Rauber, F.; Marrón-Ponce, J.A.; Machado, P.; Costa Louzada, M.L.; Andrade, G.C.; et al. Ultraprocessed Food Consumption and Dietary Nutrient Profiles Associated with Obesity: A Multicountry Study of Children and Adolescents. Obes. Rev. 2022, 23, 3387. [Google Scholar] [CrossRef] [PubMed]
- Martines, R.M.; Machado, P.P.; Neri, D.A.; Levy, R.B.; Rauber, F. Association between Watching TV Whilst Eating and Children’s Consumption of Ultraprocessed Foods in United Kingdom. Matern. Child. Nutr. 2019, 15, e12819. [Google Scholar] [CrossRef] [PubMed]
- Marrón-Ponce, J.A.; Sánchez-Pimienta, T.G.; da Costa Louzada, M.L.; Batis, C. Energy Contribution of NOVA Food Groups and Sociodemographic Determinants of Ultra-Processed Food Consumption in the Mexican Population. Public Health Nutr. 2018, 21, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Cediel, G.; Reyes, M.; da Costa Louzada, M.L.; Martinez Steele, E.; Monteiro, C.A.; Corvalán, C.; Uauy, R. Ultra-Processed Foods and Added Sugars in the Chilean Diet (2010). Public Health Nutr. 2018, 21, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, E.; Esposito, S.; Costanzo, S.; Di Castelnuovo, A.; Cerletti, C.; Donati, M.B.; de Gaetano, G.; Iacoviello, L.; Bonaccio, M. Ultra-Processed Food Consumption and Its Correlates among Italian Children, Adolescents and Adults from the Italian Nutrition & Health Survey (INHES) Cohort Study. Public Health Nutr. 2021, 24, 6258–6271. [Google Scholar] [CrossRef] [PubMed]
- Camara, S.; de Lauzon-Guillain, B.; Heude, B.; Charles, M.-A.; Botton, J.; Plancoulaine, S.; Forhan, A.; Saurel-Cubizolles, M.-J.; Dargent-Molina, P.; Lioret, S.; et al. Multidimensionality of the Relationship between Social Status and Dietary Patterns in Early Childhood: Longitudinal Results from the French EDEN Mother-Child Cohort. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 122. [Google Scholar] [CrossRef]
- Khandpur, N.; Neri, D.A.; Monteiro, C.; Mazur, A.; Frelut, M.-L.; Boyland, E.; Weghuber, D.; Thivel, D. Ultra-Processed Food Consumption among the Paediatric Population: An Overview and Call to Action from the European Childhood Obesity Group. Ann. Nutr. Metab. 2020, 76, 109–113. [Google Scholar] [CrossRef]
- Martini, D.; Godos, J.; Bonaccio, M.; Vitaglione, P.; Grosso, G. Ultra-Processed Foods and Nutritional Dietary Profile: A Meta-Analysis of Nationally Representative Samples. Nutrients 2021, 13, 3390. [Google Scholar] [CrossRef]
- Schiestl, E.T.; Rios, J.M.; Parnarouskis, L.; Cummings, J.R.; Gearhardt, A.N. A Narrative Review of Highly Processed Food Addiction across the Lifespan. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 106, 110152. [Google Scholar] [CrossRef]
- De Amicis, R.; Mambrini, S.P.; Pellizzari, M.; Foppiani, A.; Bertoli, S.; Battezzati, A.; Leone, A. Ultra-Processed Foods and Obesity and Adiposity Parameters among Children and Adolescents: A Systematic Review. Eur. J. Nutr. 2022, 61, 2297–2311. [Google Scholar] [CrossRef]
- Birch, L.L.; Doub, A.E. Learning to Eat: Birth to Age 2 y. Am. J. Clin. Nutr. 2014, 99, 723S–728S. [Google Scholar] [CrossRef]
- Luque, V.; Escribano, J.; Closa-Monasterolo, R.; Zaragoza-Jordana, M.; Ferré, N.; Grote, V.; Koletzko, B.; Totzauer, M.; Verduci, E.; ReDionigi, A.; et al. Unhealthy Dietary Patterns Established in Infancy Track to Mid-Childhood: The EU Childhood Obesity Project. J. Nutr. 2018, 148, 752–759. [Google Scholar] [CrossRef]
- Gordon, E.L.; Lent, M.R.; Merlo, L.J. The Effect of Food Composition and Behavior on Neurobiological Response to Food: A Review of Recent Research. Curr. Nutr. Rep. 2020, 9, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, C.A.; Cannon, G.; Lawrence, M.; da Costa Louzada, M.L.; Machado, P. Ultra-Processed Foods, Diet Quality, and Health Using the NOVA Classification System 2019. Available online: https://www.fao.org/3/ca5644en/ca5644en.pdf (accessed on 18 December 2022).
