Organoarsenic Compounds with In Vitro Activity against the Malaria Parasite Plasmodium falciparum
Abstract
:1. Introduction
2. Materials and Methods
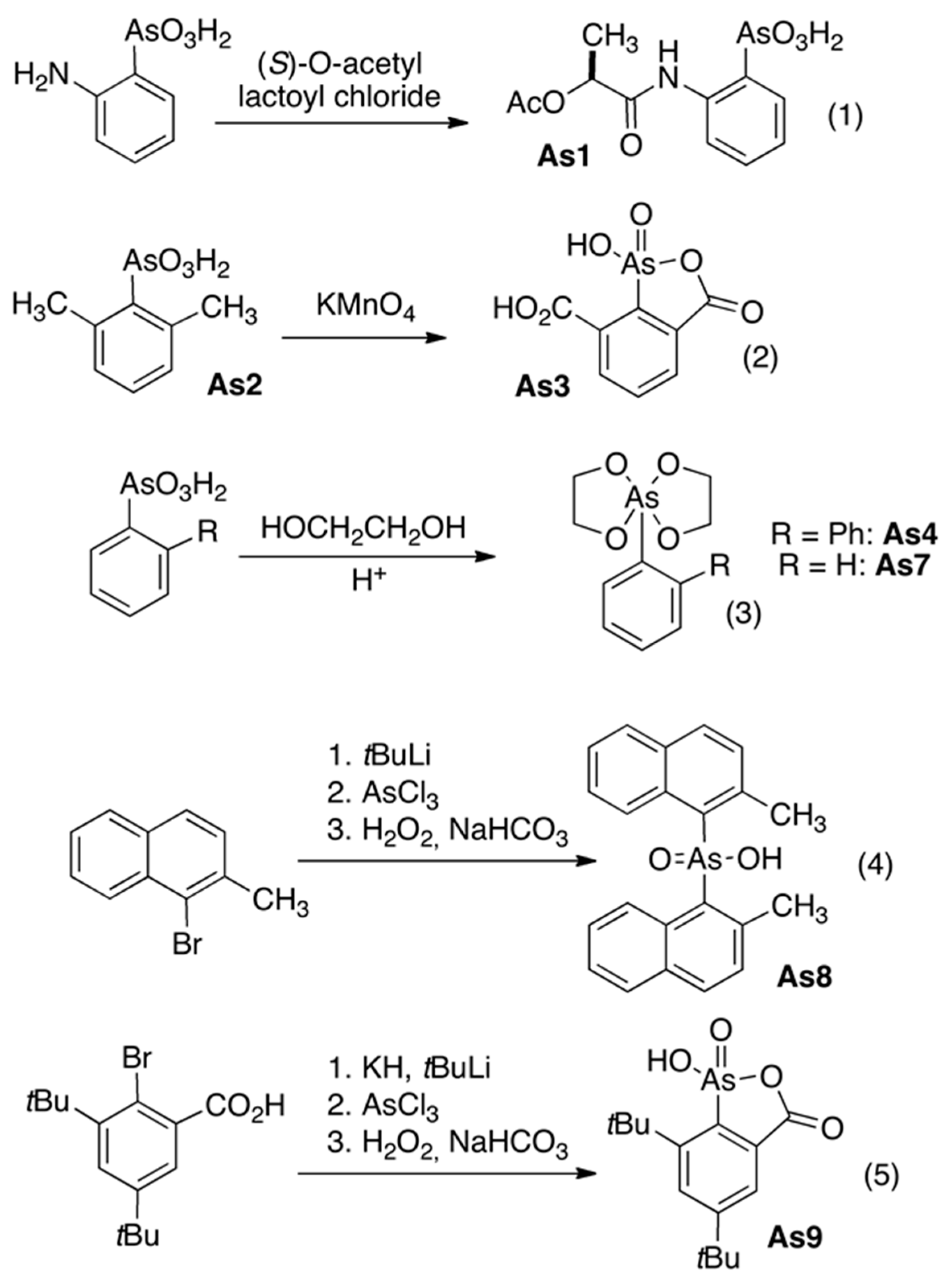
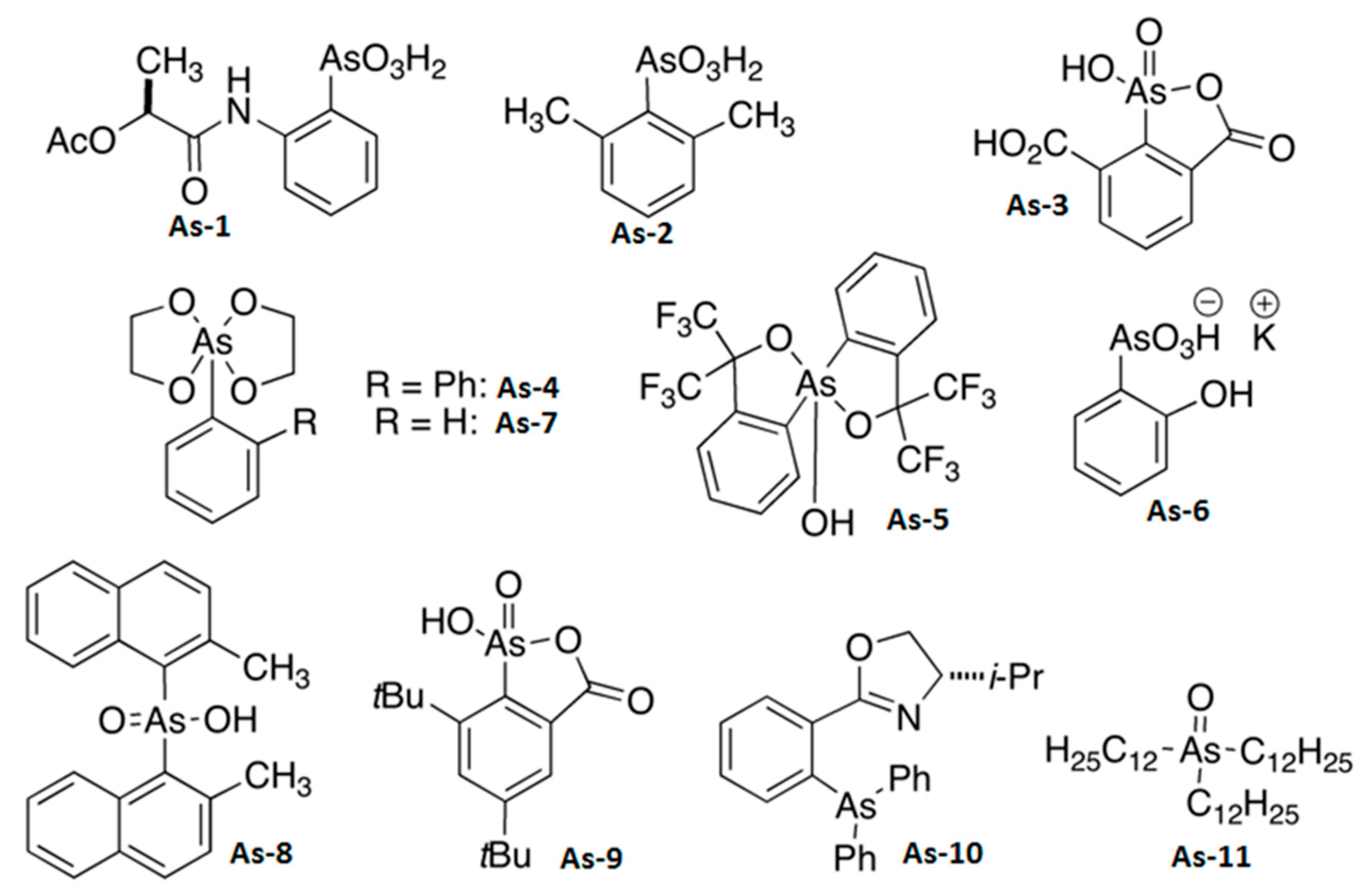
2.1. Organoarsenic Compound As-1–As-11 Synthesis
2.2. Parasite Culture
2.3. Malstat Assay
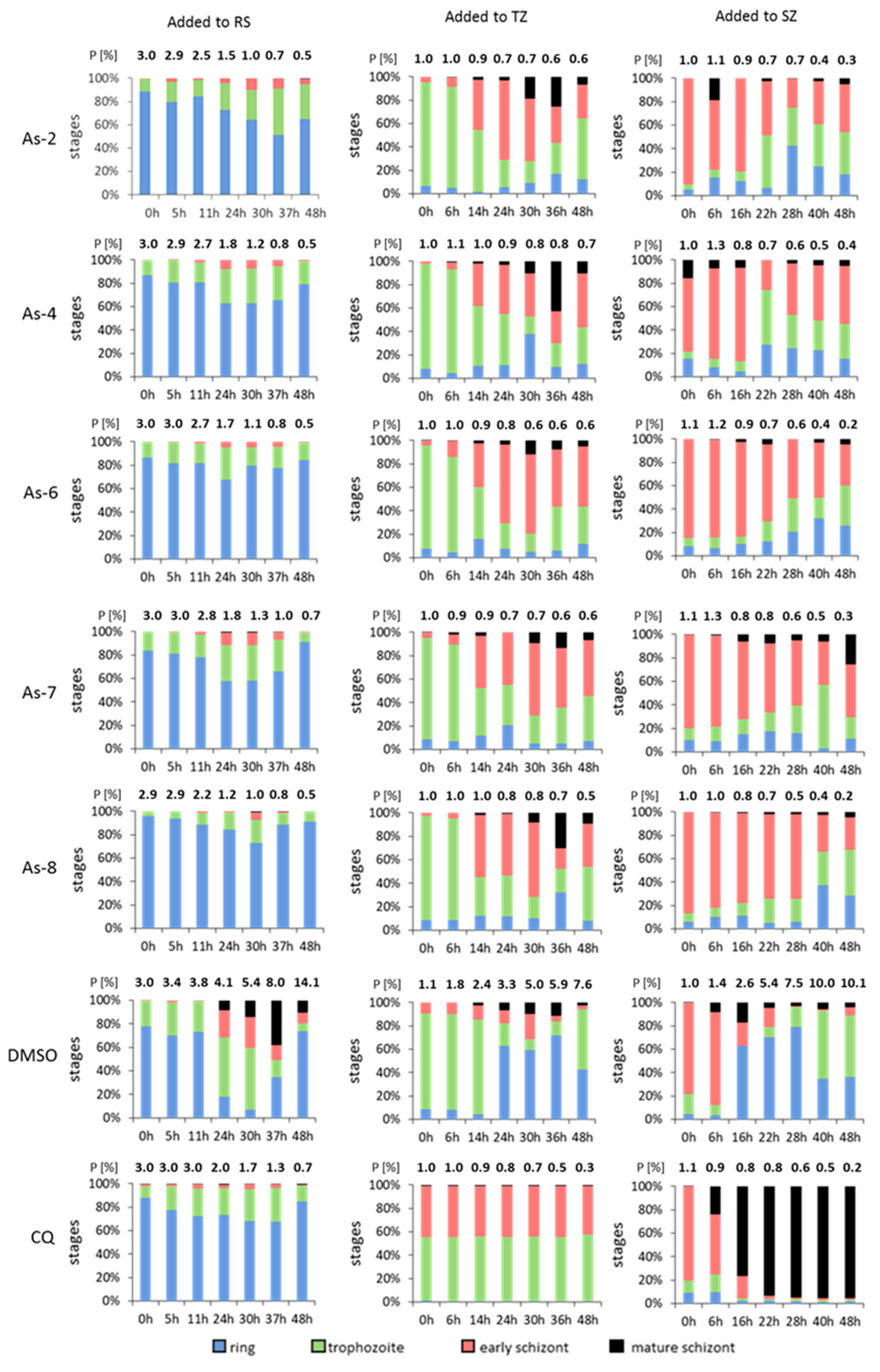
2.4. Stage-Specific Inhibition Assay
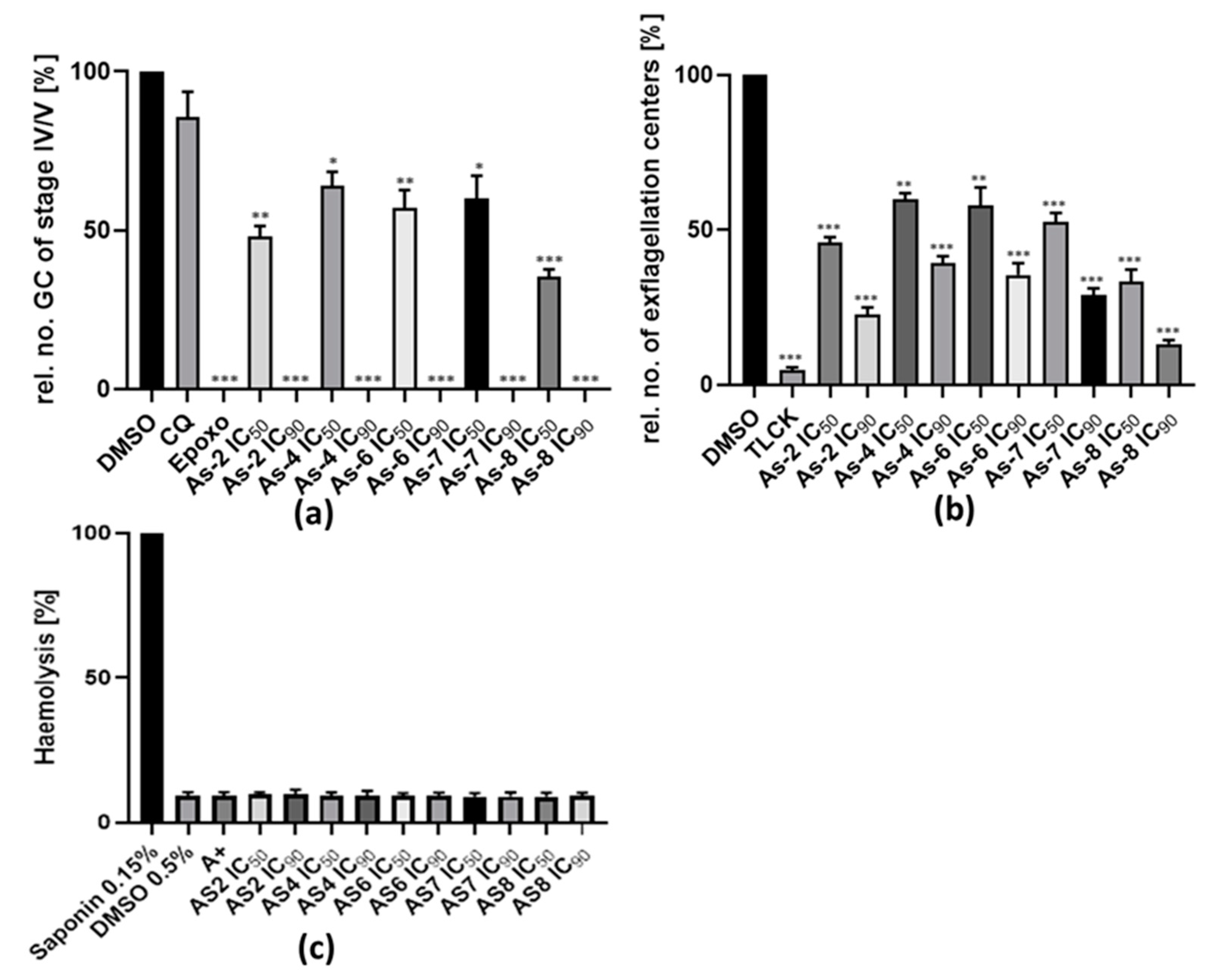
2.5. Gametocyte Toxicity Assay
2.6. Exflagellation Inhibition Assay
2.7. Hemolysis Assay
2.8. Cytotoxicity Assay
3. Results
3.1. Organoarsenic Compounds
3.2. The Organosarsenic Compounds Exhibit Antimalarial Activity
3.3. Organoarsenic Compounds Inhibit Different Asexual Blood Stages of the Malaria Parasite
3.4. Treatment with Organoarsenic Compounds Affects Gametocyte Development
3.5. The Organoarsenic Compounds Inhibit Exflagellation
3.6. The Organoarsenic Compounds Exhibit No Hemolytic or Cytotoxic Effect on Human cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CQ | Chloroquine |
| IC50 | Half maximal inhibitory concentration |
| IC90 | 90% inhibitory concentration |
| RT | Room temperature |
| DMSO | Dimethyl sulfoxide |
| APAD | 3-Acetylpyridine adenine dinucleotide |
| LDH | Lactate dehydrogenase |
| NBT | Nitro Blue Tetrazolium |
| TLCK | Tosyl-l-lysyl-chloromethane hydrochloride |
| INT | 2-[4-idophenyl]-3-[4-nitrophenyl]-5-phenyltetrazolium chloride |
| SI | Selectivity index |
References
- World Health Organization. World Malaria Report 2019; WHO Press: Geneva, Switzerland, 2019. [Google Scholar]
