Blood Neurofilament Light Chain: The Neurologist’s Troponin?
Abstract
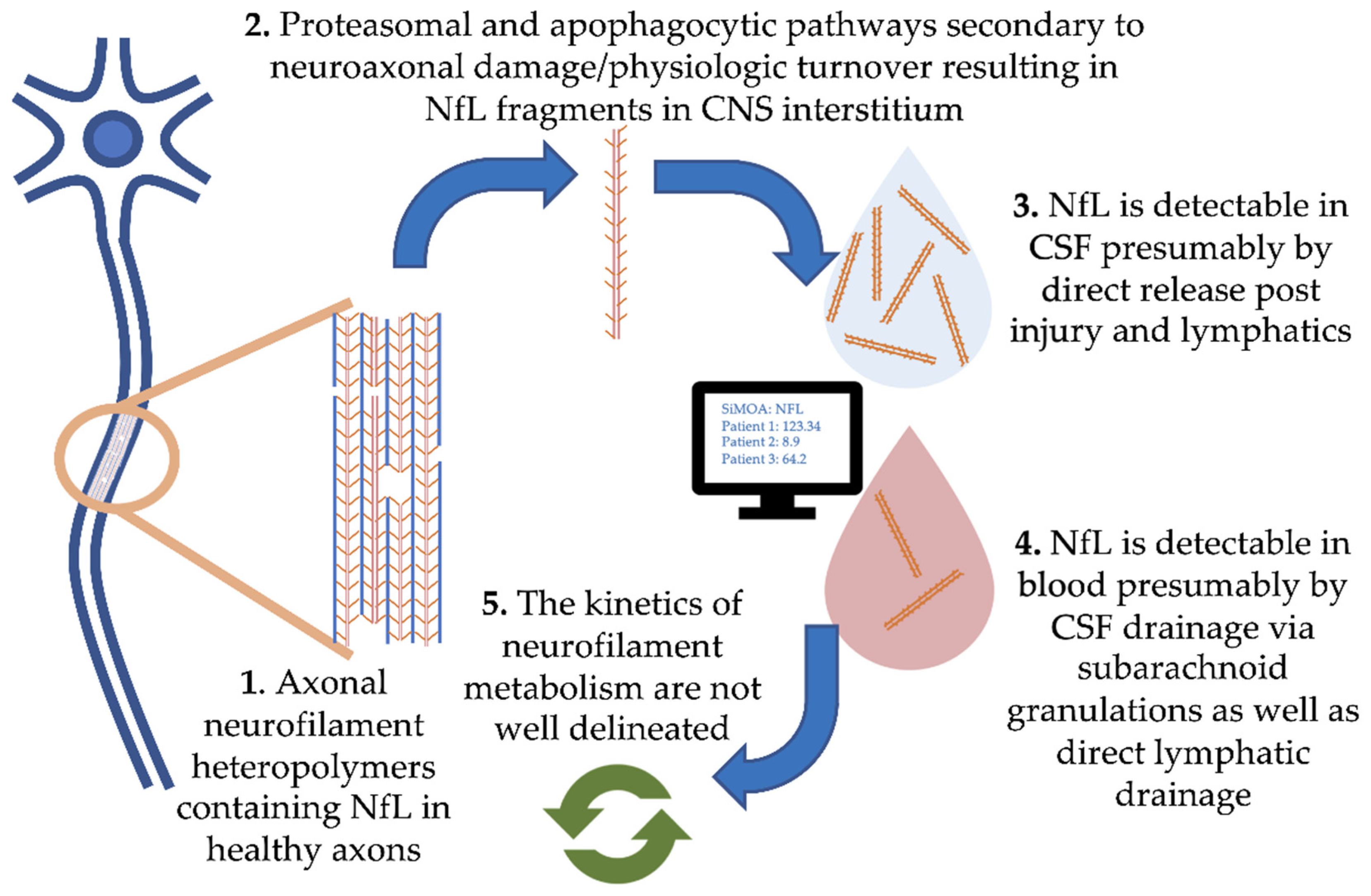
1. Neurofilament Structure and Function
2. Neurofilament Pathophysiology
3. Measurement of Blood Levels of Neurofilament Light Chain
4. Blood NfL in Neurological Diseases
5. Blood NfL in MS
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fuchs, E.; Cleveland, D.W. A structural scaffolding of intermediate filaments in health and disease. Science 1998, 279, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Rao, M.V.; Sasaki, T.; Chen, Y.; Kumar, A.; Veeranna; Liem, R.K.H.; Eyer, J.; Peterson, A.C.; Julien, J.P.; et al. α-internexin is structurally and functionally associated with the neurofilament triplet proteins in the mature CNS. J. Neurosci. 2006, 26, 10006–10019. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Sasaki, T.; Kumar, A.; Peterhoff, C.M.; Rao, M.V.; Liem, R.K.; Julien, J.P.; Nixon, R.A. Peripherin is a subunit of peripheral nerve neurofilaments: Implications for differential vulnerability of cns and peripheral nervous system axons. J. Neurosci. 2012, 32, 8501–8508. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Rao, M.V.; Veeranna; Nixon, R.A. Neurofilaments and neurofilament proteins in health and disease. Cold Spring Harb. Perspect. Biol. 2017, 9, a018309. [Google Scholar] [CrossRef] [PubMed]
- Ching, G.Y.; Liem, R.K.H. Roles of head and tail domains in α-internexin’s self-assembly and coassembly with the neurofilament triplet proteins. J. Cell Sci. 1998, 111, 321–333. [Google Scholar]
- Millecamps, S.; Gowing, G.; Corti, O.; Mallet, J.; Julien, J.P. Conditional NF-L transgene expression in mice for in vivo analysis of turnover and transport rate of neurofilaments. J. Neurosci. 2007, 27, 4947–4956. [Google Scholar] [CrossRef]
- Fliegner, K.H.; Liem, R.K.H. Cellular and Molecular Biology of Neuronal Intermediate Filaments. Int. Rev. Cytol. 1991, 131, 109–167. [Google Scholar] [CrossRef]
- Bragina, L.; Conti, F. Expression of Neurofilament Subunits at Neocortical Glutamatergic and GABAergic Synapses. Front. Neuroanat. 2018, 12, 74. [Google Scholar] [CrossRef]
- Hashimoto, R.; Nakamura, Y.; Komai, S.; Kashiwagi, Y.; Tamura, K.; Goto, T.; Aimoto, S.; Kaibuchi, K.; Shiosaka, S.; Takeda, M. Site-specific phosphorylation of neurofilament-L is mediated by calcium/calmodulin-dependent protein kinase II in the apical dendrites during long-term potentiation. J. Neurochem. 2000, 75, 373–382. [Google Scholar] [CrossRef]
- Bomont, P. Degradation of the Intermediate Filament Family by Gigaxonin. Methods Enzymol. 2016, 569, 215–231. [Google Scholar] [CrossRef]
