N-acetylcysteine Provides Cytoprotection in Murine Oligodendrocytes through Heme Oxygenase-1 Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture and Experimental Conditions
2.3. Cell Survival Assays
2.4. Evaluation of ROS Production
2.5. Determination of Intracellular GSH
2.6. Total Antioxidant Capacity Assay
2.7. Statistical Data Analysis
3. Results
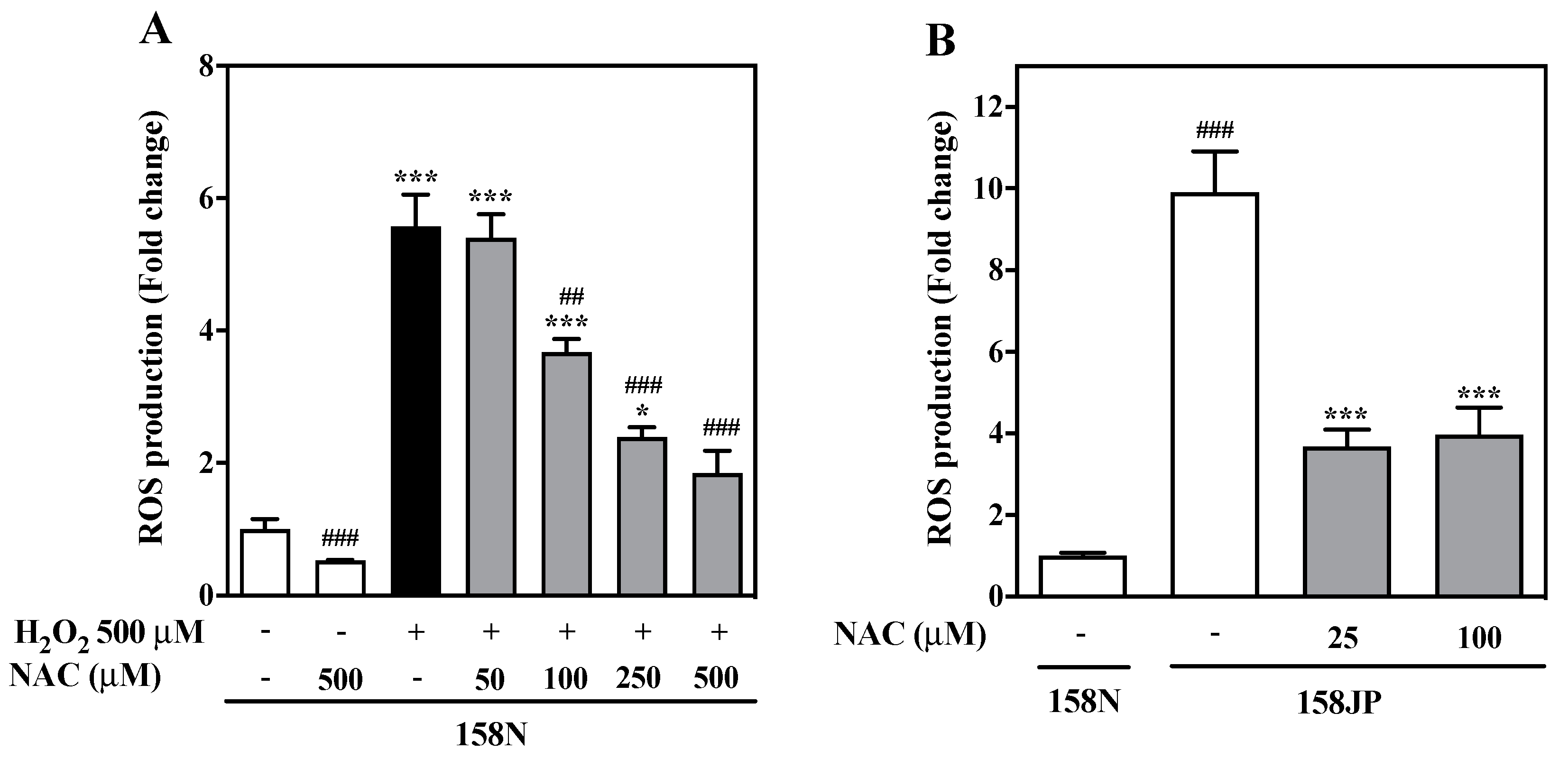
3.1. NAC Decreases ROS Content in Oligodendrocytes
3.2. NAC Improves 158N Cell Survival in Oxidative Stress
3.3. GSH Depletion Partially Reduces the Cytoprotective Effect of NAC
3.4. HO-1 Activity Mediate the Protective Effect of NAC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hsieh, H.L.; Yang, C.M. Role of redox signaling in neuroinflammation and neurodegenerative diseases. Biomed. Res. Int. 2013, 2013, 484613. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.L.; Bayraktutan, U. Oxidative stress and its role in the pathogenesis of ischaemic stroke. Int. J. Stroke 2009, 4, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Shukla, V.; Mishra, S.K.; Pant, H.C. Oxidative stress in neurodegeneration. Adv. Pharmacol. Sci. 2011, 2011, 572634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, F.; Yao, S.Y.; Whetsell, W.O., Jr.; Sriram, S. Astrogliopathy and oligodendrogliopathy are early events in CNS demyelination. Glia 2013, 61, 1261–1273. [Google Scholar] [CrossRef]
- Ahmad, S.; Khan, M.B.; Hoda, M.N.; Bhatia, K.; Haque, R.; Fazili, I.S.; Jamal, A.; Khan, J.S.; Katare, D.P. Neuroprotective effect of sesame seed oil in 6-hydroxydopamine induced neurotoxicity in mice model: Cellular, biochemical and neurochemical evidence. Neurochem. Res. 2012, 37, 516–526. [Google Scholar] [CrossRef]
- Jin, H.; Kanthasamy, A.; Ghosh, A.; Anantharam, V.; Kalyanaraman, B.; Kanthasamy, A.G. Mitochondria-targeted antioxidants for treatment of Parkinson’s disease: Preclinical and clinical outcomes. Biochim. Biophys. Acta 2014, 1842, 1282–1294. [Google Scholar] [CrossRef] [Green Version]
- Heard, K.; Green, J. Acetylcysteine therapy for acetaminophen poisoning. Curr. Pharm. Biotechnol. 2012, 13, 1917–1923. [Google Scholar] [CrossRef]
- Rushworth, G.F.; Megson, I.L. Existing and potential therapeutic uses for N-acetylcysteine: The need for conversion to intracellular glutathione for antioxidant benefits. Pharm. Ther. 2014, 141, 150–159. [Google Scholar] [CrossRef]
- Dodd, S.; Dean, O.; Copolov, D.L.; Malhi, G.S.; Berk, M. N-acetylcysteine for antioxidant therapy: Pharmacology and clinical utility. Expert Opin. Biol. Ther. 2008, 8, 1955–1962. [Google Scholar] [CrossRef]
- Schroder, H.; Warren, S.; Bargetzi, M.J.; Torti, S.V.; Torti, F.M. N-acetyl-L-cysteine protects endothelial cells but not L929 tumor cells from tumor necrosis factor-alpha-mediated cytotoxicity. Naunyn Schmiedebergs Arch. Pharm. 1993, 347, 664–666. [Google Scholar] [CrossRef]
