Anti-COVID-19 Vaccination in Patients with Autoimmune-Autoinflammatory Disorders and Primary/Secondary Immunodeficiencies: The Position of the Task Force on Behalf of the Italian Immunological Societies
Abstract
:1. Introduction
2. COVID-19 Vaccines
| Vaccines | Composition | Cellular Immunity | Neutralizing Antibodies | Doses | Efficacy | References |
|---|---|---|---|---|---|---|
| Pfizer Comirnaty | 30 µg mRNA Spike | Yes | Yes | 2, 3 weeks apart | 95% | [28] |
| Moderna Spikevax | 100 µg mRNA Spike | Yes | Yes | 2, 4 weeks apart | 94.1% | [29] |
| AstraZeneca Vaxzevria | Non-replicating viral vector Spike DNA | Yes | Yes | 2, 4–12 weeks apart | 70% | [21,22] |
| Janssen (J&J) | Non-replicating viral vector Spike DNA | Yes | Yes | 1 | 73–82% at 14–28 days, respectively | [23,24] |
| Cansino Biological Inc. | Non-replicating viral vector Spike DNA | Yes | 1 | [25] | ||
| Sinovac CoronaVac | Inactivated whole virus | Yes | 2, 2 weeks apart | 83.5% | [30,31] | |
| Gamaleya Res. Institute | Non-replicating viral vector Spike DNA | Yes | 2, 3 weeks apart | 91.6% | [26,27] | |
| BBV152 Bharat Biotech | Inactivated whole virus | Yes | 2, 2 weeks apart | [32,33] | ||
| Novavax | Recombinant Spike+ Adjuvant | Yes | Yes | 2, 3 weeks apart | 89.7% | [19,20] |
3. Safety of COVID-19 Vaccines in Patients with AIAIDs, PIDs, and SIDs
4. Immunosuppressive/Immunomodulating Therapy and COVID-19 Vaccines
5. Should Patients with AIAIDs, PIDs, and SIDs Be Prioritized in the Access to Vaccines?
6. Recommendations
- Anti-COVID-19 mass vaccination has personal and social benefits. Preventing the disease in single individuals may allow easier achievement of herd immunity, which is needed to interrupt the viral spread and protect individuals who cannot be vaccinated.
- Vaccinations are a safe and effective tool for prevention and control of infectious diseases. Patients with AIAIDs, PIDs, and SIDs are at higher risk of infections, including those by SARS-COV-2. Few studies have addressed the issue of anti-COVID vaccination in these patients, but many are available on the safety, immunogenicity, efficacy, and possible contraindications of traditional vaccines in AIAIDs, PIDs, and SIDs patients. These studies may represent the basis on which to recommend the anti-COVID-19 vaccines [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15] (Table 1, Table 2 and Table 3)
- The vaccine is more effective the lower the immunosuppression; however, the risk of a flare-up of the underlying disease after the interruption of an immunosuppressive drug is real, thus, in general, no modification of immunosuppressive therapy either during or following vaccination is advisable. In particular cases, according to the clinical picture and the drugs used, modifications and/or discontinuations of immunosuppressive therapy may be recommended by the immunology specialist.
- In general, inactivated vaccines or vaccines containing non-infectious viral sequences may be safely administered to patients with AIAIDs, PIDs, and SIDs in clinical remission [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15] (Table 1, Table 2 and Table 3). Anti-COVID-19 vaccines of the traditional type that are composed by inactivated virus [31,32,33] or recombinant Spike protein and adjuvant [19], for which wider scientific knowledge and clinical experience are available, could preferentially be indicated in patients with AIAIDs, PIDs, and SIDs; however, no traditional vaccines are approved by the European Medicines Agency (EMA) yet (Table 4).
- Viral vector non-replicating anti-COVID-19 vaccines, such as the ones from AstraZeneca (Vaxzevria) [21,22], Janssen/Johnson & Johnson [23,24], Cansino Biological Inc. [25], and Gamaleya Research Institute [26,27] (Table 2), are known based on application to the anti-Ebola vaccine, however our knowledge of them is very limited. One problem with these vaccines is the previous and effective anti-viral vector immune response, which may totally inactivate the vaccine; however, this problem involves the general population, not only patients with AIAIDs, PIDs, and SIDs, and may be reduced by using viral vectors from primates. Although during the registration studies substantial safety of these vaccines has been observed [21], lack of patients with AIAIDs, PIDs, and SIDs in these studies does not allow definitive conclusions about the safety and efficacy of these vaccines in these patients. Moreover, recently the discovery of very rare, but severe and often lethal, cases of VITT prompted the regulatory agencies of many countries to substantially limit the use of these vaccines.
- Anti-COVID-19 mRNA vaccines, such as those developed by Pfizer/Biontech [28] and Moderna [29], are brand new and the first ones to be used on a large scale. During the pre-approval studies, after four months of observation, the groups treated with the two vaccines showed an efficacy of 95% and 94.1%, respectively, compared to the group of subjects who had received placebo, and the safety was considered optimal (Table 4) [28,29]. The mRNA vaccine effectiveness has also been calculated at 91% and 81% after the complete vaccine cycle or only the first dose, respectively [126]. Although in the pre-approval studies details on the possible presence of patients with AIAIDs in the study population have not been provided, recent studies have reported substantial safety and immunogenicity of these vaccines in patients with AIAIDs [48,49,50,92,106,107,108]. Even in PIDs, two preliminary studies [98,99] have shown the substantial safety and immunogenicity of the mRNA vaccines. Finally, COVID-19 vaccines were safe and immunogenic in onco-hematologic pathologies, and mRNA vaccines were more immunogenic than the adenoviral vaccine [101]. Despite the absence of currently released definitive recommendations, the cited preliminary studies confirm that mRNA vaccines seem to be well tolerated in patients with AIAIDs, PIDs, and SIDs. In the general population as well, mRNA vaccines show a very low frequency of serious adverse events, lower than those observed in vectored vaccines. The Task Force believes that, based on the data reported above, mRNA vaccines should be chosen for use in clinically stabilized patients with AIAIDs, PIDs, and SIDs.
- There is no general agreement regarding priority criteria for anti-COVID vaccine access for patients with AIAIDs, PIDs, and SIDs, because there is no consensus on their actual risk of infection or developing a more serious disease. The best criterion to evaluate the priority level of access to COVID-19 vaccine in these patients is the clinical one [124]: in patients with AIAIDs under heavy immunosuppressive therapy and a history of serious and recurrent infections, the immunology specialist should determine the priority level, in keeping with the Raccomandazioni ad interim sui gruppi target della vaccinazione anti-SARS-CoV-2/COVID-19 (GURI 24.3.2021), elaborated by the Italian Ministry of Health. Clinical criteria should guide the management of patients with PIDs and SIDs as well. For these patients, vaccination of cohabiting relatives and healthcare providers is recommended.
- The Task Force does not consider it appropriate to interrupt/discontinue immunosuppressive therapy, excepting for high dosage CCS (≥10 mg of prednisone-equivalent [104]) and anti-CD20 mAbs, such as rituximab [103,104,105] (Table 6). Moreover, in the case of MTX [105,106], abatacept [105,106], JKI [105,106], and mycophenolate [106], the immunology specialist will evaluate the possible discontinuation based on the patients’ clinical characteristics. Regarding PIDs, the risk of non-response should be evaluated, especially in severe combined and humoral immunodeficiencies. In the case of non-response passive immunotherapy with mAbs or convalescent plasma, vaccination should carefully be considered.
- The patient’s immunology specialist should be continuously updated and available for evaluating the vaccination risk level (disease activity and immunosuppression consequent to the current therapy) of the patient. The specialist should be continuously informed and able to provide all information useful for handling treatment during the vaccination period.
- In keeping with the activity of the Italian Drug Agency (AIFA) to inform the whole population, the three Italian Immunological Societies will make available all data coming from studies carried out on immunized populations as soon as possible, using the sites or the traditional communication channels available to the members of scientific societies and the patient associations.
