Establishing Human and Canine Xenograft Murine Osteosarcoma Models for Application of Focused Ultrasound Ablation
Abstract
1. Introduction
2. Materials and Methods
2.1. Xenograft Mouse Model
2.2. Limb Function Assessment
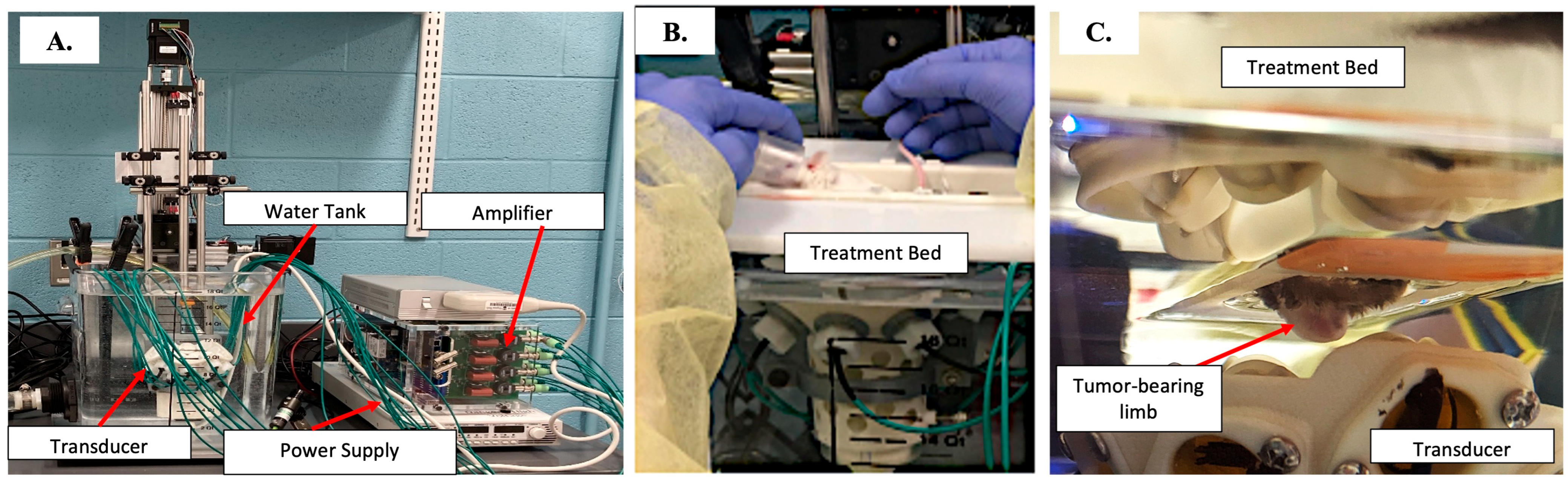
2.3. Histotripsy Treatment
2.4. Tissue Collection and Histology
2.5. Fecal Corticosterone
2.6. Magnetic Resonance Imaging (MRI)
2.7. Statistical Analysis
3. Results
3.1. Human Xenograft Model
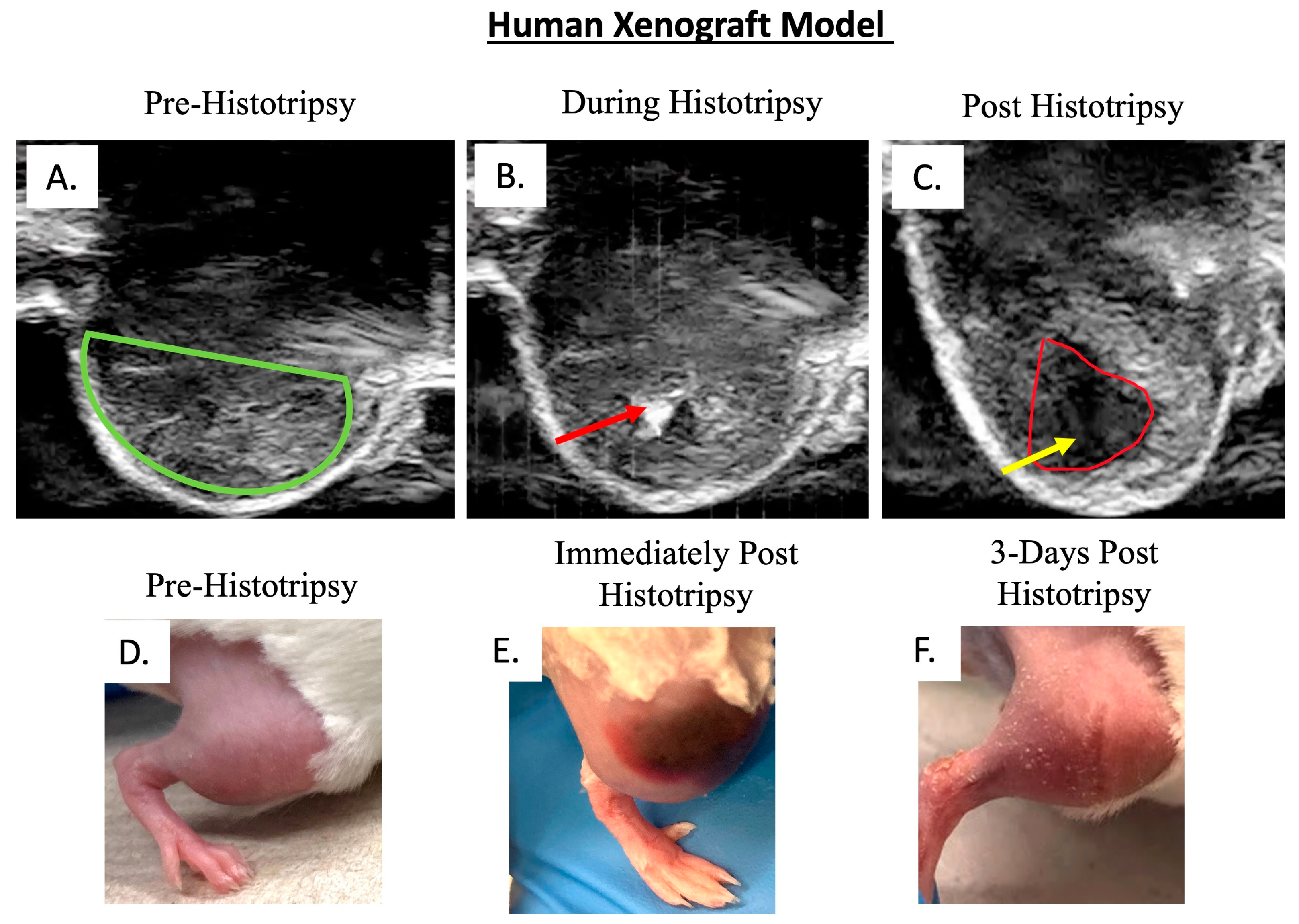
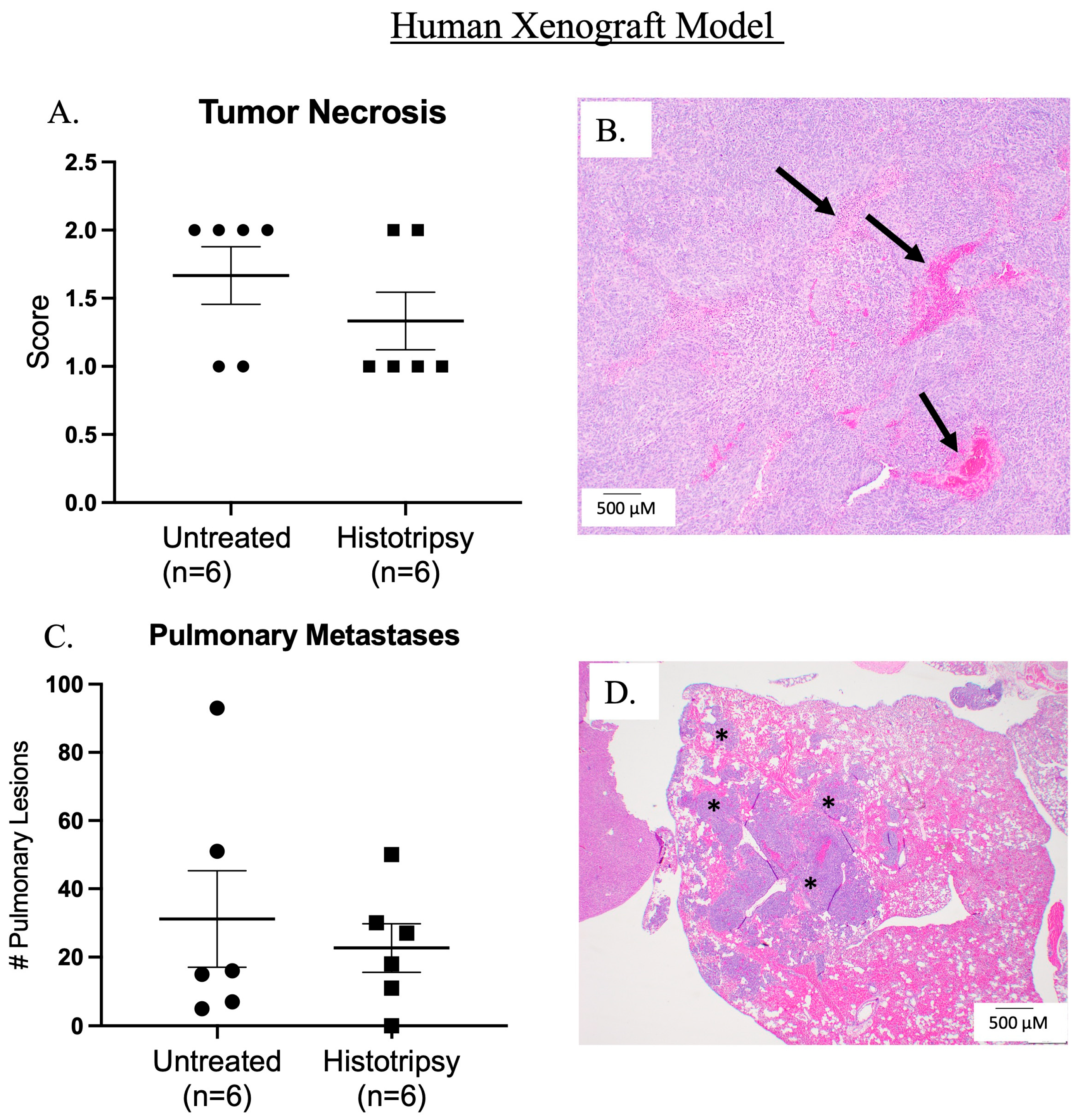
3.1.1. Ablative and Histological Outcomes; Histotripsy Results in Tumor Ablation
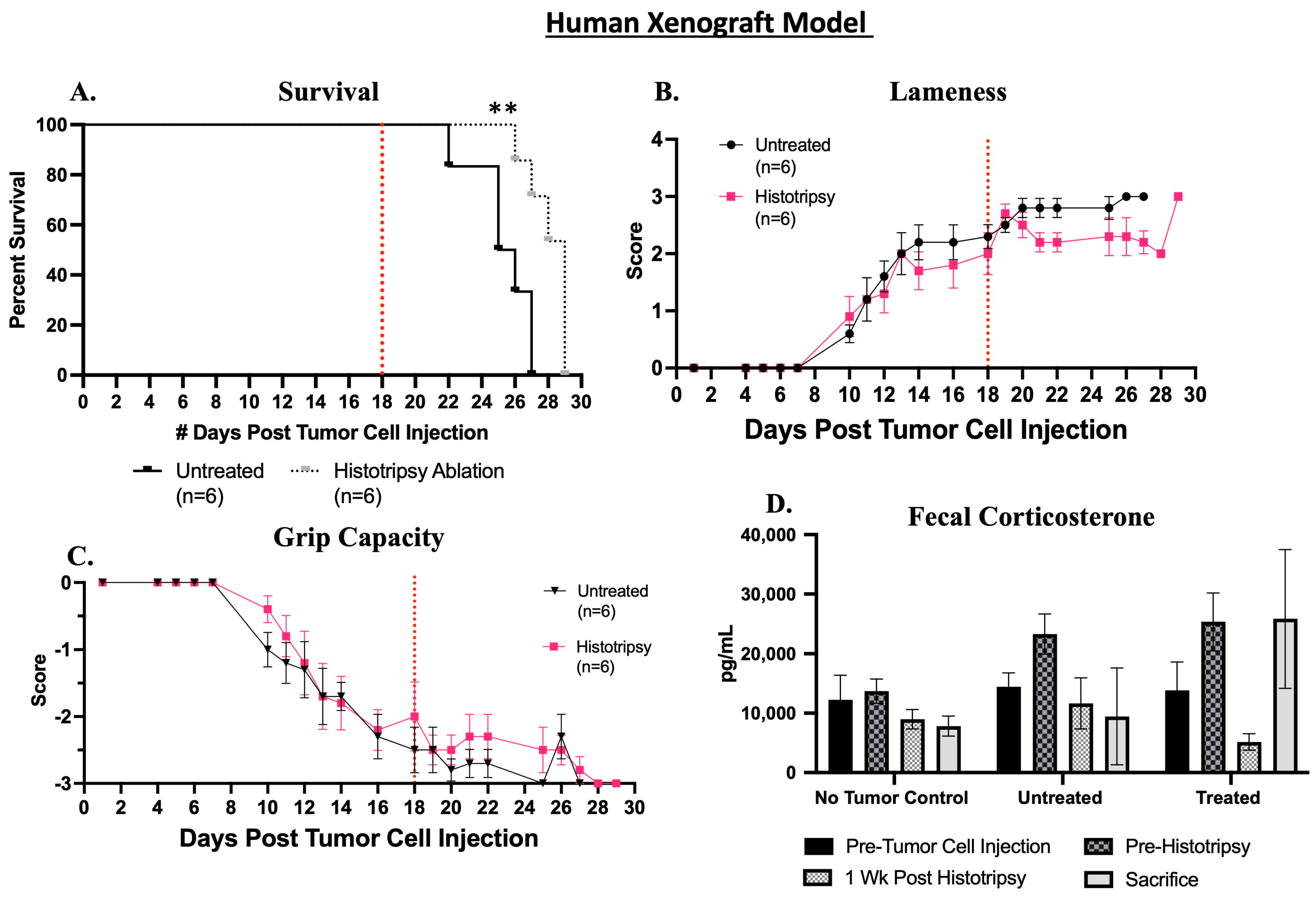
3.1.2. Clinical and Tumor-Bearing Limb Functionality Outcomes; Histotripsy Alters Tumor-Bearing Limb Function
3.2. Canine Xenograft Model
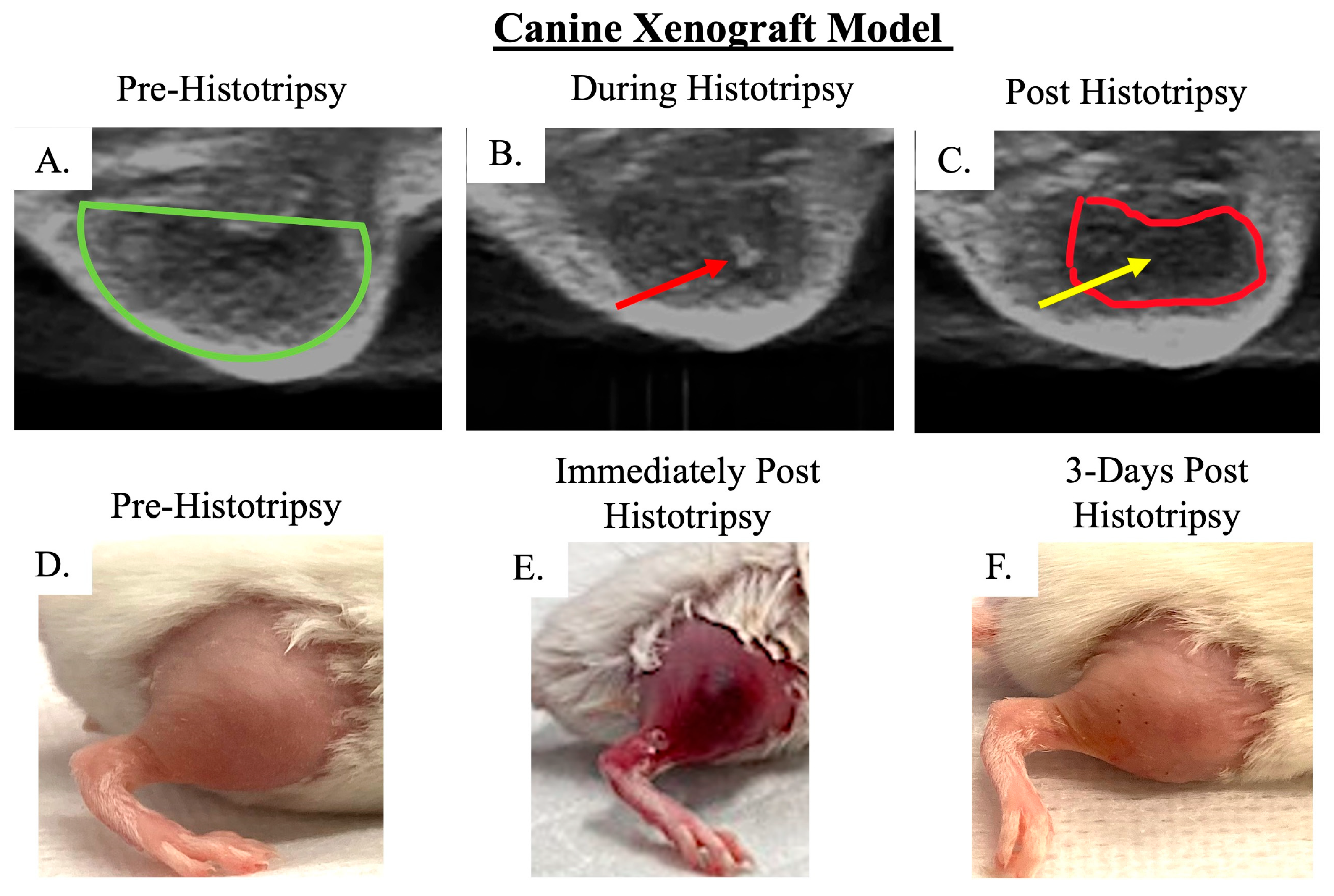
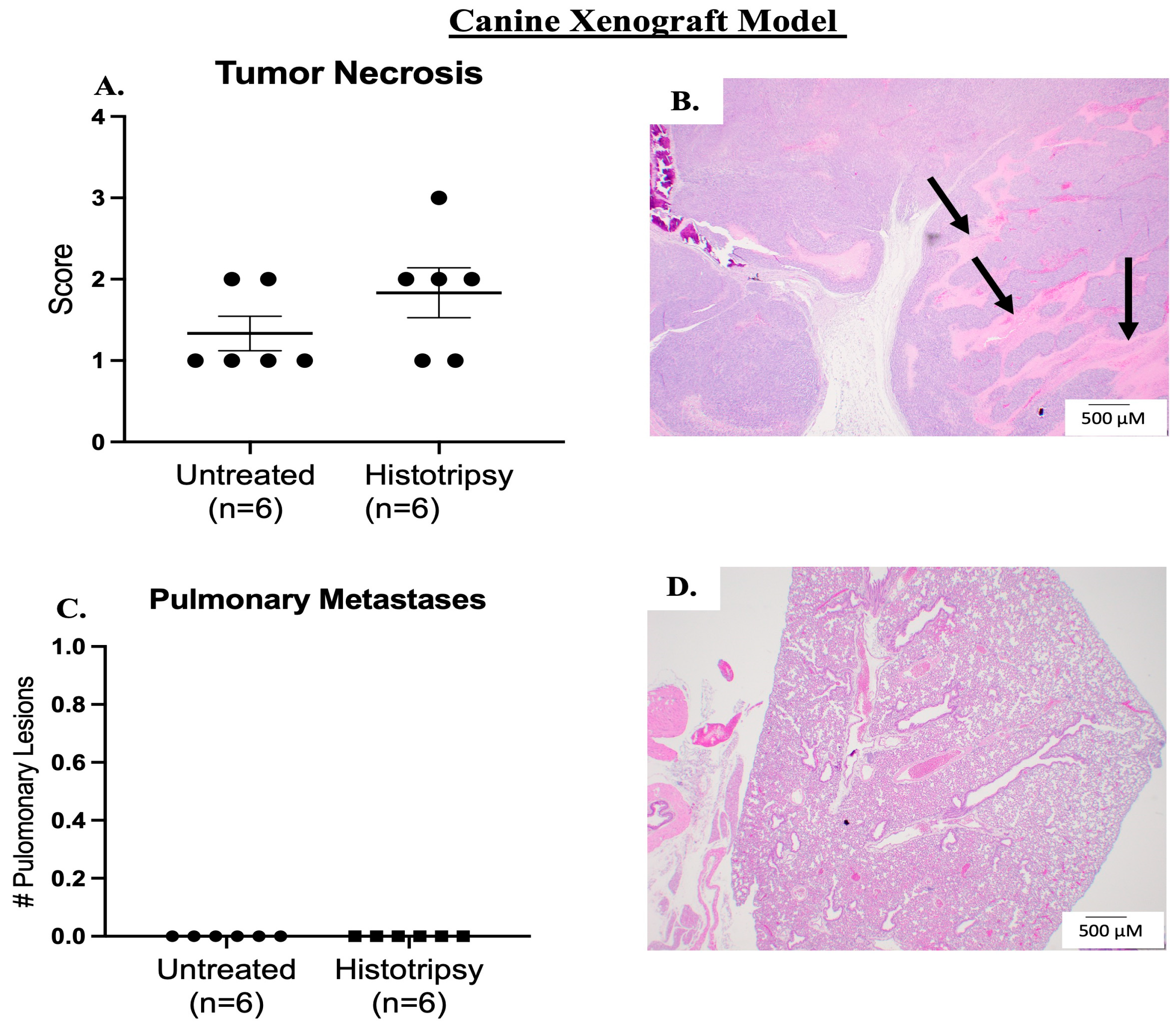
3.2.1. Ablative and Histological Outcomes; Histotripsy Results in Tumor Ablation
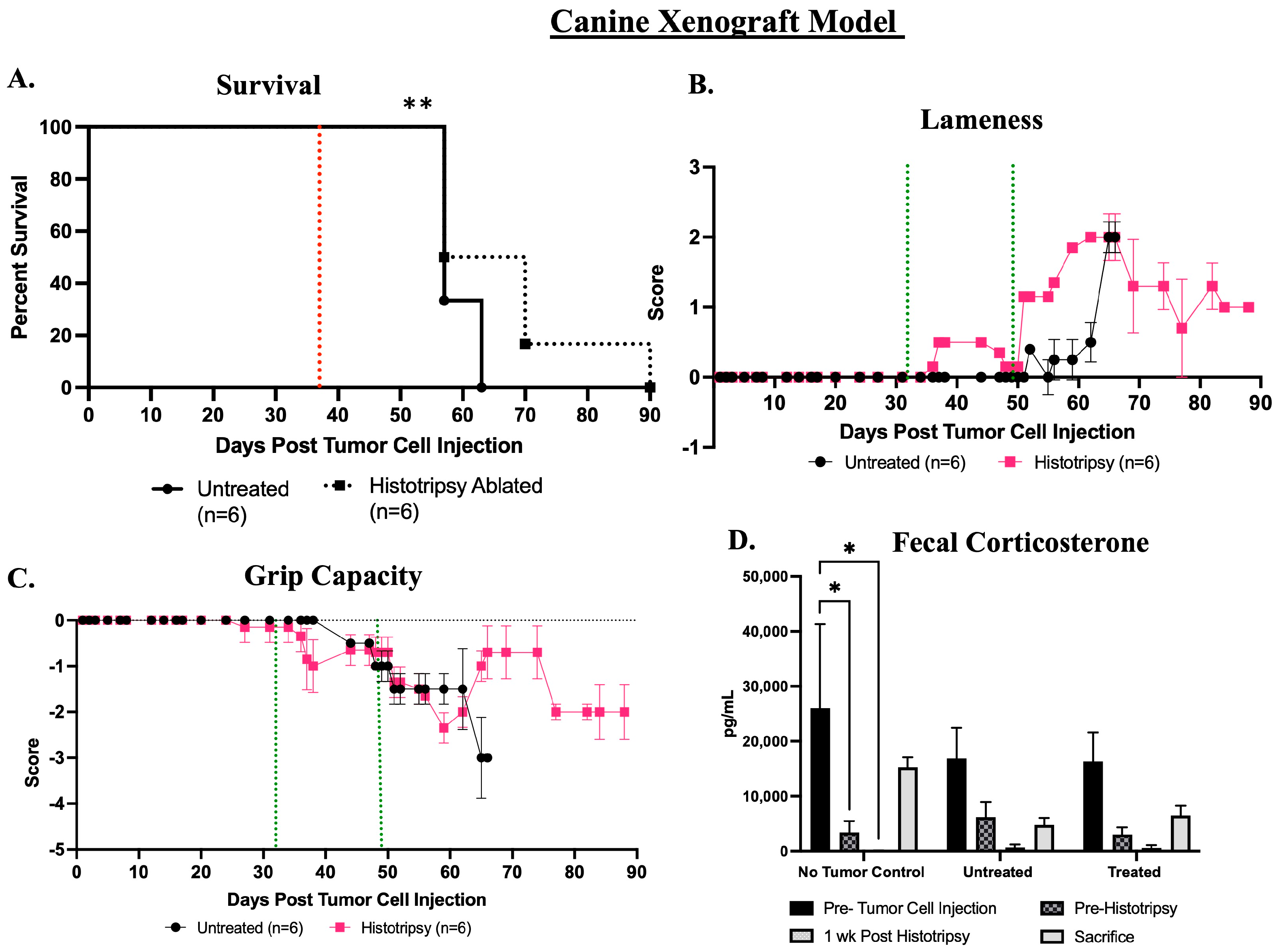
3.2.2. Clinical and Tumor-Bearing Limb Functionality Outcomes; Histotripsy Alters Tumor-Bearing Limb Function
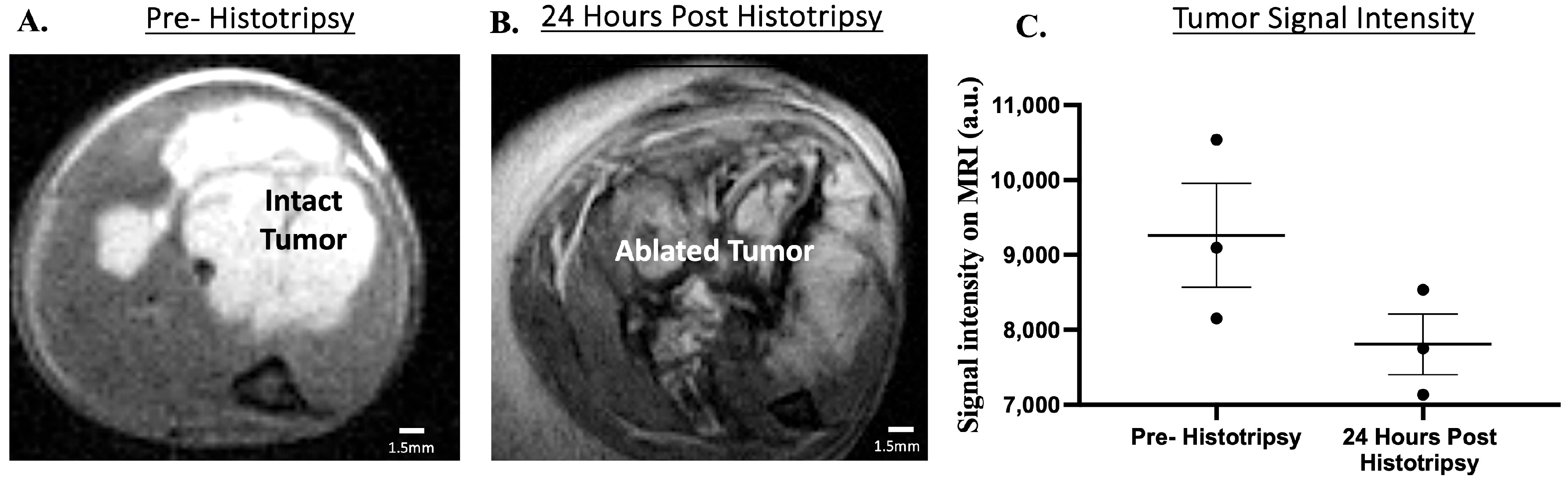
3.2.3. Investigation of Primary Tumor Monitoring with Magnetic Resonance Imaging; Histotripsy Ablation Is Evident on MRI
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simpson, S.; Dunning, M.D.; de Brot, S.; Grau-Roma, L.; Mongan, N.P.; Rutland, C.S. Comparative review of human and canine osteosarcoma: Morphology, epidemiology, prognosis, treatment and genetics. Acta Vet. Scand. 2017, 59, 71. [Google Scholar] [CrossRef] [PubMed]
