Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds
Abstract
1. Introduction
2. Mechanical Environment
2.1. Mechanical Environment on the Bone
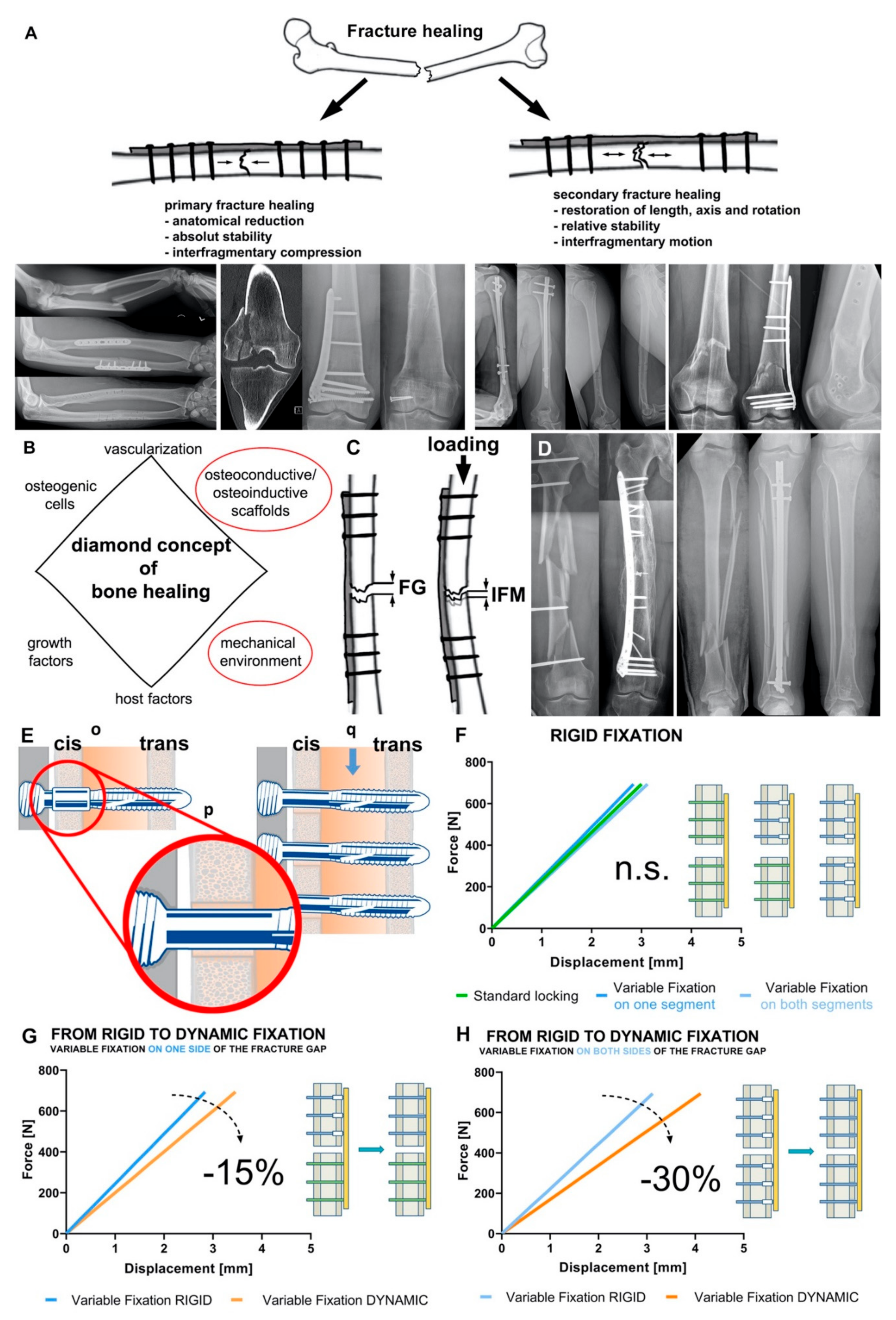
2.2. Mechanical Environment and Fracture Healing
2.3. Bone-Implant Construct Stiffness and “Dynamic” Osteosynthesis
2.4. Variable Fixation Technology
3. Bone Scaffolds Guiding Osteoinduction—An Interplay of Micro- and Nano-Topography
3.1. Natural Bone Architecture and Topography
3.2. Nanotopographical Cues of Scffolds Drive Osteoinduction and Possess Anti-Bacterial Activities
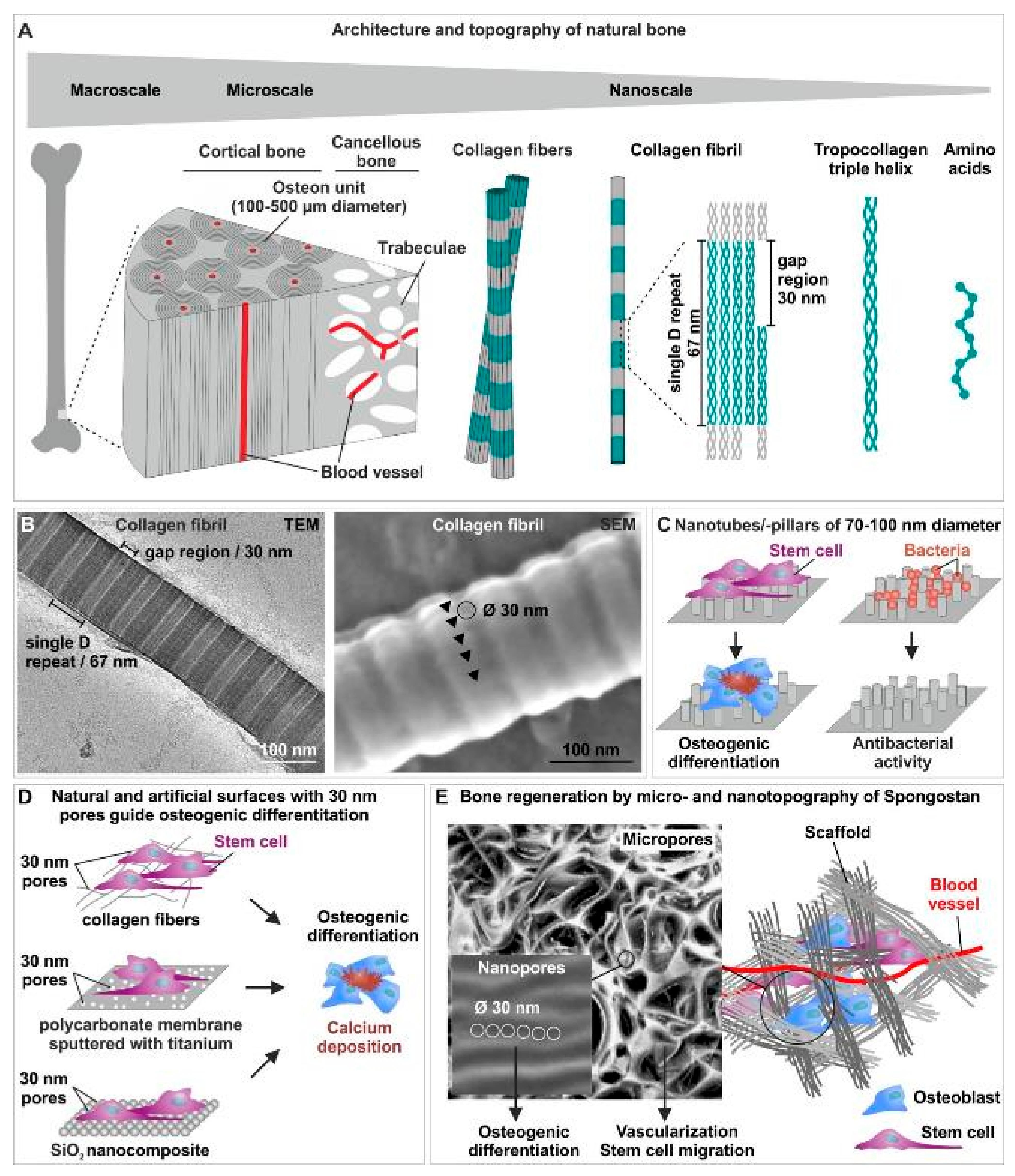
3.3. Linking the Nano- and the Microscale: Bone Scaffolds Combining an Optimal Microtopography for Vascularization and Stem Cell Migration with Nanotopographical Osteoinductive Cues
3.4. Bone Scaffolds Combining Topographical Cues with Stem Cell-Loading
3.5. Dynamizing Scaffold Design: Magnetic Scaffolds for Bone Regeneration
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Augat, P.; Hollensteiner, M.; von Ruden, C. The role of mechanical stimulation in the enhancement of bone healing. Injury 2020, 52, S78–S83. [Google Scholar] [CrossRef] [PubMed]
- Carter, D.R.; Orr, T.E. Skeletal development and bone functional adaptation. J. Bone. Miner. Res. 1992, 7 (Suppl. 2), S389–S395. [Google Scholar] [CrossRef] [PubMed]
- Wolff, J. The classic: On the inner architecture of bones and its importance for bone growth. 1870. Clin. Orthop. Relat. Res. 2010, 468, 1056–1065. [Google Scholar] [CrossRef]
- Augat, P.; Simon, U.; Liedert, A.; Claes, L. Mechanics and mechano-biology of fracture healing in normal and osteoporotic bone. Osteoporos. Int. 2005, 16 (Suppl. 2), S36–S43. [Google Scholar] [CrossRef]
- Mills, L.A.; Aitken, S.A.; Simpson, A. The risk of non-union per fracture: Current myths and revised figures from a population of over 4 million adults. Acta Orthop. 2017, 88, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Rupp, M.; Biehl, C.; Budak, M.; Thormann, U.; Heiss, C.; Alt, V. Diaphyseal long bone nonunions—Types, aetiology, economics, and treatment recommendations. Int. Orthop. 2018, 42, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Hak, D.J.; Fitzpatrick, D.; Bishop, J.A.; Marsh, J.L.; Tilp, S.; Schnettler, R.; Simpson, H.; Alt, V. Delayed union and nonunions: Epidemiology, clinical issues, and financial aspects. Injury 2014, 45 (Suppl. 2), S3–S7. [Google Scholar] [CrossRef]
- Copuroglu, C.; Calori, G.M.; Giannoudis, P.V. Fracture non-union: Who is at risk? Injury 2013, 44, 1379–1382. [Google Scholar] [CrossRef]
- Zura, R.; Xiong, Z.; Einhorn, T.; Watson, J.T.; Ostrum, R.F.; Prayson, M.J.; Della Rocca, G.J.; Mehta, S.; McKinley, T.; Wang, Z.; et al. Epidemiology of Fracture Nonunion in 18 Human Bones. JAMA Surg. 2016, 151, e162775. [Google Scholar] [CrossRef]
- Peters, R.M.; Claessen, F.M.; Doornberg, J.N.; Kolovich, G.P.; Diercks, R.L.; van den Bekerom, M.P. Union rate after operative treatment of humeral shaft nonunion—A systematic review. Injury 2015, 46, 2314–2324. [Google Scholar] [CrossRef]
- Giannoudis, P.V.; MacDonald, D.A.; Matthews, S.J.; Smith, R.M.; Furlong, A.J.; De Boer, P. Nonunion of the femoral diaphysis. The influence of reaming and non-steroidal anti-inflammatory drugs. J. Bone. Jt. Surg. Br. 2000, 82, 655–658. [Google Scholar] [CrossRef]
- Vallier, H.A.; Cureton, B.A.; Patterson, B.M. Randomized, prospective comparison of plate versus intramedullary nail fixation for distal tibia shaft fractures. J. Orthop. Trauma 2011, 25, 736–741. [Google Scholar] [CrossRef]
- Court-Brown, C.M. Reamed intramedullary tibial nailing: An overview and analysis of 1106 cases. J. Orthop. Trauma 2004, 18, 96–101. [Google Scholar] [CrossRef]
- Niikura, T.; Lee, S.Y.; Sakai, Y.; Nishida, K.; Kuroda, R.; Kurosaka, M. Causative factors of fracture nonunion: The proportions of mechanical, biological, patient-dependent, and patient-independent factors. J. Orthop. Sci. 2014, 19, 120–124. [Google Scholar] [CrossRef]
