Intra-Articular N-Acetylcysteine Reduces Synovitis Without Preventing Cartilage Degeneration in Experimental Osteoarthritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Surgical Induction of Osteoarthritis
2.2. Histological Processing and Evaluation
2.2.1. Retrieval and Handling of Synovial Fluid
2.2.2. Cartilage Tissue Extraction and Homogenization
2.2.3. Quantification of Cytokines, Enzymes, and Degradation Markers by ELISA
2.2.4. Total Antioxidant Status (TAS) and Total Oxidative Status (TOS) Assays
2.3. Statistical Analysis
3. Results
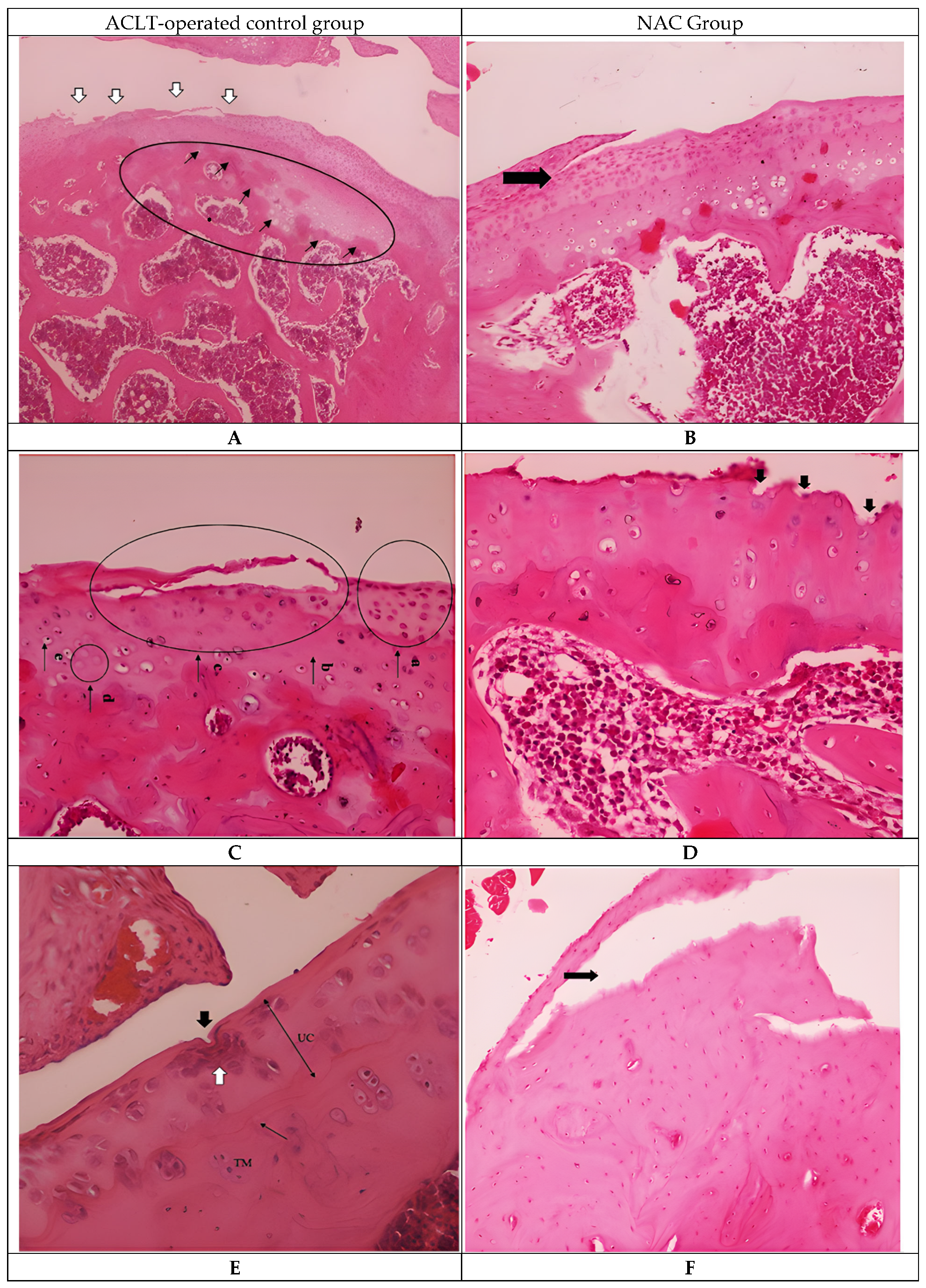
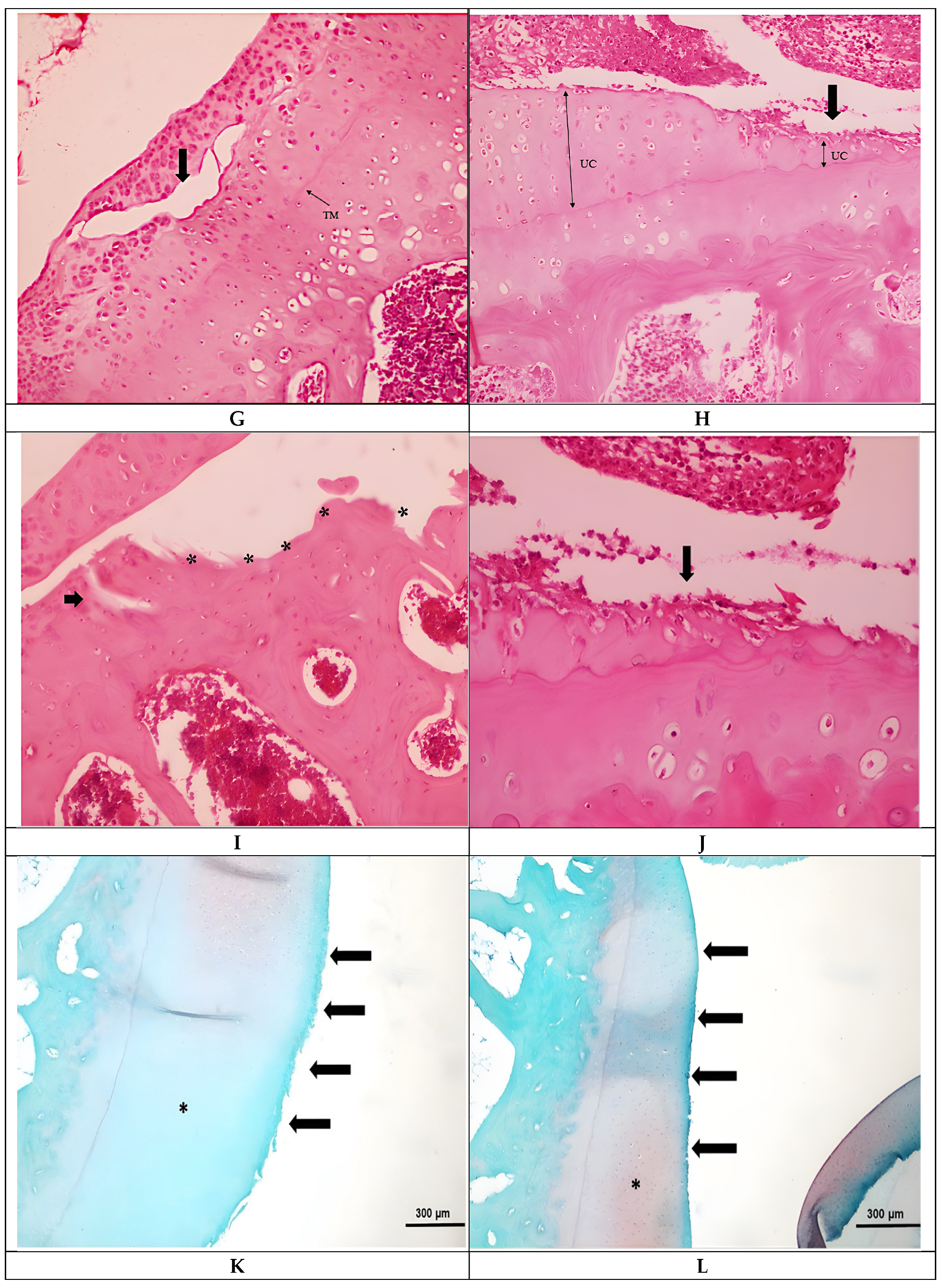
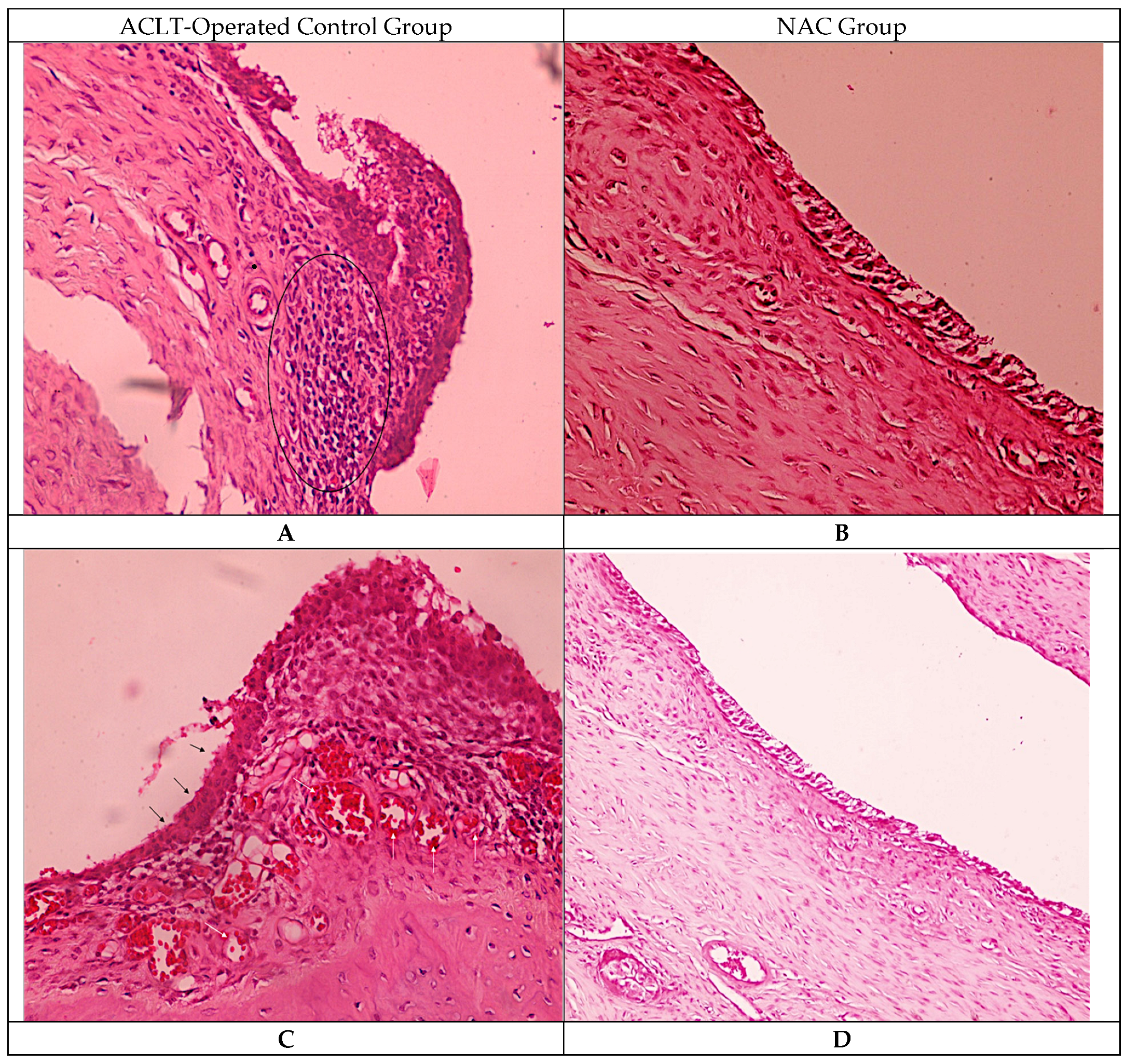
3.1. Histological Observations
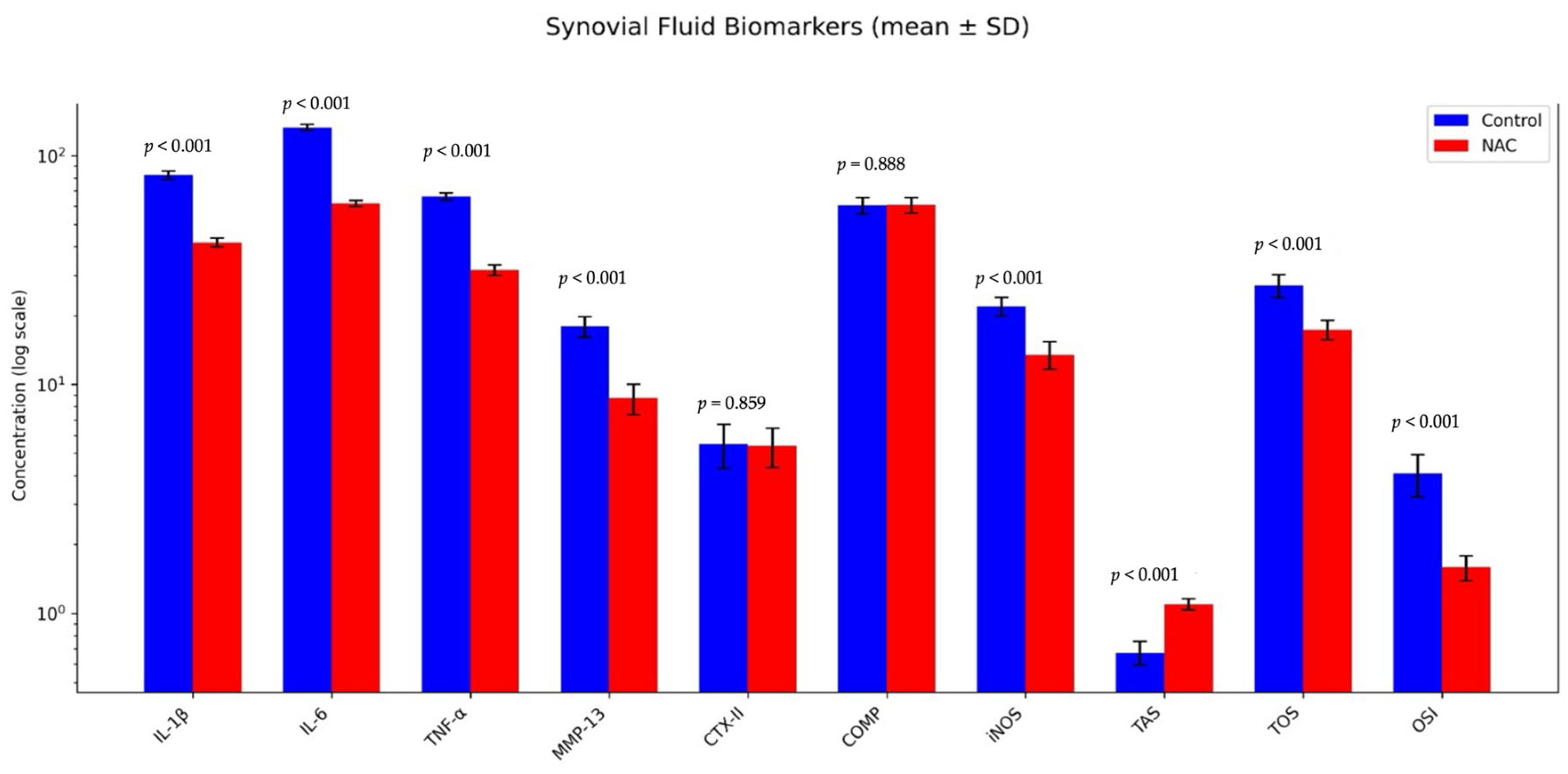
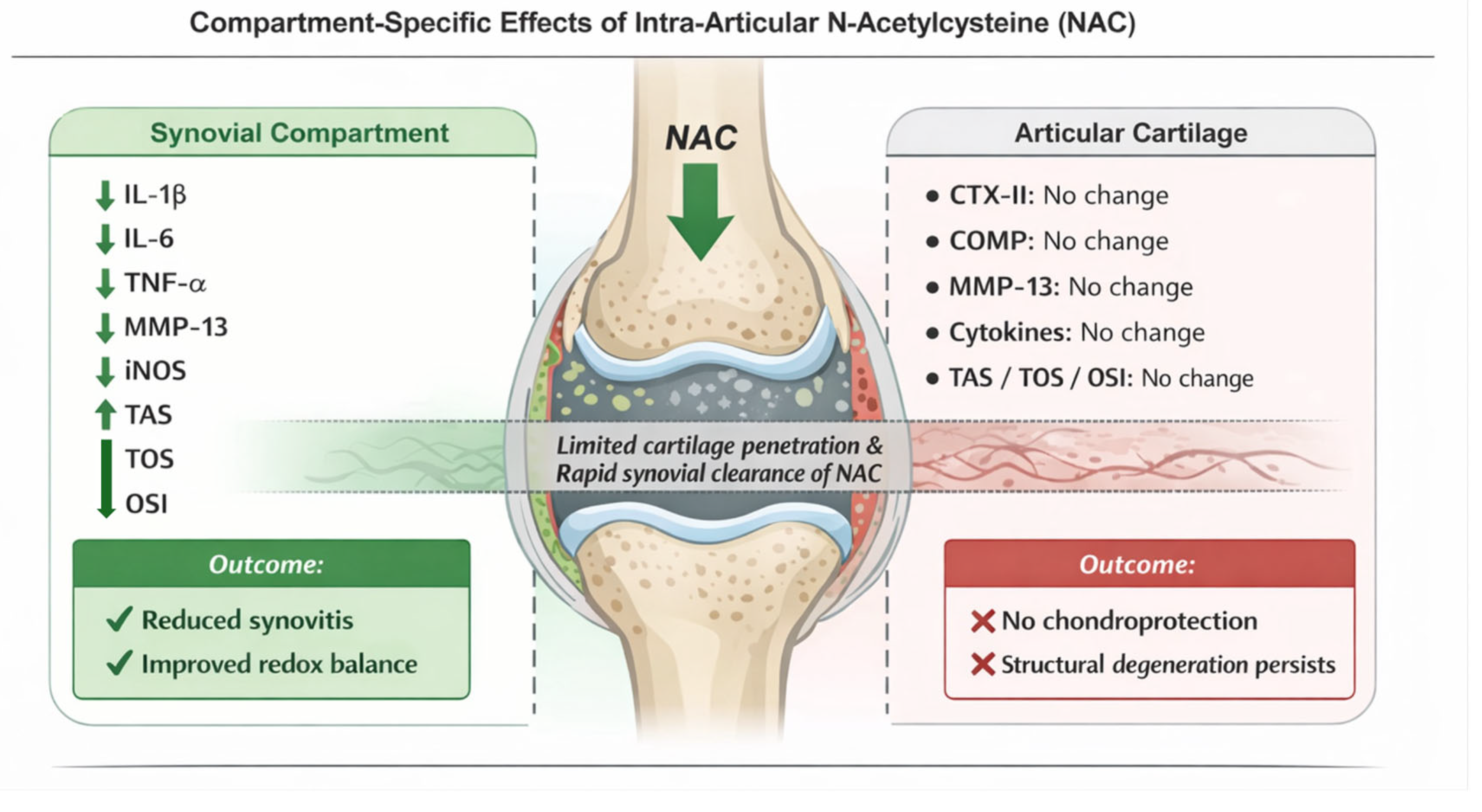
3.2. Synovial Fluid Biomarker Profiles
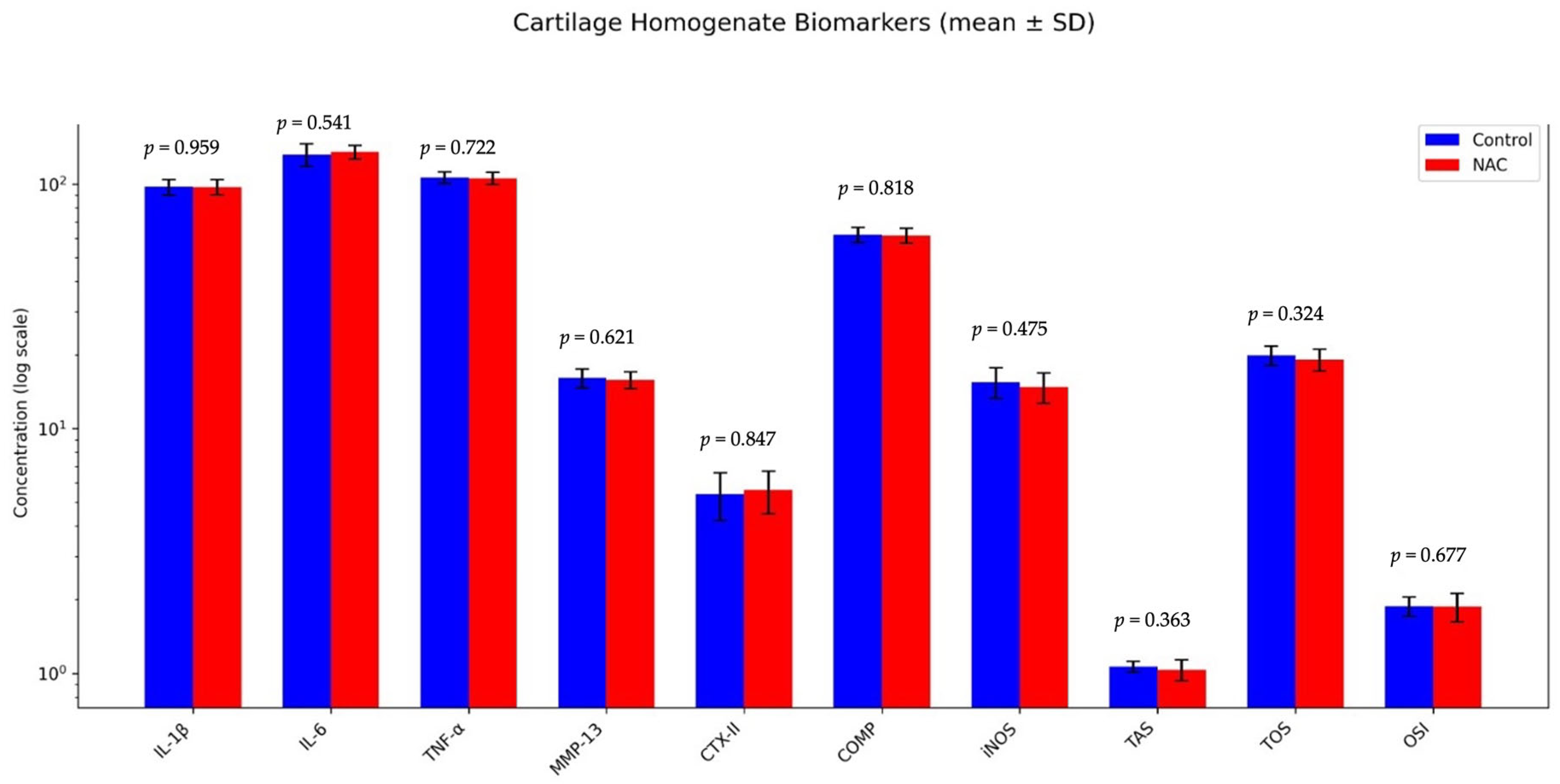
3.3. Cartilage Biochemical Profiles
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| OA | Osteoarthritis |
| KOA | Knee Osteoarthritis |
| ACLT | Anterior Cruciate Ligament Transection |
| NAC | N-acetylcysteine |
| IA | Intra-articular |
| PBS | Phosphate-Buffered Saline |
| H&E | Hematoxylin and Eosin |
| ROS | Reactive Oxygen Species |
| NO | Nitric Oxide |
| iNOS | Inducible Nitric Oxide Synthase |
| IL | Interleukin |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| TNF-α | Tumor Necrosis Factor-alpha |
| MMP-13 | Matrix Metalloproteinase-13 |
| COMP | Cartilage Oligomeric Matrix Protein |
| CTX-II | C-terminal Cross-linked Telopeptides of Type II Collagen |
| SD | Standard Deviation |
| ELISA | Enzyme-Linked Immunosorbent Assay |
References
- Ansari, M.Y.; Ahmad, N.; Haqqi, T.M. Oxidative stress and inflammation in osteoarthritis pathogenesis: Role of polyphenols. Biomed. Pharmacother. 2020, 129, 110452. [Google Scholar] [CrossRef]
- Bennell, K.L.; Hunter, D.J.; Hinman, R.S. Management of osteoarthritis of the knee. BMJ 2012, 345, e4934. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Lee, K.; Ju, J.H. Recent updates of diagnosis, pathophysiology, and treatment on osteoarthritis of the knee. Int. J. Mol. Sci. 2021, 22, 2619. [Google Scholar] [CrossRef]
- Aw, N.M.-Y.; Yeo, S.-J.; Wylde, V.; Wong, S.B.; Chan, D.; Thumboo, J.; Leung, Y.Y. Impact of pain sensitisation on the quality of life of patients with knee osteoarthritis. RMD Open 2022, 8, e001938. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.; van der Esch, M.; Hinman, R.S.; Peat, G.; de Zwart, A.; Quicke, J.; Runhaar, J.; Knoop, J.; van der Leeden, M.; de Rooij, M. How does hip osteoarthritis differ from knee osteoarthritis? Osteoarthr. Cartil. 2022, 30, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Giorgino, R.; Albano, D.; Fusco, S.; Peretti, G.M.; Mangiavini, L.; Messina, C. Knee osteoarthritis: Epidemiology, pathogenesis, and mesenchymal stem cells: What else is new? An update. Int. J. Mol. Sci. 2023, 24, 6405. [Google Scholar] [CrossRef]
- Foster, N.; Eriksson, L.; Deveza, L.; Hall, M. Osteoarthritis year in review 2022: Epidemiology & therapy. Osteoarthr. Cartil. 2023, 31, 876–883. [Google Scholar] [CrossRef]
- Loeser, R.F.; Goldring, S.R.; Scanzello, C.R.; Goldring, M.B. Osteoarthritis: A disease of the joint as an organ. Arthritis Rheum. 2012, 64, 1697. [Google Scholar] [CrossRef]
- Berenbaum, F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr. Cartil. 2013, 21, 16–21. [Google Scholar] [CrossRef]
- Clockaerts, S.; Bastiaansen-Jenniskens, Y.M.; Runhaar, J.; Van Osch, G.J.; Van Offel, J.; Verhaar, J.; De Clerck, L.; Somville, J. The infrapatellar fat pad should be considered as an active osteoarthritic joint tissue: A narrative review. Osteoarthr. Cartil. 2010, 18, 876–882. [Google Scholar] [CrossRef]