- Aoyama, K.; Nakaki, T. Impaired glutathione synthesis in neurodegeneration. Int. J. Mol. Sci. 2013, 14, 21021–21044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmay, M.J.; Terpstra, M.; Coles, L.D.; Mishra, U.; Ahlskog, M.; Oz, G.; Cloyd, J.C.; Tuite, P.J. N-acetylcysteine Boosts Brain and Blood Glutathione in Gaucher and Parkinson Diseases. Clin. Neuropharmacol. 2013, 36, 103–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zafarullah, M.; Li, W.Q.; Sylvester, J.; Ahmad, M. Molecular mechanisms of N-acetylcysteine actions. Cell Mol. Life Sci. 2003, 60, 6–20. [Google Scholar] [CrossRef]
- Ogborne, R.M.; Rushworth, S.A.; O’Connell, M.A. Alpha-lipoic acid-induced heme oxygenase-1 expression is mediated by nuclear factor erythroid 2-related factor 2 and p38 mitogen-activated protein kinase in human monocytic cells. Arter. Thromb. Vasc. Biol. 2005, 25, 2100–2105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.; Lei, S.; Liu, Y.; Gao, X.; Irwin, M.G.; Xia, Z.Y.; Hei, Z.; Gan, X.; Wang, T.; Xia, Z. Antioxidant N-acetylcysteine attenuates the reduction of Brg1 protein expression in the myocardium of type 1 diabetic rats. J. Diabetes. Res. 2013, 2013, 716219. [Google Scholar] [CrossRef]
- Abraham, N.G.; Kappas, A. Pharmacological and clinical aspects of heme oxygenase. Pharm. Rev. 2008, 60, 79–127. [Google Scholar] [CrossRef] [Green Version]
- Abraham, N.G.; Asija, A.; Drummond, G.; Peterson, S. Heme oxygenase-1 gene therapy: Recent advances and therapeutic applications. Curr. Gene Ther. 2007, 7, 89–108. [Google Scholar] [CrossRef]
- Nitti, M.; Piras, S.; Brondolo, L.; Marinari, U.M.; Pronzato, M.A.; Furfaro, A.L. Heme Oxygenase 1 in the Nervous System: Does It Favor Neuronal Cell Survival or Induce Neurodegeneration? Int. J. Mol. Sci. 2018, 19, 2260. [Google Scholar] [CrossRef] [Green Version]
- Schipper, H.M. Heme oxygenase expression in human central nervous system disorders. Free Radic. Biol. Med. 2004, 37, 1995–2011. [Google Scholar] [CrossRef]
- Fukuda, K.; Panter, S.S.; Sharp, F.R.; Noble, L.J. Induction of heme oxygenase-1 (HO-1) after traumatic brain injury in the rat. Neurosci. Lett. 1995, 199, 127–130. [Google Scholar] [CrossRef]
- Baarine, M.; Ragot, K.; Genin, E.C.; El Hajj, H.; Trompier, D.; Andreoletti, P.; Ghandour, M.S.; Menetrier, F.; Cherkaoui-Malki, M.; Savary, S.; et al. Peroxisomal and mitochondrial status of two murine oligodendrocytic cell lines (158N, 158JP): Potential models for the study of peroxisomal disorders associated with dysmyelination processes. J. Neurochem. 2009, 111, 119–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knapp, P.E.; Adjan, V.V.; Hauser, K.F. Cell-specific loss of kappa-opioid receptors in oligodendrocytes of the dysmyelinating jimpy mouse. Neurosci. Lett. 2009, 451, 114–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.; Coles, L.D.; Kartha, R.V.; Nash, N.; Mishra, U.; Lund, T.C.; Cloyd, J.C. Intravenous Administration of Stable-Labeled N-Acetylcysteine Demonstrates an Indirect Mechanism for Boosting Glutathione and Improving Redox Status. J. Pharm. Sci. 2015, 104, 2619–2626. [Google Scholar] [CrossRef] [PubMed]
