Laboratory Monitoring of Direct Oral Anticoagulants (DOACs)
Abstract
1. Introduction
2. DOAC Pharmacokinetics and Pharmacodynamics
2.1. Dabigatran
2.2. Rivaroxaban
2.3. Apixaban
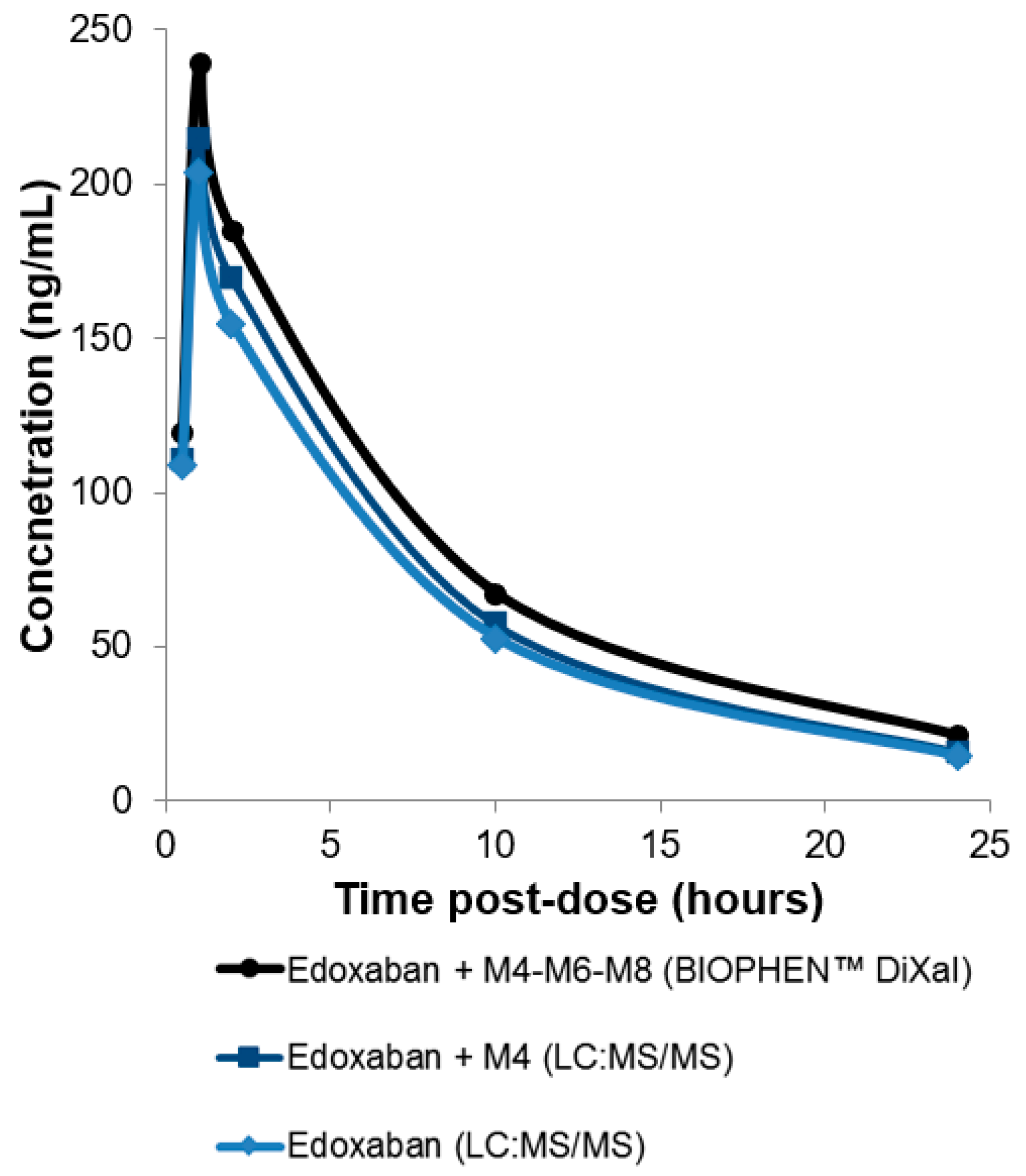
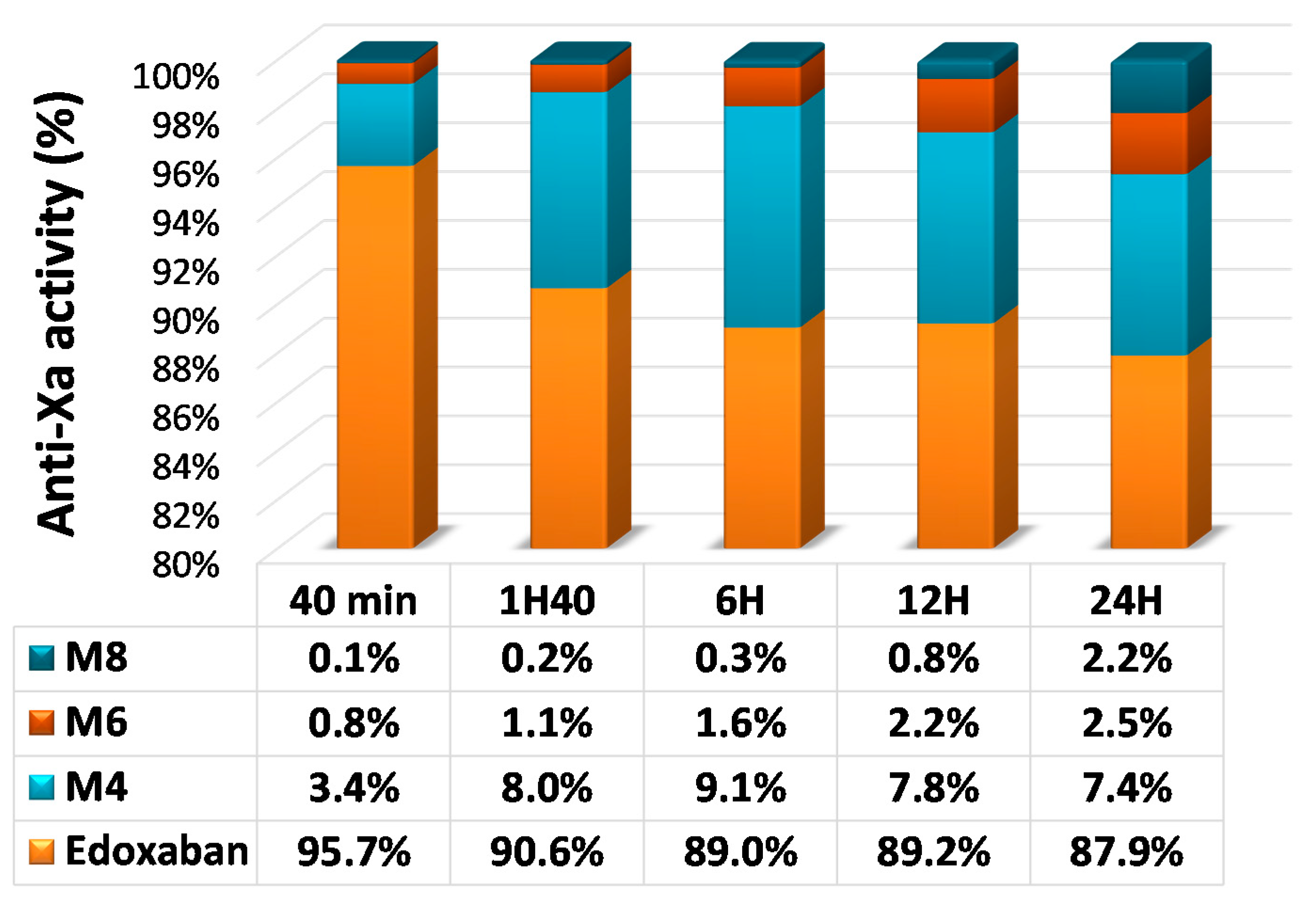
2.4. Edoxaban
2.5. Betrixaban
3. DOAC Inter-Patient Variability
3.1. Management of Patients under DOAC Therapy
3.2. Management of Patients under Multi-Drug Therapy
3.3. Pre-Operative Management of Patients under DOAC Therapy
4. DOAC Laboratory Testing
4.1. Liquid Chromatography–Mass Spectrometry (LC-MS/MS)
4.2. Routine Coagulation Screening Assays
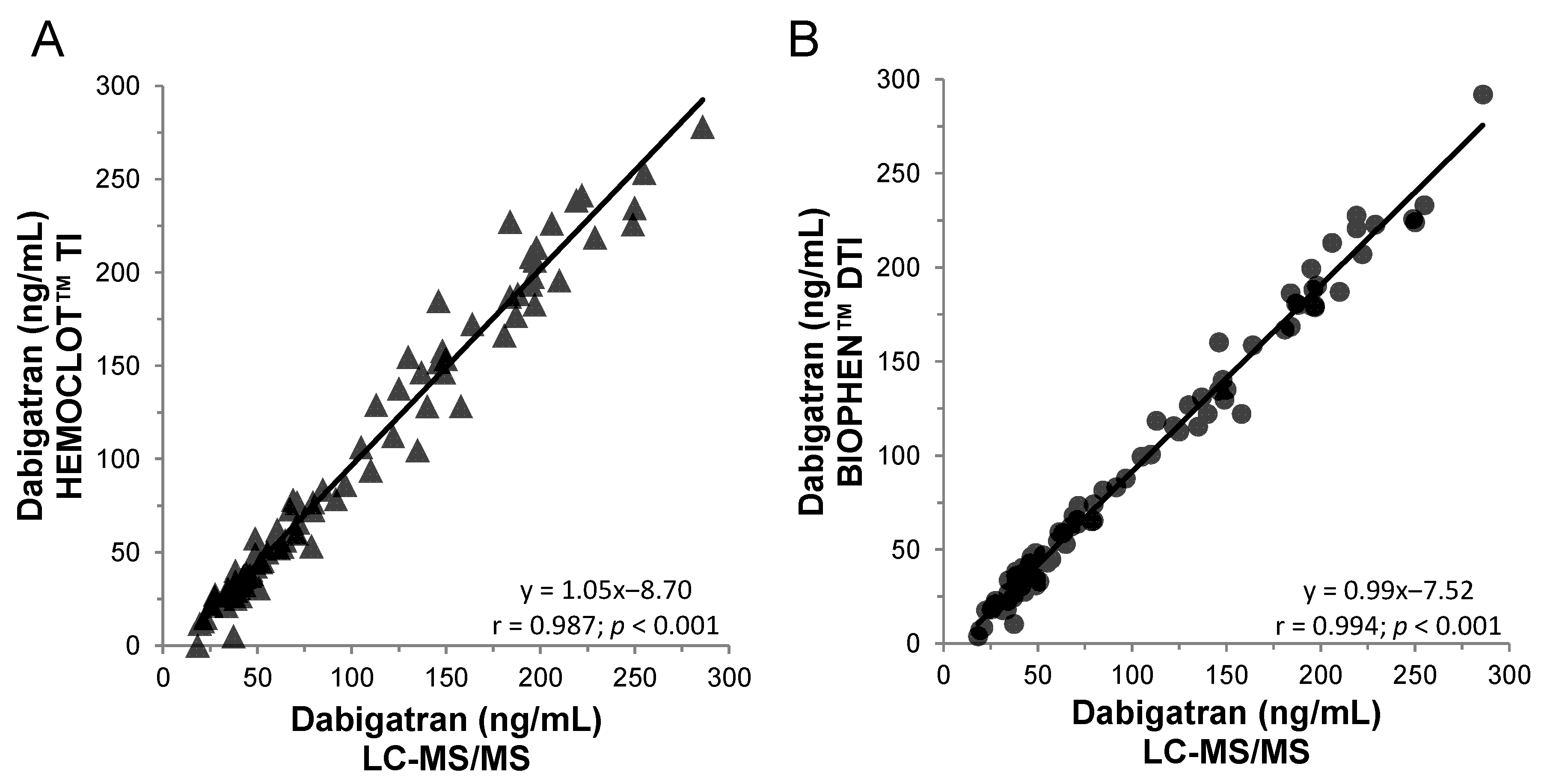
4.2.1. Direct Thrombin Inhibitors (DTIs)
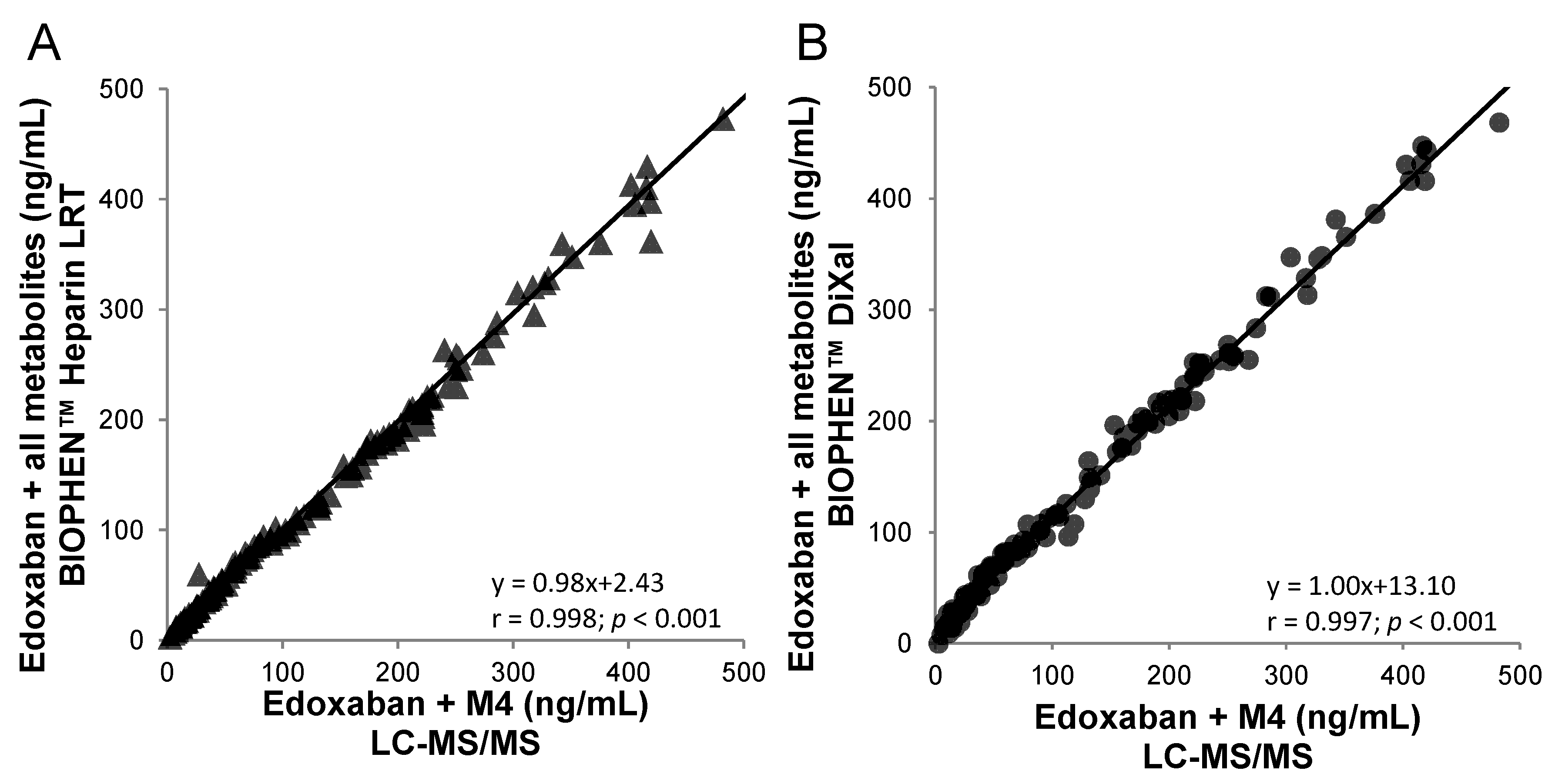
4.2.2. Direct Factor Xa Inhibitors (DiXaIs)
4.3. Quantitative Routine Assays
4.3.1. Direct Thrombin Inhibitors (DTIs)
4.3.2. Direct factor Xa Inhibitors (DiXaIs)
5. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Aronis, K.N.; Hylek, E.M. Evidence Gaps in the Era of Non-Vitamin K Oral Anticoagulants. J. Am. Heart Assoc. 2018, 7. [Google Scholar] [CrossRef]
- Kearon, C.; Akl, E.A.; Ornelas, J.; Blaivas, A.; Jimenez, D.; Bounameaux, H.; Huisman, M.; King, C.S.; Morris, T.A.; Sood, N.; et al. Antithrombotic Therapy for VTE Disease: CHEST Guideline and Expert Panel Report. Chest 2016, 149, 315–352. [Google Scholar] [CrossRef]
- Levy, J.H.; Spyropoulos, A.C.; Samama, C.M.; Douketis, J. Direct Oral Anticoagulants: New Drugs and New Concepts. JACC Cardiovasc. Interv. 2014, 7, 1333–1351. [Google Scholar] [CrossRef]
- Ruff, C.T.; Giugliano, R.P.; Braunwald, E.; Hoffman, E.B.; Deenadayalu, N.; Ezekowitz, M.D.; Camm, A.J.; Weitz, J.I.; Lewis, B.S.; Parkhomenko, A.; et al. Comparison of the Efficacy and Safety of New Oral Anticoagulants with Warfarin in Patients with Atrial Fibrillation: A Meta-Analysis of Randomised Trials. Lancet Lond. Engl. 2014, 383, 955–962. [Google Scholar] [CrossRef]
- Chai-Adisaksopha, C.; Hillis, C.; Isayama, T.; Lim, W.; Iorio, A.; Crowther, M. Mortality Outcomes in Patients Receiving Direct Oral Anticoagulants: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Thromb. Haemost. JTH 2015, 13, 2012–2020. [Google Scholar] [CrossRef]
- Hokusai-VTE Investigators; Büller, H.R.; Décousus, H.; Grosso, M.A.; Mercuri, M.; Middeldorp, S.; Prins, M.H.; Raskob, G.E.; Schellong, S.M.; Schwocho, L.; et al. Edoxaban versus Warfarin for the Treatment of Symptomatic Venous Thromboembolism. N. Engl. J. Med. 2013, 369, 1406–1415. [Google Scholar] [CrossRef]
- Connolly, S.J.; Eikelboom, J.; Dorian, P.; Hohnloser, S.H.; Gretler, D.D.; Sinha, U.; Ezekowitz, M.D. Betrixaban Compared with Warfarin in Patients with Atrial Fibrillation: Results of a Phase 2, Randomized, Dose-Ranging Study (Explore-Xa). Eur. Heart J. 2013, 34, 1498–1505. [Google Scholar] [CrossRef]
- Tripodi, A. To Measure or Not to Measure Direct Oral Anticoagulants before Surgery or Invasive Procedures: Reply. J. Thromb. Haemost. JTH 2017, 15, 202–204. [Google Scholar] [CrossRef]
- Lippi, G.; Favaloro, E.J. Recent Guidelines and Recommendations for Laboratory Assessment of the Direct Oral Anticoagulants (DOACs): Is There Consensus? Clin. Chem. Lab. Med. 2015, 53, 185–197. [Google Scholar] [CrossRef]