- Klein-Wieringa, I.; Kloppenburg, M.; Bastiaansen-Jenniskens, Y.; Yusuf, E.; Kwekkeboom, J.; El-Bannoudi, H.; Nelissen, R.; Zuurmond, A.; Stojanovic-Susulic, V.; Van Osch, G.; et al. The infrapatellar fat pad of patients with osteoarthritis has an inflammatory phenotype. Ann. Rheum. Dis. 2011, 70, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.E.; Miller, R.J.; Malfait, A.-M. Osteoarthritis joint pain: The cytokine connection. Cytokine 2014, 70, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.-P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Rose, B.J.; Kooyman, D.L. A tale of two joints: The role of matrix metalloproteases in cartilage biology. Dis. Markers 2016, 2016, 4895050. [Google Scholar] [CrossRef]
- Díaz-Gallego, L.; Prieto, J.G.; Coronel, P.; Gamazo, L.E.; Gimeno, M.; Alvarez, A.I. Apoptosis and nitric oxide in an experimental model of osteoarthritis in rabbit after hyaluronic acid treatment. J. Orthop. Res. 2005, 23, 1370–1376. [Google Scholar] [CrossRef]
- Wu, G.J.; Chen, T.G.; Chang, H.C.; Chiu, W.T.; Chang, C.C.; Chen, R.M. Nitric oxide from both exogenous and endogenous sources activates mitochondria-dependent events and induces insults to human chondrocytes. J. Cell. Biochem. 2007, 101, 1520–1531. [Google Scholar] [CrossRef]
- Han, S. Osteoarthritis year in review 2022: Biology. Osteoarthr. Cartil. 2022, 30, 1575–1582. [Google Scholar] [CrossRef]
- Sokolove, J.; Lepus, C.M. Role of inflammation in the pathogenesis of osteoarthritis: Latest findings and interpretations. Ther. Adv. Musculoskelet. Dis. 2013, 5, 77–94. [Google Scholar] [CrossRef]
- Heinegård, D.; Saxne, T. The role of the cartilage matrix in osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 50–56. [Google Scholar] [CrossRef]
- Homandberg, G.A. Cartilage damage by matrix degradation products: Fibronectin fragments. Clin. Orthop. Relat. Res. 2001, 391, S100–S107. [Google Scholar] [CrossRef]
- Scanzello, C.R.; Plaas, A.; Crow, M.K. Innate immune system activation in osteoarthritis: Is osteoarthritis a chronic wound? Curr. Opin. Rheumatol. 2008, 20, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Liu-Bryan, R.; Terkeltaub, R. Emerging regulators of the inflammatory process in osteoarthritis. Nat. Rev. Rheumatol. 2015, 11, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Kanda, V.; Bush-Joseph, C.; Verma, N.; Chubinskaya, S.; Mikecz, K.; Glant, T.T.; Malfait, A.M.; Crow, M.K.; Spear, G.T.; et al. Synovial fluid from patients with early osteoarthritis modulates fibroblast-like synoviocyte responses to toll-like receptor 4 and toll-like receptor 2 ligands via soluble CD14. Arthritis Rheum. 2012, 64, 2268–2277. [Google Scholar] [CrossRef] [PubMed]
- Henrotin, Y.; Kurz, B.; Aigner, T. Oxygen and reactive oxygen species in cartilage degradation: Friends or foes? Osteoarthr. Cartil. 2005, 13, 643–654. [Google Scholar] [CrossRef]
- Zhang, Y.; Jordan, J.M. Epidemiology of osteoarthritis. Clin. Geriatr. Med. 2010, 26, 355. [Google Scholar] [CrossRef]
- Brandl, A.; Hartmann, A.; Bechmann, V.; Graf, B.; Nerlich, M.; Angele, P. Oxidative stress induces senescence in chondrocytes. J. Orthop. Res. 2011, 29, 1114–1120. [Google Scholar] [CrossRef]
- Bolduc, J.A.; Collins, J.A.; Loeser, R.F. Reactive oxygen species, aging and articular cartilage homeostasis. Free Radic. Biol. Med. 2019, 132, 73–82. [Google Scholar] [CrossRef]
- Yudoh, K.; van Trieu, N.; Nakamura, H.; Hongo-Masuko, K.; Kato, T.; Nishioka, K. Potential involvement of oxidative stress in cartilage senescence and development of osteoarthritis: Oxidative stress induces chondrocyte telomere instability and downregulation of chondrocyte function. Arthritis Res. Ther. 2005, 7, R380. [Google Scholar] [CrossRef]
