Beneficial Biological Activities of Cinnamomum osmophloeum and Its Potential Use in the Alleviation of Oral Mucositis: A Systematic Review
Abstract
1. Introduction
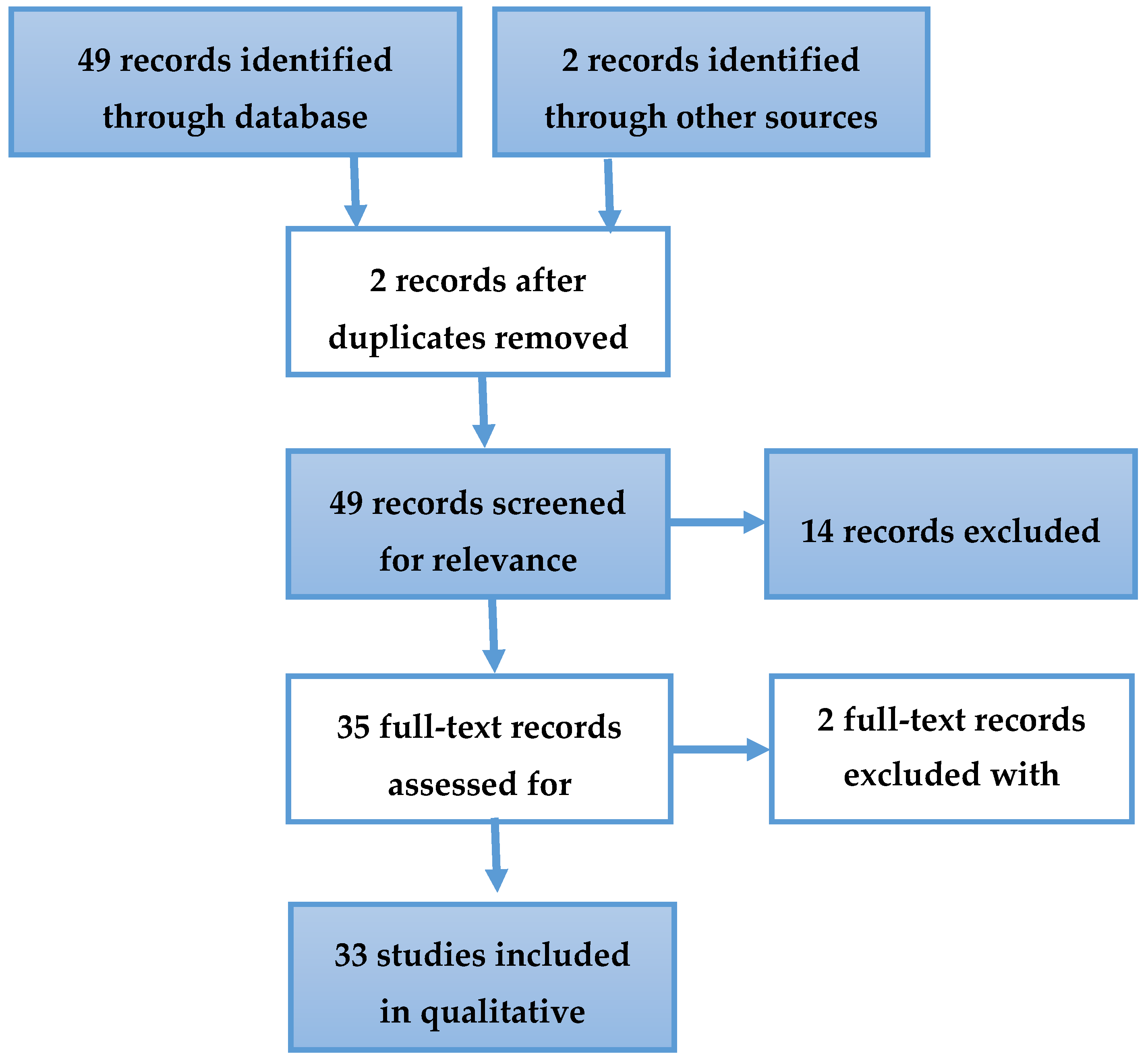
2. Materials and Methods
3. Results and Discussion
3.1. Medical-Biological Activities of C. osmophloeum
3.1.1. Anti-Inflammatory and Wound Repair Activities
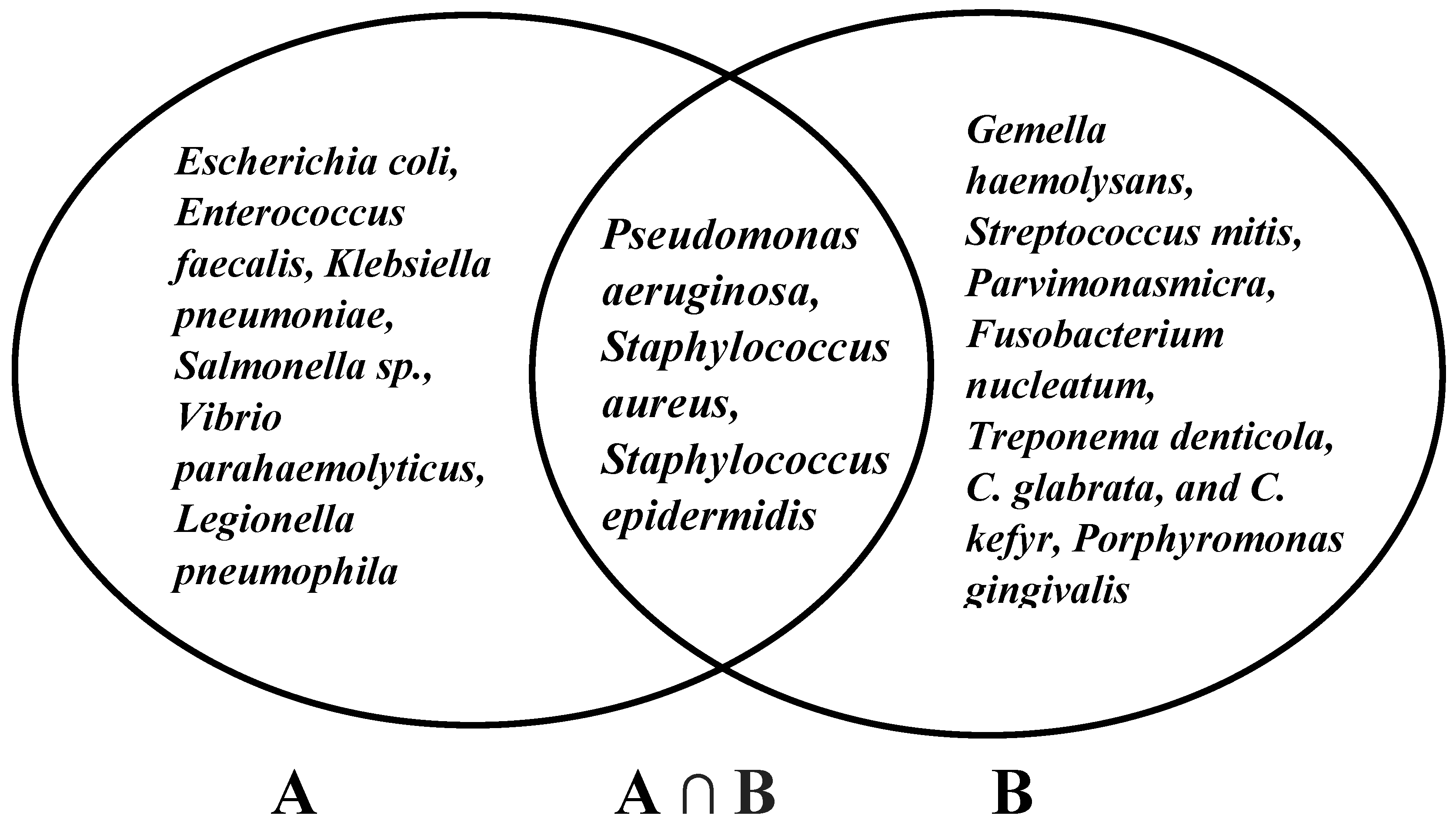
3.1.2. Antimicrobial Activities
3.1.3. Antioxidant Activities
3.1.4. Antidyslipidemic Activities
3.1.5. Anti-Hyperglycemic Activities
3.1.6. Effects on the Cardiovascular System
3.1.7. Effect on Renal Disease and Anti-Hyperuricemia
3.1.8. Anti-Tumor and Anti-Cancer Activity
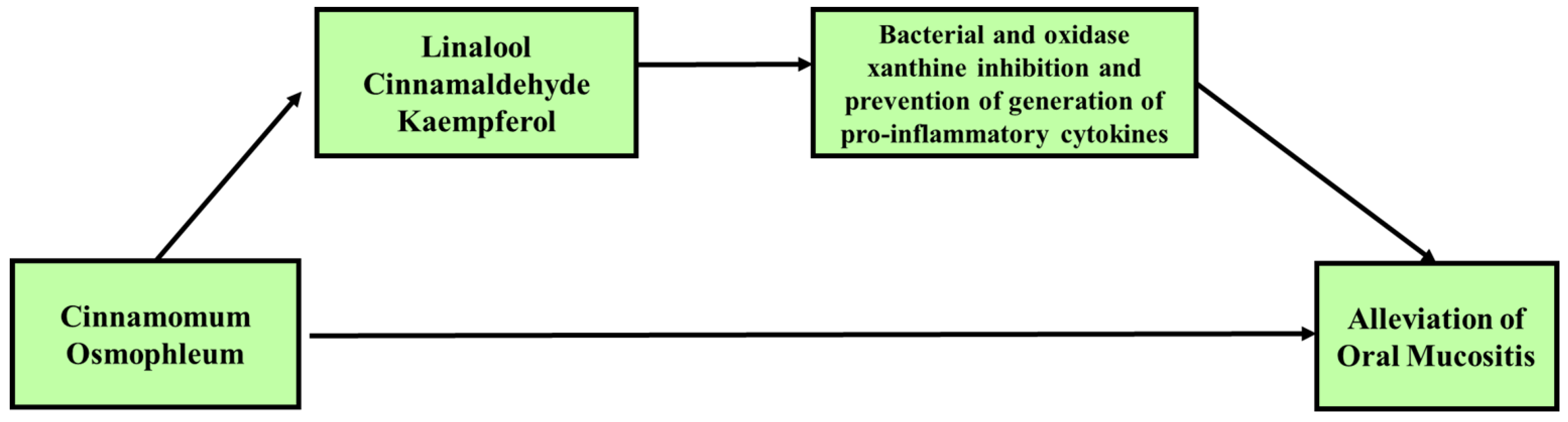
3.2. Potential Use of C. osmophloeum for the Treatment of Oral Mucositis (OM)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ribeiro-Santos, R.; Andrade, M.; Madella, D.; Martinazzo, A.P.; de Aquino Garcia Moura, L.; de Melo, N.R.; Sanches-Silva, A. Revisiting an ancient spice with medicinal purposes: Cinnamon. Trends Food Sci. Technol. 2017, 62, 154–169. [Google Scholar] [CrossRef]
- Wang, S.Y.; Yang, C.W.; Liao, J.W.; Zhen, W.W.; Chu, F.H.; Chang, S.T. Essential oil from leaves of Cinnamomum osmophloeum acts as a xanthine oxidase inhibitor and reduces the serum uric acid levels in oxonate-induced mice. Phytomedicine 2008, 15, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-C.; Xu, W.-X.; Lin, L.-Y.; Yang, J.-J.; Liu, C.-T. Chemical Composition and Hypoglycemic and Pancreas-Protective Effect of Leaf Essential Oil from Indigenous Cinnamon (Cinnamomum osmophloeum Kanehira). J. Agric. Food Chem. 2013, 61, 4905–4913. [Google Scholar] [CrossRef]
- Chao, L.K.; Chang, W.T.; Shih, Y.W.; Huang, J.S. Cinnamaldehyde impairs high glucose-induced hypertrophy in renal interstitial fibroblasts. Toxicol Appl. Pharm. 2010, 244, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.-S.; Lee, Y.-H.; Chuang, L.-Y.; Guh, J.-Y.; Hwang, J.-Y. Cinnamaldehyde and Nitric Oxide Attenuate Advanced Glycation End Products-Induced the JAK/STAT Signaling in Human Renal Tubular Cells. J. Cell. Biochem. 2015, 116, 1028–1038. [Google Scholar] [CrossRef]
- Tung, Y.-T.; Huang, C.-C.; Ho, S.-T.; Kuo, Y.-H.; Lin, C.-C.; Lin, C.-T.; Wu, J.-H. Bioactive Phytochemicals of Leaf Essential Oils of Cinnamomum osmophloeum Prevent Lipopolysaccharide/d-Galactosamine (LPS/d-GalN)-Induced Acute Hepatitis in Mice. J. Agric. Food Chem. 2011, 59, 8117–8123. [Google Scholar] [CrossRef]
- Yang, L.; Wu, Q.-Q.; Liu, Y.; Hu, Z.-F.; Bian, Z.-Y.; Tang, Q.-Z. Cinnamaldehyde attenuates pressure overload-induced cardiac hypertrophy. Int. J. Clin. Exp. Pathol. 2015, 8, 14345–14354. [Google Scholar]
- Raber-Durlacher, J.E.; Elad, S.; Barasch, A. Oral mucositis. Oral Oncol. 2010, 46, 452–456. [Google Scholar] [CrossRef]
- Elting, L.S.; Cooksley, C.D.; Chambers, M.S.; Garden, A.S. Risk, Outcomes, and Costs of Radiation-Induced Oral Mucositis Among Patients With Head-and-Neck Malignancies. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 1110–1120. [Google Scholar] [CrossRef]
- Elting, L.S.; Keefe, D.M.; Sonis, S.T.; Garden, A.S.; Spijkervet, F.K.L.; Barasch, A.; Tishler, R.B.; Canty, T.P.; Kudrimoti, M.K.; Vera-Llonch, M.; et al. Patient-reported measurements of oral mucositis in head and neck cancer patients treated with radiotherapy with or without chemotherapy. Cancer 2008, 113, 2704–2713. [Google Scholar] [CrossRef]
- Murphy, B.A.; Beaumont, J.L.; Isitt, J.; Garden, A.S.; Gwede, C.K.; Trotti, A.M.; Meredith, R.F.; Epstein, J.B.; Le, Q.-T.; Brizel, D.M.; et al. Mucositis-Related Morbidity and Resource Utilization in Head and Neck Cancer Patients Receiving Radiation Therapy With or Without Chemotherapy. J. Pain Symptom Manag. 2009, 38, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Lalla, R.V.; Sonis, S.T.; Peterson, D.E. Management of oral mucositis in patients who have cancer. Dent. Clin. North. Am. 2008, 52, 61-viii. [Google Scholar] [CrossRef] [PubMed]
- Sonis, S.; Treister, N. Oral mucositis. In Oral complications of cancer and its management, 1st ed.; Davies, A., Epstein, J., Eds.; Oxford Universirty Press: Oxford, UK; New York, NY, USA, 2010; Volume 1, pp. 141–148. [Google Scholar]
- Sonis, S.T. Pathobiology of oral mucositis: Novel insights and opportunities. J. Supportive Oncol. 2007, 5, 3–11. [Google Scholar]
- Sonis, S.T. New thoughts on the initiation of mucositis. Oral Dis. 2010, 16, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Sonis, S.T. The pathobiology of mucositis. Nat. Rev. Cancer 2004, 4, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Baharvand, M.; Jafari, S.; Mortazavi, H. Herbs in Oral Mucositis. J. Clin. Diagn Res. 2017, 11, ZE05–ZE11. [Google Scholar] [CrossRef]
- Lin, G.M.; Chen, Y.H.; Yen, P.L.; Chang, S.T. Antihyperglycemic and antioxidant activities of twig extract from Cinnamomum osmophloeum. J. Tradit Complement. Med. 2016, 6, 281–288. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Yeh, T.-F.; Hsu, F.-L.; Lin, C.-Y.; Chang, S.-T.; Chang, H.-T. Xanthine Oxidase Inhibitory Activity and Thermostability of Cinnamaldehyde-Chemotype Leaf Oil of Cinnamomum osmophloeum Microencapsulated with β-Cyclodextrin. Molecules 2018, 23, 1107. [Google Scholar] [CrossRef]