- Carare, R.O.; Bernardes-Silva, M.; Newman, T.A.; Page, A.M.; Nicoll, J.A.R.; Perry, V.H.; Weller, R.O. Solutes, but not cells, drain from the brain parenchyma along basement membranes of capillaries and arteries: Significance for cerebral amyloid angiopathy and neuroimmunology. Neuropathol. Appl. Neurobiol. 2008, 34, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Gafson, A.R.; Barthélemy, N.R.; Bomont, P.; Carare, R.O.; Durham, H.D.; Julien, J.-P.; Kuhle, J.; Leppert, D.; Nixon, R.A.; Weller, R.O.; et al. Neurofilaments: Neurobiological foundations for biomarker applications. Brain 2020, 143, 1975–1998. [Google Scholar] [CrossRef] [PubMed]
- Thebault, S.; Tessier, D.; Lee, H.; Bowman, M.; Bar-Or, A.; Arnold, D.L.; Atkins, H.; Tabard-Cossa, V.; Freedman, M.S. High Serum Neurofilament Light Chain normalises after Haematopoietic Stem Cell Transplant for MS. Neurol. Neuroimmunol. Neuroinflamm. 2019, 6, e598. [Google Scholar] [CrossRef] [PubMed]
- Kalm, M.; Boström, M.; Sandelius, Å.; Eriksson, Y.; Ek, C.J.; Blennow, K.; Björk-Eriksson, T.; Zetterberg, H. Serum concentrations of the axonal injury marker neurofilament light protein are not influenced by blood-brain barrier permeability. Brain Res. 2017, 1668, 12–19. [Google Scholar] [CrossRef]
- Uher, T.; McComb, M.; Galkin, S.; Srpova, B.; Oechtering, J.; Barro, C.; Tyblova, M.; Bergsland, N.; Krasensky, J.; Dwyer, M.; et al. Neurofilament levels are associated with blood–brain barrier integrity, lymphocyte extravasation, and risk factors following the first demyelinating event in multiple sclerosis. Mult. Scler. J. 2020, 1–12. [Google Scholar] [CrossRef]
- Bergman, J.; Dring, A.; Zetterberg, H.; Blennow, K.; Norgren, N.; Gilthorpe, J.; Bergenheim, T.; Svenningsson, A. Neurofilament light in CSF and serum is a sensitive marker for axonal white matter injury in MS. Neurol. Neuroimmunol. Neuroinflamm. 2016, 3, e271. [Google Scholar] [CrossRef]
- Akgün, K.; Kretschmann, N.; Haase, R.; Proschmann, U.; Kitzler, H.H.; Reichmann, H.; Ziemssen, T. Profiling individual clinical responses by high-frequency serum neurofilament assessment in MS. Neurol. Neuroimmunol. NeuroInflamm. 2019, 6, 1–12. [Google Scholar] [CrossRef]
- Disanto, G.; Barro, C.; Benkert, P.; Naegelin, Y.; Schädelin, S.; Giardiello, A.; Zecca, C.; Blennow, K.; Zetterberg, H.; Leppert, D.; et al. Serum Neurofilament light: A biomarker of neuronal damage in multiple sclerosis. Ann. Neurol. 2017, 81, 857–870. [Google Scholar] [CrossRef]
- Barro, C.; Benkert, P.; Disanto, G.; Tsagkas, C.; Amann, M.; Naegelin, Y.; Leppert, D.; Gobbi, C.; Granziera, C.; Yaldizli, Ö.; et al. Serum neurofilament as a predictor of disease worsening and brain and spinal cord atrophy in multiple sclerosis. Brain 2018, 141, 2382–2391. [Google Scholar] [CrossRef]
