Monitoring the Redox Status in Multiple Sclerosis
Abstract
1. Introduction
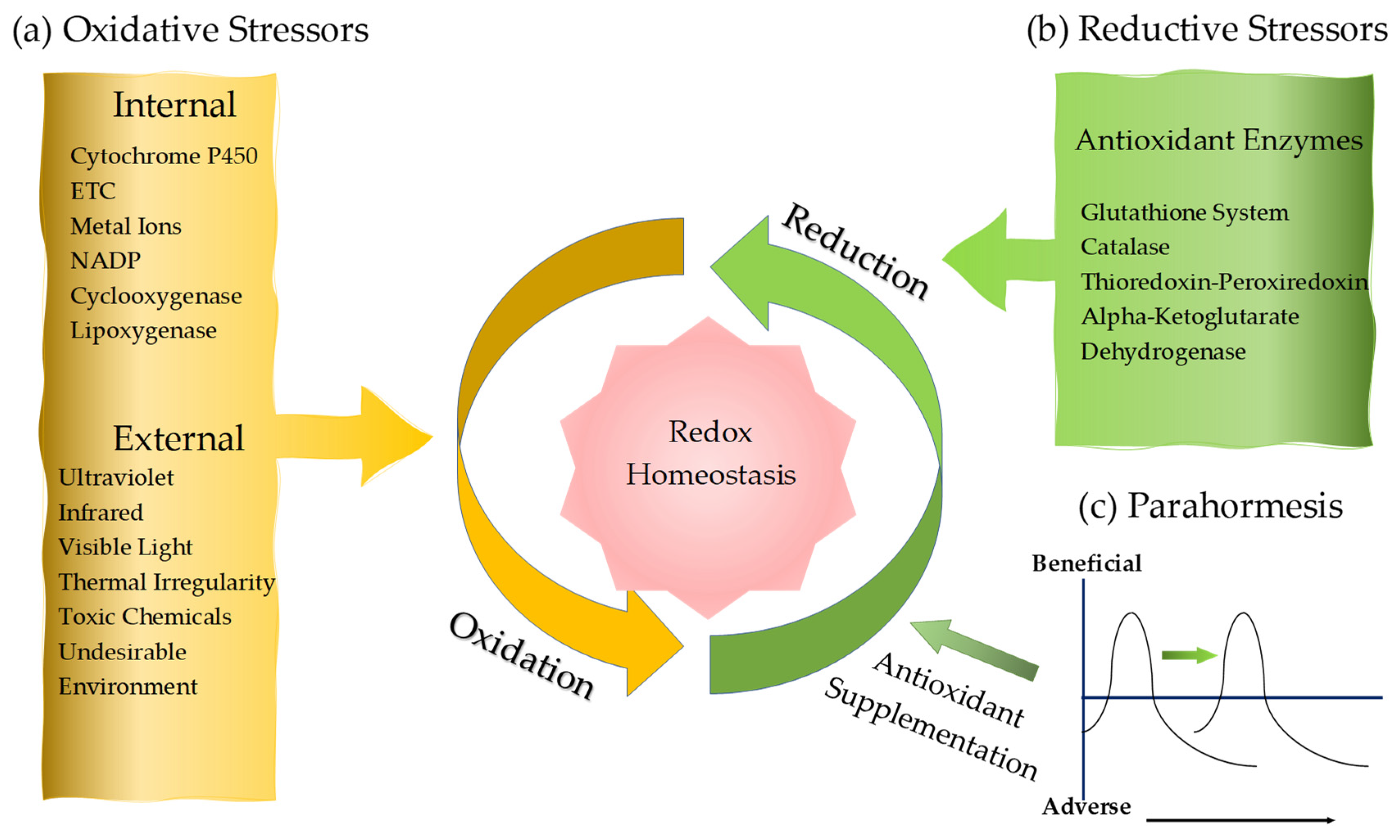
2. Oxidative Stress
2.1. Endogenous Oxidative Stressors: Oxidative Enzymes and Reactive Species
2.1.1. Oxidative Enzymes Generating Reactive Oxygen Species
2.1.2. Oxidative Enzymes Generating Reactive Nitrogen Species
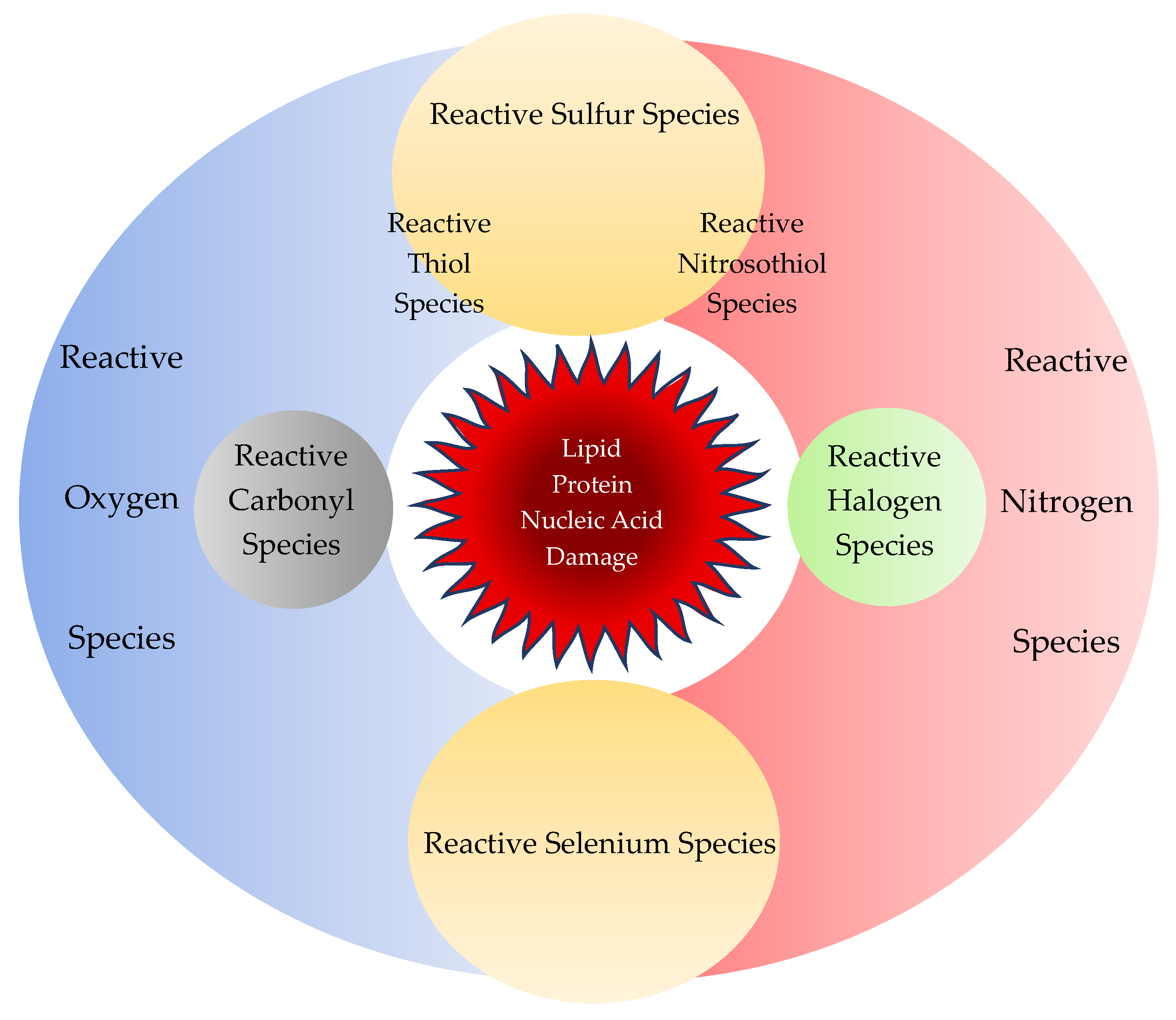
2.1.3. Reactive Sulfur Species
2.1.4. Reactive Carbonyl/Halogen/Selenium Species
2.1.5. Exogenous Oxidative Factors
3. Reductive Stress
3.1. Reactive Nucleophilic Species
3.2. Antioxidative Enzymes
3.3. Exogenous and Endogenous Antioxidants
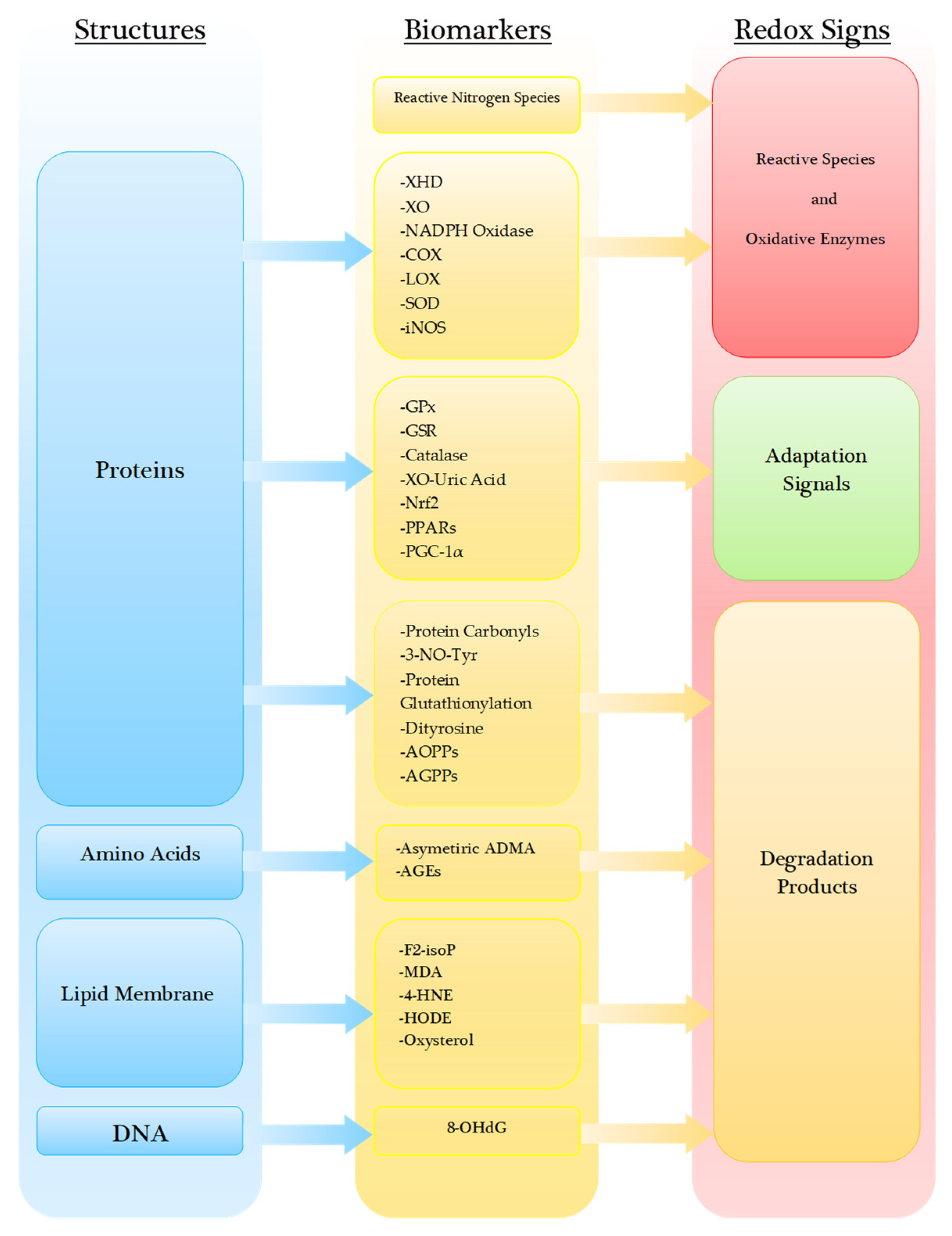
4. Degradation Products under Oxidative Stress
4.1. Proteins
4.2. Amino Acids
4.3. Lipid Membrane and Lipoproteins
4.4. Nucleic Acid
5. Conclusion and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| α | alpha |
| AD | Alzheimer’s disease |
| ADMA | asymmetric dimethylarginine |
| AGEs | advanced glycation end products |
| ALS | amyotrophic lateral sclerosis |
| AOPPs | advanced oxidation protein products |
| β | beta |
| CIS | clinically isolated syndrome |
| CNS | central nervous system |
| COX | cyclooxygenase |
| CSF | cerebrospinal fluid |
| CYP | cytochrome P450 |
| EDSS | expanded disability status scale |
| ELISA | enzyme-linked immunosorbent assay |
| ETC | electron transfer chain |
| F2-isoPs | F2-isoprostanes |
| GC/MS | gas chromatography-mass spectrometry |
| Gd+ | gadolinium |
| GPx | glutathione peroxidase |
| GSH | glutathione |
| GSSH | glutathione disulfide |
| GA | glatiramer |
| GPDH | ketoglutarat dehydrogenase |
| GSR | glutathione reductase |
| GWA | genome-wide association |
| HEL | hexanoyl-lysine |
| 4-HNE | 4-hydroxynonenal |
| HODE | hydroxyoctadecadienoic acid |
| HPLC | high-performance liquid chromatography |
| iNOS | inducible Nitric Oxide Synthase |
| IFN | Interferon |
| KEAP1 | Kelch-like ECH-associated protein 1 |
| KGDH | ketoglutarate dehydrogenase |
| LINE-1 | Long Interspersed Nuclear Element-1 |
| LOX | lipoxygenase |
| MDA | malondialdehyde |
| miRNA | micro RNA |
| MPO | myeloperoxidase |
| MRI | magnetic resonance imaging |
| MS | multiple sclerosis |
| NAD+ | nicotinamide adenine dinucleotide |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NOS | nitric oxide synthetase |
| 3-NO-Tyr | 3-nitrotyrosine |
| NOX | nicotinamide adenine dinucleotide phosphate oxidase |
| Nrf2 | nuclear factor erythroid 2-related factor |
| 8-OH2dG | 8-Hydroxy-2′-deoxyguanosine |
| 8-OHG | 8-hydroxyguanosine |
| OSMS | opticospinal phenotype of relapsing-remitting multiple sclerosis |
| oxLDL | oxidized low-density lipoprotein |
| PD | Parkinson’s disease |
| PGC-1 | peroxisome proliferator-activated receptor gamma coactivator 1 |
| PPARs | peroxisome proliferator-activated receptors |
| PUFAs | polyunsaturated fatty acids |
| RCS | reactive carbonyl species |
| redox | reduction-oxidation |
| RHS | reactive halogen species |
| RNS | reactive nitrogen species |
| ROS | reactive oxygen species |
| PPMS | primary progressive multiple sclerosis |
| PRMS | progressive-relapsing multiple sclerosis |
| RRMS | relapsing-remitting multiple sclerosis |
| RSS | reactive sulfur species |
| RSeS | reactive selenium species |
| SOD | superoxide dismutase |
| SPMS | secondary progressive multiple sclerosis |
| tNOx | total value nitric oxide |
| TRX-PRDX | thioredoxin-peroxiredoxin |
| TRXR | thioredoxin reductase |
| XDH | xanthine dehydrogenase |
| XO | xanthine oxidase |
Appendix A
Assessment of the Methodological Quality
| Classes | Types | Risk of Bias | |
|---|---|---|---|
| Reactive Species | Reactive Nitrogen Species | Unclear | |
| Oxidative Enzymes | Xanthine Dehydrogenase (XDH) | Unclear | |
| Nicotinamide Adenine Dinucleotide Phosphate (NADPH) Oxidase | Unclear | ||
| Superoxide Dismutase (SOD) | High risk | ||
| Inducible Nitric Oxide Synthase (iNOS) | High risk | ||
| Myeloperoxidase (MPO) | Unclear | ||
| Antioxidative Enzymes and Transcriptional Factors | Glutathione Peroxidase (GPx) | Low risk | |
| Glutathione Reductase (GSR) | High risk | ||
| Catalase | High risk | ||
| Xanthine oxidase (XO)-Uric Acid | Unclear | ||
| Nuclear Factor Erythroid 2-Related Factor (Nrf2) | Unclear | ||
| Peroxisome proliferator-activated receptors (PPARs) | Unclear | ||
| Peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) | Unclear | ||
| Degradation Products and End Products | Protein | Protein carbonyls | Low risk |
| 3-nitrotyrosin (3-NO-Tyr) | High risk | ||
| Protein glutathionylation | Unclear | ||
| Dityrosine | Unclear | ||
| Advanced oxidation protein products (AOPPs) | High risk | ||
| Advanced glycation end products (AGEs) | High risk | ||
| Amino acids | Asymmetric dimethylarginine (ADMA) | Unclear | |
| Lipid | F2-isoprostane (F2-isoP) | Low risk | |
| Malondialdehyde (MDA) | Low risk | ||
| 4-hydroxynonenal (4-HNE) | Unclear | ||
| Hydroxyoctadecadienoic acid (HODE) | Unclear | ||
| Oxysterol | Unclear | ||
| DNA | 8-dihydro-2′deoxyguanosine (8-oxodG) | Unclear | |
| Risk of Bias | Criteria |
|---|---|
| High risk | No meta-analysis or systematic review, fewer than five case–control and/or cohort studies, or presence of only expert review |
| Low risk | Presence of at least one meta-analysis or systematic review, without conflicting results |
| Unclear | Presence of only case–control study or cohort study, meta-analysis with conflicting results, or case–control studies with conflicting results |
References
- GBD 2016 Neurology Collaborators. Global, regional, and national burden of neurological disorders, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 459–480. [Google Scholar] [CrossRef]
- Fricska-Nagy, Z.; Füvesi, J.; Rózsa, C.; Komoly, S.; Jakab, G.; Csépány, T.; Jobbágy, Z.; Lencsés, G.; Vécsei, L.; Bencsik, K. The effects of fatigue, depression and the level of disability on the health-related quality of life of glatiramer acetate-treated relapsing-remitting patients with multiple sclerosis in Hungary. Mult. Scler. Relat. Disord. 2016, 7, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Sandi, D.; Biernacki, T.; Szekeres, D.; Füvesi, J.; Kincses, Z.T.; Rózsa, C.; Mátyás, K.; Kása, K.; Matolcsi, J.; Zboznovits, D.; et al. Prevalence of cognitive impairment among Hungarian patients with relapsing-remitting multiple sclerosis and clinically isolated syndrome. Mult. Scler. Relat. Disord. 2017, 17, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Toldi, J.; Vécsei, L. Exploring the Etiological Links behind Neurodegenerative Diseases: Inflammatory Cytokines and Bioactive Kynurenines. Int. J. Mol. Sci. 2020, 21, 2431. [Google Scholar] [CrossRef] [PubMed]
- Boeschoten, R.E.; Braamse, A.M.J.; Beekman, A.T.F.; Cuijpers, P.; van Oppen, P.; Dekker, J.; Uitdehaag, B.M.J. Prevalence of Depression and Anxiety in Multiple Sclerosis: A Systematic Review and Meta-Analysis. J. Neurol. Sci. 2017, 372, 331–341. [Google Scholar] [CrossRef]
- Hunt, C.; Macedo e Cordeiro, T.; Suchting, R.; de Dios, C.; Cuellar Leal, V.A.; Soares, J.C.; Dantzer, R.; Teixeira, A.L.; Selvaraj, S. Effect of immune activation on the kynurenine pathway and depression symptoms—A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2020, 118, 514. [Google Scholar] [CrossRef]
- Jovanovic, F.; Candido, K.D.; Knezevic, N.N. The Role of the Kynurenine Signaling Pathway in Different Chronic Pain Conditions and Potential Use of Therapeutic Agents. Int. J. Mol. Sci. 2020, 21, 6045. [Google Scholar] [CrossRef]
- Waubant, E.; Lucas, R.; Mowry, E.; Graves, J.; Olsson, T.; Alfredsson, L.; Langer-Gould, A. Environmental and genetic risk factors for MS: An integrated review. Ann. Clin. Transl. Neurol. 2019, 6, 1905–1922. [Google Scholar] [CrossRef]
- Biernacki, T.; Sandi, D.; Kincses, Z.T.; Füvesi, J.; Rózsa, C.; Mátyás, K.; Vécsei, L.; Bencsik, K. Contributing factors to health-related quality of life in multiple sclerosis. Brain Behav. 2019, 9, e01466. [Google Scholar] [CrossRef]