- Lee, M.-G.; Kuo, S.-Y.; Yen, S.-Y.; Hsu, H.-F.; Leung, C.-H.; Ma, D.-L.; Wen, Z.-H.; Wang, H.-M.D. Evaluation of Cinnamomum osmophloeum Kanehira Extracts on Tyrosinase Suppressor, Wound Repair Promoter, and Antioxidant. Sci. World J. 2015, 2015, 7. [Google Scholar] [CrossRef]
- Lin, G.-M.; Lin, H.-Y.; Hsu, C.-Y.; Chang, S.-T. Structural characterization and bioactivity of proanthocyanidins from indigenous cinnamon (Cinnamomum osmophloeum). J. Sci. Food Agric. 2016, 96, 4749–4759. [Google Scholar] [CrossRef]
- Rao, Y.K.; Fang, S.-H.; Tzeng, Y.-M. Evaluation of the anti-inflammatory and anti-proliferation tumoral cells activities of Antrodia camphorata, Cordyceps sinensis, and Cinnamomum osmophloeum bark extracts. J. Ethnopharmacol. 2007, 114, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Chao, L.K.; Hua, K.-F.; Hsu, H.-Y.; Cheng, S.-S.; Lin, I.F.; Chen, C.-J.; Chen, S.-T.; Chang, S.-T. Cinnamaldehyde inhibits pro-inflammatory cytokines secretion from monocytes/macrophages through suppression of intracellular signaling. Food Chem. Toxicol. 2008, 46, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Chao, L.K.; Hua, K.-F.; Hsu, H.-Y.; Cheng, S.-S.; Liu, J.-Y.; Chang, S.-T. Study on the Antiinflammatory Activity of Essential Oil from Leaves of Cinnamomum osmophloeum. J. Agric. Food Chem. 2005, 53, 7274–7278. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.-H.; Rao, Y.K.; Tzeng, Y.-M. Inhibitory effects of flavonol glycosides from Cinnamomum osmophloeum on inflammatory mediators in LPS/IFN-γ-activated murine macrophages. Bioorganic Med. Chem. 2005, 13, 2381–2388. [Google Scholar] [CrossRef] [PubMed]
- Ku, W.-c.; Chang, Y.-l.; Wu, S.-f.; Shih, H.-n.; Tzeng, Y.-m.; Kuo, H.-r.; Chang, K.-m.; Agrawal, D.C.; Liu, B.-l.; Chang, C.-a.; et al. A comparative proteomic study of secretomes in kaempferitrin-treated CTX TNA2 astrocytic cells. Phytomedicine 2017, 36, 137–144. [Google Scholar] [CrossRef]
- Lee, S.-C.; Hsu, J.-S.; Li, C.-C.; Chen, K.-M.; Liu, C.-T. Protective Effect of Leaf Essential Oil from Cinnamomum osmophloeum Kanehira on Endotoxin-Induced Intestinal Injury in Mice Associated with Suppressed Local Expression of Molecules in the Signaling Pathways of TLR4 and NLRP3. PLoS ONE 2015, 10, e0120700. [Google Scholar] [CrossRef]
- Lin, H.-Y.; Chang, S.-T. Kaempferol glycosides from the twigs of Cinnamomum osmophloeum and their nitric oxide production inhibitory activities. Carbohydr. Res. 2012, 364, 49–53. [Google Scholar] [CrossRef]
- Tung, Y.T.; Yen, P.L.; Lin, C.Y.; Chang, S.T. Anti-inflammatory activities of essential oils and their constituents from different provenances of indigenous cinnamon (Cinnamomum osmophloeum) leaves. Pharm. Biol. 2010, 48, 1130–1136. [Google Scholar] [CrossRef]
- Tung, Y.-T.; Chua, M.-T.; Wang, S.-Y.; Chang, S.-T. Anti-inflammation activities of essential oil and its constituents from indigenous cinnamon (Cinnamomum osmophloeum) twigs. Bioresour. Technol. 2008, 99, 3908–3913. [Google Scholar] [CrossRef]
- Chang, C.W.; Chang, W.L.; Chang, S.T.; Cheng, S.S. Antibacterial activities of plant essential oils against Legionella pneumophila. Water Res. 2008, 42, 278–286. [Google Scholar] [CrossRef]
- Chang, S.-T.; Chen, P.-F.; Chang, S.-C. Antibacterial activity of leaf essential oils and their constituents from Cinnamomum osmophloeum. J. Ethnopharmacol. 2001, 77, 123–127. [Google Scholar] [CrossRef]
- Wang, S.-Y.; Chen, P.-F.; Chang, S.-T. Antifungal activities of essential oils and their constituents from indigenous cinnamon (Cinnamomum osmophloeum) leaves against wood decay fungi. Bioresour. Technol. 2005, 96, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.-S.; Liu, J.-Y.; Hsui, Y.-R.; Chang, S.-T. Chemical polymorphism and antifungal activity of essential oils from leaves of different provenances of indigenous cinnamon (Cinnamomum osmophloeum). Bioresour. Technol. 2006, 97, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Chua, M.T.; Tung, Y.T.; Chang, S.T. Antioxidant activities of ethanolic extracts from the twigs of Cinnamomum osmophloeum. Bioresour Technol 2008, 99, 1918–1925. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.-L.; Li, W.-H.; Yu, C.-W.; Hsieh, Y.-C.; Yang, Y.-F.; Liu, J.-T.; Shih, J.; Chu, Y.-J.; Yen, P.-L.; Chang, S.-T.; et al. In Vivo Antioxidant Activities of Essential Oils and Their Constituents from Leaves of the Taiwanese Cinnamomum osmophloeum. J. Agric. Food Chem. 2012, 60, 3092–3097. [Google Scholar] [CrossRef] [PubMed]
- Yeh, H.-F.; Luo, C.-Y.; Lin, C.-Y.; Cheng, S.-S.; Hsu, Y.-R.; Chang, S.-T. Methods for Thermal Stability Enhancement of Leaf Essential Oils and Their Main Constituents from Indigenous Cinnamon (Cinnamomum osmophloeum). J. Agric. Food Chem. 2013, 61, 6293–6298. [Google Scholar] [CrossRef]
- Lin, T.-Y.; Liao, J.-W.; Chang, S.-T.; Wang, S.-Y. Antidyslipidemic Activity of Hot-water Extracts from Leaves of Cinnamomum osmophloeum Kaneh. Phytother. Res. 2011, 25, 1317–1322. [Google Scholar] [CrossRef]
- Cheng, B.H.; Sheen, L.Y.; Chang, S.T. Hypolipidemic effects of S-(+)-linalool and essential oil from Cinnamomum osmophloeum ct. linalool leaves in mice. J. Tradit Complement. Med. 2018, 8, 46–52. [Google Scholar] [CrossRef]