- Drew, R.; Miners, J.O. The effects of buthionine sulphoximine (BSO) on glutathione depletion and xenobiotic biotransformation. Biochem. Pharm. 1984, 33, 2989–2994. [Google Scholar] [CrossRef]
- Appleton, S.D.; Chretien, M.L.; McLaughlin, B.E.; Vreman, H.J.; Stevenson, D.K.; Brien, J.F.; Nakatsu, K.; Maurice, D.H.; Marks, G.S. Selective inhibition of heme oxygenase, without inhibition of nitric oxide synthase or soluble guanylyl cyclase, by metalloporphyrins at low concentrations. Drug Metab. Dispos. Biol. Fate Chem. 1999, 27, 1214–1219. [Google Scholar] [PubMed]
- Ndisang, J.F.; Jadhav, A. Up-regulating the hemeoxygenase system enhances insulin sensitivity and improves glucose metabolism in insulin-resistant diabetes in Goto-Kakizaki rats. Endocrinology 2009, 150, 2627–2636. [Google Scholar] [CrossRef]
- Ndisang, J.F.; Tiwari, S. Featured Article: Induction of heme oxygenase with hemin improves pericardial adipocyte morphology and function in obese Zucker rats by enhancing proteins of regeneration. Exp. Biol. Med. 2015, 240, 45–57. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Fu, X.; Liu, Y.; Yu, D.; Cai, S.J.; Yang, C. Blockade of Glutathione Metabolism in. Mol. Cancer Ther. 2020, 19, 221–230. [Google Scholar] [CrossRef] [Green Version]
- Shieh, P.; Jan, C.R.; Liang, W.Z. The protective effects of the antioxidant N-acetylcysteine (NAC) against oxidative stress-associated apoptosis evoked by the organophosphorus insecticide malathion in normal human astrocytes. Toxicology 2019, 417, 1–14. [Google Scholar] [CrossRef]
- Yang, X.; Yang, H.; Wu, F.; Qi, Z.; Li, J.; Xu, B.; Liu, W.; Xu, Z.; Deng, Y. Mn Inhibits GSH Synthesis via Downregulation of Neuronal EAAC1 and Astrocytic xCT to Cause Oxidative Damage in the Striatum of Mice. Oxid. Med. Cell. Longev. 2018, 2018, 4235695. [Google Scholar] [CrossRef]
- Le, W.D.; Xie, W.J.; Appel, S.H. Protective role of heme oxygenase-1 in oxidative stress-induced neuronal injury. J. Neurosci. Res. 1999, 56, 652–658. [Google Scholar] [CrossRef]
- Panahian, N.; Yoshiura, M.; Maines, M.D. Overexpression of heme oxygenase-1 is neuroprotective in a model of permanent middle cerebral artery occlusion in transgenic mice. J. Neurochem. 1999, 72, 1187–1203. [Google Scholar] [CrossRef]
- Colin-Gonzalez, A.L.; Orozco-Ibarra, M.; Chanez-Cardenas, M.E.; Rangel-Lopez, E.; Santamaria, A.; Pedraza-Chaverri, J.; Barrera-Oviedo, D.; Maldonado, P.D. Heme oxygenase-1 (HO-1) upregulation delays morphological and oxidative damage induced in an excitotoxic/pro-oxidant model in the rat striatum. Neuroscience 2013, 231, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, G.; Colín-González, A.L.; Rangel-López, E.; Chavarría, A.; Santamaría, A. Redox Signaling, Neuroinflammation, and Neurodegeneration. Antioxid. Redox. Signal. 2018, 28, 1626–1651. [Google Scholar] [CrossRef]
