Anti-Inflammatory and Antitumor Effects of Hydroxytyrosol but Not Oleuropein on Experimental Glioma In Vivo. A Putative Role for the Renin-Angiotensin System
Abstract
:1. Introduction
2. Experimental Section
2.1. Cell Culture
2.2. Animals and Treatments
2.3. Implantation of C6 Glioma Cells
2.4. Measurement of Tumor Volume and Sample Collection
2.5. Aminopeptidase Activity Assays
2.5.1. Aspartyl Aminopeptidase (ASAP) Activity Assay
2.5.2. Aminopeptidase A (APA) Activity Assay
2.5.3. Aminopeptidase N (APN) Activity Assay
2.5.4. Aminopeptidase B (APB) Activity Assay
2.5.5. Insulin-Regulated Aminopeptidase (IRAP) Activity Assay
2.6. Cytokine Production Assay
2.7. Statistical Analysis
3. Results
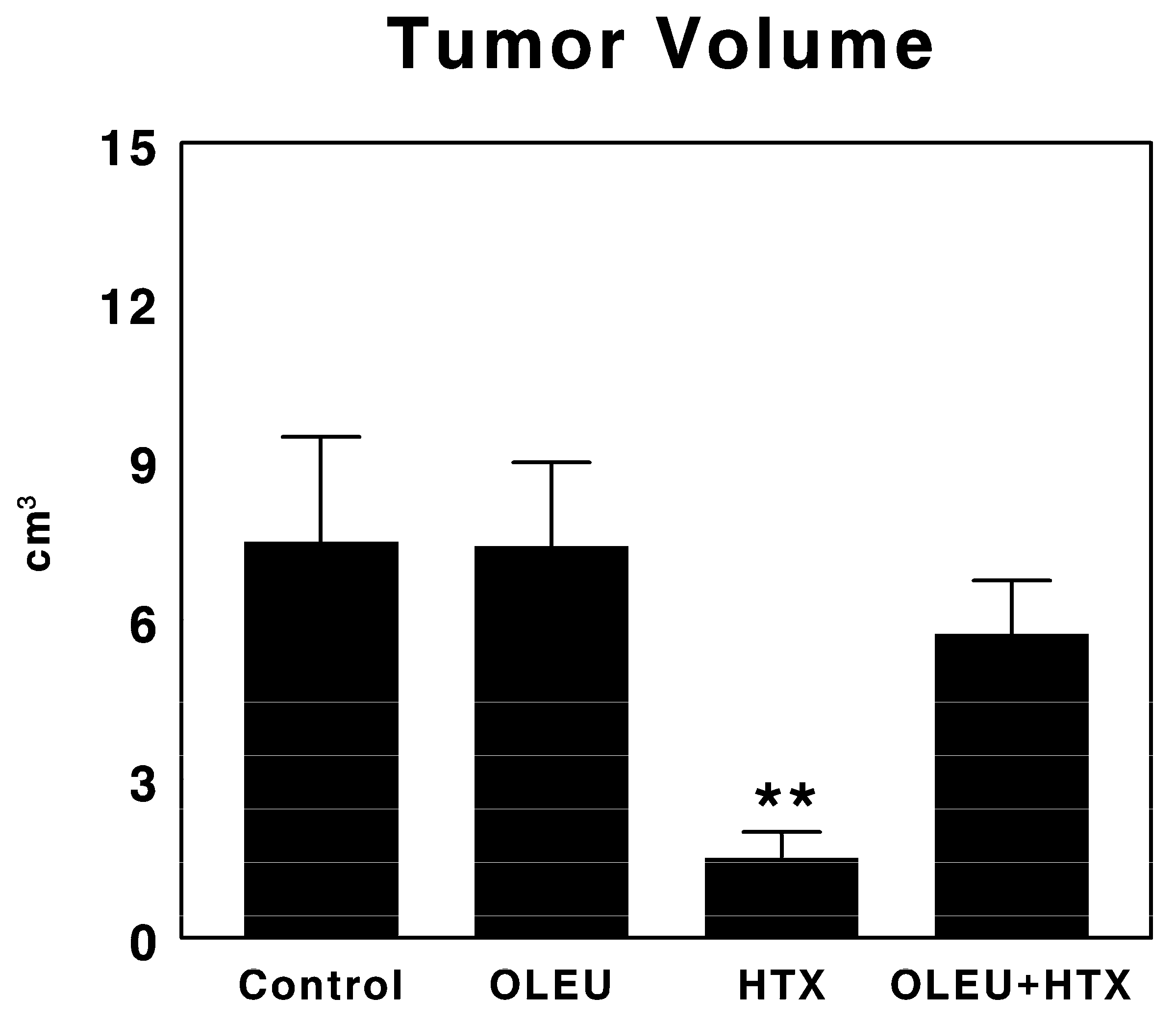
3.1. Effects of Oleuropein and Hydroxytyrosol on Tumor Growth
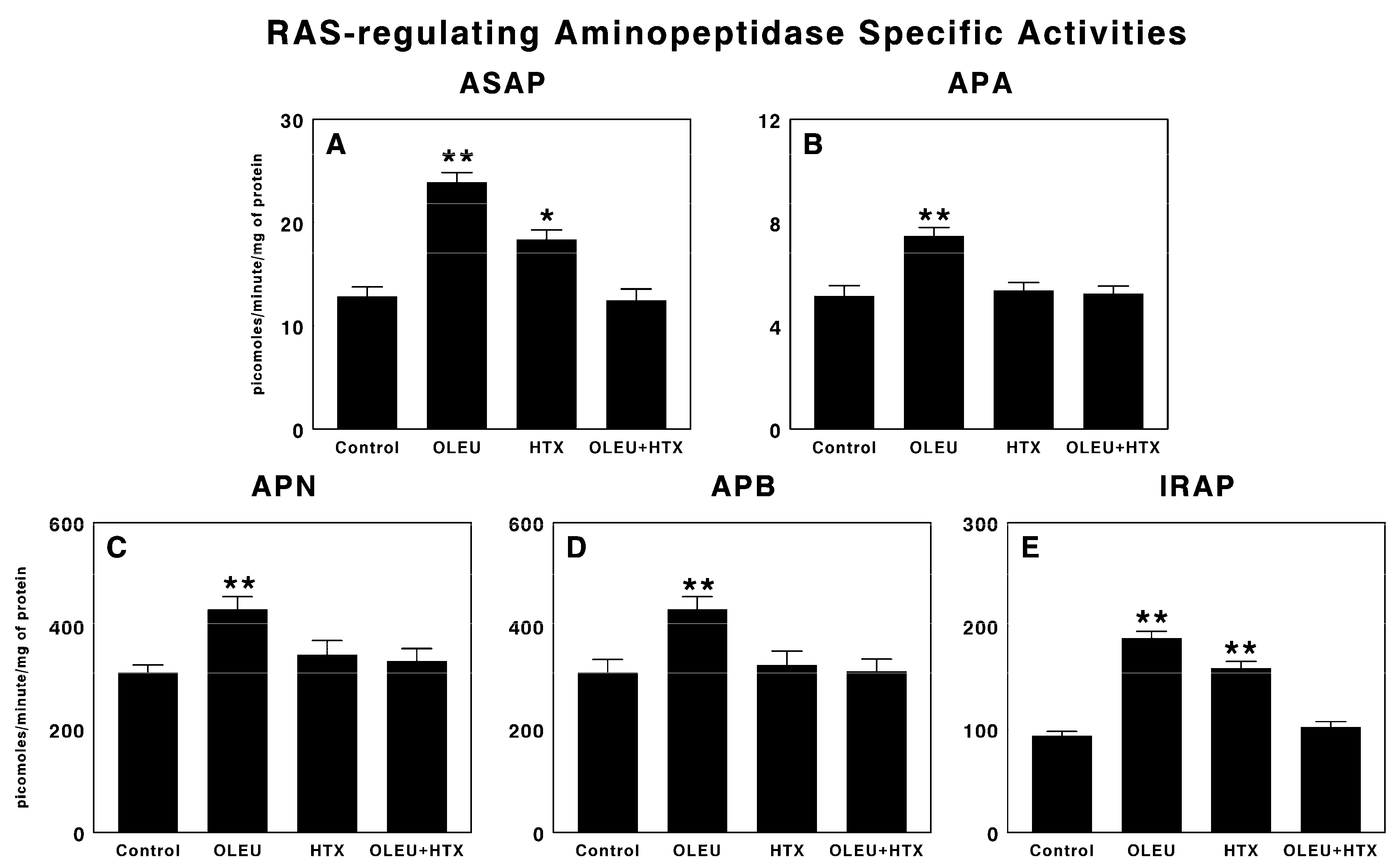
3.2. Effects of Oleuropein and Hydroxytyrosol on RAS-Regulating Aminopeptidases
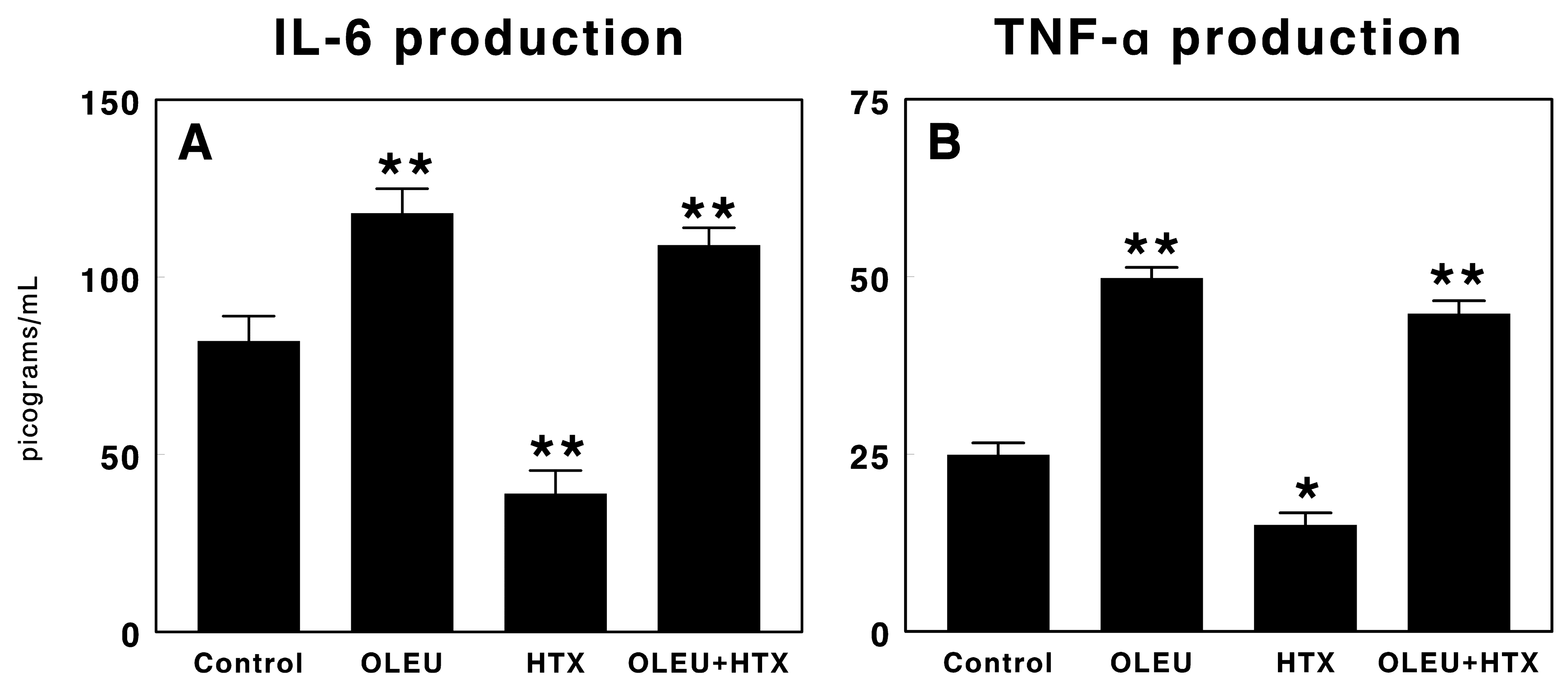
3.3. Effects of Oleuropein and Hydroxytyrosol on Cytokine Production
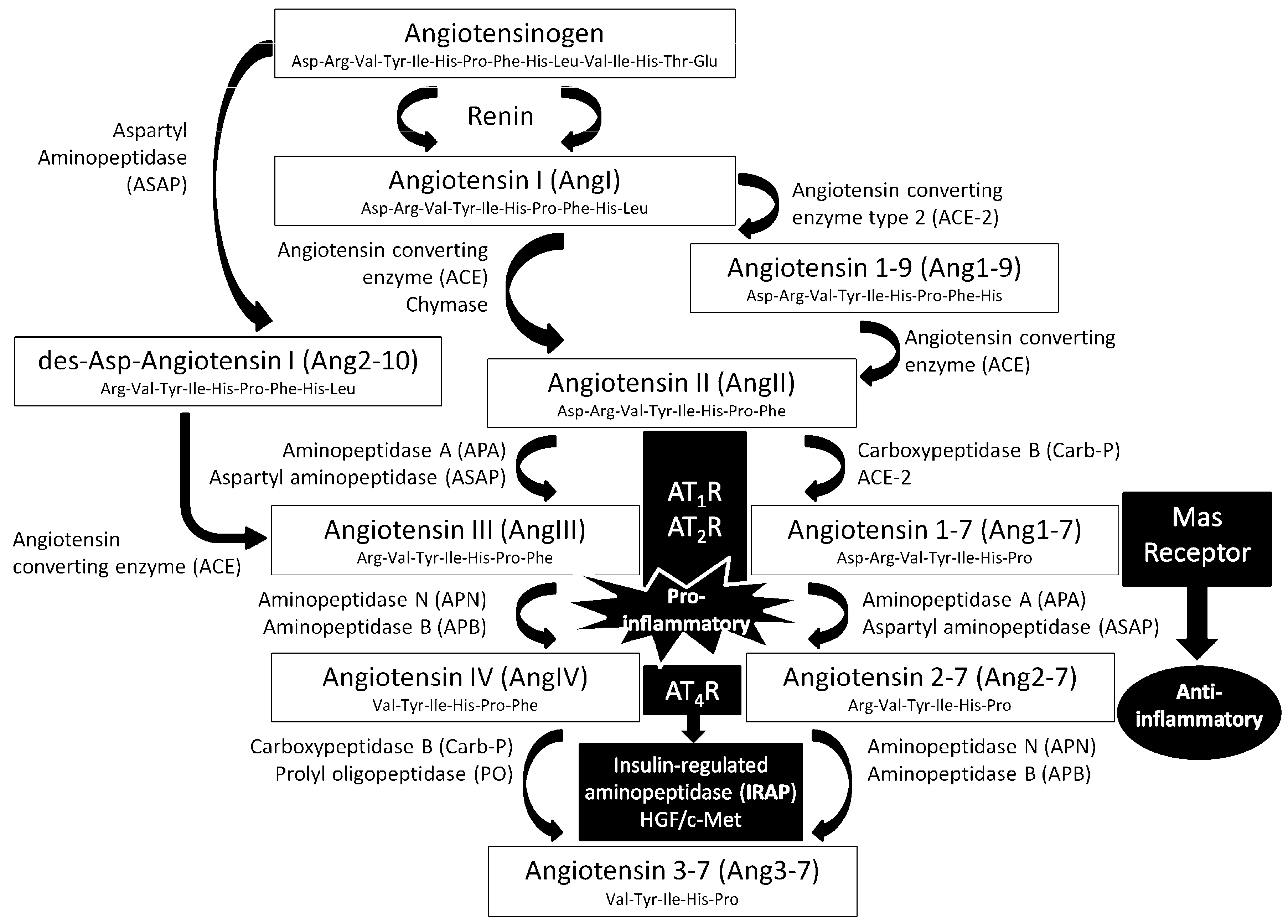
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Candido, J.; Hagemann, T. Cancer-related inflammation. J. Clin. Immunol. 2013, 33, S79–S84. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Benigni, A.; Cassis, P.; Remuzzi, G. Angiotensin ii revisited: New roles in inflammation, immunology and aging. EMBO Mol. Med. 2010, 2, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Stegbauer, J.; Coffman, T.M. New insights into angiotensin receptor actions: From blood pressure to aging. Curr. Opin. Nephrol. Hypertens. 