- Rajda, C.; Majláth, Z.; Pukoli, D.; Vécsei, L. Kynurenines and Multiple Sclerosis: The Dialogue between the Immune System and the Central Nervous System. Int. J. Mol. Sci. 2015, 16, 18270–18282. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Wang, M.C.; Wang, Y.N.; Hu, H.H.; Liu, Q.Q.; Liu, H.J.; Zhao, Y.Y. Redox signaling and Alzheimer’s disease: From pathomechanism insights to biomarker discovery and therapy strategy. Biomark. Res. 2020, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Filippi, M.; Bar-Or, A.; Piehl, F.; Preziosa, P.; Solari, A.; Vukusic, S.; Rocca, M.A. Multiple sclerosis. Nat. Rev. Dis. Primers 2018, 4, 43. [Google Scholar] [CrossRef] [PubMed]
- Kincses, Z.T.; Tóth, E.; Bankó, N.; Veréb, D.; Szabó, N.; Csete, G.; Faragó, P.; Király, A.; Bencsik, K.; Vécsei, L. Grey matter atrophy in patients suffering from multiple sclerosis. Ideggyogy Sz. 2014, 67, 293–300. [Google Scholar] [PubMed]
- Tóth, E.; Faragó, P.; Király, A.; Szabó, N.; Veréb, D.; Kocsis, K.; Kincses, B.; Sandi, D.; Bencsik, K.; Vécsei, L.; et al. The Contribution of Various MRI Parameters to Clinical and Cognitive Disability in Multiple Sclerosis. Front. Neurol. 2019, 9, 1172. [Google Scholar] [CrossRef]
- Andravizou, A.; Dardiotis, E.; Artemiadis, A.; Sokratous, M.; Siokas, V.; Tsouris, Z.; Aloizou, A.M.; Nikolaidis, I.; Bakirtzis, C.; Tsivgoulis, G.; et al. Brain atrophy in multiple sclerosis: Mechanisms, clinical relevance and treatment options. Autoimmun. Highlights 2019, 10, 7. [Google Scholar] [CrossRef]
- Hartung, H.P.; Graf, J.; Aktas, O.; Mares, J.; Barnett, M.H. Diagnosis of multiple sclerosis: Revisions of the McDonald criteria 2017—Continuity and change. Curr. Opin. Neurol. 2019, 32, 327–337. [Google Scholar] [CrossRef]
- Iacobaeus, E.; Arrambide, G.; Pia Amato, M.; Derfuss, T.; Vukusic, S.; Hemmer, B.; Tintore, M.; Brundin, L.; 2018 ECTRIMS Focused Workshop Group. Aggressive multiple sclerosis (1): Towards a definition of the phenotype. Mult. Scler. 2020, 26, 1352458520925369. [Google Scholar] [CrossRef]
- Correale, J.; Marrodan, M.; Ysrraelit, M.C. Mechanisms of Neurodegeneration and Axonal Dysfunction in Progressive Multiple Sclerosis. Biomedicines 2019, 7, 14. [Google Scholar] [CrossRef]
- Melendez-Torres, G.J.; Armoiry, X.; Court, R.; Patterson, J.; Kan, A.; Auguste, P.; Madan, J.; Counsell, C.A.R.L.; Ciccarelli, O.; Clarke, A. Comparative effectiveness of beta-interferons and glatiramer acetate for relapsing-remitting multiple sclerosis: Systematic review and network meta-analysis of trials including recommended dosages. BMC Neurol. 2018, 18, 162. [Google Scholar] [CrossRef]
- Goodman, A.D.; Anadani, N.; Gerwitz, L. Siponimod in the treatment of multiple sclerosis. Expert Opin. Investig. Drugs 2019, 28, 1051–1057. [Google Scholar] [CrossRef]
- Robertson, D.; Moreo, N. Disease-Modifying Therapies in Multiple Sclerosis: Overview and Treatment Considerations. Fed Pract. 2016, 33, 28–34. [Google Scholar] [PubMed]
- Jonasson, E.; Sejbaek, T. Diroximel fumarate in the treatment of multiple sclerosis. Neurodegener. Dis. Manag. 2020, 10, 267–276. [Google Scholar] [CrossRef]
- Hauser, S.L.; Bar-Or, A.; Cohen, J.A.; Comi, G.; Correale, J.; Coyle, P.K.; Selmaj, K. Ofatumumab versus teriflunomide in multiple sclerosis. N. Eng. J. Med. 2020, 383, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Hojati, Z.; Kay, M.; Dehghanian, F. Mechanism of Action of Interferon Beta in Treatment of Multiple Sclerosis. In Multiple Sclerosis, A Mechanistic View, 1st ed.; Minagar, A., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 365–392. [Google Scholar]
- Ziemssen, T.; Schrempf, W. Glatiramer acetate: Mechanisms of action in multiple sclerosis. Int. Rev. Neurobiol. 2007, 79, 537–570. [Google Scholar]
- De Angelis, F.; John, N.A.; Brownlee, W.J. Disease-modifying therapies for multiple sclerosis. BMJ 2018, 363, k4674. [Google Scholar] [CrossRef]
- Rajda, C.; Bergquist, J.; Vécsei, L. Kynurenines, redox disturbances and neurodegeneration in multiple sclerosis. J. Neural. Transm. Suppl. 2007, 72, 323–329. [Google Scholar]
- Rajda, C.; Pukoli, D.; Bende, Z.; Majláth, Z.; Vécsei, L. Excitotoxins, Mitochondrial and Redox Disturbances in Multiple Sclerosis. Int. J. Mol. Sci. 2017, 18, 353. [Google Scholar] [CrossRef]
- Sas, K.; Szabó, E.; Vécsei, L. Mitochondria, Oxidative Stress and the Kynurenine System, with a Focus on Ageing and Neuroprotection. Molecules 2018, 23, 191. [Google Scholar] [CrossRef]
- Fiorini, A.; Koudriavtseva, T.; Bucaj, E.; Coccia, R.; Foppoli, C.; Giorgi, A.; Schininà, M.E.; Di Domenico, F.; De Marco, F.; Perluigi, M. Involvement of oxidative stress in occurrence of relapses in multiple sclerosis: The spectrum of oxidatively modified serum proteins detected by proteomics and redox proteomics analysis. PLoS ONE 2013, 8, e65184. [Google Scholar] [CrossRef]
- Choi, I.Y.; Lee, P.; Adany, P.; Hughes, A.J.; Belliston, S.; Denney, D.R.; Lynch, S.G. In vivo evidence of oxidative stress in brains of patients with progressive multiple sclerosis. Mult. Scler. 2018, 24, 1029–1038. [Google Scholar] [CrossRef]
- Barcelos, I.P.; Troxell, R.M.; Graves, J.S. Mitochondrial Dysfunction and Multiple Sclerosis. Biology 2019, 8, 37. [Google Scholar] [CrossRef] [PubMed]
- Cortese-Krott, M.M.; Koning, A.; Kuhnle, G.G.C.; Nagy, P.; Bianco, C.L.; Pasch, A.; Wink, D.A.; Fukuto, J.M.; Jackson, A.A.; van Goor, H.; et al. The Reactive Species Interactome: Evolutionary Emergence, Biological Significance, and Opportunities for Redox Metabolomics and Personalized Medicine. Antioxid. Redox Signal 2017, 27, 684–712. [Google Scholar] [CrossRef]
- Santolini, J.; Wootton, S.A.; Jackson, A.A.; Feelisch, M. The Redox architecture of physiological function. Curr. Opin. Physiol. 2019, 9, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. On the history of oxidative stress: Concept and some aspects of current development. Curr. Opin. Toxicol. 2018, 7, 122–126. [Google Scholar] [CrossRef]
- Viña, J.; Lloret, A.; Vallés, S.L.; Borrás, C.; Badía, M.C.; Pallardó, F.V.; Sastre, J.; Alonso, M.D. Mitochondrial oxidant signalling in Alzheimer′s disease. J. Alzheimers Dis. 2007, 11, 175–181. [Google Scholar] [CrossRef]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxid. Med. Cell Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef]
- Török, N.; Majláth, Z.; Fülöp, F.; Toldi, J.; Vécsei, L. Brain Aging and Disorders of the Central Nervous System: Kynurenines and Drug Metabolism. Curr. Drug Metab. 2016, 17, 412–429. [Google Scholar] [CrossRef]
- Frijhoff, J.; Winyard, P.G.; Zarkovic, N.; Davies, S.S.; Stocker, R.; Cheng, D.; Knight, A.R.; Taylor, E.L.; Oettrich, J.; Ruskovska, T.; et al. Clinical Relevance of Biomarkers of Oxidative Stress. Antioxid. Redox Signal 2015, 23, 1144–1170. [Google Scholar] [CrossRef]
- Tanaka, M.; Bohár, Z.; Vécsei, L. Are Kynurenines Accomplices or Principal Villains in Dementia? Maintenance of Kynurenine Metabolism. Molecules 2020, 25, 564. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative Stress: An Essential Factor in the Pathogenesis of Gastrointestinal Mucosal Diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef]
- Aguilera, G.; Colín-González, A.L.; Rangel-López, E.; Chavarría, A.; Santamaría, A. Redox Signaling, Neuroinflammation, and Neurodegeneration. Antioxid. Redox Signal. 2018, 28, 1626–1651. [Google Scholar] [CrossRef] [PubMed]
- Lushchak, V.I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chem. Biol. Interact. 2014, 224, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef] [PubMed]
- Collin, F. Chemical Basis of Reactive Oxygen Species Reactivity and Involvement in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 2407. [Google Scholar] [CrossRef]
- Drulovic, J.; Dujmovic, I.; Stojsavljevic, N.; Mesaros, S.; Andjelkovic, S.; Miljkovic, D.; Peric, V.; Dragutinovic, G.; Marinkovic, J.; Levic, Z.; et al. Uric acid levels in sera from patients with multiple sclerosis. J. Neurol. 2001, 248, 121–126. [Google Scholar] [CrossRef]
- Nasyrova, R.F.; Moskaleva, P.V.; Vaiman, E.E.; Shnayder, N.A.; Blatt, N.L.; Rizvanov, A.A. Genetic Factors of Nitric Oxide’s System in Psychoneurologic Disorders. Int. J. Mol. Sci. 2020, 21, 1604. [Google Scholar] [CrossRef]