- Samuels, J.; Pillinger, M.; Jevsevar, D.; Felson, D.; Simon, L. Critical appraisal of intra-articular glucocorticoid injections for symptomatic osteoarthritis of the knee. Osteoarthr. Cartil. 2021, 29, 8–16. [Google Scholar] [CrossRef]
- Primorac, D.; Molnar, V.; Rod, E.; Jeleč, Ž.; Čukelj, F.; Matišić, V.; Vrdoljak, T.; Hudetz, D.; Hajsok, H.; Borić, I. Knee osteoarthritis: A review of pathogenesis and state-of-the-art non-operative therapeutic considerations. Genes 2020, 11, 854. [Google Scholar] [CrossRef]
- Lockwood, K.A.; Chu, B.T.; Anderson, M.J.; Haudenschild, D.R.; Christiansen, B.A. Comparison of loading rate-dependent injury modes in a murine model of post-traumatic osteoarthritis. J. Orthop. Res. 2014, 32, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.H.; Kraus, V.B.; Setton, L.A. Progress in intra-articular therapy. Nat. Rev. Rheumatol. 2014, 10, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Kuo, S.; Lin, L.; Yang, Y. The efficacy of N-acetylcysteine in chronic obstructive pulmonary disease patients: A meta-analysis. Ther. Adv. Respir. Dis. 2023, 17, 17534666231158563. [Google Scholar] [CrossRef] [PubMed]
- Nikbaf-Shandiz, M.; Adeli, S.; Faghfouri, A.H.; Khademi, F.; Jamilian, P.; Zarezadeh, M.; Ebrahimi-Mamaghani, M. The efficacy of N-acetylcysteine in improving liver function: A systematic review and meta-analysis of controlled clinical trials. PharmaNutrition 2023, 24, 100343. [Google Scholar] [CrossRef]
- Tenório, M.; Graciliano, N.G.; Moura, F.A.; Oliveira, A.C.M.; Goulart, M.O.F. N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants 2021, 10, 967. [Google Scholar] [CrossRef]
- Nakagawa, S.; Arai, Y.; Mazda, O.; Kishida, T.; Takahashi, K.A.; Sakao, K.; Saito, M.; Honjo, K.; Imanishi, J.; Kubo, T. N-acetylcysteine prevents nitric oxide-induced chondrocyte apoptosis and cartilage degeneration in an experimental model of osteoarthritis. J. Orthop. Res. 2010, 28, 156–163. [Google Scholar] [CrossRef]
- Kaneko, Y.; Tanigawa, N.; Sato, Y.; Kobayashi, T.; Nakamura, S.; Ito, E.; Soma, T.; Miyamoto, K.; Kobayashi, S.; Harato, K. Oral administration of N-acetyl cysteine prevents osteoarthritis development and progression in a rat model. Sci. Rep. 2019, 9, 18741. [Google Scholar] [CrossRef]
- Riegger, J.; Leucht, F.; Palm, H.-G.; Ignatius, A.; Brenner, R.E. Initial harm reduction by N-acetylcysteine alleviates cartilage degeneration after blunt single-impact cartilage trauma in vivo. Int. J. Mol. Sci. 2019, 20, 2916. [Google Scholar] [CrossRef]
- Roman-Blas, J.A.; Contreras-Blasco, M.A.; Largo, R.; Alvarez-Soria, M.A.; Castaneda, S.; Herrero-Beaumont, G. Differential effects of the antioxidant N-acetylcysteine on the production of catabolic mediators in IL-1β-stimulated human osteoarthritic synoviocytes and chondrocytes. Eur. J. Pharmacol. 2009, 623, 125–131. [Google Scholar] [CrossRef]
- Yeh, Y.-T.; Liang, C.-C.; Chang, C.-L.; Hsu, C.-Y.; Li, P.-C. Increased risk of knee osteoarthritis in patients using oral N-acetylcysteine: A nationwide cohort study. BMC Musculoskelet. Disord. 2020, 21, 531. [Google Scholar] [CrossRef]
- Samuni, Y.; Goldstein, S.; Dean, O.M.; Berk, M. The chemistry and biological activities of N-acetylcysteine. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2013, 1830, 4117–4129. [Google Scholar] [CrossRef]
- Atkuri, K.R.; Mantovani, J.J.; Herzenberg, L.A.; Herzenberg, L.A. N-Acetylcysteine—A safe antidote for cysteine/glutathione deficiency. Curr. Opin. Pharmacol. 2007, 7, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Rushworth, G.F.; Megson, I.L. Existing and potential therapeutic uses for N-acetylcysteine: The need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol. Ther. 2014, 141, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Gerwin, N.; Hops, C.; Lucke, A. Intraarticular drug delivery in osteoarthritis. Adv. Drug Deliv. Rev. 2006, 58, 226–242. [Google Scholar] [CrossRef] [PubMed]