- Ekegren, C.L.; Edwards, E.R.; de Steiger, R.; Gabbe, B.J. Incidence, Costs and Predictors of Non-Union, Delayed Union and Mal-Union Following Long Bone Fracture. Int. J. Environ. Res. Public Health 2018, 15, 2845. [Google Scholar] [CrossRef]
- Antonova, E.; Le, T.K.; Burge, R.; Mershon, J. Tibia shaft fractures: Costly burden of nonunions. BMC Musculoskelet. Disord. 2013, 14, 42. [Google Scholar] [CrossRef]
- Galvain, T.; Chitnis, A.; Paparouni, K.; Tong, C.; Holy, C.E.; Giannoudis, P.V. The economic burden of infections following intramedullary nailing for a tibial shaft fracture in England. BMJ Open 2020, 10, e035404. [Google Scholar] [CrossRef]
- Glatt, V.; Evans, C.H.; Tetsworth, K. A Concert between Biology and Biomechanics: The Influence of the Mechanical Environment on Bone Healing. Front. Physiol. 2016, 7, 678. [Google Scholar] [CrossRef]
- Dang, M.; Saunders, L.; Niu, X.; Fan, Y.; Ma, P.X. Biomimetic delivery of signals for bone tissue engineering. Bone Res. 2018, 6, 25. [Google Scholar] [CrossRef]
- Yuan, J.; Xin, F.; Jiang, W. Underlying Signaling Pathways and Therapeutic Applications of Pulsed Electromagnetic Fields in Bone Repair. Cell Physiol. Biochem. 2018, 46, 1581–1594. [Google Scholar] [CrossRef]
- Daish, C.; Blanchard, R.; Fox, K.; Pivonka, P.; Pirogova, E. The Application of Pulsed Electromagnetic Fields (PEMFs) for Bone Fracture Repair: Past and Perspective Findings. Ann. Biomed. Eng 2018, 46, 525–542. [Google Scholar] [CrossRef]
- Everding, J.; Freistuhler, M.; Stolberg-Stolberg, J.; Raschke, M.J.; Garcia, P. Extracorporal shock wave therapy for the treatment of pseudarthrosis: New experiences with an old technology. Unfallchirurg 2017, 120, 969–978. [Google Scholar] [CrossRef]
- Giannoudis, P.V.; Einhorn, T.A.; Marsh, D. Fracture healing: The diamond concept. Injury 2007, 38 (Suppl. 4), S3–S6. [Google Scholar] [CrossRef]
- Andrzejowski, P.; Giannoudis, P.V. The ‘diamond concept’ for long bone non-union management. J. Orthop. Traumatol. 2019, 20, 21. [Google Scholar] [CrossRef] [PubMed]
- Yavropoulou, M.P.; Yovos, J.G. The molecular basis of bone mechanotransduction. J. Musculoskelet. Neuronal. Interact. 2016, 16, 221–236. [Google Scholar] [PubMed]
- Natali, A.N.; Meroi, E.A. A review of the biomechanical properties of bone as a material. J. Biomed. Eng. 1989, 11, 266–276. [Google Scholar] [CrossRef]
- Lotz, J.C.; Gerhart, T.N.; Hayes, W.C. Mechanical properties of trabecular bone from the proximal femur: A quantitative CT study. J. Comput. Assist. Tomogr. 1990, 14, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, S.; Kothari, M.; Augat, P.; Newitt, D.C.; Link, T.M.; Lin, J.C.; Lang, T.; Lu, Y.; Genant, H.K. High-resolution magnetic resonance imaging: Three-dimensional trabecular bone architecture and biomechanical properties. Bone 1998, 22, 445–454. [Google Scholar] [CrossRef]
- Martens, M.; Van Audekercke, R.; Delport, P.; De Meester, P.; Mulier, J.C. The mechanical characteristics of cancellous bone at the upper femoral region. J. Biomech. 1983, 16, 971–983. [Google Scholar] [CrossRef]
- Cowin, S.C. The mechanical and stress adaptive properties of bone. Ann. Biomed. Eng. 1983, 11, 263–295. [Google Scholar] [CrossRef]
- Reilly, D.T.; Burstein, A.H. The elastic and ultimate properties of compact bone tissue. J. Biomech. 1975, 8, 393–405. [Google Scholar] [CrossRef]
- Suzuki, T.; Matsuura, Y.; Yamazaki, T.; Akasaka, T.; Ozone, E.; Matsuyama, Y.; Mukai, M.; Ohara, T.; Wakita, H.; Taniguchi, S.; et al. Biomechanics of callus in the bone healing process, determined by specimen-specific finite element analysis. Bone 2020, 132, 115212. [Google Scholar] [CrossRef]
- Leong, P.L.; Morgan, E.F. Measurement of fracture callus material properties via nanoindentation. Acta Biomater. 2008, 4, 1569–1575. [Google Scholar] [CrossRef]
