Transmembrane 163 (TMEM163) Protein: A New Member of the Zinc Efflux Transporter Family
Abstract
1. Introduction
2. Zinc Transport
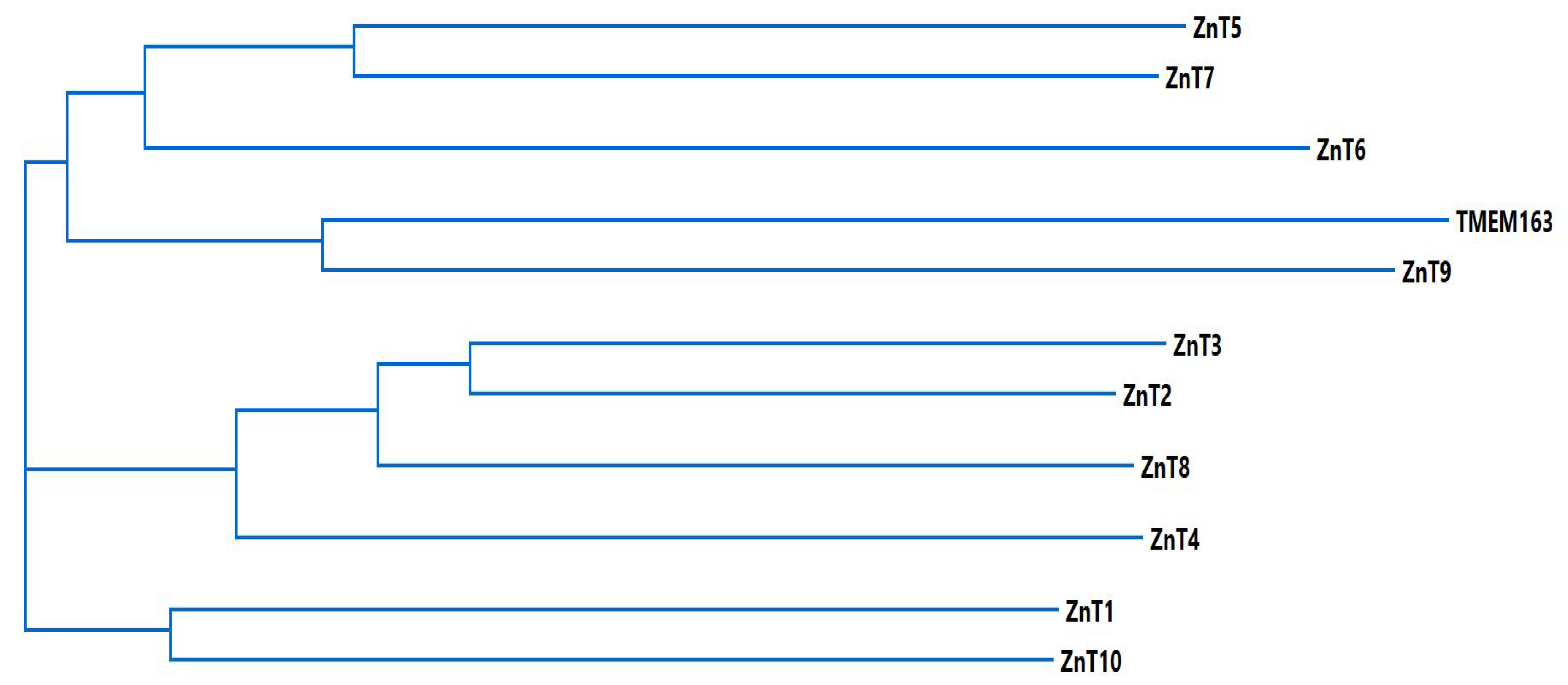
2.1. The SLC30 Family or ZnT Efflux Transporters
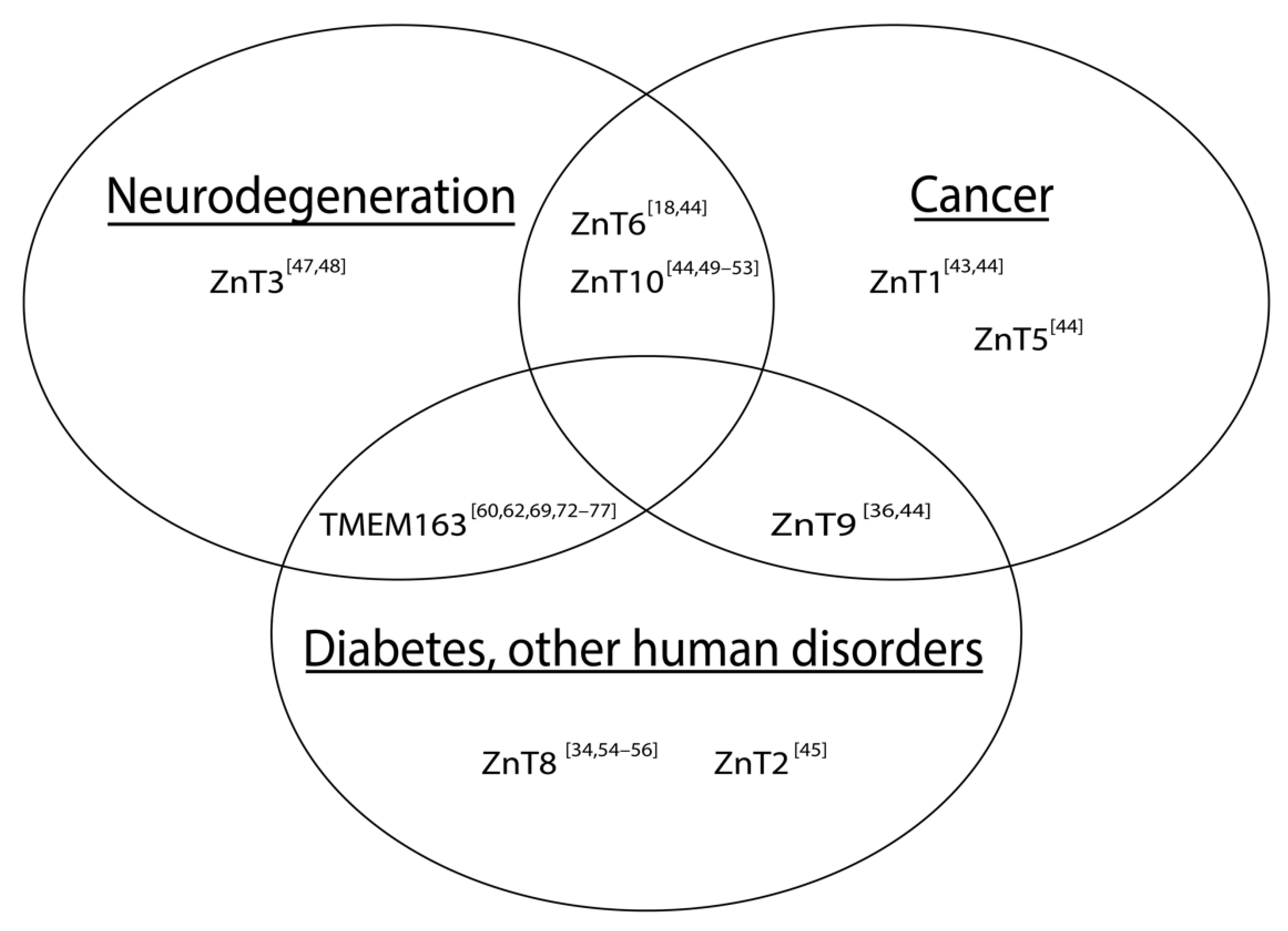
2.2. ZnTs in Human Diseases
3. TMEM163
3.1. Protein Characteristics and Function
3.2. Protein Interactome
4. Genome-Wide Association Study (GWAS) Implicating TMEM163 in Human Diseases
4.1. Diabetes Mellitus
4.2. Parkinson’s Disease
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signaling in health and diseases. JBIC J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Wessels, I.; Maywald, M.; Rink, L. Zinc as a gatekeeper of immune function. Nutrients 2017, 9, 1286. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, P.; Benedetti, G.; Albarède, F.; Miossec, P. Zinc and its role in immunity and inflammation. Autoimmun. Rev. 2015, 14, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Vallee, B.L.; Falchuk, K.H. The biochemical basis of zinc physiology. Physiol. Rev. 1993, 73, 79–118. [Google Scholar] [CrossRef]
- Zhang, N.; Duncan, F.E.; Que, E.L.; O’Halloran, T.V.; Woodruff, T.K. The fertilization-induced zinc spark is a novel biomarker of mouse embryo quality and early development. Sci. Rep. 2016, 6, 22772. [Google Scholar] [CrossRef]
- Kim, T.Y.; Hwang, J.J.; Yun, S.H.; Jung, M.W.; Koh, J.Y. Augmentation by zinc of NMDA receptor-mediated synaptic responses in CA1 of rat hippocampal slices: Mediation by Src family tyrosine kinases. Synapse 2002, 46, 49–56. [Google Scholar] [CrossRef]
- Smart, T.G.; Moss, S.J.; Xie, X.; Huganir, R.L. GABAA receptors are differentially sensitive to zinc: Dependence on subunit composition. Br. J. Pharm. 1991, 103, 1837–1839. [Google Scholar] [CrossRef]
- Uchida, K.; Tominaga, M. Extracellular zinc ion regulates transient receptor potential melastatin 5 (TRPM5) channel activation through its interaction with a pore loop domain. J. Biol. Chem. 2013, 288, 25950–25955. [Google Scholar] [CrossRef] [PubMed]
- Sensi, S.L.; Yin, H.Z.; Carriedo, S.G.; Rao, S.S.; Weiss, J.H. Preferential Zn2+ influx through Ca2+-permeable AMPA/kainate channels triggers prolonged mitochondrial superoxide production. Proc. Natl. Acad. Sci. USA 1999, 96, 2414–2419. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yan, F.; Yin, J.; Pan, R.; Shi, W.; Qi, Z.; Fang, Y.; Huang, Y.; Li, S.; Luo, Y.; et al. synergistic interaction between zinc and reactive oxygen species amplifies ischemic brain injury in rats. Stroke 2018, 49, 2200–2210. [Google Scholar] [CrossRef] [PubMed]
- Gammoh, N.Z.; Rink, L. Zinc in infection and inflammation. Nutrients 2017, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Radi, R. Oxygen radicals, nitric oxide, and peroxynitrite: Redox pathways in molecular medicine. Proc. Natl. Acad. Sci. USA 2018, 115, 5839–5848. [Google Scholar] [CrossRef] [PubMed]
