SR-B1, a Key Receptor Involved in the Progression of Cardiovascular Disease: A Perspective from Mice and Human Genetic Studies
Abstract
1. Introduction
2. SR-B1 in Lipoprotein Metabolism
3. SR-B1, an Important Participant in the Development of Cardiovascular Disease
4. Studies in Gene-Targeted Mice Related to Lipoprotein Metabolism and Cardiovascular Disease
5. Human Genetic Variants of SCARB1 in Lipoprotein Metabolism and Cardiovascular Disease
6. Genome-Wide Association Studies of SCARB1
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; De Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart Disease and Stroke Statistics’2017 Update: A Report from the American Heart Association. Circulation 2017, 135, e146–e603. [Google Scholar]
- Castelli, W.P.; Garrison, R.J.; Wilson, P.W.F.; Abbott, R.D.; Kalousdian, S.; Kannel, W.B. Incidence of Coronary Heart Disease and Lipoprotein Cholesterol Levels: The Framingham Study. JAMA J. Am. Med. Assoc. 1986, 256, 2835–2838. [Google Scholar] [CrossRef]
- Hoekstra, M. SR-BI as target in atherosclerosis and cardiovascular disease—A comprehensive appraisal of the cellular functions of SR-BI in physiology and disease. Atherosclerosis 2017, 258, 153–161. [Google Scholar] [CrossRef]
- Linton, M.F.; Tao, H.; Linton, E.F.; Yancey, P.G. SR-BI: A Multifunctional Receptor in Cholesterol Homeostasis and Atherosclerosis. Trends Endocrinol. Metab. 2017, 28, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Di Angelantonio, E.; Sarwar, N.; Perry, P.; Kaptoge, S.; Ray, K.K.; Thompson, A.; Wood, A.M.; Lewington, S.; Sattar, N.; Packard, C.J.; et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA J. Am. Med. Assoc. 2009, 302, 1993–2000. [Google Scholar] [CrossRef]
- Assmann, G.; Cullen, P.; Schulte, H. Simple scoring scheme for calculating the risk of acute coronary events based on the 10-year follow-up of the prospective cardiovascular Münster (PROCAM) study. Circulation 2002, 105, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Brunham, L.R.; Hayden, M.R. Human genetics of HDL: Insight into particle metabolism and function. Prog. Lipid Res. 2015, 58, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Helgadottir, A.; Sulem, P.; Thorgeirsson, G.; Gretarsdottir, S.; Thorleifsson, G.; Jensson, B.Ö.; Arnadottir, G.A.; Olafsson, I.; Eyjolfsson, G.I.; Sigurdardottir, O.; et al. Rare SCARB1 mutations associate with high-density lipoprotein cholesterol but not with coronary artery disease. Eur. Heart J. 2018, 39, 2172–2178. [Google Scholar] [CrossRef]
- Voight, B.F.; Peloso, G.M.; Orho-Melander, M.; Frikke-Schmidt, R.; Barbalic, M.; Jensen, M.K.; Hindy, G.; Hólm, H.; Ding, E.L.; Johnson, T.; et al. Plasma HDL cholesterol and risk of myocardial infarction: A mendelian randomisation study. Lancet 2012, 380, 572–580. [Google Scholar] [CrossRef]
- Holmes, M.V.; Ala-Korpela, M.; Smith, G.D. Mendelian randomization in cardiometabolic disease: Challenges in evaluating causality. Nat. Rev. Cardiol. 2017, 14, 577–599. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Olsson, A.G.; Abt, M.; Ballantyne, C.M.; Barter, P.J.; Brumm, J.; Chaitman, B.R.; Holme, I.M.; Kallend, D.; Leiter, L.A.; et al. Effects of dalcetrapib in patients with a recent acute coronary syndrome. N. Engl. J. Med. 2012, 367, 2089–2099. [Google Scholar] [CrossRef]
- Barter, P.J.; Caulfield, M.; Eriksson, M.; Grundy, S.M.; Kastelein, J.J.P.; Komajda, M.; Lopez-Sendon, J.; Mosca, L.; Tardif, J.C.; Waters, D.D.; et al. Effects of torcetrapib in patients at high risk for coronary events. N. Engl. J. Med. 2007, 357, 2109–2122. [Google Scholar] [CrossRef]
- Van der Steeg, W.A.; Holme, I.; Boekholdt, S.M.; Larsen, M.L.; Lindahl, C.; Stroes, E.S.G.; Tikkanen, M.J.; Wareham, N.J.; Faergeman, O.; Olsson, A.G.; et al. High-Density Lipoprotein Cholesterol, High-Density Lipoprotein Particle Size, and Apolipoprotein A-I: Significance for Cardiovascular Risk. The IDEAL and EPIC-Norfolk Studies. J. Am. Coll. Cardiol. 2008, 51, 634–642. [Google Scholar] [CrossRef]
- Chroni, A.; Kardassis, D. HDL Dysfunction Caused by Mutations in apoA-I and Other Genes that are Critical for HDL Biogenesis and Remodeling. Curr. Med. Chem. 2018, 26, 1544–1575. [Google Scholar] [CrossRef]
- Acton, S.; Rigotti, A.; Landschulz, K.T.; Xu, S.; Hobbs, H.H.; Kriegert, M. Identification of scavenger receptor SR-BI as a high density lipoprotein receptor. Science (80-.) 1996, 271, 518–520. [Google Scholar] [CrossRef]
- Acton, S.L.; Scherer, P.E.; Lodish, H.F.; Krieger, M. Expression cloning of SR-BI, a CD36-related class B scavenger receptor. J. Biol. Chem. 1994, 269, 21003–21009. [Google Scholar] [CrossRef]