- Weitz, J.I.; Eikelboom, J.W. Urgent Need to Measure Effects of Direct Oral Anticoagulants. Circulation 2016, 134, 186–188. [Google Scholar] [CrossRef]
- Spyropoulos, A.C.; Al-Badri, A.; Sherwood, M.W.; Douketis, J.D. Periprocedural Management of Patients Receiving a Vitamin K Antagonist or a Direct Oral Anticoagulant Requiring an Elective Procedure or Surgery. J. Thromb. Haemost. JTH 2016, 14, 875–885. [Google Scholar] [CrossRef]
- Reilly, P.A.; Lehr, T.; Haertter, S.; Connolly, S.J.; Yusuf, S.; Eikelboom, J.W.; Ezekowitz, M.D.; Nehmiz, G.; Wang, S.; Wallentin, L.; et al. The Effect of Dabigatran Plasma Concentrations and Patient Characteristics on the Frequency of Ischemic Stroke and Major Bleeding in Atrial Fibrillation Patients: The RE-LY Trial (Randomized Evaluation of Long-Term Anticoagulation Therapy). J. Am. Coll. Cardiol. 2014, 63, 321–328. [Google Scholar] [CrossRef]
- Ruff, C.T.; Giugliano, R.P.; Braunwald, E.; Morrow, D.A.; Murphy, S.A.; Kuder, J.F.; Deenadayalu, N.; Jarolim, P.; Betcher, J.; Shi, M.; et al. Association between Edoxaban Dose, Concentration, Anti-Factor Xa Activity, and Outcomes: An Analysis of Data from the Randomised, Double-Blind ENGAGE AF-TIMI 48 Trial. Lancet Lond. Engl. 2015, 385, 2288–2295. [Google Scholar] [CrossRef]
- Wienen, W.; Stassen, J.-M.; Priepke, H.; Ries, U.J.; Hauel, N. In-Vitro Profile and Ex-Vivo Anticoagulant Activity of the Direct Thrombin Inhibitor Dabigatran and Its Orally Active Prodrug, Dabigatran Etexilate. Thromb. Haemost. 2007, 98, 155–162. [Google Scholar]
- Blech, S.; Ebner, T.; Ludwig-Schwellinger, E.; Stangier, J.; Roth, W. The Metabolism and Disposition of the Oral Direct Thrombin Inhibitor, Dabigatran, in Humans. Drug Metab. Dispos. Biol. Fate Chem. 2008, 36, 386–399. [Google Scholar] [CrossRef]
- Stangier, J. Clinical Pharmacokinetics and Pharmacodynamics of the Oral Direct Thrombin Inhibitor Dabigatran Etexilate. Clin. Pharmacokinet. 2008, 47, 285–295. [Google Scholar] [CrossRef]
- Stangier, J.; Stähle, H.; Rathgen, K.; Fuhr, R. Pharmacokinetics and Pharmacodynamics of the Direct Oral Thrombin Inhibitor Dabigatran in Healthy Elderly Subjects. Clin. Pharmacokinet. 2008, 47, 47–59. [Google Scholar] [CrossRef]
- Byon, W.; Sweeney, K.; Frost, C.; Boyd, R.A. Population Pharmacokinetics, Pharmacodynamics, and Exploratory Exposure-Response Analyses of Apixaban in Subjects Treated for Venous Thromboembolism. CPT Pharmacomet. Syst. Pharmacol. 2017, 6, 340–349. [Google Scholar] [CrossRef]
- Gosselin, R.C.; Adcock, D.M.; Bates, S.M.; Douxfils, J.; Favaloro, E.J.; Gouin-Thibault, I.; Guillermo, C.; Kawai, Y.; Lindhoff-Last, E.; Kitchen, S. International Council for Standardization in Haematology (ICSH) Recommendations for Laboratory Measurement of Direct Oral Anticoagulants. Thromb. Haemost. 2018, 118, 437–450. [Google Scholar] [CrossRef]
- Morganroth, J.; Gretler, D.D.; Hollenbach, S.J.; Lambing, J.L.; Sinha, U. Absence of QTc Prolongation with Betrixaban: A Randomized, Double-Blind, Placebo- and Positive-Controlled Thorough ECG Study. Expert Opin. Pharmacother. 2013, 14, 5–13. [Google Scholar] [CrossRef]