- Chabosseau, P.; Rutter, G.A. Zinc and diabetes. Arch. Biochem. Biophys. 2016, 611, 79–85. [Google Scholar] [CrossRef]
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef]
- Wong, C.P.; Rinaldi, N.A.; Ho, E. Zinc deficiency enhanced inflammatory response by increasing immune cell activation and inducing IL6 promoter demethylation. Mol. Nutr. Food Res. 2015, 59, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, M.K.; Zhou, Z.; Cave, M.; Barve, A.; McClain, C.J. Zinc and liver disease. Nutr. Clin. Pract. 2012, 27, 8–20. [Google Scholar] [CrossRef]
- Kambe, T.; Tsuji, T.; Hashimoto, A.; Itsumura, N. The physiological, biochemical, and molecular roles of zinc transporters in zinc homeostasis and metabolism. Physiol. Rev. 2015, 95, 749–784. [Google Scholar] [CrossRef]
- Huang, L.; Tepaamorndech, S. The SLC30 family of zinc transporters—A review of current understanding of their biological and pathophysiological roles. Mol. Asp. Med. 2013, 34, 548–560. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.; Eide, D.J. The SLC39 family of zinc transporters. Mol. Asp. Med. 2013, 34, 612–619. [Google Scholar] [CrossRef]
- Hara, T.; Takeda, T.-A.; Takagishi, T.; Fukue, K.; Kambe, T.; Fukada, T. Physiological roles of zinc transporters: Molecular and genetic importance in zinc homeostasis. J. Physiol. Sci. 2017, 67, 283–301. [Google Scholar] [CrossRef] [PubMed]
- Lichten, L.A.; Cousins, R.J. Mammalian zinc transporters: Nutritional and physiologic regulation. Annu. Rev. Nutr. 2009, 29, 153–176. [Google Scholar] [CrossRef] [PubMed]
- Kolaj-Robin, O.; Russell, D.; Hayes, K.A.; Pembroke, J.T.; Soulimane, T. Cation diffusion facilitator family: Structure and function. Febs Lett. 2015, 589, 1283–1295. [Google Scholar] [CrossRef]
- Lu, M.; Fu, D. Structure of the zinc transporter YiiP. Science 2007, 317, 1746–1748. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Xie, T.; Zeng, W.; Jiang, Y.; Bai, X.C. Cryo-EM structures of human ZnT8 in both outward- and inward-facing conformations. eLife 2020, 9, e58823. [Google Scholar] [CrossRef]
- Bosomworth, H.J.; Thornton, J.K.; Coneyworth, L.J.; Ford, D.; Valentine, R.A. Efflux function, tissue-specific expression and intracellular trafficking of the Zn transporter ZnT10 indicate roles in adult Zn homeostasis. Metallomics 2012, 4, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Zogzas, C.E.; Aschner, M.; Mukhopadhyay, S. Structural elements in the transmembrane and cytoplasmic domains of the metal transporter SLC30A10 are required for its manganese efflux activity. J. Biol. Chem. 2016, 291, 15940–15957. [Google Scholar] [CrossRef]
- Zogzas, C.E.; Mukhopadhyay, S. Putative metal binding site in the transmembrane domain of the manganese transporter SLC30A10 is different from that of related zinc transporters. Metallomics 2018, 10, 1053–1064. [Google Scholar] [CrossRef]