- Jones, I.A.; Togashi, R.; Wilson, M.L.; Heckmann, N.; Vangsness, C.T., Jr. Intra-articular treatment options for knee osteoarthritis. Nat. Rev. Rheumatol. 2019, 15, 77–90. [Google Scholar] [CrossRef]
- Verhave, P.; van Eenige, R.; Tiebosch, I. Methods for applying blinding and randomisation in animal experiments. Lab. Anim. 2024, 58, 419–426. [Google Scholar] [CrossRef]
- Hirst, J.A.; Howick, J.; Aronson, J.K.; Roberts, N.; Perera, R.; Koshiaris, C.; Heneghan, C. The need for randomization in animal trials: An overview of systematic reviews. PLoS ONE 2014, 9, e98856. [Google Scholar] [CrossRef]
- Høegh-Andersen, P.; Tankó, L.B.; Andersen, T.L.; Lundberg, C.V.; Mo, J.A.; Heegaard, A.-M.; Delaissé, J.-M.; Christgau, S. Ovariectomized rats as a model of postmenopausal osteoarthritis: Validation and application. Arthritis Res. Ther. 2004, 6, R169. [Google Scholar] [CrossRef]
- Sniekers, Y.; Weinans, H.; Bierma-Zeinstra, S.; Van Leeuwen, J.; Van Osch, G. Animal models for osteoarthritis: The effect of ovariectomy and estrogen treatment–a systematic approach. Osteoarthr. Cartil. 2008, 16, 533–541. [Google Scholar] [CrossRef]
- Yang, Z.; Tan, Q.; Zhao, Z.; Niu, G.; Li, S.; Li, W.; Song, C.; Leng, H. Distinct pathological changes of osteochondral units in early OVX-OA involving TGF-β signaling. Front. Endocrinol. 2022, 13, 1074176. [Google Scholar] [CrossRef]
- Bapat, S.; Hubbard, D.; Munjal, A.; Hunter, M.; Fulzele, S. Pros and cons of mouse models for studying osteoarthritis. Clin. Transl. Med. 2018, 7, 36. [Google Scholar] [CrossRef]
- Lampropoulou-Adamidou, K.; Lelovas, P.; Karadimas, E.V.; Liakou, C.; Triantafillopoulos, I.K.; Dontas, I.; Papaioannou, N.A. Useful animal models for the research of osteoarthritis. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, A.; Eftimov, M.; Todorova, M.; Kuzmanova, V.; Kuzmanov, A.; Kuzmanov, K.; Vlaskovska, M.; Valcheva-Kuzmanova, S. Effects of ovariectomy-induced estrogen deficit on rat behaviour, lipid metabolism, inflammation, bone mineral density, and turnover. Folia Medica 2021, 63, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Kamekura, S.; Hoshi, K.; Shimoaka, T.; Chung, U.; Chikuda, H.; Yamada, T.; Uchida, M.; Ogata, N.; Seichi, A.; Nakamura, K. Osteoarthritis development in novel experimental mouse models induced by knee joint instability. Osteoarthr. Cartil. 2005, 13, 632–641. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-F.; Wang, J.-F.; Zhang, G. Anterior cruciate ligament transection (ACLT)-induced osteoarthritis in rats. In A Practical Manual For Musculoskeletal Research; World Scientific: Singapore, 2008; pp. 559–568. [Google Scholar]
- Oh, S.S.; Narver, H.L. Mouse and rat anesthesia and analgesia. Curr. Protoc. 2024, 4, e995. [Google Scholar] [CrossRef]
- Glasson, S.; Blanchet, T.; Morris, E. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthr. Cartil. 2007, 15, 1061–1069. [Google Scholar] [CrossRef]
- Little, C.B.; Hunter, D.J. Post-traumatic osteoarthritis: From mouse models to clinical trials. Nat. Rev. Rheumatol. 2013, 9, 485–497. [Google Scholar] [CrossRef]
- Hayami, T.; Pickarski, M.; Wesolowski, G.A.; Mclane, J.; Bone, A.; Destefano, J.; Rodan, G.A.; Duong, L.T. The role of subchondral bone remodeling in osteoarthritis: Reduction of cartilage degeneration and prevention of osteophyte formation by alendronate in the rat anterior cruciate ligament transection model. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2004, 50, 1193–1206. [Google Scholar] [CrossRef]
- Hayami, T.; Pickarski, M.; Zhuo, Y.; Wesolowski, G.A.; Rodan, G.A.; Duong, L.T. Characterization of articular cartilage and subchondral bone changes in the rat anterior cruciate ligament transection and meniscectomized models of osteoarthritis. Bone 2006, 38, 234–243. [Google Scholar] [CrossRef]