- Wadi, I.; Nath, M.; Anvikar, A.R.; Singh, P.; Sinha, A. Recent advances in transmission-blocking drugs for malaria elimination. Future Med. Chem. 2019, 11, 3047–3088. [Google Scholar] [CrossRef] [PubMed]
- Grund, S.C.; Hanusch, K.S.; Wolf, H.W. Arsenic and arsenic compounds. Ullmann’s Encycl. Ind. Chem. 2008. [Google Scholar] [CrossRef]
- Hughes, M.F.; Beck, B.D.; Chen, Y.; Lewis, A.S.; Thomas, D.J. Arsenic Exposure and Toxicology: A Historical Perspective. Toxicol. Sci. 2011, 123, 305–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibaud, S.; Jaouen, G. Arsenic-based drugs: From fowler’s solution to modern anticancer chemotherapy. Top. Organomet. Chem. 2010, 32, 1–20. [Google Scholar]
- Flora, S.J.S. Arsenic: Chemistry, Occurrence, and Exposure. In Handbook of Arsenic Toxicology; Academic Press: Cambridge, MA, USA, 2015; ISBN 9780124199552. [Google Scholar]
- Rebecca, V.W.; Nicastri, M.C.; Fennelly, C.; Chude, C.I.; Barber-Rotenberg, J.S.; Ronghe, A.; Mcafee, Q.; Mclaughlin, N.P.; Zhang, G.; Goldman, A.R.; et al. PPT1 promotes tumor growth and is the molecular target of chloroquine derivatives in cancer. Cancer Discov. 2019, 9, 220–229. [Google Scholar] [CrossRef] [Green Version]
- Nzila, A.; Okombo, J.; Becker, R.P.; Chilengi, R.; Lang, T.; Niehues, T. Anticancer agents against malaria: Time to revisit? Trends Parasitol. 2010, 26, 125–129. [Google Scholar] [CrossRef] [Green Version]
- Doak, G.O.; Steinman, H.G.; Eagle, H. The preparation of phenylarsenoxides. IV. Disubstituted compounds. J. Am. Chem. Soc. 1941, 63, 99–101. [Google Scholar] [CrossRef]