- Levy, M.; Elkoshi, N.; Barber-Zucker, S.; Hoch, E.; Zarivach, R.; Hershfinkel, M.; Sekler, I. Zinc transporter 10 (ZnT10)-dependent extrusion of cellular Mn(2+) is driven by an active Ca(2+)-coupled exchange. J. Biol. Chem. 2019, 294, 5879–5889. [Google Scholar] [CrossRef]
- Golan, Y.; Alhadeff, R.; Warshel, A.; Assaraf, Y.G. ZnT2 is an electroneutral proton-coupled vesicular antiporter displaying an apparent stoichiometry of two protons per zinc ion. Plos Comput. Biol. 2019, 15, e1006882. [Google Scholar] [CrossRef] [PubMed]
- Guffanti, A.A.; Wei, Y.; Rood, S.V.; Krulwich, T.A. An antiport mechanism for a member of the cation diffusion facilitator family: Divalent cations efflux in exchange for K+ and H+. Mol. Microbiol. 2002, 45, 145–153. [Google Scholar] [CrossRef]
- Ohana, E.; Hoch, E.; Keasar, C.; Kambe, T.; Yifrach, O.; Hershfinkel, M.; Sekler, I. Identification of the Zn2+ binding site and mode of operation of a mammalian Zn2+ transporter. J. Biol. Chem. 2009, 284, 17677–17686. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, V.B.; Ali, S.; Escobar, A.; Cuajungco, M.P. Transmembrane 163 (TMEM163) protein effluxes zinc. Arch. Biochem. Biophys. 2019, 677, 108166. [Google Scholar] [CrossRef] [PubMed]
- Valentine, R.A.; Jackson, K.A.; Christie, G.R.; Mathers, J.C.; Taylor, P.M.; Ford, D. ZnT5 variant b is a bidirectional zinc transporter and mediates zinc uptake in human intestinal caco-2 cells. J. Biol. Chem. 2007, 282, 14389–14393. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, T.J.; Bellomo, E.A.; Wijesekara, N.; Loder, M.K.; Baldwin, J.M.; Gyulkhandanyan, A.V.; Koshkin, V.; Tarasov, A.I.; Carzaniga, R.; Kronenberger, K.; et al. Insulin storage and glucose homeostasis in mice null for the granule zinc transporter ZnT8 and studies of the type 2 Diabetes-associated variants. Diabetes 2009, 58, 2070–2083. [Google Scholar] [CrossRef]
- Weijers, R.N. Three-dimensional structure of β-cell-specific zinc transporter, ZnT-8, predicted from the type 2 diabetes-associated gene variant SLC30A8 R325W. Diabetol. Metab. Syndr. 2010, 2, 33. [Google Scholar] [CrossRef]
- Perez, Y.; Shorer, Z.; Liani-Leibson, K.; Chabosseau, P.; Kadir, R.; Volodarsky, M.; Halperin, D.; Barber-Zucker, S.; Shalev, H.; Schreiber, R.; et al. SLC30A9 mutation affecting intracellular zinc homeostasis causes a novel cerebro-renal syndrome. Brain 2017, 140, 928–939. [Google Scholar] [CrossRef]
- He, D.; Wang, J.; Li, Y.; Wu, G.; Zhu, G.; Chen, L. Low-intensity pulsed ultrasound promotes aggrecan expression via ZNT-9 in temporomandibular joint chondrocytes. Gene 2021, 768, 145318. [Google Scholar] [CrossRef]
