Development of Non-Porous Silica Nanoparticles towards Cancer Photo-Theranostics
Abstract
1. Introduction
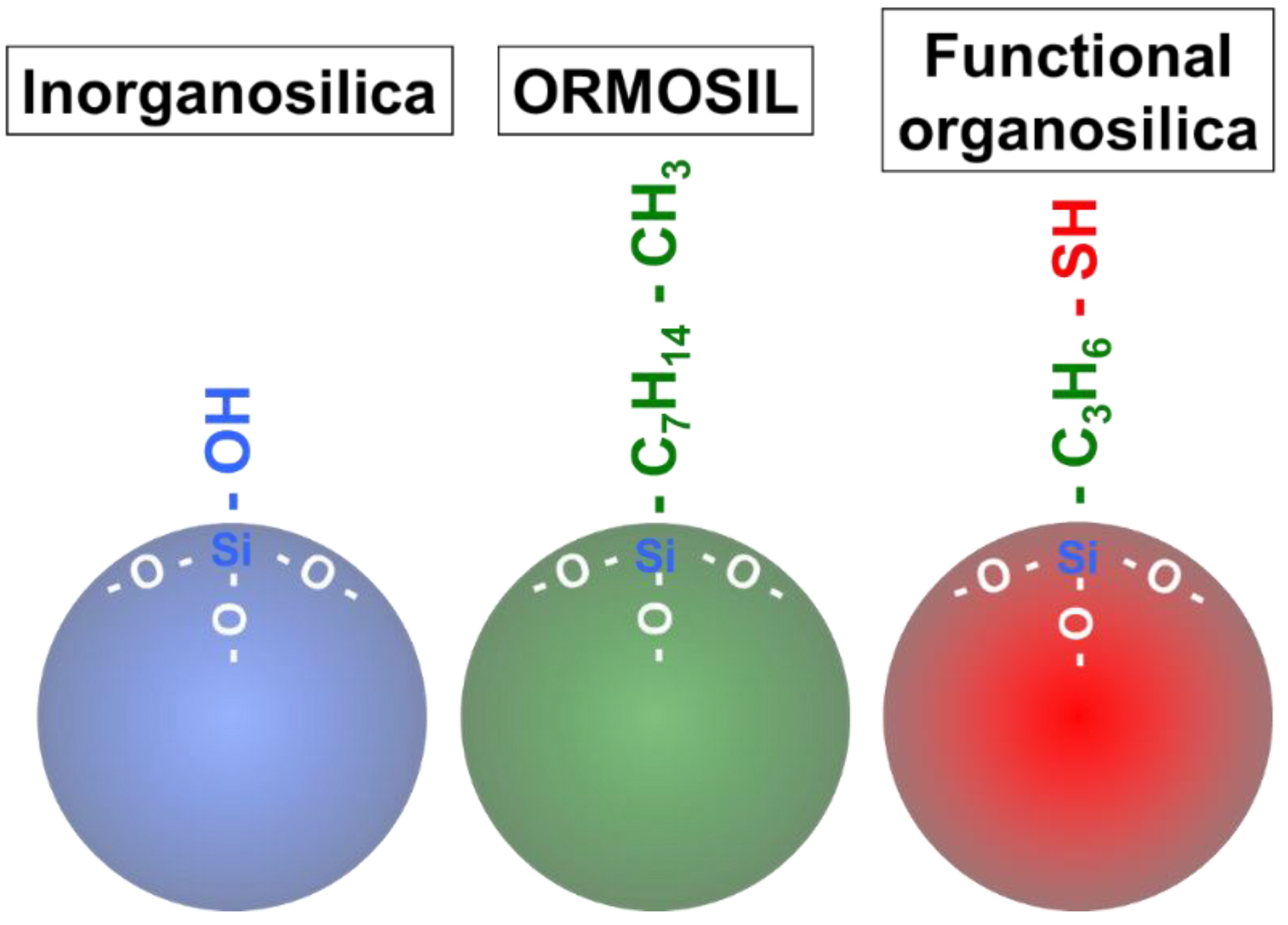
2. Silica Nanoparticles
2.1. Inorganosilica Nanoparticle
2.2. ORMOSIL
2.3. Functional Organosilica Nanoparticles
3. Multi-Functionalized Silica Nanoparticles
3.1. Fluorescence Materials
3.1.1. Organic Materials
3.1.2. Inorganic Materials
3.2. Nanoparticles
3.2.1. Silica Shell Nanoparticles
3.2.2. Silica Core Nanoparticles
4. Optical Imaging
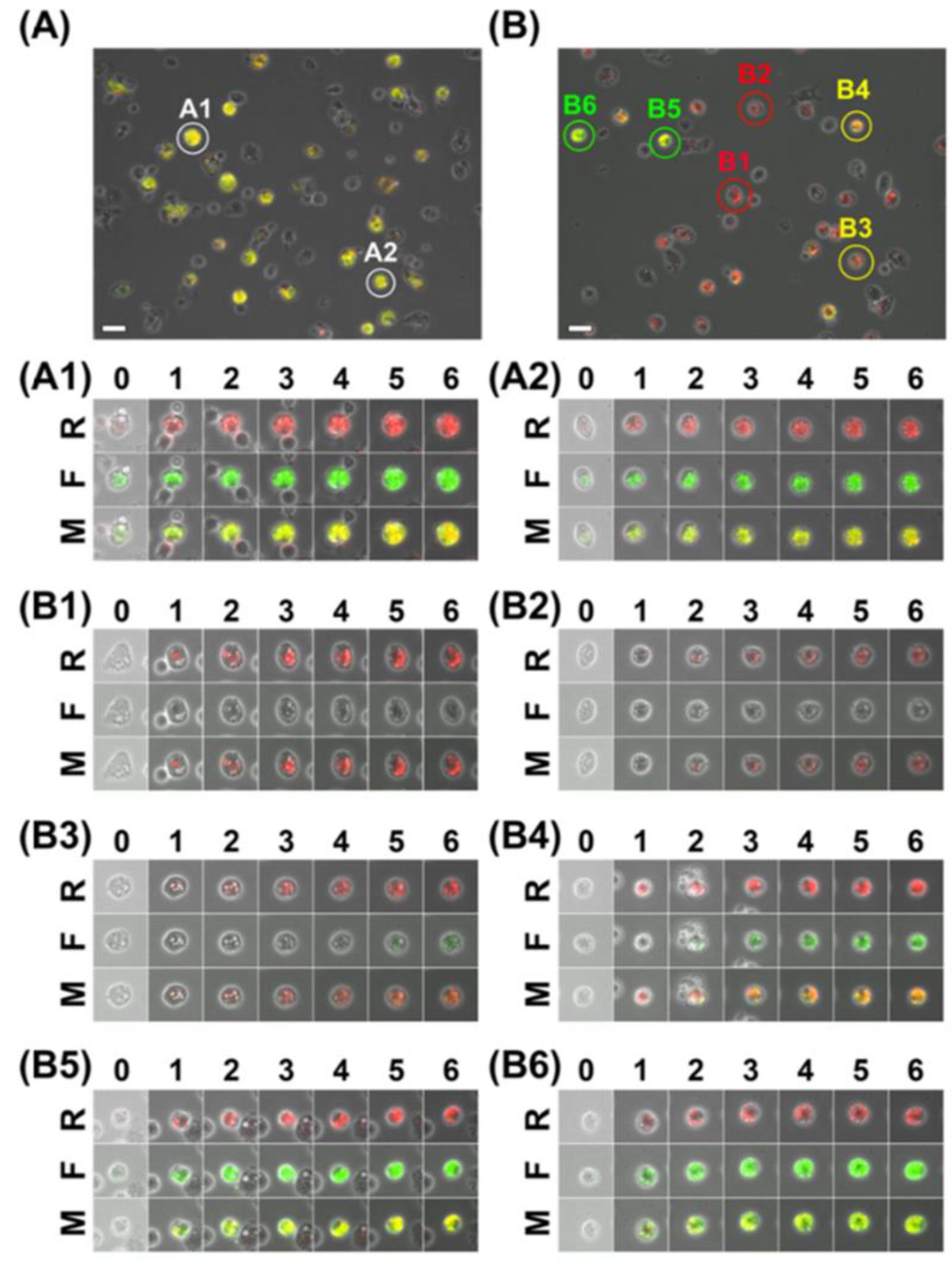
4.1. Cellular Imaging
4.1.1. Targeted Cell Imaging
4.1.2. Functional Imaging
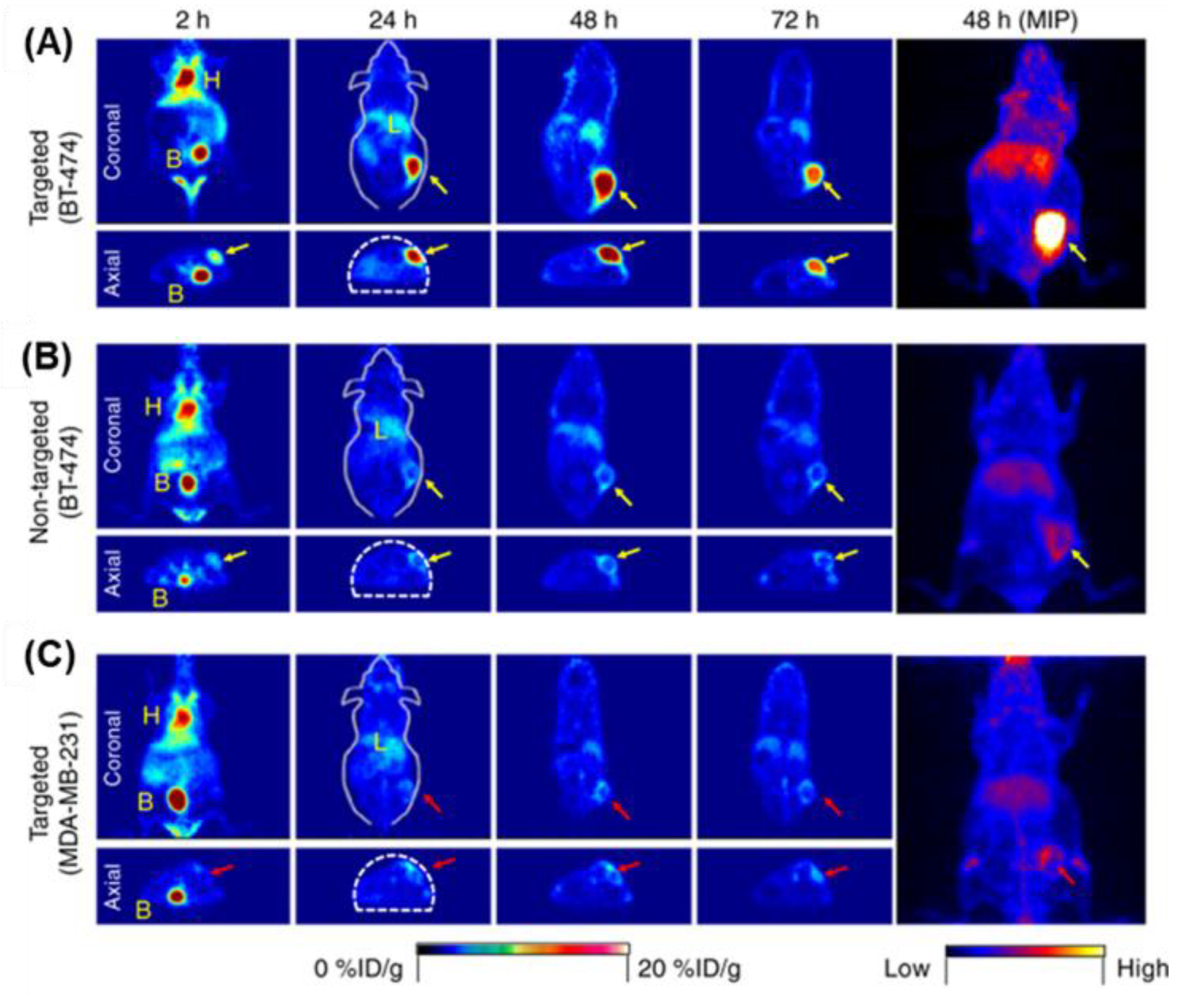
4.2. In Vivo Imaging
4.2.1. Fluorescence Imaging
4.2.2. Tumor Imaging
4.3. Multimodal Imaging
4.3.1. Optical-Magnetic Resonance (MR) Imaging
4.3.2. Optical-X ray Computed Tomography (CT) Imaging
4.3.3. Optical-Positron Emission Tomographic (PET) Imaging
5. Phototherapy
5.1. Photodynamic Therapy
5.2. Photo Hyperthermia
6. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Nanomedicine: Current status and future prospects. FASEB J. 2005, 19, 311–330. [Google Scholar] [CrossRef]
- Liu, Y.; Miyoshi, H.; Nakamura, M. Nanomedicine for drug delivery and imaging: A promising avenue for cancer therapy and diagnosis using targeted functional nanoparticles. Int. J. Cancer 2007, 120, 2527–2537. [Google Scholar] [CrossRef]
- Panwar, N.; Soehartono, A.M.; Chan, K.K.; Zeng, S.; Xu, G.; Qu, J.; Coquet, P.; Yong, K.T.; Chen, X. Nanocarbons for Biology and Medicine: Sensing, Imaging, and Drug Delivery. Chem. Rev. 2019, 119, 9559–9656. [Google Scholar] [CrossRef]
- Nam, J.; Son, S.; Park, K.S.; Zou, W.; Shea, L.D.; Moon, J.J. Cancer nanomedicine for combination cancer immunotherapy. Nat. Rev. Mater. 2019, 4, 398–414. [Google Scholar] [CrossRef]
- Yang, B.; Chen, Y.; Shi, J. Reactive oxygen species (ROS)-based nanomedicine. Chem. Rev. 2019, 119, 4881–4985. [Google Scholar] [CrossRef]
- van der Meel, R.; Sulheim, E.; Shi, Y.; Kiessling, F.; Mulder, W.J.M.; Lammers, T. Smart cancer nanomedicine. Nat. Nanotechnol. 2019, 14, 1007–1017. [Google Scholar] [CrossRef]
- Yang, H.; Tong, Z.; Sun, S.; Mao, Z. Enhancement of tumour penetration by nanomedicines through strategies based on transport processes and barriers. J. Control. Release 2020, 328, 28–44. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Zhang, Q.; Guo, J.; Zhao, X.; Zhong, Z. Robust and smart polypeptide-based nanomedicines for targeted tumor therapy. Adv. Drug Deliv. Rev. 2020, 160, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Klymchenko, A.S.; Liu, F.; Collot, M.; Anton, N. Dye-Loaded Nanoemulsions: Biomimetic Fluorescent Nanocarriers for Bioimaging and Nanomedicine. Adv. Healthc. Mater. 2020, 2001289. [Google Scholar] [CrossRef]
- Craig, M.; Jenner, A.L.; Namgung, B.; Lee, L.P.; Goldman, A. Engineering in Medicine to address the challenge of cancer drug resistance: From micro- and nanotechnologies to computational and mathematical modeling. Chem. Rev. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wong, X.Y.; Sena-Torralba, A.; Álvarez-Diduk, R.; Muthoosamy, K.; Merkoçi, A. Nanomaterials for Nanotheranostics: Tuning Their Properties According to Disease Needs. ACS Nano 2020, 14, 2585–2627. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Huang, J.; Moses, M.A. Cancer Nanomedicines in an Evolving Oncology Landscape. Trends Pharmacol. Sci. 2020, 41, 730–742. [Google Scholar] [CrossRef] [PubMed]
- Liong, M.; Lu, J.; Kovochich, M.; Xia, T.; Ruehm, S.G.; Nel, A.E.; Tamanoi, F.; Zink, J.I. Multifunctional inorganic nanoparticles for imaging, targeting, and drug delivery. ACS Nano 2008, 2, 889–896. [Google Scholar] [CrossRef]
- Martin, J.D.; Cabral, H.; Stylianopoulos, T.; Jain, R.K. Improving cancer immunotherapy using nanomedicines: Progress, opportunities and challenges. Nat. Rev. Clin. Oncol. 2020, 17, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhou, J.; Gu, Z.; Zhang, H.; Gong, Q.; Luo, K. Advances in nanomedicines for diagnosis of central nervous system disorders. Biomaterials 2020, 120492. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, C.; Yang, X.; Pu, K. Photoactivatable Protherapeutic Nanomedicine for Cancer. Adv. Mater. 2020, 32, 2002661. [Google Scholar] [CrossRef]
- Dreaden, E.C.; Alkilany, A.M.; Huang, X.; Murphy, C.J.; El-Sayed, M.A. The golden age: Gold nanoparticles for biomedicine. Chem. Soc. Rev. 2012, 41, 2740–2779. [Google Scholar] [CrossRef]
- Nakamura, M. Biomedical applications of organosilica nanoparticles toward theranostics. Nanotechnol. Rev. 2012, 1, 469–491. [Google Scholar] [CrossRef]
- Oliva, N.; Unterman, S.; Zhang, Y.; Conde, J.; Song, H.S.; Artzi, N. Personalizing Biomaterials for Precision Nanomedicine Considering the Local Tissue Microenvironment. Adv. Healthc. Mater. 2015, 4, 1584–1599. [Google Scholar] [CrossRef]
- Adiseshaiah, P.P.; Crist, R.M.; Hook, S.S.; McNeil, S.E. Nanomedicine strategies to overcome the pathophysiological barriers of pancreatic cancer. Nat. Rev. Clin. Oncol. 2016, 13, 750–765. [Google Scholar] [CrossRef]
- Kinnear, C.; Moore, T.L.; Rodriguez-Lorenzo, L.; Rothen-Rutishauser, B.; Petri-Fink, A. Form Follows Function: Nanoparticle Shape and Its Implications for Nanomedicine. Chem. Rev. 2017, 117, 11476–11521. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Kantoff, P.W.; Wooster, R.; Farokhzad, O.C. Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 2017, 17, 20–37. [Google Scholar] [CrossRef] [PubMed]
- Musetti, S.; Huang, L. Nanoparticle-Mediated Remodeling of the Tumor Microenvironment to Enhance Immunotherapy. ACS Nano 2018, 12, 11740–11755. [Google Scholar] [CrossRef] [PubMed]
- Cabral, H.; Miyata, K.; Osada, K.; Kataoka, K. Block Copolymer Micelles in Nanomedicine Applications. Chem. Rev. 2018, 118, 6844–6892. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Mammeri, F.; Ammar, S. Iron oxide and gold based magneto-plasmonic nanostructures for medical applications: A review. Nanomaterials 2018, 8, 149. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M. Organosilica Nanoparticles and Medical Imaging. Enzymes 2018, 44, 137–173. [Google Scholar]