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Salemi, S.; D’Amelio, R. Are Anti-Infectious Vaccinations Safe and Effective in Patients with Autoimmunity? Int. Rev. Immunol. 2010, 29, 270–314. [Google Scholar] [CrossRef] [PubMed]
- Kochar, B.; Herfarth, H.H. Vaccinations in Adult Patients with Inflammatory Bowel Diseases in the West. Inflamm. Intest. Dis. 2018, 3, 11–15. [Google Scholar] [CrossRef]
- Zrzavy, T.; Kollaritsch, H.; Rommer, P.S.; Boxberger, N.; Loebermann, M.; Wimmer, I.; Winkelmann, A.; Zettl, U.K. Vaccination in Multiple Sclerosis: Friend or Foe? Front. Immunol. 2019, 10, 1883. [Google Scholar] [CrossRef] [PubMed]
- Furer, V.; Rondaan, C.; Heijstek, M.W.; Agmon-Levin, N.; Van Assen, S.; Bijl, M.; Breedveld, F.C.; D’Amelio, R.; Dougados, M.; Kapetanovic, M.C.; et al. 2019 update of EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum. Dis. 2020, 79, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Rondaan, C.; Furer, V.; Heijstek, M.W.; Agmon-Levin, N.; Bijl, M.; Breedveld, F.C.; D’Amelio, R.; Dougados, M.; Kapetanovic, M.C.; Van Laar, J.M.; et al. Efficacy, immunogenicity and safety of vaccination in adult patients with autoimmune inflammatory rheumatic diseases: A systematic literature review for the 2019 update of EULAR recommendations. RMD Open 2019, 5, e001035. [Google Scholar] [CrossRef] [Green Version]
- Martire, B.; Azzari, C.; Badolato, R.; Canessa, C.; Cirillo, E.; Gallo, V.; Graziani, S.; Lorenzini, T.; Milito, C.; Panza, R.; et al. Vaccination in immunocompromised host: Recommendations of Italian Primary Immunodeficiency Network Centers (IPINET). Vaccine 2018, 36, 3541–3554. [Google Scholar] [CrossRef]
- Eibl, M.M.; Wolf, H.M. Vaccination in patients with primary immune deficiency, secondary immune deficiency and autoimmunity with immune regulatory abnormalities. Immunotherapy 2015, 7, 1273–1292. [Google Scholar] [CrossRef] [Green Version]
- Rubin, L.G.; Levin, M.J.; Ljungman, P.; Davies, E.G.; Avery, R.K.; Tomblyn, M.; Bousvaros, A.; Dhanireddy, S.; Sung, L.; Keyserling, H.; et al. Executive Summary: 2013 IDSA Clinical Practice Guideline for Vaccination of the Immunocompromised Host. Clin. Infect. Dis. 2014, 58, 309–318. [Google Scholar] [CrossRef]
- Bitterman, R.; Eliakim-Raz, N.; Vinograd, I.; Trestioreanu, A.Z.; Leibovici, L.; Paul, M. Influenza vaccines in immunosuppressed adults with cancer. Cochrane Database Syst. Rev. 2018, 2, CD008983. [Google Scholar] [CrossRef]
- Germano, V.; Cattaruzza, M.S.; Osborn, J.; Tarantino, A.; Di Rosa, R.; Salemi, S.; D’Amelio, R. Infection risk in Rheumatoid Arthritis and Spondyloarthropathy patients under treatment with DMARDs, Corticosteroids and TNF-α antagonists. J. Transl. Med. 2014, 12, 77. [Google Scholar] [CrossRef] [Green Version]
- Consiglio Superiore di Sanità. Guida Alle Controindicazioni Alle Vaccinazioni, 5th ed.; A Cura di Giovanni Gallo, Rosanna Mel, Elisa Ros e Antonietta Filia; Consiglio Superiore di Sanità: Rome, Italy, 2018. [Google Scholar]
- Caporuscio, S.; Ieraci, R.; Valesini, G.; Teloni, R.; Mariotti, S.; Spinelli, F.R.; Ferlito, C.; Salemi, S.; Diamanti, A.P.; Meneguzzi, G.; et al. Anti-polysaccharide and anti-diphtheria protective antibodies after 13-valent pneumococcal conjugate vaccination in rheumatoid arthritis patients under immunosuppressive therapy. Clin. Immunol. 2018, 195, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Arad, U.; Tzadok, S.; Amir, S.; Mandelboim, M.; Mendelson, E.; Wigler, I.; Sarbagil-Maman, H.; Paran, D.; Caspi, D.; Elkayam, O. The cellular immune response to influenza vaccination is preserved in rheumatoid arthritis patients treated with rituximab. Vaccine 2011, 29, 1643–1648. [Google Scholar] [CrossRef] [PubMed]
- Croce, E.; Hatz, C.; Jonker, E.; Visser, L.; Jaeger, V.; Bühler, S. Safety of live vaccinations on immunosuppressive therapy in patients with immune-mediated inflammatory diseases, solid organ transplantation or after bone-marrow transplantation—A systematic review of randomized trials, observational studies and case reports. Vaccine 2017, 35, 1216–1226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centers for Disease Control and Prevention; Infectious Disease Society of America; American Society of Blood and Marrow Transplantation. Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. MMWR Recomm. Rep. 2000, 49, RR-10. [Google Scholar]
- Hodgson, J. The pandemic pipeline. Nat. Biotechnol. 2020, 38, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 12 June 2021).
- Lancet Commission on COVID-19 Vaccines and Therapeutics Task Force Members. Operation Warp Speed: Implications for global vaccine security. Lancet Glob. Health 2021, 9, e1017–e1021. [Google Scholar] [CrossRef]
- Keech, C.; Albert, G.; Cho, I.; Robertson, A.; Reed, P.; Neal, S.; Plested, J.S.; Zhu, M.; Cloney-Clark, S.; Zhou, H.; et al. Phase 1–2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. N. Engl. J. Med. 2020, 383, 2320–2332. [Google Scholar] [CrossRef]
- Heath, P.T.; Galiza, E.P.; Baxter, D.N.; Boffito, M.; Browne, D.; Burns, F.; Chadwick, D.R.; Clark, R.; Cosgrove, C.; Galloway, J.; et al. Safety and Efficacy of NVX-CoV2373 Covid-19 Vaccine. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef]
- Ramasamy, M.N.; Minassian, A.M.; Ewer, K.J.; Flaxman, A.L.; Folegatti, P.M.; Owens, D.R.; Voysey, M.; Aley, P.K.; Angus, B.; Babbage, G.; et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): A single-blind, randomised, controlled, phase 2/3 trial. Lancet 2021, 396, 1979–1993. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Stephenson, K.E.; Le Gars, M.; Sadoff, J.; de Groot, A.M.; Heerwegh, D.; Truyers, C.; Atyeo, C.; Loos, C.; Chandrashekar, A.; McMahan, K.; et al. Immunogenicity of the Ad26.COV2.S Vaccine for COVID-19. JAMA 2021, 325, 1535. [Google Scholar] [CrossRef]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B.; et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.-C.; Li, Y.-H.; Guan, X.-H.; Hou, L.-H.; Wang, W.-J.; Li, J.-X.; Wu, S.-P.; Wang, B.-S.; Wang, Z.; Wang, L.; et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: A dose-escalation, open-label, non-randomised, first-in-human trial. Lancet 2020, 395, 1845–1854. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatullin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Shcheblyakov, D.V.; Tukhvatulin, A.I.; Zubkova, O.V.; Dzharullaeva, A.S.; Kovyrshina, A.V.; Lubenets, N.L.; Grousova, D.M.; Erokhova, A.S.; et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: An interim analysis of a randomised controlled phase 3 trial in Russia. Lancet 2021, 397, 671–681. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Montgomery, J.; Ryan, M.; Engler, R.; Hoffman, D.; McClenathan, B.; Collins, L.; Loran, D.; Hrncir, D.; Herring, K.; Platzer, M.; et al. Myocarditis Following Immunization with mRNA COVID-19 Vaccines in Members of the US Military. JAMA Cardiol. 2021. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, G.; Pan, H.; Li, C.; Hu, Y.; Chu, K.; Han, W.; Chen, Z.; Tang, R.; Yin, W.; et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 181–192. [Google Scholar] [CrossRef]
- Han, B.; Song, Y.; Li, C.; Yang, W.; Ma, Q.; Jiang, Z.; Li, M.; Lian, X.; Jiao, W.; Wang, L.; et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy children and adolescents: A double-blind, randomised, controlled, phase 1/2 clinical trial. Lancet. Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Tanriover, M.D.; Doğanay, H.L.; Akova, M.; Güner, H.R.; Azap, A.; Akhan, S.; Köse, Ş.; Erdinç, F.Ş.; Akalın, E.H.; Tabak, Ö.F.; et al. Efficacy and safety of an inactivated whole-virion SARS-CoV-2 vaccine (CoronaVac): Interim results of a double-blind, randomised, placebo-controlled, phase 3 trial in Turkey. Lancet 2021, 398, 213–222. [Google Scholar] [CrossRef]
- Scully, M.; Singh, D.; Lown, R.; Poles, A.; Solomon, T.; Levi, M.; Goldblatt, D.; Kotoucek, P.; Thomas, W.; Lester, W. Pathologic Antibodies to Platelet Factor 4 after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. 2021, 384, 2202–2211. [Google Scholar] [CrossRef]
- Aleem, A.; Nadeem, A.J. Coronavirus (COVID-19) Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT) [Updated 18 July 2021]. In StatPearlas [Internet]; StatPearls Publishing: Treasure Island, FL, USA. Available online: https://WWW.ncbi.nlm.nih.gov/books/NBK570605 (accessed on 18 July 2021).
