Roots of Progress: Uncovering Cerebellar Ataxias Using iPSC Models
Abstract
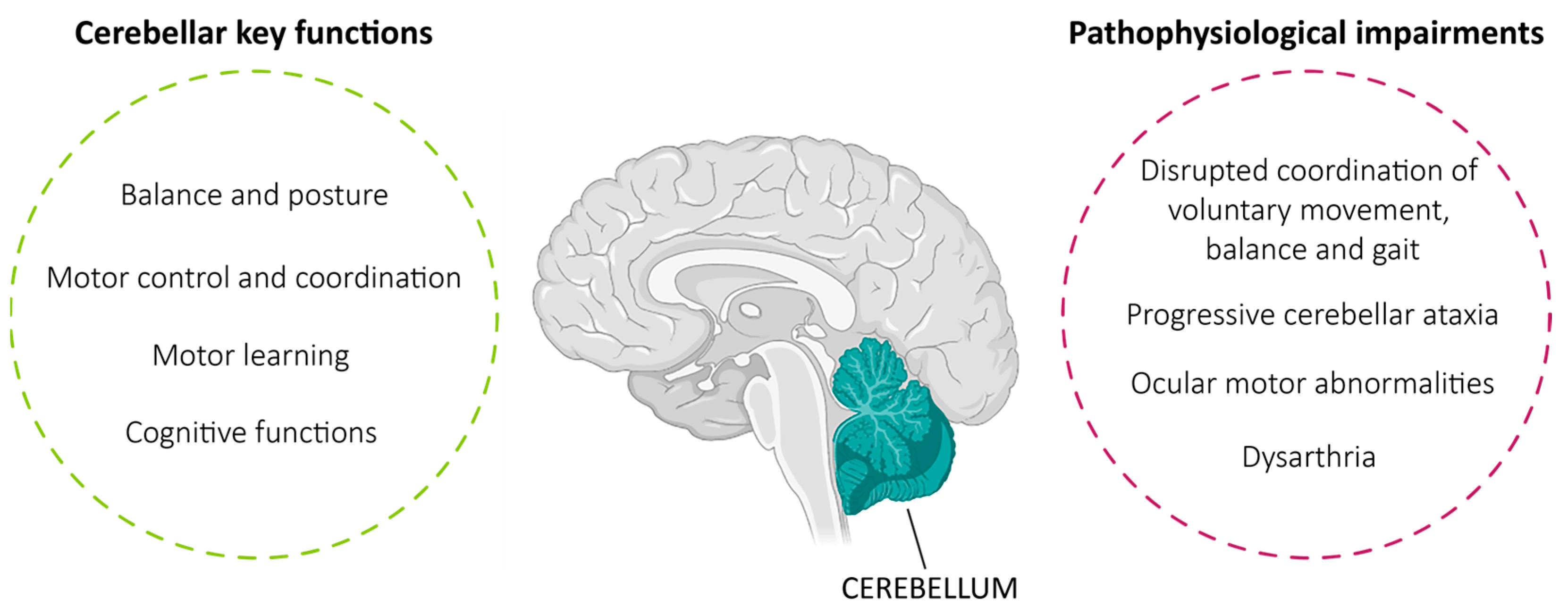
1. Introduction
2. The Developmental Blueprint of the Cerebellum
2.1. Patterning of Cerebellar Neuronal Subtypes
- Reelin, a secreted glycoprotein highly expressed in the EGL, regulating PC alignment and overall cerebellar layering. Its deficiency results in disorganized layering and ataxia-like phenotypes [62].
- Sonic hedgehog (SHH), secreted by PCs, acts as a potent mitogen for GCPs and drives expansion of the EGL that shapes cerebellar foliation [63].
2.2. Integration of Inputs and Outputs of Cerebellar Wiring
3. iPSCs for Modeling Cerebellar Ataxias
3.1. Advances and Challenges in Human iPSC-Derived Cerebellar Models
3.2. Friedreich’s Ataxia Disease Modeling Using iPSCs
3.3. Genetic Heterogeneity and iPSC-Based Models of Spinocerebellar Ataxias (SCAs)
3.4. iPSC Modeling Revealed Developmental and DNA Damage Response Defects in Ataxia–Telangiectasia
3.5. Early Advancement in Uncovering ARSACS Pathophysiology via iPSC Modeling
3.6. Dual Role of Microglia in Neuroinflammation and Neuroprotection in Cerebellar Ataxias
3.7. Expanding the Frontiers of Ataxia Research
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ito, M. Historical Review of the Significance of the Cerebellum and the Role of Purkinje Cells in Motor Learning. Ann. N. Y. Acad. Sci. 2002, 978, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Ito, M. Cerebellar Circuitry as a Neuronal Machine. Prog. Neurobiol. 2006, 78, 272–303. [Google Scholar] [CrossRef]
- Natarajan, V.; Mugundhan, K. Cerebellum. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2013; pp. 643–653. [Google Scholar]
- Hibi, M.; Shimizu, T. Development of the Cerebellum and Cerebellar Neural Circuits. Dev. Neurobiol. 2012, 72, 282–301. [Google Scholar] [CrossRef]
- Wizeman, J.W.; Guo, Q.; Wilion, E.M.; Li, J.Y. Specification of Diverse Cell Types during Early Neurogenesis of the Mouse Cerebellum. eLife 2019, 8, e42388. [Google Scholar] [CrossRef]
- Sotelo, C. Cellular and Genetic Regulation of the Development of the Cerebellar System. Prog. Neurobiol. 2004, 72, 295–339. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Krienen, F.M.; Castellanos, A.; Diaz, J.C.; Yeo, B.T.T. The Organization of the Human Cerebellum Estimated by Intrinsic Functional Connectivity. J. Neurophysiol. 2011, 106, 2322–2345. [Google Scholar] [CrossRef]
- Strick, P.L.; Dum, R.P.; Fiez, J.A. Cerebellum and Nonmotor Function. Annu. Rev. Neurosci. 2009, 32, 413–434. [Google Scholar] [CrossRef] [PubMed]
- McAfee, S.S.; Liu, Y.; Sillitoe, R.V.; Heck, D.H. Cerebellar Coordination of Neuronal Communication in Cerebral Cortex. Front. Syst. Neurosci. 2022, 15, 781527. [Google Scholar] [CrossRef]
- Phillips, J.R.; Hewedi, D.H.; Eissa, A.M.; Moustafa, A.A. The Cerebellum and Psychiatric Disorders. Front. Public Health 2015, 3, 66. [Google Scholar] [CrossRef]
- Schmahmann, J.D. Disorders of the Cerebellum: Ataxia, Dysmetria of Thought, and the Cerebellar Cognitive Affective Syndrome. J. Neuropsychiatry Clin. Neurosci. 2004, 16, 367–378. [Google Scholar] [CrossRef]
- Schmahmann, J.D. The Cerebellum and Cognition. Neurosci. Lett. 2019, 688, 62–75. [Google Scholar] [CrossRef]
- Stoodley, C.J. The Cerebellum and Neurodevelopmental Disorders. Cerebellum 2016, 15, 34–37. [Google Scholar] [CrossRef]
- Manto, M.; Gandini, J.; Feil, K.; Strupp, M. Cerebellar Ataxias: An Update. Curr. Opin. Neurol. 2020, 33, 150–160. [Google Scholar] [CrossRef]
- Parkinson, M.H.; Boesch, S.; Nachbauer, W.; Mariotti, C.; Giunti, P. Clinical Features of Friedreich’s Ataxia: Classical and Atypical Phenotypes. J. Neurochem. 2013, 126, 103–117. [Google Scholar] [CrossRef]
- Campuzano, V.; Montermini, L.; Moltò, M.D.; Pianese, L.; Cossée, M.; Cavalcanti, F.; Monros, E.; Rodius, F.; Duclos, F.; Monticelli, A.; et al. Friedreich’s Ataxia: Autosomal Recessive Disease Caused by an Intronic GAA Triplet Repeat Expansion. Science 1996, 271, 1423–1427. [Google Scholar] [CrossRef]
- Herman, D.; Jenssen, K.; Burnett, R.; Soragni, E.; Perlman, S.L.; Gottesfeld, J.M. Histone Deacetylase Inhibitors Reverse Gene Silencing in Friedreich’s Ataxia. Nat. Chem. Biol. 2006, 2, 551–558. [Google Scholar] [CrossRef]
- Koeppen, A.H.; Morral, J.A.; Davis, A.N.; Qian, J.; Petrocine, S.V.; Knutson, M.D.; Gibson, W.M.; Cusack, M.J.; Li, D. The Dorsal Root Ganglion in Friedreich’s Ataxia. Acta Neuropathol. 2009, 118, 763–776. [Google Scholar] [CrossRef]
- Koeppen, A.H.; Mazurkiewicz, J.E. Friedreich Ataxia: Neuropathology Revised. J. Neuropathol. Exp. Neurol. 2013, 72, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, R.; Yau, W.Y.; O’Connor, E.; Houlden, H. Spinocerebellar Ataxia: An Update. J. Neurol. 2019, 266, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Rousseaux, M.W.C.; Tschumperlin, T.; Lu, H.-C.; Lackey, E.P.; Bondar, V.V.; Wan, Y.-W.; Tan, Q.; Adamski, C.J.; Friedrich, J.; Twaroski, K.; et al. ATXN1-CIC Complex Is the Primary Driver of Cerebellar Pathology in Spinocerebellar Ataxia Type 1 through a Gain-of-Function Mechanism. Neuron 2018, 97, 1235–1243.e5. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, N.; Steinlein, O.; Xiao, J.; Rosenhouse-Dantsker, A.; Gerlai, R. Spinocerebellar Ataxia Type 2. In Advances in Experimental Medicine and Biology; Springer International Publishing: Cham, Switzerland, 2018; pp. 175–195. ISBN 978-3-319-71778-4. [Google Scholar]
- Costa, M.D.C.; Paulson, H.L. Toward Understanding Machado–Joseph Disease. Prog. Neurobiol. 2012, 97, 239–257. [Google Scholar] [CrossRef]
- Rezaei, N.; Steinlein, O.; Xiao, J.; Rosenhouse-Dantsker, A.; Gerlai, R. Clinical Features of Machado-Joseph Disease. In Advances in Experimental Medicine and Biology; Springer International Publishing: Cham, Switzerland, 2018; pp. 255–273. ISBN 978-3-319-71778-4. [Google Scholar]
- Natarajan, V.; Mugundhan, K. Machado–Joseph Disease/Spinocerebellar Ataxia Type 3. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2012; pp. 437–449. [Google Scholar]