- Golan, Y.; Berman, B.; Assaraf, Y.G. Heterodimerization, altered subcellular localization, and function of multiple zinc transporters in viable cells using bimolecular fluorescence complementation. J. Biol. Chem. 2015, 290, 9050–9063. [Google Scholar] [CrossRef]
- Lasry, I.; Golan, Y.; Berman, B.; Amram, N.; Glaser, F.; Assaraf, Y.G. In situ dimerization of multiple wild type and mutant zinc transporters in live cells using bimolecular fluorescence complementation. J. Biol. Chem. 2014, 289, 7275–7292. [Google Scholar] [CrossRef] [PubMed]
- Murgia, C.; Devirgiliis, C.; Mancini, E.; Donadel, G.; Zalewski, P.; Perozzi, G. Diabetes-linked zinc transporter ZnT8 is a homodimeric protein expressed by distinct rodent endocrine cell types in the pancreas and other glands. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Fukunaka, A.; Suzuki, T.; Kurokawa, Y.; Yamazaki, T.; Fujiwara, N.; Ishihara, K.; Migaki, H.; Okumura, K.; Masuda, S.; Yamaguchi-Iwai, Y.; et al. Demonstration and characterization of the heterodimerization of ZnT5 and ZnT6 in the early secretory pathway. J. Biol. Chem. 2009, 284, 30798–30806. [Google Scholar] [CrossRef]
- Patrushev, N.; Seidel-Rogol, B.; Salazar, G. Angiotensin II requires zinc and downregulation of the zinc transporters ZnT3 and ZnT10 to induce senescence of vascular smooth muscle cells. PLoS ONE 2012, 7, e33211. [Google Scholar] [CrossRef]
- Lehvy, A.I.; Horev, G.; Golan, Y.; Glaser, F.; Shammai, Y.; Assaraf, Y.G. Alterations in ZnT1 expression and function lead to impaired intracellular zinc homeostasis in cancer. Cell Death Discov. 2019, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.K.; Malas, K.M.; Tydrick, C.; Siddiqui, I.A.; Iczkowski, K.A.; Ahmad, N. Analysis of zinc-exporters expression in prostate cancer. Sci. Rep. 2016, 6, 36772. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, S.L.; Gagnon, A.; Rivera, O.C.; Hicks, S.D.; Carney, M.C.; Alam, S. Milk-derived miRNA profiles elucidate molecular pathways that underlie breast dysfunction in women with common genetic variants in SLC30A2. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Beyer, N.; Coulson, D.T.; Heggarty, S.; Ravid, R.; Irvine, G.B.; Hellemans, J.; Johnston, J.A. ZnT3 mRNA levels are reduced in Alzheimer’s disease post-mortem brain. Mol. Neurodegener 2009, 4, 53. [Google Scholar] [CrossRef]
- Chacon, J.; Rosas, L.; Cuajungco, M.P. ZnT3 expression levels are down-regulated in the brain of Mcoln1 knockout mice. Mol. Brain 2019, 12, 1–3. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, T.; Yu, D.; Feng, W.Y.; Nie, Y.X.; Stoltenberg, M.; Danscher, G.; Wang, Z.Y. Elevation of zinc transporter ZnT3 protein in the cerebellar cortex of the AbetaPP/PS1 transgenic mouse. J. Alzheimers. Dis. 2010, 20, 323–331. [Google Scholar] [CrossRef]
- Bosomworth, H.J.; Adlard, P.A.; Ford, D.; Valentine, R.A. Altered expression of ZnT10 in Alzheimer’s disease brain. PLoS ONE 2013, 8, e65475. [Google Scholar] [CrossRef]