- Calvo, D.; Gómez-Coronado, D.; Lasunción, M.A.; Vega, M.A. CLA-1 is an 85-kD plasma membrane glycoprotein that acts as a high- affinity receptor for both native (HDL, LDL, and VLDL) and modified (OxLDL and AcLDL) lipoproteins. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2341–2349. [Google Scholar] [CrossRef]
- Yang, X.P.; Amar, M.J.; Vaisman, B.; Bocharov, A.V.; Vishnyakova, T.G.; Freeman, L.A.; Kurlander, R.J.; Patterson, A.P.; Becker, L.C.; Remaley, A.T. Scavenger receptor-BI is a receptor for lipoprotein(a). J. Lipid Res. 2013, 54, 2450–2457. [Google Scholar] [CrossRef]
- Utermann, G. The mysteries of lipoprotein(a). Science (80-.) 1989, 246, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Al-Jarallah, A.; Trigatti, B.L. A role for the scavenger receptor, class B type I in high density lipoprotein dependent activation of cellular signaling pathways. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2010, 1801, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Citro, V.; Capone, D. Nonalcoholic Fatty Liver Disease: A Challenge from Mechanisms to Therapy. J. Clin. Med. 2019, 9, 15. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Liu, S.; Chen, H.T.; Yu, C.H.; Teng, X.D.; Yao, H.T.; Xu, G.Q. Upregulation of caveolin-1 and SR-B1 in mice with non-alcoholic fatty liver disease. Hepatobiliary Pancreat. Dis. Int. 2013, 12, 630–636. [Google Scholar] [CrossRef]
- Krieger, M. Charting the fate of the “good cholesterol”: Identification and characterization of the high-density lipoprotein receptor SR-BI. Annu. Rev. Biochem. 1999, 68, 523–558. [Google Scholar] [CrossRef] [PubMed]
- Connelly, M.A.; Williams, D.L. Scavenger receptor BI: A scavenger receptor with a mission to transport high density lipoprotein lipids. Curr. Opin. Lipidol. 2004, 15, 287–295. [Google Scholar] [CrossRef]
- Rigotti, A.; Miettinen, H.E.; Krieger, M. The role of the high-density lipoprotein receptor SR-BI in the lipid metabolism of endocrine and other tissues. Endocr. Rev. 2003, 24, 357–387. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.-J.; Azhar, S.; Kraemer, F.B. SR-B1: A Unique Multifunctional Receptor for Cholesterol Influx and Efflux. Annu. Rev. Physiol. 2018, 80, 95–116. [Google Scholar] [CrossRef]
- Shen, W.J.; Asthana, S.; Kraemer, F.B.; Azhar, S. Scavenger receptor B type 1: Expression, molecular regulation, and cholesterol transport function. J. Lipid Res. 2018, 59, 1114–1131. [Google Scholar] [CrossRef]
- Shen, W.J.; Hu, J.; Hu, Z.; Kraemer, F.B.; Azhar, S. Scavenger Receptor class B type i (SR-BI): A versatile receptor with multiple functions and actions. Metabolism 2014, 63, 875–886. [Google Scholar] [CrossRef]
- Gaidukov, L.; Nager, A.R.; Xu, S.; Penman, M.; Krieger, M. Glycine dimerization motif in the N-terminal transmembrane domain of the high density lipoprotein receptor SR-BI required for normal receptor oligomerization and lipid transport. J. Biol. Chem. 2011, 286, 18452–18464. [Google Scholar] [CrossRef]
- Sahoo, D.; Darlington, Y.F.; Pop, D.; Williams, D.L.; Connelly, M.A. Scavenger receptor class B Type I (SR-BI) assembles into detergent-sensitive dimers and tetramers. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2007, 1771, 807–817. [Google Scholar] [CrossRef]
- Marques, P.E.; Nyegaard, S.; Collins, R.F.; Troise, F.; Freeman, S.A.; Trimble, W.S.; Grinstein, S. Multimerization and Retention of the Scavenger Receptor SR-B1 in the Plasma Membrane. Dev. Cell 2019, 50, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Rodrigueza, W.V.; Thuahnai, S.T.; Temel, R.E.; Lund-Katz, S.; Phillips, M.C.; Williams, D.L. Mechanism of scavenger receptor class B type I-mediated selective uptake of cholesteryl esters from high density lipoprotein to adrenal cells. J. Biol. Chem. 1999, 274, 20344–20350. [Google Scholar] [CrossRef]
- Neculai, D.; Schwake, M.; Ravichandran, M.; Zunke, F.; Collins, R.F.; Peters, J.; Neculai, M.; Plumb, J.; Loppnau, P.; Pizarro, J.C.; et al. Structure of LIMP-2 provides functional insights with implications for SR-BI and CD36. Nature 2013, 504, 172–176. [Google Scholar] [CrossRef]
- Yancey, P.G.; De La Llera-Moya, M.; Swarnakar, S.; Monzo, P.; Klein, S.M.; Connelly, M.A.; Johnson, W.J.; Williams, D.L.; Rothblat, G.H. High density lipoprotein phospholipid composition is a major determinant of the bi-directional flux and net movement of cellular free cholesterol mediated by scavenger receptor BI. J. Biol. Chem. 2000, 275, 36596–36604. [Google Scholar] [CrossRef] [PubMed]
- Mineo, C. Lipoprotein Receptor Signaling in Atherosclerosis. Cardiovasc. Res. 2020, 116, 1254–1274. [Google Scholar] [CrossRef] [PubMed]