- Ebner, M.; Birschmann, I.; Peter, A.; Härtig, F.; Spencer, C.; Kuhn, J.; Rupp, A.; Blumenstock, G.; Zuern, C.S.; Ziemann, U.; et al. Limitations of Specific Coagulation Tests for Direct Oral Anticoagulants: A Critical Analysis. J. Am. Heart Assoc. 2018, 7, e009807. [Google Scholar] [CrossRef] [PubMed]
- Perzborn, E.; Strassburger, J.; Wilmen, A.; Pohlmann, J.; Roehrig, S.; Schlemmer, K.-H.; Straub, A. In Vitro and in Vivo Studies of the Novel Antithrombotic Agent BAY 59-7939—An Oral, Direct Factor Xa Inhibitor. J. Thromb. Haemost. JTH 2005, 3, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Mueck, W.; Borris, L.C.; Dahl, O.E.; Haas, S.; Huisman, M.V.; Kakkar, A.K.; Kälebo, P.; Muelhofer, E.; Misselwitz, F.; Eriksson, B.I. Population Pharmacokinetics and Pharmacodynamics of Once- and Twice-Daily Rivaroxaban for the Prevention of Venous Thromboembolism in Patients Undergoing Total Hip Replacement. Thromb. Haemost. 2008, 100, 453–461. [Google Scholar] [PubMed]
- Weinz, C.; Buetehorn, U.; Daehler, H.-P.; Kohlsdorfer, C.; Pleiss, U.; Sandmann, S.; Schlemmer, K.-H.; Schwarz, T.; Steinke, W. Pharmacokinetics of BAY 59-7939—An Oral, Direct Factor Xa Inhibitor—In Rats and Dogs. Xenobiotica Fate Foreign Compd. Biol. Syst. 2005, 35, 891–910. [Google Scholar] [CrossRef] [PubMed]
- Weinz, C.; Schwarz, T.; Kubitza, D.; Mueck, W.; Lang, D. Metabolism and Excretion of Rivaroxaban, an Oral, Direct Factor Xa Inhibitor, in Rats, Dogs, and Humans. Drug Metab. Dispos. Biol. Fate Chem. 2009, 37, 1056–1064. [Google Scholar] [CrossRef]
- Mueck, W.; Kubitza, D.; Becka, M. Co-Administration of Rivaroxaban with Drugs That Share Its Elimination Pathways: Pharmacokinetic Effects in Healthy Subjects. Br. J. Clin. Pharmacol. 2013, 76, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Gnoth, M.J.; Buetehorn, U.; Muenster, U.; Schwarz, T.; Sandmann, S. In Vitro and in Vivo P-Glycoprotein Transport Characteristics of Rivaroxaban. J. Pharmacol. Exp. Ther. 2011, 338, 372–380. [Google Scholar] [CrossRef]
- Jiang, X.; Crain, E.J.; Luettgen, J.M.; Schumacher, W.A.; Wong, P.C. Apixaban, an Oral Direct Factor Xa Inhibitor, Inhibits Human Clot-Bound Factor Xa Activity in Vitro. Thromb. Haemost. 2009, 101, 780–782. [Google Scholar] [CrossRef]
- Frost, C.; Nepal, S.; Wang, J.; Schuster, A.; Byon, W.; Boyd, R.A.; Yu, Z.; Shenker, A.; Barrett, Y.C.; Mosqueda-Garcia, R.; et al. Safety, Pharmacokinetics and Pharmacodynamics of Multiple Oral Doses of Apixaban, a Factor Xa Inhibitor, in Healthy Subjects. Br. J. Clin. Pharmacol. 2013, 76, 776–786. [Google Scholar] [CrossRef]
- Vakkalagadda, B.; Frost, C.; Byon, W.; Boyd, R.A.; Wang, J.; Zhang, D.; Yu, Z.; Dias, C.; Shenker, A.; LaCreta, F. Effect of Rifampin on the Pharmacokinetics of Apixaban, an Oral Direct Inhibitor of Factor Xa. Am. J. Cardiovasc. Drugs Drugs Devices Interv. 2016, 16, 119–127. [Google Scholar] [CrossRef]
- Upreti, V.V.; Wang, J.; Barrett, Y.C.; Byon, W.; Boyd, R.A.; Pursley, J.; LaCreta, F.P.; Frost, C.E. Effect of Extremes of Body Weight on the Pharmacokinetics, Pharmacodynamics, Safety and Tolerability of Apixaban in Healthy Subjects. Br. J. Clin. Pharmacol. 2013, 76, 908–916. [Google Scholar] [CrossRef]