- Rauber, F.; Campagnolo, P.D.B.; Hoffman, D.J.; Vitolo, M.R. Consumption of Ultra-Processed Food Products and Its Effects on Children’s Lipid Profiles: A Longitudinal Study. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 116–122. [Google Scholar] [CrossRef] [PubMed]
- da Costa Louzada, M.L.; Baraldi, L.G.; Steele, E.M.; Martins, A.P.B.; Canella, D.S.; Moubarac, J.-C.; Levy, R.B.; Cannon, G.; Afshin, A.; Imamura, F.; et al. Consumption of Ultra-Processed Foods and Obesity in Brazilian Adolescents and Adults. Prev. Med. 2015, 81, 9–15. [Google Scholar] [CrossRef]
- Mendonça, R.d.D.; Pimenta, A.M.; Gea, A.; de la Fuente-Arrillaga, C.; Martinez-Gonzalez, M.A.; Lopes, A.C.S.; Bes-Rastrollo, M. Ultraprocessed Food Consumption and Risk of Overweight and Obesity: The University of Navarra Follow-Up (SUN) Cohort Study. Am. J. Clin. Nutr. 2016, 104, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Escamilla, R.; Obbagy, J.E.; Altman, J.M.; Essery, E.V.; McGrane, M.M.; Wong, Y.P.; Spahn, J.M.; Williams, C.L. Dietary Energy Density and Body Weight in Adults and Children: A Systematic Review. J. Acad. Nutr. Diet. 2012, 112, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Rouhani, M.H.; Haghighatdoost, F.; Surkan, P.J.; Azadbakht, L. Associations between Dietary Energy Density and Obesity: A Systematic Review and Meta-Analysis of Observational Studies. Nutrition 2016, 32, 1037–1047. [Google Scholar] [CrossRef] [PubMed]
- Poti, J.M.; Braga, B.; Qin, B. Ultra-Processed Food Intake and Obesity: What Really Matters for Health—Processing or Nutrient Content? Curr. Obes. Rep. 2017, 6, 420–431. [Google Scholar] [CrossRef]
- Luiten, C.M.; Steenhuis, I.H.; Eyles, H.; Ni Mhurchu, C.; Waterlander, W.E. Ultra-Processed Foods Have the Worst Nutrient Profile, yet They Are the Most Available Packaged Products in a Sample of New Zealand Supermarkets. Public Health Nutr. 2016, 19, 530–538. [Google Scholar] [CrossRef]
- Lane, M.M.; Davis, J.A.; Beattie, S.; Gómez-Donoso, C.; Loughman, A.; O’Neil, A.; Jacka, F.; Berk, M.; Page, R.; Marx, W.; et al. Ultraprocessed Food and Chronic Noncommunicable Diseases: A Systematic Review and Meta-analysis of 43 Observational Studies. Obes. Rev. 2021, 22, 3146. [Google Scholar] [CrossRef] [PubMed]
- Martínez Steele, E.; Baraldi, L.G.; da Costa Louzada, M.L.; Moubarac, J.-C.; Mozaffarian, D.; Monteiro, C.A. Ultra-Processed Foods and Added Sugars in the US Diet: Evidence from a Nationally Representative Cross-Sectional Study. BMJ Open. 2016, 6, e009892. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Fahimi, S.; Singh, G.M.; Micha, R.; Khatibzadeh, S.; Engell, R.E.; Lim, S.; Danaei, G.; Ezzati, M.; Powles, J. Global Sodium Consumption and Death from Cardiovascular Causes. N. Engl. J. Med. 2014, 371, 624–634. [Google Scholar] [CrossRef]
- Veronese, N.; Solmi, M.; Caruso, M.G.; Giannelli, G.; Osella, A.R.; Evangelou, E.; Maggi, S.; Fontana, L.; Stubbs, B.; Tzoulaki, I. Dietary Fiber and Health Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Am. J. Clin. Nutr. 2018, 107, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, L.-G.; Wu, Q.-J.; Ma, X.; Xiang, Y.-B. Association Between Dietary Fiber and Lower Risk of All-Cause Mortality: A Meta-Analysis of Cohort Studies. Am. J. Epidemiol. 2015, 181, 83–91. [Google Scholar] [CrossRef]