- Jiang, X.D.; Matsukawa, S.; Kojima, S.; Yamamoto, Y. Synthesis and characterization of antiapicophilic arsoranes and related compounds. Inorg. Chem. 2012, 51, 10996–11006. [Google Scholar] [CrossRef]
- Jacobs, W.A.; Heidelberger, M. The isomeric hydroxyphenylarsonic acids and the direct arsenation of phenol. J. Am. Chem. Soc. 1919, 41, 1440–1450. [Google Scholar] [CrossRef] [Green Version]
- Holmes, R.R.; Day, R.O.; Sau, A.C. Synthesis and molecular structure of spiroarsoranes differing in ring unsaturation. Distortion coordinates for five-coordinated arsenic1. Phosphorus. Sulfur. Silicon Relat. Elem. 1995, 98, 399–415. [Google Scholar] [CrossRef]
- Kwong, F.Y.; Lai, C.W.; Yu, M.; Tan, D.M.; Lam, F.L.; Chan, A.S.; Chan, K.S.C. Convenient Palladium-Catalyzed Arsination: Direct Synthesis of Functionalized Aryl Arsines, Optically Active as, N Ligands, and Their Metal Complexes. Organometallics 2005, 24, 4170–4178. [Google Scholar] [CrossRef]
- Wahl, G.; Kleinhenz, D.; Schorm, A.; Sundermeyer, J.; Stowasser, R.; Rummey, C.; Bringmann, G.; Fickert, C.; Kiefer, W. Peroxomolybdenum Complexes as Epoxidation Catalysts in Biphasic Hydrogen Peroxide Activation: Raman Spectroscopic Studies and Density Functional Calculations. Chem. A Eur. J. 1999, 5, 3237–3251. [Google Scholar] [CrossRef]
- Ifediba, T.; Vanderberg, J.P. Complete in vitro maturation of Plasmodium falciparum gametocytes. Nature 1981, 294, 364–366. [Google Scholar] [CrossRef] [PubMed]
- Lambros, C.; Vanderberg, J.P. Synchronization of Plasmodium falciparum Erythrocytic Stages in Culture. J. Parasitol. 1979, 65, 418–420. [Google Scholar] [CrossRef] [PubMed]