- Mirabello, L.; Troisi, R.J.; Savage, S.A. Osteosarcoma incidence and survival rates from 1973 to 2004: Data from the Surveillance, Epidemiology, and End Results Program. Cancer 2009, 115, 1531–1543. [Google Scholar] [CrossRef]
- Ghert, M.; Mwita, W.; Mandari, F.N. Primary Bone Tumors in Children and Adolescents Treated at a Referral Center in Northern Tanzania. J. Am. Acad. Orthop. Surg. Glob. Res. Rev. 2019, 3, e045. [Google Scholar] [CrossRef]
- Jafari, F.; Javdansirat, S.; Sanaie, S.; Naseri, A.; Shamekh, A.; Rostamzadeh, D.; Dolati, S. Osteosarcoma: A comprehensive review of management and treatment strategies. Ann. Diagn. Pathol. 2020, 49, 151654. [Google Scholar] [CrossRef] [PubMed]
- Marko, T.A.; Diessner, B.J.; Spector, L.G. Prevalence of Metastasis at Diagnosis of Osteosarcoma: An International Comparison. Pediatr. Blood Cancer 2016, 63, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Mason, N.J. Comparative Immunology and Immunotherapy of Canine Osteosarcoma. Adv. Exp. Med. Biol. 2020, 1258, 199–221. [Google Scholar]
- Fenger, J.M.; London, C.A.; Kisseberth, W.C. Canine osteosarcoma: A naturally occurring disease to inform pediatric oncology. ILAR J. 2014, 55, 69–85. [Google Scholar] [CrossRef]
- Tarone, L.; Giacobino, D.; Camerino, M.; Maniscalco, L.; Iussich, S.; Parisi, L.; Giovannini, G.; Dentini, A.; Bolli, E.; Quaglino, E.; et al. A chimeric human/dog-DNA vaccine against CSPG4 induces immunity with therapeutic potential in comparative preclinical models of osteosarcoma. Mol. Ther. 2023, 31, 2342–2359. [Google Scholar] [CrossRef]
