Reliability and Validity of the 10-Meter Walk Test (10MWT) in Adolescents and Young Adults with Down Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Size
2.2. Participants
2.3. Procedure
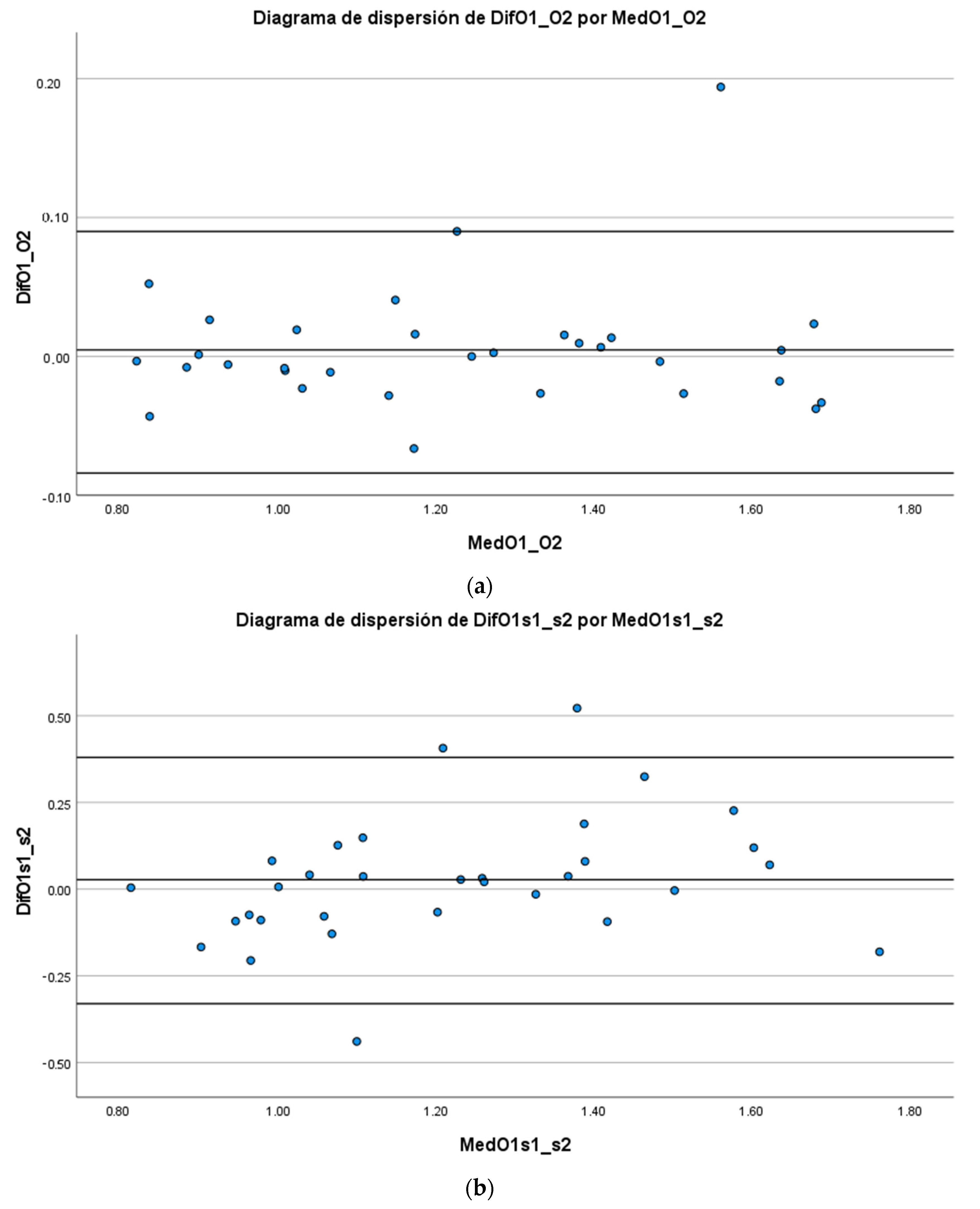
2.4. Statistical Analysis
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Looper, J.; Benjamin, D.; Nolan, M.; Schumm, L. What to measure when determining orthotic needs in children with down syndrome: A pilot study. Pediatr. Phys. Ther. 2012, 24, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Palisano, R.J.; Walter, S.D.; Russell, D.J.; Rosenbaum, P.L.; Gémus, M.; Galuppi, B.E.; Cunningham, L. Gross motor function of children with Down syndrome: Creation of motor growth curves. Arch. Phys. Med. Rehabil. 2001, 82, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Foley, C.; Killeen, O.G. Musculoskeletal anomalies in children with Down syndrome: An observational study. Arch. Dis. Child. 2001, 104, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Elshemy, S.A. Comparative study: Parameters of gait in Down syndrome versus matched obese and healthy children. Egypt. J. Med. Hum. Genet. 2013, 14, 285–291. [Google Scholar] [CrossRef]
- Galli, M.; Cimolin, V.; Pau, M.; Costici, P.; Albertini, G. Relationship between flat foot condition and gait pattern alterations in children with Down syndrome. J. Intellect. Disabil. Res. 2013, 58, 269–276. [Google Scholar] [CrossRef]
- Rigoldi, C.; Galli, M.; Albertini, G. Gait development during lifespan in subjects with Down syndrome. Res. Dev. Disabil. 2011, 32, 158–163. [Google Scholar] [CrossRef]
- Zago, M.; Duarte, N.A.C.; Grecco, L.A.C.; Condoluci, C.; Oliveira, C.S.; Galli, M. Gait and postural control patterns and rehabilitation in Down syndrome: A systematic review. J. Phys. Ther. Sci. 2020, 32, 303–314. [Google Scholar] [CrossRef]
- Skiba, A.; Marchewka, J.; Skiba, A.; Podsiadło, S.; Sulowska, I.; Chwała, W.; Marchewka, A. Evaluation of the Effectiveness of Nordic Walking Training in Improving the Gait of Persons with Down Syndrome. Biomed. Res. Int. 2019, 2019, 6353292. [Google Scholar] [CrossRef]
- Smith, B.A.; Ashton-Miller, J.A.; Ulrich, B.D. Gait adaptations in response to perturbations in adults with Down syndrome. Gait. Posture 2010, 32, 149–154. [Google Scholar] [CrossRef]
- Nicolini-Panisson, R.D.; Donadio, M.V. Normative values for the Timed ‘Up and Go’ test in children and adolescents and validation for individuals with Down syndrome. Dev. Med. Child Neurol. 2014, 56, 490–497. [Google Scholar] [CrossRef]
- de Baptista, C.R.J.A.; Vicente, A.M.; Souza, M.A.; Cardoso, J.; Ramalho, V.M.; Mattiello-Sverzut, A.C. Methods of 10-Meter Walk Test and Repercussions for Reliability Obtained in Typically Developing Children. Rehabil. Res. Pract. 2020, 2020, 4209812. [Google Scholar] [CrossRef]
- Graser, J.V.; Letsch, C.; van Hedel, H.J.A. Reliability of timed walking tests and temporo-spatial gait parameters in youths with neurological gait disorders. BMC Neurol. 2016, 16, 15. [Google Scholar] [CrossRef]