- Weinkamer, R.; Eberl, C.; Fratzl, P. Mechanoregulation of Bone Remodeling and Healing as Inspiration for Self-Repair in Materials. Biomimetics 2019, 4, 46. [Google Scholar] [CrossRef]
- Haffner-Luntzer, M.; Liedert, A.; Ignatius, A. Mechanobiology of bone remodeling and fracture healing in the aged organism. Innov. Surg. Sci. 2016, 1, 57–63. [Google Scholar] [CrossRef]
- Qin, L.; Liu, W.; Cao, H.; Xiao, G. Molecular mechanosensors in osteocytes. Bone Res. 2020, 8, 23. [Google Scholar] [CrossRef]
- Wittkowske, C.; Reilly, G.C.; Lacroix, D.; Perrault, C.M. In Vitro Bone Cell Models: Impact of Fluid Shear Stress on Bone Formation. Front. Bioeng. Biotechnol. 2016, 4, 87. [Google Scholar] [CrossRef]
- Witt, F.; Duda, G.N.; Bergmann, C.; Petersen, A. Cyclic mechanical loading enables solute transport and oxygen supply in bone healing: An in vitro investigation. Tissue Eng. Part A 2014, 20, 486–493. [Google Scholar] [CrossRef]
- Pauwels, F. A new theory on the influence of mechanical stimuli on the differentiation of supporting tissue. The tenth contribution to the functional anatomy and causal morphology of the supporting structure. Z. Anat. Entwicklungsgesch. 1960, 121, 478–515. [Google Scholar] [CrossRef]
- Perren, S.M.; Cordey, J. Tissue differences in fracture healing (author’s transl). Unfallheilkunde 1977, 80, 161–164. [Google Scholar]
- Perren, S.M. Fracture healing: Fracture healing understood as the result of a fascinating cascade of physical and biological interactions. Part II. Acta Chir. Orthop. Traumatol. Cech. 2015, 82, 13–21. [Google Scholar]
- Augat, P.; von Ruden, C. Evolution of fracture treatment with bone plates. Injury 2018, 49 (Suppl. 1), S2–S7. [Google Scholar] [CrossRef]
- Hierholzer, C.; Friederichs, J.; Augat, P.; Woltmann, A.; Trapp, O.; Buhren, V.; von Ruden, C. Evolution and principles of intramedullary locked nailing. Unfallchirurg 2018, 121, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Augat, P.; Margevicius, K.; Simon, J.; Wolf, S.; Suger, G.; Claes, L. Local tissue properties in bone healing: Influence of size and stability of the osteotomy gap. J. Orthop. Res. 1998, 16, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Claes, L.; Augat, P.; Suger, G.; Wilke, H.J. Influence of size and stability of the osteotomy gap on the success of fracture healing. J. Orthop. Res. 1997, 15, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Bottlang, M.; Doornink, J.; Lujan, T.J.; Fitzpatrick, D.C.; Marsh, J.L.; Augat, P.; von Rechenberg, B.; Lesser, M.; Madey, S.M. Effects of construct stiffness on healing of fractures stabilized with locking plates. J. Bone Jt. Surg. Am. 2010, 92 (Suppl. 2), 12–22. [Google Scholar] [CrossRef]
- Sigurdsen, U.; Reikeras, O.; Utvag, S.E. The influence of compression on the healing of experimental tibial fractures. Injury 2011, 42, 1152–1156. [Google Scholar] [CrossRef]
- Claes, L.; Augat, P.; Schorlemmer, S.; Konrads, C.; Ignatius, A.; Ehrnthaller, C. Temporary distraction and compression of a diaphyseal osteotomy accelerates bone healing. J. Orthop. Res. 2008, 26, 772–777. [Google Scholar] [CrossRef]
- Epari, D.R.; Kassi, J.P.; Schell, H.; Duda, G.N. Timely fracture-healing requires optimization of axial fixation stability. J. Bone Jt. Surg. Am. 2007, 89, 1575–1585. [Google Scholar] [CrossRef]
- Lienau, J.; Schell, H.; Duda, G.N.; Seebeck, P.; Muchow, S.; Bail, H.J. Initial vascularization and tissue differentiation are influenced by fixation stability. J. Orthop. Res. 2005, 23, 639–645. [Google Scholar] [CrossRef]
- Augat, P.; Burger, J.; Schorlemmer, S.; Henke, T.; Peraus, M.; Claes, L. Shear movement at the fracture site delays healing in a diaphyseal fracture model. J. Orthop. Res. 2003, 21, 1011–1017. [Google Scholar] [CrossRef]
- Tan, E.W.; Sirisreetreerux, N.; Paez, A.G.; Parks, B.G.; Schon, L.C.; Hasenboehler, E.A. Early Weightbearing After Operatively Treated Ankle Fractures: A Biomechanical Analysis. Foot Ankle Int. 2016, 37, 652–658. [Google Scholar] [CrossRef]
