Iron Replacement and Redox Balance in Non-Anemic and Mildly Anemic Iron Deficiency COPD Patients: Insights from a Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population, Design, and Ethics
2.2. Inclusion Criteria
2.3. Iron Replacement: Placebo and Treatment Arms
2.4. Exclusion Criteria
2.5. Anthropometric and Lung Function Assessment
2.6. Blood Samples
2.7. Molecular Biology Analysis
2.8. Statistical Analysis
3. Results
3.1. Clinical Characteristics at Baseline
3.2. Clinical Characteristics Following Iron Replacement
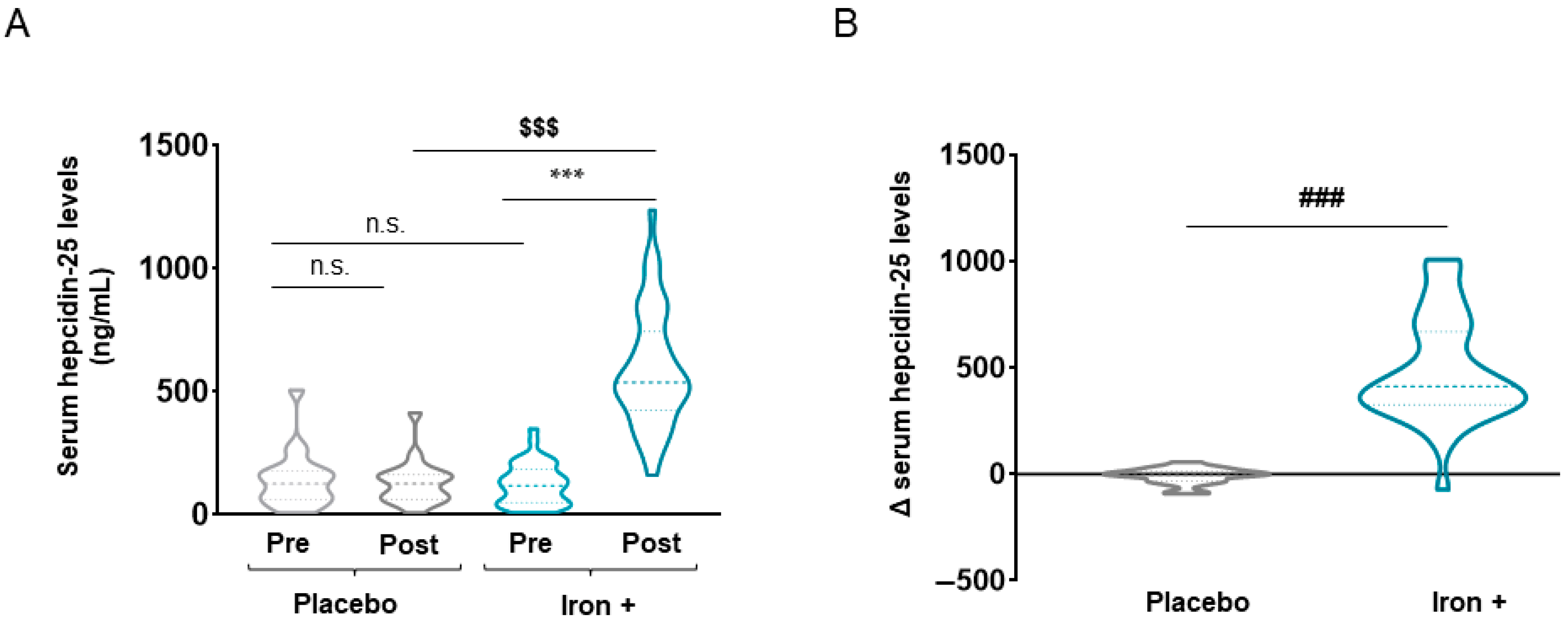
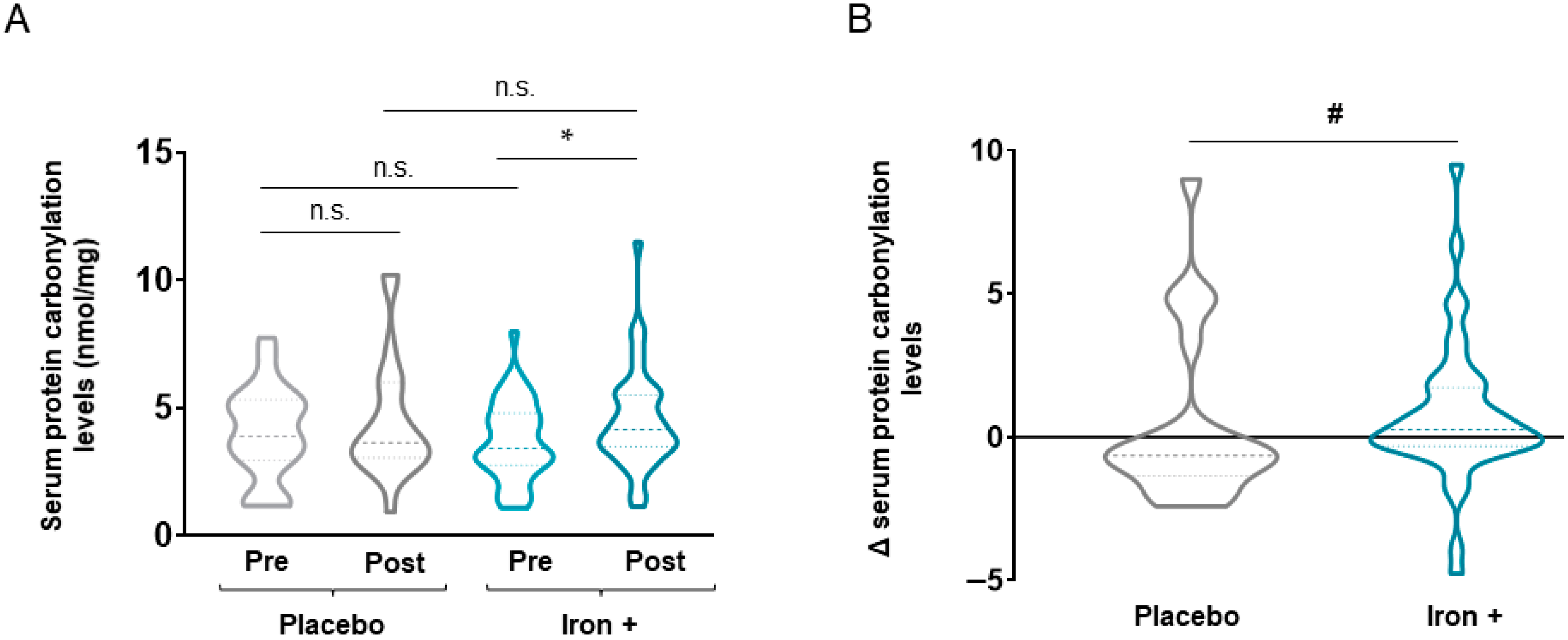
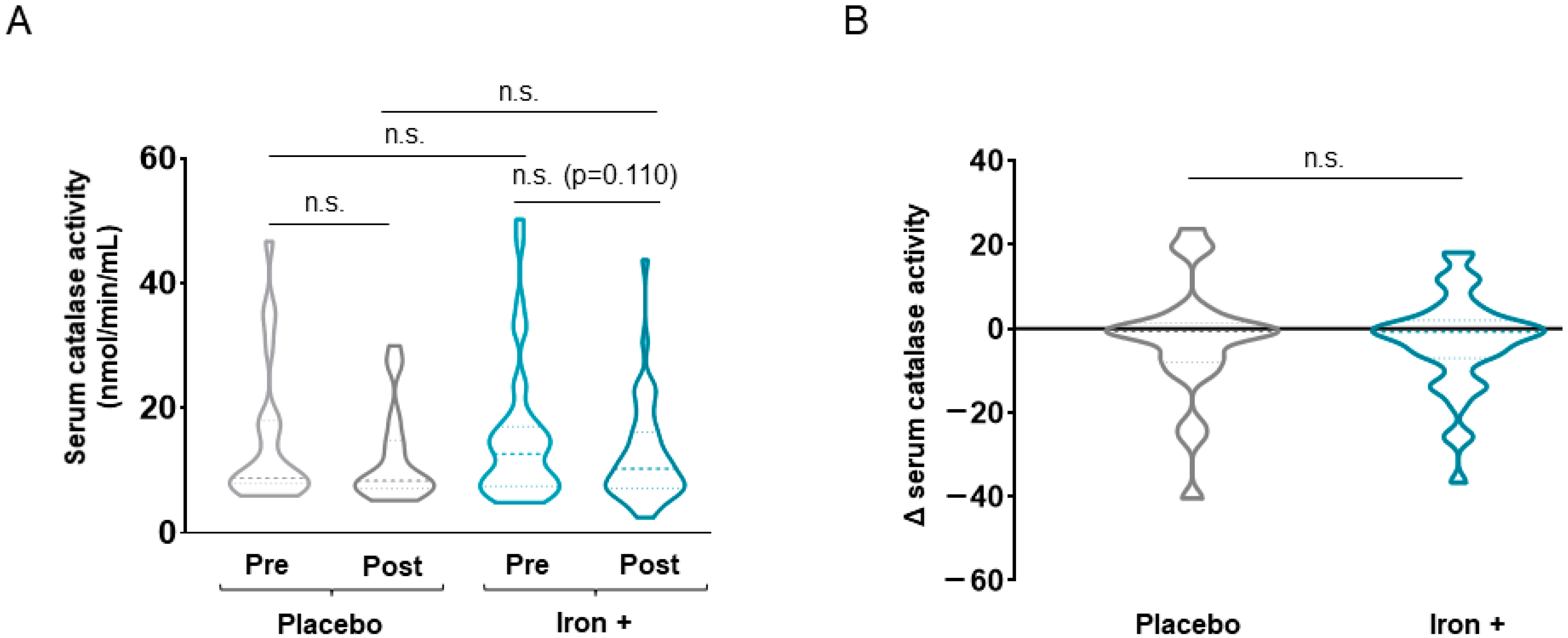
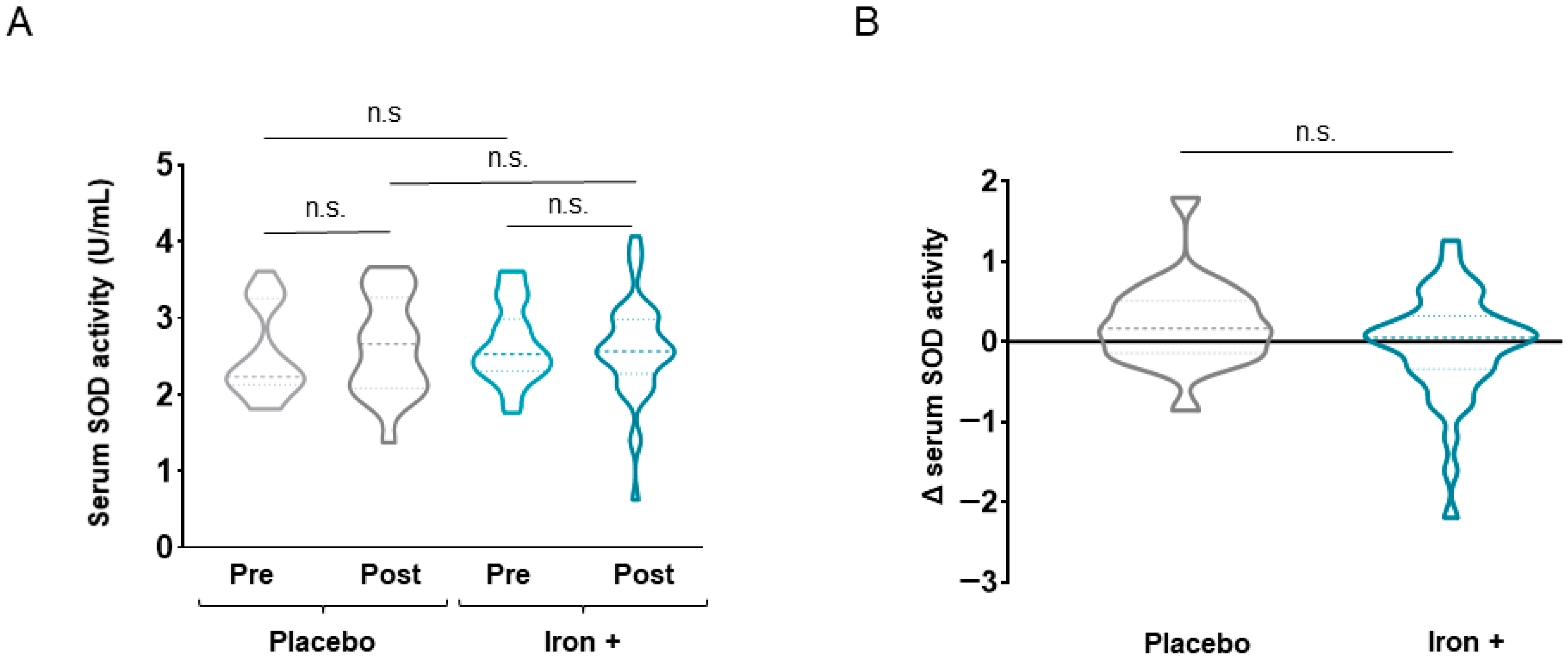
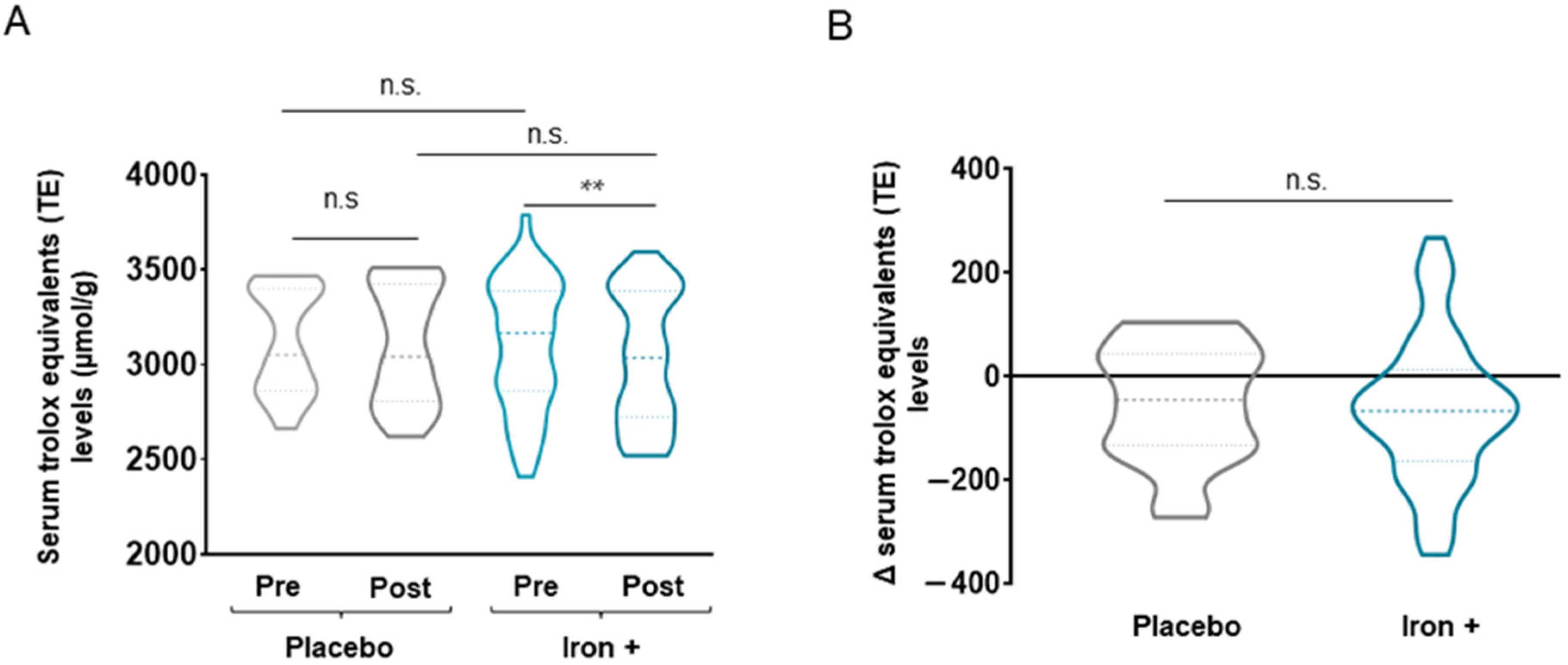
3.2.1. Oxidative Stress Markers
3.2.2. Antioxidant Markers
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jaitovich, A.; Barreiro, E. Skeletal Muscle Dysfunction in Chronic Obstructive Pulmonary Disease. What We Know and Can Do for Our Patients. Am. J. Respir. Crit. Care Med. 2018, 198, 175–186. [Google Scholar] [CrossRef]
- Kwan, H.Y.; Maddocks, M.; Nolan, C.M.; Jones, S.E.; Patel, S.; Barker, R.E.; Kon, S.S.C.; Polkey, M.I.; Cullinan, P.; Man, W.D.-C. The prognostic significance of weight loss in chronic obstructive pulmonary disease-related cachexia: A prospective cohort study. J. Cachexia Sarcopenia Muscle 2019, 10, 1330–1338. [Google Scholar] [CrossRef] [Green Version]
- Shrikrishna, D.; Patel, M.; Tanner, R.J.; Seymour, J.M.; Connolly, B.A.; Puthucheary, Z.A.; Walsh, S.L.F.; Bloch, S.A.; Sidhu, P.S.; Hart, N.; et al. Quadriceps wasting and physical inactivity in patients with COPD. Eur. Respir. J. 2012, 40, 1115–1122. [Google Scholar] [CrossRef] [Green Version]