- Pirpiris, M.; Wilkinson, A.J.; Rodda, J.; Nguyen, T.C.; Baker, R.J.; Nattrass, G.R.; Graham, H.K. Walking speed in children and young adults with neuromuscular disease: Comparison between two assessment methods. J. Pediatr. Orthop. 2003, 23, 302–307. [Google Scholar] [CrossRef]
- Walter, S.D.; Eliasziw, M.; Donner, A. Sample size and optimal designs for reliability studies. Stat. Med. 1998, 17, 101–110. [Google Scholar] [CrossRef]
- Germanotta, M.; Cruciani, A.; Pecchioli, C.; Loreti, S.; Spedicato, A.; Meotti, M.; Mosca, R.; Speranza, G.; Cecchi, F.; Giannarelli, G.; et al. Reliability, validity and discriminant ability of the instrumental indices provided by a novel planar robotic device for upper limb rehabilitation. J. Neuroeng. Rehabil. 2018, 15, 39. [Google Scholar] [CrossRef]
- Germanotta, M.; The FDG Robotic Rehabilitation Group; Gower, V.; Papadopoulou, D.; Cruciani, A.; Pecchioli, C.; Mosca, R.; Speranza, G.; Falsini, C.; Cecchi, F.; et al. Reliability, validity and discriminant ability of a robotic device for finger training in patients with subacute stroke. J. Neuroeng. Rehabil. 2020, 17, 1. [Google Scholar] [CrossRef]
- Williams, E.N.; Carroll, S.G.; Reddihough, D.S.; Phillips, B.A.; Galea, M.P. Investigation of the timed ‘up & go’ test in children. Dev. Med. Child Neurol. 2005, 47, 518–524. [Google Scholar] [CrossRef]
- Hobart, J.C.; Lamping, D.L.; Thompson, A.J. Evaluating neurological outcome measures: The bare essentials. J. Neurol. Neurosurg. Psychiatry 1996, 60, 127–130. [Google Scholar] [CrossRef]
- Gadotti, I.; Vieira, E.; Magee, D. Importance and clarification of measurement properties in rehabilitation. Rev. Bras. Fisioter. 2006, 10, 137–146. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 2016, 15, 55–163. [Google Scholar] [CrossRef]
- de Vet, H.C.W.; Terwee, C.B.; Knol, D.L.; Bouter, L.M. When to use agreement versusreliability measures. J. Clin. Epidemiol. 2006, 59, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.M.; Altman, D.G. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999, 8, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Salter, K.; Jutai, J.W.; Teasell, R.; Foley, N.C.; Bitensky, J.; Bayley, M. Issues for selection of outcome measures in stroke rehabilitation: ICF activity. Disabil. Rehabil. 2005, 27, 315–340. [Google Scholar] [CrossRef] [PubMed]
- Beerse, M.; Henderson, G.; Liang, H.; Ajisafe, T.; Wu, J. Variability of spatiotemporal gait parameters in children with and without Down syndrome during treadmill walking. Gait. Posture 2019, 68, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Selby-Silverstein, L.; Hillstrom, H.J.; Palisano, R.J. The effect of foot orthoses on standing foot posture and gait of young children with Down syndrome. Neurobiol. Rehabil. 2001, 16, 183–193. [Google Scholar] [CrossRef]
- Martin, K. Effects of supramalleolar orthoses on postural stability in children with Down syndrome. Dev. Med. Child Neurol. 2004, 48, 406–411. [Google Scholar] [CrossRef]