- Reaven, E.; Chen, Y.D.I.; Spicher, M.; Azhar, S. Morphological evidence that high density lipoproteins are not internalized by steroid-producing cells during in situ organ perfusion. J. Clin. Investig. 1984, 74, 1384–1397. [Google Scholar] [CrossRef] [PubMed]
- Oram, J.F.; Lawn, R.M.; Garvin, M.R.; Wade, D.P. ABCA1 is the cAMP-inducible apolipoprotein receptor that mediates cholesterol secretion from macrophages. J. Biol. Chem. 2000, 275, 34508–34511. [Google Scholar] [CrossRef]
- Sankaramarayanan, S.; Oram, J.F.; Asztalos, B.F.; Vaughan, A.M.; Lund-Katz, S.; Adorni, M.P.; Phillips, M.C.; Rothblat, G.H. Effects of acceptor composition and mechanism of ABCG1-mediated cellular free cholesterol efflux. J. Lipid Res. 2009, 50, 275–284. [Google Scholar] [CrossRef]
- Lim, H.Y.; Thiam, C.H.; Yeo, K.P.; Bisoendial, R.; Hii, C.S.; McGrath, K.C.Y.; Tan, K.W.; Heather, A.; Alexander, J.S.J.; Angeli, V. Lymphatic vessels are essential for the removal of cholesterol from peripheral tissues by SR-BI-Mediated transport of HDL. Cell Metab. 2013, 17, 671–684. [Google Scholar] [CrossRef]
- McGrath, K.C.Y.; Li, X.H.; Puranik, R.; Liong, E.C.; Tan, J.T.M.; Dy, V.M.; Dibartolo, B.A.; Barter, P.J.; Rye, K.A.; Heather, A.K. Role of 3β-hydroxysteroid-Δ24 reductase in mediating antiinflammatory effects of high-density lipoproteins in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 877–882. [Google Scholar] [CrossRef]
- Vergeer, M.; Korporaal, S.J.A.; Franssen, R.; Meurs, I.; Out, R.; Hovingh, G.K.; Hoekstra, M.; Sierts, J.A.; Dallinga-Thie, G.M.; Motazacker, M.M.; et al. Genetic Variant of the Scavenger Receptor BI in Humans. N. Engl. J. Med. 2011, 364, 136–145. [Google Scholar] [CrossRef]
- Yancey, P.G.; Jerome, W.G.; Yu, H.; Griffin, E.E.; Cox, B.E.; Babaev, V.R.; Fazio, S.; Linton, M.F. Severely altered cholesterol homeostasis in macrophages lacking apoE and SR-BI. J. Lipid Res. 2007, 48, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Van Eck, M.; Bos, I.S.T.; Hildebrand, R.B.; Van Rij, B.T.; Van Berkel, T.J.C. Dual role for scavenger receptor class B, type I on bone marrow-derived cells in atherosclerotic lesion development. Am. J. Pathol. 2004, 165, 785–794. [Google Scholar] [CrossRef]
- Huang, L.; Chambliss, K.L.; Gao, X.; Yuhanna, I.S.; Behling-Kelly, E.; Bergaya, S.; Ahmed, M.; Michaely, P.; Luby-Phelps, K.; Darehshouri, A.; et al. SR-B1 drives endothelial cell LDL transcytosis via DOCK4 to promote atherosclerosis. Nature 2019, 569, 565–569. [Google Scholar] [CrossRef]
- Yu, L.; Dai, Y.; Mineo, C. Novel Functions of Endothelial Scavenger Receptor Class B Type I. Curr. Atheroscler. Rep. 2021, 23. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Yancey, P.G.; Babaev, V.R.; Blakemore, J.L.; Zhang, Y.; Ding, L.; Fazio, S.; Linton, M.F. Macrophage SR-BI mediates efferocytosis via Src/PI3K/Rac1 signaling and reduces atherosclerotic lesion necrosis. J. Lipid Res. 2015, 56, 1449–1460. [Google Scholar] [CrossRef] [PubMed]
- Mineo, C.; Shaul, P.W. Novel biological functions of high-density lipoprotein cholesterol. Circ. Res. 2012, 111, 1079–1090. [Google Scholar] [CrossRef]
- Dole, V.S.; Matuskova, J.; Vasile, E.; Yesilaltay, A.; Bergmeier, W.; Bernimoulin, M.; Wagner, D.D.; Krieger, M. Thrombocytopenia and platelet abnormalities in high-density lipoprotein receptor-deficient mice. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1111–1116. [Google Scholar] [CrossRef]
- Korporaal, S.J.A.; Meurs, I.; Hauer, A.D.; Hildebrand, R.B.; Hoekstra, M.; Ten Cate, H.; Praticò, D.; Akkerman, J.W.N.; Van Berkel, T.J.C.; Kuiper, J.; et al. Deletion of the high-density lipoprotein receptor scavenger receptor BI in mice modulates thrombosis susceptibility and indirectly affects platelet function by elevation of plasma free cholesterol. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 34–42. [Google Scholar] [CrossRef]
- Barter, P.J.; Brewer, H.B.; Chapman, M.J.; Hennekens, C.H.; Rader, D.J.; Tall, A.R. Cholesteryl ester transfer protein: A novel target for raising HDL and inhibiting atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 160–167. [Google Scholar] [CrossRef]
- Tai, E.S.; Ordovas, J.M.; Carmena-Ramon, R.; Real, J.; Corella, D.; Ascaso, J.; Carmena, R. Polymorphisms at the SRBI locus are associated with lipoprotein levels in subjects with heterozygous familial hypercholesterolemia. Clin. Genet. 2003, 63, 53–58. [Google Scholar] [CrossRef]
- Tsutsumi, K.; Hagi, A.; Inoue, Y. The relationship between plasma high density lipoprotein cholesterol levels and cholesteryl ester transfer protein activity in six species of healthy experimental animals. Biol. Pharm. Bull. 2001, 24, 579–581. [Google Scholar] [CrossRef]