- Volani, C.; Paglia, G.; Smarason, S.; Pramstaller, P.; Demetz, E.; Pfeifhofer-Obermair, C.; Weiss, G. Metabolic Signature of Dietary Iron Overload in a Mouse Model. Cells 2018, 7, 264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muckenthaler, M.; Rivella, S.; Hentze, M.; Galy, B. A Red Carpet for Iron Metabolism. Cell 2017, 168, 344–361. [Google Scholar] [CrossRef] [Green Version]
- Pizzini, A.; Aichner, M.; Sonnweber, T.; Tancevski, I.; Weiss, G.; Löffler-Ragg, J. The Significance of iron deficiency and anemia in a real-life COPD cohort. Int. J. Med. Sci. 2020, 17, 2232. [Google Scholar] [CrossRef]
- Cloonan, S.M.; Mumby, S.; Adcock, I.M.; Choi, A.M.K.; Chung, K.F.; Quinlan, G.J. The iron-y of iron overload and iron deficiency in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2017, 196, 1103–1112. [Google Scholar] [CrossRef] [Green Version]
- Boutou, A.K.; Karrar, S.; Hopkinson, N.S.; Polkey, M.I. Anemia and Survival in Chronic Obstructive Pulmonary Disease: A Dichotomous rather than a Continuous Predictor. Respiration 2013, 85, 126–131. [Google Scholar] [CrossRef]
- Martinez-Rivera, C.; Portillo, K.; Muñoz-Ferrer, A.; Martínez-Ortiz, M.L.; Molins, E.; Serra, P.; Ruiz-Manzano, J.; Morera, J. Anemia is a Mortality Predictor in Hospitalized Patients for COPD Exacerbation. COPD J. Chronic Obstr. Pulm. Dis. 2012, 9, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Portillo, K.; Martinez-Rivera, C.; Ruiz-Manzano, J. Anaemia in chronic obstructive pulmonary disease. Does it really matter? Int. J. Clin. Pract. 2013, 67, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Cappellini, M.D.; Comin-Colet, J.; de Francisco, A.; Dignass, A.; Doehner, W.; Lam, C.S.; Macdougall, I.C.; Rogler, G.; Camaschella, C.; Kadir, R.; et al. Iron deficiency across chronic inflammatory conditions: International expert opinion on definition, diagnosis, and management. Am. J. Hematol. 2017, 92, 1068–1078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, G. Iron and anemia of chronic disease. Kidney Int. 1999, 55, S12–S17. [Google Scholar] [CrossRef] [Green Version]