- Watson, M.L. Refining the ten-metre walking test for use with neurologically impaired people. Physiotherapy 2002, 88, 386–397. [Google Scholar] [CrossRef]
- van Loo, M.A.; Moseley, A.M.; Bosman, J.M.; de Bie, R.A.; Hassett, L. Test-re-test reliability of walking speed, step length and step width measurement after traumatic brain injury: A pilot study. Brain Inj. 2004, 18, 1041–1048. [Google Scholar] [CrossRef]
- Chrysagis, N.; Skordilis, E.K.; Koutsouki, D. Validity and clinical utility of functional assessments in children with cerebral palsy. Arch. Phys. Med. Rehabil. 2014, 95, 369–374. [Google Scholar] [CrossRef]
- Drouin, L.M.; Malouin, F.; Richards, C.L.; Marcoux, S. Correlation between the gross motor function measure scores and gait spatiotemporal measures in children with neurological impairments. Dev. Med. Child Neurol. 1996, 38, 1007–1019. [Google Scholar] [CrossRef]
- Thompson, P.; Beath, T.; Bell, J.; Jacobson, G.; Phair, T.; Salbach, N.M.; Wright, F.V. Test-retest reliability of the 10-metre fast walk test and 6-minute walk test in ambulatory school-aged children with cerebral palsy. Dev. Med. Child Neurol. 2008, 50, 370–376. [Google Scholar] [CrossRef]
- Wagner, J.M.; Rhodes, J.A.; Patten, C. Reproducibility and minimal detectable change of three-dimensional kinematic analysis of reaching tasks in people with hemiparesis after stroke. Phys. Ther. 2008, 88, 652–663. [Google Scholar] [CrossRef]
- Steffen, T.; Seney, M. Test-retest reliability and minimal detectable change on balance and ambulation tests, the 36-item short-form health survey, and the unified Parkinson disease rating scale in people with parkinsonism. Phys. Ther. 2008, 88, 733–746. [Google Scholar] [CrossRef]
- Lam, T.; Noonan, V.K.; Eng, J.J.; SCIRE Research Team. A systematic review of functional ambulation outcome measures in spinal cord injury. Spinal. Cord. 2008, 46, 246–254. [Google Scholar] [CrossRef]
- Perera, S.; Mody, S.; Woodman, R.C.; Studenski, S.A. Meaningful change and responsiveness in common physical performance measures in older adults. J. Am. Geriatr. Soc. 2006, 54, 743–749. [Google Scholar] [CrossRef]
- Hiengkaew, V.; Jitaree, K.; Chaiyawat, P. Minimal detectable changes of the Berg Balance Scale, Fugl-Meyer Assessment Scale, Timed “Up & Go” Test, gait speeds, and 2-minute walk test in individuals with chronic stroke with different degrees of ankle plantarflexor tone. Arch. Phys. Med. Rehabil. 2012, 93, 1201–1208. [Google Scholar]
- Paltamaa, J.; Sarasoja, T.; Leskinen, E.; Wikstrom, J.; Malkia, E. Measuring deterioration in international classification of functioning domains of people with multiple sclerosis who are ambulatory. Phys. Ther. 2008, 88, 176–190. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The timed ‘Up & Go’: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar]
- Pau, M.; Condoluci, C.; Zago, M.; Galli, M. Men and women with Down syndrome exhibit different kinematic (but not spatio-temporal) gait patterns. J. Intellect. Disabil. Res. 2018, 63, 64–71. [Google Scholar] [CrossRef]