- Aminake, M.N.; Schoof, S.; Sologub, L.; Leubner, M.; Kirschner, M.; Arndt, H.D.; Pradel, G. Thiostrepton and derivatives exhibit antimalarial and gametocytocidal activity by dually targeting parasite proteasome and apicoplast. Antimicrob. Agents Chemother. 2011, 55, 1338–1348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngwa, C.J.; Kiesow, M.J.; Papst, O.; Orchard, L.M.; Filarsky, M.; Rosinski, A.N.; Voss, T.S.; Llinás, M.; Pradel, G. Transcriptional Profiling Defines Histone Acetylation as a Regulator of Gene Expression during Human-to-Mosquito Transmission of the Malaria Parasite Plasmodium falciparum. Front. Cell. Infect. Microbiol. 2017, 7, 320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngwa, C.J.; Kiesow, M.J.; Orchard, L.M.; Farrukh, A.; Llinás, M.; Pradel, G. The g9a histone methyltransferase inhibitor BIX-01294 modulates gene expression during plasmodium falciparum gametocyte development and transmission. Int. J. Mol. Sci. 2019, 20, 5087. [Google Scholar] [CrossRef] [Green Version]
- Garcia, G.E.; Wirtz, R.A.; Barr, J.R.; Woolfitt, A.; Rosenberg, R. Xanthurenic acid induces gametogenesis in Plasmodium, the malaria parasite. J. Biol. Chem. 1998, 273, 12003–12005. [Google Scholar] [CrossRef] [Green Version]
- Billker, O.; Lindo, V.; Panico, M.; Etienne, A.E.; Paxton, T.; Dell, A.; Rogers, M.; Sinden, R.E.; Morris, H.R. Identification of xanthurenic acid as the putative inducer of malaria development in the mosquito. Nature 1998, 392, 289–292. [Google Scholar] [CrossRef]