- Kim, D.; Shin, K.; Kwon, S.G.; Hyeon, T. Synthesis and Biomedical Applications of Multifunctional Nanoparticles. Adv. Mater. 2018, 30, 1802309. [Google Scholar] [CrossRef]
- Bian, F.; Sun, L.; Cai, L.; Wang, Y.; Zhao, Y. Quantum dots from microfluidics for nanomedical application. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1567. [Google Scholar] [CrossRef]
- Ding, X.; Li, D.; Jiang, J. Gold-based inorganic nanohybrids for nanomedicine applications. Theranostics 2020, 10, 8061–8079. [Google Scholar] [CrossRef]
- Ryu, J.H.; Yoon, H.Y.; Sun, I.C.; Kwon, I.C.; Kim, K. Tumor-Targeting Glycol Chitosan Nanoparticles for Cancer Heterogeneity. Adv. Mater. 2020, 32, e2002197. [Google Scholar] [CrossRef]
- Soetaert, F.; Korangath, P.; Serantes, D.; Fiering, S.; Ivkov, R. Cancer therapy with iron oxide nanoparticles: Agents of thermal and immune therapies. Adv. Drug Deliv. Rev. 2020, 163–164, 65–83. [Google Scholar] [CrossRef] [PubMed]
- Alsaab, H.O.; Alghamdi, M.S.; Alotaibi, A.S.; Alzhrani, R.; Alwuthaynani, F.; Althobaiti, Y.S.; Almalki, A.H.; Sau, S.; Iyer, A.K. Progress in clinical trials of photodynamic therapy for solid tumors and the role of nanomedicine. Cancers 2020, 12, 2793. [Google Scholar] [CrossRef] [PubMed]
- Gomez, S.; Tsung, A.; Hu, Z. Current Targets and Bioconjugation Strategies in Photodynamic Diagnosis and Therapy of Cancer. Molecules 2020, 25, 4964. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Du, Y.; Zhang, K.; Liang, Z.; Li, J.; Yu, H.; Ren, R.; Feng, J.; Jin, Z.; Li, F.; et al. Tau-Targeted Multifunctional Nanocomposite for Combinational Therapy of Alzheimer’s Disease. ACS Nano 2018, 12, 1321–1338. [Google Scholar] [CrossRef]
- Cui, Z.; Bu, W.; Fan, W.; Zhang, J.; Ni, D.; Liu, Y.; Wang, J.; Liu, J.; Yao, Z.; Shi, J. Sensitive imaging and effective capture of Cu2+: Towards highly efficient theranostics of Alzheimer’s disease. Biomaterials 2016, 104, 158–167. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, S.M.; Park, K.H.; Mun, C.H.; Park, Y.B.; Yoo, K.H. Drug-loaded gold/iron/gold plasmonic nanoparticles for magnetic targeted chemo-photothermal treatment of rheumatoid arthritis. Biomaterials 2015, 61, 95–102. [Google Scholar] [CrossRef]
- Lu, Y.; Li, L.; Lin, Z.; Wang, L.; Lin, L.; Li, M.; Zhang, Y.; Yin, Q.; Li, Q.; Xia, H. A New Treatment Modality for Rheumatoid Arthritis: Combined Photothermal and Photodynamic Therapy Using Cu7.2S4 Nanoparticles. Adv. Healthc. Mater. 2018, 7, 1800013. [Google Scholar] [CrossRef]
- Sun, X.; Li, W.; Zhang, X.; Qi, M.; Zhang, Z.; Zhang, X.E.; Cui, Z. In Vivo Targeting and Imaging of Atherosclerosis Using Multifunctional Virus-Like Particles of Simian Virus 40. Nano Lett. 2016, 16, 6164–6171. [Google Scholar] [CrossRef]
- Nandwana, V.; Ryoo, S.R.; Kanthala, S.; McMahon, K.M.; Rink, J.S.; Li, Y.; Venkatraman, S.S.; Thaxton, C.S.; Dravid, V.P. High-Density Lipoprotein-like Magnetic Nanostructures (HDL-MNS): Theranostic Agents for Cardiovascular Disease. Chem. Mater. 2017, 29, 2276–2282. [Google Scholar] [CrossRef]
- Maria Vallet-Regi, F.T. Overview of Studies Regarding Mesoporous Silica Nanomaterials and Their Biomedical Application. Enzym. 2003, 2018, 1–10. [Google Scholar]
- Mekaru, H.; Lu, J.; Tamanoi, F. Development of mesoporous silica-based nanoparticles with controlled release capability for cancer therapy. Adv. Drug Deliv. Rev. 2015, 95, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Chen, W.; Cheng, C.A.; Deng, T.; Wang, R.; Zink, J.I. Stimuli-Responsive Nanomachines and Caps for Drug Delivery. Enzymes 2018, 43, 31–65. [Google Scholar]
- Barkat, A.; Beg, S.; Panda, S.K.; Alharbi, K.S.; Rahman, M.; Ahmed, F.J. Functionalized mesoporous silica nanoparticles in anticancer therapeutics. Semin. Cancer Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Thi, T.T.H.; Cao, V.D.; Nguyen, T.N.Q.; Hoang, D.T.; Ngo, V.C.; Nguyen, D.H. Functionalized mesoporous silica nanoparticles and biomedical applications. Mater. Sci. Eng. C 2019, 99, 631–656. [Google Scholar]
- Li, T.; Shi, S.; Goel, S.; Shen, X.; Xie, X.; Chen, Z.; Zhang, H.; Li, S.; Qin, X.; Yang, H.; et al. Recent advancements in mesoporous silica nanoparticles towards therapeutic applications for cancer. Acta Biomater. 2019, 89, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Kankala, R.K.; Han, Y.H.; Na, J.; Lee, C.H.; Sun, Z.; Wang, S.B.; Kimura, T.; Ok, Y.S.; Yamauchi, Y.; Chen, A.Z.; et al. Nanoarchitectured Structure and Surface Biofunctionality of Mesoporous Silica Nanoparticles. Adv. Mater. 2020, 32, 1907035. [Google Scholar] [CrossRef] [PubMed]
- Baeza, A.; Vallet-Regí, M. Mesoporous silica nanoparticles as theranostic antitumoral nanomedicines. Pharmaceutics 2020, 12, 957. [Google Scholar] [CrossRef] [PubMed]
- Alyassin, Y.; Sayed, E.G.; Mehta, P.; Ruparelia, K.; Arshad, M.S.; Rasekh, M.; Shepherd, J.; Kucuk, I.; Wilson, P.B.; Singh, N.; et al. Application of mesoporous silica nanoparticles as drug delivery carriers for chemotherapeutic agents. Drug Discov. Today 2020, 25, 1513–1520. [Google Scholar] [CrossRef]
- Tang, L.; Cheng, J. Nonporous silica nanoparticles for nanomedicine application. Nano Today 2013, 8, 290–312. [Google Scholar] [CrossRef]
- Xu, Z.; Ma, X.; Gao, Y.E.; Hou, M.; Xue, P.; Li, C.M.; Kang, Y. Multifunctional silica nanoparticles as a promising theranostic platform for biomedical applications. Mater. Chem. Front. 2017, 1, 1257–1272. [Google Scholar] [CrossRef]
- Blanco, I. Polysiloxanes in theranostics and drug delivery: A review. Polymers 2018, 10, 755. [Google Scholar] [CrossRef] [PubMed]
- Stöber, W.; Fink, A.; Bohn, E. Controlled Growth of Monodisperse Silica Spheres in the Micron Size Range. J. Colloid Interface Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Osseo-Asare, K.; Arriagada, F.J. Preparation of SiO2 nanoparticles in a non-ionic reverse micellar system. Colloids Surf. 1990, 50, 321–339. [Google Scholar] [CrossRef]