- Doğan, H.O.; Yildiz, Ö.K. Serum NADPH oxidase concentrations and the associations with iron metabolism in relapsing remitting multiple sclerosis. J. Trace Elem. Med. Biol. 2019, 55, 39–43. [Google Scholar] [CrossRef]
- Inarrea, P.; Alarcia, R.; Alava, M.A.; Capablo, J.L.; Casanova, A.; Iñiguez, C.; Iturralde, M.; Larrodé, P.; Martín, J.; Mostacero, E.; et al. Mitochondrial complex enzyme activities and cytochrome C expression changes in multiple sclerosis. Mol. Neurobiol. 2014, 49, 1–9. [Google Scholar] [CrossRef]
- Ljubisavljevic, S.; Stojanovic, I.; Vojinovic, S.; Stojanov, D.; Stojanovic, S.; Cvetkovic, T.; Savic, D.; Pavlovic, D. The patients with clinically isolated syndrome and relapsing remitting multiple sclerosis show different levels of advanced protein oxidation products and total thiol content in plasma and CSF. Neurochem. Int. 2013, 62, 988–997. [Google Scholar] [CrossRef]
- Damiano, S.; Sasso, A.; De Felice, B.; Terrazzano, G.; Bresciamorra, V.; Carotenuto, A.; Orefice, N.S.; Orefice, G.; Vacca, G.; Belfiore, A.; et al. The IFN-beta 1b effect on Cu Zn superoxide dismutase (SOD1) in peripheral mononuclear blood cells of relapsing-remitting multiple sclerosis patients and in neuroblastoma SK-N-BE cells. Brain Res. Bull. 2015, 118, 1–6. [Google Scholar] [CrossRef]
- Tasset, I.; Bahamonde, C.; Agüera, E.; Conde, C.; Cruz, A.H.; Pérez-Herrera, A.; Gascón, F.; Giraldo, A.I.; Ruiz, M.C.; Lillo, R.; et al. Effect of natalizumab on oxidative damage biomarkers in relapsing-remitting multiple sclerosis. Pharmacol. Rep. 2013, 65, 624–631. [Google Scholar] [CrossRef]
- Gliozzi, M.; Scicchitano, M.; Bosco, F.; Musolino, V.; Carresi, C.; Scarano, F.; Maiuolo, J.; Nucera, S.; Maretta, A.; Paone, S.; et al. Modulation of Nitric Oxide Synthases by Oxidized LDLs: Role in Vascular Inflammation and Atherosclerosis Development. Int. J. Mol. Sci. 2019, 20, 3294. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Moratalla, N.; Gonzalez, A.; Aymerich, M.S.; López-Zabalza, M.J.; Pío, R.; de Castro, P.; Santiago, E. Monocyte inducible nitric oxide synthase in multiple sclerosis: Regulatory role of nitric oxide. Nitric Oxide 1997, 1, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Scapagnini, G.; Ravagna, A.; Bella, R.; Foresti, R.; Bates, T.E.; Giuffrida Stella, A.M.; Pennisi, G. Nitric oxide synthase is present in the cerebrospinal fluid of patients with active multiple sclerosis and is associated with increases in cerebrospinal fluid protein nitrotyrosine and S-nitrosothiols and with changes in glutathione levels. J. Neurosci. Res. 2002, 70, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Tasset, I.; Aguera, E.; Sanchez-Lopez, F.; Feijóo, M.; Giraldo, A.I.; Cruz, A.H.; Gascón, F.; Túnez, I. Peripheral oxidative stress in relapsing remitting multiple sclerosis. Clin. Biochem. 2012, 45, 440–444. [Google Scholar] [CrossRef]
- Cupp-Sutton, K.A.; Ashby, M.T. Biological Chemistry of Hydrogen Selenide. Antioxidants 2016, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Boylan, M.; Selvam, A.; Spallholz, J.E.; Björnstedt, M. Redox-Active Selenium Compounds—From Toxicity and Cell Death to Cancer Treatment. Nutrients 2015, 7, 3536–3556. [Google Scholar] [CrossRef]
- Ljubisavljevic, S.; Stojanovic, I.; Cvetkovic, T.; Vojinovic, S.; Stojanov, D.; Stojanovic, D.; Bojanic, V.; Stokanovic, D.; Pavlovic, D. Glutathione homeostasis disruption of erythrocytes, but not glutathione peroxidase activity change, is closely accompanied with neurological and radiological scoring of acute CNS inflammation. Neuroimmunomodulation 2014, 21, 13–20. [Google Scholar] [CrossRef]
- Zachara, B.; Gromadzinska, J.; Czernicki, J.; Maciejek, Z.; Chmielewski, H. Red blood cell glutathione peroxidase activity in multiple sclerosis. Klin. Wochenschr. 1984, 62, 179–182. [Google Scholar] [CrossRef]
- Calabrese, V.; Raffaele, R.; Cosentino, E.; Rizza, V. Changes in cerebrospinal fluid levels of malondialdehyde and glutathione reductase activity in multiple sclerosis. Int. J. Clin. Pharmacol. Res. 1994, 14, 119–123. [Google Scholar]
- Jensen, G.E.; Gissel-Nielsen, G.; Clausen, J. Leucocyte glutathione peroxidase activity and selenium level in multiple sclerosis. J. Neurol. Sci. 1980, 48, 61–67. [Google Scholar] [CrossRef]
- Hirsch, H.E.; Blanco, C.E.; Parks, M.E. Glycerol phosphate dehydrogenase: Reduced activity in multiple sclerosis plaques confirms localization in oligodendrocytes. J. Neurochem. 1980, 34, 760–762. [Google Scholar] [CrossRef] [PubMed]
- Ferret-Sena, V.; Capela, C.; Sena, A. Metabolic Dysfunction and Peroxisome Proliferator-Activated Receptors (PPAR) in Multiple Sclerosis. Int. J. Mol. Sci. 2018, 19, 1639. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.R.; Kallaur, A.P.; Simão, A.N.; Morimoto, H.K.; Lopes, J.; Panis, C.; Petenucci, D.L.; da Silva, E.; Cecchini, R.; Kaimen-Maciel, D.R.; et al. Oxidative stress in multiple sclerosis patients in clinical remission: Association with the expanded disability status scale. J. Neurol. Sci. 2012, 321, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Walczak, A.; Saluk, J.; Ponczek, M.B.; Majsterek, I. Oxidative modification of patient’s plasma proteins and its role in pathogenesis of multiple sclerosis. Clin. Biochem. 2012, 45, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Sadowska-Bartosz, I.; Adamczyk-Sowa, M.; Gajewska, A.; Bartosz, G. Oxidative modification of blood serum proteins in multiple sclerosis after interferon or mitoxantrone treatment. J. Neuroimmunol. 2014, 266, 7–74. [Google Scholar] [CrossRef] [PubMed]
- Zabaleta, M.; Marino, R.; Borges, J.; Camargo, B.; Ordaz, P.; De Sanctis, J.B.; Bianco, N.E. Activity profile in multiple sclerosis: An integrative approach. A preliminary report. Mult. Scler. 2002, 8, 343–349. [Google Scholar] [CrossRef]
- Iarlori, C.; Gambi, D.; Lugaresi, A.; Patruno, A.; Felaco, M.; Salvatore, M.; Speranza, L.; Reale, M. Reduction of free radicals in multiple sclerosis: Effect of glatiramer acetate (Copaxone). Mult. Scler. 2008, 14, 739–748. [Google Scholar] [CrossRef]
- Seven, A.; Aslan, M.; Incir, S.; Altintas, A. Evaluation of oxidative and nitrosative stress in relapsing remitting multiple sclerosis: Effect of corticosteroid therapy. Folia Neuropathol. 2013, 51, 58–64. [Google Scholar] [CrossRef]
- Stojanovic, I.; Vojinovic, S.; Ljubisavljevic, S.; Pavlovic, R.; Basic, J.; Pavlovic, D.; Ilic, A.; Cvetkovic, T.; Stukalov, M. INF-β1b therapy modulates L-arginine and nitric oxide metabolism in patients with relapse remittent multiple sclerosis. J. Neurol. Sci. 2012, 323, 187–192. [Google Scholar] [CrossRef]
- Teixeira, D.; Fernandes, R.; Prudêncio, C.; Vieira, M. 3-Nitrotyrosine quantification methods: Current concepts and future challenges. Biochimie 2016, 125, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Kukkuta Sarma, G.R.; Dsouza, D.S.; Muralidharan, M.; Srinivasan, K.; Mandal, A.K. Characterization of residue-specific glutathionylation of CSF proteins in multiple sclerosis—A MS-based approach. Anal Biochem. 2019, 564-565, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk-Sowa, M.; Galiniak, S.; Żyracka, E.; Grzesik, M.; Naparło, K.; Sowa, P.; Bartosz, G.; Sadowska-Bartosz, I. Oxidative Modification of Blood Serum Proteins in Multiple Sclerosis after Interferon Beta and Melatonin Treatment. Oxid. Med. Cell Longev. 2017, 2017, 7905148. [Google Scholar] [CrossRef] [PubMed]
- Wetzels, S.; Vanmierlo, T.; Scheijen, J.L.J.M.; van Horssen, J.; Amor, S.; Somers, V.; Schalkwijk, C.G.; Hendriks, J.J.A.; Wouters, K. Methylglyoxal-Derived Advanced Glycation Endproducts Accumulate in Multiple Sclerosis Lesions. Front. Immunol. 2019, 10, 855. [Google Scholar] [CrossRef] [PubMed]
- Haghikia, A.; Kayacelebi, A.A.; Beckmann, B.; Hanff, E.; Gold, R.; Haghikia, A.; Tsikas, D. Serum and cerebrospinal fluid concentrations of homoarginine, arginine, asymmetric and symmetric dimethylarginine, nitrite and nitrate in patients with multiple sclerosis and neuromyelitis optica. Amino Acids 2015, 47, 1837–1845. [Google Scholar] [CrossRef]