- Czesny, B.; Goshu, S.; Cook, J.L.; Williamson, K.C. The proteasome inhibitor epoxomicin has potent Plasmodium falciparum gametocytocidal activity. Antimicrob. Agents Chemother. 2009, 53, 4080–4085. [Google Scholar] [CrossRef] [Green Version]
- Rupp, I.; Bosse, R.; Schirmeister, T.; Pradel, G. Effect of protease inhibitors on exflagellation in Plasmodium falciparum. Mol. Biochem. Parasitol. 2008, 158, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Sologub, L.; Kuehn, A.; Kern, S.; Przyborski, J.; Schillig, R.; Pradel, G. Malaria proteases mediate inside-out egress of gametocytes from red blood cells following parasite transmission to the mosquito. Cell. Microbiol. 2011, 13, 897–912. [Google Scholar] [CrossRef] [PubMed]
- Baragaña, B.; Hallyburton, I.; Lee, M.C.S.; Norcross, N.R.; Grimaldi, R.; Otto, T.D.; Proto, W.R.; Blagborough, A.M.; Meister, S.; Wirjanata, G.; et al. A novel multiple-stage antimalarial agent that inhibits protein synthesis. Nature 2015, 522, 315–320. [Google Scholar] [CrossRef]
- Kato, N.; Comer, E.; Sakata-Kato, T.; Sharma, A.; Sharma, M.; Maetani, M.; Bastien, J.; Brancucci, N.M.; Bittker, J.A.; Corey, V.; et al. Diversity-oriented synthesis yields novel multistage antimalarial inhibitors. Nature 2016, 538, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Plouffe, D.M.; Wree, M.; Du, A.Y.; Meister, S.; Li, F.; Patra, K.; Lubar, A.; Okitsu, S.L.; Flannery, E.L.; Kato, N.; et al. High-Throughput Assay and Discovery of Small Molecules that Interrupt Malaria Transmission. Cell Host Microbe 2016, 19, 114–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fairlamb, A.H. Chemotherapy of human African trypanosomiasis: Current and future prospects. Trends Parasitol. 