- Venditti, F.; Angelico, R.; Palazzo, G.; Colafemmina, G.; Ceglie, A.; Lopez, F. Preparation of nanosize silica in reverse micelles: Ethanol produced during TEOS hydrolysis affects the microemulsion structure. Langmuir 2007, 23, 10063–10068. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.A.; Padavettan, V. Synthesis of Silica nanoparticles by Sol-Gel: Size-dependent properties, surface modification, and applications in silica-polymer nanocompositesa review. J. Nanomater. 2012, 2012, 132424. [Google Scholar] [CrossRef]
- Das, S.; Jain, T.K.; Maitra, A. Inorganic-organic hybrid nanoparticles from n-octyl triethoxy silane. J. Colloid Interface Sci. 2002, 252, 82–88. [Google Scholar] [CrossRef]
- Sharma, R.K.; Das, S.; Maitra, A. Surface modified ormosil nanoparticles. J. Colloid Interface Sci. 2004, 277, 342–346. [Google Scholar] [CrossRef]
- Nakamura, M.; Ishimura, K. Synthesis and characterization of organosilica nanoparticles prepared from 3-mercaptopropyltrimethoxysilane as the single silica source. J. Phys. Chem. C 2007, 111, 18892–18898. [Google Scholar] [CrossRef]
- Vogel, R.; Surawski, P.P.T.; Littleton, B.N.; Miller, C.R.; Lawrie, G.A.; Battersby, B.J.; Trau, M. Fluorescent organosilica micro- and nanoparticles with controllable size. J. Colloid Interface Sci. 2007, 310, 144–150. [Google Scholar] [CrossRef]
- Nakamura, M.; Ishimura, K. One-pot synthesis and characterization of three kinds of thiol—Organosilica nanoparticles. Langmuir 2008, 24, 5099–5108. [Google Scholar] [CrossRef]
- Nakamura, M.; Ishimura, K. Size-controlled, one-pot synthesis, characterization, and biological applications of epoxy-organosilica particles possessing positive zeta potential. Langmuir 2008, 24, 12228–12234. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Maeda, H. A New Concept for Macromolecular Therapeutics in Cancer Chemotherapy: Mechanism of Tumoritropic Accumulation of Proteins and the Antitumor Agent Smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar] [PubMed]
- Shi, Y.; van der Meel, R.; Chen, X.; Lammers, T. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 2020, 10, 7921–7924. [Google Scholar] [CrossRef] [PubMed]
- Croissant, J.G.; Fatieiev, Y.; Khashab, N.M. Degradability and Clearance of Silicon, Organosilica, Silsesquioxane, Silica Mixed Oxide, and Mesoporous Silica Nanoparticles. Adv. Mater. 2017, 29, 1604634. [Google Scholar] [CrossRef]
- Ehrlich, H.; Demadis, K.D.; Pokrovsky, O.S.; Koutsoukos, P.G. Modern views on desilicification: Biosilica and abiotic silica dissolution in natural and artificial environments. Chem. Rev. 2010, 110, 4656–4689. [Google Scholar] [CrossRef]
- Hadipour Moghaddam, S.P.; Mohammadpour, R.; Ghandehari, H. In vitro and in vivo evaluation of degradation, toxicity, biodistribution, and clearance of silica nanoparticles as a function of size, porosity, density, and composition. J. Control. Release 2019, 311–312, 1–15. [Google Scholar] [CrossRef]
- Mohammadpour, R.; Cheney, D.L.; Grunberger, J.W.; Yazdimamaghani, M.; Jedrzkiewicz, J.; Isaacson, K.J.; Dobrovolskaia, M.A.; Ghandehari, H. One-year chronic toxicity evaluation of single dose intravenously administered silica nanoparticles in mice and their Ex vivo human hemocompatibility. J. Control. Release 2020, 324, 471–481. [Google Scholar] [CrossRef]
- Mohammadpour, R.; Yazdimamaghani, M.; Cheney, D.L.; Jedrzkiewicz, J.; Ghandehari, H. Subchronic toxicity of silica nanoparticles as a function of size and porosity. J. Control. Release 2019, 304, 216–232. [Google Scholar] [CrossRef]
- Braley, S. The Chemistry and Properties of the Medical-Grade Silicones. J. Macromol. Sci. Part A Chem. 1970, 4, 529–544. [Google Scholar] [CrossRef]
- Schmidt, H.; Scholze, H.; Kaiser, A. Principles of hydrolysis and condensation reaction of alkoxysilanes. J. Non. Cryst. Solids 1984, 63, 1–11. [Google Scholar] [CrossRef]
- Schmidt, H. New type of non-crystalline solids between inorganic and organic materials. J. Non. Cryst. Solids 1985, 73, 681–691. [Google Scholar] [CrossRef]
- Wilkes, G.L.; Orler, B.; Huang, H. Polym. Prepr. Chem. Soc. Dir. Polym. Chem. 1985, 26, 300. [Google Scholar]
- Doura, T.; Nishio, T.; Tamanoi, F.; Nakamura, M. Relationship between the glutathione-responsive degradability of thiol-organosilica nanoparticles and the chemical structures. J. Mater. Res. 2019, 34, 1266–1278. [Google Scholar] [CrossRef]
- Zhao, X.; Tapec-Dytioco, R.; Tan, W. Ultrasensitive DNA detection using highly fluorescent bioconjugated nanoparticles. J. Am. Chem. Soc. 2003, 125, 11474–11475. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhou, J. Improving the signal sensitivity and photostability of DNA hybridizations on microarrays by using dye-doped core-shell silica nanoparticles. Anal. Chem. 2004, 76, 5302–5312. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, W.; Tan, W. Bioconjugated silica nanoparticles: Development and applications. Nano Res. 2008, 1, 99–115. [Google Scholar] [CrossRef]
- Burns, A.; Ow, H.; Wiesner, U. Fluorescent core–shell silica nanoparticles: Towards “Lab on a Particle” architectures for nanobiotechnology. Chem. Soc. Rev. 2006, 35, 1028–1042. [Google Scholar] [CrossRef]
- Wang, F.; Tan, W.B.; Zhang, Y.; Fan, X.; Wang, M. Luminescent nanomaterials for biological labelling. Nanotechnology 2006, 17, R1. [Google Scholar] [CrossRef]
- Quan, B.; Choi, K.; Kim, Y.H.; Kang, K.W.; Chung, D.S. Near infrared dye indocyanine green doped silica nanoparticles for biological imaging. Talanta 2012, 99, 387–393. [Google Scholar] [CrossRef]
- Nakamura, M.; Awaad, A.; Hayashi, K.; Ochiai, K.; Ishimura, K. Thiol-organosilica particles internally functionalized with propidium iodide as a multicolor fluorescence and X-ray computed tomography probe and application for non-invasive functional gastrointestinal tract imaging. Chem. Mater. 2012, 24, 3772–3779. [Google Scholar] [CrossRef]