- See, I.; Su, J.R.; Lale, A.; Woo, E.J.; Guh, A.Y.; Shimabukuro, T.T.; Streiff, M.B.; Rao, A.K.; Wheeler, A.P.; Beavers, S.F.; et al. US Case Reports of Cerebral Venous Sinus Thrombosis with Thrombocytopenia After Ad26.COV2.S Vaccination, March 2 to April 21, 2021. JAMA 2021, 325, 2448. [Google Scholar] [CrossRef] [PubMed]
- Kardani, K.; Bolhassani, A.; Shahbazi, S. Prime-boost vaccine strategy against viral infections: Mechanisms and benefits. Vaccine 2016, 34, 413–423. [Google Scholar] [CrossRef]
- Ferlito, C.; Biselli, R.; Visco, V.; Cattaruzza, M.; Capobianchi, M.; Castilletti, C.; Lapa, D.; Nicoletti, L.; Marchi, A.; Magurano, F.; et al. Immunogenicity of Viral Vaccines in the Italian Military. Biomedicines 2021, 9, 87. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Mao, Q.; An, C.; Zhang, J.; Gao, F.; Bian, L.; Li, C.; Liang, Z.; Xu, M.; Wang, J. Heterologous prime-boost: Breaking the protective immune response bottleneck of COVID-19 vaccine candidates. Emerg. Microbes Infect. 2021, 10, 629–637. [Google Scholar] [CrossRef]
- Spencer, A.J.; McKay, P.F.; Belij-Rammerstorfer, S.; Ulaszewska, M.; Bissett, C.D.; Hu, K.; Samnuan, K.; Blakney, A.K.; Wright, D.; Sharpe, H.R.; et al. Heterologous vaccination regimens with self-amplifying RNA and adenoviral COVID vaccines induce robust immune responses in mice. Nat. Commun. 2021, 12, 2893. [Google Scholar] [CrossRef]
- Callaway, E. Mix-and-match COVID vaccines trigger potent immune response. Nature 2021, 593, 491. [Google Scholar] [CrossRef] [PubMed]
- Normark, J.; Vikström, L.; Gwon, Y.-D.; Persson, I.-L.; Edin, A.; Björsell, T.; Dernstedt, A.; Christ, W.; Tevell, S.; Evander, M.; et al. Heterologous ChAdOx1 nCoV-19 and mRNA-1273 Vaccination. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Shaw, R.H.; Stuart, A.; Greenland, M.; Liu, X.; Van-Tam, J.S.N.; Snape, M.D.; Com-COV Study Group. Heterologous prime-boost COVID-19 vaccination: Initial reactogenicity data. Lancet 2021, 397, 2043–2046. [Google Scholar] [CrossRef]
- Hillus, D.; Schwarz, T.; Tober-Lau, P.; Vanshylla, K.; Hastor, H.; Thibeault, C.; Jentzsch, S.; Helbig, E.T.; Lippert, L.J.; Tscheak, P.; et al. Safety, reactogenicity, and immunogenicity of homologous and heterologous prime-boost immunisation with ChAdOx1 nCoV-19 and BNT162b2: A prospective cohort study. Lancet Respir. Med. 2021. [Google Scholar] [CrossRef]
- Groß, R.; Zanoni, M.; Seidel, A.; Conzelmann, C.; Gilg, A.; Krnavek, D.; Erdemci-Evin, S.; Mayer, B.; Hoffmann, M.; Pöh-lmann, S.; et al. Heterologous ChAdOx1 nCoV-19 and BNT162b2 prime-boost vaccination elicits potent neutralizing anti-body responses and T cell reactivity. medRxiv 2021. [Google Scholar] [CrossRef]
- Tregoning, J.S.; Brown, E.S.; Cheeseman, H.M.; Flight, K.E.; Higham, S.L.; Lemm, N.; Pierce, B.F.; Stirling, D.C.; Wang, Z.; Pollock, K.M. Vaccines for COVID-19. Clin. Exp. Immunol. 2020, 202, 162–192. [Google Scholar] [CrossRef]
- Castells, M.C.; Phillips, E.J. Maintaining Safety with SARS-CoV-2 Vaccines. N. Engl. J. Med. 2021, 384, 643–649. [Google Scholar] [CrossRef]
- Connolly, C.M.; Ruddy, J.A.; Boyarsky, B.J.; Avery, R.K.; Werbel, W.A.; Segev, D.L.; Garonzik-Wang, J.; Paik, J.J. Safety of the first dose of mRNA SARS-CoV-2 vaccines in patients with rheumatic and musculoskeletal diseases. Ann. Rheum. Dis. 2021, 80, 1100–1101. [Google Scholar] [CrossRef]
- Geisen, U.M.; Berner, D.K.; Tran, F.; Sümbül, M.; Vullriede, L.; Ciripoi, M.; Reid, H.M.; Schaffarzyk, A.; Longardt, A.C.; Franzenburg, J.; et al. Immunogenicity and safety of anti-SARS-CoV-2 mRNA vaccines in patients with chronic inflammatory conditions and immunosuppressive therapy in a monocentric cohort. Ann. Rheum. Dis. 2021, annrheumdis-2021-220272. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Tascilar, K.; Fagni, F.; Krönke, G.; Kleyer, A.; Meder, C.; Atreya, R.; Leppkes, M.; Kremer, A.E.; Ramming, A.; et al. SARS-CoV-2 vaccination responses in untreated, conventionally treated and anticytokine-treated patients with immune-mediated inflammatory diseases. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Cines, D.B.; Gernsheimer, T.; Kessler, C.; Michel, M.; Tarantino, M.D.; Semple, J.W.; Arnold, D.M.; Godeau, B.; Lambert, M.P.; et al. Thrombocytopenia following Pfizer and Moderna SARS-CoV-2 vaccination. Am. J. Hematol. 2021, 96, 534–537. [Google Scholar] [CrossRef]
- Albert, E.; Aurigemma, G.; Saucedo, J.; Gerson, D.S. Myocarditis following COVID-19 vaccination. Radiol. Case Rep. 2021, 16, 2142–2145. [Google Scholar] [CrossRef]
- Ella, R.; Reddy, S.; Jogdand, H.; Sarangi, V.; Ganneru, B.; Prasad, S.; Das, D.; Raju, D.; Praturi, U.; Sapkal, G.; et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: Interim results from a double-blind, randomised, multicentre, phase 2 trial, and 3-month follow-up of a double-blind, randomised phase 1 trial. Lancet Infect. Dis. 2021, 21, 950–961. [Google Scholar] [CrossRef]
- Yadav, P.D.; Ella, R.; Kumar, S.; Patil, D.R.; Mohandas, S.; Shete, A.M.; Vadrevu, K.M.; Bhati, G.; Sapkal, G.; Kaushal, H.; et al. Immunogenicity and protective efficacy of inactivated SARS-CoV-2 vaccine candidate, BBV152 in rhesus macaques. Nat. Commun. 2021, 12, 1386. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.G.; Abbott, S.; Barnard, R.C.; Jarvis, C.I.; Kucharski, A.J.; Munday, J.D.; Pearson, C.A.B.; Russell, T.W.; Tully, D.C.; Washburne, A.D.; et al. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science 2021, 372, eabg3055. [Google Scholar] [CrossRef]
- Madhi, S.A.; Baillie, V.; Cutland, C.L.; Voysey, M.; Koen, A.L.; Fairlie, L.; Padayachee, S.D.; Dheda, K.; Barnabas, S.L.; Bhorat, Q.E.; et al. Efficacy of the ChAdOx1 nCoV-19 Covid-19 Vaccine against the B.1.351 Variant. N. Engl. J. Med. 2021, 384, 1885–1898. [Google Scholar] [CrossRef]
- Faria, N.R.; Mellan, T.A.; Whittaker, C.; Claro, I.M.; Candido, D.D.S.; Mishra, S.; Crispim, M.A.E.; Sales, F.C.S.; Hawryluk, I.; McCrone, J.T.; et al. Genomics and epidemiology of the P.1 SARS-CoV-2 lineage in Manaus, Brazil. Science 2021, 372, 815–821. [Google Scholar] [CrossRef]
- Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/988619/Variants_of_Concern_VOC_Technical_Briefing_12_England.pdf (accessed on 3 July 2021).