2011, 20, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.W.; Harding, J.W. The brain renin-angiotensin system: A diversity of functions and implications for cns diseases. Pflugers Arch. 2013, 465, 133–151. [Google Scholar] [CrossRef] [PubMed]
- Escobar, E.; Rodriguez-Reyna, T.S.; Arrieta, O.; Sotelo, J. Angiotensin ii, cell proliferation and angiogenesis regulator: Biologic and therapeutic implications in cancer. Curr. Vasc. Pharmacol. 2004, 2, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, O.; Pineda-Olvera, B.; Guevara-Salazar, P.; Hernandez-Pedro, N.; Morales-Espinosa, D.; Ceron-Lizarraga, T.L.; Gonzalez-De la Rosa, C.H.; Rembao, D.; Segura-Pacheco, B.; Sotelo, J. Expression of AT1 and AT2 angiotensin receptors in astrocytomas is associated with poor prognosis. Br. J. Cancer 2008, 99, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Juillerat-Jeanneret, L.; Celerier, J.; Chapuis Bernasconi, C.; Nguyen, G.; Wostl, W.; Maerki, H.P.; Janzer, R.C.; Corvol, P.; Gasc, J.M. Renin and angiotensinogen expression and functions in growth and apoptosis of human glioblastoma. Br. J. Cancer 2004, 90, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Juillerat-Jeanneret, L.; Lohm, S.; Hamou, M.F.; Pinet, F. Regulation of aminopeptidase a in human brain tumor vasculature: Evidence for a role of transforming growth factor-beta. Lab. Investig. 