2003, 19, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Chou, W.C.; Dang, C.V. Acute promyelocytic leukemia: Recent advances in therapy and molecular basis of response to arsenic therapies. Curr. Opin. Hematol. 2005, 12, 1–6. [Google Scholar] [CrossRef]
- Swindell, E.P.; Hankins, P.L.; Chen, H.; Miodragović, C.D.S.U.; O’Halloran, T.V. Anticancer activity of small-molecule and nanoparticulate arsenic(III) complexes. Inorg. Chem. 2013, 52, 12292–12304. [Google Scholar] [CrossRef] [Green Version]
- Cioloboc, D.; Kurtz, D.M. Targeted cancer cell delivery of arsenate as a reductively activated prodrug. J. Biol. Inorg. Chem. 2020, 25, 441–449. [Google Scholar] [CrossRef]
- Aminake, M.N.; Arndt, H.-D.D.; Pradel, G. The proteasome of malaria parasites: A multi-stage drug target for chemotherapeutic intervention? Int. J. Parasitol. Drugs Drug Resist. 2012, 2, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Trenholme, K.; Marek, L.; Duffy, S.; Pradel, G.; Fisher, G.; Hansen, F.K.; Skinner-Adams, T.S.; Butterworth, A.; Ngwa, C.J.; Moecking, J.; et al. Lysine acetylation in sexual stage malaria parasites is a target for antimalarial small molecules. Antimicrob. Agents Chemother. 2014, 58, 3666–3678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duffin, W.M.; Rollo, I.M. Antimalarial activity of hydroxy-substituted naphthalene compounds. Br. J. Pharmacol. Chemother. 1957, 12, 171–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Compounds | IC50 [µM] | IC90 [µM] | CC50 [µM] | SI |
|---|---|---|---|---|
| CQ-sensitive 3D7 | ||||