- He, K.; Luettgen, J.M.; Zhang, D.; He, B.; Grace, J.E.; Xin, B.; Pinto, D.J.P.; Wong, P.C.; Knabb, R.M.; Lam, P.Y.S.; et al. Preclinical Pharmacokinetics and Pharmacodynamics of Apixaban, a Potent and Selective Factor Xa Inhibitor. Eur. J. Drug Metab. Pharmacokinet. 2011, 36, 129–139. [Google Scholar] [CrossRef]
- Raghavan, N.; Frost, C.E.; Yu, Z.; He, K.; Zhang, H.; Humphreys, W.G.; Pinto, D.; Chen, S.; Bonacorsi, S.; Wong, P.C.; et al. Apixaban Metabolism and Pharmacokinetics after Oral Administration to Humans. Drug Metab. Dispos. Biol. Fate Chem. 2009, 37, 74–81. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, D.; Raghavan, N.; Yao, M.; Ma, L.; Frost, C.E.; Frost, C.A.; Maxwell, B.D.; Chen, S.; He, K.; et al. In Vitro Assessment of Metabolic Drug-Drug Interaction Potential of Apixaban through Cytochrome P450 Phenotyping, Inhibition, and Induction Studies. Drug Metab. Dispos. Biol. Fate Chem. 2010, 38, 448–458. [Google Scholar] [CrossRef]
- Zhang, D.; He, K.; Herbst, J.J.; Kolb, J.; Shou, W.; Wang, L.; Balimane, P.V.; Han, Y.-H.; Gan, J.; Frost, C.E.; et al. Characterization of Efflux Transporters Involved in Distribution and Disposition of Apixaban. Drug Metab. Dispos. Biol. Fate Chem. 2013, 41, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Frost, C.; Wang, J.; Nepal, S.; Schuster, A.; Barrett, Y.C.; Mosqueda-Garcia, R.; Reeves, R.A.; LaCreta, F. Apixaban, an Oral, Direct Factor Xa Inhibitor: Single Dose Safety, Pharmacokinetics, Pharmacodynamics and Food Effect in Healthy Subjects. Br. J. Clin. Pharmacol. 2013, 75, 476–487. [Google Scholar] [CrossRef]
- Ogata, K.; Mendell-Harary, J.; Tachibana, M.; Masumoto, H.; Oguma, T.; Kojima, M.; Kunitada, S. Clinical Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of the Novel Factor Xa Inhibitor Edoxaban in Healthy Volunteers. J. Clin. Pharmacol. 2010, 50, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Zahir, H.; Matsushima, N.; Halim, A.-B.; He, L.; Zhang, G.; Lee, F.; Worland, V.; Mendell, J. Edoxaban Administration Following Enoxaparin: A Pharmacodynamic, Pharmacokinetic, and Tolerability Assessment in Human Subjects. Thromb. Haemost. 2012, 108, 166–175. [Google Scholar] [CrossRef]
- Bathala, M.S.; Masumoto, H.; Oguma, T.; He, L.; Lowrie, C.; Mendell, J. Pharmacokinetics, Biotransformation, and Mass Balance of Edoxaban, a Selective, Direct Factor Xa Inhibitor, in Humans. Drug Metab. Dispos. Biol. Fate Chem. 2012, 40, 2250–2255. [Google Scholar] [CrossRef]
- Matsushima, N.; Lee, F.; Sato, T.; Weiss, D.; Mendell, J. Bioavailability and Safety of the Factor Xa Inhibitor Edoxaban and the Effects of Quinidine in Healthy Subjects. Clin. Pharmacol. Drug Dev. 2013, 2, 358–366. [Google Scholar] [CrossRef]
- Mikkaichi, T.; Yoshigae, Y.; Masumoto, H.; Imaoka, T.; Rozehnal, V.; Fischer, T.; Okudaira, N.; Izumi, T. Edoxaban Transport via P-Glycoprotein Is a Key Factor for the Drug’s Disposition. Drug Metab. Dispos. Biol. Fate Chem. 2014, 42, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Mendell, J.; Zahir, H.; Matsushima, N.; Noveck, R.; Lee, F.; Chen, S.; Zhang, G.; Shi, M. Drug-Drug Interaction Studies of Cardiovascular Drugs Involving P-Glycoprotein, an Efflux Transporter, on the Pharmacokinetics of Edoxaban, an Oral Factor Xa Inhibitor. Am. J. Cardiovasc. Drugs Drugs Devices Interv. 