2000, 80, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Mayas, M.D.; Ramirez-Exposito, M.J.; Carrera, M.P.; Cobo, M.; Martinez-Martos, J.M. Renin-angiotensin system-regulating aminopeptidases in tumor growth of rat c6 gliomas implanted at the subcutaneous region. Anticancer Res. 2012, 32, 3675–3682. [Google Scholar] [PubMed]
- Genest, J.; Cantin, M.; Garcia, R.; Thibault, G.; Gutkowska, J.; Schiffrin, E.; Kuchel, O.; Hamet, P. Extrarenal angiotensin-forming enzymes. Clin. Exp. Hypertens. A 1983, 5, 1065–1080. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.I. Ace inhibitors and the kidney. Nephron 1990, 55, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Exposito, M.J.; Martinez, J.M.; Prieto, I.; Alba, F.; Ramirez, M. Comparative distribution of glutamyl and aspartyl aminopeptidase activities in mouse organs. Horm. Metab. Res. 2000, 32, 161–163. [Google Scholar] [CrossRef] [PubMed]
- Kambayashi, Y.; Bardhan, S.; Takahashi, K.; Tsuzuki, S.; Inui, H.; Hamakubo, T.; Inagami, T. Molecular cloning of a novel angiotensin ii receptor isoform involved in phosphotyrosine phosphatase inhibition. J. Biol. Chem. 1993, 268, 24543–24546. [Google Scholar] [PubMed]
- Mukoyama, M.; Nakajima, M.; Horiuchi, M.; Sasamura, H.; Pratt, R.E.; Dzau, V.J. Expression cloning of type 2 angiotensin ii receptor reveals a unique class of seven-transmembrane receptors. J. Biol. Chem. 1993, 268, 24539–24542. [Google Scholar] [CrossRef] [PubMed]