- Quadri, M.; Federico, A.; Zhao, T.; Guido, C.; Battisti, C.; Delnooz, L.-A.; Severijnen Lara, A.; Mignarri, L.; Monti, A.; Sanna, P.; et al. Mutations in slc30a10 cause parkinsonism and dystonia with hypermanganesemia, polycythemia, and chronic liver disease. Am. J. Hum. Genet. 2012, 90, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Stamelou, M.; Tuschl, K.; Chong, W.K.; Burroughs, A.K.; Mills, P.B.; Bhatia, K.P.; Clayton, P.T. Dystonia with brain manganese accumulation resulting from SLC30A10 mutations: A new treatable disorder. Mov. Disord. 2012, 27, 1317–1322. [Google Scholar] [CrossRef] [PubMed]
- Tuschl, K.; Clayton, P.T.; Gospe, S.M.; Gulab, S., Jr.; Ibrahim, S.; Singhi, P.; Aulakh, R.; Ribeiro, R.T.; Barsottini, O.G.; Zaki, M.S.; et al. Mills. Syndrome of hepatic cirrhosis, dystonia, polycythemia, and hypermanganesemia caused by mutations in SLC30A10, a manganese transporter in man. Am. J. Hum. Genet. 2012, 90, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Lambrianides, S.; Nicolaou, P.; Michaelidou, M.; Kakouris, P.; Votsi, C.; Petrou, P.P.; Drousiotou, A.; Minaidou, A.; Demetriou, P.; Voulgaris, C.; et al. A novel SLC30A10 missense variant associated with parkinsonism and dystonia without hypermanganesemia. J. Neurol. Sci. 2020, 418, 117101. [Google Scholar] [CrossRef] [PubMed]
- Chimienti, F.; Devergnas, S.; Pattou, F.; Schuit, F.; Garcia-Cuenca, R.; Vandewalle, B.; Kerr-Conte, J.; Van Lommel, L.; Grunwald, D.; Favier, A.; et al. In vivo expression and functional characterization of the zinc transporter ZnT8 in glucose-induced insulin secretion. J. Cell Sci. 2006, 119, 4199–4206. [Google Scholar] [CrossRef]
- Dwivedi, O.P.; Lehtovirta, M.; Hastoy, B.; Chandra, V.; Krentz, N.A.J.; Kleiner, S.; Jain, D.; Richard, A.-M.; Abaitua, F.; Beer, N.L.; et al. Loss of ZnT8 function protects against diabetes by enhanced insulin secretion. Nat. Genet. 2019, 51, 1596–1606. [Google Scholar] [CrossRef]
- Yi, B.; Huang, G.; Zhou, Z. Different role of zinc transporter 8 between type 1 diabetes mellitus and type 2 diabetes mellitus. J. Diabetes Investig. 2016, 7, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Burré, J.; Zimmermann, H.; Volknandt, W. Identification and characterization of SV31, a novel synaptic vesicle membrane protein and potential transporter. J. Neurochem. 2007, 103, 276–287. [Google Scholar] [CrossRef]
- Barth, J.; Zimmermann, H.; Volknandt, W. SV31 is a Zn2+-binding synaptic vesicle protein. J. Neurochem. 2011, 118, 558–570. [Google Scholar] [CrossRef]
- Frederickson, C.J. Neurobiology of zinc and zinc-containing neurons. Int. Rev. Neurobiol. 1989, 31, 145–238. [Google Scholar]