2013, 13, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Mendell, J.; Chen, S.; He, L.; Desai, M.; Parasramupria, D.A. The Effect of Rifampin on the Pharmacokinetics of Edoxaban in Healthy Adults. Clin. Drug Investig. 2015, 35, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, B.I.; Quinlan, D.J.; Weitz, J.I. Comparative Pharmacodynamics and Pharmacokinetics of Oral Direct Thrombin and Factor Xa Inhibitors in Development. Clin. Pharmacokinet. 2009, 48, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Chan, N.C.; Bhagirath, V.; Eikelboom, J.W. Profile of Betrixaban and Its Potential in the Prevention and Treatment of Venous Thromboembolism. Vasc. Health Risk Manag. 2015, 11, 343–351. [Google Scholar] [CrossRef]
- Stangier, J.; Rathgen, K.; Stähle, H.; Gansser, D.; Roth, W. The Pharmacokinetics, Pharmacodynamics and Tolerability of Dabigatran Etexilate, a New Oral Direct Thrombin Inhibitor, in Healthy Male Subjects. Br. J. Clin. Pharmacol. 2007, 64, 292–303. [Google Scholar] [CrossRef]
- Gustafson, W.L.; Saunders, J.; Vazquez, S.R.; Jones, A.E.; Witt, D.M. Real-World Study of Direct Oral Anticoagulant Dosing Patterns in Patients with Atrial Fibrillation. Pharm. Pract. 2019, 17, 1709. [Google Scholar] [CrossRef]
- Serper, M.; Weinberg, E.M.; Cohen, J.B.; Reese, P.P.; Taddei, T.H.; Kaplan, D.E. Mortality and Hepatic Decompensation in Patients with Cirrhosis and Atrial Fibrillation Treated With Anticoagulation. Hepatology 2020. [Google Scholar] [CrossRef]
- Coons, J.C.; Albert, L.; Bejjani, A.; Iasella, C.J. Effectiveness and Safety of Direct Oral Anticoagulants versus Warfarin in Obese Patients with Acute Venous Thromboembolism. Pharmacotherapy 2020, 40, 204–210. [Google Scholar] [CrossRef]
- Gibson, C.M.; Halaby, R.; Korjian, S.; Daaboul, Y.; Arbetter, D.F.; Yee, M.K.; Goldhaber, S.Z.; Hull, R.; Hernandez, A.F.; Lu, S.-P.; et al. The Safety and Efficacy of Full- versus Reduced-Dose Betrixaban in the Acute Medically Ill VTE (Venous Thromboembolism) Prevention with Extended-Duration Betrixaban (APEX) Trial. Am. Heart J. 2017, 185, 93–100. [Google Scholar] [CrossRef]
- Gu, T.M.; Garcia, D.A.; Sabath, D.E. Assessment of Direct Oral Anticoagulant Assay Use in Clinical Practice. J. Thromb. Thrombolysis 2019, 47, 403–408. [Google Scholar] [CrossRef]
- Wright, C.; Brown, R.; Cuker, A. Laboratory Measurement of the Direct Oral Anticoagulants: Indications and Impact on Management in Clinical Practice. Int. J. Lab. Hematol. 2017, 39 (Suppl. S1), 31–36. [Google Scholar] [CrossRef]
- Martin, K.; Moll, S. Direct Oral Anticoagulant Drug Level Testing in Clinical Practice: A Single Institution Experience. Thromb. Res. 2016, 143, 40–44. [Google Scholar] [CrossRef]
- Takatsuki, S.; Kimura, T.; Sugimoto, K.; Misaki, S.; Nakajima, K.; Kashimura, S.; Kunitomi, A.; Katsumata, Y.; Nishiyama, T.; Nishiyama, N.; et al. Real-World Monitoring of Direct Oral Anticoagulants in Clinic and Hospitalization Settings. SAGE Open Med. 2017, 5, 2050312117734773. [Google Scholar] [CrossRef]