- Baker, P.; Machado, P.; Santos, T.; Sievert, K.; Backholer, K.; Hadjikakou, M.; Russell, C.; Huse, O.; Bell, C.; Scrinis, G.; et al. Ultra-Processed Foods and the Nutrition Transition: Global, Regional and National Trends, Food Systems Transformations and Political Economy Drivers. Obes. Rev. 2020, 21, e13126. [Google Scholar] [CrossRef]
- Boyland, E.J.; Nolan, S.; Kelly, B.; Tudur-Smith, C.; Jones, A.; Halford, J.C.; Robinson, E. Advertising as a Cue to Consume: A Systematic Review and Meta-Analysis of the Effects of Acute Exposure to Unhealthy Food and Nonalcoholic Beverage Advertising on Intake in Children and Adults. Am. J. Clin. Nutr. 2016, 103, 519–533. [Google Scholar] [CrossRef]
- Robinson, E.; Aveyard, P.; Daley, A.; Jolly, K.; Lewis, A.; Lycett, D.; Higgs, S. Eating Attentively: A Systematic Review and Meta-Analysis of the Effect of Food Intake Memory and Awareness on Eating. Am. J. Clin. Nutr. 2013, 97, 728–742. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Moubarac, J.-C.; Levy, R.B.; Louzada, M.L.C.; Jaime, P.C. The UN Decade of Nutrition, the NOVA Food Classification and the Trouble with Ultra-Processing. Public Health Nutr. 2018, 21, 5–17. [Google Scholar] [CrossRef]
- English, L.K.; Fearnbach, S.N.; Wilson, S.J.; Fisher, J.O.; Savage, J.S.; Rolls, B.J.; Keller, K.L. Food Portion Size and Energy Density Evoke Different Patterns of Brain Activation in Children. Am. J. Clin. Nutr. 2017, 105, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Gearhardt, A.N.; Hebebrand, J. The Concept of “Food Addiction” Helps Inform the Understanding of Overeating and Obesity: YES. Am. J. Clin. Nutr. 2021, 113, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Gramza-Michałowska, A. The Effects of Ultra-Processed Food Consumption-Is There Any Action Needed? Nutrients 2020, 12, 2556. [Google Scholar] [CrossRef]
- Zhang, Y.; Venkitasamy, C.; Pan, Z.; Liu, W.; Zhao, L. Novel Umami Ingredients: Umami Peptides and Their Taste. J. Food Sci. 2017, 82, 16–23. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, L.; Venkitasamy, C.; Pan, Z.; Ke, H.; Guo, S.; Wu, D.; Wu, W.; Zhao, L. Potential Effects of Umami Ingredients on Human Health: Pros and Cons. Crit. Rev. Food Sci. Nutr. 2020, 60, 2294–2302. [Google Scholar] [CrossRef]
- Teo, P.S.; Tso, R.; van Dam, R.M.; Forde, C.G. Taste of Modern Diets: The Impact of Food Processing on Nutrient Sensing and Dietary Energy Intake. J. Nutr. 2022, 152, 200–210. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Moubarac, J.-C.; Levy, R.B.; Canella, D.S.; da Costa Louzada, M.L.; Cannon, G. Household Availability of Ultra-Processed Foods and Obesity in Nineteen European Countries. Public Health Nutr. 2018, 21, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.; Grimes, C.; Baker, P.; Sievert, K.; Lawrence, M.A. The Drivers, Trends and Dietary Impacts of Non-Nutritive Sweeteners in the Food Supply: A Narrative Review. Nutr. Res. Rev. 2021, 34, 185–208. [Google Scholar] [CrossRef]
- Shum, B.; Georgia, S. The Effects of Non-Nutritive Sweetener Consumption in the Pediatric Populations: What We Know, What We Don’t, and What We Need to Learn. Front. Endocrinol. 2021, 12, 625415. [Google Scholar] [CrossRef]