- Rigotti, A.; Trigatti, B.L.; Penman, M.; Rayburn, H.; Herz, J.; Krieger, M. A targeted mutation in the murine gene encoding the high density lipoprotein (HDL) receptor scavenger receptor class B type I reveals its key role in HDL metabolism. Proc. Natl. Acad. Sci. USA 1997, 94, 12610–12615. [Google Scholar] [CrossRef] [PubMed]
- Van Eck, M.; Twisk, J.; Hoekstra, M.; Van Rij, B.T.; Van der Lans, C.A.C.; Bos, I.S.T.; Kruijt, J.K.; Kuipers, F.; Van Berkel, T.J.C. Differential effects of scavenger receptor BI deficiency on lipid metabolism in cells of the arterial wall and in the liver. J. Biol. Chem. 2003, 278, 23699–23705. [Google Scholar] [CrossRef]
- Trigatti, B.L.; Krieger, M.; Rigotti, A. Influence of the HDL receptor SR-BI on lipoprotein metabolism and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1732–1738. [Google Scholar] [CrossRef] [PubMed]
- Kozarsky, K.F.; Donahee, M.H.; Glick, J.M.; Krieger, M.; Rader, D.J. Gene transfer and hepatic overexpression of the HDL receptor SR-BI reduces atherosclerosis in the cholesterol-fed LDL receptor-deficient mouse. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Ueda, Y.; Royer, L.; Gong, E.; Zhang, J.; Cooper, P.N.; Francone, O.; Rubin, E.M. Lower plasma levels and accelerated clearance of high density lipoprotein (HDL) and non-HDL cholesterol in scavenger receptor class B type I transgenic mice. J. Biol. Chem. 1999, 274, 7165–7171. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, R.B.; Lammers, B.; Meurs, I.; Korporaal, S.J.A.; De Haan, W.; Zhao, Y.; Kruijt, J.K.; Praticò, D.; Schimmel, A.W.M.; Holleboom, A.G.; et al. Restoration of high-density lipoprotein levels by cholesteryl ester transfer protein expression in scavenger receptor class B Type i (SR-BI) knockout mice does not normalize pathologies associated with SR-BI deficiency. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1439–1445. [Google Scholar] [CrossRef][Green Version]
- Ji, Y.; Wang, N.; Ramakrishnan, R.; Sehayek, E.; Huszar, D.; Breslow, J.L.; Tall, A.R. Hepatic scavenger receptor BI promotes rapid clearance of high density lipoprotein free cholesterol and its transport into bile. J. Biol. Chem. 1999, 274, 33398–33402. [Google Scholar] [CrossRef]
- Mineo, C.; Shaul, P.W. Role of High-Density Lipoprotein and Scavenger Receptor B Type I in the Promotion of Endothelial Repair. Trends Cardiovasc. Med. 2007, 17, 156–161. [Google Scholar] [CrossRef]
- Varban, M.L.; Rinninger, F.; Wang, N.; Fairchild-Huntress, V.; Dunmore, J.H.; Fang, Q.; Gosselin, M.L.; Dixon, K.L.; Deeds, J.D.; Acton, S.L.; et al. Targeted mutation reveals a central role for SR-BI in hepatic selective uptake of high density lipoprotein cholesterol. Proc. Natl. Acad. Sci. USA 1998, 95, 4619–4624. [Google Scholar] [CrossRef]
- Huby, T.; Doucet, C.; Dachet, C.; Ouzilleau, B.; Ueda, Y.; Afzal, V.; Rubin, E.; Chapman, M.J.; Lesnik, P. Knockdown expression and hepatic deficiency reveal an atheroprotective role for SR-BI in liver and peripheral tissues. J. Clin. Investig. 2006, 116, 2767–2776. [Google Scholar] [CrossRef]
- Huszar, D.; Varban, M.L.; Rinninger, F.; Feeley, R.; Arai, T.; Fairchild-Huntress, V.; Donovan, M.J.; Tall, A.R. Increased LDL cholesterol and atherosclerosis in LDL receptor-deficient mice with attenuated expression of scavenger receptor B1. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1068–1073. [Google Scholar] [CrossRef] [PubMed]
- Covey, S.D.; Krieger, M.; Wang, W.; Penman, M.; Trigatti, B.L. Scavenger receptor class B type I-mediated protection against atherosclerosis in LDL receptor-negative mice involves its expression in bone marrow-derived cells. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1589–1594. [Google Scholar] [CrossRef] [PubMed]
- Braun, A.; Trigatti, B.L.; Post, M.J.; Sato, K.; Simons, M.; Edelberg, J.M.; Rosenberg, R.D.; Schrenzel, M.; Krieger, M. Loss of SR-BI expression leads to the early onset of occlusive atherosclerotic coronary artery disease, spontaneous myocardial infarctions, severe cardiac dysfunction, and premature death in apolipoprotein E-deficient mice. Circ. Res. 2002, 90, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Arai, T.; Wang, N.; Bezouevski, M.; Welch, C.; Tall, A.R. Decreased atherosclerosis in heterozygous low density lipoprotein receptor-deficient mice expressing the scavenger receptor BI transgene. J. Biol. Chem. 1999, 274, 2366–2371. [Google Scholar] [CrossRef] [PubMed]