- Little, C.B.; Smith, M.M. Animal models of osteoarthritis. Curr. Rheumatol. Rev. 2008, 4, 175–182. [Google Scholar] [CrossRef]
- Teeple, E.; Jay, G.D.; Elsaid, K.A.; Fleming, B.C. Animal models of osteoarthritis: Challenges of model selection and analysis. AAPS J. 2013, 15, 438–446. [Google Scholar] [CrossRef]
- Kuyinu, E.L.; Narayanan, G.; Nair, L.S.; Laurencin, C.T. Animal models of osteoarthritis: Classification, update, and measurement of outcomes. J. Orthop. Surg. Res. 2016, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Basting, R.T.; Napimoga, M.H.; De Lima, J.M.; De Freitas, N.S.; Clemente-Napimoga, J.T. Fast and accurate protocol for histology and immunohistochemistry reactions in temporomandibular joint of rats. Arch. Oral Biol. 2021, 126, 105115. [Google Scholar] [CrossRef] [PubMed]
- Bogoevski, K.; Woloszyk, A.; Blackwood, K.; Woodruff, M.A.; Glatt, V. Tissue morphology and antigenicity in mouse and rat tibia: Comparing 12 different decalcification conditions. J. Histochem. Cytochem. 2019, 67, 545–561. [Google Scholar] [CrossRef] [PubMed]
- Savi, F.M.; Brierly, G.I.; Baldwin, J.; Theodoropoulos, C.; Woodruff, M.A. Comparison of different decalcification methods using rat mandibles as a model. J. Histochem. Cytochem. 2017, 65, 705–722. [Google Scholar] [CrossRef]
- Manyu, G.; Waithaka, P. End-Point Determination In Acid Decalcification Of Bone: A New Novel Method. Int. J. Sci. Res. Publ. 2024, 14, 25–27. [Google Scholar] [CrossRef]
- Wehrle, E.; Günther, D.; Mathavan, N.; Singh, A.; Müller, R. Protocol for preparing formalin-fixed paraffin-embedded musculoskeletal tissue samples from mice for spatial transcriptomics. STAR Protoc. 2024, 5, 102986. [Google Scholar] [CrossRef]
- Slaoui, M.; Fiette, L. Histopathology procedures: From tissue sampling to histopathological evaluation. In Drug Safety Evaluation: Methods and Protocols; Springer: Berlin/Heidelberg, Germany, 2010; pp. 69–82. [Google Scholar]
- Fischer, A.H.; Jacobson, K.A.; Rose, J.; Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harb. Protoc. 2008, 2008, pdb-prot4986. [Google Scholar] [CrossRef]
- Wall, A.; Board, T. Chemical basis for the histological use of safranin O in the study of articular cartilage. In Classic Papers in Orthopaedics; Springer: Berlin/Heidelberg, Germany, 2013; pp. 433–435. [Google Scholar]
- Moody, H.R.; Heard, B.J.; Frank, C.B.; Shrive, N.G.; Oloyede, A.O. Investigating the potential value of individual parameters of histological grading systems in a sheep model of cartilage damage: The Modified Mankin method. J. Anat. 2012, 221, 47–54. [Google Scholar] [CrossRef]
- Barton, N.J.; Stevens, D.A.; Hughes, J.P.; Rossi, A.G.; Chessell, I.P.; Reeve, A.J.; McQueen, D.S. Demonstration of a novel technique to quantitatively assess inflammatory mediators and cells in rat knee joints. J. Inflamm. 2007, 4, 13. [Google Scholar] [CrossRef]
- Núñez-Carro, C.; Blanco-Blanco, M.; Montoya, T.; Villagrán-Andrade, K.M.; Hermida-Gómez, T.; Blanco, F.J.; De Andrés, M.C. Histone extraction from human articular cartilage for the study of epigenetic regulation in osteoarthritis. Int. J. Mol. Sci. 2022, 23, 3355. [Google Scholar] [CrossRef] [PubMed]
- Bundgaard, L.; Åhrman, E.; Malmström, J.; auf dem Keller, U.; Walters, M.; Jacobsen, S. Effective protein extraction combined with data independent acquisition analysis reveals a comprehensive and quantifiable insight into the proteomes of articular cartilage and subchondral bone. Osteoarthr. Cartil. 2022, 30, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Kruger, N.J. The Bradford method for protein quantitation. In The Protein Protocols Handbook; Humana Press: Totowa, NJ, USA, 2009; pp. 17–24. [Google Scholar]