- Khalil, M.; Pirpamer, L.; Hofer, E.; Voortman, M.M.; Barro, C.; Leppert, D.; Benkert, P.; Ropele, S.; Enzinger, C.; Fazekas, F.; et al. Serum neurofilament light levels in normal aging and their association with morphologic brain changes. Nat. Commun. 2020, 11, 1–9. [Google Scholar] [CrossRef]
- Manouchehrinia, A.; Piehl, F.; Hillert, J.; Kuhle, J.; Alfredsson, L.; Olsson, T.; Kockum, I. Confounding effect of blood volume and body mass index on blood neurofilament light chain levels. Ann. Clin. Transl. Neurol. 2020, 7, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Korley, F.K.; Goldstick, J.; Mastali, M.; Van Eyk, J.E.; Barsan, W.; Meurer, W.J.; Sussman, J.; Falk, H.; Levine, D. Serum NfL (Neurofilament Light Chain) Levels and Incident Stroke in Adults with Diabetes Mellitus. Stroke 2019, 50, 1669–1675. [Google Scholar] [CrossRef] [PubMed]
- Bomont, P.; Cavalier, L.; Blondeau, F.; Hamida, C. Ben; Belal, S.; Tazir, M.; Demir, E.; Topaloglu, H.; Korinthenberg, R.; Tüysüz, B.; et al. The gene encoding gigaxonin, a new member of the cytoskeletal BTB/kelch repeat family, is mutated in giant axonal neuropathy. Nat. Genet. 2000, 26, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Mersiyanova, I.V.; Perepelov, A.V.; Polyakov, A.V.; Sitnikov, V.F.; Dadali, E.L.; Oparin, R.B.; Petrin, A.N.; Evgrafov, O.V. A new variant of Charcot-Marie-Tooth disease type 2 is probably the result of a mutation in the neurofilament-light gene. Am. J. Hum. Genet. 2000, 67, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Figlewicz, D.A.; Krizus, A.; Martinoli, M.G.; Meininger, V.; Dib, M.; Rouleau, G.A.; Julien, J.P. Variants of the heavy neurofilament subunit are associated with the development of amyotrophic lateral sclerosis. Hum. Mol. Genet. 1994, 3, 1757–1761. [Google Scholar] [CrossRef]
- Silber, E.; Semra, Y.K.; Gregson, N.A.; Sharief, M.K. Patients with progressive multiple sclerosis have elevated antibodies to neurofilament subunit. Neurology 2002, 58, 1372–1381. [Google Scholar] [CrossRef]
- Sádaba, M.C.; Tzartos, J.; Paíno, C.; García-Villanueva, M.; Álvarez-Cermeño, J.C.; Villar, L.M.; Esiri, M.M. Axonal and oligodendrocyte-localized IgM and IgG deposits in MS lesions. J. Neuroimmunol. 2012, 247, 86–94. [Google Scholar] [CrossRef]
- Puentes, F.; van der Star, B.J.; Boomkamp, S.D.; Kipp, M.; Boon, L.; Bosca, I.; Raffel, J.; Gnanapavan, S.; van der Valk, P.; Stephenson, J.; et al. Neurofilament light as an immune target for pathogenic antibodies. Immunology 2017, 152, 580–588. [Google Scholar] [CrossRef]
- Eikelenboom, M.J.; Petzold, A.; Lazeron, R.H.C.; Silber, E.; Sharief, M.; Thompson, E.J.; Barkhof, F.; Giovannoni, G.; Polman, C.H.; Uitdehaag, B.M.J. Multiple sclerosis: Neurofilament light chain antibodies are correlated to cerebral atrophy. Neurology 2003, 60, 219–223. [Google Scholar] [CrossRef]