- Lee, S.-C.; Wang, S.-Y.; Li, C.-C.; Liu, C.-T. Anti-inflammatory effect of cinnamaldehyde and linalool from the leaf essential oil of Cinnamomum osmophloeum Kanehira in endotoxin-induced mice. J. Food Drug Anal. 2018, 26, 211–220. [Google Scholar] [CrossRef]
- Yu, C.-W.; Li, W.-H.; Hsu, F.-L.; Yen, P.-L.; Chang, S.-T.; Liao, V.H.-C. Essential Oil Alloaromadendrene from Mixed-Type Cinnamomum osmophloeum Leaves Prolongs the Lifespan in Caenorhabditis elegans. J. Agric. Food Chem. 2014, 62, 6159–6165. [Google Scholar] [CrossRef]
- Chen, T.H.; Huang, Y.H.; Lin, J.J.; Liau, B.C.; Wang, S.Y.; Wu, Y.C.; Jong, T.T. Cytotoxic lignan esters from Cinnamomum osmophloeum. Planta Med. 2010, 76, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-J.; Rao, Y.K.; Chen, K.; Lee, Y.-C.; Tzeng, Y.-M. Effect of flavonol glycosides from cinnamomum osmophloeum leaves on adiponectin secretion and phosphorylation of insulin receptor-β in 3t3-l1 adipocytes. J. Ethnopharmacol. 2009, 126, 79–85. [Google Scholar] [CrossRef]
- Cheng, B.-H.; Sheen, L.-Y.; Chang, S.-T. Evaluation of anxiolytic potency of essential oil and s-(+)-linalool from cinnamomum osmophloeum ct. Linalool leaves in mice. J. Tradit. Complement Med. 2014, 5, 27–34. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, S.-S.C.; Lu, T.-M.; Chao, P.-C.; Lai, Y.-Y.; Tsai, H.-T.; Chen, C.-S.; Lee, Y.-P.; Chen, S.-C.; Chou, M.-C.; Yang, C.-C. In Vivo Cytokine Modulatory Effects of Cinnamaldehyde, the Major Constituent of Leaf Essential Oil from Cinnamomum osmophloeum Kaneh. Phytother. Res. 2011, 25, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Theurer, M.; Shaik, N.; Lang, F. Stimulation of suicidal erythrocyte death by trans-cinnamaldehyde. Phytomedicine 2013, 20, 1119–1123. [Google Scholar] [CrossRef] [PubMed]
- Mdoe, F.P.; Cheng, S.-S.; Msangi, S.; Nkwengulila, G.; Chang, S.-T.; Kweka, E.J. Activity of Cinnamomum osmophloeum leaf essential oil against Anopheles gambiae s.s. Parasit Vectors 2014, 7, 209. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.-S.; Liu, J.-Y.; Tsai, K.-H.; Chen, W.-J.; Chang, S.-T. Chemical Composition and Mosquito Larvicidal Activity of Essential Oils from Leaves of Different Cinnamomum osmophloeum Provenances. J. Agric. Food Chem. 2004, 52, 4395–4400. [Google Scholar] [CrossRef]
- Cheng, S.-S.; Liu, J.-Y.; Lin, C.-Y.; Hsui, Y.-R.; Lu, M.-C.; Wu, W.-J.; Chang, S.-T. Terminating red imported fire ants using Cinnamomum osmophloeum leaf essential oil. Bioresour. Technol. 2008, 99, 889–893. [Google Scholar] [CrossRef]
- Cheng, S.-S.; Liu, J.-Y.; Huang, C.-G.; Hsui, Y.-R.; Chen, W.-J.; Chang, S.-T. Insecticidal activities of leaf essential oils from cinnamomum osmophloeum against three mosquito species. Bioresour. Technol. 2009, 100, 457–464. [Google Scholar] [CrossRef]
- Shimizu, M.; Ito, T.; Terashima, S.; Hayashi, T.; Arisawa, M.; Morita, N.; Kurokawa, S.; Ito, K.; Hashimoto, Y. Inhibition of lens aldose reductase by flavonoids. Phytochemistry 1984, 23, 1885–1888. [Google Scholar] [CrossRef]
- Przygodzka, M.; Zielińska, D.; Ciesarová, Z.; Kukurová, K.; Zieliński, H. Comparison of methods for evaluation of the antioxidant capacity and phenolic compounds in common spices. LWT Food Sci. Technol. 2014, 58, 321–326. [Google Scholar] [CrossRef]
- Guo, J.-Y.; Huo, H.-R.; Zhao, B.-S.; Liu, H.-B.; Li, L.-F.; Ma, Y.-Y.; Guo, S.-Y.; Jiang, T.-L. Cinnamaldehyde reduces IL-1β-induced cyclooxygenase-2 activity in rat cerebral microvascular endothelial cells. Eur. J. Pharmacol. 2006, 537, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Gan, F.F.; Chua, Y.S.; Scarmagnani, S.; Palaniappan, P.; Franks, M.; Poobalasingam, T.; Bradshaw, T.D.; Westwell, A.D.; Hagen, T. Structure–activity analysis of 2′-modified cinnamaldehyde analogues as potential anticancer agents. Biochem. Biophys. Res. Commun. 2009, 387, 741–747. [Google Scholar] [CrossRef]
- Wu, S.-J.; Ng, L.-T. MAPK inhibitors and pifithrin-alpha block cinnamaldehyde-induced apoptosis in human PLC/PRF/5 cells. Food Chem. Toxicol. 2007, 45, 2446–2453. [Google Scholar] [CrossRef]
- Curra, M.; Soares Junior, L.A.V.; Martins, M.D.; Santos, P.S.d.S. Chemotherapy protocols and incidence of oral mucositis. An integrative review. Einstein (Sao Paulo) 2018, 16, eRW4007. [Google Scholar] [CrossRef] [PubMed]
- Köstler, W.J.; Hejna, M.; Wenzel, C.; Zielinski, C.C. Oral Mucositis Complicating Chemotherapy and/or Radiotherapy: Options for Prevention and Treatment. CA: A Cancer J. Clin. 2001, 51, 290–315. [Google Scholar] [CrossRef]
- Lalla, R.V.; Bowen, J.; Barasch, A.; Elting, L.; Epstein, J.; Keefe, D.M.; McGuire, D.B.; Migliorati, C.; Nicolatou-Galitis, O.; Peterson, D.E.; et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 2014, 120, 1453–1461. [Google Scholar] [CrossRef]
- Molania, T.; Moghadamnia, A.A.; Pouramir, M.; Aghel, S.; Moslemi, D.; Ghassemi, L.; Motallebnejad, M. The effect of Cinnamaldehyde on mucositis and salivary antioxidant capacity in gamma-irradiated rats (a preliminary study). Daru 2012, 20, 89. [Google Scholar] [CrossRef]
- Parkins, C.S.; Fowler, J.F.; Yu, S. A murine model of lip epidermal/mucosal reactions to X-irradiation. Radiother. Oncol. 1983, 1, 159–165. [Google Scholar] [CrossRef]