- Oblinger, J.L.; Wang, J.; Wetherell, G.D.; Agarwal, G.; Wilson, T.A.; Benson, N.R.; Fenger, J.M.; Fuchs, J.R.; Kinghorn, A.D.; Chang, L.-S. Anti-tumor effects of the eIF4A inhibitor didesmethylrocaglamide and its derivatives in human and canine osteosarcomas. Sci. Rep. 2024, 14, 19349. [Google Scholar] [CrossRef]
- Blattmann, C.; Thiemann, M.; Stenzinger, A.; Roth, E.K.; Dittmar, A.; Witt, H.; Lehner, B.; Renker, E.; Jugold, M.; Eichwald, V.; et al. Establishment of a patient-derived orthotopic osteosarcoma mouse model. J. Transl. Med. 2015, 13, 136. [Google Scholar] [CrossRef]
- Beck, J.; Ren, L.; Huang, S.; Berger, E.; Bardales, K.; Mannheimer, J.; Mazcko, C.; LeBlanc, A. Canine and murine models of osteosarcoma. Vet. Pathol. 2022, 59, 399–414. [Google Scholar] [CrossRef]
- Stewart, E.; Federico, S.M.; Chen, X.; Shelat, A.A.; Bradley, C.; Gordon, B.; Karlstrom, A.; Twarog, N.R.; Clay, M.R.; Bahrami, A.; et al. Orthotopic patient-derived xenografts of paediatric solid tumours. Nature 2017, 549, 96–100. [Google Scholar] [CrossRef]
- Scott, M.C.; Tomiyasu, H.; Garbe, J.R.; Cornax, I.; Amaya, C.; O’Sullivan, M.G.; Subramanian, S.; Bryan, B.A.; Modiano, J.F. Heterotypic mouse models of canine osteosarcoma recapitulate tumor heterogeneity and biological behavior. Dis. Model. Mech. 2016, 9, 1435–1444. [Google Scholar] [CrossRef]
- Xu, Z.; Khokhlova, T.D.; Cho, C.S.; Khokhlova, V.A. Histotripsy: A Method for Mechanical Tissue Ablation with Ultrasound. Annu. Rev. Biomed. Eng. 2024, 26, 141–167. [Google Scholar] [CrossRef]