- Cavone, F.; Cappelli, S.; Bonuccelli, A.; D’Elios, S.; Costagliola, G.; Peroni, D.; Orsini, A.; Consolini, R. Ataxia Telangiectasia Arising as Immunodeficiency: The Intriguing Differential Diagnosis. J. Clin. Med. 2023, 12, 6041. [Google Scholar] [CrossRef]
- Lavin, M.F.; Shiloh, Y. The genetic defect in ataxia-telangiectasia. Annu. Rev. Immunol. 1997, 15, 177–202. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Du, L.; Nagabayashi, G.; Seeger, R.C.; Gatti, R.A. ATM Is Down-Regulated by N-Myc–Regulated microRNA-421. Proc. Natl. Acad. Sci. USA 2010, 107, 1506–1511. [Google Scholar] [CrossRef]
- Stagni, V.; Oropallo, V.; Fianco, G.; Antonelli, M.; Cinà, I.; Barilà, D. Tug of War between Survival and Death: Exploring ATM Function in Cancer. Int. J. Mol. Sci. 2014, 15, 5388–5409. [Google Scholar] [CrossRef]
- Cheng, A.; Tse, K.-H.; Chow, H.-M.; Gan, Y.; Song, X.; Ma, F.; Qian, Y.X.Y.; She, W.; Herrup, K. ATM Loss Disrupts the Autophagy-Lysosomal Pathway. Autophagy 2021, 17, 1998–2010. [Google Scholar] [CrossRef]
- Aida, I.; Ozawa, T.; Fujinaka, H.; Goto, K.; Ohta, K.; Nakajima, T. Autosomal Recessive Spastic Ataxia of Charlevoix-Saguenay without Spasticity. Intern. Med. 2021, 60, 3963–3967. [Google Scholar] [CrossRef] [PubMed]
- Gentil, B.J.; Lai, G.-T.; Menade, M.; Larivière, R.; Minotti, S.; Gehring, K.; Chapple, J.-P.; Brais, B.; Durham, H.D. Sacsin, Mutated in the Ataxia ARSACS, Regulates Intermediate Filament Assembly and Dynamics. FASEB J. 2019, 33, 2982–2994. [Google Scholar] [CrossRef] [PubMed]
- Salem, I.H.; Blais, M.; Zuluaga-Sánchez, V.M.; Rouleau, L.; Becker, E.B.E.; Dupré, N. ARSACS: Clinical Features, Pathophysiology and iPS-Derived Models. Cerebellum 2025, 24, 24. [Google Scholar] [CrossRef] [PubMed]
- Synofzik, M.; Soehn, A.S.; Gburek-Augustat, J.; Schicks, J.; Karle, K.N.; Schüle, R.; Haack, T.B.; Schöning, M.; Biskup, S.; Rudnik-Schöneborn, S.; et al. Autosomal Recessive Spastic Ataxia of Charlevoix Saguenay (ARSACS): Expanding the Genetic, Clinical and Imaging Spectrum. Orphanet J. Rare Dis. 2013, 8, 41. [Google Scholar] [CrossRef]
- Campbell, A.; Krupp, B.; Bushman, J.; Noble, M.; Pröschel, C.; Mayer-Pröschel, M. A Novel Mouse Model for Ataxia-Telangiectasia with a N-Terminal Mutation Displays a Behavioral Defect and a Low Incidence of Lymphoma but No Increased Oxidative Burden. Hum. Mol. Genet. 2015, 24, 6331–6349. [Google Scholar] [CrossRef]
- Damrath, E.; Heck, M.V.; Gispert, S.; Azizov, M.; Nowock, J.; Seifried, C.; Rüb, U.; Walter, M.; Auburger, G. ATXN2-CAG42 Sequesters PABPC1 into Insolubility and Induces FBXW8 in Cerebellum of Old Ataxic Knock-In Mice. PLoS Genet. 2012, 8, e1002920. [Google Scholar] [CrossRef]
- Huynh, D.P.; Figueroa, K.; Hoang, N.; Pulst, S.-M. Nuclear Localization or Inclusion Body Formation of Ataxin-2 Are Not Necessary for SCA2 Pathogenesis in Mouse or Human. Nat. Genet. 2000, 26, 44–50. [Google Scholar] [CrossRef]
- Aguiar, J.; Fernández, J.; Aguilar, A.; Mendoza, Y.; Vázquez, M.; Suárez, J.; Berlanga, J.; Cruz, S.; Guillén, G.; Herrera, L.; et al. Ubiquitous Expression of Human SCA2 Gene under the Regulation of the SCA2 Self Promoter Cause Specific Purkinje Cell Degeneration in Transgenic Mice. Neurosci. Lett. 2006, 392, 202–206. [Google Scholar] [CrossRef]
- Liu, J.; Tang, T.-S.; Tu, H.; Nelson, O.; Herndon, E.; Huynh, D.P.; Pulst, S.M.; Bezprozvanny, I. Deranged Calcium Signaling and Neurodegeneration in Spinocerebellar Ataxia Type 2. J. Neurosci. 2009, 29, 9148–9162. [Google Scholar] [CrossRef]
- Ross, C.A.; Akimov, S.S. Human-Induced Pluripotent Stem Cells: Potential for Neurodegenerative Diseases. Hum. Mol. Genet. 2014, 23, R17–R26. [Google Scholar] [CrossRef]
- Atamian, A.; Birtele, M.; Hosseini, N.; Quadrato, G. Generation and Long-Term Culture of Human Cerebellar Organoids from Pluripotent Stem Cells. Nat. Protoc. 2025, 20, 1584–1615. [Google Scholar] [CrossRef]
- Lago, C.; Gianesello, M.; Santomaso, L.; Leva, G.; Ballabio, C.; Anderle, M.; Antonica, F.; Tiberi, L. Medulloblastoma and High-Grade Glioma Organoids for Drug Screening, Lineage Tracing, Co-Culture and In Vivo Assay. Nat. Protoc. 2023, 18, 2143–2180. [Google Scholar] [CrossRef] [PubMed]
- Muguruma, K.; Nishiyama, A.; Kawakami, H.; Hashimoto, K.; Sasai, Y. Self-Organization of Polarized Cerebellar Tissue in 3D Culture of Human Pluripotent Stem Cells. Cell Rep. 2015, 10, 537–550. [Google Scholar] [CrossRef] [PubMed]
- Zink, A.; Lisowski, P.; Prigione, A. Generation of Human iPSC-Derived Neural Progenitor Cells (NPCs) as Drug Discovery Model for Neurological and Mitochondrial Disorders. Bio-Protocol 2021, 11, e3939. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, M.A.; Renner, M.; Martin, C.-A.; Wenzel, D.; Bicknell, L.S.; Hurles, M.E.; Homfray, T.; Penninger, J.M.; Jackson, A.P.; Knoblich, J.A. Cerebral Organoids Model Human Brain Development and Microcephaly. Nature 2013, 501, 373–379. [Google Scholar] [CrossRef]
- McQuade, A.; Coburn, M.; Tu, C.H.; Hasselmann, J.; Davtyan, H.; Blurton-Jones, M. Development and Validation of a Simplified Method to Generate Human Microglia from Pluripotent Stem Cells. Mol. Neurodegener. 2018, 13, 67. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Gutierrez, C.; Xue, T.; Hampton, C.; Vergara, M.N.; Cao, L.-H.; Peters, A.; Park, T.S.; Zambidis, E.T.; Meyer, J.S.; et al. Generation of Three-Dimensional Retinal Tissue with Functional Photoreceptors from Human iPSCs. Nat. Commun. 2014, 5, 4047. [Google Scholar] [CrossRef]
- Kalpana, K.; Rao, C.; Semrau, S.; Zhang, B.; Noggle, S.; Fossati, V. Generating Neuroimmune Assembloids Using Human Induced Pluripotent Stem Cell (iPSC)-Derived Cortical Organoids and Microglia. In Organoids; Turksen, K., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2024; Volume 2951, pp. 139–158. ISBN 978-1-0716-4681-6. [Google Scholar]