- Cummings, J.R.; Schiestl, E.T.; Tomiyama, A.J.; Mamtora, T.; Gearhardt, A.N. Highly Processed Food Intake and Immediate and Future Emotions in Everyday Life. Appetite 2022, 169, 105868. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.S.; Del-Ponte, B.; Assunção, M.C.F.; Santos, I.S. Consumption of Ultra-Processed Foods and Body Fat during Childhood and Adolescence: A Systematic Review. Public Health Nutr. 2018, 21, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Coricelli, C.; Toepel, U.; Notter, M.-L.; Murray, M.M.; Rumiati, R.I. Distinct Brain Representations of Processed and Unprocessed Foods. Eur. J. Neurosci. 2019, 50, 3389–3401. [Google Scholar] [CrossRef] [PubMed]
- Gibney, M.J.; Forde, C.G.; Mullally, D.; Gibney, E.R. Ultra-Processed Foods in Human Health: A Critical Appraisal. Am. J. Clin. Nutr. 2017, 106, 717–724. [Google Scholar] [CrossRef]
- Pineda, E.; Poelman, M.P.; Aaspõllu, A.; Bica, M.; Bouzas, C.; Carrano, E.; De Miguel-Etayo, P.; Djojosoeparto, S.; Blenkuš, M.G.; Graca, P.; et al. Policy Implementation and Priorities to Create Healthy Food Environments Using the Healthy Food Environment Policy Index (Food-EPI): A Pooled Level Analysis across Eleven European Countries. Lancet Reg. Health Eur. 2022, 23, 100522. [Google Scholar] [CrossRef]
- Popkin, B.M.; Barquera, S.; Corvalan, C.; Hofman, K.J.; Monteiro, C.; Ng, S.W.; Swart, E.C.; Taillie, L.S. Towards Unified and Impactful Policies to Reduce Ultra-Processed Food Consumption and Promote Healthier Eating. Lancet Diabetes Endocrinol. 2021, 9, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Cotter, T.; Kotov, A.; Wang, S.; Murukutla, N. “Warning: Ultra-Processed”-A Call for Warnings on Foods That Aren’t Really Foods. BMJ Glob. Health 2021, 6, e007240. [Google Scholar] [CrossRef] [PubMed]
- Dereń, K.; Dembiński, Ł.; Wyszyńska, J.; Mazur, A.; Weghuber, D.; Łuszczki, E.; Hadjipanayis, A.; Koletzko, B. Front-Of-Pack Nutrition Labelling: A Position Statement of the European Academy of Paediatrics and the European Childhood Obesity Group. Ann. Nutr. Metab. 2021, 77, 23–28. [Google Scholar] [CrossRef]
- Scharf, R.J.; DeBoer, M.D. Sugar-Sweetened Beverages and Children’s Health. Annu. Rev. Public Health 2016, 37, 273–293. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.S.; Bensignor, M.O.; Hsia, D.S.; Shoemaker, A.H.; Shih, W.; Peterson, C.; Varghese, S.T. Phentermine/Topiramate for the Treatment of Adolescent Obesity. NEJM Evid. 2022, 1, 14. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calcaterra, V.; Cena, H.; Rossi, V.; Santero, S.; Bianchi, A.; Zuccotti, G. Ultra-Processed Food, Reward System and Childhood Obesity. Children 2023, 10, 804. https://doi.org/10.3390/children10050804
Calcaterra V, Cena H, Rossi V, Santero S, Bianchi A, Zuccotti G. Ultra-Processed Food, Reward System and Childhood Obesity. Children. 2023; 10(5):804. https://doi.org/10.3390/children10050804
Chicago/Turabian StyleCalcaterra, Valeria, Hellas Cena, Virginia Rossi, Sara Santero, Alice Bianchi, and Gianvincenzo Zuccotti. 2023. "Ultra-Processed Food, Reward System and Childhood Obesity" Children 10, no. 5: 804. https://doi.org/10.3390/children10050804
APA StyleCalcaterra, V., Cena, H., Rossi, V., Santero, S., Bianchi, A., & Zuccotti, G. (2023). Ultra-Processed Food, Reward System and Childhood Obesity. Children, 10(5), 804. https://doi.org/10.3390/children10050804