- Collier, D.A.; De Marco, A.; Ferreira, I.A.T.M.; Meng, B.; Datir, R.P.; Walls, A.C.; Kemp, S.A.; Bassi, J.; Pinto, D.; Fregni, C.S.; et al. Sensitivity of SARS-CoV-2 B.1.1.7 to mRNA vaccine-elicited antibodies. Nature 2021, 593, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.G.; Jarvis, C.I.; CMMID COVID-19 Working Group; Edmunds, W.J.; Jewell, N.P.; Diaz-Ordaz, K.; Keogh, R.H. Increased mortality in community-tested cases of SARS-CoV-2 lineage B.1.1.7. Nature 2021, 593, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://cmmid.github.io/topics/covid19/reports/sa-novel-variant/ 2021_01_11_Transmissibil-ity_and_severity_of_501Y_V2_in_SA.pdf (accessed on 3 July 2021).
- Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/975742/Variants_of_Concern_VOC_Technical_Briefing_8_England.pdf (accessed on 3 July 2021).
- Sheikh, A.; McMenamin, J.; Taylor, B.; Robertson, C.; Public Health Scotland and the EAVE II Collaborators. SARS-CoV-2 Delta VOC in Scotland: Demographics, risk of hospital admission, and vaccine effectiveness. Lancet 2021, 397, 2461–2462. [Google Scholar] [CrossRef]
- ECDC-European Center for Disease Control. SARS-CoV-2 Variants of Concern as of 6 May 2021. Available online: https://www.ecdc.europa.eu/en/covid-19/variants-concern (accessed on 3 July 2021).
- Liu, Y.; Liu, J.; Xia, H.; Zhang, X.; Fontes-Garfias, C.R.; Swanson, K.A.; Cai, H.; Sarkar, R.; Chen, W.; Cutler, M.; et al. Neutralizing Activity of BNT162b2-Elicited Serum. N. Engl. J. Med. 2021, 384, 1466–1468. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Werner, A.P.; Koch, M.; Choi, A.; Narayanan, E.; Stewart-Jones, G.B.; Colpitts, T.; Bennett, H.; Boyoglu-Barnum, S.; Shi, W.; et al. Serum Neutralizing Activity Elicited by mRNA-1273 Vaccine. N. Engl. J. Med. 2021, 384, 1468–1470. [Google Scholar] [CrossRef] [PubMed]
- Woldemeskel, B.A.; Garliss, C.C.; Blankson, J.N. SARS-CoV-2 mRNA vaccines induce broad CD4+ T cell responses that recognize SARS-CoV-2 variants and HCoV-NL63. J. Clin. Investig. 2021, 131, 2149335. [Google Scholar] [CrossRef] [PubMed]
- Rubin, R. COVID-19 Vaccines vs Variants—Determining How Much Immunity Is Enough. JAMA 2021, 325, 1241. [Google Scholar] [CrossRef]
- Shinde, V.; Bhikha, S.; Hoosain, Z.; Archary, M.; Bhorat, Q.; Fairlie, L.; Lalloo, U.; Masilela, M.S.; Moodley, D.; Hanley, S.; et al. Efficacy of NVX-CoV2373 Covid-19 Vaccine against the B.1.351 Variant. N. Engl. J. Med. 2021, 384, 1899–1909. [Google Scholar] [CrossRef] [PubMed]
- Abu-Raddad, L.J.; Chemaitelly, H.; Butt, A.A.; National Study Group for COVID-19 Vaccination. Effectiveness of the BNT162b2 Covid-19 Vaccine against the B.1.1.7 and B.1.351 Variants. N. Engl. J. Med. 2021, 385, 187–189. [Google Scholar] [CrossRef]
- Dejnirattisai, W.; Zhou, D.; Supasa, P.; Liu, C.; Mentzer, A.J.; Ginn, H.M.; Zhao, Y.; Duyvesteyn, H.M.; Tuekprakhon, A.; Nutalai, R.; et al. Antibody evasion by the P.1 strain of SARS-CoV-2. Cell 2021, 184, 2939–2954.e9. [Google Scholar] [CrossRef]
- Bernal, J.L.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar] [CrossRef]
- Boyarsky, B.J.; Werbel, W.A.; Avery, R.K.; Tobian, A.A.R.; Massie, A.B.; Segev, D.L.; Garonzik-Wang, J.M. Antibody Response to 2-Dose SARS-CoV-2 mRNA Vaccine Series in Solid Organ Transplant Recipients. JAMA 2021, 325, 2204–2206. [Google Scholar] [CrossRef] [PubMed]
- Sattler, A.; Schrezenmeier, E.; Weber, U.A.; Potekhin, A.; Bachmann, F.; Straub-Hohenbleicher, H.; Budde, K.; Storz, E.; Proß, V.; Bergmann, Y.; et al. Impaired humoral and cellular immunity after SARS-CoV-2 BNT162b2 (tozinameran) prime-boost vaccination in kidney transplant recipients. J. Clin. Investig. 2021, 131, e150175. [Google Scholar] [CrossRef]
- Kamar, N.; Abravanel, F.; Marion, O.; Couat, C.; Izopet, J.; Del Bello, A. Three Doses of an mRNA Covid-19 Vaccine in Solid-Organ Transplant Recipients. N. Engl. J. Med. 2021, 385, 661–662. [Google Scholar] [CrossRef]
- Albach, F.N.; Burmester, G.R.; Biesen, R. Successful BNT162b2 booster vaccinations in a patient with rheumatoid arthritis and initially negative antibody response. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef]
- Stamatatos, L.; Czartoski, J.; Wan, Y.H.; Homad, L.J.; Rubin, V.; Glantz, H.; Neradilek, M.; Seydoux, E.; Jennewein, M.F.; MacCamy, A.J.; et al. mRNA vaccination boosts cross-variant neutralizing antibodies elicited by SARS-CoV-2 infection. Science 2021, 25, eabg9175. [Google Scholar] [CrossRef]
- Wang, Z.; Muecksch, F.; Schaefer-Babajew, D.; Finkin, S.; Viant, C.; Gaebler, C.; Hoffmann, H.H.; Barnes, C.O.; Cipolla, M.; Ramos, V.; et al. Naturally enhanced neutralizing breadth against SARS-CoV-2 one year after infection. Nature 2021, 595, 426–431. [Google Scholar] [CrossRef]
- Mazzoni, A.; Di Lauria, N.; Maggi, L.; Salvati, L.; Vanni, A.; Capone, M.; Lamacchia, G.; Mantengoli, E.; Spinicci, M.; Zammarchi, L.; et al. First-dose mRNA vaccination is sufficient to reactivate immunological memory to SARS-CoV-2 in subjects who have recovered from COVID-19. J. Clin. Investig. 2021, 131, e149150. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Peng, H.; Zhu, Z.; Li, G.; Huang, Z.; Zhao, Z.; Koup, R.A.; Bailer, R.T.; Wu, C. Persistent memory CD4+ and CD8+ T-cell responses in recovered severe acute respiratory syndrome (SARS) patients to SARS coronavirus M antigen. J. Gen. Virol. 2007, 88 Pt 10, 2740–2748. [Google Scholar] [CrossRef]
- Ng, O.-W.; Chia, A.; Tan, A.T.; Jadi, R.S.; Leong, H.N.; Bertoletti, A.; Tan, Y.-J. Memory T cell responses targeting the SARS coronavirus persist up to 11 years post-infection. Vaccine 2016, 34, 2008–2014. [Google Scholar] [CrossRef]
- Dispinseri, S.; Secchi, M.; Pirillo, M.F.; Tolazzi, M.; Borghi, M.; Brigatti, C.; De Angelis, M.L.; Baratella, M.; Bazzigaluppi, E.; Venturi, G.; et al. Neutralizing antibody responses to SARS-CoV-2 in symptomatic COVID-19 is persistent and critical for survival. Nat. Commun. 2021, 12, 2670. [Google Scholar] [CrossRef]