- Blair-West, J.R.; Coghlan, J.P.; Denton, D.A.; Funder, J.W.; Scoggins, B.A.; Wright, R.D. The effect of the heptapeptide (2-8) and hexapeptide (3-8) fragments of angiotensin ii on aldosterone secretion. J. Clin. Endocrinol. Metab. 1971, 32, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Donoghue, M.; Hsieh, F.; Baronas, E.; Godbout, K.; Gosselin, M.; Stagliano, N.; Donovan, M.; Woolf, B.; Robison, K.; Jeyaseelan, R.; et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ace2) converts angiotensin i to angiotensin 1-9. Circ. Res. 2000, 87, E1–E9. [Google Scholar] [CrossRef] [PubMed]
- Nie, W.; Yan, H.; Li, S.; Zhang, Y.; Yu, F.; Zhu, W.; Fan, F.; Zhu, J. Angiotensin-(1-7) enhances angiotensin ii induced phosphorylation of erk1/2 in mouse bone marrow-derived dendritic cells. Mol. Immunol. 2009, 46, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Carrera-Gonzalez, M.P.; Ramirez-Exposito, M.J.; Mayas, M.D.; Martinez-Martos, J.M. Protective role of oleuropein and its metabolite hydroxytyrosol on cancer. Trends Food Sci. Technol. 2013, 31, 92–99. [Google Scholar] [CrossRef]
- Martinez-Martos, J.M.; Mayas, M.D.; Carrera, P.; de Saavedra, J.M.A.; Sanchez-Agesta, R.; Arrazola, M.; Ramirez-Exposito, M.J. Phenolic compounds oleuropein and hydroxytyrosol exert differential effects on glioma development via antioxidant defense systems. J. Funct. Foods 2014, 11, 221–234. [Google Scholar] [CrossRef]
- Garcia, G.E. Ang ii receptor antagonists as modulators of macrophages polarization. Am. J. Physiol.-Renal Phys. 2010, 298, F868–F869. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.A.; Ferreira, A.J.; Simoes, E.S.A.C. Recent advances in the angiotensin-converting enzyme 2-angiotensin(1-7)-mas axis. Exp. Physiol. 