- McCrimmon, C.M.; Toker, D.; Pahos, M.; Lozano, K.; Lin, J.J.; Parent, J.; Tidball, A.; Zheng, J.; Molnár, L.; Mody, I.; et al. Modeling Cortical Versus Hippocampal Network Dysfunction in a Human Brain Assembloid Model of Epilepsy and Intellectual Disability. bioRxiv 2024. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Hibi, M. Development and Evolution of Cerebellar Neural Circuits. Dev. Growth Differ. 2012, 54, 373–389. [Google Scholar] [CrossRef]
- Acampora, D.; Avantaggiato, V.; Tuorto, F.; Simeone, A. Genetic Control of Brain Morphogenesis through Otx Gene Dosage Requirement. Development 1997, 124, 3639–3650. [Google Scholar] [CrossRef]
- Liu, A.; Joyner, A.L. Early Anterior/Posterior Patterning of the Midbrain and Cerebellum. Annu. Rev. Neurosci. 2001, 24, 869–896. [Google Scholar] [CrossRef]
- Dickinson, M.E.; Krumlauf, R.; McMahon, A.P. Evidence for a Mitogenic Effect of Wnt-1 in the Developing Mammalian Central Nervous System. Development 1994, 120, 1453–1471. [Google Scholar] [CrossRef]
- Sepp, M.; Leiss, K.; Murat, F.; Okonechnikov, K.; Joshi, P.; Leushkin, E.; Spänig, L.; Mbengue, N.; Schneider, C.; Schmidt, J.; et al. Cellular Development and Evolution of the Mammalian Cerebellum. Nature 2024, 625, 788–796. [Google Scholar] [CrossRef]
- Chizhikov, V.V.; Lindgren, A.G.; Currle, D.S.; Rose, M.F.; Monuki, E.S.; Millen, K.J. The Roof Plate Regulates Cerebellar Cell-Type Specification and Proliferation. Development 2006, 133, 2793–2804. [Google Scholar] [CrossRef] [PubMed]
- Tam, W.Y.; Wang, X.; Cheng, A.S.K.; Cheung, K.-K. In Search of Molecular Markers for Cerebellar Neurons. Int. J. Mol. Sci. 2021, 22, 1850. [Google Scholar] [CrossRef] [PubMed]
- Redies, C.; Neudert, F.; Lin, J. Cadherins in Cerebellar Development: Translation of Embryonic Patterning into Mature Functional Compartmentalization. Cerebellum 2011, 10, 393–408. [Google Scholar] [CrossRef]
- Consalez, G.G.; Goldowitz, D.; Casoni, F.; Hawkes, R. Origins, Development, and Compartmentation of the Granule Cells of the Cerebellum. Front. Neural Circuits 2021, 14, 611841. [Google Scholar] [CrossRef] [PubMed]
- Leto, K.; Arancillo, M.; Becker, E.B.E.; Buffo, A.; Chiang, C.; Ding, B.; Dobyns, W.B.; Dusart, I.; Haldipur, P.; Hatten, M.E.; et al. Consensus Paper: Cerebellar Development. Cerebellum 2016, 15, 789–828. [Google Scholar] [CrossRef]
- Renaud, J.; Kerjan, G.; Sumita, I.; Zagar, Y.; Georget, V.; Kim, D.; Fouquet, C.; Suda, K.; Sanbo, M.; Suto, F.; et al. Plexin-A2 and Its Ligand, Sema6A, Control Nucleus-Centrosome Coupling in Migrating Granule Cells. Nat. Neurosci. 2008, 11, 440–449. [Google Scholar] [CrossRef]
- Maier, V.; Jolicoeur, C.; Rayburn, H.; Takegahara, N.; Kumanogoh, A.; Kikutani, H.; Tessier-Lavigne, M.; Wurst, W.; Friedel, R.H. Semaphorin 4C and 4G Are Ligands of Plexin-B2 Required in Cerebellar Development. Mol. Cell. Neurosci. 2011, 46, 419–431. [Google Scholar] [CrossRef]
- Nakamura, K.; Beppu, M.; Sakai, K.; Yagyu, H.; Matsumaru, S.; Kohno, T.; Hattori, M. The C-Terminal Region of Reelin Is Necessary for Proper Positioning of a Subset of Purkinje Cells in the Postnatal Cerebellum. Neuroscience 2016, 336, 20–29. [Google Scholar] [CrossRef]
- Dahmane, N.; Altaba, A.R.I. Sonic Hedgehog Regulates the Growth and Patterning of the Cerebellum. Development 1999, 126, 3089–3100. [Google Scholar] [CrossRef]
- Chédotal, A.; Bloch-Gallego, E.; Sotelo, C. The Embryonic Cerebellum Contains Topographic Cues That Guide Developing Inferior Olivary Axons. Development 1997, 124, 861–870. [Google Scholar] [CrossRef]
- Sotelo, C.; Bourrat, F.; Triller, A. Postnatal Development of the Inferior Olivary Complex in the Rat. II. Topographic Organization of the Immature Olivocerebellar Projection. J. Comp. Neurol. 1984, 222, 177–199. [Google Scholar] [CrossRef] [PubMed]
- Beckinghausen, J.; Sillitoe, R.V. Insights into Cerebellar Development and Connectivity. Neurosci. Lett. 2019, 688, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Schilling, K.; Oberdick, J.; Rossi, F.; Baader, S.L. Besides Purkinje Cells and Granule Neurons: An Appraisal of the Cell Biology of the Interneurons of the Cerebellar Cortex. Histochem. Cell Biol. 2008, 130, 601–615. [Google Scholar] [CrossRef]
- Natarajan, V.; Mugundhan, K. Physiology of the Cerebellum. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2018; pp. 85–108. [Google Scholar]
- Schweighofer, N.; Lang, E.J.; Kawato, M. Role of the Olivo-Cerebellar Complex in Motor Learning and Control. Front. Neural Circuits 2013, 7, 94. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Duan, L.; Ou, Y.; Ling, Q.; Cao, L.; Qian, H.; Zhang, J.; Wang, J.; Yuan, X. The Cerebellum and Cognitive Neural Networks. Front. Hum. Neurosci. 2023, 17, 1197459. [Google Scholar] [CrossRef]
- Perlman, S. Hereditary Ataxia Overview. In GeneReviews®; Adam, M.P., Feldman, J., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Watson, L.M.; Wong, M.M.K.; Becker, E.B.E. Induced Pluripotent Stem Cell Technology for Modelling and Therapy of Cerebellar Ataxia. Open Biol. 2015, 5, 150056. [Google Scholar] [CrossRef]
- Muguruma, K.; Nishiyama, A.; Ono, Y.; Miyawaki, H.; Mizuhara, E.; Hori, S.; Kakizuka, A.; Obata, K.; Yanagawa, Y.; Hirano, T.; et al. Ontogeny-Recapitulating Generation and Tissue Integration of ES Cell–Derived Purkinje Cells. Nat. Neurosci. 2010, 13, 1171–1180. [Google Scholar] [CrossRef]
- Higuera, G.A.; Iaffaldano, G.; Bedar, M.; Shpak, G.; Broersen, R.; Munshi, S.T.; Dupont, C.; Gribnau, J.; De Vrij, F.M.S.; Kushner, S.A.; et al. An Expandable Embryonic Stem Cell-Derived Purkinje Neuron Progenitor Population That Exhibits in Vivo Maturation in the Adult Mouse Cerebellum. Sci. Rep. 2017, 7, 8863. [Google Scholar] [CrossRef]
- Watson, L.M.; Wong, M.M.K.; Vowles, J.; Cowley, S.A.; Becker, E.B.E. A Simplified Method for Generating Purkinje Cells from Human-Induced Pluripotent Stem Cells. Cerebellum 2018, 17, 419–427. [Google Scholar] [CrossRef]