- Veilleux, M.-P.; Grenier, D. Determination of the effects of cinnamon bark fractions on Candida albicans and oral epithelial cells. BMC Complement. Altern. Med. 2019, 19, 303. [Google Scholar] [CrossRef]
- Feghali, K.; Feldman, M.; La, V.D.; Santos, J.; Grenier, D. Cranberry Proanthocyanidins: Natural Weapons against Periodontal Diseases. J. Agric. Food Chem. 2012, 60, 5728–5735. [Google Scholar] [CrossRef] [PubMed]
- Raeessi, M.A.; Raeessi, N.; Panahi, Y.; Gharaie, H.; Davoudi, S.M.; Saadat, A.; Karimi Zarchi, A.A.; Raeessi, F.; Ahmadi, S.M.; Jalalian, H. “Coffee plus honey” versus “topical steroid” in the treatment of chemotherapy-induced oral mucositis: A randomised controlled trial. BMC Complement. Altern. Med. 2014, 14, 293. [Google Scholar] [CrossRef] [PubMed]
- Fidler, P.; Loprinzi, C.L.; O’Fallon, J.R.; Leitch, J.M.; Lee, J.K.; Hayes, D.L.; Novotny, P.; Clemens-Schutjer, D.; Bartel, J.; Michalak, J.C. Prospective evaluation of a chamomile mouthwash for prevention of 5-FU-induced oral mucositis. Cancer 1996, 77, 522–525. [Google Scholar] [CrossRef]
- Mazokopakis, E.E.; Vrentzos, G.E.; Papadakis, J.A.; Babalis, D.E.; Ganotakis, E.S. Wild chamomile (Matricaria recutita L.) mouthwashes in methotrexate-induced oral mucositis. Phytomedicine 2005, 12, 25–27. [Google Scholar] [CrossRef]
- Pourdeghatkar, F.; Motaghi, M.; Darbandi, B.; BagherSalimi, A. The Effect of Chamomile Mouthwash on the Prevention of Oral Mucositis Caused by Chemotherapy in Children with Acute Lymphoblastic Leukemia. SSU 2017, 7, 76–81. [Google Scholar]
- Aoyama, T.; Nishikawa, K.; Takiguchi, N.; Tanabe, K.; Imano, M.; Fukushima, R.; Sakamoto, J.; Oba, M.S.; Morita, S.; Kono, T.; et al. Double-blind, placebo-controlled, randomized phase II study of TJ-14 (hangeshashinto) for gastric cancer chemotherapy-induced oral mucositis. Cancer Chemother Pharm. 2014, 73, 1047–1054. [Google Scholar] [CrossRef]
- Kono, T.; Kaneko, A.; Matsumoto, C.; Miyagi, C.; Ohbuchi, K.; Mizuhara, Y.; Miyano, K.; Uezono, Y. Multitargeted Effects of Hangeshashinto for Treatment of Chemotherapy-Induced Oral Mucositis on Inducible Prostaglandin E2 Production in Human Oral Keratinocytes. Integr. Cancer Ther. 2014, 13, 435–445. [Google Scholar] [CrossRef]
- You, W.C.; Hsieh, C.C.; Huang, J.T. Effect of Extracts from Indigowood Root (Isatis indigotica Fort.) on Immune Responses in Radiation-Induced Mucositis. J. Altern. Complementary Med. 2009, 15, 771–778. [Google Scholar] [CrossRef]
- Tiemann, P.; Toelg, M.; Ramos, F.M.H. Administration of Ratanhia-based herbal oral care products for the prophylaxis of oral mucositis in cancer chemotherapy patients: A clinical trial. Evid Based Complement. Altern. Med. 2007, 4, 361–366. [Google Scholar] [CrossRef]
- Sahebjamee, M.; Mansourian, A.; Hajimirzamohammad, M.; Zadeh, M.T.; Bekhradi, R.; Kazemian, A.; Manifar, S.; Ashnagar, S.; Doroudgar, K. Comparative Efficacy of Aloe vera and Benzydamine Mouthwashes on Radiation-induced Oral Mucositis: A Triple-blind, Randomised, Controlled Clinical Trial. Oral Health Prev. Dent. 2015, 13, 309–315. [Google Scholar] [CrossRef]
- Hawley, P.; Hovan, A.; McGahan, C.E.; Saunders, D. A randomized placebo-controlled trial of manuka honey for radiation-induced oral mucositis. Supportive Care Cancer 2014, 22, 751–761. [Google Scholar] [CrossRef]
- Miranzadeh, S.; Adib-Hajbaghery, M.; Soleymanpoor, L.; Ehsani, M. A New mouthwash for Chemotherapy Induced Stomatitis. Nurs. Midwifery Stud. 2014, 3, e20249. [Google Scholar] [CrossRef] [PubMed]
- Babaee, N.; Moslemi, D.; Khalilpour, M.; Vejdani, F.; Moghadamnia, Y.; Bijani, A.; Baradaran, M.; Kazemi, M.T.; Khalilpour, A.; Pouramir, M.; et al. Antioxidant capacity of calendula officinalis flowers extract and prevention of radiation induced oropharyngeal mucositis in patients with head and neck cancers: A randomized controlled clinical study. Daru 2013, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Khanal, B.; Baliga, M.; Uppal, N. Effect of topical honey on limitation of radiation-induced oral mucositis: An intervention study. Int. J. Oral Maxillofac. Surg. 2010, 39, 1181–1185. [Google Scholar] [CrossRef] [PubMed]
- Kuduban, O.; Mazlumoglu, M.R.; Kuduban, S.D.; Erhan, E.; Cetin, N.; Kukula, O.; Yarali, O.; Cimen, F.K.; Cankaya, M. The effect of hippophae rhamnoides extract on oral mucositis induced in rats with methotrexate. J. Appl Oral Sci 2016, 24, 423–430. [Google Scholar] [CrossRef][Green Version]
- Motallebnejad, M.; Akram, S.; Moghadamnia, A.; Moulana, Z.; Omidi, S. The effect of topical application of pure honey on radiation-induced mucositis: A randomized clinical trial. J. Contemp. Dent. Pract. 2008, 9, 40–47. [Google Scholar] [CrossRef]
- Rashad, U.M.; Al-Gezawy, S.M.; El-Gezawy, E.; Azzaz, A.N. Honey as topical prophylaxis against radiochemotherapy-induced mucositis in head and neck cancer. J. Laryngol. Otol. 2009, 123, 223–228. [Google Scholar] [CrossRef]
- Lambros, M.P.; Kondapalli, L.; Parsa, C.; Mulamalla, H.C.; Orlando, R.; Pon, D.; Huang, Y.; Chow, M.S.S. Molecular signatures in the prevention of radiation damage by the synergistic effect of n-acetyl cysteine and qingre liyan decoction, a traditional chinese medicine, using a 3-dimensional cell culture model of oral mucositis. Evid. Based Complement Alternat. Med. 2015, 2015, 425760. [Google Scholar] [CrossRef]