- Cuajungco, M.P.; Basilio, L.C.; Silva, J.; Hart, T.; Tringali, J.; Chen, C.-C.; Biel, M.; Grimm, C. Cellular zinc levels are modulated by TRPML1-TMEM163 interaction. Traffic 2014, 15, 1247–1265. [Google Scholar] [CrossRef]
- Chakraborty, S.; Vellarikkal, S.K.; Sivasubbu, S.; Roy, S.S.; Tandon, N.; Bharadwaj, D. Role of Tmem163 in zinc-regulated insulin storage of MIN6 cells: Functional exploration of an Indian type 2 diabetes GWAS associated gene. Biochem. Biophys. Res. Commun. 2020, 522, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Cuajungco, M.P.; Kiselyov, K. The mucolipin-1 TRPML1 ion channel transmembrane-163 TMEM163 protein and lysosomal zinc handling. Front. Biosci. 2017, 22, 1330–1343. [Google Scholar]
- Waberer, L.; Henrich, E.; Peetz, O.; Morgner, N.; Dötsch, V.; Bernhard, F.; Volknandt, W. The synaptic vesicle protein SV31 assembles into a dimer and transports Zn2+. J. Neurochem. 2017, 140, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Cuajungco, M.P. Protocol for quantifying zinc flux in cultured cells using fluorescent indicators. Star Protoc. 2020, 1, 100050. [Google Scholar] [CrossRef]
- Fine, M.; Schmiege, P.; Li, X. Structural basis for PtdInsP2-mediated human TRPML1 regulation. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, K.; Gustafson, A.M.; Sidransky, E.; Goldin, E. Mucolipidosis type IV: An update. Mol. Genet. Metab. 2011, 104, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Eichelsdoerfer, J.L.; Evans, J.A.; Slaugenhaupt, S.A.; Cuajungco, M.P. Zinc dyshomeostasis is linked with the loss of mucolipidosis IV-associated TRPML1 ion channel. J. Biol. Chem. 2010, 285, 34304–34308. [Google Scholar] [CrossRef] [PubMed]
- Kukic, I.; Jeffrey, J.; Coblentz, S.; Kiselyov, K. Zinc-dependent lysosomal enlargement in TRPML1-deficient cells involves MTF-1 transcription factor and ZnT4 (Slc30a4) transporter. Biochem. J. 2013, 451, 155–163. [Google Scholar] [CrossRef]
- Salm, E.J.; Dunn, P.J.; Shan, L.; Yamasaki, M.; Malewicz, N.M.; Miyazaki, T.; Park, J.; Sumioka, A.; Hamer, R.R.L.; He, W.-W.; et al. TMEM163 regulates ATP-Gated P2X receptor and behavior. Cell Rep. 2020, 31, 107704. [Google Scholar] [CrossRef] [PubMed]
- Khakh, B.S. Molecular physiology of p2x receptors and atp signalling at synapses. Nat. Rev. Neurosci. 2001, 2, 165–174. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Kim, J.H.; Stallcup, M.R. GAC63, a GRIP1-dependent nuclear receptor coactivator. Mol. Cell. Biol. 2005, 25, 5965–5972. [Google Scholar] [CrossRef] [PubMed]
- Tabassum, R.; Chauhan, G.; Dwivedi, O.P.; Mahajan, A.; Jaiswal, A.; Kaur, I.; Bandesh, K.; Singh, T.; Mathai, B.J.; Pandey, Y.; et al. Genome-wide association study for Type 2 Diabetes in indians identifies a new susceptibility locus at 2q21. Diabetes 2013, 62, 977–986. [Google Scholar] [CrossRef]