- Amor, S.; van der Star, B.J.; Bosca, I.; Raffel, J.; Gnanapavan, S.; Watchorn, J.; Kuhle, J.; Giovannoni, G.; Baker, D.; Malaspina, A.; et al. Neurofilament light antibodies in serum reflect response to natalizumab treatment in multiple sclerosis. Mult. Scler. J. 2014, 20, 1355–1362. [Google Scholar] [CrossRef]
- Schlaepfer, W.W.; Lee, C.; Lee, V.M.; Zimmerman, U.-J.P. An Immunoblot Study of Neurofilament Degradation In Situ and During Calcium-Activated Proteolysis. J. Neurochem. 1985, 44, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, M.E.; Sternberger, N.H.; Sternberger, L.A. Phosphorylation protects neurofilaments against proteolysis. J. Neuroimmunol. 1987, 14, 149–160. [Google Scholar] [CrossRef]
- Pant, H.C. Dephosphorylation of neurofilament proteins enhances their susceptibility to degradation by calpain. Biochem. J. 1988, 256, 665–668. [Google Scholar] [CrossRef] [PubMed]
- Koel-Simmelink, M.J.A.; Teunissen, C.E.; Behradkia, P.; Blankenstein, M.A.; Petzold, A. The neurofilament light chain is not stable in vitro. Ann. Neurol. 2011, 69, 1065–1066. [Google Scholar] [CrossRef] [PubMed]
- Petzold, A.; Keir, G.; Green, A.J.E.; Giovannoni, G.; Thompson, E.J. A specific ELISA for measuring neurofilament heavy chain phosphoforms. J. Immunol. Methods 2003, 278, 179–190. [Google Scholar] [CrossRef]
- Leppert, D.; Petzold, A.; Regeniter, A.; Schindler, C.; Mehling, M.; Anthony, D.C.; Kappos, L.; Lindberg, R.L.P. Neurofilament heavy chain in CSF correlates with relapses and disability in multiple sclerosis. Neurology 2011, 76, 1206–1213. [Google Scholar]
- Kuhle, J.; Plattner, K.; Bestwick, J.P.; Lindberg, R.L.; Ramagopalan, S.V.; Norgren, N.; Nissim, A.; Malaspina, A.; Leppert, D.; Giovannoni, G.; et al. A comparative study of CSF neurofilament light and heavy chain protein in MS. Mult. Scler. J. 2013, 19, 1597–1603. [Google Scholar] [CrossRef]
- Kuhle, J.; Barro, C.; Andreasson, U.; Derfuss, T.; Lindberg, R.; Sandelius, Å.; Liman, V.; Norgren, N.; Blennow, K.; Zetterberg, H. Comparison of three analytical platforms for quantification of the neurofilament light chain in blood samples: ELISA, electrochemiluminescence immunoassay and Simoa. Clin. Chem. Lab. Med. 2016, 54, 1655–1661. [Google Scholar] [CrossRef]
- Wilson, D.H.; Rissin, D.M.; Kan, C.W.; Fournier, D.R.; Piech, T.; Campbell, T.G.; Meyer, R.E.; Fishburn, M.W.; Cabrera, C.; Patel, P.P.; et al. The Simoa HD-1 Analyzer: A Novel Fully Automated Digital Immunoassay Analyzer with Single-Molecule Sensitivity and Multiplexing. J. Lab. Autom. 2016, 21, 533–547. [Google Scholar] [CrossRef]
- Carvalho, T. Siemens Healthineers, Novartis Partner to Develop NfL Diagnostic Test. 2020. Available online: https://multiplesclerosisnewstoday.com/news-posts/2020/09/21/siemens-healthineers-novartis-partner-to-develop-new-nfl-diagnostic-test-for-ms/ (accessed on 28 October 2020).