- Loo, W.T.Y.; Jin, L.J.; Chow, L.W.C.; Cheung, M.N.B.; Wang, M. Rhodiola algida improves chemotherapy-induced oral mucositis in breast cancer patients. Expert Opin. Investig. Drugs 2010, 19, S91–S100. [Google Scholar] [CrossRef]
- Wu, M.-h.; Yuan, B.; Liu, Q.-f.; Wang, Q. Study of qingre liyan decoction (清热利咽汤) in treating and preventing acute radioactive oral mucositis. Chin. J. Integr. Med. 2007, 13, 280–284. [Google Scholar] [CrossRef]
- Puataweepong, P.; Dhanachai, M.; Dangprasert, S.; Sithatani, C.; Sawangsilp, T.; Narkwong, L.; Puttikaran, P.; Intragumtornchai, T. The efficacy of oral aloe vera juice for radiation induced mucositis in head and neck cancer patients: A double-blind placebo-controlled study. Asian Biomedicine 2009, 3, 375–382. [Google Scholar]
- De Freitas Cuba, L.; Braga Filho, A.; Cherubini, K.; Salum, F.G.; Figueiredo, M.A.Z.d. Topical application of aloe vera and vitamin e on induced ulcers on the tongue of rats subjected to radiation: Clinical and histological evaluation. Support. Care Cancer 2016, 24, 2557–2564. [Google Scholar] [CrossRef] [PubMed]
- Sencer, S.F.; Zhou, T.; Freedman, L.S.; Ives, J.A.; Chen, Z.; Wall, D.; Nieder, M.L.; Grupp, S.A.; Yu, L.C.; Sahdev, I.; et al. Traumeel s in preventing and treating mucositis in young patients undergoing sct: A report of the children’s oncology group. Bone Marrow Transplant 2012, 47, 1409–1414. [Google Scholar] [CrossRef] [PubMed]
- Braga, F.T.M.M.; Santos, A.C.F.; Bueno, P.C.P.; Silveira, R.C.C.P.; Santos, C.B.; Bastos, J.K.; Carvalho, E.C. Use of chamomilla recutita in the prevention and treatment of oral mucositis in patients undergoing hematopoietic stem cell transplantation: A randomized, controlled, phase ii clinical trial. Cancer Nursing 2015, 38, 322–329. [Google Scholar] [CrossRef]
- Napeñas, J.J.; Brennan, M.T.; Coleman, S.; Kent, M.L.; Noll, J.; Frenette, G.; Nussbaum, M.L.; Mougeot, J.-L.; Paster, B.J.; Lockhart, P.B.; et al. Molecular methodology to assess the impact of cancer chemotherapy on the oral bacterial flora: A pilot study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2010, 109, 554–560. [Google Scholar] [CrossRef]
- Sonalika, W.G.; Amsavardani Tayaar, S.; Bhat, K.G.; Patil, B.R.; Muddapur, M.V. Oral microbial carriage in oral squamous cell carcinoma patients at the time of diagnosis and during radiotherapy – A comparative study. Oral Oncol. 2012, 48, 881–886. [Google Scholar] [CrossRef]
- Panghal, M.; Kaushal, V.; Kadayan, S.; Yadav, J.P. Incidence and risk factors for infection in oral cancer patients undergoing different treatments protocols. BMC Oral Health 2012, 12, 22. [Google Scholar] [CrossRef]
- Laheij, A.M.G.A.; de Soet, J.J.; von dem Borne, P.A.; Kuijper, E.J.; Kraneveld, E.A.; van Loveren, C.; Raber-Durlacher, J.E. Oral bacteria and yeasts in relationship to oral ulcerations in hematopoietic stem cell transplant recipients. Supportive Care Cancer 2012, 20, 3231–3240. [Google Scholar] [CrossRef]
- Cechinel-Zanchett, C.C.; Boeing, T.; Somensi, L.B.; Steimbach, V.M.B.; Campos, A.; Krueger, C.d.M.A.; Schultz, C.; Sant’ana, D.d.M.G.; Cechinel-Filho, V.; Mota da Silva, L.; et al. Flavonoid-rich fraction of Bauhinia forficata Link leaves prevents the intestinal toxic effects of irinotecan chemotherapy in IEC-6 cells and in mice. Phytother. Res. 2019, 33, 90–106. [Google Scholar] [CrossRef]
- Liu, Z.; Xi, J.; Schröder, S.; Wang, W.; Xie, T.; Wang, Z.; Bao, S.; Fei, J. Chimonanthus nitens var. salicifolius Aqueous Extract Protects against 5-Fluorouracil Induced Gastrointestinal Mucositis in a Mouse Model. Evid. -Based Complementary Altern. Med. 2013, 2013, 12. [Google Scholar] [CrossRef]
- Vladimir, R.A.S.N.; Gustavo, S.A.; Rafael, T.G.; Samara, H.I.; Maralice, C.B.; Evandro, N.A.; Efigenia Ferreira e, F.; Ana Cristina Viana, C.; Alexandre, A.S.; Sheila, R.L.A.; et al. Mucoadhesive Propolis Gel for Prevention of Radiation-Induced Oral Mucositis. Curr. Clin. Pharmacol. 2014, 9, 359–364. [Google Scholar] [CrossRef]
- Georgakopoulou, E.A. Cinnamon contact stomatitis. J. Derm. Case Rep. 2010, 4, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, S.; Avon, S.L. Contact allergy to cinnamon: Case report. J. Can. Dent. Assoc. 2008, 74, 445–461. [Google Scholar] [PubMed]
- Calapai, G.; Miroddi, M.; Mannucci, C.; Minciullo, P.L.; Gangemi, S. Oral adverse reactions due to cinnamon-flavoured chewing gums consumption. Oral Dis. 2014, 20, 637–643. [Google Scholar] [CrossRef]
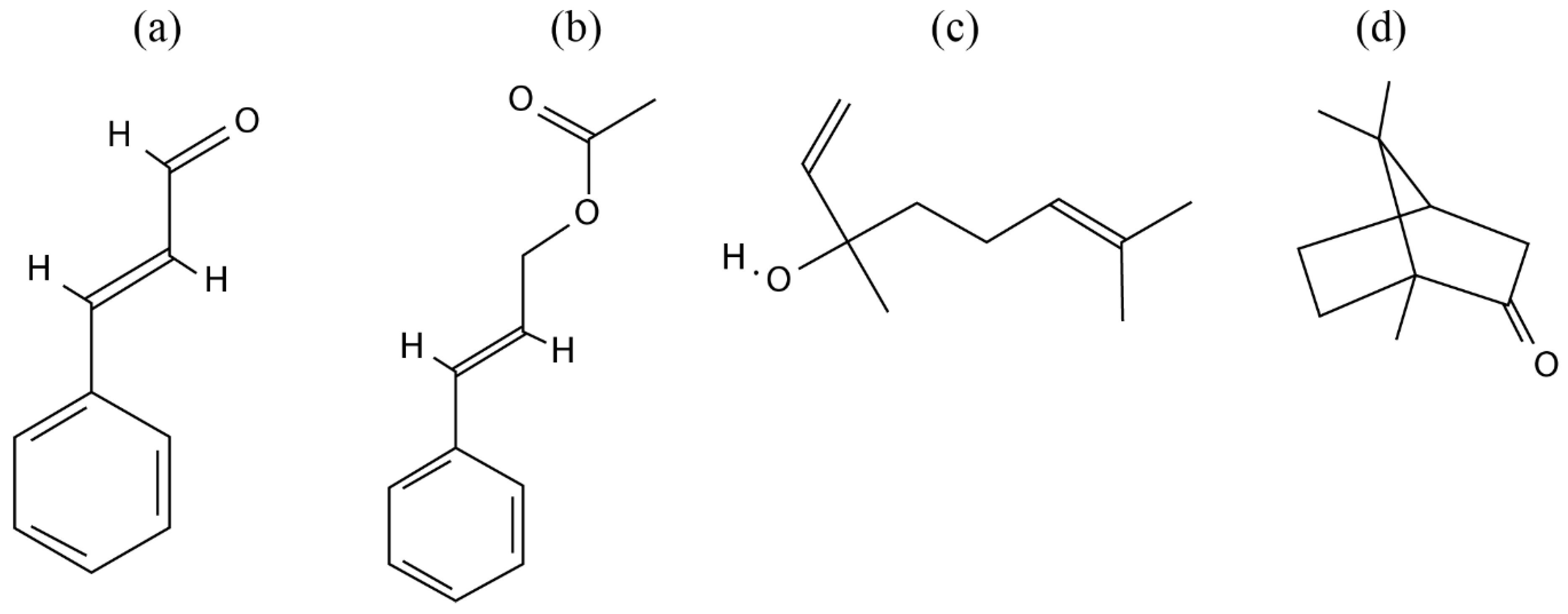
| Bioactivity | Chemical Identification | C. osmophloeum Parts | Constituent (s) | Study | Mechanisms | Reference |
|---|---|---|---|---|---|---|
| Anti-inflammatory effect | LC-MS/MS | Leaves | Kaempferitrin | In vitro | Down-regulate the extracellular LDL-R (chronic inflammation-related diabetes mellitus) | Ku et al., (2017) [26] |
| GC-MS | Twigs | Trans-cinnamaldehyde, caryophyllene oxide, L-borneol, L-bornyl acetate, eugenol, β-caryophyllene, E-nerolidol, and cinnamyl | In vitro | Suppressing nitric oxide synthesis by LPS-stimulated macrophages | Tung et al. (2008) [30] | |
| GC-MS | Leaves | Trans-cinnamaldehyde,(-)-aromadendrene, caryophyllene oxide, T-cadinol, and α-cadinol | In vitro | Suppressing nitric oxide production by LPS-stimulated macrophages | Tung et al. (2010) [29] | |