- Duru, S.; Bilgin, E.; Ardiç, S. Hepcidin: A useful marker in chronic obstructive pulmonary disease. Ann. Thorac. Med. 2012, 7, 31. [Google Scholar] [CrossRef] [PubMed]
- Kossiva, L.; Soldatou, A.; Gourgiotis, D.I.; Stamati, L.; Tsentidis, C. Serum hepcidin: Indication of its role as an “acute phase” marker in febrile children. Ital. J. Pediatr. 2013, 39, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, J.E.; Shanthi, B.; Selvi, V.S.K. Serum Hepcidin as an Inflammatory Marker in Chronic Kidney Disease. J. Pharm. Res. Int. 2020, 32, 40–45. [Google Scholar] [CrossRef]
- Chen, L.; Ma, B.; Liu, X.; Hao, Y.; Yang, X.; Liu, M. H2O2 induces oxidative stress damage through the BMP-6/SMAD/hepcidin axis. Dev. Growth Differ. 2020, 62, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Imam, M.U.; Zhang, S.; Ma, J.; Wang, H.; Wang, F. Antioxidants Mediate Both Iron Homeostasis and Oxidative Stress. Nutrients 2017, 9, 671. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, P.J. Regulation of Iron Metabolism by Hepcidin under Conditions of Inflammation *. J. Biol. Chem. 2015, 290, 18975–18983. [Google Scholar] [CrossRef] [Green Version]
- Trinder, D.; Ayonrinde, O.T.; Olynyk, J.K. HCV, Iron, and Oxidative Stress: The New Choreography of Hepcidin. Gastroenterology 2008, 134, 348–351. [Google Scholar] [CrossRef]
- Hecker, L. Mechanisms and consequences of oxidative stress in lung disease: Therapeutic implications for an aging populace. Am. J. Physiol. Cell. Mol. Physiol. 2018, 314, L642–L653. [Google Scholar] [CrossRef]
- Thimmulappa, R.K.; Chattopadhyay, I.; Rajasekaran, S. Oxidative Stress Mechanisms in the Pathogenesis of Environmental Lung Diseases. Oxidative Stress Lung Dis. 2020, 2, 103–137. [Google Scholar] [CrossRef]