- Khalil, M.; Teunissen, C.E.; Otto, M.; Piehl, F.; Sormani, M.P.; Gattringer, T.; Barro, C.; Kappos, L.; Comabella, M.; Fazekas, F.; et al. Neurofilaments as biomarkers in neurological disorders. Nat. Rev. Neurol. 2018, 14, 577–589. [Google Scholar] [CrossRef]
- Forgrave, L.M.; Ma, M.; Best, J.R.; DeMarco, M. The diagnostic performance of neurofilament light chain in CSF and blood for Alzheimer’s disease, frontotemporal dementia, and amyotrophic lateral sclerosis: A systematic review and meta-analysis. Alzheimer’s Dement. Diagnosis Assess. Dis. Monit. 2019, 11, 730–743. [Google Scholar] [CrossRef] [PubMed]
- Quiroz, Y.T.; Zetterberg, H.; Reiman, E.M.; Chen, Y.; Su, Y.; Fox-Fuller, J.T.; Garcia, G.; Villegas, A.; Sepulveda-Falla, D.; Villada, M.; et al. Plasma neurofilament light chain in the presenilin 1 E280A autosomal dominant Alzheimer’s disease kindred: A cross-sectional and longitudinal cohort study. Lancet Neurol. 2020, 19, 513–521. [Google Scholar] [CrossRef]
- Wilke, C.; dos Santos, M.C.T.; Schulte, C.; Deuschle, C.; Scheller, D.; Verbelen, M.; Brockmann, K.; von Thaler, A.K.; Sünkel, U.; Roeben, B.; et al. Intraindividual Neurofilament Dynamics in Serum Mark the Conversion to Sporadic Parkinson’s Disease. Mov. Disord. 2020, 35, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, H.H.; Soares, C.B.; Høgedal, S.S.; Madsen, J.S.; Hansen, R.B.; Christensen, A.A.; Madsen, C.; Clausen, B.H.; Frich, L.H.; Degn, M.; et al. Acute Neurofilament Light Chain Plasma Levels Correlate With Stroke Severity and Clinical Outcome in Ischemic Stroke Patients. Front. Neurol. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, G.C.; Alder, M.L.; Smothers, C.G.; Still, C.H.; Webel, A.R.; Moore, S.M. Diagnosis of ischemic stroke using circulating levels of brain-specific proteins measured via high-sensitivity digital ELISA. Brain Res. 2020, 1739. [Google Scholar] [CrossRef]
- Ameres, M.; Brandstetter, S.; Toncheva, A.A.; Kabesch, M.; Leppert, D.; Kuhle, J.; Wellmann, S. Association of neuronal injury blood marker neurofilament light chain with mild-to-moderate COVID-19. J. Neurol. 2020. [Google Scholar] [CrossRef]
- Confavreux, C.; Vukusic, S. The clinical course of multiple sclerosis. Handb. Clin. Neurol. 2014, 122, 343–369. [Google Scholar] [CrossRef]
- Igra, M.S.; Paling, D.; Wattjes, M.P.; Connolly, D.J.A.; Hoggard, N. Multiple sclerosis update: Use of MRI for early diagnosis, disease monitoring and assessment of treatment related complications. Br. J. Radiol. 2017, 90, 20160721. [Google Scholar] [CrossRef]
- Wattjes, M.P.; Steenwijk, M.D.; Stangel, M. MRI in the Diagnosis and Monitoring of Multiple Sclerosis: An Update. Clin. Neuroradiol. 2015, 25, 157–165. [Google Scholar] [CrossRef]
- Wattjes, M.P.; Rovira, À.; Miller, D.; Yousry, T.A.; Sormani, M.P.; De Stefano, N.; Tintoré, M.; Auger, C.; Tur, C.; Filippi, M.; et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis—Establishing disease prognosis and monitoring patients. Nat. Rev. Neurol. 2015, 11, 597–606. [Google Scholar] [CrossRef]
- Freedman, M.S.; Thompson, E.J.; Deisenhammer, F.; Giovannoni, G.; Grimsley, G.; Keir, G.; Öhman, S.; Racke, M.K.; Sharief, M.; Sindic, C.J.M.; et al. Recommended standard of cerebrospinal fluid analysis in the diagnosis of multiple sclerosis: A consensus statement. Arch. Neurol. 2005, 62, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Lennon, V.A.; Wingerchuk, D.M.; Kryzer, T.J.; Pittock, S.J.; Lucchinetti, C.F.; Fujihara, K.; Nakashima, I.; Weinshenker, B.G. A serum autoantibody marker of neuromyelitis optica: Distinction from multiple sclerosis. Lancet 2004, 364, 2106–2112. [Google Scholar] [CrossRef]