- Corbett, K.S.; Flynn, B.; Foulds, K.E.; Francica, J.R.; Boyoglu-Barnum, S.; Werner, A.P.; Flach, B.; O’Connell, S.; Bock, K.W.; Minai, M.; et al. Evaluation of the mRNA-1273 Vaccine against SARS-CoV-2 in Nonhuman Primates. N. Engl. J. Med. 2020, 383, 1544–1555. [Google Scholar] [CrossRef] [PubMed]
- Van Doremalen, N.; Lambe, T.; Spencer, A.; Belij-Rammerstorfer, S.; Purushotham, J.N.; Port, J.R.; Avanzato, V.A.; Bushmaker, T.; Flaxman, A.; Ulaszewska, M.; et al. ChAdOx1 nCoV-19 vaccine prevents SARS-CoV-2 pneumonia in rhesus macaques. Nature 2020, 586, 578–582. [Google Scholar] [CrossRef]
- Guebre-Xabier, M.; Patel, N.; Tian, J.-H.; Zhou, B.; Maciejewski, S.; Lam, K.; Portnoff, A.D.; Massare, M.J.; Frieman, M.B.; Piedra, P.A.; et al. NVX-CoV2373 vaccine protects cynomolgus macaque upper and lower airways against SARS-CoV-2 challenge. Vaccine 2020, 38, 7892–7896. [Google Scholar] [CrossRef] [PubMed]
- Ketas, T.J.; Chaturbhuj, D.; Portillo, V.M.C.; Francomano, E.; Golden, E.; Chandrasekhar, S.; Debnath, G.; Diaz-Tapia, R.; Yasmeen, A.; Kramer, K.D.; et al. Antibody Responses to SARS-CoV-2 mRNA Vaccines Are Detectable in Saliva. Pathog. Immun. 2021, 6, 116–134. [Google Scholar] [CrossRef] [PubMed]
- Prévost, J.; Gasser, R.; Beaudoin-Bussières, G.; Richard, J.; Duerr, R.; Laumaea, A.; Anand, S.P.; Goyette, G.; Benlarbi, M.; Ding, S.; et al. Cross-Sectional Evaluation of Humoral Responses against SARS-CoV-2 Spike. Cell Rep. Med. 2020, 1, 100126. [Google Scholar] [CrossRef]
- Shimabukuro, T.T.; Kim, S.Y.; Myers, T.R.; Moro, P.L.; Oduyebo, T.; Panagiotakopoulos, L.; Marquez, P.L.; Olson, C.K.; Liu, R.; Chang, K.T.; et al. Preliminary Findings of mRNA Covid-19 Vaccine Safety in Pregnant Persons. N. Engl. J. Med. 2021, 384, 2273–2282. [Google Scholar] [CrossRef] [PubMed]
- Collier, A.-R.Y.; McMahan, K.; Yu, J.; Tostanoski, L.H.; Aguayo, R.; Ansel, J.; Chandrashekar, A.; Patel, S.; Bondzie, E.A.; Sellers, D.; et al. Immunogenicity of COVID-19 mRNA Vaccines in Pregnant and Lactating Women. JAMA 2021, 325, 2370–2380. [Google Scholar] [CrossRef]
- Perl, S.H.; Uzan-Yulzari, A.; Klainer, H.; Asiskovich, L.; Youngster, M.; Rinott, E.; Youngster, I. SARS-CoV-2–Specific Antibodies in Breast Milk After COVID-19 Vaccination of Breastfeeding Women. JAMA 2021, 325, 2013–2014. [Google Scholar] [CrossRef]
- Wang, Z.; Lorenzi, J.C.C.; Muecksch, F.; Finkin, S.; Viant, C.; Gaebler, C.; Cipolla, M.; Hoffmann, H.-H.; Oliveira, T.Y.; Oren, D.A.; et al. Enhanced SARS-CoV-2 neutralization by dimeric IgA. Sci. Transl. Med. 2020, 13, eabf1555. [Google Scholar] [CrossRef]
- Frenck, R.W.; Klein, N.P.; Kitchin, N.; Gurtman, A.; Absalon, J.; Lockhart, S.; Perez, J.L.; Walter, E.B.; Senders, S.; Bailey, R.; et al. Safety, Immunogenicity, and Efficacy of the BNT162b2 Covid-19 Vaccine in Adolescents. N. Engl. J. Med. 2021, 385, 239–250. [Google Scholar] [CrossRef]
- Furer, V.; Eviatar, T.; Zisman, D.; Peleg, H.; Paran, D.; Levartovsky, D.; Zisapel, M.; Elalouf, O.; Kaufman, I.; Meidan, R.; et al. Immunogenicity and safety of the BNT162b2 mRNA COVID-19 vaccine in adult patients with autoimmune inflammatory rheumatic diseases and in the general population: A multicentre study. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef]
- Furer, V.; Zisman, D.; Kibari, A.; Rimar, D.; Paran, Y.; Elkayam, O. Herpes zoster following BNT162b2 mRNA COVID-19 vaccination in patients with autoimmune inflammatory rheumatic diseases: A case series. Rheumatology 2021, keab345. [Google Scholar] [CrossRef]
- Warny, M.; Helby, J.; Nordestgaard, B.G.; Birgens, H.; Bojesen, S.E. Lymphopenia and risk of infection and infection-related death in 98,344 individuals from a prospective Danish population-based study. PLoS Med. 2018, 15, e1002685. [Google Scholar] [CrossRef] [Green Version]
- Martin, M.; Guffroy, A.; Argemi, X.; Martin, T. Lupus érythémateux systémique et lymphopénie: Aspects cliniques et physiopathologiques. [Systemic lupus erythematosus and lymphopenia: Clinical and pathophysiological features]. Rev. Med. Interne 2017, 38, 603–613. [Google Scholar] [CrossRef]
- Carli, L.; Tani, C.; Vagnani, S.; Signorini, V.; Mosca, M. Leukopenia, lymphopenia, and neutropenia in systemic lupus erythematosus: Prevalence and clinical impact—A systematic literature review. Semin. Arthritis Rheum. 2015, 45, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Squire, J.; Joshi, A. Seroconversion Following COVID-19 Vaccination in Immune Deficient Patients. Ann. Allergy Asthma Immunol. 2021. [Google Scholar] [CrossRef]
- Hagin, D.; Freund, T.; Navon, M.; Halperin, T.; Adir, D.; Marom, R.; Levi, I.; Benor, S.; Alcalay, Y.; Freund, N.T. Immunogenicity of Pfizer-BioNTech COVID-19 vaccine in patients with inborn errors of immunity. J. Allergy Clin. Immunol. 2021. [Google Scholar] [CrossRef]
- Fernandez Salinas, A.; Piano Mortari, E.; Terreri, S.; Quintarelli, C.; Pulvirenti, F.; Di Cecca, S.; Guercio, M.; Milito, C.; Bonanni, L.; Auria, S.; et al. SARS-CoV-2 Vaccine Induced Atypical Immune Responses in Antibody Defects: Everybody does their best. medRxiv 2021. [Google Scholar] [CrossRef]
- Thakkar, A.; Gonzalez-Lugo, J.D.; Goradia, N.; Gali, R.; Shapiro, L.C.; Pradhan, K.; Rahman, S.; Kim, S.Y.; Ko, B.; Sica, R.A.; et al. Seroconversion rates following COVID-19 vaccination among patients with cancer. Cancer Cell 2021, 39, 1081–1090.e2. [Google Scholar] [CrossRef]
- Monin, L.; Laing, A.G.; Muñoz-Ruiz, M.; McKenzie, D.R.; Barrio, I.D.M.D.; Alaguthurai, T.; Domingo-Vila, C.; Hayday, T.S.; Graham, C.; Seow, J.; et al. Safety and immunogenicity of one versus two doses of the COVID-19 vaccine BNT162b2 for patients with cancer: Interim analysis of a prospective observational study. Lancet Oncol. 2021, 22, 765–778. [Google Scholar] [CrossRef]