- Van der Vliet, A.; Janssen-Heininger, Y.M.W.; Anathy, V. Oxidative stress in chronic lung disease: From mitochondrial dysfunction to dysregulated redox signaling. Mol. Asp. Med. 2018, 63, 59–69. [Google Scholar] [CrossRef]
- Barreiro, E.; Peinado, V.I.; Galdiz, J.B.; Ferrer, E.; Marin-Corral, J.; Sánchez, F.; Gea, J.; Barberà, J.A. Cigarette Smoke–induced Oxidative Stress. Am. J. Respir. Crit. Care Med. 2012, 182, 477–488. [Google Scholar] [CrossRef] [Green Version]
- Barnes, P.J. Oxidative stress-based therapeutics in COPD. Redox Biol. 2020, 33, 101544. [Google Scholar] [CrossRef]
- Emerit, J.; Beaumont, C.; Trivin, F. Iron metabolism, free radicals, and oxidative injury. Biomed. Pharmacother. 2001, 55, 333–339. [Google Scholar] [CrossRef]
- Papanikolao, G.; Pantopoulos, K. Iron metabolism and toxicity. Toxicol. Appl. Pharmacol. 2005, 202, 199–211. [Google Scholar] [CrossRef]
- Prats, M.; Font, R.; García, C.; Muñoz-Cortés, M.; Cabré, C.; Jariod, M.; Romeu, M.; Giralt, M.; Martinez-Vea, A. Oxidative stress markers in predicting response to treatment with ferric carboxymaltose in nondialysis chronic kidney disease patients. Clin. Nephrol. 2014, 81, 419–426. [Google Scholar] [CrossRef]
- Altun, D.; Kurekci, A.E.; Gursel, O.; Hacıhamdioglu, D.O.; Kurt, I.; Aydın, A.; Ozcan, O. Malondialdehyde, Antioxidant Enzymes, and Renal Tubular Functions in Children with Iron Deficiency or Iron-Deficiency Anemia. Biol. Trace Elem. Res. 2014, 161, 48–56. [Google Scholar] [CrossRef]
- Barreiro, E.; Fermoselle, C.; Mateu-Jimenez, M.; Sánchez-Font, A.; Pijuan, L.; Gea, J.; Curull, V. Oxidative stress and inflammation in the normal airways and blood of patients with lung cancer and COPD. Free Radic. Biol. Med. 2013, 65, 859–871. [Google Scholar] [CrossRef]