- Teunissen, C.E.; Sombekke, M.; van Winsen, L.; Killestein, J.; Barkhof, F.; Polman, C.H.; Dijkstra, C.D.; Blankenstein, M.A.; Pratico, D. Increased plasma 8,12-iso-iPF2alpha- VI levels in relapsing multiple sclerosis patients are not predictive of disease progression. Mult. Scler. 2012, 18, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Mrowicka, M.; Saluk-Juszczak, J.; Ireneusz, M. The level of isoprostanes as a non-invasive marker for in vivo lipid peroxidation in secondary progressive multiple sclerosis. Neurochem. Res. 2011, 36, 1012–1016. [Google Scholar] [CrossRef]
- Gonzalo, H.; Brieva, L.; Tatzber, F.; Jové, M.; Cacabelos, D.; Cassanyé, A.; Lanau-Angulo, L.; Boada, J.; Serrano, J.C.; González, C.; et al. Lipidome analysis in multiple sclerosis reveals protein lipoxidative damage as a potential pathogenic mechanism. J. Neurochem. 2012, 123, 622–634. [Google Scholar] [CrossRef]
- Greco, A.; Minghetti, L.; Sette, G.; Fieschi, C.; Levi, G. Cerebrospinal fluid isoprostane shows oxidative stress in patients with multiple sclerosis. Neurology 1999, 53, 1876–1879. [Google Scholar] [CrossRef]
- Pohl, E.E.; Jovanovic, O. The Role of Phosphatidylethanolamine Adducts in Modification of the Activity of Membrane Proteins under Oxidative Stress. Molecules 2019, 24, 4545. [Google Scholar] [CrossRef]
- Mitosek-Szewczyk, K.; Gordon-Krajcer, W.; Walendzik, P.; Stelmasiak, Z. Free radical peroxidation products in cerebrospinal fluid and serum of patients with multiple sclerosis after glucocorticoid therapy. Folia Neuropathol. 2010, 48, 116–122. [Google Scholar] [PubMed]
- Tavazzi, B.; Batocchi, A.P.; Amorini, A.M.; Nociti, V.; D′Urso, S.; Longo, S.; Gullotta, S.; Picardi, M.; Lazzarino, G. Serum Metabolic Profile in Multiple Sclerosis Patients. Mult. Scler. Int. 2011, 2011, 167156. [Google Scholar] [CrossRef] [PubMed]
- Ljubisavljevic, S.; Stojanovic, I.; Vojinovic, S.; Stojanov, D.; Stojanovic, S.; Kocic, G.; Savic, D.; Cvetkovic, T.; Pavlovic, D. Cerebrospinal fluid and plasma oxidative stress biomarkers in different clinical phenotypes of neuroinflammatory acute attacks. Conceptual accession: From fundamental to clinic. Cell Mol. Neurobiol. 2013, 33, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Ghabaee, M.; Jabedari, B.; Al-E-Eshagh, N.; Ghaffarpour, M.; Asadi, F. Serum and cerebrospinal fluid antioxidant activity and lipid peroxidation in Guillain-Barre syndrome and multiple sclerosis patients. Int. J. Neurosci. 2010, 120, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Pawlowski, J.; Shukla, P.; Bielekova, B. Identifying CSF Biomarkers of Oxidative Stress in Patients with Multiple Sclerosis. 2011. Available online: https://www.researchgate.net/publication/290998239_Identifying_CSF_Biomarkers_of_Oxidative_Stress_in_Patients_with_Multiple_Sclerosis (accessed on 28 August 2020).
- Villoslada, P.; Alonso, C.; Agirrezabal, I.; Kotelnikova, E.; Zubizarreta, I.; Pulido-Valdeolivas, I.; Saiz, A.; Comabella, M.; Montalban, X.; Villar, L.; et al. Metabolomic signatures associated with disease severity in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 2017, 4, e321. [Google Scholar] [CrossRef] [PubMed]
- Fellows Maxwell, K.; Bhattacharya, S.; Bodziak, M.L.; Jakimovski, D.; Hagemeier, J.; Browne, R.W.; Weinstock-Guttman, B.; Zivadinov, R.; Ramanathan, M. Oxysterols and apolipoproteins in multiple sclerosis: A 5 year follow-up study. J. Lipid Res. 2019, 60, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.; Balukova, A.; Pospíšil, P. Triplet Excited Carbonyls and Singlet Oxygen Formation during Oxidative Radical Reaction in Skin. Front. Physiol. 2018, 9, 1109. [Google Scholar] [CrossRef]
- Nordzieke, D.E.; Medraño-Fernandez, I. The Plasma Membrane: A Platform for Intra- and Intercellular Redox Signaling. Antioxidants 2018, 7, 168. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef]
- Azadmanesh, J.; Borgstahl, G.E.O. A Review of the Catalytic Mechanism of Human Manganese Superoxide Dismutase. Antioxidants 2018, 7, 25. [Google Scholar] [CrossRef]
- Pospíšil, P.; Prasad, A.; Rác, M. Mechanism of the Formation of Electronically Excited Species by Oxidative Metabolic Processes: Role of Reactive Oxygen Species. Biomolecules 2019, 9, 258. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, N.; Chisci, E.; Giovannoni, R. The Role of Hydrogen Peroxide in Redox-Dependent Signaling: Homeostatic and Pathological Responses in Mammalian Cells. Cells 2018, 7, 156. [Google Scholar] [CrossRef] [PubMed]
- Case, A.J. On the Origin of Superoxide Dismutase: An Evolutionary Perspective of Superoxide-Mediated Redox Signaling. Antioxidants 2017, 6, 82. [Google Scholar] [CrossRef]
- Tang, J.X.; Thompson, K.; Taylor, R.W.; Oláhová, M. Mitochondrial OXPHOS Biogenesis: Co-Regulation of Protein Synthesis, Import, and Assembly Pathways. Int. J. Mol. Sci. 2020, 21, 3820. [Google Scholar] [CrossRef] [PubMed]
- Weidinger, A.; Kozlov, A.V. Biological Activities of Reactive Oxygen and Nitrogen Species: Oxidative Stress versus Signal Transduction. Biomolecules 2015, 5, 472–484. [Google Scholar] [CrossRef]
- Ježek, J.; Cooper, K.F.; Strich, R. Reactive Oxygen Species and Mitochondrial Dynamics: The Yin and Yang of Mitochondrial Dysfunction and Cancer Progression. Antioxidants 2018, 7, 13. [Google Scholar] [CrossRef]
- Klivenyi, P.; Karg, E.; Rozsa, C.; Horvath, R.; Komoly, S.; Nemeth, I.; Turi, S.; Vecsei, L. alpha-Tocopherol/lipid ratio in blood is decreased in patients with Leber’s hereditary optic neuropathy and asymptomatic carriers of the 11,778 mtDNA mutation. J. Neurol. Neurosurg. Psychiatry 2001, 70, 359–362. [Google Scholar] [CrossRef]
- Ahmad, W.; Ijaz, B.; Shabbiri, K.; Ahmed, F.; Rehman, S. Oxidative toxicity in diabetes and Alzheimer′s disease: Mechanisms behind ROS/ RNS generation. J. Biomed. Sci. 2017, 24, 76. [Google Scholar] [CrossRef]
- Ljubisavljevic, S.; Stojanovic, I.; Cvetkovic, T.; Vojinovic, S.; Stojanov, D.; Stojanovic, D.; Stefanovic, N.; Pavlovic, D. Erythrocytes′ antioxidative capacity as a potential marker of oxidative stress intensity in neuroinflammation. J. Neurol. Sci. 2014, 337, 8–13. [Google Scholar] [CrossRef]
- Spinello, A.; Ritacco, I.; Magistrato, A. The Catalytic Mechanism of Steroidogenic Cytochromes P450 from All-Atom Simulations: Entwinement with Membrane Environment, Redox Partners, and Post-Transcriptional Regulation. Catalysts 2019, 9, 81. [Google Scholar] [CrossRef]
- Irazabal, M.V.; Torres, V.E. Reactive Oxygen Species and Redox Signaling in Chronic Kidney Disease. Cells 2020, 9, 1342. [Google Scholar] [CrossRef] [PubMed]
- Onukwufor, J.O.; Berry, B.J.; Wojtovich, A.P. Physiologic Implications of Reactive Oxygen Species Production by Mitochondrial Complex I Reverse Electron Transport. Antioxidants 2019, 8, 285. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, V.; Tuli, H.S.; Varol, A.; Thakral, F.; Yerer, M.B.; Sak, K.; Varol, M.; Jain, A.; Khan, M.A.; Sethi, G. Role of Reactive Oxygen Species in Cancer Progression: Molecular Mechanisms and Recent Advancements. Biomolecules 2019, 9, 735. [Google Scholar] [CrossRef] [PubMed]
- Siendones, E.; Ballesteros, M.; Navas, P. Cellular and Molecular Mechanisms of Recessive Hereditary Methaemoglobinaemia Type II. J. Clin. Med. 2018, 7, 341. [Google Scholar] [CrossRef]
- Lismont, C.; Revenco, I.; Fransen, M. Peroxisomal Hydrogen Peroxide Metabolism and Signaling in Health and Disease. Int. J. Mol. Sci. 2019, 20, 3673. [Google Scholar] [CrossRef]
- Chu, R.; Lin, Y.; Reddy, K.C.; Pan, J.; Rao, M.S.; Reddy, J.K.; Yeldandi, A.V. Transformation of epithelial cells stably transfected with H2O2-generating peroxisomal urate oxidase. Cancer Res. 1996, 56, 4846–4852. [Google Scholar]
- Gray, E.; Rice, C.; Hares, K.; Redondo, J.; Kemp, K.; Williams, M.; Brown, A.; Scolding, N.; Wilkins, A. Reductions in neuronal peroxisomes in multiple sclerosis grey matter. Mult. Scler. 2014, 20, 651–659. [Google Scholar] [CrossRef]