- van Blaaderen, A.; Vrij, A. Synthesis and Characterization of Colloidal Dispersions of Fluorescent, Monodisperse Silica Spheres. Langmuir 1992, 8, 2921–2931. [Google Scholar] [CrossRef]
- Nyffenegger, R.; Quellet, C.; Ricka, J. Synthesis of fluorescent, monodisperse, colloidal silica particles. J. Colloid Interface Sci. 1993, 159, 150–157. [Google Scholar] [CrossRef]
- Nakamura, M.; Shono, M.; Ishimura, K. Synthesis, characterization, and biological applications of multifluorescent silica nanoparticles. Anal. Chem. 2007, 79, 6507–6514. [Google Scholar] [CrossRef]
- Yari, A.; Saidikhah, M. Dye doped eosin yellowish silica nanoparticles as novel fluorophore for a peroxyoxalate chemiluminescence system. J. Fluoresc. 2012, 22, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Fabian, J.; Nakazumi, H.; Matsuoka, M. Near-infrared absorbing dyes. Chem. Rev. 2002, 92, 1197–1226. [Google Scholar] [CrossRef]
- Accomasso, L.; Rocchietti, E.C.; Raimondo, S.; Catalano, F.; Alberto, G.; Giannitti, A.; Minieri, V.; Turinetto, V.; Orlando, L.; Saviozzi, S.; et al. Fluorescent silica nanoparticles improve optical imaging of stem cells allowing direct discrimination between live and early-stage apoptotic cells. Small 2012, 8, 3192–3200. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Ma, K.; Madajewski, B.; Zhuang, L.; Zhang, L.; Rickert, K.; Marelli, M.; Yoo, B.; Turker, M.Z.; Overholtzer, M.; et al. Ultrasmall targeted nanoparticles with engineered antibody fragments for imaging detection of HER2-overexpressing breast cancer. Nat. Commun. 2018, 9, 1–11. [Google Scholar]
- Murray, K.; Cao, Y.C.; Ali, S.; Hanley, Q. Lanthanide doped silica nanoparticles applied to multiplexed immunoassays. Analyst 2010, 135, 2132–2138. [Google Scholar] [CrossRef]
- Shao, Y.Z.; Liu, L.Z.; Song, S.Q.; Cao, R.H.; Liu, H.; Cui, C.Y.; Li, X.; Bie, M.J.; Li, L. A novel one-step synthesis of Gd3+-incorporated mesoporous SiO2 nanoparticles for use as an efficient MRI contrast agent. Contrast Media Mol. Imaging 2011, 6, 110–118. [Google Scholar] [CrossRef]
- Davies, G.L.; O’Brien, J.; Gun’ko, Y.K. Rare Earth Doped Silica Nanoparticles via Thermolysis of a Single Source Metallasilsesquioxane Precursor. Sci. Rep. 2017, 7, 45862. [Google Scholar] [CrossRef]
- Yang, J.; Sandoval, S.; Alfaro, J.G.; Aschemeyer, S.; Liberman, A.; Martin, D.T.; Makale, M.; Kummel, A.C.; Trogler, W.C. Red-luminescent europium (III) doped silica nanoshells: Synthesis, characterization, and their interaction with HeLa cells. J. Biomed. Opt. 2011, 16, 066012. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wu, Z.; Xu, J.; Liang, J.; Li, J.; Yang, W. Tuning the emission properties of Ru(phen)32+ doped silica nanoparticles by changing the addition time of the dye during the stöber process. Langmuir 2010, 26, 6657–6662. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Z.; Cai, L.; Dong, X.M.; Tang, H.W.; Pang, D.W. Covalent conjugation of avidin with dye-doped silica nanopaticles and preparation of high density avidin nanoparticles as photostable bioprobes. Biosens. Bioelectron. 2012, 37, 75–81. [Google Scholar] [CrossRef]
- Zhou, L.; Huang, J.; Yu, B.; Liu, Y.; You, T. A Novel Electrochemiluminescence Immunosensor for the Analysis of HIV-1 p24 Antigen Based on P-RGO@Au@Ru-SiO2 Composite. ACS Appl. Mater. Interfaces 2015, 7, 24438–24445. [Google Scholar] [CrossRef]
- Bagwe, R.P.; Yang, C.; Hilliard, L.R.; Tan, W. Optimization of dye-doped silica nanoparticles prepared using a reverse microemulsion method. Langmuir 2004, 20, 8336–8342. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.M.; Shi, L.; Quina, F.H.; Rosenzweig, Z. Stöber synthesis of monodispersed luminescent silica nanoparticles for bioanalytical assays. Langmuir 2005, 21, 4277–4280. [Google Scholar] [CrossRef]
- Zanarini, S.; Rampazzo, E.; Ciana, L.D.; Marcaccio, M.; Marzocchi, E.; Montalti, M.; Paolucci, F.; Prodi, L. Ru(bpy)3 covalently doped silica nanoparticles as multicenter tunable structures for electrochemiluminescence amplification. J. Am. Chem. Soc. 2009, 131, 2260–2267. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Yang, X. One-step synthesis of Ru(2,2′-Bipyridine)3Cl2-immobilized silica nanoparticles for use in electrogenerated chemiluminescence detection. Adv. Funct. Mater. 2007, 17, 1353–1358. [Google Scholar] [CrossRef]
- Nakamura, M.; Ozaki, S.; Abe, M.; Matsumoto, T.; Ishimura, K. One-pot synthesis and characterization of dual fluorescent thiol-organosilica nanoparticles as non-photoblinking quantum dots and their applications for biological imaging. J. Mater. Chem. 2011, 21, 4689–4695. [Google Scholar] [CrossRef]
- Selvan, S.T.; Yang Tan, T.T.; Kee Yi, D.; Jana, N.R. Functional and multifunctional nanoparticles for bioimaging and biosensing. Langmuir 2010, 26, 11631–11641. [Google Scholar] [CrossRef]
- Law, W.C.; Yong, K.T.; Roy, I.; Xu, G.X.; Ding, H.; Bergey, E.J.; Zeng, H.; Prasad, P.N. Optically and magnetically doped organically modified silica nanoparticles as efficient magnetically guided biomarkers for two-photon imaging of live cancer cells. J. Phys. Chem. C 2008, 112, 7972–7977. [Google Scholar] [CrossRef]
- Jun, B.H.; Hwang, D.W.; Jung, H.S.; Jang, J.; Kim, H.; Kang, H.; Kang, T.; Kyeong, S.; Lee, H.; Jeong, D.H.; et al. Ultrasensitive, biocompatible, quantum-dot-embedded silica nanoparticles for bioimaging. Adv. Funct. Mater. 2012, 22, 1843–1849. [Google Scholar] [CrossRef]
- Ow, H.; Larson, D.R.; Srivastava, M.; Baird, B.A.; Webb, W.W.; Wiesner, U. Bright and stable core-shell fluorescent silica nanoparticles. Nano Lett. 2005, 5, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Wang, D.; Cai, F.; Zhan, Q.; Wang, Y.; He, S. Photosensitizer encapsulated organically modified silica nanoparticles for direct two-photon photodynamic therapy and In Vivo functional imaging. Biomaterials 2012, 33, 4851–4860. [Google Scholar] [CrossRef]