- Puig-Vilanova, E.; Rodriguez, D.A.; Lloreta, J.; Ausin, P.; Pascual-Guardia, S.; Broquetas, J.; Roca, J.; Gea, J.; Barreiro, E. Oxidative stress, redox signaling pathways, and autophagy in cachectic muscles of male patients with advanced COPD and lung cancer. Free Radic. Biol. Med. 2015, 79, 91–108. [Google Scholar] [CrossRef] [PubMed]
- Zinellu, E.; Zinellu, A.; Fois, A.; Carru, C.; Pirina, P. Circulating biomarkers of oxidative stress in chronic obstructive pulmonary disease: A systematic review. Respir. Res. 2016, 17, 150. [Google Scholar] [CrossRef] [Green Version]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and Clinical Significance of Biomarkers of Oxidative Stress in Humans. Oxid. Med. Cell. Longev. 2017, 2017, 6501046. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef] [Green Version]
- Kasapoglu, M.; Ozben, T. Alterations of antioxidant enzymes and oxidative stress markers in aging. Exp. Gerontol. 2001, 36, 209–220. [Google Scholar] [CrossRef]
- Martín, C.; Rodó, A.; Echeverría, D.; Admetlló, M.; Duran, X.; Alvarado, M.; Gea, J.; Barreiro, E.; Rodríguez, D. Intravenous iron replacement improves exercise tolerance in COPD: A randomised trial. Arch. Bronconeumol. 2021. [Google Scholar] [CrossRef]
- Kurtoglu, E.; Ugur, A.; Baltaci, A.K.; Undar, L. Effect of iron supplementation on oxidative stress and antioxidant status in iron-deficiency anemia. Biol. Trace Elem. Res. 2003, 96, 117–123. [Google Scholar] [CrossRef]
- Sundaram, R.; Selvaraj, N.; Vijayan, G.; Bobby, Z.; Hamide, A.; Rattina Dasse, N. Increased plasma malondialdehyde and fructosamine in iron deficiency anemia: Effect of treatment. Biomed. Pharmacother. 2007, 61, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gøtzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, 869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Global Initiative for Chronic Obstructive Lung Disease. GOLD 2021. 2021. Available online: www.goldcopd.org (accessed on 7 September 2021).