| GC-MS and HPLC | Leaves | Cinnamaldehyde | In vitro | Cinnamaldehyde inhibits LPS-mediated pro-inflammatory cytokine production | Chao et al. (2008) [23] | |
| TLC | Leaves | NA | In vitro | Inhibition of the production of NO and cytokines (TNF-a and IL-12), from LPS/IFNc-activated macrophages | Fang, Rao & Tzeng (2005) [25] | |
| CC, HPLC, TLC, ESIMS, and GC-MS | Twigs | Kaempferol glycosides | In vitro | Nitric oxide production inhibitory activities | Lin & Chang(2012) [28] | |
| GC-MS | Leaves | Linalool and cinnamaldehyde | In vivo | Inhibition of the expression of molecules in both TLR4 and NLRP3 signaling pathways | Lee et al. (2018) [40] | |
| GC-MS | Leaves | 21 compounds were identified | In vitro | Inhibition of IL-1â and IL-6 production | Chao et al. (2005) [24] | |
| Antibacterial activity | GC | Leaves | Cinnamaldehyde | In vitro | Bactericidal | Chang, Chen & Chang (2001) [32] |
| GC-MS | Leaves | Cinnamaldehyde, cinnamic acid, cinnamyl alcohol, and cinnamyl acetate | In vitro | Bacterial inhibition | Chang et al. (2008) [31] | |
| Antifungal activity | GC-MS | Leaves | Cinnamaldehyde | In vitro | NA | Cheng et al. (2006) [34] |
| GC-MS | Leaves | Cinnamaldehyde | In vitro | NA | Wang, Chen & Chang (2005) [33] | |
| Antioxidant activities | ESIMS | Twigs | Kaempferol-7-O-rhamnoside | In vitro | NA | Chua, Tung, & Chang (2008) [35] |
| GC-MS and GC−FID | Leaves | Alloaromadendrene | In vitro | NA | Yu et al. (2014) [41] | |
| GC-MS and GC-FID | Leaves | Trans-cinnamaldehyde | In vitro | NA | Yeh et al. (2013) [37] | |
| GC-MS and GC-FID | Leaves | Trans cinnamaldehyde | In vivo | Expression of antioxidative-related genes was pointedly affected by essential oils from C. osmophloeum. | Hsu et al. (2012) [36] | |
| Antidyslipidemic activity | HPLC | Leaves | Kaempferol and kaempferitrin | In vivo | Cholesterol-lowering activity | Lin et al. (2011) [38] |
| Anti-hyperglycemic and antioxidant activities | A modified vanillin-H2SO4 assay A modified acid-butanol assay The AlCl3 method | Twigs | Proanthocyanidin and tannin contents | In vitro | CoTE has PTP1B inhibitory activity to improve insulin or leptin resistance | Lin et al. (2016) [18] |
| Hepatoprotective effects | NA | Leaves | trans-cinnamaldehyde, ()-aromadendrene, T-cadinol, or R-cadinol | In vivo | The modulation of anti-inflammatory activities (decreased the aspartate aminotransferase (AST), alanine aminotransferase (ALT), tumor necrosis factor-R (TNF-R), and interleukin 6 (IL-6) levels in serum) | Tung et al. (2011) [6] |
| Pancreas Protective Effect and Hypoglycemic activity | GC/MS | Leaves | Linalool | In vivo | 1. Decreased pancreatic values of thiobarbituric acid reactive substances and activities of superoxide dismutase and glutathione reductase 2. Decreased pancreatic levels of interleukin-1β and nitric oxide | Lee et al. (2013) [3] |
| Prevent Cardiac Hypertrophy | HPLC | Leaves | Cinnamaldehyde | In vivo | The protective role of cinnamaldehyde related to the ERK1/2 signaling pathway. | Yang et al. (2015) [7] |
| Treatment of renal interstitial fibroblasts | NA | Leaves | Cinnamaldehyde | In vitro | Inhibit high glucose-induced hypertrophy (decreased cell size; cellular hypertrophy index; and protein levels of collagen IV, fibronectin, and α-smooth muscle actin). | Chao et al. (2010) [4] |
| Anticancer (liver and oral cancer) | TLC, CC and HPLC | Heart wood and roots | Lignan Esters | In vitro | Tumor cell growth inhibition | Chen et al. (2010) [42] |
| Anti-diabetes | TLC | Twigs | Kaempferol glycosides CO-1 and CO-2 | In vitro | Enhanced adiponectin secretion, and activation of the insulin signaling pathway | Lee et al. (2009) [43] |
| Anti-hyperuricemia effect | GC-MS | Leaves | Cinnamaldehyde | In vivo | Acts as a xanthine oxidase inhibitor and reduces the serum uric acid levels | Wang et al. (2008) [2] |
| Anxiolytic properties | HPLC | Leaves | Linalool | In vivo | Reduced the amount of 5-HT, DA and NE and increased the level of dopamine in striatum | Cheng et al. (2014) [44] |
| Wound Repair Promoter and Antioxidant | NA | Leaves | NA | In vitro and in vivo | Inhibited tyrosinase activity and reduced melanin content | Lee et al. (2015) [20] |
| Anti-inflammatory and anti-cancer properties | NA | Barks | NA | In vivo | The growth inhibition of NO, TNF-, and IL-12, and tumor cell proliferation | Rao et al. (2007) [22] |
| Hypolipidemic effects | NA | Leaves | S-(þ)-linalool | In vivo | Inhibited lipid accumulation through downregulation of 3T3-L1 adipocyte differentiation | Cheng et al. (2018) [39] |
| Effect on the human immune system | HS-GC/MS and HPLC | Leaves | Cinnamaldehyde | In vivo | Cytokines modulatory effect | Lin et al. (2011) [45] |
| Potential skin-whitening and protective agent | NA | Leaves | Cinnamaldehyde and cinnamylacetate | In vitro | Neutralized the IBMX-induced increase in melanin content in B16-F10 cells by inhibiting tyrosinase gene expression at the level of transcription | Lee et al. (2015) [20] |
| Anti-inflammatory effect in intestine | GC/MS | Leaves | Linalool | In vivo | The suppression of the TLR4 pathway by CO and partly by the inhibitory effect of CO on the activity of xanthine oxidase | Lee et al. (2015) [27] |