- Galle-Treger, L.; Moreau, M.; Ballaire, R.; Poupel, L.; Huby, T.; Sasso, E.; Troise, F.; Poti, F.; Lesnik, P.; Le Goff, W.; et al. Targeted invalidation of SR-B1 in macrophages reduces macrophage apoptosis and accelerates atherosclerosis. Cardiovasc. Res. 2020, 116, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Pennings, M.; Hildebrand, R.B.; Ye, D.; Calpe-Berdiel, L.; Out, R.; Kjerrulf, M.; Hurt-Camejo, E.; Groen, A.K.; Hoekstra, M.; et al. Enhanced foam cell formation, atherosclerotic lesion development, and inflammation by combined deletion of ABCA1 and SR-BI in bone marrow-derived cells in LDL receptor knockout mice on western-type diet. Circ. Res. 2010, 107, e20–e31. [Google Scholar] [CrossRef]
- Vaisman, B.L.; Vishnyakova, T.G.; Freeman, L.A.; Amar, M.J.; Demosky, S.J.; Liu, C.; Stonik, J.A.; Sampson, M.L.; Pryor, M.; Bocharov, A.V.; et al. Endothelial Expression of Scavenger Receptor Class B, Type I Protects against Development of Atherosclerosis in Mice. Biomed Res. Int. 2015, 2015, 607120. [Google Scholar] [CrossRef]
- Acton, S.; Osgood, D.; Donoghue, M.; Corella, D.; Pocovi, M.; Cenarro, A.; Mozas, P.; Keilty, J.; Squazzo, S.; Woolf, E.A.; et al. Association of polymorphisms at the SR-BI gene locus with plasma lipid levels and body mass index in a white population. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 1734–1743. [Google Scholar] [CrossRef]
- Richard, E.; Von Muhlen, D.; Barrett-Connor, E.; Alcaraz, J.; Davis, R.; McCarthy, J.J. Modification of the effects of estrogen therapy on HDL cholesterol levels by polymorphisms of the HDL-C receptor, SR-BI: The Rancho Bernardo Study. Atherosclerosis 2005, 180, 255–262. [Google Scholar] [CrossRef]
- Roberts, C.G.P.; Shen, H.; Mitchell, B.D.; Damcott, C.M.; Shuldiner, A.R.; Rodriguez, A. Variants in scavenger receptor class B type I gene are associated with HDL cholesterol levels in younger women. Hum. Hered. 2007, 64, 107–113. [Google Scholar] [CrossRef]
- Rodríguez-Esparragón, F.; Rodríguez-Pérez, J.C.; Hernández-Trujillo, Y.; Macías-Reyes, A.; Medina, A.; Caballero, A.; Ferrario, C.M. Allelic variants of the human scavenger receptor class B type 1 and paraoxonase 1 on coronary heart disease: Genotype-phenotype correlations. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 854–860. [Google Scholar] [CrossRef]
- De Andrade, F.M.; Fiegenbaum, M.; De Almeida, S.; Hutz, M.H. Influência de combinações genéticas nos níveis de HDL-C em uma população do sul do Brasil. Arq. Bras. Cardiol. 2010, 95, 430–435. [Google Scholar] [CrossRef]
- Osgood, D.; Corella, D.; Demissie, S.; Cupples, L.A.; Wilson, P.W.F.; Meigs, J.B.; Schaefer, E.J.; Coltell, O.; Ordovas, J.M. Genetic variation at the scavenger receptor class B type I gene locus determines plasma lipoprotein concentrations and particle size and interacts with type 2 diabetes: The Framingham study. J. Clin. Endocrinol. Metab. 2003, 88, 2869–2879. [Google Scholar] [CrossRef]
- Morabia, A.; Ross, B.M.; Costanza, M.C.; Cayanis, E.; Flaherty, M.S.; Alvin, G.B.; Das, K.; James, R.; Yang, A.S.; Evagrafov, O.; et al. Population-based study of SR-BI genetic variation and lipid profile. Atherosclerosis 2004, 175, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Hsu, L.A.; Ko, Y.L.; Wu, S.; Teng, M.S.; Peng, T.Y.; Chen, C.F.; Chen, C.F.; Lee, Y.S. Association between a novel 11-base pair deletion mutation in the promoter region of the scavenger receptor class B type I gene and plasma HDL cholesterol levels in Taiwanese Chinese. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1869–1874. [Google Scholar] [CrossRef] [PubMed]
- Rejeb, J.; Omezzine, A.; Boumaiza, I.; Rebhi, L.; Kacem, S.; Rejeb, N.B.; Nabli, N.; Abdelaziz, A.B.; Boughzala, E.; Bouslama, A. Association of three polymorphisms of scavenger receptor class BI gene (exon8, exon1, intron5) with coronary stenosis in a coronary Tunisian population. Gene 2012, 511, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Stanislovaitiene, D.; Lesauskaite, V.; Zaliuniene, D.; Smalinskiene, A.; Gustiene, O.; Zaliaduonyte-Peksiene, D.; Tamosiunas, A.; Luksiene, D.; Petkeviciene, J.; Zaliunas, R. SCARB1 single nucleotide polymorphism (rs5888) is associated with serum lipid profile and myocardial infarction in an age- and gender-dependent manner. Lipids Health Dis. 2013, 12, 24. [Google Scholar] [CrossRef] [PubMed]
- Niemsiri, V.; Wang, X.; Pirim, D.; Radwan, Z.H.; Hokanson, J.E.; Hamman, R.F.; Barmada, M.M.; Demirci, F.Y.; Kamboh, M.I. Impact of genetic variants in human scavenger receptor class B type i (SCARB1) on plasma lipid traits. Circ. Cardiovasc. Genet. 2014, 7, 838–847. [Google Scholar] [CrossRef][Green Version]