- Kamei, T.; Tamada, A.; Kimura, T.; Kakizuka, A.; Asai, A.; Muguruma, K. Survival and Process Outgrowth of Human iPSC-Derived Cells Expressing Purkinje Cell Markers in a Mouse Model for Spinocerebellar Degenerative Disease. Exp. Neurol. 2023, 369, 114511. [Google Scholar] [CrossRef]
- Muguruma, K. Self-Organized Cerebellar Tissue from Human Pluripotent Stem Cells and Disease Modeling with Patient-Derived iPSCs. Cerebellum 2018, 17, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Bird, M.J.; Needham, K.; Frazier, A.E.; Van Rooijen, J.; Leung, J.; Hough, S.; Denham, M.; Thornton, M.E.; Parish, C.L.; Nayagam, B.A.; et al. Functional Characterization of Friedreich Ataxia iPS-Derived Neuronal Progenitors and Their Integration in the Adult Brain. PLoS ONE 2014, 9, e101718. [Google Scholar] [CrossRef]
- Koeppen, A.H. Friedreich’s Ataxia: Pathology, Pathogenesis, and Molecular Genetics. J. Neurol. Sci. 2011, 303, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.; Brown, J.M.; Buckle, V.J.; Wade-Martins, R.; Lufino, M.M.P. Expanded GAA Repeats Impair FXN Gene Expression and Reposition the FXN Locus to the Nuclear Lamina in Single Cells. Hum. Mol. Genet. 2015, 24, 3457–3471. [Google Scholar] [CrossRef] [PubMed]
- Vilsbøll, T.; Knop, F.K. Long-Acting GLP-1 Analogs for the Treatment of Type 2 Diabetes Mellitus. BioDrugs 2008, 22, 251–257. [Google Scholar] [CrossRef]
- Drucker, D.J.; Philippe, J.; Mojsov, S.; Chick, W.L.; Habener, J.F. Glucagon-like Peptide I Stimulates Insulin Gene Expression and Increases Cyclic AMP Levels in a Rat Islet Cell Line. Proc. Natl. Acad. Sci. USA 1987, 84, 3434–3438. [Google Scholar] [CrossRef]
- Fernández-Millán, E.; Martín, M.A.; Goya, L.; Lizárraga-Mollinedo, E.; Escrivá, F.; Ramos, S.; Álvarez, C. Glucagon-like Peptide-1 Improves Beta-Cell Antioxidant Capacity via Extracellular Regulated Kinases Pathway and Nrf2 Translocation. Free Radic. Biol. Med. 2016, 95, 16–26. [Google Scholar] [CrossRef]
- Igoillo-Esteve, M.; Oliveira, A.F.; Cosentino, C.; Fantuzzi, F.; Demarez, C.; Toivonen, S.; Hu, A.; Chintawar, S.; Lopes, M.; Pachera, N.; et al. Exenatide Induces Frataxin Expression and Improves Mitochondrial Function in Friedreich Ataxia. JCI Insight 2020, 5, e134221. [Google Scholar] [CrossRef]
- Hommersom, M.P.; Buijsen, R.A.M.; Van Roon-Mom, W.M.C.; Van De Warrenburg, B.P.C.; Van Bokhoven, H. Human Induced Pluripotent Stem Cell-Based Modelling of Spinocerebellar Ataxias. Stem Cell Rev. Rep. 2022, 18, 441–456. [Google Scholar] [CrossRef]
- Luo, Y.; Fan, Y.; Zhou, B.; Xu, Z.; Chen, Y.; Sun, X. Generation of Induced Pluripotent Stem Cells from Skin Fibroblasts of a Patient with Olivopontocerebellar Atrophy. Tohoku J. Exp. Med. 2012, 226, 151–159. [Google Scholar] [CrossRef]
- Kawaguchi, S.; Hirano, T. Gating of Long-term Depression by Ca2+/Calmodulin-dependent Protein Kinase II through Enhanced cGMP Signalling in Cerebellar Purkinje Cells. J. Physiol. 2013, 591, 1707–1730. [Google Scholar] [CrossRef]
- Egorova, P.A.; Bezprozvanny, I.B. Molecular Mechanisms and Therapeutics for Spinocerebellar Ataxia Type 2. Neurotherapeutics 2019, 16, 1050–1073. [Google Scholar] [CrossRef]
- Raghunathan, N.; Sankaran, S.; Miteu, G.D. A Comprehensive Review of iPS Cell Line-Based Disease Modelling of the Polyglutamine Spinocerebellar Ataxias 2 and 3: A Focus on the Research Outcomes. Ann. Med. Surg. 2024, 86, 3487–3498. [Google Scholar] [CrossRef]
- Xia, G.; Santostefano, K.; Hamazaki, T.; Liu, J.; Subramony, S.H.; Terada, N.; Ashizawa, T. Generation of Human-Induced Pluripotent Stem Cells to Model Spinocerebellar Ataxia Type 2 In Vitro. J. Mol. Neurosci. 2013, 51, 237–248. [Google Scholar] [CrossRef]
- Kiehl, T.-R.; Nechiporuk, A.; Figueroa, K.P.; Keating, M.T.; Huynh, D.P.; Pulst, S.-M. Generation and Characterization of Sca2 (Ataxin-2) Knockout Mice. Biochem. Biophys. Res. Commun. 2006, 339, 17–24. [Google Scholar] [CrossRef]
- Kumar, R.; Donakonda, S.; Müller, S.A.; Lichtenthaler, S.F.; Bötzel, K.; Höglinger, G.U.; Koeglsperger, T. Basic Fibroblast Growth Factor 2-Induced Proteome Changes Endorse Lewy Body Pathology in Hippocampal Neurons. iScience 2020, 23, 101349. [Google Scholar] [CrossRef]
- Chuang, C.-Y.; Yang, C.-C.; Soong, B.-W.; Yu, C.-Y.; Chen, S.-H.; Huang, H.-P.; Kuo, H.-C. Modeling Spinocerebellar Ataxias 2 and 3 with iPSCs Reveals a Role for Glutamate in Disease Pathology. Sci. Rep. 2019, 9, 1166. [Google Scholar] [CrossRef]
- Marthaler, A.G.; Tubsuwan, A.; Schmid, B.; Poulsen, U.B.; Hyttel, P.; Nielsen, J.E.; Nielsen, T.T.; Holst, B. Generation of Spinocerebellar Ataxia Type 2 Patient-Derived iPSC Line H271. Stem Cell Res. 2016, 16, 159–161. [Google Scholar] [CrossRef]
- Marthaler, A.G.; Schmid, B.; Tubsuwan, A.; Poulsen, U.B.; Engelbrecht, A.F.; Mau-Holzmann, U.A.; Hyttel, P.; Nielsen, J.E.; Nielsen, T.T.; Holst, B. Generation of an Isogenic, Gene-Corrected Control Cell Line of the Spinocerebellar Ataxia Type 2 Patient-Derived iPSC Line H196. Stem Cell Res. 2016, 16, 162–165. [Google Scholar] [CrossRef]
- Marthaler, A.G.; Schmid, B.; Tubsuwan, A.; Poulsen, U.B.; Hyttel, P.; Nielsen, T.T.; Nielsen, J.E.; Holst, B. Generation of Spinocerebellar Ataxia Type 2 Patient-Derived iPSC Line H266. Stem Cell Res. 2016, 16, 166–169. [Google Scholar] [CrossRef]
- Marthaler, A.G.; Schmid, B.; Tubsuwan, A.; Poulsen, U.B.; Engelbrecht, A.F.; Mau-Holzmann, U.A.; Hyttel, P.; Nielsen, J.E.; Nielsen, T.T.; Holst, B. Generation of an Isogenic, Gene-Corrected Control Cell Line of the Spinocerebellar Ataxia Type 2 Patient-Derived iPSC Line H271. Stem Cell Res. 2016, 16, 180–183. [Google Scholar] [CrossRef]
- Marthaler, A.G.; Schmid, B.; Tubsuwan, A.; Poulsen, U.B.; Hyttel, P.; Nielsen, T.T.; Nielsen, J.E.; Holst, B. Generation of Spinocerebellar Ataxia Type 2 Patient-Derived iPSC Line H196. Stem Cell Res. 2016, 16, 199–201. [Google Scholar] [CrossRef]
- Marthaler, A.G.; Tubsuwan, A.; Schmid, B.; Poulsen, U.B.; Engelbrecht, A.F.; Mau-Holzmann, U.A.; Hyttel, P.; Nielsen, T.T.; Nielsen, J.E.; Holst, B. Generation of an Isogenic, Gene-Corrected Control Cell Line of the Spinocerebellar Ataxia Type 2 Patient-Derived iPSC Line H266. Stem Cell Res. 2016, 16, 202–205. [Google Scholar] [CrossRef]