- Sharma, V.; Sharma, I.; Sethi, I.; Mahajan, A.; Singh, G.; Angural, A.; Bhanwer, A.J.S.; Dhar, M.K.; Singh, V.; Rai, E.; et al. Replication of newly identified type 2 Diabetes susceptible loci in northwest Indian population. Diabetes Res. Clin. Pract. 2017, 126, 160–163. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.-X.; Hu, S.-M.; You, Y.-P.; Yang, G.-L.; Wang, W. Replication of previous genome-wide association studies of HKDC1, BACE2, SLC16A11 and TMEM163 SNPs in a gestational diabetes mellitus case-control sample from Han Chinese population. Diabetes Metab. Syndr. Obes. 2019, 12, 983–989. [Google Scholar] [CrossRef]
- Bai, H.; Liu, H.; Suyalatu, S.; Guo, X.; Chu, S.; Chen, Y.; Lan, T.; Borjigin, B.; Orlov, Y.L.; Posukh, O.L.; et al. Association analysis of genetic variants with type 2 Diabetes in a Mongolian population in China. J. Diabetes Res. 2015, 2015, 1–7. [Google Scholar] [CrossRef]
- Chang, K.-H.; Chen, C.-M.; Chen, Y.-C.; Fung, H.-C.; Wu, Y.-R. Polymorphisms of ACMSD-TMEM163, MCCC1, and BCKDK-STX1B are not associated with Parkinson’s disease in Taiwan. Parkinson’s Dis. 2019, 2019, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ou, R.; Chen, Y.; Gu, X.; Wei, Q.; Cao, B.; Zhang, L.; Hou, Y.; Liu, K.; Chen, X.; et al. Mutation analysis of TMEM family members for early-onset Parkinson’s disease in Chinese population. Neurobiol. Aging 2020. [Google Scholar] [CrossRef]
- Nalls, M.A.; Blauwendraat, C.; Vallerga, C.L.; Heilbron, K.; Bandres-Ciga, S.; Chang, D.; Tan, M.; Kia, D.A.; Noyce, A.J.; Xue, A.; et al. System genomics of Parkinson’s Disease, C. International Parkinson’s disease genomics. Identification of novel risk loci, causal insights, and heritable risk for Parkinson’s disease: A meta-analysis of genome-wide association studies. Lancet Neurol. 2019, 18, 1091–1102. [Google Scholar] [CrossRef]
- Nalls, M.A.; Pankratz, N.; Lill, C.M.; Do, C.B.; Hernandez, D.G.; Saad, M.; Destefano, A.L.; Kara, E.; Bras, J.; Sharma, M.; et al. Large-scale meta-analysis of genome-wide association data identifies six new risk loci for Parkinson’s disease. Nat. Genet. 2014, 46, 989–993. [Google Scholar] [CrossRef]
- Thirtamara-Rajamani, K.; Li, P.; Escobar Galvis, M.L.; Labrie, V.; Brundin, P.; Brundin, L. Is the enzyme ACMSD a novel therapeutic target in Parkinson’s disease? J. Parkinson’s Dis. 2017, 7, 577–587. [Google Scholar] [CrossRef]
- Carmona, A.; Zogzas, C.E.; Roudeau, S.; Porcaro, F.; Garrevoet, J.; Spiers, K.M.; Salome, M.; Cloetens, P.; Mukhopadhyay, S.; Ortega, R. SLC30A10 mutation involved in parkinsonism results in manganese accumulation within nanovesicles of the golgi apparatus. ACS Chem. Neurosci. 2019, 10, 599–609. [Google Scholar] [CrossRef]
- Cuajungco, M.P.; Lees, G.J. Zinc metabolism in the brain: Relevance to human neurodegenerative disorders. Neurobiol. Dis. 1997, 4, 137–169. [Google Scholar] [CrossRef] [PubMed]