- Willie, B.M.; Blakytny, R.; Glockelmann, M.; Ignatius, A.; Claes, L. Temporal variation in fixation stiffness affects healing by differential cartilage formation in a rat osteotomy model. Clin. Orthop. Relat. Res. 2011, 469, 3094–3101. [Google Scholar] [CrossRef]
- Pfeufer, D.; Zeller, A.; Mehaffey, S.; Bocker, W.; Kammerlander, C.; Neuerburg, C. Weight-bearing restrictions reduce postoperative mobility in elderly hip fracture patients. Arch. Orthop. Trauma Surg. 2019, 139, 1253–1259. [Google Scholar] [CrossRef]
- Kammerlander, C.; Pfeufer, D.; Lisitano, L.A.; Mehaffey, S.; Bocker, W.; Neuerburg, C. Inability of Older Adult Patients with Hip Fracture to Maintain Postoperative Weight-Bearing Restrictions. J. Bone Jt. Surg. Am. 2018, 100, 936–941. [Google Scholar] [CrossRef]
- Consigliere, P.; Iliopoulos, E.; Ads, T.; Trompeter, A. Early versus delayed weight bearing after surgical fixation of distal femur fractures: A non-randomized comparative study. Eur. J. Orthop. Surg. Traumatol. 2019, 29, 1789–1794. [Google Scholar] [CrossRef]
- Dehghan, N.; McKee, M.D.; Jenkinson, R.J.; Schemitsch, E.H.; Stas, V.; Nauth, A.; Hall, J.A.; Stephen, D.J.; Kreder, H.J. Early Weightbearing and Range of Motion Versus Non-Weightbearing and Immobilization After Open Reduction and Internal Fixation of Unstable Ankle Fractures: A Randomized Controlled Trial. J. Orthop. Trauma 2016, 30, 345–352. [Google Scholar] [CrossRef]
- Hoegel, F.W.; Hoffmann, S.; Weninger, P.; Buhren, V.; Augat, P. Biomechanical comparison of locked plate osteosynthesis, reamed and unreamed nailing in conventional interlocking technique, and unreamed angle stable nailing in distal tibia fractures. J. Trauma Acute Care Surg. 2012, 73, 933–938. [Google Scholar] [CrossRef]
- Kuhn, S.; Greenfield, J.; Arand, C.; Jarmolaew, A.; Appelmann, P.; Mehler, D.; Rommens, P.M. Treatment of distal intraarticular tibial fractures: A biomechanical evaluation of intramedullary nailing vs. angle-stable plate osteosynthesis. Injury 2015, 46 (Suppl. 4), S99–S103. [Google Scholar] [CrossRef]
- Nourisa, J.; Rouhi, G. Biomechanical evaluation of intramedullary nail and bone plate for the fixation of distal metaphyseal fractures. J. Mech. Behav. Biomed. Mater. 2016, 56, 34–44. [Google Scholar] [CrossRef]
- Bonyun, M.; Nauth, A.; Egol, K.A.; Gardner, M.J.; Kregor, P.J.; McKee, M.D.; Wolinsky, P.R.; Schemitsch, E.H. Hot topics in biomechanically directed fracture fixation. J. Orthop. Trauma 2014, 28 (Suppl. 1), S32–S35. [Google Scholar] [CrossRef]
- Bottlang, M.; Lesser, M.; Koerber, J.; Doornink, J.; von Rechenberg, B.; Augat, P.; Fitzpatrick, D.C.; Madey, S.M.; Marsh, J.L. Far cortical locking can improve healing of fractures stabilized with locking plates. J. Bone Jt. Surg. Am. 2010, 92, 1652–1660. [Google Scholar] [CrossRef] [PubMed]
- Bottlang, M.; Tsai, S.; Bliven, E.K.; von Rechenberg, B.; Kindt, P.; Augat, P.; Henschel, J.; Fitzpatrick, D.C.; Madey, S.M. Dynamic Stabilization of Simple Fractures With Active Plates Delivers Stronger Healing Than Conventional Compression Plating. J. Orthop. Trauma 2017, 31, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Bottlang, M.; Tsai, S.; Bliven, E.K.; von Rechenberg, B.; Klein, K.; Augat, P.; Henschel, J.; Fitzpatrick, D.C.; Madey, S.M. Dynamic Stabilization with Active Locking Plates Delivers Faster, Stronger, and More Symmetric Fracture-Healing. J. Bone Jt. Surg. Am. 2016, 98, 466–474. [Google Scholar] [CrossRef]
- Hofmann-Fliri, L.; Epari, D.R.; Schwyn, R.; Zeiter, S.; Windolf, M. Biphasic Plating—In vivo study of a novel fixation concept to enhance mechanobiological fracture healing. Injury 2020, 51, 1751–1758. [Google Scholar] [CrossRef] [PubMed]