- Lin, K.-J.; Lin, K.-L.; Chen, S.-D.; Liou, C.-W.; Chuang, Y.-C.; Lin, H.-Y.; Lin, T.-K. The Overcrowded Crossroads: Mitochondria, Alpha-Synuclein, and the Endo-Lysosomal System Interaction in Parkinson’s Disease. Int. J. Mol. Sci. 2019, 20, 5312. [Google Scholar] [CrossRef]
- Chobot, V.; Hadacek, F.; Kubicova, L. Effects of Selected Dietary Secondary Metabolites on Reactive Oxygen Species Production Caused by Iron(II) Autoxidation. Molecules 2014, 19, 20023–20033. [Google Scholar] [CrossRef]
- Onyango, A.N. Endogenous Generation of Singlet Oxygen and Ozone in Human and Animal Tissues: Mechanisms, Biological Significance, and Influence of Dietary Components. Oxid. Med. Cell Longev. 2016, 2016, 2398573. [Google Scholar] [CrossRef]
- Adams, L.; Franco, M.C.; Estevez, A.G. Reactive nitrogen species in cellular signaling. Exp. Biol. Med. 2015, 240, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and Clinical Significance of Biomarkers of Oxidative Stress in Humans. Oxid. Med. Cell Longev. 2017, 2017, 6501046. [Google Scholar] [CrossRef] [PubMed]
- Rejdak, K.; Petzold, A.; Stelmasiak, Z.; Giovannoni, G. Cerebrospinal fluid brain specific proteins in relation to nitric oxide metabolites during relapse of multiple sclerosis. Mult. Scler. 2008, 14, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Giovannoni, G.; Miller, D.H.; Losseff, N.A.; Sailer, M.; Lewellyn-Smith, N.; Thompson, A.J.; Thompson, E.J. Serum inflammatory markers and clinical/MRI markers of disease progression in multiple sclerosis. J. Neurol. 2001, 248, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Peltola, J.; Ukkonen, M.; Moilanen, E.; Elovaara, I. Increased nitric oxide products in CSF in primary progressive MS may reflect brain atrophy. Neurology 2001, 57, 895–896. [Google Scholar] [CrossRef]
- Acar, G.; Idiman, F.; Idiman, E.; Kirkali, G.; Cakmakci, H.; Ozakbas, S. Nitric oxide as an activity marker in multiple sclerosis. J. Neurol. 2003, 250, 588–592. [Google Scholar] [CrossRef]
- Danilov, A.I.; Andersson, M.; Bavand, N.; Wiklund, N.P.; Olsson, T.; Brundin, L. Nitric oxide metabolite determinations reveal continuous inflammation in multiple sclerosis. J. Neuroimmunol. 2003, 136, 112–118. [Google Scholar] [CrossRef]
- Svenningsson, A.; Petersson, A.S.; Andersen, O.; Hansson, G.K. Nitric oxide metabolites in CSF of patients with MS are related to clinical disease course. Neurology 1999, 53, 1880–1882. [Google Scholar] [CrossRef]
- Brundin, L.; Morcos, E.; Olsson, T.; Wiklund, N.P.; Andersson, M. Increased intrathecal nitric oxide formation in multiple sclerosis; cerebrospinal fluid nitrite as activity marker. Eur. J. Neurol. 1999, 6, 585–590. [Google Scholar] [CrossRef]
- Xue, Q.; Yan, Y.; Zhang, R.; Xiong, H. Regulation of iNOS on Immune Cells and Its Role in Diseases. Int. J. Mol. Sci. 2018, 19, 3805. [Google Scholar] [CrossRef]
- Fernando, V.; Zheng, X.; Walia, Y.; Sharma, V.; Letson, J.; Furuta, S. S-Nitrosylation: An Emerging Paradigm of Redox Signaling. Antioxidants 2019, 8, 404. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Torres, I.; Manzano-Pech, L.; Rubio-Ruíz, M.E.; Soto, M.E.; Guarner-Lans, V. Nitrosative Stress and Its Association with Cardiometabolic Disorders. Molecules 2020, 25, 2555. [Google Scholar] [CrossRef] [PubMed]
- Bryll, A.; Skrzypek, J.; Krzyściak, W.; Szelągowska, M.; Śmierciak, N.; Kozicz, T.; Popiela, T. Oxidative-Antioxidant Imbalance and Impaired Glucose Metabolism in Schizophrenia. Biomolecules 2020, 10, 384. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, D.; Sun, W.; Wang, T. The Adaptive Mechanism of Plants to Iron Deficiency via Iron Uptake, Transport, and Homeostasis. Int. J. Mol. Sci. 2019, 20, 2424. [Google Scholar] [CrossRef] [PubMed]
- Venditti, P.; Di Meo, S. The Role of Reactive Oxygen Species in the Life Cycle of the Mitochondrion. Int. J. Mol. Sci. 2020, 21, 2173. [Google Scholar] [CrossRef]
- López-Gambero, A.J.; Sanjuan, C.; Serrano-Castro, P.J.; Suárez, J.; Rodríguez de Fonseca, F. The Biomedical Uses of Inositols: A Nutraceutical Approach to Metabolic Dysfunction in Aging and Neurodegenerative Diseases. Biomedicines 2020, 8, 295. [Google Scholar] [CrossRef]
- O’Day, D.H. Calmodulin Binding Proteins and Alzheimer’s Disease: Biomarkers, Regulatory Enzymes and Receptors That Are Regulated by Calmodulin. Int. J. Mol. Sci. 2020, 21, 7344. [Google Scholar] [CrossRef]
- Giles, G.I.; Nasim, M.J.; Ali, W.; Jacob, C. The Reactive Sulfur Species Concept: 15 Years On. Antioxidants 2017, 6, 38. [Google Scholar] [CrossRef]
- Schöneich, C. Thiyl Radical Reactions in the Chemical Degradation of Pharmaceutical Proteins. Molecules 2019, 24, 4357. [Google Scholar] [CrossRef]
- Ramírez, R.E.; García-Martínez, C.; Méndez, F. Understanding the Nucleophilic Character and Stability of the Carbanions and Alkoxides of 1-(9-Anthryl)ethanol and Derivatives. Molecules 2013, 18, 10254–10265. [Google Scholar] [CrossRef]
- Bjørklund, G.; Crisponi, G.; Nurchi, V.M.; Cappai, R.; Buha Djordjevic, A.; Aaseth, J. A Review on Coordination Properties of Thiol-Containing Chelating Agents towards Mercury, Cadmium, and Lead. Molecules 2019, 24, 3247. [Google Scholar] [CrossRef] [PubMed]
- Głowacka, U.; Brzozowski, T.; Magierowski, M. Synergisms, Discrepancies and Interactions between Hydrogen Sulfide and Carbon Monoxide in the Gastrointestinal and Digestive System Physiology, Pathophysiology and Pharmacology. Biomolecules 2020, 10, 445. [Google Scholar] [CrossRef]
- Benchoam, D.; Cuevasanta, E.; Möller, M.N.; Alvarez, B. Hydrogen Sulfide and Persulfides Oxidation by Biologically Relevant Oxidizing Species. Antioxidants 2019, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- McBean, G.J. Cysteine, Glutathione, and Thiol Redox Balance in Astrocytes. Antioxidants 2017, 6, 62. [Google Scholar] [CrossRef] [PubMed]
- Marozkina, N.; Gaston, B. An Update on Thiol Signaling: S-Nitrosothiols, Hydrogen Sulfide and a Putative Role for Thionitrous Acid. Antioxidants 2020, 9, 225. [Google Scholar] [CrossRef]
- McNeil, N.M.R.; McDonnell, C.; Hambrook, M.; Back, T.G. Oxidation of Disulfides to Thiolsulfinates with Hydrogen Peroxide and a Cyclic Seleninate Ester Catalyst. Molecules 2015, 20, 10748–10762. [Google Scholar] [CrossRef]
- Grman, M.; Nasim, M.J.; Leontiev, R.; Misak, A.; Jakusova, V.; Ondrias, K.; Jacob, C. Inorganic Reactive Sulfur-Nitrogen Species: Intricate Release Mechanisms or Cacophony in Yellow, Blue and Red? Antioxidants 2017, 6, 14. [Google Scholar] [CrossRef]
- Nagahara, N.; Wróbel, M. H2S, Polysulfides, and Enzymes: Physiological and Pathological Aspects. Biomolecules 2020, 10, 640. [Google Scholar] [CrossRef]
- Kolluru, G.K.; Shen, X.; Kevil, C.G. Reactive Sulfur Species: A New Redox Player in Cardiovascular Pathophysiology. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 874–884. [Google Scholar] [CrossRef]
- Bild, W.; Ciobica, A.; Padurariu, M.; Bild, V. The interdependence of the reactive species of oxygen, nitrogen, and carbon. J. Physiol. Biochem. 2013, 69, 147–154. [Google Scholar] [CrossRef]
- Monti, D.A.; Zabrecky, G.; Leist, T.P.; Wintering, N.; Bazzan, A.J.; Zhan, T.; Newberg, A.B. N-acetyl Cysteine Administration Is Associated with Increased Cerebral Glucose Metabolism in Patients With Multiple Sclerosis: An Exploratory Study. Front. Neurol. 2020, 11, 88. [Google Scholar] [CrossRef] [PubMed]
- Krysko, K.; Bischof, A.; Nourbakhsh, B.; Henry, R.; Revirajan, N.; Manguinao, M.; Li, Y.; Waubant, E. N-acetyl cysteine for fatigue in progressive multiple sclerosis: A pilot randomized double-blind placebo-controlled trial (P5.2-093). Neurology 2019, 92 (Suppl. 15), P5.2-093. [Google Scholar]
- Singhal, N.K.; Freeman, E.; Arning, E.; Wasek, B.; Clements, R.; Sheppard, C.; Blake, P.; Bottiglieri, T.; McDonough, J. Dysregulation of methionine metabolism in multiple sclerosis. Neurochem. Int. 2018, 112, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Methionine Metabolism Disrupted in MS. Available online: https://www.medpagetoday.com/meetingcoverage/sfn/69274 (accessed on 24 June 2020).