- Aydin, S.; Emre, E.; Ugur, K.; Aydin, M.A.; Sahin, İ.; Cinar, V.; Akbulut, T. An overview of ELISA: A review and update on best laboratory practices for quantifying peptides and proteins in biological fluids. J. Int. Med. Res. 2025, 53, 03000605251315913. [Google Scholar] [CrossRef] [PubMed]
- Alhajj, M.; Zubair, M.; Farhana, A. Enzyme Linked Immunosorbent Assay; StatPearls: Tampa, FL, USA, 2023. [Google Scholar]
- Erel, O. A novel automated method to measure total antioxidant response against potent free radical reactions. Clin. Biochem. 2004, 37, 112–119. [Google Scholar] [CrossRef]
- Erel, O. A new automated colorimetric method for measuring total oxidant status. Clin. Biochem. 2005, 38, 1103–1111. [Google Scholar] [CrossRef]
- Kim, J.E.; Song, D.-h.; Kim, S.H.; Jung, Y.; Kim, S.J. Development and characterization of various osteoarthritis models for tissue engineering. PLoS ONE 2018, 13, e0194288. [Google Scholar] [CrossRef]
- Namhong, S.; Wongdee, K.; Suntornsaratoon, P.; Teerapornpuntakit, J.; Hemstapat, R.; Charoenphandhu, N. Knee osteoarthritis in young growing rats is associated with widespread osteopenia and impaired bone mineralization. Sci. Rep. 2020, 10, 15079. [Google Scholar] [CrossRef]
- Gigout, A.; Harazin, D.; Topping, L.M.; Merciris, D.; Lindemann, S.; Brenneis, C.; Nissim, A. Early detection of osteoarthritis in the rat with an antibody specific to type II collagen modified by reactive oxygen species. Arthritis Res. Ther. 2021, 23, 113. [Google Scholar] [CrossRef]
- Pickarski, M.; Hayami, T.; Zhuo, Y.; Duong, L.T. Molecular changes in articular cartilage and subchondral bone in the rat anterior cruciate ligament transection and meniscectomized models of osteoarthritis. BMC Musculoskelet. Disord. 2011, 12, 197. [Google Scholar] [CrossRef]
- Ozcamdalli, M.; Misir, A.; Kizkapan, T.B.; Uzun, E.; Duygulu, F.; Yazici, C.; Kafadar, I.H. Comparison of intra-articular injection of hyaluronic acid and N-acetyl cysteine in the treatment of knee osteoarthritis: A pilot study. Cartilage 2017, 8, 384–390. [Google Scholar] [CrossRef]
- Özdemir, M.; Birinci, B.; Haberal, B.; Atılgan, A.O.; Demirkale, I. In vivo study of the role of hyaluronic acid, N-acetyl cysteine, and deproteinized calf serum on injury-induced cartilage degeneration. Jt. Dis. Relat. Surg. 2022, 34, 158. [Google Scholar] [CrossRef] [PubMed]
- Dycus, D.L.; Au, A.Y.; Grzanna, M.W.; Wardlaw, J.L.; Frondoza, C.G. Modulation of inflammation and oxidative stress in canine chondrocytes. Am. J. Vet. Res. 2013, 74, 983–989. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morici, L.; Allémann, E.; Rodríguez-Nogales, C.; Jordan, O. Cartilage-targeted drug nanocarriers for osteoarthritis therapy. Int. J. Pharm. 2024, 666, 124843. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Ma, Y.; Tao, Y.; Lin, W.; Wang, P. Intra-articular drug delivery for osteoarthritis treatment. Pharmaceutics 2021, 13, 2166. [Google Scholar] [CrossRef]
- Huang, H.; Lou, Z.; Zheng, S.; Wu, J.; Yao, Q.; Chen, R.; Kou, L.; Chen, D. Intra-articular drug delivery systems for osteoarthritis therapy: Shifting from sustained release to enhancing penetration into cartilage. Drug Deliv. 2022, 29, 767–791. [Google Scholar] [CrossRef]
- Ferrell, W.R.; Najafipour, H. Changes in synovial PO2 and blood flow in the rabbit knee joint due to stimulation of the posterior articular nerve. J. Physiol. 1992, 449, 607–617. [Google Scholar] [CrossRef]
- Frede, S.; Berchner-Pfannschmidt, U.; Fandrey, J. Regulation of hypoxia-inducible factors during inflammation. Methods Enzymol. 2007, 435, 403–419. [Google Scholar]
- Görlach, A.; Kietzmann, T. Superoxide and derived reactive oxygen species in the regulation of hypoxia-inducible factors. Methods Enzymol. 2007, 435, 421–446. [Google Scholar]