- Barberan-Garcia, A.; Rodríguez, D.A.; Blanco, I.; Gea, J.; Torralba, Y.; Arbillaga-Etxarri, A.; Barberà, J.A.; Vilarõ, J.; Roca, J.; Orozco-Levi, M. Non-anaemic iron deficiency impairs response to pulmonary rehabilitation in COPD. Respirology 2015, 20, 1089–1095. [Google Scholar] [CrossRef]
- Anker, S.D.; Comin Colet, J.; Filippatos, G.; Willenheimer, R.; Dickstein, K.; Drexler, H.; Lüscher, T.F.; Bart, B.; Banasiak, W.; Niegowska, J.; et al. Ferric Carboxymaltose in Patients with Heart Failure and Iron Deficiency. N. Engl. J. Med. 2009, 361, 2436–2448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Barreiro, E.; Salazar-Degracia, A.; Sancho-Muñoz, A.; Gea, J. Endoplasmic reticulum stress and unfolded protein response profile in quadriceps of sarcopenic patients with respiratory diseases. J. Cell. Physiol. 2019, 234, 11315–11329. [Google Scholar] [CrossRef]
- Puig-Vilanova, E.; Martínez-Llorens, J.; Ausin, P.; Roca, J.; Gea, J.; Barreiro, E. Quadriceps muscle weakness and atrophy are associated with a differential epigenetic profile in advanced COPD. Clin. Sci. 2015, 128, 905–921. [Google Scholar] [CrossRef]
- Roca, J.; Burgos, F.; Barberà, J.A.; Sunyer, J.; Rodriguez-Roisin, R.; Castellsagué, J.; Sanchis, J.; Antóo, J.M.; Casan, P.; Clausen, J.L. Prediction equations for plethysmographic lung volumes. Respir. Med. 1998, 92, 454–460. [Google Scholar] [CrossRef] [Green Version]
- Roca, J.; Burgos, F.; Sunyer, J.; Saez, M.; Chinn, S.; Anto, J.; Rodriguez-Roisin, R.; Quanjer, P.; Nowak, D.; Burney, P. References values for forced spirometry. Group of the European Community Respiratory Health Survey. Eur. Respir. J. 1998, 11, 1354–1362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roca, J.; Vargas, C.; Cano, I.; Selivanov, V.; Barreiro, E.; Maier, D.; Falciani, F.; Wagner, P.; Cascante, M.; Garcia-Aymerich, J.; et al. Chronic Obstructive Pulmonary Disease heterogeneity: Challenges for health risk assessment, stratification and management. J. Transl. Med. 2014, 12, S3. [Google Scholar] [CrossRef] [PubMed]
- Taheri, N.; Mojerloo, M.; Hadad, M.; Mirkarimi, H.; Nejad, R.; Joshaghani, H. Comparison of serum levels of hepcidin and pro-hepcidin in hemodialysis patients and healthy subjects. Saudi J. Kidney Dis. Transpl. 2015, 26, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Mateu-Jiménez, M.; Sánchez-Font, A.; Rodríguez-Fuster, A.; Aguiló, R.; Pijuan, L.; Fermoselle, C.; Gea, J.; Curull, V.; Barreiro, E. Redox imbalance in lung cancer of patients with underlying chronic respiratory conditions. Mol. Med. 2016, 22, 85–98. [Google Scholar] [CrossRef]
- Barreiro, E.; Rabinovich, R.; Marin-Corral, J.; Barberà, J.A.; Gea, J.; Roca, J. Chronic endurance exercise induces quadriceps nitrosative stress in patients with severe COPD. Thorax 2009, 64, 13–19. [Google Scholar] [CrossRef] [Green Version]
- Assouline, B.; Benoliel, A.; Zamberg, I.; Legouis, D.; Delhumeau, C.; Favre, M.; Andrès, A.; Toso, C.; Samii, K.; Schiffer, E. Intravenous iron supplementation after liver surgery: Impact on anemia, iron, and hepcidin levels—a randomized controlled trial. Surgery 2021, 170, 813–821. [Google Scholar] [CrossRef]
- Onken, J.E.; Bregman, D.B.; Harrington, R.A.; Morris, D.; Acs, P.; Akright, B.; Barish, C.; Bhaskar, B.S.; Smith-Nguyen, G.N.; Butcher, A.; et al. A multicenter, randomized, active-controlled study to investigate the efficacy and safety of intravenous ferric carboxymaltose in patients with iron deficiency anemia. Transfusion 2014, 54, 306–315. [Google Scholar] [CrossRef]