| Anti-tumor | NA | Leaves | Trans-cinnamaldehyde | In vitro | Trans-cinnamaldehyde triggers suicidal death oferythrocytes, i.e., cells devoid of mitochondria and gene expression. | Theurer et al. (2013) [46] |
| Dietary supplements and treatment of hyperuricemia and gout | GC-MS and GC-FID | Leaves | Cinnamaldehyde | In vitro | The xanthine oxidase inhibitory activity | Huang et al. (2018) [19] |
| Anti-hyperglycemic and antioxidant activities | (MALDI/MS) (RP-HPLC) /MS/MS | Twigs | Proanthocyanidin | In vitro | The proanthocyanidins in CoTE mainly consisted of (epi)catechin and contained at least one A-type linkage. The inhibitory activity of α-glucosidase and α-amylase | Lin et al. (2016) [21] |
| No | Chemical Compositions | References |
|---|---|---|
| Cinnamaldehyde type | ||
| 1 | Cinnamaldehyde, Geranyl acetate, Benzaldehyde. | Chang et al. (2001), Wang et al. (2005) [32,33] |
| 2 | trans-Cinnamaldehyde, Benzenepropanal, 4-Allylanisole. | Chang et al. (2008) [31] |
| 3 | trans-Cinnamaldehyde, Cinnamyl acetate, 3-Phenylpropionaldehyde. | Tung et al. (2010) [29] |
| 4 | trans-Cinnamaldehyde, Cinnamyl acetate, 3-Phenylpropionaldehyde. | Tung et al. (2010) [29] |
| 5 | trans-Cinnamaldehyde, Cinnamyl acetate, 3-Phenylpropionaldehyde. | Tung et al. (2010) [29] |
| 6 | trans-Cinnamaldehyde, Cinnamyl acetate, 3-Pheaylpionaldehyde. | Wang et al. (2008) [2] |
| 7 | trans-Cinnamaldehyde, trans-Cinnamyl acetate, 3-Phenylpropionaldehyde. | Hsu et al. (2012) [36] |
| 8 | trans-Cinnamaldehyde, Benzenepropanal, Benzaldehyde. | Yeh et al. (2013), Cheng (2008) [37,49] |
| 9 | trans-Cinnamaldehyde, Benzaldehyde, Benzenepropanal. | Yeh et al. (2013) [37] |
| 10 | trans-Cinnamaldehyde, Benzaldehyde, Benzenepropanal. | Yeh et al. (2013) [37] |
| 11 | trans-Cinnamaldehyde, Benzaldehyde, Benzenepropanal. | Yeh et al. (2013) [37] |
| 12 | trans-Cinnamaldehyde, Linalool, trans-Cinnamyl acetate. | Yeh et al. (2013) [37] |
| 13 | trans-Cinnamaldehyde, Benzaldehyde, 3-Phenylpropionaldehyde. | Huang et al. (2018) [19] |
| 14 | trans-Cinnamaldehyde, trans-Cinnamyl acetate, Benzenepropanal. | Mdoe et al. (2014) [47] |
| 15 | Cinnamaldehyde, Geranyl acetate, Benzaldehyde. | Wang et al. (2005) [33] |
| 16 | trans-Cinnamaldehyde, Cinnamyl acetate, β-Caryophyllene. | Cheng et al. (2004) [48] |
| 17 | trans-Cinnamaldehyde, Benzenepropanal, benzaldehyde. | Cheng et al. (2004) [48] |
| 18 | trans-Cinnamaldehyde, Benzenepropanal, benzaldehyde. | Cheng et al. (2004) [48] |
| 19 | trans-Cinnamaldehyde, Benzenepropanal, 4-Allylanisole. | Cheng et al. (2009) [50] |
| 20 | trans-Cinnamaldehyde, Cinnamyl acetate, Bornyl acetate. | Cheng et al. (2006) [34] |
| 21 | trans-Cinnamaldehyde, Benzenepropanal, Eugenol. | Cheng et al. (2006) [34] |
| 22 | trans-Cinnamaldehyde, Benzenepropanal, Cinnamyl acetate. | Cheng et al. (2006) [34] |
| Cinnamaldehyde/Cinnamyl acetate type | ||
| 23 | Cinnamyl acetate, trans-Cinnamaldehyde, Benzenepropanal. | Chang et al. (2008) [31] |
| 24 | trans-Cinnamaldehyde, Cinnamyl acetate, Benzenepropanal. | Cheng et al. (2004) [48] |
| 25 | Cinnamyl acetate, trans-Cinnamaldehyde, Camphene. | Cheng et al. (2004) [48] |
| 26 | Cinnamyl acetate, trans-Cinnamaldehyde, Benzenepropanal. | Cheng et al. (2009) [50] |
| 27 | trans-Cinnamaldehyde, Cinnamyl acetate, Benzenepropanal. | Cheng et al. (2006) [34] |
| 28 | Cinnamyl acetate, trans-Cinnamaldehyde, Camphene. | Cheng et al. (2006) [34] |
| Cinnamyl acetate type | ||
| 29 | Cinnamyl acetate, 2-Methylbenzofuran, Geranyl acetate. | Chang et al. (2008) [31] |
| 30 | Cinnamyl acetate, trans-Cinnamaldehyde, Caryophyllene oxide. | Tung et al. (2010) [29] |
| 31 | Cinnamyl acetate, 2-Methylbenzofuran, Geranyl acetate. | Cheng et al. (2009) [50] |
| 32 | Cinnamyl acetate, 2-Methylbenzofuran, Geranyl acetate. | Cheng et al. (2006) [34] |
| Linalool type | ||
| 33 | Linalool, trans-Cinnamaldehyde, Cinnamyl acetate. | Chang et al. (2008) [31] |
| 34 | Linalool, β-Caryophyllene, 4-Allylanisole. | Tung et al. (2010) [29] |
| 35 | Linalool, Cinnamaldehyde, 3-phenyl-2-propenal. | Lee et al. (2013) [3] |
| 36 | Linalool, Citral, Coumarin. | Cheng et al. (2004) [48] |
| 37 | Linalool, trans-Cinnamaldehyde, 4-Allylanisole. | Cheng et al. (2009) [50] |
| 38 | Linalool, Coumarin, trans-Cinnamaldehyde. | Cheng et al. (2006) [34] |
| Camphor type | ||
| 39 | Camphor, L-Bornyl acetate, (+)-Limonene. | Chang et al. (2008) [31] |
| 40 | D-(+)-Camphor, L-Bornyl acetate, α-Terpineol | Tung et al. (2010) [29] |
| 41 | D-(+)-Camphor, L-Bornyl acetate, α-Terpineol. | Hsu et al. (2012) [36] |
| 42 | Camphor, bornyl acetate, Limonene. | Cheng et al. (2004) [48] |
| 43 | Camphor, L-Bornyl acetate, (+)-Limonene. | Cheng et al. (2009) [50] |
| 44 | Camphor, Bornyl acetate, Limonene. | Cheng et al. (2006) [34] |
| Mixed type | ||
| 45 | Spathulenol, Linalool, α-Terpineol. | Chang et al. (2008) [31] |
| 46 | Neral, 1.8-Cineol, Linalool. | Chang et al. (2001), Wang et al. (2005) [32,33] |
| 47 | L-Bornyl acetate, Caryophyllene oxide, γ-Eudesmol. | Tung et al. (2008) [30] |
| 48 | L-Bornyl acetate, α-Cadinol, T-Cadinol. | Tung et al. (2010) [29] |
| 49 | L-bornyl acetate, T-Cadinol, α -Cadinol. | Yu et al. (2014) [41] |