- Zitt, E.; Davidovic, T.; Schimpf, J.; Abbassi-Nik, A.; Mutschlechner, B.; Ulmer, H.; Benda, M.A.; Sprenger-Mähr, H.; Winder, T.; Lhotta, K. The Safety and Immunogenicity of the mRNA-BNT162b2 SARS-CoV-2 Vaccine in Hemodialysis Patients. Front. Immunol. 2021, 12, 704773. [Google Scholar] [CrossRef]
- Bijlsma, J.W. EULAR December 2020 viewpoints on SARS-CoV-2 vaccination in patients with RMDs. Ann. Rheum. Dis. 2021, 80, 411–412. [Google Scholar] [CrossRef] [PubMed]
- American College of Rheumatology. COVID-19 Vaccine Clinical Guidance Summary for Patients with Rheumatic and Mus-culoskeletal Diseases. Developed by the ACR COVID-19 Vaccine Clinical Guidance Task Force. This draft summary was ap-proved by the ACR Board of Directors on February 8, 2021, and updated on March 4, 2021. A full manuscript is pending journal peer review. Arthritis Rheumatol. 2021, 73, 1093–1107. [Google Scholar]
- Curtis, J.R.; Johnson, S.R.; Anthony, D.D.; Arasaratnam, R.J.; Baden, L.R.; Bass, A.R.; Calabrese, C.; Gravallese, E.M.; Harpaz, R.; Kroger, A.; et al. American College of Rheumatology Guidance for COVID-19 Vaccination in Patients With Rheumatic and Musculoskeletal Diseases: Version 2. Arthritis Rheumatol. 2021, 73. [Google Scholar] [CrossRef]
- Spiera, R.; Jinich, S.; Jannat-Khah, D. Rituximab, but not other antirheumatic therapies, is associated with impaired serological response to SARS- CoV-2 vaccination in patients with rheumatic diseases. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef]
- Ruddy, J.A.; Connolly, C.M.; Boyarsky, B.J.; Werbel, W.A.; Christopher-Stine, L.; Garonzik-Wang, J.; Segev, D.L.; Paik, J.J. High antibody response to two-dose SARS-CoV-2 messenger RNA vaccination in patients with rheumatic and musculoskeletal diseases. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef]
- Haberman, R.H.; Herati, R.; Simon, D.; Samanovic, M.; Blank, R.B.; Tuen, M.; Koralov, S.B.; Atreya, R.; Tascilar, K.; Allen, J.R.; et al. Methotrexate hampers immunogenicity to BNT162b2 mRNA COVID-19 vaccine in immune-mediated inflammatory disease. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef]
- Bonelli, M.M.; Mrak, D.; Perkmann, T.; Haslacher, H.; Aletaha, D. SARS-CoV-2 vaccination in rituximab-treated patients: Evidence for impaired humoral but inducible cellular immune response. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef]
- Westhoff, T.H.; Seibert, F.S.; Anft, M.; Blazquez-Navarro, A.; Skrzypczyk, S.; Doevelaar, A.; Hölzer, B.; Paniskaki, K.; Dolff, S.; Wilde, B.; et al. Correspondence on ‘SARS-CoV-2 vaccination in rituximab-treated patients: Evidence for impaired humoral but inducible cellular immune response’. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Sarzi-Puttini, P.; Giorgi, V.; Sirotti, S.; Marotto, D.; Ardizzone, S.; Rizzardini, G.; Antinori, S.; Galli, M. COVID-19, cytokines and immunosuppression: What can we learn from severe acute respiratory syndrome? Clin. Exp. Rheumatol. 2020, 38, 337–342. [Google Scholar]
- Park, J.K.; Lee, E.B.; Shin, K.; Sung, Y.-K.; Kim, T.H.; Kwon, S.-R.; Lee, M.S.; Hong, S.-J.; Choi, B.Y.; Lee, S.-S.; et al. COVID-19 Vaccination in Patients with Autoimmune Inflammatory Rheumatic Diseases: Clinical Guidance of the Korean College of Rheumatology. J. Korean Med. Sci. 2021, 36, e95. [Google Scholar] [CrossRef]
- Deepak, P.; Kim, W.; Paley, M.A.; Yang, M.; Carvidi, A.B.; El-Qunni, A.A.; Haile, A.; Huang, K.; Kinnett, B.; Liebeskind, M.J.; et al. Glucocorticoids and B Cell Depleting Agents Substantially Impair Immunogenicity of mRNA Vaccines to SARS-CoV-2. medRxiv 2021. [Google Scholar] [CrossRef]
- Díez, J.-M.; Romero, C.; Gajardo, R. Currently available intravenous immunoglobulin contains antibodies reacting against severe acute respiratory syndrome coronavirus 2 antigens. Immunotherapy 2020, 12, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Pisani, G.; Cristiano, K.; Simeoni, M.; Martina, A.; Pati, I.; Carocci, A.; Gaggioli, A.; Marino, F.; Adriani, D.; Pupella, S.; et al. Detection of antibodies against SARS-CoV-2 both in plasma pools for fractionation and in commercial intravenous immunoglobulins produced from plasma collected in Italy during the pandemic. Blood Transfus. 2021. [Google Scholar] [CrossRef]
- Akiyama, S.; Hamdeh, S.; Micic, D.; Sakuraba, A. Prevalence and clinical outcomes of COVID-19 in patients with autoimmune diseases: A systematic review and meta-analysis. Ann. Rheum. Dis. 2020, 80, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Druyan, A.; Lidar, M.; Brodavka, M.; Levy, I.; Barzilai, A.; Pavlotsky, F. The risk for severe COVID 19 in patients with autoimmune and/or inflammatory diseases: First wave lessons. Dermatol. Ther. 2021, 34, e14627. [Google Scholar] [CrossRef]
- Quinti, I.; Mezzaroma, I.; Milito, C. Clinical management of patients with primary immunodeficiencies during the COVID-19 pandemic. Expert Rev. Clin. Immunol. 2021, 17, 163–168. [Google Scholar] [CrossRef]
- Quinti, I.; Lougaris, V.; Milito, C.; Cinetto, F.; Pecoraro, A.; Mezzaroma, I.; Mastroianni, C.M.; Turriziani, O.; Bondioni, M.P.; Filippini, M.; et al. A possible role for B cells in COVID-19? Lesson from patients with agammaglobulinemia. J. Allergy Clin. Immunol. 2020, 146, 211–213.e4. [Google Scholar] [CrossRef] [PubMed]
- Meyts, I.; Bucciol, G.; Quinti, I.; Neven, B.; Fischer, A.; Seoane, E.; Lopez-Granados, E.; Gianelli, C.; Robles-Marhuenda, A.; Jeandel, P.-Y.; et al. Coronavirus disease 2019 in patients with inborn errors of immunity: An international study. J. Allergy Clin. Immunol. 2020, 147, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Shields, A.M.; Burns, S.O.; Savic, S.; Richter, A.G.; Anantharachagan, A.; Arumugakani, G.; Baker, K.; Bahal, S.; Bermingham, W.; Bhole, M.; et al. COVID-19 in patients with primary and secondary immunodeficiency: The United Kingdom experience. J. Allergy Clin. Immunol. 2021, 147, 870–875.e1. [Google Scholar] [CrossRef]