- Richter, H.; Plecko, M.; Andermatt, D.; Frigg, R.; Kronen, P.W.; Klein, K.; Nuss, K.; Ferguson, S.J.; Stockle, U.; von Rechenberg, B. Dynamization at the near cortex in locking plate osteosynthesis by means of dynamic locking screws: An experimental study of transverse tibial osteotomies in sheep. J. Bone Jt. Surg. Am. 2015, 97, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Ghiasi, M.S.; Chen, J.; Vaziri, A.; Rodriguez, E.K.; Nazarian, A. Bone fracture healing in mechanobiological modeling: A review of principles and methods. Bone Rep. 2017, 6, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Augat, P.; Merk, J.; Ignatius, A.; Margevicius, K.; Bauer, G.; Rosenbaum, D.; Claes, L. Early, full weightbearing with flexible fixation delays fracture healing. Clin. Orthop. Relat. Res. 1996, 328, 194–202. [Google Scholar] [CrossRef]
- Claes, L.; Eckert-Hubner, K.; Augat, P. The effect of mechanical stability on local vascularization and tissue differentiation in callus healing. J. Orthop. Res. 2002, 20, 1099–1105. [Google Scholar] [CrossRef]
- Salisbury Palomares, K.T.; Gerstenfeld, L.C.; Wigner, N.A.; Lenburg, M.E.; Einhorn, T.A.; Morgan, E.F. Transcriptional profiling and biochemical analysis of mechanically induced cartilaginous tissues in a rat model. Arthritis Rheum. 2010, 62, 1108–1118. [Google Scholar] [CrossRef]
- Palomares, K.T.; Gleason, R.E.; Mason, Z.D.; Cullinane, D.M.; Einhorn, T.A.; Gerstenfeld, L.C.; Morgan, E.F. Mechanical stimulation alters tissue differentiation and molecular expression during bone healing. J. Orthop. Res. 2009, 27, 1123–1132. [Google Scholar] [CrossRef]
- Weaver, A.S.; Su, Y.P.; Begun, D.L.; Miller, J.D.; Alford, A.I.; Goldstein, S.A. The effects of axial displacement on fracture callus morphology and MSC homing depend on the timing of application. Bone 2010, 47, 41–48. [Google Scholar] [CrossRef]
- Tufekci, P.; Tavakoli, A.; Dlaska, C.; Neumann, M.; Shanker, M.; Saifzadeh, S.; Steck, R.; Schuetz, M.; Epari, D. Early mechanical stimulation only permits timely bone healing in sheep. J. Orthop. Res. 2018, 36, 1790–1796. [Google Scholar] [CrossRef]
- Glatt, V.; Miller, M.; Ivkovic, A.; Liu, F.; Parry, N.; Griffin, D.; Vrahas, M.; Evans, C. Improved healing of large segmental defects in the rat femur by reverse dynamization in the presence of bone morphogenetic protein-2. J. Bone Jt. Surg. Am. 2012, 94, 2063–2073. [Google Scholar] [CrossRef]
- Klein, P.; Schell, H.; Streitparth, F.; Heller, M.; Kassi, J.P.; Kandziora, F.; Bragulla, H.; Haas, N.P.; Duda, G.N. The initial phase of fracture healing is specifically sensitive to mechanical conditions. J. Orthop. Res. 2003, 21, 662–669. [Google Scholar] [CrossRef]
- Plecko, M.; Lagerpusch, N.; Andermatt, D.; Frigg, R.; Koch, R.; Sidler, M.; Kronen, P.; Klein, K.; Nuss, K.; Burki, A.; et al. The dynamisation of locking plate osteosynthesis by means of dynamic locking screws (DLS)-an experimental study in sheep. Injury 2013, 44, 1346–1357. [Google Scholar] [CrossRef]
- Frank, A.; Brianza, S.; Plecko, M.; Raschke, M.J.; Wahnert, D. Variable Fixation Technology Provides Rigid as Well as Progressive Dynamic Fixation: A Biomechanical Investigation. J. Bone Jt. Surg. Am. 2020, 102, e115. [Google Scholar] [CrossRef]
- Plecko, M.; Klein, K.; Planzer, K.; Wahnert, D.; Behm, P.; Ferguson, S.J.; Brianza, S.; Stadelmann, V.A.; von Rechenberg, B. Variable fixation promotes callus formation: An experimental study on transverse tibial osteotomies stabilized with locking plates. BMC Musculoskelet. Disord. 2020, 21, 806. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Luo, D.; Liu, Y. Effect of the nano/microscale structure of biomaterial scaffolds on bone regeneration. Int. J. Oral Sci. 2020, 12, 6. [Google Scholar] [CrossRef]
- Fillingham, Y.; Jacobs, J. Bone grafts and their substitutes. Bone Jt. J. 2016, 98-B, 6–9. [Google Scholar] [CrossRef]
- Du, Y.; Guo, J.L.; Wang, J.; Mikos, A.G.; Zhang, S. Hierarchically designed bone scaffolds: From internal cues to external stimuli. Biomaterials 2019, 218, 119334. [Google Scholar] [CrossRef] [PubMed]