- Durst, R.; Colombo, R.; Shpitzen, S.; Avi, L.B.; Friedlander, Y.; Wexler, R.; Raal, F.J.; Marais, D.A.; Defesche, J.C.; Mandelshtam, M.Y.; et al. Recent origin and spread of a common lithuanian mutation, G197del LDLR, causing familial hypercholesterolemia: Positive selection is not always necessary to account for disease incidence among Ashkenazi Jews. Am. J. Hum. Genet. 2001, 68, 1172–1188. [Google Scholar] [CrossRef][Green Version]
- Goodarzynejad, H.; Boroumand, M.; Behmanesh, M.; Ziaee, S.; Jalali, A. The rs5888 single nucleotide polymorphism in scavenger receptor class B type 1 (SCARB1) gene and the risk of premature coronary artery disease: A case-control study. Lipids Health Dis. 2016, 15, 7. [Google Scholar] [CrossRef]
- Manichaikul, A.; Naj, A.C.; Herrington, D.; Post, W.; Rich, S.S.; Rodriguez, A. Association of SCARB1 variants with subclinical atherosclerosis and incident cardiovascular disease: The multi-ethnic study of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1991–1999. [Google Scholar] [CrossRef]
- Pérez-Martínez, P.; López-Miranda, J.; Ordovás, J.M.; Bellido, C.; Marín, C.; Gómez, P.; Paniagua, J.A.; Moreno, J.A.; Fuentes, F.; Pérez-Jiménez, F. Postprandial lipemia is modified by the presence of the polymorphism present in the exon 1 variant at the SR-BI gene locus. J. Mol. Endocrinol. 2004, 32, 237–245. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tanaka, T.; Delgado-Lista, J.; Lopez-Miranda, J.; Perez-Jimenez, F.; Marin, C.; Perez-Martinez, P.; Gomez, P.; Ordovas, J.M. Scavenger Receptor Class B Type I (SCARB1) c.1119C>T Polymorphism Affects Postprandial Triglyceride Metabolism in Men. J. Nutr. 2007, 137, 578–582. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, Y.; Ordovas, J.M.; Gao, G.; Province, M.; Straka, R.J.; Tsai, M.Y.; Lai, C.Q.; Zhang, K.; Borecki, I.; Hixson, J.E.; et al. The SCARB1 gene is associated with lipid response to dietary and pharmacological interventions. J. Hum. Genet. 2008, 53, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Cerda, Á.; Genvigir, F.D.V.; Arazi, S.S.; Hirata, M.H.; Dorea, E.L.; Bernik, M.M.S.; Bertolami, M.C.; Faludi, A.A.; Hirata, R.D.C. Influence of SCARB1 polymorphisms on serum lipids of hypercholesterolemic individuals treated with atorvastatin. Clin. Chim. Acta 2010, 411, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Constantineau, J.; Greason, E.; West, M.; Filbin, M.; Kieft, J.S.; Carletti, M.Z.; Christenson, L.K.; Rodriguez, A. A synonymous variant in scavenger receptor, class B, type I gene is associated with lower SR-BI protein expression and function. Atherosclerosis 2010, 210, 177–182. [Google Scholar] [CrossRef]
- Yang, X.; Sethi, A.; Yanek, L.R.; Knapper, C.; Nordestgaard, B.G.; Tybjærg-Hansen, A.; Becker, D.M.; Mathias, R.A.; Remaley, A.T.; Becker, L.C. SCARB1 Gene Variants Are Associated with the Phenotype of Combined High High-Density Lipoprotein Cholesterol and High Lipoprotein (a). Circ. Cardiovasc. Genet. 2016, 9, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Naj, A.C.; West, M.; Rich, S.S.; Post, W.; Linda Kao, W.H.; Wasserman, B.A.; Herrington, D.M.; Rodriguez, A. Association of scavenger receptor class B type I polymorphisms with subclinical atherosclerosis: The multi-ethnic study of atherosclerosis. Circ. Cardiovasc. Genet. 2010, 3, 47–52. [Google Scholar] [CrossRef]
- West, M.; Greason, E.; Kolmakova, A.; Jahangiri, A.; Asztalos, B.; Pollin, T.I.; Rodriguez, A. Scavenger receptor class B type i protein as an independent predictor of high-density lipoprotein cholesterol levels in subjects with hyperalphalipoproteinemia. J. Clin. Endocrinol. Metab. 2009, 94, 1451–1457. [Google Scholar] [CrossRef] [PubMed]
- Zeng, T.T.; Tang, D.J.; Ye, Y.X.; Su, J.; Jiang, H. Influence of SCARB1 gene SNPs on serum lipid levels and susceptibility to coronary heart disease and cerebral infarction in a Chinese population. Gene 2017, 626, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Teslovich, T.M.; Musunuru, K.; Smith, A.V.; Edmondson, A.C.; Stylianou, I.M.; Koseki, M.; Pirruccello, J.P.; Ripatti, S.; Chasman, D.I.; Willer, C.J.; et al. Biological, clinical and population relevance of 95 loci for blood lipids. Nature 2010, 466, 707–713. [Google Scholar] [CrossRef]
- Chiba-Falek, O.; Nichols, M.; Suchindran, S.; Guyton, J.; Ginsburg, G.S.; Barrett-Connor, E.; McCarthy, J.J. Impact of gene variants on sex-specific regulation of human scavenger receptor class B type 1 (SR-BI) expression in liver and association with lipid levels in a population-based study. BMC Med. Genet. 2010, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Zanoni, P.; Khetarpal, S.A.; Larach, D.B.; Hancock-Cerutti, W.F.; Millar, J.S.; Cuchel, M.; DerOhannessian, S.; Kontush, A.; Surendran, P.; Saleheen, D.; et al. Rare variant in scavenger receptor BI raises HDL cholesterol and increases risk of coronary heart disease. Science (80-.) 2016, 351, 1166–1171. [Google Scholar] [CrossRef]