- Maguire, J.A.; Gagne, A.L.; Gonzalez-Alegre, P.; Davidson, B.L.; Shakkottai, V.; Gadue, P.; French, D.L. Generation of Spinocerebellar Ataxia Type 2 Induced Pluripotent Stem Cell Lines, CHOPi002-A and CHOPi003-A, from Patients with Abnormal CAG Repeats in the Coding Region of the ATXN2 Gene. Stem Cell Res. 2019, 34, 101361. [Google Scholar] [CrossRef]
- Li, P.P.; Sun, X.; Xia, G.; Arbez, N.; Paul, S.; Zhu, S.; Peng, H.B.; Ross, C.A.; Koeppen, A.H.; Margolis, R.L.; et al. ATXN2-AS, a Gene Antisense to ATXN2, Is Associated with Spinocerebellar Ataxia Type 2 and Amyotrophic Lateral Sclerosis. Ann. Neurol. 2016, 80, 600–615. [Google Scholar] [CrossRef]
- Du, X.; Wang, J.; Zhu, H.; Rinaldo, L.; Lamar, K.-M.; Palmenberg, A.C.; Hansel, C.; Gomez, C.M. Second Cistron in CACNA1A Gene Encodes a Transcription Factor Mediating Cerebellar Development and SCA6. Cell 2013, 154, 118–133. [Google Scholar] [CrossRef]
- Du, X.; Semler, B.L.; Gomez, C.M. Revelations from a Bicistronic Calcium Channel Gene. Cell Cycle 2014, 13, 875–876. [Google Scholar] [CrossRef][Green Version]
- Ishida, Y.; Kawakami, H.; Kitajima, H.; Nishiyama, A.; Sasai, Y.; Inoue, H.; Muguruma, K. Vulnerability of Purkinje Cells Generated from Spinocerebellar Ataxia Type 6 Patient-Derived iPSCs. Cell Rep. 2017, 18, 1075–1076. [Google Scholar] [CrossRef]
- Bavassano, C.; Eigentler, A.; Stanika, R.; Obermair, G.J.; Boesch, S.; Dechant, G.; Nat, R. Bicistronic CACNA1A Gene Expression in Neurons Derived from Spinocerebellar Ataxia Type 6 Patient-Induced Pluripotent Stem Cells. Stem Cells Dev. 2017, 26, 1612–1625. [Google Scholar] [CrossRef]
- Durr, A. Autosomal Dominant Cerebellar Ataxias: Polyglutamine Expansions and Beyond. Lancet Neurol. 2010, 9, 885–894. [Google Scholar] [CrossRef]
- Morino, H.; Matsuda, Y.; Muguruma, K.; Miyamoto, R.; Ohsawa, R.; Ohtake, T.; Otobe, R.; Watanabe, M.; Maruyama, H.; Hashimoto, K.; et al. A Mutation in the Low Voltage-Gated Calcium Channel CACNA1G Alters the Physiological Properties of the Channel, Causing Spinocerebellar Ataxia. Mol. Brain 2015, 8, 89. [Google Scholar] [CrossRef]
- Wong, M.M.K.; Hoekstra, S.D.; Vowles, J.; Watson, L.M.; Fuller, G.; Németh, A.H.; Cowley, S.A.; Ansorge, O.; Talbot, K.; Becker, E.B.E. Neurodegeneration in SCA14 Is Associated with Increased PKCγ Kinase Activity, Mislocalization and Aggregation. Acta Neuropathol. Commun. 2018, 6, 99. [Google Scholar] [CrossRef]
- Buijsen, R.A.M.; Hu, M.; Sáez-González, M.; Notopoulou, S.; Mina, E.; Koning, W.; Gardiner, S.L.; Van Der Graaf, L.M.; Daoutsali, E.; Pepers, B.A.; et al. Spinocerebellar Ataxia Type 1 Characteristics in Patient-Derived Fibroblast and iPSC-Derived Neuronal Cultures. Mov. Disord. 2023, 38, 1428–1442. [Google Scholar] [CrossRef]
- Fryer, J.D.; Yu, P.; Kang, H.; Mandel-Brehm, C.; Carter, A.N.; Crespo-Barreto, J.; Gao, Y.; Flora, A.; Shaw, C.; Orr, H.T.; et al. Exercise and Genetic Rescue of SCA1 via the Transcriptional Repressor Capicua. Science 2011, 334, 690–693. [Google Scholar] [CrossRef]
- Fan, J.-B.; Arimoto, K.; Motamedchaboki, K.; Yan, M.; Wolf, D.A.; Zhang, D.-E. Identification and Characterization of a Novel ISG15-Ubiquitin Mixed Chain and Its Role in Regulating Protein Homeostasis. Sci. Rep. 2015, 5, 12704. [Google Scholar] [CrossRef]
- Homma, H.; Yoshioka, Y.; Fujita, K.; Shirai, S.; Hama, Y.; Komano, H.; Saito, Y.; Yabe, I.; Okano, H.; Sasaki, H.; et al. Dynamic Molecular Network Analysis of iPSC-Purkinje Cells Differentiation Delineates Roles of ISG15 in SCA1 at the Earliest Stage. Commun. Biol. 2024, 7, 413. [Google Scholar] [CrossRef]
- Ito, H.; Fujita, K.; Tagawa, K.; Chen, X.; Homma, H.; Sasabe, T.; Shimizu, J.; Shimizu, S.; Tamura, T.; Muramatsu, S.; et al. HMGB1 Facilitates Repair of MitochondrialDNADamage and Extends the Lifespan of Mutant Ataxin-1 Knock-in Mice. EMBO Mol. Med. 2015, 7, 78–101. [Google Scholar] [CrossRef]
- Qi, M.-L.; Tagawa, K.; Enokido, Y.; Yoshimura, N.; Wada, Y.; Watase, K.; Ishiura, S.; Kanazawa, I.; Botas, J.; Saitoe, M.; et al. Proteome Analysis of Soluble Nuclear Proteins Reveals That HMGB1/2 Suppress Genotoxic Stress in Polyglutamine Diseases. Nat. Cell Biol. 2007, 9, 402–414. [Google Scholar] [CrossRef]
- Pérez Ortiz, J.M.; Mollema, N.; Toker, N.; Adamski, C.J.; O’Callaghan, B.; Duvick, L.; Friedrich, J.; Walters, M.A.; Strasser, J.; Hawkinson, J.E.; et al. Reduction of Protein Kinase A-Mediated Phosphorylation of ATXN1-S776 in Purkinje Cells Delays Onset of Ataxia in a SCA1 Mouse Model. Neurobiol. Dis. 2018, 116, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Fagan, K.J.; Chillon, G.; Carrell, E.M.; Waxman, E.A.; Davidson, B.L. Cas9 Editing of ATXN1 in a Spinocerebellar Ataxia Type 1 Mice and Human iPSC-Derived Neurons. Mol. Ther.-Nucleic Acids 2024, 35, 102317. [Google Scholar] [CrossRef] [PubMed]
- Sheeler, C.; Labrada, E.; Duvick, L.; Thompson, L.M.; Zhang, Y.; Orr, H.T.; Cvetanovic, M. Expanded ATXN1 Alters Transcription and Calcium Signaling in SCA1 Human Motor Neurons Differentiated from Induced Pluripotent Stem Cells. Neurobiol. Dis. 2024, 201, 106673. [Google Scholar] [CrossRef] [PubMed]
- Evert, B.O.; Nalavade, R.; Jungverdorben, J.; Matthes, F.; Weber, S.; Rajput, A.; Bonn, S.; Brüstle, O.; Peitz, M.; Krauß, S. Upregulation of miR-370 and miR-543 Is Associated with Reduced Expression of Heat Shock Protein 40 in Spinocerebellar Ataxia Type 3. PLoS ONE 2018, 13, e0201794. [Google Scholar] [CrossRef]
- Onofre, I.; Mendonça, N.; Lopes, S.; Nobre, R.; De Melo, J.B.; Carreira, I.M.; Januário, C.; Gonçalves, A.F.; De Almeida, L.P. Fibroblasts of Machado Joseph Disease Patients Reveal Autophagy Impairment. Sci. Rep. 2016, 6, 28220. [Google Scholar] [CrossRef]
- Martier, R.; Sogorb-Gonzalez, M.; Stricker-Shaver, J.; Hübener-Schmid, J.; Keskin, S.; Klima, J.; Toonen, L.J.; Juhas, S.; Juhasova, J.; Ellederova, Z.; et al. Development of an AAV-Based MicroRNA Gene Therapy to Treat Machado-Joseph Disease. Mol. Ther.-Methods Clin. Dev. 2019, 15, 343–358. [Google Scholar] [CrossRef]