2008, 93, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Ma, Y.; Xu, Z.; Wang, J.; Wang, F.; Wang, D.; Pan, S.; Wu, Y.; Pan, H.; Xu, D.; et al. Hydroxytyrosol, a natural molecule from olive oil, suppresses the growth of human hepatocellular carcinoma cells via inactivating akt and nuclear factor-kappa b pathways. Cancer Lett. 2014, 347, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Rosignoli, P.; Fuccelli, R.; Sepporta, M.V.; Fabiani, R. In vitro chemo-preventive activities of hydroxytyrosol: The main phenolic compound present in extra-virgin olive oil. Food Funct. 2016, 7, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Lopez de Las Hazas, M.C.; Pinol, C.; Macia, A.; Motilva, M.J. Hydroxytyrosol and the colonic metabolites derived from virgin olive oil intake induce cell cycle arrest and apoptosis in colon cancer cells. J. Agric. Food Chem. 2017, 65, 6467–6476. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Han, Z.; Ma, Y.; Song, R.; Pei, T.; Zheng, T.; Wang, J.; Xu, D.; Fang, X.; Jiang, H.; et al. Hydroxytyrosol inhibits cholangiocarcinoma tumor growth: An in vivo and in vitro study. Oncol. Rep. 2014, 31, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R. Anti-cancer properties of olive oil secoiridoid phenols: A systematic review of in vivo studies. Food Funct. 2016, 7, 4145–4159. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Liu, Y.; Jiang, Y. Podocalyxin promotes glioblastoma multiforme cell invasion and proliferation by inhibiting angiotensin-(1-7)/mas signaling. Oncol. Rep. 2015, 33, 2583–2591. [Google Scholar] [CrossRef] [PubMed]