- Xu, Z.; Hall, T.L.; Vlaisavljevich, E.; Lee, F.T., Jr. Histotripsy: The first noninvasive, non-ionizing, non-thermal ablation technique based on ultrasound. Int. J. Hyperth. 2021, 38, 561–575. [Google Scholar] [CrossRef] [PubMed]
- Ruger, L.N.; Hay, A.N.; Gannon, J.M.; Sheppard, H.O.; Coutermarsh-Ott, S.L.; Daniel, G.B.; Kierski, K.R.; Ciepluch, B.J.; Vlaisavljevich, E.; Tuohy, J.L. Histotripsy Ablation of Spontaneously Occurring Canine Bone Tumors In Vivo. IEEE Trans. Biomed. Eng. 2022, 70, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Ruger, L.N.; Hay, A.N.; Vickers, E.R.; Coutermarsh-Ott, S.L.; Gannon, J.M.; Covell, H.S.; Daniel, G.B.; Laeseke, P.F.; Ziemlewicz, T.J.; Kierski, K.R.; et al. Characterizing the Ablative Effects of Histotripsy for Osteosarcoma: In Vivo Study in Dogs. Cancers 2023, 15, 741. [Google Scholar] [CrossRef] [PubMed]
- Arnold, L.; Hendricks-Wenger, A.; Coutermarsh-Ott, S.; Gannon, J.; Hay, A.N.; Dervisis, N.; Klahn, S.; Allen, I.C.; Tuohy, J.; Vlaisavljevich, E. Histotripsy Ablation of Bone Tumors: Feasibility Study in Excised Canine Osteosarcoma Tumors. Ultrasound Med. Biol. 2021, 47, 3435–3446. [Google Scholar] [CrossRef]
- Iwanicki, I.; Wu, L.L.; Flores-Guzman, F.; Sundland, R.; Viza-Gomes, P.; Nordgren, R.; Centner, C.S.; Kandel, J.J.; Applebaum, M.A.; Bader, K.B.; et al. Histotripsy induces apoptosis and reduces hypoxia in a neuroblastoma xenograft model. Int. J. Hyperth. 2023, 40, 2222941. [Google Scholar] [CrossRef]