- Gong, T.; Xie, J.; Liao, J.; Zhang, T.; Lin, S.; Lin, Y. Nanomaterials and bone regeneration. Bone Res. 2015, 3, 15029. [Google Scholar] [CrossRef] [PubMed]
- Hodge, A.J.; Petruska, J.A. Aspects of Protein Structure; Ramachandran, G.N., Ed.; Academic Press: New York, NY, USA, 1963; pp. 289–300. [Google Scholar]
- Fang, M.; Holl, M.M. Variation in type I collagen fibril nanomorphology: The significance and origin. BoneKEy Rep. 2013, 2, 394. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Lee, B.; Thomopoulos, S.; Jun, Y.-S. The role of confined collagen geometry in decreasing nucleation energy barriers to intrafibrillar mineralization. Nat. Commun. 2018, 9, 962. [Google Scholar] [CrossRef]
- Greiner, J.F.; Gottschalk, M.; Fokin, N.; Buker, B.; Kaltschmidt, B.P.; Dreyer, A.; Vordemvenne, T.; Kaltschmidt, C.; Hutten, A.; Kaltschmidt, B. Natural and synthetic nanopores directing osteogenic differentiation of human stem cells. Nanomedicine 2019, 17, 319–328. [Google Scholar] [CrossRef]
- Vordemvenne, T.; Wahnert, D.; Koettnitz, J.; Merten, M.; Fokin, N.; Becker, A.; Buker, B.; Vogel, A.; Kronenberg, D.; Stange, R.; et al. Bone Regeneration: A Novel Osteoinductive Function of Spongostan by the Interplay between Its Nano- and Microtopography. Cells 2020, 9, 654. [Google Scholar] [CrossRef]
- Dalby, M.J.; Gadegaard, N.; Tare, R.; Andar, A.; Riehle, M.O.; Herzyk, P.; Wilkinson, C.D.; Oreffo, R.O. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat. Mater. 2007, 6, 997–1003. [Google Scholar] [CrossRef]
- Oh, S.; Brammer, K.S.; Li, Y.S.; Teng, D.; Engler, A.J.; Chien, S.; Jin, S. Stem cell fate dictated solely by altered nanotube dimension. Proc. Natl. Acad. Sci. USA 2009, 106, 2130–2135. [Google Scholar] [CrossRef]
- Altuntas, S.; Dhaliwal, H.K.; Bassous, N.J.; Radwan, A.E.; Alpaslan, P.; Webster, T.; Buyukserin, F.; Amiji, M. Nanopillared Chitosan/Gelatin Films: A Biomimetic Approach for Improved Osteogenesis. ACS Biomater. Sci. Eng. 2019, 5, 4311–4322. [Google Scholar] [CrossRef]
- Hendricks, K.J.; Burd, T.A.; Anglen, J.O.; Simpson, A.W.; Christensen, G.D.; Gainor, B.J. Synergy between Staphylococcus aureus and Pseudomonas aeruginosa in a rat model of complex orthopaedic wounds. J. Bone Jt. Surg. Am. Vol. 2001, 83, 855–861. [Google Scholar] [CrossRef]
- Wu, S.; Zuber, F.; Maniura-Weber, K.; Brugger, J.; Ren, Q. Nanostructured surface topographies have an effect on bactericidal activity. J. Nanobiotechnol. 2018, 16, 20. [Google Scholar] [CrossRef]
- Jaggessar, A.; Shahali, H.; Mathew, A.; Yarlagadda, P.K.D.V. Bio-mimicking nano and micro-structured surface fabrication for antibacterial properties in medical implants. J. Nanobiotechnol. 2017, 15, 64. [Google Scholar] [CrossRef]
- Park, J.; Bauer, S.; von der Mark, K.; Schmuki, P. Nanosize and vitality: TiO2 nanotube diameter directs cell fate. Nano Lett. 2007, 7, 1686–1691. [Google Scholar] [CrossRef]
- Shih, Y.-R.V.; Tseng, K.-F.; Lai, H.-Y.; Lin, C.-H.; Lee, O.K. Matrix stiffness regulation of integrin-mediated mechanotransduction during osteogenic differentiation of human mesenchymal stem cells. J. Bone Miner. Res. 2011, 26, 730–738. [Google Scholar] [CrossRef]
- Niu, H.; Lin, D.; Tang, W.; Ma, Y.; Duan, B.; Yuan, Y.; Liu, C. Surface Topography Regulates Osteogenic Differentiation of MSCs via Crosstalk between FAK/MAPK and ILK/β-Catenin Pathways in a Hierarchically Porous Environment. ACS Biomater. Sci. Eng. 2017, 3, 3161–3175. [Google Scholar] [CrossRef]
- Schurmann, M.; Wolff, A.; Widera, D.; Hauser, S.; Heimann, P.; Hutten, A.; Kaltschmidt, C.; Kaltschmidt, B. Interaction of adult human neural crest-derived stem cells with a nanoporous titanium surface is sufficient to induce their osteogenic differentiation. Stem Cell Res. 2014, 13, 98–110. [Google Scholar] [CrossRef][Green Version]
- de Barros, E.; Lima Bueno, R.; Ponce, K.J.; Dias, A.P.; Guadarrama Bello, D.; Brunski, J.B.; Nanci, A. Influence of Nanotopography on Early Bone Healing during Controlled Implant Loading. Nanomaterials 2020, 10, 2191. [Google Scholar] [CrossRef]