- Roy, D.; Chen, J.; Mamane, V. Methionine Metabolism Shapes T Helper Cell Responses through Regulation of Epigenetic Reprogramming. Cell Metab. 2020, 31, 250–266. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, B.; Mendes, F.; Osório, N.; Caseiro, A.; Gabriel, A.; Valado, A. Glutathione in multiple sclerosis. Br. J. Biomed. Sci. 2013, 70, 75–79. [Google Scholar] [CrossRef]
- Karg, E.; Klivényi, P.; Németh, I.; Bencsik, K.; Pintér, S.; Vécsei, L. Nonenzymatic antioxidants of blood in multiple sclerosis. J. Neurol. 1999, 246, 533–539. [Google Scholar] [CrossRef]
- Fominykh, V.; Onufriev, M.V.; Vorobyeva, A.; Brylev, L.; Yakovlev, A.A.; Zakharova, M.N.; Gulyaeva, N.V. Increased S-nitrosothiols are associated with spinal cord injury in multiple sclerosis. J. Clin. Neurosci. 2016, 28, 38–42. [Google Scholar] [CrossRef]
- Antognelli, C.; Perrelli, A.; Armeni, T.; Nicola Talesa, V.; Retta, S.F. Dicarbonyl Stress and S-Glutathionylation in Cerebrovascular Diseases: A Focus on Cerebral Cavernous Malformations. Antioxidants 2020, 9, 124. [Google Scholar] [CrossRef]
- Hwang, S.W.; Lee, Y.-M.; Aldini, G.; Yeum, K.-J. Targeting Reactive Carbonyl Species with Natural Sequestering Agents. Molecules 2016, 21, 280. [Google Scholar] [CrossRef]
- Pérez-Pérez, A.; Sánchez-Jiménez, F.; Vilariño-García, T.; Sánchez-Margalet, V. Role of Leptin in Inflammation and Vice Versa. Int. J. Mol. Sci. 2020, 21, 5887. [Google Scholar] [CrossRef]
- Khan, A.A.; Alsahli, M.A.; Rahmani, A.H. Myeloperoxidase as an Active Disease Biomarker: Recent Biochemical and Pathological Perspectives. Med. Sci. 2018, 6, 33. [Google Scholar] [CrossRef]
- Mannino, M.H.; Patel, R.S.; Eccardt, A.M.; Janowiak, B.E.; Wood, D.C.; He, F.; Fisher, J.S. Reversible Oxidative Modifications in Myoglobin and Functional Implications. Antioxidants 2020, 9, 549. [Google Scholar] [CrossRef] [PubMed]
- Gonos, E.S.; Kapetanou, M.; Sereikaite, J.; Bartosz, G.; Naparło, K.; Grzesik, M.; Sadowska-Bartosz, I. Origin and pathophysiology of protein carbonylation, nitration and chlorination in age-related brain diseases and aging. Aging (Albany NY) 2018, 10, 868–901. [Google Scholar] [CrossRef] [PubMed]
- Mostert, J.P.; Ramsaransing, G.S.; Heersema, D.J.; Heerings, M.; Wilczak, N.; De Keyser, J. Serum uric acid levels and leukocyte nitric oxide production in multiple sclerosis patients outside relapses. J. Neurol. Sci. 2005, 231, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Minohara, M.; Matsuoka, T.; Li, W.; Osoegawa, M.; Ishizu, T.; Ohyagi, Y.; Kira, J. Upregulation of myeloperoxidase in patients with opticospinal multiple sclerosis: Positive correlation with disease severity. J. Neuroimmunol. 2006, 178, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Zoidis, E.; Seremelis, I.; Kontopoulos, N.; Danezis, G.P. Selenium-Dependent Antioxidant Enzymes: Actions and Properties of Selenoproteins. Antioxidants 2018, 7, 66. [Google Scholar] [CrossRef]
- Socha, K.; Kochanowicz, J.; Karpińska, E.; Soroczyńska, J.; Jakoniuk, M.; Mariak, Z.; Borawska, M.H. Dietary habits and selenium, glutathione peroxidase and total antioxidant status in the serum of patients with relapsing-remitting multiple sclerosis. Nutr. J. 2014, 13, 62. [Google Scholar] [CrossRef]
- Grandi, C.; D’Ovidio, M.C. Balance between Health Risks and Benefits for Outdoor Workers Exposed to Solar Radiation: An Overview on the Role of Near Infrared Radiation Alone and in Combination with Other Solar Spectral Bands. Int. J. Environ. Res. Public Health 2020, 17, 1357. [Google Scholar] [CrossRef]
- Karimi, A.; Bahrampour, K.; Momeni Moghaddam, M.A.; Asadikaram, G.; Ebrahimi, G.; Torkzadeh-Mahani, M.; Esmaeili Tarzi, M.; Nematollahi, M.H. Evaluation of lithium serum level in multiple sclerosis patients: A neuroprotective element. Mult. Scler. Relat. Disord. 2017, 17, 244–248. [Google Scholar] [CrossRef]
- Juybari, K.B.; Ebrahimi, G.; Momeni Moghaddam, M.A.; Asadikaram, G.; Torkzadeh-Mahani, M.; Akbari, M.; Mirzamohammadi, S.; Karimi, A.; Nematollahi, M.H. Evaluation of serum arsenic and its effects on antioxidant alterations in relapsing-remitting multiple sclerosis patients. Mult. Scler. Relat. Disord. 2018, 19, 79–84. [Google Scholar] [CrossRef]
- Carroll, L.D.; Davies, M.J. Reaction of Selenium Compounds with Oxygen Species and the Control of Oxidative Stress. In Organoselenium Compounds in Biology and Medicine: Synthesis, Biological and Therapeutic Treatments; Jain, V.K., Priyadarsini, K.I., Eds.; Royal Society of Chemistry: London, UK, 2018; pp. 254–275. [Google Scholar]
- Xiao, W.; Loscalzo, J. Metabolic Responses to Reductive Stress. Antioxid. Redox Signal 2020, 32, 1330–1347. [Google Scholar] [CrossRef] [PubMed]
- Korge, P.; Calmettes, G.; Weiss, J.N. Increased reactive oxygen species production during reductive stress: The roles of mitochondrial glutathione and thioredoxin reductases. Biochim. Biophys. Acta 2015, 1847, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, P.C. Cytoplasmic and Mitochondrial NADPH-Coupled Redox Systems in the Regulation of Aging. Nutrients 2019, 11, 504. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Torres, I.; Guarner-Lans, V.; Rubio-Ruiz, M.E. Reductive Stress in Inflammation-Associated Diseases and the Pro-Oxidant Effect of Antioxidant Agents. Int. J. Mol. Sci. 2017, 18, 2098. [Google Scholar] [CrossRef] [PubMed]
- Jozefczak, M.; Remans, T.; Vangronsveld, J.; Cuypers, A. Glutathione Is a Key Player in Metal-Induced Oxidative Stress Defenses. Int. J. Mol. Sci. 2012, 13, 3145–3175. [Google Scholar] [CrossRef] [PubMed]
- Singhal, S.S.; Singh, S.P.; Singhal, P.; Horne, D.; Singhal, J.; Awasthi, S. Antioxidant Role of Glutathione S-Transferases: 4-Hydroxynonenal, a Key Molecule in Stress-Mediated Signaling. Toxicol. Appl. Pharmacol. 2015, 289, 361–370. [Google Scholar] [CrossRef]
- Bocedi, A.; Noce, A.; Marrone, G.; Noce, G.; Cattani, G.; Gambardella, G.; Di Lauro, M.; Di Daniele, N.; Ricci, G. Glutathione Transferase P1-1 an Enzyme Useful in Biomedicine and as Biomarker in Clinical Practice and in Environmental Pollution. Nutrients 2019, 11, 1741. [Google Scholar] [CrossRef]
- Shukla, V.K.; Jensen, G.E.; Clausen, J. Erythrocyte glutathione perioxidase deficiency in multiple sclerosis. Acta Neurol. Scand. 1977, 56, 542–550. [Google Scholar] [CrossRef]
- Szeinberg, A.; Golan, R.; Ben Ezzer, J.; Sarova-Pinhas, I.; Sadeh, M.; Braham, J. Decreased erythrocyte glutathione peroxidase activity in multiple sclerosis. Acta Neurol. Scand. 1979, 60, 265–271. [Google Scholar] [CrossRef]
- Szeinberg, A.; Golan, R.; Ben-Ezzer, J.; Sarova-Pinhas, I.; Kindler, D. Glutathione peroxidase activity in various types of blood cells in multiple sclerosis. Acta Neurol. Scand. 1981, 63, 67–75. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Rosales-Corral, S.; Galano, A.; Zhou, X.J.; Xu, B. Mitochondria: Central Organelles for Melatonin′s Antioxidant and Anti-Aging Actions. Molecules 2018, 23, 509. [Google Scholar] [CrossRef] [PubMed]
- Jensen, G.E.; Clausen, J. Glutathione peroxidase and reductase, glucose-6-phosphate dehydrogenase and catalase activities in multiple sclerosis. J. Neurol. Sci. 1984, 63, 45–53. [Google Scholar] [CrossRef]
- Belcastro, E.; Gaucher, C.; Corti, A.; Leroy, P.; Lartaud, I.; Pompella, A. Regulation of Protein Function by S-nitrosation and S-glutathionylation: Processes and Targets in Cardiovascular Pathophysiology. Biol. Chem. 2017, 398, 1267–1293. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudian, E.; Khalilnezhad, A.; Gharagozli, K.; Amani, D. Thioredoxin-1, redox factor-1 and thioredoxin-interacting protein, mRNAs are differentially expressed in Multiple Sclerosis patients exposed and non-exposed to interferon and immunosuppressive treatments. Gene 2017, 634, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Voigt, D.; Scheidt, U.; Derfuss, T.; Brück, W.; Junker, A. Expression of the Antioxidative Enzyme Peroxiredoxin 2 in Multiple Sclerosis Lesions in Relation to Inflammation. Int. J. Mol. Sci. 2017, 18, 760. [Google Scholar] [CrossRef]
- Todisco, S.; Convertini, P.; Iacobazzi, V.; Infantino, V. TCA Cycle Rewiring as Emerging Metabolic Signature of Hepatocellular Carcinoma. Cancers 2020, 12, 68. [Google Scholar] [CrossRef]
- Schaur, R.J.; Siems, W.; Bresgen, N.; Eckl, P.M. 4-Hydroxy-nonenal—A Bioactive Lipid Peroxidation Product. Biomolecules 2015, 5, 2247–2337. [Google Scholar] [CrossRef]
- McArdle, B.; Mackenzie, I.C.; Webster, G.R. STUDIES ON INTERMEDIATE CARBOHYDRATE METABOLISM IN MULTIPLE SCLEROSIS. J. Neurol. Neurosurg. Psychiatry 1960, 23, 127–132. [Google Scholar] [CrossRef]
- Nijland, P.G.; Molenaar, R.J.; van der Pol, S.M.; van der Valk, P.; van Noorden, C.J.; de Vries, H.E.; van Horssen, J. Differential expression of glucose-metabolizing enzymes in multiple sclerosis lesions. Acta Neuropathol. Commun. 2015, 3, 79. [Google Scholar] [CrossRef]
- Cores, Á.; Piquero, M.; Villacampa, M.; León, R.; Menéndez, J.C. NRF2 Regulation Processes as a Source of Potential Drug Targets against Neurodegenerative Diseases. Biomolecules 2020, 10, 904. [Google Scholar] [CrossRef]
- Orrù, C.; Perra, A.; Kowalik, M.A.; Rizzolio, S.; Puliga, E.; Cabras, L.; Giordano, S.; Columbano, A. Distinct Mechanisms Are Responsible for Nrf2-Keap1 Pathway Activation at Different Stages of Rat Hepatocarcinogenesis. Cancers 2020, 12, 2305. [Google Scholar] [CrossRef] [PubMed]
- Lamichane, S.; Dahal Lamichane, B.; Kwon, S.-M. Pivotal Roles of Peroxisome Proliferator-Activated Receptors (PPARs) and Their Signal Cascade for Cellular and Whole-Body Energy Homeostasis. Int. J. Mol. Sci. 2018, 19, 949. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Zhang, Y.; Zhu, S.; Luo, Y.; Xu, P.; Huang, Z. PPAR-Mediated Toxicology and Applied Pharmacology. Cells 2020, 9, 352. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Mendoza, N.; Morales-González, Á.; Madrigal-Santillán, E.O.; Madrigal-Bujaidar, E.; Álvarez-González, I.; García-Melo, L.F.; Anguiano-Robledo, L.; Fregoso-Aguilar, T.; Morales-Gonzalez, J.A. Antioxidant and Adaptative Response Mediated by Nrf2 during Physical Exercise. Antioxidants 2019, 8, 196. [Google Scholar] [CrossRef] [PubMed]
- Witte, M.E.; Nijland, P.G.; Drexhage, J.A.; Gerritsen, W.; Geerts, D.; van Het Hof, B.; Reijerkerk, A.; de Vries, H.E.; van der Valk, P.; van Horssen, J. Reduced expression of PGC-1α partly underlies mitochondrial changes and correlates with neuronal loss in multiple sclerosis cortex. Acta Neuropathol. 2013, 125, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Del Bo’, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic Review on Polyphenol Intake and Health Outcomes: Is there Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern? Nutrients 2019, 11, 1355. [Google Scholar]
- Shenkin, A. Micronutrients in health and disease. Postgrad. Med. J. 2006, 82, 559–567. [Google Scholar] [CrossRef]
- Leri, M.; Scuto, M.; Ontario, M.L.; Calabrese, V.; Calabrese, E.J.; Bucciantini, M.; Stefani, M. Healthy Effects of Plant Polyphenols: Molecular Mechanisms. Int. J. Mol. Sci. 2020, 21, 1250. [Google Scholar] [CrossRef]
- Brunetti, G.; Di Rosa, G.; Scuto, M.; Leri, M.; Stefani, M.; Schmitz-Linneweber, C.; Calabrese, V.; Saul, N. Healthspan Maintenance and Prevention of Parkinson’s-like Phenotypes with Hydroxytyrosol and Oleuropein Aglycone in C. elegans. Int. J. Mol. Sci. 2020, 21, 2588. [Google Scholar] [CrossRef]