- Bandrés-Ciga, S.; Price, T.R.; Barrero, T.R.; Escamilla-Sevilla, F.; Pelegrina, J.; Arepalli, S.; Hernández, S.; Gutiérrez, B.; Cervilla, J.; Rivera, M.; et al. Genome-wide assessment of Parkinson’s disease in a southern Spanish population. Neurobiol. Aging 2016, 45, 213.e3–213.e9. [Google Scholar]
- Tejera-Parrado, C.; Jesús, S.; Periñán, M.T.; Buiza-Rueda, D.; Oliva-Ariza, G.; Adarmes-Gómez, A.D.; Macías-García, D.; Gómez-Garre, P.; Mir, P. A replication study of GWAS-genetic risk variants associated with Parkinson’s disease in a Spanish population. Neurosci. Lett. 2019, 712, 134425. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, N.-N.; Lu, Z.-J.; Li, J.-Y.; Peng, J.-X.; Duan, L.-R.; Peng, R. Association of three candidate genetic variants in ACMSD/TMEM163, GPNMB and BCKDK/STX1B with sporadic Parkinson’s disease in Han Chinese. Neurosci. Lett. 2019, 703, 45–48. [Google Scholar] [CrossRef]
| TMEM163 | ZnT1 | ZnT2 | ZnT3 | ZnT4 | ZnT5 | ZnT6 | ZnT7 | ZnT8 | ZnT9 | ZnT10 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TMEM163 | 0 | 17.6 | 10.3 | 11.5 | 11.2 | 9.5 | 11.7 | 14.7 | 10.8 | 24.2 | 18.9 |
| ZnT1 | 36.5 | 0 | 30.5 | 27.1 | 25.8 | 25.6 | 16.1 | 26.5 | 27.3 | 14.5 | 38.9 |
| ZnT2 | 35.3 | 50.8 | 0 | 53.7 | 38.4 | 22.6 | 18.4 | 22.5 | 50.6 | 14.9 | 24.8 |
| ZnT3 | 28.6 | 45.4 | 67.5 | 0 | 35.1 | 20.5 | 20.2 | 22.3 | 44.5 | 13.4 | 22.7 |
| ZnT4 | 29.4 | 49.4 | 60.8 | 57.4 | 0 | 22.6 | 18.5 | 22.4 | 39.2 | 15.4 | 25.2 |
| ZnT5 | 32.1 | 48.8 | 45.0 | 42.5 | 44.2 | 0 | 24.3 | 43.6 | 21.6 | 16.0 | 25.9 |
| ZnT6 | 29.4 | 34.3 | 38.1 | 37.1 | 39.7 | 47.0 | 0 | 24.6 | 18.2 | 14.5 | 16.7 |
| ZnT7 | 36.8 | 48.8 | 46.4 | 42.6 | 47.8 | 63.0 | 44.5 | 0 | 21.8 | 15.4 | 27.8 |
| ZnT8 | 30.8 | 52.3 | 69.1 | 64.3 | 61.0 | 44.1 | 37.9 | 42.9 | 0 | 14.9 | 25.7 |
| ZnT9 | 46.1 | 36.6 | 36.1 | 31.4 | 34.0 | 34.4 | 30.1 | 35.2 | 33.4 | 0 | 17.3 |
| ZnT10 | 36.0 | 54.1 | 47.6 | 44.5 | 48.8 | 43.7 | 38.0 | 45.8 | 50.0 | 37.3 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Styrpejko, D.J.; Cuajungco, M.P. Transmembrane 163 (TMEM163) Protein: A New Member of the Zinc Efflux Transporter Family. Biomedicines 2021, 9, 220. https://doi.org/10.3390/biomedicines9020220
Styrpejko DJ, Cuajungco MP. Transmembrane 163 (TMEM163) Protein: A New Member of the Zinc Efflux Transporter Family. Biomedicines. 2021; 9(2):220. https://doi.org/10.3390/biomedicines9020220
Chicago/Turabian StyleStyrpejko, Daniel J., and Math P. Cuajungco. 2021. "Transmembrane 163 (TMEM163) Protein: A New Member of the Zinc Efflux Transporter Family" Biomedicines 9, no. 2: 220. https://doi.org/10.3390/biomedicines9020220
APA StyleStyrpejko, D. J., & Cuajungco, M. P. (2021). Transmembrane 163 (TMEM163) Protein: A New Member of the Zinc Efflux Transporter Family. Biomedicines, 9(2), 220. https://doi.org/10.3390/biomedicines9020220