- Pollack, C.V.; Reilly, P.A.; van Ryn, J.; Eikelboom, J.W.; Glund, S.; Bernstein, R.A.; Dubiel, R.; Huisman, M.V.; Hylek, E.M.; Kam, C.-W.; et al. Idarucizumab for Dabigatran Reversal—Full Cohort Analysis. N. Engl. J. Med. 2017, 377, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Connolly, S.J.; Milling, T.J.; Eikelboom, J.W.; Gibson, C.M.; Curnutte, J.T.; Gold, A.; Bronson, M.D.; Lu, G.; Conley, P.B.; Verhamme, P.; et al. Andexanet Alfa for Acute Major Bleeding Associated with Factor Xa Inhibitors. N. Engl. J. Med. 2016, 375, 1131–1141. [Google Scholar] [CrossRef]
- Lange, U.; Nowak, G.; Bucha, E. Ecarin Chromogenic Assay—A New Method for Quantitative Determination of Direct Thrombin Inhibitors like Hirudin. Pathophysiol. Haemost. Thromb. 2003, 33, 184–191. [Google Scholar] [CrossRef]
- Skeppholm, M.; Hjemdahl, P.; Antovic, J.P.; Muhrbeck, J.; Eintrei, J.; Rönquist-Nii, Y.; Pohanka, A.; Beck, O.; Malmström, R.E. On the Monitoring of Dabigatran Treatment in “Real Life” Patients with Atrial Fibrillation. Thromb. Res. 2014, 134, 783–789. [Google Scholar] [CrossRef]
- Annesley, T.; Majzoub, J.; Hsing, A.; Wu, A.; Rockwood, A.; Mason, D. Mass Spectrometry in the Clinical Laboratory: How Have We Done, and Where Do We Need to Be? Clin. Chem. 2009, 55, 1236–1239. [Google Scholar] [CrossRef]
- Schmohl, M.; Gansser, D.; Moschetti, V.; Stangier, J. Measurement of Dabigatran Plasma Concentrations by Calibrated Thrombin Clotting Time in Comparison to LC-MS/MS in Human Volunteers on Dialysis. Thromb. Res. 2015, 135, 532–536. [Google Scholar] [CrossRef]
- Rohde, G. Determination of Rivaroxaban—A Novel, Oral, Direct Factor Xa Inhibitor-in Human Plasma by High-Performance Liquid Chromatography-Tandem Mass Spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2008, 872, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Kitchen, S.; Gray, E.; Mackie, I.; Baglin, T.; Makris, M. BCSH committee Measurement of Non-Coumarin Anticoagulants and Their Effects on Tests of Haemostasis: Guidance from the British Committee for Standards in Haematology. Br. J. Haematol. 2014, 166, 830–841. [Google Scholar] [CrossRef]
- Douxfils, J.; Mullier, F.; Robert, S.; Chatelain, C.; Chatelain, B.; Dogné, J.-M. Impact of Dabigatran on a Large Panel of Routine or Specific Coagulation Assays. Laboratory Recommendations for Monitoring of Dabigatran Etexilate. Thromb. Haemost. 2012, 107, 985–997. [Google Scholar] [CrossRef]
- Cuker, A.; Siegal, D.M.; Crowther, M.A.; Garcia, D.A. Laboratory Measurement of the Anticoagulant Activity of the Non-Vitamin K Oral Anticoagulants. J. Am. Coll. Cardiol. 2014, 64, 1128–1139. [Google Scholar] [CrossRef]
- Dale, B.J.; Chan, N.C.; Eikelboom, J.W. Laboratory Measurement of the Direct Oral Anticoagulants. Br. J. Haematol. 2016, 172, 315–336. [Google Scholar] [CrossRef]
- Siddiqui, F.; Hoppensteadt, D.; Jeske, W.; Iqbal, O.; Tafur, A.; Fareed, J. Factor Xa Inhibitory Profile of Apixaban, Betrixaban, Edoxaban, and Rivaroxaban Does Not Fully Reflect Their Biologic Spectrum. Clin. Appl. Thromb. Off. J. Int. Acad. Clin. Appl. Thromb. 2019, 25, 1076029619847524. [Google Scholar] [CrossRef]
- Barrett, Y.C.; Wang, Z.; Frost, C.; Shenker, A. Clinical Laboratory Measurement of Direct Factor Xa Inhibitors: Anti-Xa Assay Is Preferable to Prothrombin Time Assay. Thromb. Haemost. 2010, 104, 1263–1271. [Google Scholar] [CrossRef]