- Samadi, S.; Farjami, Z.; Hosseini, Z.S.; Ferns, G.A.; Mohammadpour, A.H.; Tayefi, M.; Fal-Soleiman, H.; Moohebati, M.; Ghayour-Mobarhan, M.; Esmaily, H.; et al. Rare P376L variant in the SR-BI gene associates with HDL dysfunction and risk of cardiovascular disease. Clin. Biochem. 2019, 73, 44–49. [Google Scholar] [CrossRef]
- May, S.C.; Dron, J.S.; Hegele, R.A.; Sahoo, D. Human Variant of Scavenger Receptor BI (R174C) Exhibits Impaired Cholesterol Transport Functions. J. Lipid Res. 2021, 62, 100045. [Google Scholar] [CrossRef] [PubMed]
- Brunham, L.R.; Tietjen, I.; Bochem, A.E.; Singaraja, R.R.; Franchini, P.L.; Radomski, C.; Mattice, M.; Legendre, A.; Hovingh, G.K.; Kastelein, J.J.P.; et al. Novel mutations in scavenger receptor BI associated with high HDL cholesterol in humans. Clin. Genet. 2011, 79, 575–581. [Google Scholar] [CrossRef]
- Ljunggren, S.A.; Levels, J.H.M.; Hovingh, K.; Holleboom, A.G.; Vergeer, M.; Argyri, L.; Gkolfinopoulou, C.; Chroni, A.; Sierts, J.A.; Kastelein, J.J.; et al. Lipoprotein profiles in human heterozygote carriers of a functional mutation P297S in scavenger receptor class B1. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2015, 1851, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, A.C.; Sahoo, D. Functional Characterization of Newly-Discovered Mutations in Human SR-BI. PLoS ONE 2012, 7, e45660. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Trigatti, B.L.; Hegele, R.A. Rare genetic variants and high-density lipoprotein. Arterioscler. Thromb. Vasc. Biol. 2016, 36, e53–e55. [Google Scholar] [CrossRef][Green Version]
- Fuchs, M.; Ivandic, B.; Müller, O.; Schalla, C.; Scheibner, J.; Bartsch, P.; Stange, E.F. Biliary cholesterol hypersecretion in gallstone-susceptible mice is associated with hepatic up-regulation of the high-density lipoprotein receptor SRBI. Hepatology 2001, 33, 1451–1459. [Google Scholar] [CrossRef]
- Webb, T.R.; Erdmann, J.; Stirrups, K.E.; Stitziel, N.O.; Masca, N.G.D.; Jansen, H.; Kanoni, S.; Nelson, C.P.; Ferrario, P.G.; König, I.R.; et al. Systematic Evaluation of Pleiotropy Identifies 6 Further Loci Associated With Coronary Artery Disease. J. Am. Coll. Cardiol. 2017, 69, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Howson, J.M.M.; Zhao, W.; Barnes, D.R.; Ho, W.K.; Young, R.; Paul, D.S.; Waite, L.L.; Freitag, D.F.; Fauman, E.B.; Salfati, E.L.; et al. Fifteen new risk loci for coronary artery disease highlight arterial-wall-specific mechanisms. Nat. Genet. 2017, 49, 1113–1119. [Google Scholar] [CrossRef]
- Vitali, C.; Khetarpal, S.A.; Rader, D.J. HDL Cholesterol Metabolism and the Risk of CHD: New Insights from Human Genetics. Curr. Cardiol. Rep. 2017, 19, 132. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A. High HDL-Cholesterol Paradox: SCARB1-LAG3-HDL Axis. Curr. Atheroscler. Rep. 2021, 23, 5. [Google Scholar] [CrossRef] [PubMed]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; John Chapman, M.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; Pedersen, T.R.; et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias. Eur. Heart J. 2016, 37, 2999–3058l. [Google Scholar] [CrossRef] [PubMed]
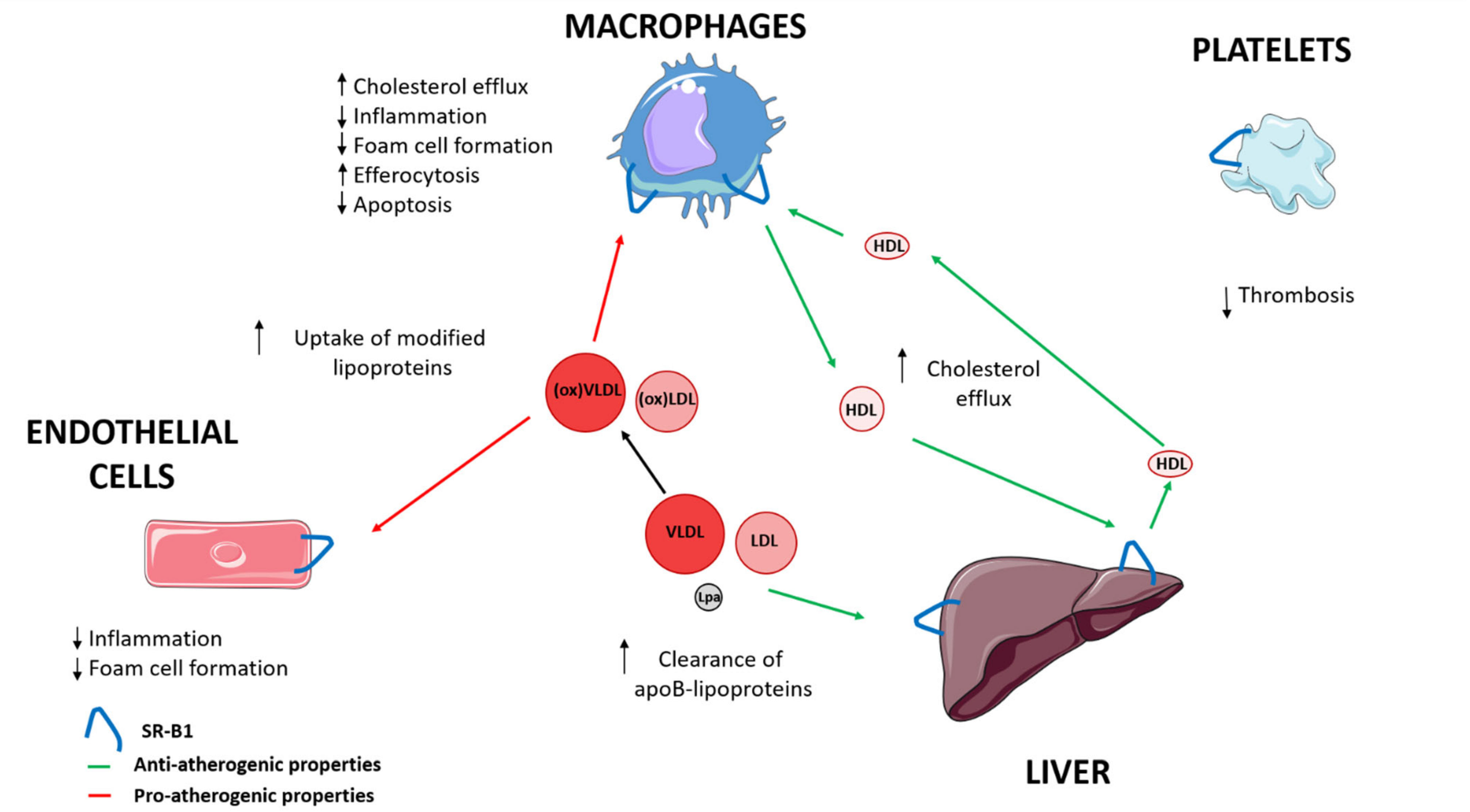
| Anti-Atherogenic Properties | Pro-Atherogenic Properties | |
|---|---|---|
| Liver | ↑ Cholesterol efflux [16,17,18,19] | |
| ↑ Clearance LDL, VLDL and Lp(a) [16,17,18,19] | ||
| Macrophages | ↑ Cholesterol efflux [16,17,18,19] | ↑ Uptake modified lipoproteins [43,44,45] |
| ↓ Inflammation [39] | ||
| ↓ Foam cell formation [40] | ||
| ↑ Efferocytosis [47] | ||
| ↓ Apoptosis [46] | ||
| Endothelial cells | ↓ Inflammation [40] | ↑ Uptake modified lipoproteins [43,44,45] |
| ↓ Foam cell formation [41] | ||
| Platelets | ↓ Thrombosis [48,49] |
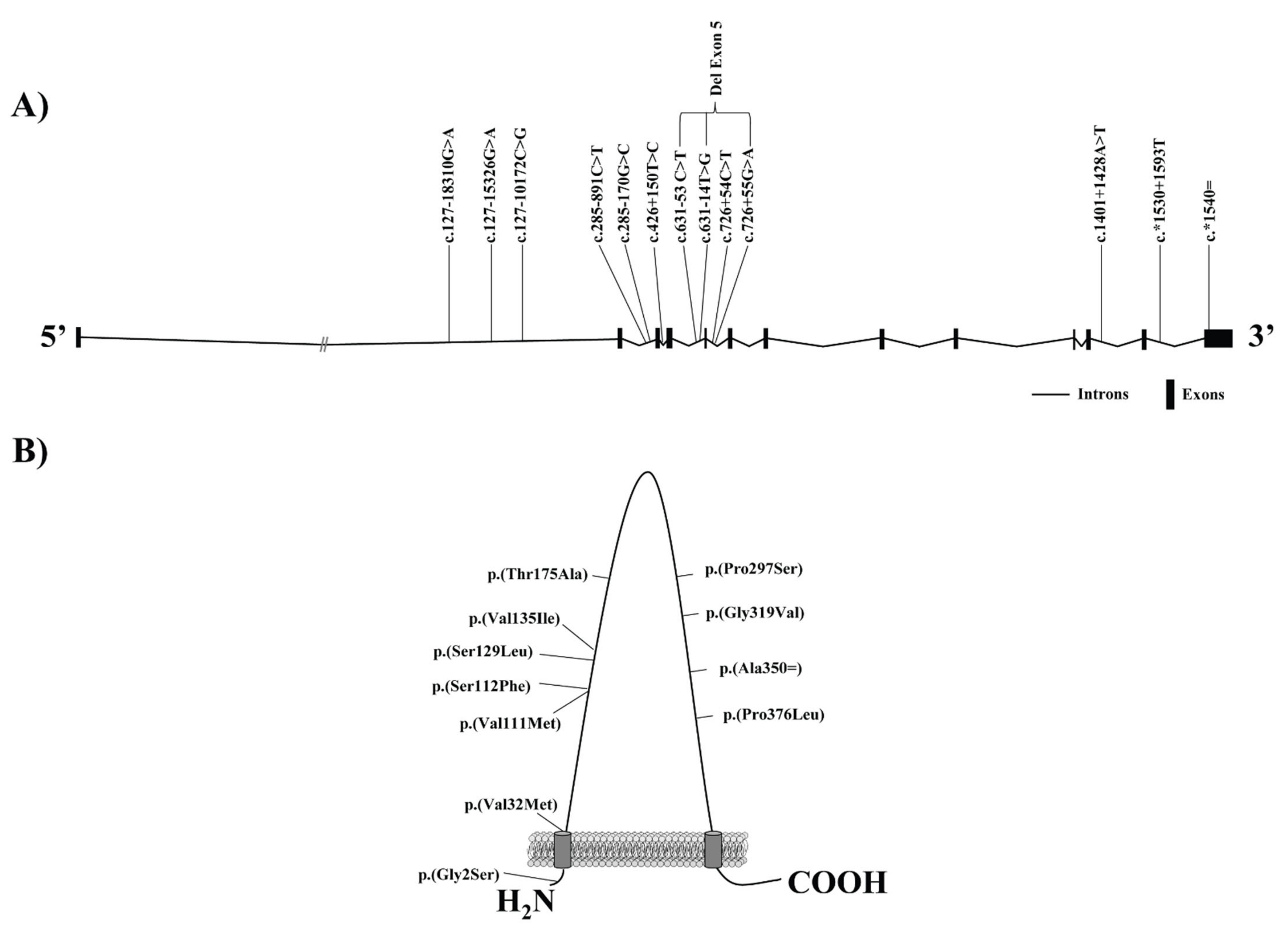
| DNA Variant † | Protein Variant | Study Subjects | rsID | Exon/Intron | Variant Effect | Reference |
|---|---|---|---|---|---|---|
| c.4G > A | p.(Gly2Ser) | Spanish Caucasians | 4238001 | Exon 1 | ↑ HDLc in men | Acton et al., 1999 * [70] |
| ↓ LDLc levels in men | ||||||
| c.1050T > G | p.(Ala350Ala) | 5888 | Exon 8 | ↓ LDLc values in women | ||
| 4c.795 + 54C > T | NA | NA | Intron 5 | ↑ body mass index in women | ||
| ↓ TG levels in men | ||||||
| c.-140_-150del | NA | Taiwanese Chinese population | NA | NA | ↑ levels of HDLc | Hsu et al., 2003 [77] |
| ↓ promotor activity (in vitro) | ||||||
| c.403G > A | p.(Val135Ile) | Amish population | 5891 | Exon 3 | ↑ levels of HDLc in women | Roberts et al., 2007 [72] |