- Kartha, R.V.; Zhou, J.; Basso, L.; Schröder, H.; Orchard, P.J.; Cloyd, J. Mechanisms of Antioxidant Induction with High-Dose N-Acetylcysteine in Childhood Cerebral Adrenoleukodystrophy. CNS Drugs 2015, 29, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Escartin, C.; Won, S.J.; Malgorn, C.; Auregan, G.; Berman, A.E.; Chen, P.C.; Déglon, N.; Johnson, J.A.; Suh, S.W.; Swanson, R.A. Nuclear factor erythroid 2-related factor 2 facilitates neuronal glutathione synthesis by upregulating neuronal excitatory amino acid transporter 3 expression. J. Neurosci. 2011, 31, 7392–7401. [Google Scholar] [CrossRef] [Green Version]
- Santos, C.L.; Bobermin, L.D.; Souza, D.G.; Bellaver, B.; Bellaver, G.; Arús, B.A.; Souza, D.O.; Gonçalves, C.A.; Quincozes-Santos, A. Lipoic acid and N-acetylcysteine prevent ammonia-induced inflammatory response in C6 astroglial cells: The putative role of ERK and HO1 signaling pathways. Toxicol. In Vitro 2015, 29, 1350–1357. [Google Scholar] [CrossRef] [Green Version]
- Deon, M.; Marchetti, D.P.; Donida, B.; Wajner, M.; Vargas, C. Oxidative Stress in Patients with X-Linked Adrenoleukodystrophy. Cell Mol. Neurobiol. 2016, 36, 497–512. [Google Scholar] [CrossRef]
- Harauz, G.; Ishiyama, N.; Hill, C.M.; Bates, I.R.; Libich, D.S.; Fares, C. Myelin basic protein-diverse conformational states of an intrinsically unstructured protein and its roles in myelin assembly and multiple sclerosis. Micron 2004, 35, 503–542. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, T.; Ziegler, A.C.; Dimitrion, P.; Zuo, L. Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxid. Med. Cell Longev. 2017, 2017, 2525967. [Google Scholar] [CrossRef]
- van Zandwijk, N. N-acetylcysteine (NAC) and glutathione (GSH): Antioxidant and chemopreventive properties, with special reference to lung cancer. J. Cell. Biochem. Suppl. 1995, 22, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Fang, Y.Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione metabolism and its implications for health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, I.Y.; Lee, S.P.; Denney, D.R.; Lynch, S.G. Lower levels of glutathione in the brains of secondary progressive multiple sclerosis patients measured by 1H magnetic resonance chemical shift imaging at 3 T. Mult. Scler. 2011, 17, 289–296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oz, G.; Tkac, I.; Charnas, L.R.; Choi, I.Y.; Bjoraker, K.J.; Shapiro, E.G.; Gruetter, R. Assessment of adrenoleukodystrophy lesions by high field MRS in non-sedated pediatric patients. Neurology 2005, 64, 434–441. [Google Scholar] [CrossRef] [Green Version]
- Petrillo, S.; Piemonte, F.; Pastore, A.; Tozzi, G.; Aiello, C.; Pujol, A.; Cappa, M.; Bertini, E. Glutathione imbalance in patients with X-linked adrenoleukodystrophy. Mol. Genet. Metab. 2013, 109, 366–370. [Google Scholar] [CrossRef]
- Casasnovas, C.; Ruiz, M.; Schlüter, A.; Naudí, A.; Fourcade, S.; Veciana, M.; Castañer, S.; Albertí, A.; Bargalló, N.; Johnson, M.; et al. Biomarker Identification, Safety, and Efficacy of High-Dose Antioxidants for Adrenomyeloneuropathy: A Phase II Pilot Study. Neurotherapeutics 2019, 16, 1167–1182. [Google Scholar] [CrossRef] [Green Version]