- Waters, P.; Woodhall, M.; O’Connor, K.C.; Reindl, M.; Lang, B.; Sato, D.K.; Jurynczyk, M.; Tackley, G.; Rocha, J.; Takahashi, T.; et al. MOG cell-based assay detects non-MS patients with inflammatory neurologic disease. Neurol. Neuroimmunol. NeuroInflamm. 2015, 2, e89. [Google Scholar] [CrossRef] [PubMed]
- Subramanyam, M.; Goelz, S.; Natarajan, A.; Lee, S.; Plavina, T.; Scanlon, J.V.; Sandrock, A.; Bozic, C. Risk of Natalizumab-Associated Progressive Multifocal Leukoencephalopathy. N. Engl. J. Med. 2012, 366, 1870–1880. [Google Scholar]
- Delcoigne, B.; Manouchehrinia, A.; Barro, C.; Benkert, P.; Michalak, Z.; Kappos, L.; Leppert, D.; Tsai, J.A.; Plavina, T.; Kieseier, B.C.; et al. Blood neurofilament light levels segregate treatment effects in multiple sclerosis. Neurology 2020, 94, e1201–e1212. [Google Scholar] [CrossRef]
- Novakova, L.; Zetterberg, H.; Sundström, P.; Axelsson, M.; Khademi, M.; Gunnarsson, M.; Malmeström, C.; Svenningsson, A.; Olsson, T.; Piehl, F.; et al. Monitoring disease activity in multiple sclerosis using serum neurofilament light protein. Neurology 2017, 89, 2230–2237. [Google Scholar] [CrossRef]
- Bittner, S.; Steffen, F.; Uphaus, T.; Muthuraman, M.; Fleischer, V.; Salmen, A.; Luessi, F.; Berthele, A.; Klotz, L.; Meuth, S.G.; et al. Clinical implications of serum neurofilament in newly diagnosed MS patients: A longitudinal multicentre cohort study. EBioMedicine 2020, 56, 1–13. [Google Scholar] [CrossRef]
- Varhaug, K.N.; Barro, C.; Bjørnevik, K.; Myhr, K.M.; Torkildsen, Ø.; Wergeland, S.; Bindoff, L.A.; Kuhle, J.; Vedeler, C. Neurofilament light chain predicts disease activity in relapsing-remitting MS. Neurol. Neuroimmunol. NeuroInflamm. 2018, 5, e422. [Google Scholar] [CrossRef]
- Siller, N.; Kuhle, J.; Muthuraman, M.; Barro, C.; Uphaus, T.; Groppa, S.; Kappos, L.; Zipp, F.; Bittner, S. Serum neurofilament light chain is a biomarker of acute and chronic neuronal damage in early multiple sclerosis. Mult. Scler. J. 2018. [Google Scholar] [CrossRef]
- Kuhle, J.; Kropshofer, H.; Haering, D.A.; Kundu, U.; Meinert, R.; Barro, C.; Dahlke, F.; Tomic, D.; Leppert, D.; Kappos, L. Blood neurofilament light chain as a biomarker of MS disease activity and treatment response. Neurology 2019, 92, E1007–E1015. [Google Scholar] [CrossRef]
- Bitsch, A.; Schuchardt, J.; Bunkowski, S.; Kuhlmann, T.; Brück, W. Acute axonal injury in multiple sclerosis. Correlation with demyelination and inflammation. Brain 2000, 123, 1174–1183. [Google Scholar] [CrossRef] [PubMed]
- Yaldizli, O. Value of serum neurofilament light chain levels as a biomarker of suboptimal treatment response in MS clinical practice. ECTRIMS Online Library, 10 December 2018. [Google Scholar]
- Ferraro, D.; Guicciardi, C.; De Biasi, S.; Pinti, M.; Bedin, R.; Camera, V.; Vitetta, F.; Nasi, M.; Meletti, S.; Sola, P. Plasma neurofilaments correlate with disability in progressive multiple sclerosis patients. Acta Neurol. Scand. 2020, 141, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Högel, H.; Rissanen, E.; Barro, C.; Matilainen, M.; Nylund, M.; Kuhle, J.; Airas, L. Serum glial fibrillary acidic protein correlates with multiple sclerosis disease severity. Mult. Scler. J. 2020, 26, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Filippi, P.; Vestenická, V.; Siarnik, P.; Sivakova, M.; Čopíková-Cudráková, D.; Belan, V.; Hanes, J.; Novák, M.; Kollar, B.; Turcani, P. Neurofilament light chain and MRI volume parameters as markers of neurodegeneration in multiple sclerosis. Neuro Endocrinol. Lett. 2020, 41, 17–26. [Google Scholar] [PubMed]