- Soppi, E. Intravenous iron—Personal experience of 1000 consecutive infusions. Clin. Case Rep. Rev. 2020, 6. [Google Scholar] [CrossRef]
- Nickol, A.H.; Frise, M.C.; Cheng, H.-Y.; McGahey, A.; McFadyen, B.M.; Harris-Wright, T.; Bart, N.K.; Curtis, M.K.; Khandwala, S.; O’Neill, D.P.; et al. A cross-sectional study of the prevalence and associations of iron deficiency in a cohort of patients with chronic obstructive pulmonary disease. BMJ Open 2015, 5, e007911. [Google Scholar] [CrossRef] [Green Version]
- Silverberg, D.S.; Mor, R.; Weu, M.T.; Schwartz, D.; Schwartz, I.F.; Chernin, G. Anemia and iron deficiency in COPD patients: Prevalence and the effects of correction of the anemia with erythropoiesis stimulating agents and intravenous iron. BMC Pulm. Med. 2014, 14, 24. [Google Scholar] [CrossRef] [Green Version]
- Weiss, G.; Goodnough, L.T. Anemia of Chronic Disease. N. Engl. J. Med. 2009, 352, 1011–1023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ponikowski, P.; van Veldhuisen, D.J.; Comin-Colet, J.; Ertl, G.; Komajda, M.; Mareev, V.; McDonagh, T.; Parkhomenko, A.; Tavazzi, L.; Levesque, V.; et al. Beneficial effects of long-term intravenous iron therapy with ferric carboxymaltose in patients with symptomatic heart failure and iron deficiency. Eur. Heart J. 2015, 36, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.S.; Song, S.H.; Lee, J.H.; Kim, H.-J.; Yang, H.R. Serum hepcidin levels and iron parameters in children with iron deficiency. Korean J. Hematol. 2012, 47, 286–292. [Google Scholar] [CrossRef] [Green Version]
- Cherian, S.; Forbes, D.; Cook, A.; Sanfilippo, F.; Kemna, E.; Swinkels, D.; Burgner, D. An insight into the relationships between hepcidin, anemia, infections and inflammatory cytokines in pediatric refugees: A cross-sectional study. PLoS ONE 2008, 3, e4030. [Google Scholar] [CrossRef] [Green Version]
- Sanad, M.; Gharib, A. Urinary hepcidin level as an early predictor of iron deficiency in children: A case control study. Ital. J. Pediatr. 2011, 37, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, D.R.; Eid, R.; Miller, K.A.; Boucher, E.; Mandato, C.A.; Greenwood, M.T. Intracellular second messengers mediate stress inducible hormesis and Programmed Cell Death: A review. Biochim. Biophys. Acta Mol. Cell Res. 2019, 1866, 773–792. [Google Scholar] [CrossRef] [PubMed]
- Galaris, D.; Barbouti, A.; Pantopoulos, K. Iron homeostasis and oxidative stress: An intimate relationship. Biochim. Biophys. Acta Mol. Cell Res. 2019, 1866, 118535. [Google Scholar] [CrossRef] [PubMed]
- Mimić-Oka, J.; Savić-Radojević, A.; Pljesa-Ercegovac, M.; Opacić, M.; Simić, T.; Dimković, N.; Simić, D. Evaluation of oxidative stress after repeated intravenous iron supplementation. Ren. Fail. 2005, 27, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, A.; Mahdi, A.; Chandyan, S.; Zahra, F.; Godbole, M.; Jaiswar, S.; Srivastava, V.; Negi, M. Oral iron supplementation leads to oxidative imbalance in anemic women: A prospective study. Clin. Nutr. 2011, 30, 188–193. [Google Scholar] [CrossRef] [PubMed]




| Placebo (N = 22) | Iron (N = 44) | |
|---|---|---|
| Anthropometry | ||
| Age, years | 69.2 (5.4) | 66.8 (7.3) |
| Males/females | 16/6 | 26/18 |
| BMI, kg/m2 | 25.1 (2.9) | 24.4 (3.4) |
| FFMI, kg/m2 | 16.14 (1.8) | 15.9 (2.1) |
| Smoking status | ||
| Ex-smoker, N (%) | 13 (59.1) | 20 (45.5) |