- Wang, Y.; Cheng, Y.; Sun, H.; Wang, Z.; Chen, N.; Shi, C.; Liu, H.; Yang, J.; Xu, Y. Genome Editing in Spinocerebellar Ataxia Type 3 Cells Improves Golgi Apparatus Structure. Sci. Rep. 2025, 15, 12106. [Google Scholar] [CrossRef]
- Song, G.; Ma, Y.; Gao, X.; Zhang, X.; Zhang, F.; Tian, C.; Hou, J.; Liu, Z.; Zhao, Z.; Tian, Y. CRISPR/Cas9-Mediated Genetic Correction Reverses Spinocerebellar Ataxia 3 Disease-Associated Phenotypes in Differentiated Cerebellar Neurons. Life Med. 2022, 1, 27–44. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S.; Helm, J.; Kraft, M.; Korneck, M.; Hübener-Schmid, J.; Schöls, L. Allele-Specific Targeting of Mutant Ataxin-3 by Antisense Oligonucleotides in SCA3-iPSC-Derived Neurons. Mol. Ther. Nucleic Acids 2022, 27, 99–108. [Google Scholar] [CrossRef]
- Yang, H.-H.; Chiang, I.-T.; Liu, J.-W.; Hsieh, J.; Lee, J.-H.; Lu, H.-E.; Tso, H.-S.; Deng, Y.-C.; Kao, J.-C.; Wu, J.-R.; et al. Anti-Excitotoxic Effects of N-Butylidenephthalide Revealed by Chemically Insulted Purkinje Progenitor Cells Derived from SCA3 iPSCs. Int. J. Mol. Sci. 2022, 23, 1391. [Google Scholar] [CrossRef]
- Lee, J.-H.; Lin, S.-Y.; Liu, J.-W.; Lin, S.-Z.; Harn, H.-J.; Chiou, T.-W. N-Butylidenephthalide Modulates Autophagy to Ameliorate Neuropathological Progress of Spinocerebellar Ataxia Type 3 through mTOR Pathway. Int. J. Mol. Sci. 2021, 22, 6339. [Google Scholar] [CrossRef]
- Rajamani, K.; Liu, J.-W.; Wu, C.-H.; Chiang, I.-T.; You, D.-H.; Lin, S.-Y.; Hsieh, D.-K.; Lin, S.-Z.; Harn, H.-J.; Chiou, T.-W. N-Butylidenephthalide Exhibits Protection against Neurotoxicity through Regulation of Tryptophan 2, 3 Dioxygenase in Spinocerebellar Ataxia Type 3. Neuropharmacology 2017, 117, 434–446. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, L.S.; Nóbrega, C.; Tavino, S.; Brinkhaus, M.; Matos, C.; Tomé, S.; Moreira, R.; Henriques, D.; Kaspar, B.K.; Pereira De Almeida, L. Ibuprofen Enhances Synaptic Function and Neural Progenitors Proliferation Markers and Improves Neuropathology and Motor Coordination in Machado–Joseph Disease Models. Hum. Mol. Genet. 2019, 28, 3691–3703. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.M.; Li, H.; Chung, D.L.; Mak, A.T.L.; Leung, F.P.; Chan, H.Y.E.; Wong, W.T. IL-4/STAT6 Axis Observed to Reverse Proliferative Defect in SCA3 Patient-derived Neural Progenitor Cells. Clin. Exp. Pharmacol. Physiol. 2024, 51, 30–39. [Google Scholar] [CrossRef]
- Chen, I.-C.; Chang, K.-H.; Chen, Y.-J.; Chen, Y.-C.; Lee-Chen, G.-J.; Chen, C.-M. Pueraria Lobata and Daidzein Reduce Cytotoxicity by Enhancing Ubiquitin-Proteasome System Function in SCA3-iPSC-Derived Neurons. Oxid. Med. Cell. Longev. 2019, 2019, 8130481. [Google Scholar] [CrossRef]
- Pellerin, D.; Méreaux, J.-L.; Boluda, S.; Danzi, M.C.; Dicaire, M.-J.; Davoine, C.-S.; Genis, D.; Spurdens, G.; Ashton, C.; Hammond, J.M.; et al. Somatic Instability of the FGF14-SCA27B GAA TTC Repeat Reveals a Marked Expansion Bias in the Cerebellum. Brain 2025, 148, 1258–1270. [Google Scholar] [CrossRef]
- Leeson, H.C.; Hunter, Z.; Chaggar, H.K.; Lavin, M.F.; Mackay-Sim, A.; Wolvetang, E.J. Ataxia Telangiectasia iPSC Line Generated from a Patient Olfactory Biopsy Identifies Novel Disease-Causing Mutations. Stem Cell Res. 2021, 56, 102528. [Google Scholar] [CrossRef] [PubMed]
- Nayler, S.; Gatei, M.; Kozlov, S.; Gatti, R.; Mar, J.C.; Wells, C.A.; Lavin, M.; Wolvetang, E. Induced Pluripotent Stem Cells from Ataxia-Telangiectasia Recapitulate the Cellular Phenotype. Stem Cells Transl. Med. 2012, 1, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Nayler, S.P.; Powell, J.E.; Vanichkina, D.P.; Korn, O.; Wells, C.A.; Kanjhan, R.; Sun, J.; Taft, R.J.; Lavin, M.F.; Wolvetang, E.J. Human iPSC-Derived Cerebellar Neurons from a Patient with Ataxia-Telangiectasia Reveal Disrupted Gene Regulatory Networks. Front. Cell. Neurosci. 2017, 11, 321. [Google Scholar] [CrossRef] [PubMed]
- Corti, A.; Sota, R.; Dugo, M.; Calogero, R.A.; Terragni, B.; Mantegazza, M.; Franceschetti, S.; Restelli, M.; Gasparini, P.; Lecis, D.; et al. DNA Damage and Transcriptional Regulation in iPSC-Derived Neurons from Ataxia Telangiectasia Patients. Sci. Rep. 2019, 9, 651. [Google Scholar] [CrossRef]
- Bradshaw, T.Y.; Romano, L.E.L.; Duncan, E.J.; Nethisinghe, S.; Abeti, R.; Michael, G.J.; Giunti, P.; Vermeer, S.; Chapple, J.P. A Reduction in Drp1-Mediated Fission Compromises Mitochondrial Health in Autosomal Recessive Spastic Ataxia of Charlevoix Saguenay. Hum. Mol. Genet. 2016, 25, 3232–3244. [Google Scholar] [CrossRef]
- Louit, A.; Beaudet, M.-J.; Blais, M.; Gros-Louis, F.; Dupré, N.; Berthod, F. In Vitro Characterization of Motor Neurons and Purkinje Cells Differentiated from Induced Pluripotent Stem Cells Generated from Patients with Autosomal Recessive Spastic Ataxia of Charlevoix-Saguenay. Stem Cells Int. 2023, 2023, 1496597. [Google Scholar] [CrossRef]
- Ginhoux, F.; Lim, S.; Hoeffel, G.; Low, D.; Huber, T. Origin and Differentiation of Microglia. Front. Cell. Neurosci. 2013, 7, 45. [Google Scholar] [CrossRef]
- Marín-Teva, J.L.; Dusart, I.; Colin, C.; Gervais, A.; Van Rooijen, N.; Mallat, M. Microglia Promote the Death of Developing Purkinje Cells. Neuron 2004, 41, 535–547. [Google Scholar] [CrossRef]
- Salter, M.W.; Stevens, B. Microglia Emerge as Central Players in Brain Disease. Nat. Med. 2017, 23, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Matcovitch-Natan, O.; Winter, D.R.; Giladi, A.; Vargas Aguilar, S.; Spinrad, A.; Sarrazin, S.; Ben-Yehuda, H.; David, E.; Zelada González, F.; Perrin, P.; et al. Microglia Development Follows a Stepwise Program to Regulate Brain Homeostasis. Science 2016, 353, aad8670. [Google Scholar] [CrossRef]
- Pantovic-Stefanovic, M.; Velimirovic, M.; Jurisic, V.; Puric, M.; Gostiljac, M.; Dodic, S.; Minic, I.; Nesic, M.; Nikolic, T.; Petronijevic, N.; et al. Exploring the Role of TNF-α, TGF-β, and IL-6 Serum Levels in Categorical and Noncategorical Models of Mood and Psychosis. Sci. Rep. 2024, 14, 23117. [Google Scholar] [CrossRef]