- Mattioli, F.; Bellomi, F.; Stampatori, C.; Mariotto, S.; Ferrari, S.; Monaco, S.; Mancinelli, C.; Capra, R. Longitudinal serum neurofilament light chain (sNfL) concentration relates to cognitive function in multiple sclerosis patients. J. Neurol. 2020. [Google Scholar] [CrossRef]
- Bridel, C.; van Wieringen, W.N.; Zetterberg, H.; Tijms, B.M.; Teunissen, C.E.; Alvarez-Cermeño, J.C.; Andreasson, U.; Axelsson, M.; Bäckström, D.C.; Bartos, A.; et al. Diagnostic Value of Cerebrospinal Fluid Neurofilament Light Protein in Neurology. JAMA Neurol. 2019, 76, 1035. [Google Scholar] [CrossRef]
- Jakimovski, D.; Zivadinov, R.; Ramanthan, M.; Hagemeier, J.; Weinstock-Guttman, B.; Tomic, D.; Kropshofer, H.; Fuchs, T.A.; Barro, C.; Leppert, D.; et al. Serum neurofilament light chain level associations with clinical and cognitive performance in multiple sclerosis: A longitudinal retrospective 5-year study. Mult. Scler. J. 2019. [Google Scholar] [CrossRef]
- Reinert, M.C.; Benkert, P.; Wuerfel, J.; Michalak, Z.; Ruberte, E.; Barro, C.; Huppke, P.; Stark, W.; Kropshofer, H.; Tomic, D.; et al. Serum neurofilament light chain is a useful biomarker in pediatric multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 2020, 7. [Google Scholar] [CrossRef]
- Sejbaek, T.; Nielsen, H.H.; Penner, N.; Plavina, T.; Mendoza, J.P.; Martin, N.A.; Elkjaer, M.L.; Ravnborg, M.H.; Illes, Z. Dimethyl fumarate decreases neurofilament light chain in CSF and blood of treatment naïve relapsing MS patients. J. Neurol. Neurosurg. Psychiatry 2019, 90, 1324–1330. [Google Scholar] [CrossRef]
- Piehl, F.; Kockum, I.; Khademi, M.; Blennow, K.; Lycke, J.; Zetterberg, H.; Olsson, T. Plasma neurofilament light chain levels in patients with MS switching from injectable therapies to fingolimod. Mult. Scler. J. 2017. [Google Scholar] [CrossRef]
- Gunnarsson, M.; Malmeström, C.; Axelsson, M.; Sundström, P.; Dahle, C.; Vrethem, M.; Olsson, T.; Piehl, F.; Norgren, N.; Rosengren, L.; et al. Axonal damage in relapsing multiple sclerosis is markedly reduced by natalizumab. Ann. Neurol. 2011, 69, 83–89. [Google Scholar] [CrossRef] [PubMed]
- de Flon, P.; Laurell, K.; Sundström, P.; Blennow, K.; Söderström, L.; Zetterberg, H.; Gunnarsson, M.; Svenningsson, A. Comparison of plasma and cerebrospinal fluid neurofilament light in a multiple sclerosis trial. Acta Neurol. Scand. 2019, 139, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Cross, A. Ocrelizumab treatment reduced levels of neurofilament light chain and numbers of B cells in the cerebrospinal fluid of patients with relapsing multiple sclerosis in the OBOE study S56.008. Neurology 2019, 92, 52. [Google Scholar]
- Hauser, S.L.; Bar-Or, A.; Cohen, J.A.; Comi, G.; Correale, J.; Coyle, P.K.; Cross, A.H.; de Seze, J.; Leppert, D.; Montalban, X.; et al. Ofatumumab versus Teriflunomide in Multiple Sclerosis. N. Engl. J. Med. 2020, 383, 546–557. [Google Scholar] [CrossRef]
- Kuhle, J.; Kropshofer, H.; Barro, C.; Meinert, R.; Haring, D.A.; Leppert, D.; Tomic, D.; Dahlke, F.; Kuhle, J.; Kropshofer, H.; et al. Siponimod Reduces Neurofilament Light Chain Blood Levels in Secondary Progressive Multiple Sclerosis Patients. Neurology 2018, 90 (Suppl. 15), S8.006. [Google Scholar]
- Bar-Or, A. Blood neurofilament light levels are lowered to a healthy donor range in patients with RMS and PPMS following ocrelizumab treatment. ECTRIMS Online Library, 9 December 2019. [Google Scholar]
- Kapoor, R.; Sellebjerg, F.; Hartung, H.-P.; Arnold, D.L.; Freedman, M.S.; Jeffery, D.; Miller, A.; Edwards, K.R.; Singh, C.M.; Chang, I.; et al. Natalizumab reduced serum levels of neurofilament light chain in secondary progressive multiple sclerosis patients from the phase 3 ASCEND study. Mult. Scler. J. 2018, 24, 988. [Google Scholar] [CrossRef]