- Fellows, C.R.; Matta, C.; Mobasheri, A. Applying proteomics to study crosstalk at the cartilage-subchondral bone interface in osteoarthritis: Current status and future directions. eBioMedicine 2016, 11, 2–4. [Google Scholar] [CrossRef]
- Zhou, X.; Cao, H.; Yuan, Y.; Wu, W. Biochemical signals mediate the crosstalk between cartilage and bone in osteoarthritis. BioMed Res. Int. 2020, 2020, 5720360. [Google Scholar] [CrossRef]
- Chou, C.-H.; Jain, V.; Gibson, J.; Attarian, D.E.; Haraden, C.A.; Yohn, C.B.; Laberge, R.-M.; Gregory, S.; Kraus, V.B. Synovial cell cross-talk with cartilage plays a major role in the pathogenesis of osteoarthritis. Sci. Rep. 2020, 10, 10868. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Wang, K.; Ding, S.; Zhang, M. Cross-talk of inflammation and cellular senescence: A new insight into the occurrence and progression of osteoarthritis. Bone Res. 2024, 12, 69. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zhu, Y.; Xiao, W.; Hu, Y.; Li, Y. Instability and excessive mechanical loading mediate subchondral bone changes to induce osteoarthritis. Ann. Transl. Med. 2020, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Buckwalter, J.A.; Anderson, D.D.; Brown, T.D.; Tochigi, Y.; Martin, J.A. The roles of mechanical stresses in the pathogenesis of osteoarthritis: Implications for treatment of joint injuries. Cartilage 2013, 4, 286–294. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, X.; Wang, S.; Jing, Y.; Su, J. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 2021, 9, 20. [Google Scholar] [CrossRef]
- van der Kraan, P.M.; van den Berg, W.B. Osteoarthritis in the context of ageing and evolution: Loss of chondrocyte differentiation block during ageing. Ageing Res. Rev. 2008, 7, 106–113. [Google Scholar] [CrossRef]
- Luyten, F.; Bierma-Zeinstra, S.; Dell’Accio, F.; Kraus, V.; Nakata, K.; Sekiya, I.; Arden, N.; Lohmander, L. Toward classification criteria for early osteoarthritis of the knee. In Seminars in Arthritis and Rheumatism; WB Saunders: Philadelphia, PA, USA, 2018; pp. 457–463. [Google Scholar]
- Xia, B.; Chen, D.; Zhang, J.; Hu, S.; Jin, H.; Tong, P. Osteoarthritis pathogenesis: A review of molecular mechanisms. Calcif. Tissue Int. 2014, 95, 495–505. [Google Scholar] [CrossRef]
- Vincent, T.L.; Alliston, T.; Kapoor, M.; Loeser, R.F.; Troeberg, L.; Little, C.B. Osteoarthritis pathophysiology: Therapeutic target discovery may require a multifaceted approach. Clin. Geriatr. Med. 2022, 38, 193–219. [Google Scholar] [CrossRef]
- Bijlsma, J.W.; Berenbaum, F.; Lafeber, F.P. Osteoarthritis: An update with relevance for clinical practice. Lancet 2011, 377, 2115–2126. [Google Scholar] [CrossRef]
- Mobasheri, A.; Rayman, M.P.; Gualillo, O.; Sellam, J.; Van Der Kraan, P.; Fearon, U. The role of metabolism in the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 2017, 13, 302–311. [Google Scholar] [CrossRef]
- Kraus, V.B.; Blanco, F.J.; Englund, M.; Karsdal, M.A.; Lohmander, L.S. Call for standardized definitions of osteoarthritis and risk stratification for clinical trials and clinical use. Osteoarthr. Cartil. 2015, 23, 1233–1241. [Google Scholar] [CrossRef]
| Marker | Sensitivity | Detection Range |
|---|---|---|
| IL-1β | 18.75 pg/mL | 31.25–2000 pg/mL |
| IL-6 | 7.5 pg/mL | 12.5–800 pg/mL |
| TNF-α | 9.38 pg/mL | 15.63–1000 pg/mL |
| MMP-13 | 0.19 ng/mL | 0.31–20 ng/mL |
| CTX-II | 0.1 ng/mL | 0.16–10 ng/mL |
| COMP | 0.94 ng/mL | 1.56–100 ng/mL |
| iNOS | 0.19 ng/mL | 0.31–20 ng/mL |
| ACLT-Operated Control Group | NAC Group | p Value | Effect Size | |
|---|---|---|---|---|
| Structure | 5.0 (4–6) (5.0 ± 0.82) | 5.0 (4–6) (4.9 ± 0.74) | 0.776 * | 0.06 |
| Cellularity | 2.0 (1–3) (2.3 ± 0.67) | 2.0 (2–3) (2.1 ± 0.32) | 0.322 * | 0.22 |