- Manna, C.; D’Angelo, S.; Migliardi, V.; Loffredi, E.; Mazzoni, O.; Morrica, P.; Galletti, P.; Zappia, V. Protective effect of the phenolic fraction from virgin olive oils against oxidative stress in human cells. J. Agric. Food Chem. 2002, 50, 6521–6526. [Google Scholar] [CrossRef] [PubMed]
- De la Puerta, R.; Martinez Dominguez, M.E.; Ruiz-Gutierrez, V.; Flavill, J.A.; Hoult, J.R. Effects of virgin olive oil phenolics on scavenging of reactive nitrogen species and upon nitrergic neurotransmission. Life Sci. 2001, 69, 1213–1222. [Google Scholar] [CrossRef]
- Valko, M.; Izakovic, M.; Mazur, M.; Rhodes, C.J.; Telser, J. Role of oxygen radicals in DNA damage and cancer incidence. Mol. Cell. Biochem. 2004, 266, 37–56. [Google Scholar] [CrossRef] [PubMed]
- Hamdi, H.K.; Castellon, R. Oleuropein, a non-toxic olive iridoid, is an anti-tumor agent and cytoskeleton disruptor. Biochem. Biophys. Res. Commun. 2005, 334, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Li, X.; Lin, J.; Zheng, W.; Hu, Z.; Xuan, J.; Ni, W.; Pan, X. Oleuropein inhibits the il-1beta-induced expression of inflammatory mediators by suppressing the activation of nf-kappab and mapks in human osteoarthritis chondrocytes. Food Funct. 2017, 8, 3737–3744. [Google Scholar] [CrossRef] [PubMed]
- Qabaha, K.; AL-Rimawi, F.; Qasem, A.; Naser, S.A. Oleuropein is responsible for the major anti-inflammatory effects of olive leaf extract. J. Med. Food 2017. [Google Scholar] [CrossRef] [PubMed]