- Thorat, A.V.; Ghoshal, T.; Chen, L.; Holmes, J.D.; Morris, M.A. Synthesis and stability of IR-820 and FITC doped silica nanoparticles. J. Colloid Interface Sci. 2017, 490, 294–302. [Google Scholar] [CrossRef]
- Nakamura, M.; Hayashi, K.; Nakamura, J.; Mochizuki, C.; Murakami, T.; Miki, H.; Ozaki, S.; Abe, M. Near-Infrared Fluorescent Thiol-Organosilica Nanoparticles That Are Functionalized with IR-820 and Their Applications for Long-Term Imaging of in Situ Labeled Cells and Depth-Dependent Tumor in Vivo Imaging. Chem. Mater. 2020, 32, 7201–7214. [Google Scholar] [CrossRef]
- Bouzigues, C.; Gacoin, T.; Alexandrou, A. Biological applications of rare-earth based nanoparticles. ACS Nano 2011, 5, 8488–8505. [Google Scholar] [CrossRef]
- Guerrero-Martínez, A.; Pérez-Juste, J.; Liz-Marzán, L.M. Recent progress on silica coating of nanoparticles and related nanomaterials. Adv. Mater. 2010, 22, 1182–1195. [Google Scholar] [CrossRef]
- Ohmori, M.; Matijević, E. Preparation and properties of uniform coated colloidal particles. VII. Silica on hematite. J. Colloid Interface Sci. 1992, 150, 594–598. [Google Scholar] [CrossRef]
- Liz-Marzán, L.M.; Giersig, M.; Mulvaney, P. Synthesis of nanosized gold-silica core-shell particles. Langmuir 1996, 12, 4329–4335. [Google Scholar] [CrossRef]
- Lee, J.; Lee, Y.; Youn, J.K.; Na, H.B.; Yu, T.; Kim, H.; Lee, S.M.; Koo, Y.M.; Kwak, J.H.; Park, H.G.; et al. Simple synthesis of functionalized superparamagnetic magnetite/silica core/shell nanoparticles and their application as magnetically separable high-performance biocatalysts. Small 2008, 4, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Graf, C.; Vossen, D.L.J.; Imhof, A.; Van Blaaderen, A. A general method to coat colloidal particles with silica. Langmuir 2003, 19, 6693–6700. [Google Scholar] [CrossRef]
- Yoon, T.J.; Yu, K.N.; Kim, E.; Kim, J.S.; Kim, B.G.; Yun, S.H.; Sohn, B.H.; Cho, M.H.; Lee, J.K.; Park, S.B. Specific targeting, cell sorting, and bioimaging with smart magnetic silica core-shell nanomaterials. Small 2006, 2, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, S.J.; Jackson, J.B.; Westcott, S.L.; Halas, N.J. Infrared extinction properties of gold nanoshells. Appl. Phys. Lett. 1999, 75, 2897–2899. [Google Scholar] [CrossRef]
- Pham, T.; Jackson, J.B.; Halas, N.J.; Lee, T.R. Preparation and characterization of gold nanoshells coated with self-assembled monolayers. Langmuir 2002, 18, 4915–4920. [Google Scholar] [CrossRef]
- Ji, X.; Shao, R.; Elliott, A.M.; Jason Stafford, R.; Esparza-Coss, E.; Bankson, J.A.; Liang, G.; Luo, Z.P.; Park, K.; Markert, J.T.; et al. Bifunctional Gold Nanoshells with a Superparamagnetic Iron Oxide-Silica Core Suitable for Both MR Imaging and Photothermal Therapy. J. Phys. Chem. C 2007, 111, 6245–6251. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Chen, E.; Ye, Y.; Xu, S.; Guo, T. Highly Stabilized Gradient Alloy Quantum Dots and Silica Hybrid Nanospheres by Core Double Shells for Photoluminescence Devices. J. Phys. Chem. Lett. 2020, 11, 1428–1434. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.C.; Wang, H.; Qing, M.; Qiu, C.W.; Yang, Y.; Li, Y.W. Preparation and characterization of SiO2@Fe2O3 core-shell catalysts. Ranliao Huaxue Xuebao/J. Fuel Chem. Technol. 2015, 43, 692–700. [Google Scholar] [CrossRef]
- Choma, J.; Dziura, A.; Jamioła, D.; Nyga, P.; Jaroniec, M. Preparation and properties of silica-gold core-shell particles. Colloids Surf. A Physicochem. Eng. Asp. 2011, 373, 167–171. [Google Scholar] [CrossRef]
- Kwok, W.L.J.; Crivoi, D.G.; Chen, C.; Buffet, J.C.; O’Hare, D. Silica@layered double hydroxide core-shell hybrid materials. Dalt. Trans. 2018, 47, 143–149. [Google Scholar] [CrossRef]
- Insin, N.; Tracy, J.B.; Lee, H.; Zimmer, J.P.; Westervelt, R.M.; Bawendi, M.G. Incorporation of iron oxide nanoparticles and quantum dots into silica microspheres. ACS Nano 2008, 2, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Herr, J.K.; Smith, J.E.; Medley, C.D.; Shangguan, D.; Tan, W. Aptamer-conjugated nanoparticles for selective collection and detection of cancer cells. Anal. Chem. 2006, 78, 2918–2924. [Google Scholar] [CrossRef]
- Medley, C.D.; Bamrungsap, S.; Tan, W.; Smith, J.E. Aptamer-conjugated nanoparticles for cancer cell detection. Anal. Chem. 2011, 83, 727–734. [Google Scholar] [CrossRef]
- Cai, L.; Chen, Z.Z.; Chen, M.Y.; Tang, H.W.; Pang, D.W. MUC-1 aptamer-conjugated dye-doped silica nanoparticles for MCF-7 cells detection. Biomaterials 2013, 34, 371–381. [Google Scholar] [CrossRef]
- Kumar, R.; Roy, I.; Ohulchanskyy, T.Y.; Goswami, L.N.; Bonoiu, A.C.; Bergey, E.J.; Tramposch, K.M.; Maitra, A.; Prasad, P.N. Covalently dye-linked, surface-controlled, and bioconjugated organically modified silica nanoparticles as targeted probes for optical imaging. ACS Nano 2008, 2, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Miyamoto, K.; Hayashi, K.; Awaad, A.; Ochiai, M.; Ishimura, K. Time-lapse fluorescence imaging and quantitative single cell and endosomal analysis of peritoneal macrophages using fluorescent organosilica nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Hayashi, K.; Nakano, M.; Kanadani, T.; Miyamoto, K.; Kori, T.; Horikawa, K. Identification of polyethylene glycol-resistant macrophages on stealth imaging in vitro using fluorescent organosilica nanoparticles. ACS Nano 2015, 9, 1058–1071. [Google Scholar] [CrossRef] [PubMed]
- Alberto, G.; Miletto, I.; Viscardi, G.; Caputo, G.; Latterini, L.; Coluccia, S.; Martra, G. Hybrid Cyanine-Silica Nanoparticles: Homogeneous Photoemission Behavior of Entrapped Fluorophores and Consequent High Brightness Enhancement. J. Phys. Chem. C 2009, 113, 21048–21053. [Google Scholar] [CrossRef]
- Wu, C.; Wu, Y.; Jin, Y.; Zhu, P.; Shi, W.; Li, J.; Wu, Q.; Zhang, Q.; Han, Y.; Zhao, X. Endosomal/lysosomal location of organically modified silica nanoparticles following caveolae-mediated endocytosis. RSC Adv. 2019, 9, 13855–13862. [Google Scholar] [CrossRef]