- Kuhle, J.; Kropshofer, H.; Haring, D.A.; Barro, C.; Dahlke, F.; Leppert, D.; Tomic, D. Neurofilament light levels in the blood of patients with secondary progressive MS are higher than in primary progressive MS and may predict brain atrophy in both MS subtypes. Mult. Scler. J. 2018, 24, 111. [Google Scholar] [CrossRef]
- Kapoor, R.; Smith, K.E.; Allegretta, M.; Arnold, D.L.; Carroll, W.; Comabella, M.; Furlan, R.; Harp, C.; Kuhle, J.; Leppert, D.; et al. Serum neurofilament light as a biomarker in progressive multiple sclerosis. Neurology 2020. [Google Scholar] [CrossRef]
- Kuhle, J.; Nourbakhsh, B.; Grant, D.; Morant, S.; Barro, C.; Yaldizli, Ö.; Pelletier, D.; Giovannoni, G.; Waubant, E.; Gnanapavan, S. Serum neurofilament is associated with progression of brain atrophy and disability in early MS. Neurology 2017, 88, 826–831. [Google Scholar] [CrossRef]
- Jakimovski, D.; Kuhle, J.; Ramanathan, M.; Barro, C.; Tomic, D.; Hagemeier, J.; Kropshofer, H.; Bergsland, N.; Leppert, D.; Dwyer, M.G.; et al. Serum neurofilament light chain levels associations with gray matter pathology: A 5-year longitudinal study. Ann. Clin. Transl. Neurol. 2019, 6, 1757–1770. [Google Scholar] [CrossRef]
- Manouchehrinia, A.; Stridh, P.; Khademi, M.; Leppert, D.; Barro, C.; Michalak, Z.; Benkert, P.; Lycke, J.; Alfredsson, L.; Kappos, L.; et al. Plasma neurofilament light levels are associated with risk of disability in multiple sclerosis. Neurology 2020, 94, e2457–e2467. [Google Scholar] [CrossRef] [PubMed]
- Lublin, F.D. Disease activity free status in MS. Mult. Scler. Relat. Disord. 2012, 1, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Thebault, S.; Abdoli, M.; Fereshtehnejad, S.M.; Tessier, D.; Tabard-Cossa, V.; Freedman, M.S. Serum neurofilament light chain predicts long term clinical outcomes in multiple sclerosis. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chitnis, T.; Gonzalez, C.; Healy, B.C.; Saxena, S.; Rosso, M.; Barro, C.; Michalak, Z.; Paul, A.; Kivisakk, P.; Diaz-Cruz, C.; et al. Neurofilament light chain serum levels correlate with 10-year MRI outcomes in multiple sclerosis. Ann. Clin. Transl. Neurol. 2018, 1478–1491. [Google Scholar] [CrossRef]
- Novakova, L.; Axelsson, M.; Khademi, M.; Zetterberg, H.; Blennow, K.; Malmeström, C.; Piehl, F.; Olsson, T.; Lycke, J. Cerebrospinal fluid biomarkers as a measure of disease activity and treatment efficacy in relapsing-remitting multiple sclerosis. J. Neurochem. 2017, 141, 296–304. [Google Scholar] [CrossRef]
- Bsteh, G.; Hegen, H.; Teuchner, B.; Amprosi, M.; Berek, K.; Ladstätter, F.; Wurth, S.; Auer, M.; Di Pauli, F.; Deisenhammer, F.; et al. Peripapillary retinal nerve fibre layer as measured by optical coherence tomography is a prognostic biomarker not only for physical but also for cognitive disability progression in multiple sclerosis. Mult. Scler. J. 2019, 25, 196–203. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thebault, S.; Booth, R.A.; Freedman, M.S. Blood Neurofilament Light Chain: The Neurologist’s Troponin? Biomedicines 2020, 8, 523. https://doi.org/10.3390/biomedicines8110523
Thebault S, Booth RA, Freedman MS. Blood Neurofilament Light Chain: The Neurologist’s Troponin? Biomedicines. 2020; 8(11):523. https://doi.org/10.3390/biomedicines8110523
Chicago/Turabian StyleThebault, Simon, Ronald A. Booth, and Mark S. Freedman. 2020. "Blood Neurofilament Light Chain: The Neurologist’s Troponin?" Biomedicines 8, no. 11: 523. https://doi.org/10.3390/biomedicines8110523
APA StyleThebault, S., Booth, R. A., & Freedman, M. S. (2020). Blood Neurofilament Light Chain: The Neurologist’s Troponin? Biomedicines, 8(11), 523. https://doi.org/10.3390/biomedicines8110523