| CQ | 0.002 ± 0.0003 | 0.0189 | ||
| As-1 | - | - | ||
| As-2 | 8.63 ± 0.606 | 77.67 | >100 | >12 |
| As-3 | - | - | ||
| As-4 | 8.64 ± 0.963 | 77.76 | >100 | >12 |
| As-5 | 16.57 ± 3.092 | 149.13 | ||
| As-6 | 7.55 ± 0.580 | 67.95 | >100 | >13 |
| As-7 | 7.64 ± 0.387 | 68.76 | >100 | >13 |
| As-8 | 1.52 ± 0.089 | 13.68 | >100 | >66 |
| As-9 | 45.65 ± 0.435 | 410.85 | ||
| As-10 | 18.13 ± 2.996 | 163.17 | ||
| As-11 | 43.04 ± 5.700 | 387.36 | ||
| CQ-resistant Dd2 | ||||
| CQ | 0.28 ± 0.022 | 2.52 | ||
| As-2 | 7.88 ± 0.261 | 70.92 | >100 | >13 |
| As-4 | 30.05 ± 2.071 | 270.45 | >100 | >3 |
| As-6 | 8.27 ± 1.330 | 74.43 | >100 | >12 |
| As-7 | 34.92 ± 2.120 | 314.28 | >100 | >3 |
| As-8 | 0.35 ± 0.099 | 3.15 | >100 | >286 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basova, S.; Wilke, N.; Koch, J.C.; Prokop, A.; Berkessel, A.; Pradel, G.; Ngwa, C.J. Organoarsenic Compounds with In Vitro Activity against the Malaria Parasite Plasmodium falciparum. Biomedicines 2020, 8, 260. https://doi.org/10.3390/biomedicines8080260
Basova S, Wilke N, Koch JC, Prokop A, Berkessel A, Pradel G, Ngwa CJ. Organoarsenic Compounds with In Vitro Activity against the Malaria Parasite Plasmodium falciparum. Biomedicines. 2020; 8(8):260. https://doi.org/10.3390/biomedicines8080260
Chicago/Turabian StyleBasova, Sofia, Nathalie Wilke, Jan Christoph Koch, Aram Prokop, Albrecht Berkessel, Gabriele Pradel, and Che Julius Ngwa. 2020. "Organoarsenic Compounds with In Vitro Activity against the Malaria Parasite Plasmodium falciparum" Biomedicines 8, no. 8: 260. https://doi.org/10.3390/biomedicines8080260