- Choi, J.; Burns, A.A.; Williams, R.M.; Zhou, Z.; Flesken-Nikitin, A.; Zipfel, W.R.; Wiesner, U.; Nikitin, A.Y. Core-shell silica nanoparticles as fluorescent labels for nanomedicine. J. Biomed. Opt. 2007, 12, 064007. [Google Scholar] [CrossRef]
- Hayashi, K.; Nakamura, M.; Ishimura, K. Silica-porphyrin hybrid nanotubes for in vivo cell tracking by near-infrared fluorescence imaging. Chem. Commun. 2012, 48, 3830–3832. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Song, F.; Zhang, B.; Ning, H.; Cui, J.; Peng, X. Improving the brightness and photostability of NIR fluorescent silica nanoparticles through rational fine-tuning of the covalent encapsulation methods. J. Mater. Chem. B 2017, 5, 5278–5283. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Kim, G.R.; Yoon, J.; Kim, S.E.; Yoo, J.S.; Piao, Y. In vivo delineation of glioblastoma by targeting tumor-associated macrophages with near-infrared fluorescent silica coated iron oxide nanoparticles in orthotopic xenografts for surgical guidance. Sci. Rep. 2018, 8, 1–12. [Google Scholar]
- Dong, W.; Wen, J.; Li, Y.; Wang, C.; Sun, S.; Shang, D. Targeted antimicrobial peptide delivery in vivo to tumor with near infrared photoactivated mesoporous silica nanoparticles. Int. J. Pharm. 2020, 588, 119767. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Roy, I.; Ohulchanskky, T.Y.; Vathy, L.A.; Bergey, E.J.; Sajjad, M.; Prasad, P.N. In vivo biodistribution and clearance studies using multimodal organically modified silica nanoparticles. ACS Nano 2010, 4, 699–708. [Google Scholar] [CrossRef]
- He, X.; Nie, H.; Wang, K.; Tan, W.; Wu, X.; Zhang, P. In vivo study of biodistribution and urinary excretion of surface-modified silica nanoparticles. Anal. Chem. 2008, 80, 9597–9603. [Google Scholar] [CrossRef] [PubMed]
- Adumeau, L.; Genevois, C.; Roudier, L.; Schatz, C.; Couillaud, F.; Mornet, S. Impact of surface grafting density of PEG macromolecules on dually fluorescent silica nanoparticles used for the in vivo imaging of subcutaneous tumors. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 1587–1596. [Google Scholar] [CrossRef]
- Tang, L.; Fan, T.M.; Borst, L.B.; Cheng, J. Synthesis and biological response of size-specific, monodisperse drug-silica nanoconjugates. ACS Nano 2012, 6, 3954–3966. [Google Scholar] [CrossRef]
- Awaad, A.; Nakamura, M.; Ishimura, K. Imaging of size-dependent uptake and identification of novel pathways in mouse Peyer’s patches using fluorescent organosilica particles. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 627–636. [Google Scholar] [CrossRef]
- Karaman, D.Ş.; Sarparanta, M.P.; Rosenholm, J.M.; Airaksinen, A.J. Multimodality Imaging of Silica and Silicon Materials In Vivo. Adv. Mater. 2018, 30, 1703651. [Google Scholar] [CrossRef]
- Nakamura, M.; Hayashi, K.; Kubo, H.; Kanadani, T.; Harada, M.; Yogo, T. Relaxometric property of organosilica nanoparticles internally functionalized with iron oxide and fluorescent dye for multimodal imaging. J. Colloid Interface Sci. 2017, 492, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Hayashi, K.; Kubo, H.; Harada, M.; Izumi, K.; Tsuruo, Y.; Yogo, T. Mesoscopic Multimodal Imaging Provides New Insight to Tumor Tissue Evaluation: An Example of Macrophage Imaging of Hepatic Tumor using Organosilica Nanoparticles. Sci. Rep. 2017, 7, 1–10. [Google Scholar]
- Li, Y.; Xu, D.; Chan, H.N.; Poon, C.Y.; Ho, S.L.; Li, H.W.; Wong, M.S. Dual-Modal NIR-Fluorophore Conjugated Magnetic Nanoparticle for Imaging Amyloid-β Species In Vivo. Small 2018, 14, 1800901. [Google Scholar] [CrossRef]
- Shao, D.; Xu, K.; Song, X.; Hu, J.; Yang, W.; Wang, C. Effective adsorption and separation of lysozyme with PAA-modified Fe3O4@silica core/shell microspheres. J. Colloid Interface Sci. 2009, 336, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Hui, C.; Shen, C.; Tian, J.; Bao, L.; Ding, H.; Li, C.; Tian, Y.; Shi, X.; Gao, H.J. Core-shell Fe3O4@SiO2 nanoparticles synthesized with well-dispersed hydrophilic Fe3O4 seeds. Nanoscale 2011, 3, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.L.; Zhang, Y.X.; Wang, S.; Xu, J.M.; Xu, S.C.; Li, G.H. Fe3O4@SiO2 core/shell nanoparticles: The silica coating regulations with a single core for different core sizes and shell thicknesses. Chem. Mater. 2012, 24, 4572–4580. [Google Scholar] [CrossRef]
- Alipour, A.; Soran-Erdem, Z.; Utkur, M.; Sharma, V.K.; Algin, O.; Saritas, E.U.; Demir, H.V. A new class of cubic SPIONs as a dual-mode T1 and T2 contrast agent for MRI. Magn. Reson. Imaging 2018, 49, 16–24. [Google Scholar] [CrossRef]
- Santra, S.; Bagwe, R.P.; Dutta, D.; Stanley, J.T.; Walter, G.A.; Tan, W.; Moudgil, B.M.; Mericle, R.A. Synthesis and characterization of fluorescent, radio-opaque, and paramagnetic silica nanoparticles for multimodal bioimaging applications. Adv. Mater. 2005, 17, 2165–2169. [Google Scholar] [CrossRef]
- Zhao, C.; Zhu, D.; Gao, W.; Han, T.; Peng, L.; Tu, M. Effect of doping Gd3+ on crystal structure and luminescent properties of Sr2SiO4:Eu2+ phosphor. J. Rare Earths 2015, 33, 693–699. [Google Scholar] [CrossRef]
- Hayashi, K.; Nakamura, M.; Miki, H.; Ozaki, S.; Abe, M.; Matsumoto, T.; Ishimura, K. Gold nanoparticle cluster-plasmon-enhanced fluorescent silica core-shell nanoparticles for X-ray computed tomography-fluorescence dual-mode imaging of tumors. Chem. Commun. 2013, 49, 5334–5336. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Wataru, S.; Yogo, T. Iodinated silica/porphyrin hybrid nanoparticles for X-ray computed tomography/fluorescence dual-modal imaging of tumors. J. Asian Ceram. Soc. 2014, 2, 429–434. [Google Scholar] [CrossRef][Green Version]