| 50 | 1,8-cineole, spathulenol, santolina triene. | Chao et al. (2005) [24] |
| 51 | T-Cadinol, α -Cadinol., bornyl acetate. | Cheng et al. (2004) [48] |
| 52 | Geranial, Neral, 1,8-Cineole. | Cheng et al. (2009) [50] |
| 53 | T-Cadinol, α -Cadinol, bornyl acetate. | Cheng et al. (2006) [34] |
| Intervention | Protocol | Population | Evidence of Effectiveness |
|---|---|---|---|
| Basic oral care | Tooth brushing, flossing, and one mouth rinse | All age groups and across all cancer treatment modalities | Not strong evidence |
| Growth factors and cytokines | Palifermin (keratinocyte growth factor-1) | Patients receiving high-dose chemotherapy and total body irradiation, followed by autologous stem cell transplantation for hematological malignancies | Strong evidence |
| Anti-inflammatory agents | Benzydamine mouthwash | Patients with head and neck cancer receiving moderate-dose radiation therapy (up to 50 Grays), without concomitant chemotherapy | Strong evidence |
| Laser and other light therapy | Low-level laser therapy (LLLT) | Patients receiving high-dose chemotherapy for HSCT with or without total body irradiation | Strong evidence |
| Cryotherapy | The placement of ice chips in the mouth | Patients receiving bolus dosing of 5-fluorouracil | Strong evidence |
| Natural and miscellaneous agents | Systemic zinc supplements administered orally (antioxidant effect) | Patients with oral cancer undergoing radiotherapy or chemoradiation | Not strong evidence |
| Natural Agents | Bioactivity | References |
|---|---|---|
| Yarrow Plant (Achillea millefolium) | Anti-bacterial and anti-inflammatory effect | Mirazandeh et al. (2014) [73] |
| Manuka Honey (Leptospermum scoparium) | Wound healing and anti-microbial | Hawley et al. (2013) [72] |
| Weleda Pflanzen-Zahngel and Weleda Ratanhia-Mundwasser | Anti-inflammatory, anti-bacterial, and lesion healing | Tiemann et al. (2007) [70] |
| Calendula officinalis flowers | Anti-inflammatory, anti-bacterial, and anti-oxidant | Babaee et al. (2013) [74] |
| Honey and coffee | Antioxidant, anti-microbial, and anti-inflammatory | Raeessi et al. (2014) [63] |
| Aloe vera | Anti-inflammatory, bactericidal, and wound healing | Sahebjamee et al. (2015) [71] |
| Hangeshashinto: Pinelliae tuber, Scutellariae Radix, Glycyrrhizae Radix, Zizyphi Fructus, Ginseng Radix, Zingiberis Processum rhizoma, and Coptidis rhizome | Anti-inflammatory | Aoyama et al. (2014) [67] |
| Indigowood Root (Isatis indigotica Fort.) | Anti-inflammatory | You et al. (2009) [69] |
| Topical Honey | Anti-inflammatory, anti-microbial, and wound healing | Khanal et al. (2010) [75] |
| Hippophae rhamnoides L. plant | Anti-oxidant, anti-ulcerogenic, anti-inflammatory, anti-microbial, and proinflammatory cytokine Antagonist | Kuduban et al. (2016) [76] |
| Honey from the clover plant Trifolium alexandrenum | Anti-microbial | Rashad et al. (2009) [78] |
| Qingre Liyan decoction | Anti-oxidant and anti-inflammatory | Lambros et al. (2014) [79] |
| Hangeshashinto | Anti-inflammatory and anti-microbial | Kono et al. (2014) [65] |
| Chamomile | Anti-inflammatory, anti-bacterial, and antifungal | Fidler et al. (1996) [64] |
| Rhodila algida | Anti-oxidant and immunostimulant | Loo et al. (2010) [80] |
| Qingre Liyan Decoction | Enhancing body immunity and promoting salivary EGF | Wu et al. (2007) [81] |
| Chamomile | Anti-inflammatory, anti-bacterial, and anti-fungal | Pourdeghatkar et al. (2017) [66] |
| Pure Honey | Anti-bacterial and anti-inflammatory | Motallebnejad et al. (2008) [77] |
| Aloe vera | Anti-inflammatory, anti-bacterial, and anti-fungal | Puataweepong et al. (2009) [82] |
| Aloe vera and vitamin E | Antioxidant, anti-inflammatory, and healing properties | Cuba et al. (2015) [83] |
| Traumeel S | Anti-inflammatory | Sencer et al. (2012) [84] |
| Chamomilla recutita | Anti-inflammatory | Braga et al. (2015) [85] |
| Wild chamomile (Matricaria recutita L.) | Anti-inflammatory, anti-bacterial, and anti-fungal | Mazokopakis et al. (2003) [65] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakar, A.; Yao, P.-C.; Ningrum, V.; Liu, C.-T.; Lee, S.-C. Beneficial Biological Activities of Cinnamomum osmophloeum and Its Potential Use in the Alleviation of Oral Mucositis: A Systematic Review. Biomedicines 2020, 8, 3. https://doi.org/10.3390/biomedicines8010003
Bakar A, Yao P-C, Ningrum V, Liu C-T, Lee S-C. Beneficial Biological Activities of Cinnamomum osmophloeum and Its Potential Use in the Alleviation of Oral Mucositis: A Systematic Review. Biomedicines. 2020; 8(1):3. https://doi.org/10.3390/biomedicines8010003
Chicago/Turabian StyleBakar, Abu, Pin-Chuan Yao, Valendriyani Ningrum, Cheng-Tzu Liu, and Shih-Chieh Lee. 2020. "Beneficial Biological Activities of Cinnamomum osmophloeum and Its Potential Use in the Alleviation of Oral Mucositis: A Systematic Review" Biomedicines 8, no. 1: 3. https://doi.org/10.3390/biomedicines8010003
APA StyleBakar, A., Yao, P.-C., Ningrum, V., Liu, C.-T., & Lee, S.-C. (2020). Beneficial Biological Activities of Cinnamomum osmophloeum and Its Potential Use in the Alleviation of Oral Mucositis: A Systematic Review. Biomedicines, 8(1), 3. https://doi.org/10.3390/biomedicines8010003