- Hendricks-Wenger, A.; Saunier, S.; Simon, A.; Grider, D.; Luyimbazi, D.; Allen, I.C.; Vlaisavljevich, E. Histotripsy for the Treatment of Cholangiocarcinoma in a Patient-Derived Xenograft Mouse Model. Ultrasound Med. Biol. 2022, 48, 293–303. [Google Scholar] [CrossRef]
- Worlikar, T.; Vlaisavljevich, E.; Gerhardson, T.; Greve, J.; Wan, S.; Kuruvilla, S.; Lundt, J.; Ives, K.; Hall, T.; Welling, T.H.; et al. Histotripsy for Non-Invasive Ablation of Hepatocellular Carcinoma (HCC) Tumor in a Subcutaneous Xenograft Murine Model. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 6064–6067. [Google Scholar]
- Hay, A.N.; Imran, K.M.; Hendricks-Wenger, A.; Gannon, J.M.; Sereno, J.; Simon, A.; Lopez, V.A.; Coutermarsh-Ott, S.; Vlaisavljevich, E.; Allen, I.C.; et al. Ablative and Immunostimulatory Effects of Histotripsy Ablation in a Murine Osteosarcoma Model. Biomedicines 2023, 11, 2737. [Google Scholar] [CrossRef]
- Hendricks-Wenger, A.; Sereno, J.; Gannon, J.; Zeher, A.; Brock, R.M.; Beitel-White, N.; Simon, A.; Davalos, R.V.; Coutermarsh-Ott, S.; Vlaisavljevich, E.; et al. Histotripsy Ablation Alters the Tumor Microenvironment and Promotes Immune System Activation in a Subcutaneous Model of Pancreatic Cancer. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 68, 2987–3000. [Google Scholar] [CrossRef]
- Miwa, S.; Hiroshima, Y.; Yano, S.; Zhang, Y.; Matsumoto, Y.; Uehara, F.; Yamamoto, M.; Kimura, H.; Hayashi, K.; Bouvet, M.; et al. Fluorescence-guided surgery improves outcome in an orthotopic osteosarcoma nude-mouse model. J. Orthop. Res. 2014, 32, 1596–1601. [Google Scholar] [CrossRef]