- Yamaguchi, H.; Tsuchimochi, M.; Hayama, K.; Kawase, T.; Tsubokawa, N. Dual-labeled near-infrared/99mTc imaging probes using PAMAM-coated silica nanoparticles for the imaging of HER2-expressing cancer cells. Int. J. Mol. Sci. 2016, 17, 1086. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.; Choi, J.W.; Ko, G.; Baik, S.; Kim, D.; Park, O.K.; Lee, K.; Cho, H.R.; Han, S.I.; Lee, S.H.; et al. Multifunctional nanoparticles as a tissue adhesive and an injectable marker for image-guided procedures. Nat. Commun. 2017, 8, 1–12. [Google Scholar]
- Hayashi, K.; Nakamura, M.; Ishimura, K. Near-Infrared Fluorescent Silica-Coated Gold Nanoparticle Clusters for X-ray Computed Tomography/Optical Dual Modal Imaging of the Lymphatic System. Adv. Healthc. Mater. 2013, 2, 756–763. [Google Scholar] [CrossRef]
- Shaffer, T.M.; Harmsen, S.; Khwaja, E.; Kircher, M.F.; Drain, C.M.; Grimm, J. Stable Radiolabeling of Sulfur-Functionalized Silica Nanoparticles with Copper-64. Nano Lett. 2016, 16, 5601–5604. [Google Scholar] [CrossRef] [PubMed]
- Phillips, E.; Penate-Medina, O.; Zanzonico, P.B.; Carvajal, R.D.; Mohan, P.; Ye, Y.; Humm, J.; Gönen, M.; Kalaigian, H.; Schöder, H.; et al. Clinical translation of an ultrasmall inorganic optical-PET imaging nanoparticle probe. Sci. Transl. Med. 2014, 6, 260. [Google Scholar] [CrossRef]
- Chen, F.; Ma, K.; Benezra, M.; Zhang, L.; Cheal, S.M.; Phillips, E.; Yoo, B.; Pauliah, M.; Overholtzer, M.; Zanzonico, P.; et al. Cancer-Targeting Ultrasmall Silica Nanoparticles for Clinical Translation: Physicochemical Structure and Biological Property Correlations. Chem. Mater. 2017, 29, 8766–8779. [Google Scholar] [CrossRef] [PubMed]
- Dolmans, D.E.J.G.J.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar] [CrossRef]
- Overholt, B.F.; Wang, K.K.; Burdick, J.S.; Lightdale, C.J.; Kimmey, M.; Nava, H.R.; Sivak, M.V.; Nishioka, N.; Barr, H.; Marcon, N.; et al. Five-year efficacy and safety of photodynamic therapy with Photofrin in Barrett’s high-grade dysplasia. Gastrointest. Endosc. 2007, 66, 460–468. [Google Scholar] [CrossRef]
- Wieder, M.E.; Hone, D.C.; Cook, M.J.; Handsley, M.M.; Gavrilovic, J.; Russell, D.A. Intracellular photodynamic therapy with photosensitizer-nanoparticle conjugates: Cancer therapy using a ‘Trojan horse’. Photochem. Photobiol. Sci. 2006, 5, 727–734. [Google Scholar] [CrossRef]
- Roy, I.; Ohulchanskyy, T.Y.; Pudavar, H.E.; Bergey, E.J.; Oseroff, A.R.; Morgan, J.; Dougherty, T.J.; Prasad, P.N. Ceramic-based nanoparticles entrapping water-insoluble photosensitizing anticancer drugs: A novel drug-carrier system for photodynamic therapy. J. Am. Chem. Soc. 2003, 125, 7860–7865. [Google Scholar] [CrossRef] [PubMed]
- Couleaud, P.; Morosini, V.; Frochot, C.; Richeter, S.; Raehm, L.; Durand, J.O. Silica-based nanoparticles for photodynamic therapy applications. Nanoscale 2010, 2, 1083–1095. [Google Scholar] [CrossRef]
- Cho, M.H.; Li, Y.; Lo, P.C.; Lee, H.; Choi, Y. Fucoidan-Based Theranostic Nanogel for Enhancing Imaging and Photodynamic Therapy of Cancer. Nano Micro Lett. 2020, 12, 47. [Google Scholar] [CrossRef]
- Yan, F.; Kopelman, R. The Embedding of Meta-tetra(Hydroxyphenyl)-Chlorin into Silica Nanoparticle Platforms for Photodynamic Therapy and Their Singlet Oxygen Production and pH-dependent Optical Properties. Photochem. Photobiol. 2003, 78, 587. [Google Scholar] [CrossRef]
- Li, Z.; Wang, J.; Chen, J.; Lei, W.; Wang, X.; Zhang, B. Hypocrellin B doped and pH-responsive silica nanoparticles for photodynamic therapy. Sci. China Chem. 2010, 53, 1994–1999. [Google Scholar] [CrossRef]
- Simon, V.; Devaux, C.; Darmon, A.; Donnet, T.; Thiénot, E.; Germain, M.; Honnorat, J.; Duval, A.; Pottier, A.; Borghi, E.; et al. Pp IX silica nanoparticles demonstrate differential interactions with in vitro tumor cell lines and in vivo mouse models of human cancers. Photochem. Photobiol. 2010, 86, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Kohle, F.F.E.; Li, S.; Turker, M.Z.; Wiesner, U.B. Ultrasmall PEGylated and Targeted Core-Shell Silica Nanoparticles Carrying Methylene Blue Photosensitizer. ACS Biomater. Sci. Eng. 2020, 6, 256–264. [Google Scholar] [CrossRef]
- Kim, S.; Ohulchanskyy, T.Y.; Pudavar, H.E.; Pandey, R.K.; Prasad, P.N. Organically modified silica nanoparticles co-encapsulating photosensitizing drug and aggregation-enhanced two-photon absorbing fluorescent dye aggregates for two-photon photodynamic therapy. J. Am. Chem. Soc. 2007, 129, 2669–2675. [Google Scholar] [CrossRef]
- Croissant, J.G.; Zink, J.I.; Raehm, L.; Durand, J.O. Two-Photon-Excited Silica and Organosilica Nanoparticles for Spatiotemporal Cancer Treatment. Adv. Healthc. Mater. 2018, 7, 1701248. [Google Scholar] [CrossRef]
- Roti, J.L. Cellular responses to hyperthermia (40–46 °C): Cell killing and molecular events. Int. J. Hyperth. 2008, 24, 3–15. [Google Scholar] [CrossRef]
- Huang, L.; Li, Y.; Du, Y.; Zhang, Y.; Wang, X.; Ding, Y.; Yang, X.; Meng, F.; Tu, J.; Luo, L.; et al. Mild photothermal therapy potentiates anti-PD-L1 treatment for immunologically cold tumors via an all-in-one and all-in-control strategy. Nat. Commun. 2019, 10, 1–15. [Google Scholar]
- Liang, S.; Zhao, Y.; Xu, S.; Wu, X.; Chen, J.; Wu, M.; Zhao, J.X. A silica-gold-silica nanocomposite for photothermal therapy in the near-infrared region. ACS Appl. Mater. Interfaces 2015, 7, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, L.R.; Stafford, R.J.; Bankson, J.A.; Sershen, S.R.; Rivera, B.; Price, R.E.; Hazle, J.D.; Halas, N.J.; West, J.L. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc. Natl. Acad. Sci. USA 2003, 100, 13549–13554. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, D.P.; Hirsch, L.R.; Halas, N.J.; Payne, J.D.; West, J.L. Photo-thermal tumor ablation in mice using near infrared-absorbing nanoparticles. Cancer Lett. 2004, 209, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Shan, G.; Weissleder, R.; Hilderbrand, S.A. Upconverting organic dye doped core-shell nano-composites for dual-modality NIR imaging and photo-thermal therapy. Theranostics 2013, 3, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Nagesetti, A.; McGoron, A.J. Multifunctional organically modified silica nanoparticles for chemotherapy, adjuvant hyperthermia and near infrared imaging. Colloids Surf. B Biointerfaces 2016, 147, 492–500. [Google Scholar] [CrossRef]