- Barone, E.; Di Domenico, F.; Mancuso, C.; Butterfield, D.A. The Janus face of the heme oxygenase/biliverdin reductase system in Alzheimer disease: It’s time for reconciliation. Neurobiol. Dis. 2013, 62C, 144–159. [Google Scholar] [CrossRef] [Green Version]
- Chien, W.L.; Lee, T.R.; Hung, S.Y.; Kang, K.H.; Lee, M.J.; Fu, W.M. Impairment of oxidative stress-induced heme oxygenase-1 expression by the defect of Parkinson-related gene of PINK1. J. Neurochem. 2011, 117, 643–653. [Google Scholar] [CrossRef]
- Choi, A.M.; Alam, J. Heme oxygenase-1: Function, regulation, and implication of a novel stress-inducible protein in oxidant-induced lung injury. Am. J. Respir. Cell Mol. Biol. 1996, 15, 9–19. [Google Scholar] [CrossRef]
- Ryter, S.W.; Choi, A.M. Heme oxygenase-1: Redox regulation of a stress protein in lung and cell culture models. Antioxid. Redox Signal. 2005, 7, 80–91. [Google Scholar] [CrossRef]
- Grosser, N.; Abate, A.; Oberle, S.; Vreman, H.J.; Dennery, P.A.; Becker, J.C.; Pohle, T.; Seidman, D.S.; Schroder, H. Heme oxygenase-1 induction may explain the antioxidant profile of aspirin. Biochem. Biophys. Res. Commun. 2003, 308, 956–960. [Google Scholar] [CrossRef]
- Li, M.; Liu, Y.; Shi, H.; Zhang, Y.; Wang, G.; Xu, J.; Lu, J.; Zhang, D.; Xie, X.; Han, D.; et al. Statins inhibit pulmonary artery smooth muscle cell proliferation by upregulation of HO-1 and p21WAF1. Naunyn Schmiedebergs Arch. Pharm. 2012, 385, 961–968. [Google Scholar] [CrossRef] [PubMed]
- D’Araio, E.; Shaw, N.; Millward, A.; Demaine, A.; Whiteman, M.; Hodgkinson, A. Hydrogen sulfide induces heme oxygenase-1 in human kidney cells. Acta Diabetol. 2014, 51, 155–157. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Lai, Y.S.; Chou, T.C. The protective effect of alpha-lipoic Acid in lipopolysaccharide-induced acute lung injury is mediated by heme oxygenase-1. Evid. Based Complement. Altern. Med. 2013, 2013, 590363. [Google Scholar] [CrossRef] [Green Version]
- Erdmann, K.; Cheung, B.W.; Immenschuh, S.; Schroder, H. Heme oxygenase-1 is a novel target and antioxidant mediator of S-adenosylmethionine. Biochem. Biophys. Res. Commun. 2008, 368, 937–941. [Google Scholar] [CrossRef]
- Son, Y.; Lee, J.H.; Chung, H.T.; Pae, H.O. Therapeutic Roles of Heme Oxygenase-1 in Metabolic Diseases: Curcumin and Resveratrol Analogues as Possible Inducers of Heme Oxygenase-1. Oxid. Med. Cell Longev. 2013, 2013, 639541. [Google Scholar] [CrossRef] [Green Version]
- Erdmann, K.; Grosser, N.; Schroder, H. L-methionine reduces oxidant stress in endothelial cells: Role of heme oxygenase-1, ferritin, and nitric oxide. AAPS J. 2005, 7, E195–E200. [Google Scholar] [CrossRef] [Green Version]
- Kweon, M.H.; Jung, M.J.; Sung, H.C. Cytoprotective effects of heme oxygenase-1 induction by 3-O-caffeoyl-1-methylquinic acid. Free Radic. Biol. Med. 2004, 36, 40–52. [Google Scholar] [CrossRef]