| Safronin-O staining | 3.0 (2–4) (3.0 ± 0.82) | 3.0 (2–4) (2.7 ± 0.67) | 0.392 * | 0.19 |
| Tidemark | 1.0 (0–1) (0.8 ± 0.42) | 1.0 (0–1) (0.6 ± 0.52) | 0.342 * | 0.21 |
| Total | 11.0 (8–14) (11.1 ± 1.97) | 11.0 (8–13) (10.3 ± 1.57) | 0.328 ** | 0.45 |
| Synovial Fluid | ACLT-Operated Control Group | NAC Group | p Value | Effect Size |
|---|---|---|---|---|
| IL-1 (pg/mL) | 81.7 (77.5–88.1) (82.2 ± 3.50) | 41.9 (38.7–44.1) (41.7 ± 1.92) | <0.001 * | 0.85 |
| IL-6 (pg/mL) | 131.8 (127.5–140.1) (132.6 ± 3.97) | 61.8 (58.7–64.3) (61.7 ± 1.86) | <0.001 * | 0.85 |
| TNF-α (pg/mL) | 65.7 (62.5–70.1) (66.2 ± 2.55) | 31.2 (29.3–34.1) (31.6 ± 1.70) | <0.001 * | 0.85 |
| MMP-13 (ng/mL) | 17.8 (15.2–21.4) (17.9 ± 1.87) | 8.7 (6.6–10.9) (8.7 ± 1.31) | <0.001 * | 0.85 |
| CTX-II (ng/mL) | 5.5 (3.4–7.7) (5.5 ± 1.20) | 5.4 (3.9–7.0) (5.4 ± 1.05) | 0.859 ** | 0.08 |
| COMP (ng/mL) | 60.0 (52.8–68.3) (60.5 ± 5.01) | 60.8 (54.7–67.8) (60.8 ± 4.67) | 0.888 ** | 0.06 |
| iNOS (ng/mL) | 22.1 (18.5–24.9) (22.0 ± 2.06) | 13.5 (11.1–16.5) (13.5 ± 1.84) | <0.001 ** | 4.36 |
| TAS (mmol Trolox/L) | 0.674 (0.54–0.79) (0.674 ± 0.079) | 1.098 (1.01–1.20) (1.098 ± 0.060) | <0.001 * | 0.85 |
| TOS (µmol H2O2/L) | 27.03 (23.1–32.6) (27.03 ± 3.12) | 17.37 (15.1–19.7) (17.37 ± 1.70) | <0.001 * | 0.85 |
| OSI (arbitrary unit) | 4.08 (3.31–6.04) (4.08 ± 0.85) | 1.59 (1.34–1.88) (1.59 ± 0.20) | <0.001 * | 0.85 |
| Cartilage Homogenate | ACLT-Operated Control Group | NAC Group | p Value | Effect Size |
|---|---|---|---|---|
| IL-1 (pg/mL) | 96.9 (88.5–110.1) (97.2 ± 7.23) | 96.7 (87.3–108.9) (97.1 ± 6.97) | 0.959 ** | 0.02 |
| IL-6 (pg/mL) | 136.4 (102.8–150.2) (131.9 ± 13.9) | 136.8 (119.6–148.9) (135.2 ± 8.85) | 0.541 ** | 0.28 |
| TNF-α (pg/mL) | 106.4 (98.3–115.7) (106.4 ± 5.74) | 105.6 (97.5–116.2) (105.5 ± 5.91) | 0.722 ** | 0.16 |
| MMP-13 (ng/mL) | 16.2 (13.9–18.5) (16.1 ± 1.42) | 15.9 (14.2–18.2) (15.8 ± 1.26) | 0.621 ** | 0.23 |
| CTX-II (ng/mL) | 5.4 (4.1–7.4) (5.4 ± 1.19) | 5.6 (3.9–7.3) (5.6 ± 1.11) | 0.847 ** | 0.09 |
| COMP (ng/mL) | 61.6 (56.3–70.1) (62.0 ± 4.55) | 61.3 (55.4–68.4) (61.6 ± 4.35) | 0.818 ** | 0.11 |
| iNOS (ng/mL) | 15.4 (12.1–19.0) (15.5 ± 2.21) | 14.8 (12.1–18.2) (14.8 ± 2.08) | 0.475 ** | 0.33 |
| TAS (mmol Trolox/L) | 1.064 (0.99–1.15) (1.064 ± 0.054) | 1.033 (0.92–1.20) (1.033 ± 0.101) | 0.363 * | 0.20 |
| TOS (µmol H2O2/L) | 19.94 (16.9–22.0) (19.94 ± 1.82) | 19.16 (16.7–22.1) (19.16 ± 1.98) | 0.324 * | 0.22 |
| OSI (arbitrary unit) | 1.88 (1.61–2.14) (1.88 ± 0.17) | 1.87 (1.39–2.13) (1.87 ± 0.25) | 0.677 * | 0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Dinç, M.; Bayrak, H.Ç.; Karasu, R.; Aykaç, B.; Soydemir, Ö.C.; Saricetin, A. Intra-Articular N-Acetylcysteine Reduces Synovitis Without Preventing Cartilage Degeneration in Experimental Osteoarthritis. Biomedicines 2026, 14, 86. https://doi.org/10.3390/biomedicines14010086
Dinç M, Bayrak HÇ, Karasu R, Aykaç B, Soydemir ÖC, Saricetin A. Intra-Articular N-Acetylcysteine Reduces Synovitis Without Preventing Cartilage Degeneration in Experimental Osteoarthritis. Biomedicines. 2026; 14(1):86. https://doi.org/10.3390/biomedicines14010086
Chicago/Turabian StyleDinç, Mustafa, Hünkar Çağdaş Bayrak, Recep Karasu, Bilal Aykaç, Ömer Cevdet Soydemir, and Aysun Saricetin. 2026. "Intra-Articular N-Acetylcysteine Reduces Synovitis Without Preventing Cartilage Degeneration in Experimental Osteoarthritis" Biomedicines 14, no. 1: 86. https://doi.org/10.3390/biomedicines14010086
APA StyleDinç, M., Bayrak, H. Ç., Karasu, R., Aykaç, B., Soydemir, Ö. C., & Saricetin, A. (2026). Intra-Articular N-Acetylcysteine Reduces Synovitis Without Preventing Cartilage Degeneration in Experimental Osteoarthritis. Biomedicines, 14(1), 86. https://doi.org/10.3390/biomedicines14010086