| US non-Hispanic white with extreme HDL-C level | ↑ apo B levels | Niemsiri et al., 2014 [79] | ||||
| c.127-18310G > A | NA | US non-Hispanic white with extreme HDL-C level | 11057844 | Intron 1 | Associated to HDLc | Niemsiri et al., 2014 [80] |
| c.*1530 + 1593T | NA | 701106 | Intron 12 | |||
| c.*1540 = | NA | 838880 | 3 prime UTR | |||
| c.285–891C > T | NA | US non-Hispanic white with extreme HDL-C level | 2343394 | Intron 2 | ↑apo B levels | Niemsiri et al., 2014 [80] |
| Multiethnic groups | Associated with carotid intima-media thickness | Naj et al., 2010 [90] | ||||
| Related to CHD | ||||||
| ↓ SCARB1 protein levels | West et al., 2009 [91] | |||||
| c.127–15326G > A | NA | Multiethnic groups | 10744182 | Intron 1 | ↑ common carotid intima-media thickness | Naj et al., 2010 [90] |
| Hen Chinese population | No association | Zeng et al., 2017 [92] | ||||
| c.127–10172C > G | NA | Multiethnic groups | 10846744 | Intron 1 | ↑ common carotid intima-media thickness independent of lipid levels | Naj et al., 2010 [93] |
| Hen Chinese population | ↑ HDLc levels | Zeng et al., 2017 [92] | ||||
| ↑ CHD risk | ||||||
| c.1401 + 1428A > T | NA | The suburban community of Rancho Bernardo | 838893 | Intron 11 | Related to endogenous estradiol levels, HDLc, TG, and the ratio TG:HDLc in postmenopausal Caucasian women | Chiba-Falek et al., 2010 [94] |
| Associated with ↓level of liver SR-B1 in women under the age of 45 | ||||||
| Hen Chinese population | No association | Zeng et al., 2017 [92] | ||||
| c.889C > T | p.(Pro297Ser) | Caucasian population with HDLc above the 95th percentile (Netherlands) | 387906791 | Exon 7 | ↑ HDLc levels | Vergeer et al., 2011 [41] |
| ↓ Cholesterol efflux macrophages (in vitro) | ||||||
| ↓ adrenal steroigenesis (in vitro) | ||||||
| Changes in platelet function (in vitro) | ||||||
| No alterations in carotid intima-media thickness | ||||||
| Changes in HDL, LDL and VLDL composition | ||||||
| c.335C > T | p.(Ser112Phe) | Caucasian ancestry population with high levels of HDLc | 397514572 | Exon 3 | Alterations in HDL binding (in vitro) | Brunham et al., 2011 [7] |
| c.523A > G | p.(Thr175Ala) | 187831231 | Exon 4 | Modifications in selective uptake of HDL-CE (in vitro) | ||
| Changes in the delivery of FC from cells to HDL (in vitro) | ||||||
| c.386C > T | p.(Ser129Leu) | Multiethnic population with high HDLc and high Lp(a) | 150222965 | Exon 3 | ↓CE uptake from HDL and Lp(a) (in vitro) | Yang et al., 2016 [89] |
| c.631–14T > G | delExon5 | 113910315 | Intron 4 | |||
| c.631–53 C > T c.726+55 G > A | delExon5 | 77740046 59809936 | Introns 4,5 | |||
| c.1127 C > T | p.(Pro376Leu) | European ancestry with extremely elevated HDLc levels (Ashkenazi Jews) | 74830677 | Exon 8 | Alteration in posttranscriptional processing of SR-B1 (in vitro) | Zanoni et al., 2016 [95] |
| Abolishment of selective uptake of HDL-CE (in vitro and in vivo) | ||||||
| ↑ CVD risk | ||||||
| CVD patients from MASHAD cohort | No differences found in HDLc levels | Samadi et al., 2019 [96] | ||||
| HDL lipid peroxidation | ||||||
| c.956G > T | p.(Gly319Val) | Homogenous population of Iceland | 150728540 | Exon 7 | ↑ HDLc levels | Helgadottir et al., 2018 [8] |
| c.331G > A | p.(Val111Met) | 5890 | Exon 3 | ↓ hepatic reverse cholesterol | ||
| c.94G > A | p.(Val32Met) | 771247110 | Exon 1 | No ↑ CVD risk | ||
| c.520C > T | p.(Arg174Cys) | Patients with extreme levels of HDLc (Canada) | 367669186 | Exon 4 | ↓ Cholesterol transport | May et al., 2021 [97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gracia-Rubio, I.; Martín, C.; Civeira, F.; Cenarro, A. SR-B1, a Key Receptor Involved in the Progression of Cardiovascular Disease: A Perspective from Mice and Human Genetic Studies. Biomedicines 2021, 9, 612. https://doi.org/10.3390/biomedicines9060612
Gracia-Rubio I, Martín C, Civeira F, Cenarro A. SR-B1, a Key Receptor Involved in the Progression of Cardiovascular Disease: A Perspective from Mice and Human Genetic Studies. Biomedicines. 2021; 9(6):612. https://doi.org/10.3390/biomedicines9060612
Chicago/Turabian StyleGracia-Rubio, Irene, César Martín, Fernando Civeira, and Ana Cenarro. 2021. "SR-B1, a Key Receptor Involved in the Progression of Cardiovascular Disease: A Perspective from Mice and Human Genetic Studies" Biomedicines 9, no. 6: 612. https://doi.org/10.3390/biomedicines9060612
APA StyleGracia-Rubio, I., Martín, C., Civeira, F., & Cenarro, A. (2021). SR-B1, a Key Receptor Involved in the Progression of Cardiovascular Disease: A Perspective from Mice and Human Genetic Studies. Biomedicines, 9(6), 612. https://doi.org/10.3390/biomedicines9060612