- Marino, A.; Filippeschi, C.; Genchi, G.G.; Mattoli, V.; Mazzolai, B.; Ciofani, G. The Osteoprint: A bioinspired two-photon polymerized 3-D structure for the enhancement of bone-like cell differentiation. Acta Biomater. 2014, 10, 4304–4313. [Google Scholar] [CrossRef]
- Hayashi, K.; Ishikawa, K. Effects of nanopores on the mechanical strength, osteoclastogenesis, and osteogenesis in honeycomb scaffolds. J. Mater. Chem. B 2020, 8, 8536–8545. [Google Scholar] [CrossRef]
- Dutta, R.C.; Dey, M.; Dutta, A.K.; Basu, B. Competent processing techniques for scaffolds in tissue engineering. Biotechnol. Adv. 2017, 35, 240–250. [Google Scholar] [CrossRef]
- Janson, I.A.; Putnam, A.J. Extracellular matrix elasticity and topography: Material-based cues that affect cell function via conserved mechanisms. J. Biomed. Mater. Res. A 2015, 103, 1246–1258. [Google Scholar] [CrossRef]
- Cipitria, A.; Lange, C.; Schell, H.; Wagermaier, W.; Reichert, J.C.; Hutmacher, D.W.; Fratzl, P.; Duda, G.N. Porous scaffold architecture guides tissue formation. J. Bone Miner. Res. 2012, 27, 1275–1288. [Google Scholar] [CrossRef]
- Lan Levengood, S.K.; Polak, S.J.; Wheeler, M.B.; Maki, A.J.; Clark, S.G.; Jamison, R.D.; Wagoner Johnson, A.J. Multiscale osteointegration as a new paradigm for the design of calcium phosphate scaffolds for bone regeneration. Biomaterials 2010, 31, 3552–3563. [Google Scholar] [CrossRef]
- Petersen, A.; Princ, A.; Korus, G.; Ellinghaus, A.; Leemhuis, H.; Herrera, A.; Klaumünzer, A.; Schreivogel, S.; Woloszyk, A.; Schmidt-Bleek, K.; et al. A biomaterial with a channel-like pore architecture induces endochondral healing of bone defects. Nat. Commun. 2018, 9, 4430. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, S.; Luo, D.; Xue, Z.; Yang, X.; Gu, L.; Zhou, Y.; Wang, T. Hierarchically Staggered Nanostructure of Mineralized Collagen as a Bone-Grafting Scaffold. Adv. Mater. 2016, 28, 8740–8748. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.S.; He, D.Q.; Luo, D.; Wang, Y.; Yu, M.; Guan, B.; Fu, Y.; Li, Z.X.; Zhang, T.; Zhou, Y.H.; et al. A Biomimetic Hierarchical Nanointerface Orchestrates Macrophage Polarization and Mesenchymal Stem Cell Recruitment To Promote Endogenous Bone Regeneration. ACS Nano 2019, 13, 6581–6595. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, N.; Qi, Y.P.; Dai, L.; Bryan, T.E.; Mao, J.; Pashley, D.H.; Tay, F.R. Intrafibrillar collagen mineralization produced by biomimetic hierarchical nanoapatite assembly. Adv. Mater. 2011, 23, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Liu, S.; Cui, S.J.; Kou, X.X.; Wang, X.D.; Liu, X.M.; Sun, Y.; Wang, G.N.; Liu, Y.; Zhou, Y.H. Surface Chemistry of Nanoscale Mineralized Collagen Regulates Periodontal Ligament Stem Cell Fate. ACS Appl. Mater. Interfaces 2016, 8, 15958–15966. [Google Scholar] [CrossRef]
- Wahnert, D.; Koettnitz, J.; Merten, M.; Kronenberg, D.; Stange, R.; Greiner, J.F.W.; Kaltschmidt, C.; Vordemvenne, T.; Kaltschmidt, B. Spongostan™ Leads to Increased Regeneration of a Rat Calvarial Critical Size Defect Compared to NanoBone((R)) and Actifuse. Materials 2021, 14, 1961. [Google Scholar] [CrossRef]
- Abshagen, K.; Schrodi, I.; Gerber, T.; Vollmar, B. In vivo analysis of biocompatibility and vascularization of the synthetic bone grafting substitute NanoBone®. J. Biomed. Mater. Res. Part A 2009, 91A, 557–566. [Google Scholar] [CrossRef]
- Oryan, A.; Baghaban Eslaminejad, M.; Kamali, A.; Hosseini, S.; Moshiri, A.; Baharvand, H. Mesenchymal stem cells seeded onto tissue-engineered osteoinductive scaffolds enhance the healing process of critical-sized radial bone defects in rat. Cell Tissue Res. 2018, 374, 63–81. [Google Scholar] [CrossRef]
- Tortelli, F.; Tasso, R.; Loiacono, F.; Cancedda, R. The development of tissue-engineered bone of different origin through endochondral and intramembranous ossification following the implantation of mesenchymal stem cells and osteoblasts in a murine model. Biomaterials 2010, 31, 242–249. [Google Scholar] [CrossRef]