- McCaughan, G.; Di Ciaccio, P.; Ananda-Rajah, M.; Gilroy, N.; MacIntyre, R.; Teh, B.; Weinkove, R.; Curnow, J.; Szer, J.; Enjeti, A.K.; et al. COVID -19 vaccination in haematology patients: An Australian and New Zealand consensus position statement. Intern. Med. J. 2021, 51, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, F.; Cannatelli, R.; Benucci, M.; Carmagnola, S.; Clementi, E.; Danelli, P.; Dilillo, D.; Fiorina, P.; Galli, M.; Gallieni, M.; et al. How to Manage COVID-19 Vaccination in Immune-Mediated Inflammatory Diseases: An Expert Opinion by IMIDs Study Group. Front. Immunol. 2021, 12, 656362. [Google Scholar] [CrossRef]
- Choi, B.; Choudhary, M.C.; Regan, J.; Sparks, J.A.; Padera, R.F.; Qiu, X.; Solomon, I.H.; Kuo, H.-H.; Boucau, J.; Bowman, K.; et al. Persistence and Evolution of SARS-CoV-2 in an Immunocompromised Host. N. Engl. J. Med. 2020, 383, 2291–2293. [Google Scholar] [CrossRef]
- Thompson, M.G.; Burgess, J.L.; Naleway, A.L.; Tyner, H.; Yoon, S.K.; Meece, J.; Olsho, L.E.; Caban-Martinez, A.J.; Fowlkes, A.L.; Lutrick, K.; et al. Prevention and Attenuation of Covid-19 with the BNT162b2 and mRNA-1273 Vaccines. N. Engl. J. Med. 2021, 385, 320–329. [Google Scholar] [CrossRef] [PubMed]
| AIAIDs | Corticosteroids | IVIg/scDMARDs | bDMARDs/tsDMARDs | Inactivated Vaccines | Live Vaccines |
|---|---|---|---|---|---|
| High doses: ≥20 mg/day prednisone-equivalent for 1–2 weeks; non immunosuppressive doses: 7.5 mg/day | Sulphasalazine, hydroxichloroquine, azathioprine, mycophenolate, methotrexate, leflunomide, cyclosporine, tacrolimus, cyclophosphamide | infliximab, adalimumab, etanercept, golimumab, certolizumab, rituximab, tocilizumab, abatacept, anakinra, canakinumab, belimumab, secukinumab, ixekizumab, ustekinumab, tofacitinib, baricitinib | Hepatitis A/B, human papillomavirus, influenza, herpes zoster, inactivated poliovirus, pneumococcus, tetanus/diphtheria/pertussis, polysaccharide typhoid fever, Hib, meningococcus | Measles/Mumps/Rubella, Varicella, BCG, Ty21A, Yellow fever | |
| Rheumatoid arthritis, Systemic lupus, erythematosus Sjögren Syndrome, Anti-phospholipid syndrome, Systemic sclerosis, Polymyositis/ dermatomyositis, Vasculitis, Psoriatic arthritis, Spondyloarthritis, Familiar Mediterranean Fever, Periodical fever syndromes, Type 1 diabetes, Inflammatory bowel diseases, Multiple sclerosis | At high doses, they seem to interfere with the immune response to vaccines. Combination steroids/anti-TNF-α is particularly associated with the infection risk [10]. | IVIg should not be administered together with live vaccines due to the risk of vaccine inactivation [11], or with an inactivated vaccine because evaluating vaccine immunogenicity become simpossible. csDAMRDs are generally well tolerated at the doses generally used in IMIDs and they do not seem to interfere with the immune response. For methotrexate, a negative interference with the pneumococcal vaccine has been described, which has not been confirmed with the conjugate vaccine [12]. | When used alone, they are well tolerated and do not induce immunosuppression; immunosuppression is induced when they are used in combination. Abatacept and tofacitinib have been associated with a slightly reduced response to influenza and pneumococcus vaccines. Rituximab markedly reduces antibody response, but it does not seem to modify the adaptive cellular one [13]. Vaccination should be carried out before starting therapy with rituximab; in case of impossibility, it should be carried out 6 months after the last infusion of rituximab and 1 month before the next one. | Generally allowed, influenza and pneumococcus recommended, and, in particular subjects, hepatitis B, papillomavirus and herpes zoster. | Generally contraindicated, Measles/Mumps/Rubella seems well tolerated. Caution for Yellow fever, even though a recent review seems to partially reduce these fears [14]. |
| PIDs | Corticosteroids | IVIg/SCIg | HSCT/Gene therapy | Inactivated Vaccines | Live Vaccines |
|---|---|---|---|---|---|
| High doses: ≥20 mg/day prednisone-equivalent for 1–2 weeks; non immunosuppressive doses: 7.5 mg/day | Hepatitis A/B, human papillomavirus, influenza, herpes zoster, inactivated poliovirus, pneumococcus, tetanus/diphtheria/pertussis, polysaccharide typhoid fever, Hib, meningococcus, | Measles/Mumps/Rubella, Varicella, BCG, Ty21A, Yellow fever | |||
| Major antibody defects (XLA, CVID) Minor antibody defects (Defect of: IgA, IgG subclasses, specific antibodies) SCID—CID MSMD Invasive bacterial infections CMCD Defects of TLR Defects of IL12/IFN-ɣ pathway Defects of complement Congenital phagocyte defects Complete DiGeorge Syndrome Partial DiGeorge Syndrome Ataxia-Telangiectasia Wiskott-Aldrich Syndrome Hyper-IgE Syndrome IPEX Syndrome APECED Syndrome | At high doses, they seem to interfere with the immune response to vaccines. | IVIg should not be administered together with live vaccines due to the risk of vaccine inactivation [11], or with an inactivated vaccine because evaluating vaccine immunogenicity becomes impossible. | After 1 year following engraftment and lack of GVHD, it is possible to set the vaccination schedule with inactivated vaccines. Live vaccines should not be set before 2 years from transplant [15]. | Generally allowed in all the PIDs, excepting SCID and complete DiGeorge syndrome, in which only the polysaccharide vaccines (meningococcus, pneumococcus, Hib) are allowed. | Generally contraindicated, MMR and Varicella seem well tolerated in the minor antibody defects, complement defects, congenital phagocyte defects, partial DiGeorge syndrome, ataxia-telangiectasia, and hyper-IgE syndrome. Caution should be used for Yellow fever vaccine. |
| SIDs | Corticosteroids | IVIg/SCIg | Chemotherapy/Biologics/Janus Kinase Inhibitors | Inactivated Vaccines | Live Vaccines |
|---|---|---|---|---|---|