| Active, N (%) | 9 (40.9) | 24 (54.5) |
| Packs-year | 56.3 (36.0) | 51.5 (25.8) |
| Pulmonary Function | ||
| FEV1, L | 1.3 (0.3) | 1.4 (0.6) |
| FEV1, % predicted | 47.0 (10.6) | 51.6 (13.5) |
| FVC, L | 3.0 (0.7) | 2.8 (0.8) |
| FVC, % | 84.5 (13.1) | 80.7 (12.6) |
| FEV1/FVC, % | 43.5 (9.1) | 48.7 (10.4) |
| RV, % | 175.9 (40.6) | 176.3 (44.4) |
| RV/TLC, % | 59.3 (7.1) | 60.0 (12.8) |
| TLC, % | 108.5 (15.1) | 110.4 (16.1) |
| DLCO, % | 53.0 (16.7) | 52.3 (12.6) |
| KCO, % | 57.1 (16.2) | 55.0 (12.4) |
| Arterial blood gas | ||
| PaO2, kPa | 10.4 (0.9) | 10.2 (1.1) |
| PaCO2, kPa | 5.1 (0.6) | 5.1 (0.5) |
| Placebo (N = 22) | Iron (N = 44) | |||||
|---|---|---|---|---|---|---|
| Pre | Post | Δ | Pre | Post | Δ | |
| Iron status | ||||||
| Hemoglobin, g/dL | 14.9 (1.2) | 15.0 (1.2) | +0.1 (0.7) | 14.5 (1.2) | 15.7 (4.8) | +1.3 (4.8) |
| Hematocrit, % | 45.0 (3.5) | 45.5 (3.6) | +0.5 (2.8) | 43.6 (3.7) | 45.1 (4.0) *** | +1.6 (2.3) |
| MCV, fl | 90.3 (4.7) | 89.8 (5.7) | −0.5 (2.2) | 90.6 (3.7) | 91.9 (3.8) *** | +1.4 (1.8) ### |
| MCH, pg | 29.8 (2.1) | 29.6 (2.3) | −0.3 (0.7) | 30.1 (1.7) | 30.6 (1.5) ***,$ | +0.5 (0.8) ### |
| MCHC, g/dL | 33.0 (1.1) | 32.9 (1.2) | −0.2 (1.1) | 33.2 (1.0) | 33.3 (1.1) | +0.1 (0.8) |
| Serum iron, µg/dL | 83.8 (36.4) | 89.3 (26.0) | +5.4 (37.1) | 82.5 (32.5) | 108.7 (31.2) ***,$ | +26.2 (36.4) # |
| Ferritin, ng/mL | 69.0 (42.4) | 64.3 (41.6) | −4.6 (18.4) | 61.9 (36.2) | 342.2 (176.0) ***,$$$ | +280.3 (178.9) ### |
| Transferrin saturation, % | 22.6 (10.1) | 23.2 (7.4) | +0.5 (9.6) | 22.5 (11.1) | 34.8 (11.1) ***,$$$ | +12.2 (11.0) ### |
| Transferrin, g/dL | 271.3 (36.9) | 277.0 (29.2) | +8.6 (23.7) | 277.0 (41.9) | 227.5 (33.5) ***,$$$ | −49.5 (38.0) ### |
| Soluble transferrin receptor, mg/L | 3.2 (1.6) | 3.3 (1.8) | +0.3 (0.8) | 3.3 (2.1) | 2.7 (1.4) ** | −0.6 (1.1) ### |
| Inflammatory marker | ||||||
| IL-6, pg/mL | 14.0 (17.6) | 12.5 (17.7) | −0.7 (13.2) | 12.9 (19.9) | 11.4 (9.8) | 1.8 (8.9) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Peiró, M.; Martín-Ontiyuelo, C.; Rodó-Pi, A.; Piccari, L.; Admetlló, M.; Durán, X.; Rodríguez-Chiaradía, D.A.; Barreiro, E. Iron Replacement and Redox Balance in Non-Anemic and Mildly Anemic Iron Deficiency COPD Patients: Insights from a Clinical Trial. Biomedicines 2021, 9, 1191. https://doi.org/10.3390/biomedicines9091191
Pérez-Peiró M, Martín-Ontiyuelo C, Rodó-Pi A, Piccari L, Admetlló M, Durán X, Rodríguez-Chiaradía DA, Barreiro E. Iron Replacement and Redox Balance in Non-Anemic and Mildly Anemic Iron Deficiency COPD Patients: Insights from a Clinical Trial. Biomedicines. 2021; 9(9):1191. https://doi.org/10.3390/biomedicines9091191
Chicago/Turabian StylePérez-Peiró, Maria, Clara Martín-Ontiyuelo, Anna Rodó-Pi, Lucilla Piccari, Mireia Admetlló, Xavier Durán, Diego A. Rodríguez-Chiaradía, and Esther Barreiro. 2021. "Iron Replacement and Redox Balance in Non-Anemic and Mildly Anemic Iron Deficiency COPD Patients: Insights from a Clinical Trial" Biomedicines 9, no. 9: 1191. https://doi.org/10.3390/biomedicines9091191
APA StylePérez-Peiró, M., Martín-Ontiyuelo, C., Rodó-Pi, A., Piccari, L., Admetlló, M., Durán, X., Rodríguez-Chiaradía, D. A., & Barreiro, E. (2021). Iron Replacement and Redox Balance in Non-Anemic and Mildly Anemic Iron Deficiency COPD Patients: Insights from a Clinical Trial. Biomedicines, 9(9), 1191. https://doi.org/10.3390/biomedicines9091191