- Cvetanovic, M.; Ingram, M.; Orr, H.; Opal, P. Early Activation of Microglia and Astrocytes in Mouse Models of Spinocerebellar Ataxia Type 1. Neuroscience 2015, 289, 289–299. [Google Scholar] [CrossRef]
- Seki, T.; Sato, M.; Kibe, Y.; Ohta, T.; Oshima, M.; Konno, A.; Hirai, H.; Kurauchi, Y.; Hisatsune, A.; Katsuki, H. Lysosomal Dysfunction and Early Glial Activation Are Involved in the Pathogenesis of Spinocerebellar Ataxia Type 21 Caused by Mutant Transmembrane Protein 240. Neurobiol. Dis. 2018, 120, 34–50. [Google Scholar] [CrossRef]
- Shen, Y.; McMackin, M.Z.; Shan, Y.; Raetz, A.; David, S.; Cortopassi, G. Frataxin Deficiency Promotes Excess Microglial DNA Damage and Inflammation That Is Rescued by PJ34. PLoS ONE 2016, 11, e0151026. [Google Scholar] [CrossRef]
- Aikawa, T.; Mogushi, K.; Iijima-Tsutsui, K.; Ishikawa, K.; Sakurai, M.; Tanaka, H.; Mizusawa, H.; Watase, K. Loss of MyD88 Alters Neuroinflammatory Response and Attenuates Early Purkinje Cell Loss in a Spinocerebellar Ataxia Type 6 Mouse Model. Hum. Mol. Genet. 2015, 24, 4780–4791. [Google Scholar] [CrossRef]
- Lai, J.; Demirbas, D.; Kim, J.; Jeffries, A.M.; Tolles, A.; Park, J.; Chittenden, T.W.; Buckley, P.G.; Yu, T.W.; Lodato, M.A.; et al. ATM-Deficiency-Induced Microglial Activation Promotes Neurodegeneration in Ataxia-Telangiectasia. Cell Rep. 2024, 43, 113622. [Google Scholar] [CrossRef] [PubMed]
- Broccoletti, T.; Del Giudice, E.; Cirillo, E.; Vigliano, I.; Giardino, G.; Ginocchio, V.M.; Bruscoli, S.; Riccardi, C.; Pignata, C. Efficacy of Very-Low-Dose Betamethasone on Neurological Symptoms in Ataxia-Telangiectasia: Efficacy of Betamethasone on Ataxia-Telangiectasia. Eur. J. Neurol. 2011, 18, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Quek, H.; Luff, J.; Cheung, K.; Kozlov, S.; Gatei, M.; Lee, C.S.; Bellingham, M.C.; Noakes, P.G.; Lim, Y.C.; Barnett, N.L.; et al. A Rat Model of Ataxia-Telangiectasia: Evidence for a Neurodegenerative Phenotype. Hum. Mol. Genet. 2016, 26, 109–123. [Google Scholar] [CrossRef]
- Ferro, A.; Sheeler, C.; Rosa, J.-G.; Cvetanovic, M. Role of Microglia in Ataxias. J. Mol. Biol. 2019, 431, 1792–1804. [Google Scholar] [CrossRef]
- Schafer, D.P.; Lehrman, E.K.; Kautzman, A.G.; Koyama, R.; Mardinly, A.R.; Yamasaki, R.; Ransohoff, R.M.; Greenberg, M.E.; Barres, B.A.; Stevens, B. Microglia Sculpt Postnatal Neural Circuits in an Activity and Complement-Dependent Manner. Neuron 2012, 74, 691–705. [Google Scholar] [CrossRef]
- Asthana, P.; Kumar, G.; Milanowski, L.M.; Au, N.P.B.; Chan, S.C.; Huang, J.; Feng, H.; Kwan, K.M.; He, J.; Chan, K.W.Y.; et al. Cerebellar Glutamatergic System Impacts Spontaneous Motor Recovery by Regulating Gria1 Expression. npj Regen. Med. 2022, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Fanciulli, A.; Wenning, G.K. Multiple-System Atrophy. N. Engl. J. Med. 2015, 372, 249–263. [Google Scholar] [CrossRef]
- Lee, H.-J.; Ricarte, D.; Ortiz, D.; Lee, S.-J. Models of Multiple System Atrophy. Exp. Mol. Med. 2019, 51, 139. [Google Scholar] [CrossRef]
- Nakamoto, F.K.; Okamoto, S.; Mitsui, J.; Sone, T.; Ishikawa, M.; Yamamoto, Y.; Kanegae, Y.; Nakatake, Y.; Imaizumi, K.; Ishiura, H.; et al. The Pathogenesis Linked to Coenzyme Q10 Insufficiency in iPSC-Derived Neurons from Patients with Multiple-System Atrophy. Sci. Rep. 2018, 8, 14215. [Google Scholar] [CrossRef] [PubMed]
- Mitchison, H.M.; Valente, E.M. Motile and Non-motile Cilia in Human Pathology: From Function to Phenotypes. J. Pathol. 2017, 241, 294–309. [Google Scholar] [CrossRef]
- De Mori, R.; Tardivo, S.; Pollara, L.; Giliani, S.C.; Ali, E.; Giordano, L.; Leuzzi, V.; Fischetto, R.; Gener, B.; Diprima, S.; et al. Joubert Syndrome-Derived Induced Pluripotent Stem Cells Show Altered Neuronal Differentiation in Vitro. Cell Tissue Res. 2024, 396, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Shimada, H.; Lu, Q.; Insinna-Kettenhofen, C.; Nagashima, K.; English, M.A.; Semler, E.M.; Mahgerefteh, J.; Cideciyan, A.V.; Li, T.; Brooks, B.P.; et al. In Vitro Modeling Using Ciliopathy-Patient-Derived Cells Reveals Distinct Cilia Dysfunctions Caused by CEP290 Mutations. Cell Rep. 2017, 20, 384–396. [Google Scholar] [CrossRef]
- Becker, E. Recent Advances in Modelling of Cerebellar Ataxia Using Induced Pluripotent Stem Cells. J. Neurol. Neuromed. 2017, 2, 11–15. [Google Scholar] [CrossRef]
- Doss, M.X.; Sachinidis, A. Current Challenges of iPSC-Based Disease Modeling and Therapeutic Implications. Cells 2019, 8, 403. [Google Scholar] [CrossRef] [PubMed]
- Haubenreich, C.; Lenz, M.; Schuppert, A.; Peitz, M.; Koch, P.; Zenke, M.; Brüstle, O. Epigenetic and Transcriptional Shifts in Human Neural Stem Cells after Reprogramming into Induced Pluripotent Stem Cells and Subsequent Redifferentiation. Int. J. Mol. Sci. 2024, 25, 3214. [Google Scholar] [CrossRef]
- Cerneckis, J.; Cai, H.; Shi, Y. Induced Pluripotent Stem Cells (iPSCs): Molecular Mechanisms of Induction and Applications. Signal Transduct. Target. Ther. 2024, 9, 112. [Google Scholar] [CrossRef] [PubMed]
- Valdes, P.; Henry, K.W.; Fitzgerald, M.Q.; Muralidharan, K.; Caldwell, A.B.; Ramachandran, S.; Goldstein, L.S.B.; Mobley, W.C.; Galasko, D.R.; Subramaniam, S. Limitations of the Human iPSC-Derived Neuron Model for Early-Onset Alzheimer’s Disease. Mol. Brain 2023, 16, 75. [Google Scholar] [CrossRef]
- Son, J.; Park, S.J.; Ha, T.; Lee, S.-N.; Cho, H.-Y.; Choi, J.-W. Electrophysiological Monitoring of Neurochemical-Based Neural Signal Transmission in a Human Brain–Spinal Cord Assembloid. ACS Sens. 2022, 7, 409–414. [Google Scholar] [CrossRef]
- Sun, X.; Kofman, S.; Ogbolu, V.C.; Karch, C.M.; Ibric, L.; Qiang, L. Vascularized Brain Assembloids with Enhanced Cellular Complexity Provide Insights Into the Cellular Deficits of Tauopathy. Stem Cells 2024, 42, 107–115. [Google Scholar] [CrossRef]