- Moradi, S.L.; Golchin, A.; Hajishafieeha, Z.; Khani, M.-M.; Ardeshirylajimi, A. Bone tissue engineering: Adult stem cells in combination with electrospun nanofibrous scaffolds. J. Cell. Physiol. 2018, 233, 6509–6522. [Google Scholar] [CrossRef]
- Hai-Yan XU, N.G. Magnetic responsive scaffolds and magnetic fields in bone repair and regeneration. Front. Mater. Sci. 2014, 8, 20–31. [Google Scholar] [CrossRef]
- Jiang, P.; Zhang, Y.; Zhu, C.; Zhang, W.; Mao, Z.; Gao, C. Fe3O4/BSA particles induce osteogenic differentiation of mesenchymal stem cells under static magnetic field. Acta Biomater. 2016, 46, 141–150. [Google Scholar] [CrossRef]
- Xia, Y.; Sun, J.; Zhao, L.; Zhang, F.; Liang, X.J.; Guo, Y.; Weir, M.D.; Reynolds, M.A.; Gu, N.; Xu, H.H.K. Magnetic field and nano-scaffolds with stem cells to enhance bone regeneration. Biomaterials 2018, 183, 151–170. [Google Scholar] [CrossRef]
- Panseri, S.; Russo, A.; Sartori, M.; Giavaresi, G.; Sandri, M.; Fini, M.; Maltarello, M.C.; Shelyakova, T.; Ortolani, A.; Visani, A.; et al. Modifying bone scaffold architecture in vivo with permanent magnets to facilitate fixation of magnetic scaffolds. Bone 2013, 56, 432–439. [Google Scholar] [CrossRef]
- Russo, A.; Bianchi, M.; Sartori, M.; Parrilli, A.; Panseri, S.; Ortolani, A.; Sandri, M.; Boi, M.; Salter, D.M.; Maltarello, M.C.; et al. Magnetic forces and magnetized biomaterials provide dynamic flux information during bone regeneration. J. Mater. Sci. Mater. Med. 2016, 27, 51. [Google Scholar] [CrossRef]
- Castro, P.S.; Bertotti, M.; Naves, A.F.; Catalani, L.H.; Cornejo, D.R.; Bloisi, G.D.; Petri, D.F.S. Hybrid magnetic scaffolds: The role of scaffolds charge on the cell proliferation and Ca2+ ions permeation. Colloids Surf. B Biointerfaces 2017, 156, 388–396. [Google Scholar] [CrossRef]
- Fanti, A.; Lodi, M.B.; Mazzarella, G. Enhancement of Cell Migration Rate Toward a Superparamagnetic Scaffold Using LF Magnetic Fields. IEEE Trans. Magn. 2016, 52, 1–8. [Google Scholar] [CrossRef]
- Lodi, M.B.; Fanti, A.; Muntoni, G.; Mazzarella, G. A Multiphysic Model for the Hyperthermia Treatment of Residual Osteosarcoma Cells in Upper Limbs Using Magnetic Scaffolds. IEEE J. Multiscale Multiphysics Comput. Tech. 2019, 4, 337–347. [Google Scholar] [CrossRef]
- Baeza, A.; Arcos, D.; Vallet-Regi, M. Thermoseeds for interstitial magnetic hyperthermia: From bioceramics to nanoparticles. J. Phys. Condens. Matter. 2013, 25, 484003. [Google Scholar] [CrossRef]
| Trabecular Bone | Cortical Bone | Callus | Granulation Tissue | |
|---|---|---|---|---|
| Peak load (MPa) | 2.2–6.8 [27,28,29] | 109–205 [30,31] | 5.3 [32] | - |
| E-modulus (MPa) | 74–900 [27,28,29] | 10,460–17,100 [30,31] | 98 [32] | - |
| Indentation modulus (MPa) | - | 132 [33] | 2.89 [33] | 0.99 [33] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wähnert, D.; Greiner, J.; Brianza, S.; Kaltschmidt, C.; Vordemvenne, T.; Kaltschmidt, B. Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds. Biomedicines 2021, 9, 746. https://doi.org/10.3390/biomedicines9070746
Wähnert D, Greiner J, Brianza S, Kaltschmidt C, Vordemvenne T, Kaltschmidt B. Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds. Biomedicines. 2021; 9(7):746. https://doi.org/10.3390/biomedicines9070746
Chicago/Turabian StyleWähnert, Dirk, Johannes Greiner, Stefano Brianza, Christian Kaltschmidt, Thomas Vordemvenne, and Barbara Kaltschmidt. 2021. "Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds" Biomedicines 9, no. 7: 746. https://doi.org/10.3390/biomedicines9070746
APA StyleWähnert, D., Greiner, J., Brianza, S., Kaltschmidt, C., Vordemvenne, T., & Kaltschmidt, B. (2021). Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds. Biomedicines, 9(7), 746. https://doi.org/10.3390/biomedicines9070746