- Di Rosa, G.; Brunetti, G.; Scuto, M.; Trovato Salinaro, A.; Calabrese, E.J.; Crea, R.; Schmitz-Linneweber, C.; Calabrese, V.; Saul, N. Healthspan Enhancement by Olive Polyphenols in C. elegans Wild Type and Parkinson’s Models. Int. J. Mol. Sci. 2020, 21, 3893. [Google Scholar] [CrossRef]
- Siracusa, R.; Scuto, M.; Fusco, R.; Trovato, A.; Ontario, M.L.; Crea, R.; Di Paola, R.; Cuzzocrea, S.; Calabrese, V. Anti-inflammatory and Anti-oxidant Activity of Hidrox® in Rotenone-Induced Parkinson’s Disease in Mice. Antioxidants 2020, 9, 824. [Google Scholar] [CrossRef] [PubMed]
- Scuto, M.; Di Mauro, P.; Ontario, M.L.; Amato, C.; Modafferi, S.; Ciavardelli, D.; Trovato Salinaro, A.; Maiolino, L.; Calabrese, V. Nutritional Mushroom Treatment in Meniere’s Disease with Coriolus versicolor: A Rationale for Therapeutic Intervention in Neuroinflammation and Antineurodegeneration. Int. J. Mol. Sci. 2020, 21, 284. [Google Scholar] [CrossRef] [PubMed]
- Miquel, S.; Champ, C.; Day, J.; Aarts, E.; Bahr, B.A.; Bakker, M.; Bánáti, D.; Calabrese, V.; Cederholm, T.; Cryan, J.; et al. Poor cognitive ageing: Vulnerabilities, mechanisms and the impact of nutritional interventions. Ageing Res. Rev. 2018, 42, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Preconditioning is hormesis part II: How the conditioning dose mediates protection: Dose optimization within temporal and mechanistic frameworks. Pharmacol. Res. 2016, 110, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Koola, M.M. Galantamine-Memantine combination in the treatment of Alzheimer’s disease and beyond. Psychiatry Res. 2020, 293, 113409. [Google Scholar] [CrossRef] [PubMed]
- Koola, M.M.; Jafarnejad, S.; Looney, S.; Praharaj, S.; Pillai, A.; Ahmed, A.; Slifstein, M. Meta-Analyses of Randomized Controlled Trials and Potential Novel Combination Treatments in Schizophrenia. Biol. Psychiatry 2020, 87, S306. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Zhang, J. Curcumin in antidepressant treatments: An overview of potential mechanisms, pre-clinical/clinical trials and ongoing challenges. Basic Clin. Pharmacol. Toxicol. 2020, 127, 243–253. [Google Scholar] [CrossRef]
- Török, N.; Maszlag-Török, R.; Molnár, K.; Szolnoki, Z.; Somogyvári, F.; Boda, K.; Tanaka, M.; Klivényi, P.; Vécsei, L. Single Nucleotide Polymorphisms of Indoleamine 2,3-Dioxygenase 1 Influenced the Age Onset of Parkinson’s Disease. Preprints 2020, 2020090470. [Google Scholar] [CrossRef]
- Carrillo-Mora, P.; Pérez-De la Cruz, V.; Estrada-Cortés, B.; Toussaint-González, P.; Martínez-Cortéz, J.A.; Rodríguez-Barragán, M.; Quinzaños-Fresnedo, J.; Rangel-Caballero, F.; Gamboa-Coria, G.; Sánchez-Vázquez, I.; et al. Serum Kynurenines Correlate With Depressive Symptoms and Disability in Poststroke Patients: A Cross-sectional Study. Neurorehabil. Neural Repair 2020, 154596832095367. [Google Scholar] [CrossRef]
- Tanaka, M.; Bohár, Z.; Martos, D.; Telegdy, G.; Vécsei, L. Antidepressant-like effects of kynurenic acid in a modified forced swim test. Pharmacol. Rep. 2020, 72, 449–455. [Google Scholar] [CrossRef]
- Peris, E.; Micallef, P.; Paul, A.; Palsdottir, V.; Enejder, A.; Bauzá-Thorbrügge, M.; Olofsson, C.S.; Asterholm, W.I. Antioxidant treatment induces reductive stress associated with mitochondrial dysfunction in adipocyte. J. Biol. Chem. 2019, 294, 2340–2352. [Google Scholar] [CrossRef] [PubMed]
- Becerril, S.; Rodríguez, A.; Catalán, V.; Ramírez, B.; Unamuno, X.; Portincasa, P.; Gómez-Ambrosi, J.; Frühbeck, G. Functional Relationship between Leptin and Nitric Oxide in Metabolism. Nutrients 2019, 11, 2129. [Google Scholar] [CrossRef] [PubMed]
- Feige, J.; Moser, T.; Bieler, L.; Schwenker, K.; Hauer, L.; Sellner, J. Vitamin D Supplementation in Multiple Sclerosis: A Critical Analysis of Potentials and Threats. Nutrients 2020, 12, 783. [Google Scholar] [CrossRef] [PubMed]
- Morel, A.; Bijak, M.; Niwald, M.; Miller, E.; Saluk, J. Markers of oxidative/nitrative damage of plasma proteins correlated with EDSS and BDI scores in patients with secondary progressive multiple sclerosis. Redox Rep. 2017, 22, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Rommer, P.S.; Greilberger, J.; Salhofer-Polanyi, S.; Auff, E.; Leutmezer, F.; Herwig, R. Elevated levels of carbonyl proteins in cerebrospinal fluid of patients with neurodegenerative diseases. Tohoku J. Exp. Med. 2014, 234, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Irani, D.N. Cerebrospinal fluid protein carbonylation identifies oxidative damage in autoimmune demyelination. Ann. Clin. Transl. Neurol. 2016, 4, 145–150. [Google Scholar] [CrossRef]
- Poerschke, R.L.; Fritz, K.S.; Franklin, C.C. Methods to detect protein glutathionylation. Curr. Protoc. Toxicol. 2013, 57, 6.17.1–6.17.18. [Google Scholar] [CrossRef]
- Garibaldi, S.; Barisione, C.; Marengo, B.; Ameri, P.; Brunelli, C.; Balbi, M.; Ghigliotti, G. Advanced Oxidation Protein Products-Modified Albumin Induces Differentiation of RAW264.7 Macrophages into Dendritic-Like Cells Which Is Modulated by Cell Surface Thiols. Toxins 2017, 9, 27. [Google Scholar] [CrossRef]
- Hányšová, S.; Čierny, D.; Petráš, M.; Lehotský, J. Elevated plasma levels of advanced oxidation protein products in Slovak multiple sclerosis patients: Possible association with different disability states. Act Nerv. Super Rediviva. 2017, 59, 45–50. [Google Scholar]
- Gill, V.; Kumar, V.; Singh, K.; Kumar, A.; Kim, J.-J. Advanced Glycation End Products (AGEs) May Be a Striking Link Between Modern Diet and Health. Biomolecules 2019, 9, 888. [Google Scholar] [CrossRef]
- Tain, Y.; Hsu, C. Toxic Dimethylarginines: Asymmetric Dimethylarginine (ADMA) and Symmetric Dimethylarginine (SDMA). Toxins 2017, 9, 92. [Google Scholar] [CrossRef] [PubMed]
- Zarkovic, N. Antioxidants and Second Messengers of Free Radicals. Antioxidants 2018, 7, 158. [Google Scholar] [CrossRef]
- Ito, F.; Sono, Y.; Ito, T. Measurement and Clinical Significance of Lipid Peroxidation as a Biomarker of Oxidative Stress: Oxidative Stress in Diabetes, Atherosclerosis, and Chronic Inflammation. Antioxidants 2019, 8, 72. [Google Scholar] [CrossRef] [PubMed]
- Håkansson, I.; Gouveia-Figueira, S.; Ernerudh, J.; Vrethem, M.; Ghafouri, N.; Ghafouri, B.; Nording, M. Oxylipins in cerebrospinal fluid in clinically isolated syndrome and relapsing remitting multiple sclerosis. Prostaglandins Other Lipid Mediat. 2018, 138, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Palavra, F.; Marado, D.; Mascarenhas-Melo, F.; Sereno, J.; Teixeira-Lemos, E.; Nunes, C.C.; Gonçalves, G.; Teixeira, F.; Reis, F. New markers of early cardiovascular risk in multiple sclerosis patients: Oxidized-LDL correlates with clinical staging. Dis. Mark. 2013, 34, 341–348. [Google Scholar] [CrossRef]
- Besler, H.T.; Comoğlu, S. Lipoprotein oxidation, plasma total antioxidant capacity and homocysteine level in patients with multiple sclerosis. Nutr. Neurosci. 2003, 6, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Salemi, G.; Gueli, M.C.; Vitale, F.; Battaglieri, F.; Guglielmini, E.; Ragonese, P.; Trentacosti, A.; Massenti, M.F.; Savettieri, G.; Bono, A. Blood lipids, homocysteine, stress factors, and vitamins in clinically stable multiple sclerosis patients. Lipids Health Dis. 2010, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Meyers, L.; Groover, C.J.; Douglas, J.; Lee, S.; Brand, D.; Levin, M.C.; Gardner, L.A. A role for Apolipoprotein A-I in the pathogenesis of multiple sclerosis. J. Neuroimmunol. 2014, 277, 176–185. [Google Scholar] [CrossRef] [PubMed]
- van de Kraats, C.; Killestein, J.; Popescu, V.; Rijkers, E.; Vrenken, H.; Lütjohann, D.; Barkhof, F.; Polman, C.H.; Teunissen, C.E. Oxysterols and cholesterol precursors correlate to magnetic resonance imaging measures of neurodegeneration in multiple sclerosis. Mult. Scler. 2014, 20, 412–417. [Google Scholar] [CrossRef]
- Graille, M.; Wild, P.; Sauvain, J.-J.; Hemmendinger, M.; Guseva Canu, I.; Hopf, N.B. Urinary 8-OHdG as a Biomarker for Oxidative Stress: A Systematic Literature Review and Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 3743. [Google Scholar] [CrossRef]
- Ibitoye, R.; Kemp, K.C.; Rice, C.M.; Hares, K.M.; Scolding, N.J.; Wilkins, A. Oxidative stress-related biomarkers in multiple sclerosis: A review. Biomark. Med. 2016, 10, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, Y.-H.; Wang, H. Exploring MicroRNA Biomarkers for Parkinson’s Disease from mRNA Expression Profiles. Cells 2018, 7, 245. [Google Scholar] [CrossRef] [PubMed]
- Brito, L.M.; Ribeiro-dos-Santos, Â.; Vidal, A.F.; de Araújo, G.S.; on behalf of the Alzheimer’s Disease Neuroimaging Initiative. Differential Expression and miRNA–Gene Interactions in Early and Late Mild Cognitive Impairment. Biology 2020, 9, 251. [Google Scholar] [CrossRef]
- Catanesi, M.; d’Angelo, M.; Tupone, M.G.; Benedetti, E.; Giordano, A.; Castelli, V.; Cimini, A. MicroRNAs Dysregulation and Mitochondrial Dysfunction in Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 5986. [Google Scholar] [CrossRef]
- Mohammed, E.M.A. Environmental Influencers, MicroRNA, and Multiple Sclerosis. J. Cent. Nerv. Syst. Dis. 2020, 12, 1179573519894955. [Google Scholar] [CrossRef] [PubMed]
- Martinez, B.; Peplow, P.V. MicroRNAs in blood and cerebrospinal fluid as diagnostic biomarkers of multiple sclerosis and to monitor disease progression. Neural. Regen. Res. 2020, 15, 606–619. [Google Scholar]
- Pfaff, A.L.; Bubb, V.J.; Quinn, J.P.; Koks, S. An increased burden of highly active 3 retrotransposition competent L1s is associated with 4 Parkinson’s disease risk and progression in the PPMI 5 cohort. Int. J. Mol. Sci. 2020, 21, 6562. [Google Scholar] [CrossRef]
- Geis, F.K.; Goff, S.P. Silencing and Transcriptional Regulation of Endogenous Retroviruses: An Overview. Viruses 2020, 12, 884. [Google Scholar] [CrossRef]
- Török, N.; Molnár, K.; Füvesi, J.; Karácsony, M.; Zsiros, V.; Fejes-Szabó, A.; Fiatal, S.; Ádány, R.; Somogyvári, F.; Stojiljković, O.; et al. Chemokine receptor V Δ32 deletion in multiple sclerosis patients in Csongrád County in Hungary and the North-Bácska region in Serbia. Hum Immunol. 2015, 76, 59–64. [Google Scholar] [CrossRef]
- Hall, A.; Bandres-Ciga, S.; Diez-Fairen, M.; Billingsley, K.J. Genetic risk profiling in Parkinson’s disease and utilizing genetics to gain insight into disease-related biological pathways. Int. J. Mol. Sci. 2020, 19, 7332. [Google Scholar] [CrossRef]
- Dhama, K.; Latheef, S.K.; Dadar, M.; Samad, H.A.; Munjal, A.; Khandia, R.; Karthik, K.; Tiwari, R.; Yatoo, M.I.; Bhatt, P.; et al. Biomarkers in Stress Related Diseases/Disorders: Diagnostic, Prognostic, and Therapeutic Values. Front. Mol. Biosci. 2019, 6, 91. [Google Scholar] [CrossRef] [PubMed]
 : oxidative biomarkers;
: oxidative biomarkers;  : degradation biomarkers;
: degradation biomarkers;  : antioxidative biomarkers; ↑: increase; ↓: decrease; and 0: unchanged.
: antioxidative biomarkers; ↑: increase; ↓: decrease; and 0: unchanged.
 : oxidative biomarkers;
: oxidative biomarkers;  : degradation biomarkers;
: degradation biomarkers;  : antioxidative biomarkers; ↑: increase; ↓: decrease; and 0: unchanged.