| High doses: ≥20 mg/day prednisone-equivalent for 1–2 weeks; non immunosuppressive doses: 7.5 mg/day | Hepatitis A/B, human papillomavirus, influenza, herpes zoster, inactivated poliovirus, pneumococcus, tetanus/diphtheria/pertussis, polysaccharide typhoid fever, Hib, meningococcus | Measles/Mumps/Rubella, Varicella, BCG, Ty21A, Yellow fever | |||
| Transplanted patients Hematological patients Oncological patients Patients with IMIDs on immunosuppression Dialysis patientsSevere asthma/COPD Splenectomized patients HIV-infected patients | At high doses they seem to interfere with the immune response to vaccines. | IVIg should not be administered together with live vaccines due to the risk of vaccine inactivation [11], or with an inactivated vaccine because evaluating vaccine immunogenicity becomes impossible. | In case of high-level immunosuppression by chemotherapy in onco-hematological pathologies, inactivated vaccines should preferably be administered either before or after, but not during, the treatment. | Generally allowed in all the reported SIDs. Polysaccharide vaccines (pneumococcus, meningococcus, Hib) are specifically recommended in the splenectomized patients. Polysaccharide and influenza vaccines are recommended in transplanted patients and in the other SIDs. Hepatitis A and B in liver transplanted patients. Hepatitis B in HIV-infected patients. Two months after the transplant it is possible to plan vaccinations with inactivated vaccines [7]. In Rituximab-treated patients, vaccination should be carried out at least 6 months after the last infusion. | Generally contraindicated, Measles/Mumps/Rubella and Varicella are allowed in HIV-infected patients, provided that they have CD4 ≥ 200/µL. Caution for Yellow fever, even though a recent review seems to partially reduce these fears [14]. |
| WHO Label | Lineage | Country of Isolation | Transmissibility | Disease Severity | Vaccine Protection |
|---|---|---|---|---|---|
| Alpha | B.1.1.7 | United Kingdom | Increased | Increased | Unmodified |
| Beta | B.1.351 | South Africa | Increased | Increased | Reduced |
| Gamma | P.1 | Brazil | Increased | Increased | Reduced |
| Delta | B.1.617.2 | India | Increased | Increased | Reduced |
| Drug | Modification of Therapy | Modification of Therapy in Relation to Vaccination |
|---|---|---|
| Hydroxychloroquine | NO | |
| Apremilast | NO | |
| IVIG | NO | |
| Glucocorticoids (Prednisone-equivalent < 10 mg/day) | NO | |
| Glucocorticoids (Prednisone-equivalent ≥ 10 mg/day) | The dose should be reduced to <10 mg/die, if possible, before each vaccine dose | |
| Sulphasalazine | NO | |
| Leflunomide | NO | |
| Mycophenolate mofetil | Delay the dose, if >20 mg/week, for 1 week for each vaccine dose, in case of stable disease * | |
| Azathioprine | NO | |
| Cyclophosphamide (Oral) | NO | |
| TNFα inhibitors (Adalimumab, Infliximab, Golimumab, Certolizumab, Etanercept) | NO | |
| Anti-IL-6R moAb (Tocilizumab) | NO | |
| IL-1β inhibitors (Anakinra, Canakinumab) | NO | |
| Anti-IL-17A moAbs (Secukinumab, Ixekizumab) | NO | |
| Anti-IL-12/23 moAb (Ustekinumab) | NO | |
| Anti-IL-23 moAbs (Tildrakizumab, Guselkumab, Risankizumab) | NO | |
| Anti-Blys moAb (Belimumab) | NO | |
| Calcineurin inhibitors (oral) | NO | |
| Methotrexate | Delay the dose, if >20 mg/week, for 1 week for each vaccine dose, in case of stable disease * | |
| JAK inhibitors | Delay the dose for 1 week for each vaccine dose * | |
| Abatacept sc | Temporary interruption 1 week before and 1 week after each vaccine dose * | |
| Abatacept iv | Each vaccine dose should be administered 4 weeks after the last infusion and the next infusion should be postponed 1 week * | |
| Cyclophosphamide iv | The infusion should be administered 1 week after vaccine * | |
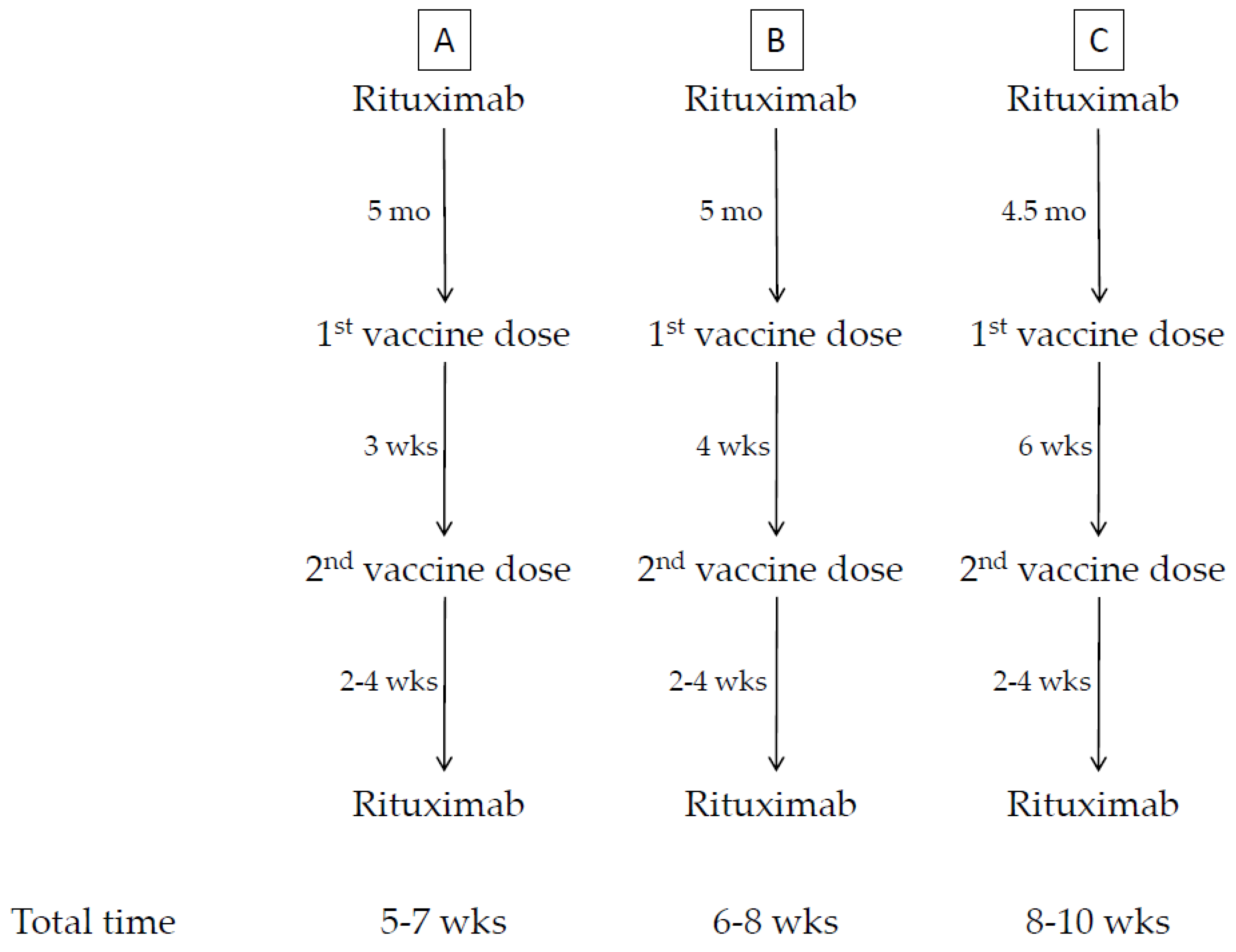
| Anti-CD20 moAb (Rituximab) | The first vaccine dose should be administered 4 weeks before the next scheduled cycle. Rituximab may be administered not before 3 weeks after the second vaccine dose, provided that the patient’s clinical condition allows. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D'Amelio, R.; Asero, R.; Cassatella, M.A.; Laganà, B.; Lunardi, C.; Migliorini, P.; Nisini, R.; Parronchi, P.; Quinti, I.; Racanelli, V.; et al. Anti-COVID-19 Vaccination in Patients with Autoimmune-Autoinflammatory Disorders and Primary/Secondary Immunodeficiencies: The Position of the Task Force on Behalf of the Italian Immunological Societies. Biomedicines 2021, 9, 1163. https://doi.org/10.3390/biomedicines9091163
D'Amelio R, Asero R, Cassatella MA, Laganà B, Lunardi C, Migliorini P, Nisini R, Parronchi P, Quinti I, Racanelli V, et al. Anti-COVID-19 Vaccination in Patients with Autoimmune-Autoinflammatory Disorders and Primary/Secondary Immunodeficiencies: The Position of the Task Force on Behalf of the Italian Immunological Societies. Biomedicines. 2021; 9(9):1163. https://doi.org/10.3390/biomedicines9091163
Chicago/Turabian StyleD'Amelio, Raffaele, Riccardo Asero, Marco Antonio Cassatella, Bruno Laganà, Claudio Lunardi, Paola Migliorini, Roberto Nisini, Paola Parronchi, Isabella Quinti, Vito Racanelli, and et al. 2021. "Anti-COVID-19 Vaccination in Patients with Autoimmune-Autoinflammatory Disorders and Primary/Secondary Immunodeficiencies: The Position of the Task Force on Behalf of the Italian Immunological Societies" Biomedicines 9, no. 9: 1163. https://doi.org/10.3390/biomedicines9091163
APA StyleD'Amelio, R., Asero, R., Cassatella, M. A., Laganà, B., Lunardi, C., Migliorini, P., Nisini, R., Parronchi, P., Quinti, I., Racanelli, V., Senna, G., Vacca, A., & Maggi, E. (2021). Anti-COVID-19 Vaccination in Patients with Autoimmune-Autoinflammatory Disorders and Primary/Secondary Immunodeficiencies: The Position of the Task Force on Behalf of the Italian Immunological Societies. Biomedicines, 9(9), 1163. https://doi.org/10.3390/biomedicines9091163