- Pantovic-Stefanovic, M.; Karanovic, J.; Jurisic, V.; Dunjic-Kostic, B.; Nesic, M.; Dodic, S.; Gostiljac, M.; Puric, M.; Savic Pavicevic, D.; Ivkovic, M. Mood Disorders and 5-HTR2A Genetic Variants—The Moderator Effect of Inflammation on Expression of Affective Polarity Phenotype. BMC Psychiatry 2024, 24, 747. [Google Scholar] [CrossRef]
- Pantovic-Stefanovic, M.; Petronijevic, N.; Dunjic-Kostic, B.; Velimirovic, M.; Jurisic, V.; Nikolic, T.; Dodic, S.; Ivkovic, M. Differentiating Stages of Bipolar and Unipolar Depression—The Possible Role of sICAM-1 and sVCAM-1. Cells 2024, 13, 1213. [Google Scholar] [CrossRef] [PubMed]



| Disease | Gene | Models | Main Results | References |
|---|---|---|---|---|
| Friedrich’s Ataxia | FXN | FRDA patients’ iPSC-derived neurons | Exenatide treatments enhance frataxin expression, improve oxidative stress and alleviate mitochondrial dysfunction. Frataxin deficiency-induced DNA damage leads to microglial activation. | [78,84,144] |
| Spinocerebellar Ataxia 1 (SCA1) | ATXN1 ISG15 | SCA1 patients’ iPSC-derived neurons and Purkinje cells | Expanded ATXN1 protein forms a hyperactive complex with CIC, leading to altered gene expression that contributes to cerebellar degeneration. ISG15 could be an early SCA1 biomarker. PKA-S776 signaling could be a promising therapeutic target, as PKA seems to be an upstream regulator of ATXN1. | [21,112,115] |
| Spinocerebellar Ataxia 2 (SCA2) | ATXN2 | SCA2 patients’ iPSC-derived neurons | Relevant screening platforms for evaluating new therapeutic targets. Disruptions to calcium channel function lead to excitotoxicity and impaired LTD. SCA2 NSCs suggest early developmental alterations. Transcriptome analysis reveals downregulation of glutamate receptor genes. | [88,90,93,100] |
| Spinocerebellar Ataxia 3 (Machado–Joseph Ataxia or SCA3) | ATXN3 | SCA3 patients’ iPSC-derived neurons, cerebellar cells and progenitors | Disrupted autophagy and chaperone pathways, with reduced Beclin-1 and DNAJB1 expressions. Mutant ATXN3 aggregation is more pronounced in mature neurons than in progenitors. AAV-based gene therapy using engineered miRNAs silences both mutant and wild-type ATXN3. N-butylidenephtalide reverses excitotoxic stress. Ibuprofen treatments reduce pro-inflammatory cytokine levels and decrease ATXN3 accumulation. IL-4 activates STAT6 signaling, thereby restoring proliferation and enhancing autophagy-related gene expression. Daidzein alleviates oxidative stress and apoptosis. | [118,120,124,127,128,129] |
| Spinocerebellar Ataxia 6 (SCA6) | CACNA1A | SCA6 patients’ iPSC-derived Purkinje cells | PolyQ-expanded α1ACT protein fails to translocate to the nucleus, reducing expression of neuroprotective genes. T3 decrement leads to simplified dendritic arborizations and cell death, rescued by TRH and Riluzole. Increased susceptibility to glutamate-induced excitotoxicity. Genetic ablation of MyD88 reduces microglial activation and slows disease progression. | [77,102,103,104,105,145] |
| Spinocerebellar Ataxia 42 (SCA42) | CACNA1G | SCA patients’ iPSC-derived Purkinje cells | Channel Cav3.1 exhibits a shift in the voltage sensitivity toward more depolarized potentials, disrupting calcium signaling. | [107] |
| Spinocerebellar Ataxia 14 (SCA14) | PRKCG | SCA14 patients’ iPSC-derived neurons | PKCγ fails to translocate properly to the membrane, leading to cytoplasmic aggregation and impaired degradation, resulting in membrane-level loss-of-function and cytoplasmic gain-of-function effects. | [108] |
| Spinocerebellar Ataxia 27B (SCA27B) | FGF14 | SCA27B-patient-derived iPSCs | A GAA-TCC intronic repeat expansion was identified, with larger expansions linked to reduced FGF14 expression, likely due to heterochromatin-mediated silencing. | [130] |
| Ataxia–Telangiectasia (A-T) | ATM | A-T patients’ iPSC-derived neurons | Olfactory mucosal biopsy for generating iPSC lines. Derived neurons analyses show impaired cerebellar development, synaptic signaling and oxidative stress pathways. Loss of ATM function leads to a hyperactive microglial phenotype, promoting neurodegeneration, which seems to be reduced by betamethasone treatment. | [131,132,133,146,148] |
| Autosomal Recessive Spastic Ataxia of Charlevoix–Saguenay | SACS | ARSACS patients’ iPSC-derived Purkinje cells and motor neurons | Derived neuron types revealed reduced sacsin protein expression and abnormal accumulation of NFM along neurites. | [136] |
| Peripheral Nerve Injury (PNI) | GRIA1 | SCA6 patients’ iPSCs-derived PCs and A-T patients’ cerebellar tissue | Reduced GRIA1 expression in ataxia patients disrupts the excitatory-inhibitory balance in DCN, impairing motor recovery after PNI. | [151] |
| Multiple System Atrophy (MSA) | MSA patients’ iPSC-derived neurons | Derived neurons show α-synuclein accumulation, glial cytoplasmic inclusions and alterations in neuroinflammatory responses | [153] | |
| Joubert Syndrome (JS) | CEP290 | CEP290-mutant iPSC-derived NPCs | Mutant cells show reduced expression of neural maturation markers, fail to form mature neurons and neuronal networks. | [156] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giacich, M.; Naef, V.; Santorelli, F.M.; Damiani, D. Roots of Progress: Uncovering Cerebellar Ataxias Using iPSC Models. Biomedicines 2025, 13, 2121. https://doi.org/10.3390/biomedicines13092121
Giacich M, Naef V, Santorelli FM, Damiani D. Roots of Progress: Uncovering Cerebellar Ataxias Using iPSC Models. Biomedicines. 2025; 13(9):2121. https://doi.org/10.3390/biomedicines13092121
Chicago/Turabian StyleGiacich, Michela, Valentina Naef, Filippo Maria Santorelli, and Devid Damiani. 2025. "Roots of Progress: Uncovering Cerebellar Ataxias Using iPSC Models" Biomedicines 13, no. 9: 2121. https://doi.org/10.3390/biomedicines13092121
APA StyleGiacich, M., Naef, V., Santorelli, F. M., & Damiani, D. (2025). Roots of Progress: Uncovering Cerebellar Ataxias Using iPSC Models. Biomedicines, 13(9), 2121. https://doi.org/10.3390/biomedicines13092121