- Patel, J.P.; Roberts, L.N.; Chitongo, P.B.; Patel, R.K.; Arya, R. More on Normal Prothrombin Times in the Presence of Therapeutic Levels of Rivaroxaban—Early Experience from King’s College Hospital. Br. J. Haematol. 2013, 162, 717–718. [Google Scholar] [CrossRef]
- Patel, J.P.; Chitongo, P.B.; Czuprynska, J.; Roberts, L.N.; Patel, R.K.; Arya, R. Normal Prothrombin Times in the Presence of Therapeutic Levels of Apixaban—In-Vivo Experience from King’s College Hospital. Br. J. Haematol. 2015, 169, 152–153. [Google Scholar] [CrossRef]
- Patel, J.P.; Chitongo, P.B.; Dighe, P.; Roberts, L.N.; Vadher, B.; Patel, R.K.; Arya, R. Prothrombin Times in the Presence of Edoxaban—In-Vivo Experience from King’s College Hospital. Br. J. Haematol. 2019, 184, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Samama, M.M.; Martinoli, J.-L.; LeFlem, L.; Guinet, C.; Plu-Bureau, G.; Depasse, F.; Perzborn, E. Assessment of Laboratory Assays to Measure Rivaroxaban--an Oral, Direct Factor Xa Inhibitor. Thromb. Haemost. 2010, 103, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Douxfils, J.; Lessire, S.; Dincq, A.-S.; Hjemdahl, P.; Rönquist-Nii, Y.; Pohanka, A.; Gourdin, M.; Chatelain, B.; Dogné, J.-M.; Mullier, F. Estimation of Dabigatran Plasma Concentrations in the Perioperative Setting. An Ex Vivo Study Using Dedicated Coagulation Assays. Thromb. Haemost. 2015, 113, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Chin, P.K.L.; Wright, D.F.B.; Patterson, D.M.; Doogue, M.P.; Begg, E.J. A Proposal for Dose-Adjustment of Dabigatran Etexilate in Atrial Fibrillation Guided by Thrombin Time. Br. J. Clin. Pharmacol. 2014, 78, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Amiral, J.; Dunois, C.; Amiral, C.; Seghatchian, J. An Update on Laboratory Measurements of Dabigatran: Smart Specific and Calibrated Dedicated Assays for Measuring Anti-IIa Activity in Plasma. Transfus. Apher. Sci. Off. J. World Apher. Assoc. Off. J. Eur. Soc. Haemapheresis 2016, 54, 428–437. [Google Scholar] [CrossRef]
- Samama, M.M.; Poller, L. Contemporary Laboratory Monitoring of Low Molecular Weight Heparins. Clin. Lab. Med. 1995, 15, 119–123. [Google Scholar] [CrossRef]
- Samama, M.M.; Amiral, J.; Guinet, C.; Perzborn, E.; Depasse, F. An Optimised, Rapid Chromogenic Assay, Specific for Measuring Direct Factor Xa Inhibitors (Rivaroxaban) in Plasma. Thromb. Haemost. 2010, 104, 1078–1079. [Google Scholar] [CrossRef]
- Lessire, S.; Douxfils, J.; Pochet, L.; Dincq, A.-S.; Larock, A.-S.; Gourdin, M.; Dogné, J.-M.; Chatelain, B.; Mullier, F. Estimation of Rivaroxaban Plasma Concentrations in the Perioperative Setting in Patients With or Without Heparin Bridging. Clin. Appl. Thromb. Off. J. Int. Acad. Clin. Appl. Thromb. 2018, 24, 129–138. [Google Scholar] [CrossRef] [PubMed]
| Dabigatran | Rivaroxaban | Apixaban | Edoxaban | Betrixaban | |
|---|---|---|---|---|---|
| Action | Anti-IIa | Anti-Xa | Anti-Xa | Anti-Xa | Anti-Xa |
| Frequency of intake | Twice daily | Once daily | Twice daily | Once daily | Once daily |
| Tmax (h) | 1.5–3 | 2–4 | 3–4 | 1–2 | 3–4 |
| Ttrough (h) | 12 | 24 | 12 | 24 | 24 |
| Half-life (h) | 12–14 | 5–9 | 10–14 | 9–11 | 35–45 |
| Bioavailability | 3–7% | 80–100% | 50% | 62% | 34% |
| Protein binding | 35% | 92–95% | 87% | 40–59% | 60% |