- Jacques, C.; Renema, N.; Lezot, F.; Ory, B.; Walkley, C.R.; Grigoriadis, A.E.; Heymann, D. Small animal models for the study of bone sarcoma pathogenesis:characteristics, therapeutic interests and limitations. J. Bone Oncol. 2018, 12, 7–13. [Google Scholar] [CrossRef]
- Sottnik, J.L.; Duval, D.L.; Ehrhart, E.J.; Thamm, D.H. An orthotopic, postsurgical model of luciferase transfected murine osteosarcoma with spontaneous metastasis. Clin. Exp. Metastasis 2010, 27, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Hay, A.N.; Ruger, L.; Hsueh, A.; Vickers, E.; Klahn, S.; Vlaisavljevich, E.; Tuohy, J. A review of the development of histotripsy for extremity tumor ablation with a canine comparative oncology model to inform human treatments. Int. J. Hyperth. 2023, 40, 2274802. [Google Scholar] [CrossRef] [PubMed]
- Vlaisavljevich, E.; Greve, J.; Cheng, X.; Ives, K.; Shi, J.; Jin, L.; Arvidson, A.; Hall, T.; Welling, T.H.; Owens, G.; et al. Non-Invasive Ultrasound Liver Ablation Using Histotripsy: Chronic Study in an In Vivo Rodent Model. Ultrasound Med. Biol. 2016, 42, 1890–1902. [Google Scholar] [CrossRef]
- Cruz Lebrón, A.; Blackwell, E.; Balbuena Almodóvar, P.; Syakirah Faiez, T.; Mummert, L.A.; Sfanos, K.S. Use of Micropipette-Guided Drug Administration as an Alternative Method to Oral Gavage in Rodent Models. J. Vis. Exp. 2024, 209, e66836. [Google Scholar] [CrossRef]
- Sidwell, A.B.; Girard, B.M.; Campbell, S.E.; Vizzard, M.A. TRPV1 and mast cell involvement in repeated variate stress-induced urinary bladder dysfunction in adult female mice. Am. J. Physiol. Renal Physiol. 2024, 327, F476–F488. [Google Scholar] [CrossRef]