: antioxidative biomarkers; ↑: increase; ↓: decrease; and 0: unchanged.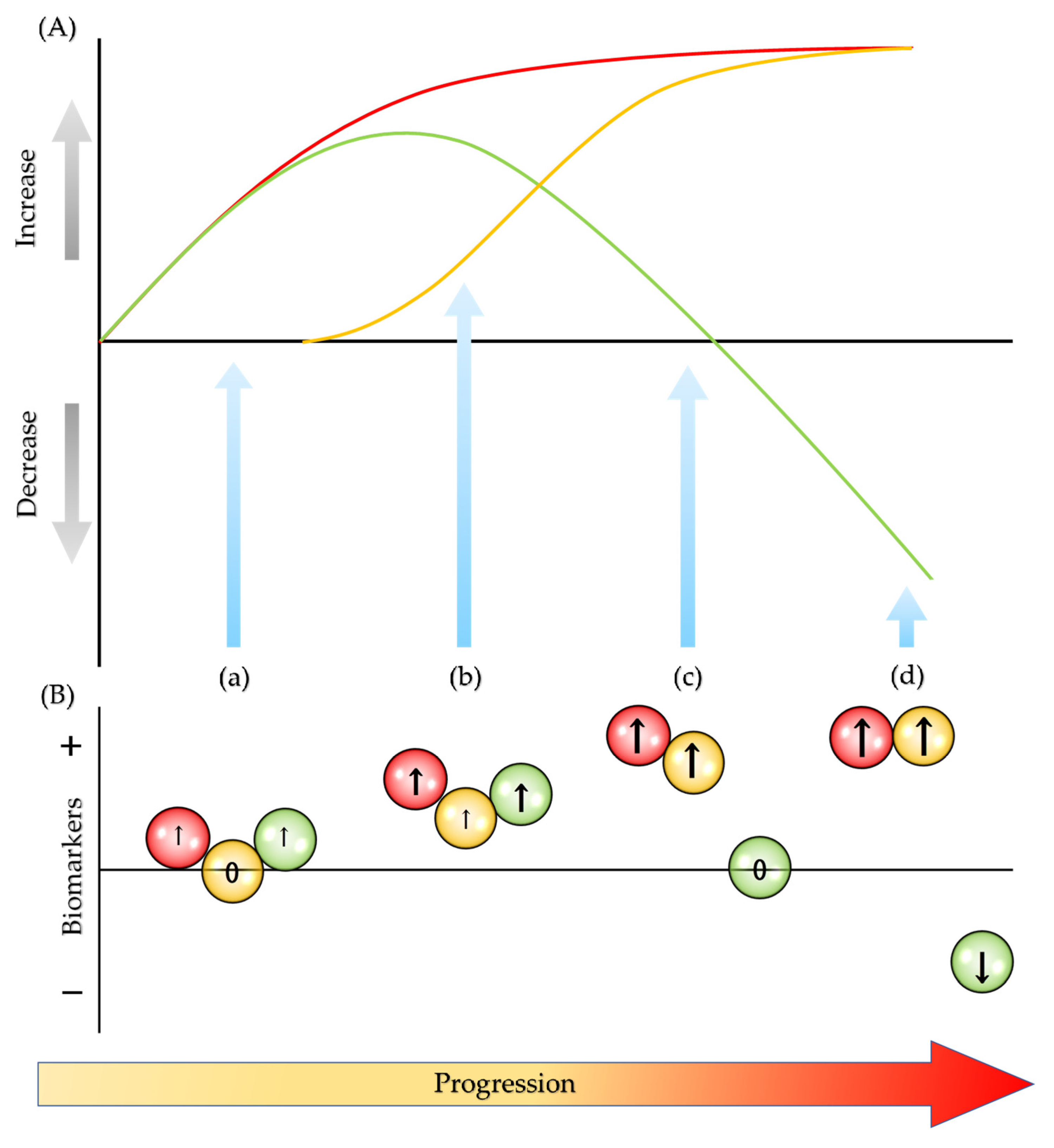
| Class | Drugs | Indications |
|---|---|---|
| Immunomodulators | Interferon beta (IFN-β) | CIS Active RRMS |
| Methylprednisolone | Acute flare-ups RRMS Relapse prevention | |
| Glatiramer acetate (GA) | CIS Active RRMS | |
| Dimethyl fumarate | Active RRMS | |
| Diroximel fumarate | CIS RRMS Active SPMS | |
| Fingolimod | Active RRMS High-active RRMS | |
| Teriflunomide | Active RRMS | |
| Siponimod | CIS RRMS SPMS | |
| Immunosuppressors | Alemtuzumab | Active RRMS |
| Natalizumab | High-active RRMS | |
| Ocrelizumab | Active RRMS High-active RRMS PPMS | |
| Ofatumumab | CIS RRMS Active SPMS | |
| Cytotoxic Agents | Cladribine | High-active RRMS |
| Mitoxantrone | High-active RRMS |
| Reactive Chemical Species | |
|---|---|
| Reactive Oxygen Species (ROS) | Free Radicals |
| Superoxide anion (O2−•), Hydroxyl radical (OH•), Alkoxyl radical (RO•), Peroxyl radical (ROO•) | |
| Nonradicals | |
| Hydrogen peroxide (H2O2), Organic hydroperoxide (ROOH), Organic peroxide (ROOR), Singlet oxygen (O21Δg), Ozone (O3) | |
| Reactive Nitrogen Species (RNS) | Free Radicals |
| Nitric oxide radical (NO•), Nitrogen dioxide radical (NO2•) | |
| Nonradicals | |
| Nitrite (NO2−), Nitrate (NO3−), Nitroxyl anion (NO−), Nitrosyl cation (NO+), Peroxynitrite (ONOO−), Peroxynitrate (O2NOO−), Nitrosoperoxycarbonate (ONOOCO2−), Dinitrogen trioxide (N2O3), Dinitrogen tetraoxide (N2O4), Nitryl chloride (NClO2) | |
| Reactive Sulfur Species (RSN) | Free Radicals |
| Thiyl radical (RS•), Peroxysulphenyl radical (RSOO•) | |
| Nonradicals | |
| Hydrogen sulfide (H2S), Thiolate anion (RS−), Thiol (RSH), Hydropersulfide (RSSH), Disulfide (RSSR), Hydropolysulfide (RSSnH), Dialkyl polysulfide (RSSnR), Polysulfide (H2Sx), Sulfenate (RSO−), Sulfinate (RSO2−), Sulfonate (RSO3−), Thiosulmonate (S2O32−), Sulfite (RS2O32−), Sulfate (SO42 −), Thiosulfinate (C6H10OS2), S-nitrosothiols (RSNOs), Nitrosopersulfide (SSNO−), Dinitrosylated sulfite adduct (SULFI/NO) | |
| Reactive Carbonyl Species (RCS) | Nonradicals |
| Acetaldehyde (CH3CHO), Acrolein (Proponel+: C3H4O), Methylglyoxal 4-Hydroxy-nonenal (C9H16O2), 3-Deoxyglucosone (C6H10O5), Glyoxal (C2H2O2), Methylgyoxal (C3H4O2), Electronically excited triplet carbonyls (3L=O*) | |
| Reactive Halogen Species (RHS) | Free Radicals |
| Atomic chlorine (Cl•), Atomic bromine (Br•) | |
| Nonradicals | |
| Hypochlorite (OCl−), Chloramines (RNHCl), Hypobromite (OBr−), Hypoiodite (IO−), Hypohalogenite (XO−; X = F, Cl, Br, or I) | |
| Reactive Selenium Species (RSeS) | Nonradicals |
| Selenite (O3Se−2), Selenate (SeO42−), Selenocysteine (C3H7NO2Se), Selenomethionine (C5H11NO2Se) | |
| Reactive Nucleophilic Species | Free Radicals |
| Superoxide anion (O2−•) | |
| Nonradicals | |
| Hydrogen sulfide (H2S), Thiolate (RS−), Hydropersulfide (RSS−), Disulfide (RSSR), Selenite (O3Se−2), Selenate (SeO42−), Selenocysteine (C3H7NO2Se), Selenomethionine (C5H11NO2Se) | |
| Classes | Types | Human Samples | Reference | ||
|---|---|---|---|---|---|
| Blood | CSF | ||||
| Reactive Species | Reactive Nitrogen Species | ↑ | ↑ | [47] | |
| Oxidative Enzymes | Xanthine Dehydrogenase (XDH) | ↓ | - | [46] | |
| Nicotinamide Adenine Dinucleotide Phosphate (NADPH) Oxidase | mixed | - | [48] | ||
| Superoxide Dismutase (SOD) | ↑ | ↓ | [49,50,51,52] | ||
| Inducible Nitric Oxide Synthase (iNOS) | ↑ | ↑ | [18,53,54,55] | ||
| Myeloperoxidase (MPO) | mixed | - | [48] | ||
| Antioxidative Enzymes and Transcriptional Factors | Glutathione Peroxidase (GPx) | ↑(relapse) ↓(remission) | ↓ | [56,57,58,59,60,61] | |
| Glutathione Reductase (GSR) | - | ↑ | [61,62] | ||
| Catalase | ↓ | - | [61,62] | ||
| Xanthine oxidase (XO)-Uric Acid | ↓ | - | [46] | ||
| Nuclear Factor Erythroid 2-Related Factor (Nrf2) | ↑ | - | [52] | ||
| Peroxisome proliferator-activated receptors (PPARs) | - | ↑ | [63] | ||
| Peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) | ↓ | - | [64] | ||
| Degradation Products and End Products | Protein | Protein carbonyls | ↑ | - | [65,66,67,68,69,70,71] |
| 3-nitrotyrosin (3-NO-Tyr) | ↑ | - | [68,69,70,72] | ||
| Protein glutathionylation | - | ↑ | [73] | ||
| Dityrosine | ↑ | - | [74] | ||
| Advanced oxidation protein products (AOPPs) | ↑ | - | [49,74,75] | ||
| Advanced glycation end products (AGEs) | ↑ | ↑ | [74,75] | ||
| Amino acids | Asymmetric dimethylarginine (ADMA) | ↓ | - | [76] | |
| Lipid | F2-isoprostane (F2-isoP) | ↑ | ↑ | [77,78,79,80,81] | |
| Malondialdehyde (MDA) | mixed | ↑ | [61,82,83,84,85] | ||
| 4-hydroxynonenal (4-HNE) | - | ↑ | [86] | ||
| Hydroxyoctadecadienoic acid (HODE) | ↑ | ↑ | [87] | ||
| Oxysterol | mixed | ↑ | [88] | ||
| DNA | 8-dihydro-2′deoxyguanosine (8-oxodG) | ↑ | - | [56] | |
| Class | Components | Biomarkers | |||
|---|---|---|---|---|---|
| Diagnostic | Prognostic | Predictive | Therapeutic | ||
| Reactive Chemical Species | Total nitrite (NO2−)/nitrite (NO3−) value (tNOx) | PPMS, RRMS, Relapse, SPMS | RRMS | - | - |
| S-nitrosothiol | RRMS, SPMS | Spinal injury | - | - | |
| Oxidative Enzymes | Superoxide dismutase (SOD) | CIS, RRMS | CIS, RRMS | RRMS | RRMS |
| Myeloperoxidase (MPO) | RRMS | RRMS | - | - | |
| Inducible nitric oxide synthase (iNOS) | RRMS | - | - | - | |
| Antioxidants and Antioxidative Enzymes | Xanthine oxidase (XO)-Uric acid | PPMM, RRMM, SPMM | - | - | - |
| Selenium | RRMS | - | - | - | |
| Glutathione reductase (GSR) | mixedMM | MixedMM | - | - | |
| Catalase | CIS, RRMS | RRMS | - | - | |
| Thioredoxin-Peroxiredoxin (TRX-PRDX) | MS | - | - | - | |
| Nuclear factor erythroid 2-related factor (Nrf2) | RRMS | - | RRMS | - | |
| Peroxisome proliferator-activated receptors (PPARs) | RRMS | - | - | - | |
| Peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) | PPMS, SPMS | - | - | - | |
| Degradation Products and End Products | Protein carbonyls | RRMS, SPMS | RRMS, SPMS | RRMS | - |
| 3-nitrotyrosine (3-NO-Tyr) | RRMS, SPMS | - | RRMS | - | |
| Glutathionylation | Acute attack | - | - | - | |
| Dityrosine | RRSM | - | - | - | |
| Advanced oxidation protein products (AOPPs) | CIS, RRMS | RRMS | RRMS | - | |
| Advanced glycation end products (AGEs) | RRMS | - | - | - | |
| Asymmetric dimethylarginine (ADMA) | RRMS, SPMS | - | - | - | |
| F2-isoprostane (F2-isoP) | RRMS, SPMS | SPMS | - | - | |
| Malondialdehyde (MDA) | RRMS | RRMS | RRMS | - | |
| 4-hydroxynonenal (4-HNE) | PPMS, RRMS, SPMS | - | - | - | |
| Hydroxyoctadecadienoic acid (HODE) | CIS, RRMS | - | - | - | |
| Oxocholesterols | MixedMS | SPMS | - | - | |
| Oxidized low-density lipoprotein (oxLDL) | RRMS, SPMS | - | - | - | |
| 8-OH2dG | RRMS | - | - | - | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanaka, M.; Vécsei, L. Monitoring the Redox Status in Multiple Sclerosis. Biomedicines 2020, 8, 406. https://doi.org/10.3390/biomedicines8100406
Tanaka M, Vécsei L. Monitoring the Redox Status in Multiple Sclerosis. Biomedicines. 2020; 8(10):406. https://doi.org/10.3390/biomedicines8100406
Chicago/Turabian StyleTanaka, Masaru, and László Vécsei. 2020. "Monitoring the Redox Status in Multiple Sclerosis" Biomedicines 8, no. 10: 406. https://doi.org/10.3390/biomedicines8100406
APA StyleTanaka, M., & Vécsei, L. (2020). Monitoring the Redox Status in Multiple Sclerosis. Biomedicines, 8(10), 406. https://doi.org/10.3390/biomedicines8100406