| Renal clearance (%) | 80% | 40% | 27% | 50% | 6–13% |
| Dabigatran a | Rivaroxaban b | Apixaban c,d | Edoxaban e | Betrixaban f | ||
|---|---|---|---|---|---|---|
| Frequency of intake | Twice daily | Once daily | Twice daily | Twice daily | Once daily | Once daily |
| Dose | 150 mg | 20 mg | 5 mg | 10 mg | 60 mg | 80 mg |
| Cmax (ng/mL) | 175 (117–275) | 249 (184–343) | 171 (91–321) | 251 (111–572) | 170 (125–245) | 46 (5–117) |
| Ctrough (ng/mL) | 91 (61–143) | 44 (12–137) | 103 (41–230) | 120 (41–335) | 36 (19–62) | 17 (16–22) |
| Dabigatran | Rivaroxaban | Apixaban | Edoxaban | |
|---|---|---|---|---|
| AF dosage | 150 mg twice daily | 20 mg once daily | 5 mg twice daily | 60 mg once daily |
| Criteria for adjustment | CrCL 30–50 mL/min No recommendation: CrCL < 30 mL/min or HD | CrCL 15–50 mL/min No recommendation: CrCL < 15 mL/min or HD | 2 of 3 required: body weight ≤ 60 kg age ≥ 80 years SCr ≥ 1.5 mg/dL | CrCL 15–50 mL/min Avoid use if: CrCL < 15 mL/min CrCL > 95 mL/min No recommendation for HD |
| Dose adjustment | 110 mg twice daily | 15 mg once daily | 2.5 mg twice daily | 30 mg once daily |
| VTE treatment Recurrent VTE VTE prophylaxis | 150 mg twice daily 150 mg twice daily 150 mg twice daily | 15 mg (21 days) then 20 mg once daily 10 or 20 mg once daily 10 mg once daily | 10 mg (7 days) then 5 mg twice daily 2.5 mg twice daily 2.5 mg twice daily | 60 mg once daily |
| Criteria for adjustment | CrCL 30–50 mL/min No recommendation: CrCL < 30 mL/min | Avoid use if: CrCL < 30 mL/min or HD | No recommendation: CrCL < 25 mL/min | CrCL: 15–50 mL/min or body weight ≤ 60 kg Avoid use if: CrCL < 15 mL/min |
| Dose adjustment | 110 mg twice daily | 30 mg once daily |
| Dabigatran | Rivaroxaban | Apixaban | Edoxaban | Betrixaban | |
|---|---|---|---|---|---|
| Drug interaction (inhibitors) | P-gp | Strong CYP3A4+P-gp | Strong CYP3A4+P-gp | P-gp | P-gp |
| Plasma concentration | ↑ | ↑ | ↑ | ↑ | ↑ |
| Dose adjustment | Reduce or avoid | Avoid | Reduce or avoid | Reduce (VTE) | Avoid: CrCL < 30 mL/min |
| Drug interaction (inducers) | P-gp | Strong CYP3A4 or P-gp | Strong CYP3A4 or P-gp | P-gp | P-gp |
| Plasma concentration | ↓ | ↓ | ↓ | ↓ | ↓ |
| Dose adjustment | Avoid | Avoid | Avoid | Avoid with Rifampin | No recommendation |
| Dabigatran | Rivaroxaban | Apixaban | Edoxaban | Betrixaban | |
|---|---|---|---|---|---|
| PT/INR a,b | ↑ at Cmax | ↑ | Slight or no change | ↑ | ↑ |
| APTT a,b | ↑ | ↑at Cmax | Slight or no change | ↑ at Cmax | ↑ at Cmax |
| TT | ↑at Cthrough | No change | No change | No change | No change |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dunois, C. Laboratory Monitoring of Direct Oral Anticoagulants (DOACs). Biomedicines 2021, 9, 445. https://doi.org/10.3390/biomedicines9050445
Dunois C. Laboratory Monitoring of Direct Oral Anticoagulants (DOACs). Biomedicines. 2021; 9(5):445. https://doi.org/10.3390/biomedicines9050445
Chicago/Turabian StyleDunois, Claire. 2021. "Laboratory Monitoring of Direct Oral Anticoagulants (DOACs)" Biomedicines 9, no. 5: 445. https://doi.org/10.3390/biomedicines9050445
APA StyleDunois, C. (2021). Laboratory Monitoring of Direct Oral Anticoagulants (DOACs). Biomedicines, 9(5), 445. https://doi.org/10.3390/biomedicines9050445