- Bazzocchi, A.; Aparisi Gómez, M.P.; Taninokuchi Tomassoni, M.; Napoli, A.; Filippiadis, D.; Guglielmi, G. Musculoskeletal oncology and thermal ablation: The current and emerging role of interventional radiology. Skelet. Radiol. 2023, 52, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhang, W.; Fan, W.; Huang, J.; Zhang, F.; Wu, P. Noninvasive treatment of malignant bone tumors using high-intensity focused ultrasound. Cancer 2010, 116, 3934–3942. [Google Scholar] [CrossRef]
- Scipione, R.; Anzidei, M.; Bazzocchi, A.; Gagliardo, C.; Catalano, C.; Napoli, A. HIFU for Bone Metastases and other Musculoskeletal Applications. Semin. Interv. Radiol. 2018, 35, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Baal, J.D.; Chen, W.C.; Baal, U.; Wagle, S.; Baal, J.H.; Link, T.M.; Bucknor, M.D. Efficacy and safety of magnetic resonance-guided focused ultrasound for the treatment of painful bone metastases: A systematic review and meta-analysis. Skelet. Radiol. 2021, 50, 2459–2469. [Google Scholar] [CrossRef]
- Nascimento, D.; Suchard, G.; Hatem, M.; de Abreu, A. The role of magnetic resonance imaging in the evaluation of bone tumours and tumour-like lesions. Insights Imaging 2014, 5, 419–440. [Google Scholar] [CrossRef]
- Cè, M.; Cellina, M.; Ueanukul, T.; Carrafiello, G.; Manatrakul, R.; Tangkittithaworn, P.; Jaovisidha, S.; Fuangfa, P.; Resnick, D. Multimodal Imaging of Osteosarcoma: From First Diagnosis to Radiomics. Cancers 2025, 17, 599. [Google Scholar] [CrossRef]