- Kotronoulas, A.; Pizarro, N.; Serra, A.; Robledo, P.; Joglar, J.; Rubio, L.; Hernaez, A.; Tormos, C.; Motilva, M.J.; Fito, M.; et al. Dose-dependent metabolic disposition of hydroxytyrosol and formation of mercapturates in rats. Pharmacol. Res. 2013, 77, 47–56. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Expósito, M.J.; Martínez-Martos, J.M. Anti-Inflammatory and Antitumor Effects of Hydroxytyrosol but Not Oleuropein on Experimental Glioma In Vivo. A Putative Role for the Renin-Angiotensin System. Biomedicines 2018, 6, 11. https://doi.org/10.3390/biomedicines6010011
Ramírez-Expósito MJ, Martínez-Martos JM. Anti-Inflammatory and Antitumor Effects of Hydroxytyrosol but Not Oleuropein on Experimental Glioma In Vivo. A Putative Role for the Renin-Angiotensin System. Biomedicines. 2018; 6(1):11. https://doi.org/10.3390/biomedicines6010011
Chicago/Turabian StyleRamírez-Expósito, María Jesús, and José Manuel Martínez-Martos. 2018. "Anti-Inflammatory and Antitumor Effects of Hydroxytyrosol but Not Oleuropein on Experimental Glioma In Vivo. A Putative Role for the Renin-Angiotensin System" Biomedicines 6, no. 1: 11. https://doi.org/10.3390/biomedicines6010011
APA StyleRamírez-Expósito, M. J., & Martínez-Martos, J. M. (2018). Anti-Inflammatory and Antitumor Effects of Hydroxytyrosol but Not Oleuropein on Experimental Glioma In Vivo. A Putative Role for the Renin-Angiotensin System. Biomedicines, 6(1), 11. https://doi.org/10.3390/biomedicines6010011