- Zago, M.; Condoluci, C.; Pau, M.; Galli, M. Sex differences in the gait kinematics of patients with Down Syndrome: A preliminary report. J. Rehabil. Med. 2019, 51, 144–146. [Google Scholar] [CrossRef]
- Cimolin, V.; Galli, M.; Grugni, G.; Vismara, L.; Albertini, G.; Rigoldi, C.; Capodaglio, P. Gait patterns in Prader-Willi and Down Syndrome patients. J. Neuroeng. Rehabil. 2010, 7, 28. [Google Scholar] [CrossRef] [PubMed]
- Solway, S.; Brooks, D.; Lacasse, Y.; Thomas, S. A qualitative systematic overview of the measurement properties of functional walk tests used in the cardiorespiratory domain. Chest 2001, 119, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Rintala, P.; McCubbin, J.A.; Downs, S.B.; Fox, S.D. Cross validation of the 1-mile walking test for men with mental retardation. Med. Sci. Sports Exerc. 1997, 29, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Draheim, C.C.; Laurie, N.E.; McCubbin, J.A.; Perkins, J.L. Validity of a modified aerobic fitness test for adults with mental retardation. Med. Sci. Sports Exerc. 1999, 31, 1849–1854. [Google Scholar] [CrossRef]
| Session 1 | Session 2 | ICC | 95% CI | p-Value | |
|---|---|---|---|---|---|
| 10MWT | 1.23 (0.28) | 1.20 (0.22) | 0.857 | 0.987 to 0.997 | <0.01 * |
| 10MWT | RRater 1 vs. Rater 2 | |||
|---|---|---|---|---|
| Rater 1 | Rater 2 | ICC | 95% CI | p-Value |
| 1.23 (0.28) | 1.22 (0.27) | 0.994 | 0.989 to 0.997 | <0.01 * |
| Rater 1 vs. TUG | Rater 2 vs. TUG | |||||||
|---|---|---|---|---|---|---|---|---|
| Rater 1 | Rater 2 | TUG | r | p-Value | 95% CI | r | p-Value | 95% CI |
| 5.12 (1.19) | 5.12 (1.18) | 7.63 (1.35) | 0.553 | <0.01 * | −0.756 to −0.253 | 0.522 | 0.002 * | −0.737 to −0.212 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-González, J.L.; Llamas-Ramos, I.; Llamas-Ramos, R.; Molina-Rueda, F.; Carratalá-Tejada, M.; Cuesta-Gómez, A. Reliability and Validity of the 10-Meter Walk Test (10MWT) in Adolescents and Young Adults with Down Syndrome. Children 2023, 10, 655. https://doi.org/10.3390/children10040655
Sánchez-González JL, Llamas-Ramos I, Llamas-Ramos R, Molina-Rueda F, Carratalá-Tejada M, Cuesta-Gómez A. Reliability and Validity of the 10-Meter Walk Test (10MWT) in Adolescents and Young Adults with Down Syndrome. Children. 2023; 10(4):655. https://doi.org/10.3390/children10040655
Chicago/Turabian StyleSánchez-González, Juan Luis, Inés Llamas-Ramos, Rocío Llamas-Ramos, Francisco Molina-Rueda, María Carratalá-Tejada, and Alicia Cuesta-Gómez. 2023. "Reliability and Validity of the 10-Meter Walk Test (10MWT) in Adolescents and Young Adults with Down Syndrome" Children 10, no. 4: 655. https://doi.org/10.3390/children10040655
APA StyleSánchez-González, J. L., Llamas-Ramos, I., Llamas-Ramos, R., Molina-Rueda, F., Carratalá-Tejada, M., & Cuesta-Gómez, A. (2023). Reliability and Validity of the 10-Meter Walk Test (10MWT) in Adolescents and Young Adults with Down Syndrome. Children, 10(4), 655. https://doi.org/10.3390/children10040655