- Majid, A.S.; Yin, Z.Q.; Ji, D. Sulphur antioxidants inhibit oxidative stress induced retinal ganglion cell death by scavenging reactive oxygen species but influence nuclear factor (erythroid-derived 2)-like 2 signalling pathway differently. Biol. Pharm. Bull. 2013, 36, 1095–1110. [Google Scholar] [CrossRef] [Green Version]
- Kim, N.; Hwangbo, C.; Lee, S.; Lee, J.H. Eupatolide Inhibits PDGF-induced Proliferation and Migration of Aortic Smooth Muscle Cells Through ROS-dependent Heme Oxygenase-1 Induction. Phytother. Res. 2013, 27, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Rensing, H.; Bauer, I.; Peters, I.; Wein, T.; Silomon, M.; Jaeschke, H.; Bauer, M. Role of reactive oxygen species for hepatocellular injury and heme oxygenase-1 gene expression after hemorrhage and resuscitation. Shock 1999, 12, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Aruoma, O.I.; Halliwell, B.; Hoey, B.M.; Butler, J. The antioxidant action of N-acetylcysteine: Its reaction with hydrogen peroxide, hydroxyl radical, superoxide, and hypochlorous acid. Free Radic. Biol. Med. 1989, 6, 593–597. [Google Scholar] [CrossRef]
- Cotgreave, I.A. N-acetylcysteine: Pharmacological considerations and experimental and clinical applications. Adv. Pharm. 1997, 38, 205–227. [Google Scholar]
- Panchenko, M.V.; Farber, H.W.; Korn, J.H. Induction of heme oxygenase-1 by hypoxia and free radicals in human dermal fibroblasts. Am. J. Physiol. Cell Physiol. 2000, 278, C92–C101. [Google Scholar] [CrossRef]
- Applegate, L.A.; Luscher, P.; Tyrrell, R.M. Induction of heme oxygenase: A general response to oxidant stress in cultured mammalian cells. Cancer Res. 1991, 51, 974–978. [Google Scholar]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, J.; Terluk, M.R.; Basso, L.; Mishra, U.R.; Orchard, P.J.; Cloyd, J.C.; Schröder, H.; Kartha, R.V. N-acetylcysteine Provides Cytoprotection in Murine Oligodendrocytes through Heme Oxygenase-1 Activity. Biomedicines 2020, 8, 240. https://doi.org/10.3390/biomedicines8080240
Zhou J, Terluk MR, Basso L, Mishra UR, Orchard PJ, Cloyd JC, Schröder H, Kartha RV. N-acetylcysteine Provides Cytoprotection in Murine Oligodendrocytes through Heme Oxygenase-1 Activity. Biomedicines. 2020; 8(8):240. https://doi.org/10.3390/biomedicines8080240
Chicago/Turabian StyleZhou, Jie, Marcia R. Terluk, Lisa Basso, Usha R. Mishra, Paul J. Orchard, James C. Cloyd, Henning Schröder, and Reena V. Kartha. 2020. "N-acetylcysteine Provides Cytoprotection in Murine Oligodendrocytes through Heme Oxygenase-1 Activity" Biomedicines 8, no. 8: 240. https://doi.org/10.3390/biomedicines8080240
APA StyleZhou, J., Terluk, M. R., Basso, L., Mishra, U. R., Orchard, P. J., Cloyd, J. C., Schröder, H., & Kartha, R. V. (2020). N-acetylcysteine Provides Cytoprotection in Murine Oligodendrocytes through Heme Oxygenase-1 Activity. Biomedicines, 8(8), 240. https://doi.org/10.3390/biomedicines8080240