- Zhou, Z.; Sun, Y.; Shen, J.; Wei, J.; Yu, C.; Kong, B.; Liu, W.; Yang, H.; Yang, S.; Wang, W. Iron/iron oxide core/shell nanoparticles for magnetic targeting MRI and near-infrared photothermal therapy. Biomaterials 2014, 35, 7470–7478. [Google Scholar] [CrossRef]
- Li, X.; Lovell, J.F.; Yoon, J.; Chen, X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 2020, 17, 657–674. [Google Scholar] [CrossRef]
- Nemec, S.; Kralj, S.; Wilhelm, C.; Abou-Hassan, A.; Rols, M.P.; Kolosnjaj-Tabi, J. Comparison of iron oxide nanoparticles in photothermia and magnetic hyperthermia: Effects of clustering and silica encapsulation on nanoparticles’ heating yield. Appl. Sci. 2020, 10, 7322. [Google Scholar] [CrossRef]
- Benezra, M.; Penate-Medina, O.; Zanzonico, P.B.; Schaer, D.; Ow, H.; Burns, A.; DeStanchina, E.; Longo, V.; Herz, E.; Iyer, S.; et al. Multimodal silica nanoparticles are effective cancer-targeted probes in a model of human melanoma. J. Clin. Investig. 2011, 121, 2768–2780. [Google Scholar] [CrossRef]
- Mignot, A.; Truillet, C.; Lux, F.; Sancey, L.; Louis, C.; Denat, F.; Boschetti, F.; Bocher, L.; Gloter, A.; Stéphan, O.; et al. A top-down synthesis route to ultrasmall multifunctional Gd-based silica nanoparticles for theranostic applications. Chem. A Eur. J. 2013, 19, 6122–6136. [Google Scholar] [CrossRef] [PubMed]
- Kotb, S.; Detappe, A.; Lux, F.; Appaix, F.; Barbier, E.L.; Tran, V.L.; Plissonneau, M.; Gehan, H.; Lefranc, F.; Rodriguez-Lafrasse, C.; et al. Gadolinium-based nanoparticles and radiation therapy for multiple brain melanoma metastases: Proof of concept before phase I trial. Theranostics 2016, 6, 418–427. [Google Scholar] [CrossRef]
- Detappe, A.; Thomas, E.; Tibbitt, M.W.; Kunjachan, S.; Zavidij, O.; Parnandi, N.; Reznichenko, E.; Lux, F.; Tillement, O.; Berbeco, R. Ultrasmall Silica-Based Bismuth Gadolinium Nanoparticles for Dual Magnetic Resonance-Computed Tomography Image Guided Radiation Therapy. Nano Lett. 2017, 17, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Lux, F.; Tran, V.L.; Thomas, E.; Dufort, S.; Rossetti, F.; Martini, M.; Truillet, C.; Doussineau, T.; Bort, G.; Denat, F.; et al. AGuiX® from bench to bedside-transfer of an ultrasmall theranostic gadolinium-based nanoparticle to clinical medicine. Br. J. Radiol. 2019, 92, 20180365. [Google Scholar] [CrossRef] [PubMed]
- Bort, G.; Lux, F.; Dufort, S.; Crémillieux, Y.; Verry, C.; Tillement, O. EPR-mediated tumor targeting using ultrasmall-hybrid nanoparticles: From animal to human with theranostic AGuIX nanoparticles. Theranostics 2020, 10, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Day, E.; Ehrhardt, G.J. Therapeutic Use of 90Y Microspheres. Int. J. Radiat. Appl. Instrum. Part B Nucl. Med. Biol. 1987, 14, 233–242. [Google Scholar]
- Andrews, J.C.; Walker, S.C.; Ackermann, R.J.; Cotton, L.A.; Ensminger, W.D.; Shapiro, B.; Feinendegen, L.E. Hepatic radioembolization with Yttrium-90 containing glass microspheres: Preliminary results and clinical follow-up. J. Nucl. Med. 1994, 35, 1637–1646. [Google Scholar]
- Carr, B.I. Hepatic arterial 90Yttrium glass microspheres (therasphere) for unresectable hepatocellular carcinoma: Interim safety and survival data on 65 patients. Liver Transplant. 2004, 10, 107–110. [Google Scholar] [CrossRef]
- Lewandowski, R.J.; Kulik, L.M.; Riaz, A.; Senthilnathan, S.; Mulcahy, M.F.; Ryu, R.K.; Ibrahim, S.M.; Sato, K.T.; Baker, T.; Miller, F.H.; et al. A comparative analysis of transarterial downstaging for hepatocellular carcinoma: Chemoembolization versus radioembolization. Am. J. Transplant. 2009, 9, 1920–1928. [Google Scholar] [CrossRef]
- Salem, R.; Lewandowski, R.J.; Kulik, L.; Wang, E.; Riaz, A.; Ryu, R.K.; Sato, K.T.; Gupta, R.; Nikolaidis, P.; Miller, F.H.; et al. Radioembolization results in longer time-to-progression and reduced toxicity compared with chemoembolization in patients with hepatocellular carcinoma. Gastroenterology 2011, 140, 497–507. [Google Scholar] [CrossRef]
- Manas, D.; Bell, J.K.; Mealing, S.; Davies, H.; Baker, H.; Holmes, H.; Hubner, R.A. The cost-effectiveness of therasphere in patients with hepatocellular carcinoma who are eligible for transarterial embolization. Eur. J. Surg. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]

| Fluorescent Materials | Reference | ||
|---|---|---|---|
| Organic | Fluorescent dye | Fluorescein isothiocyanate (FTIC), Rhodamine B, Eosin yellowish, Propidium iodide, Cyanine dyes (Cy3, Cy5, Indocyanine green (ICG), IRDye 800CW, IR820) | [60,61,74,79,80,81,82,83,84,85,86,87] |
| Inorganic | Rare earth | Eu, In | [88,89,90,91] |
| Transition metal complex | tris(1,10-phenanthroline) ruthenium (II) chloride (Ru(phen)32+), tris(2,2′-bipyridyl) dichlororuthenium(II) (Ru(bpy)32+) | [92,93,94,95,96,97,98] | |
| Quantum dots | CdSe/ZnS, CdTe, CdSe/CdS/ZnS | [78,99,100,101,102] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mochizuki, C.; Nakamura, J.; Nakamura, M. Development of Non-Porous Silica Nanoparticles towards Cancer Photo-Theranostics. Biomedicines 2021, 9, 73. https://doi.org/10.3390/biomedicines9010073
Mochizuki C, Nakamura J, Nakamura M. Development of Non-Porous Silica Nanoparticles towards Cancer Photo-Theranostics. Biomedicines. 2021; 9(1):73. https://doi.org/10.3390/biomedicines9010073
Chicago/Turabian StyleMochizuki, Chihiro, Junna Nakamura, and Michihiro Nakamura. 2021. "Development of Non-Porous Silica Nanoparticles towards Cancer Photo-Theranostics" Biomedicines 9, no. 1: 73. https://doi.org/10.3390/biomedicines9010073
APA StyleMochizuki, C., Nakamura, J., & Nakamura, M. (2021). Development of Non-Porous Silica Nanoparticles towards Cancer Photo-Theranostics. Biomedicines, 9(1), 73. https://doi.org/10.3390/biomedicines9010073