- Zhu, J.; Sun, K.; Shi, Q.; Huang, S.; Li, D.; Guo, W.; Tang, X.; Ji, T. Magnetic resonance imaging assessment of epiphyseal invasion in paediatric osteosarcoma. Skelet. Radiol. 2025, 54, 2077–2085. [Google Scholar] [CrossRef]
- Brennecke, P.; Arlt, M.J.; Campanile, C.; Husmann, K.; Gvozdenovic, A.; Apuzzo, T.; Thelen, M.; Born, W.; Fuchs, B. CXCR4 antibody treatment suppresses metastatic spread to the lung of intratibial human osteosarcoma xenografts in mice. Clin. Exp. Metastasis 2014, 31, 339–349. [Google Scholar] [CrossRef]
- Yuan, J.; Ossendorf, C.; Szatkowski, J.P.; Bronk, J.T.; Maran, A.; Yaszemski, M.; Bolander, M.E.; Sarkar, G.; Fuchs, B. Osteoblastic and osteolytic human osteosarcomas can be studied with a new xenograft mouse model producing spontaneous metastases. Cancer Investig. 2009, 27, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Ohishi, T.; Kaneko, M.K.; Takei, J.; Mizuno, T.; Kawada, M.; Saito, M.; Suzuki, H.; Kato, Y. Defucosylated Mouse-Dog Chimeric Anti-EGFR Antibody Exerts Antitumor Activities in Mouse Xenograft Models of Canine Tumors. Cells 2021, 10, 3599. [Google Scholar] [CrossRef]
- Chaffee, B.K.; Allen, M.J. A clinically relevant mouse model of canine osteosarcoma with spontaneous metastasis. In Vivo 2013, 27, 599–603. [Google Scholar] [PubMed]
- Barroga, E.F.; Kadosawa, T.; Okumura, M.; Fujinaga, T. Establishment and characterization of the growth and pulmonary metastasis of a highly lung metastasizing cell line from canine osteosarcoma in nude mice. J. Vet. Med. Sci. 1999, 61, 361–367. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Higuchi, T.; Igarashi, K.; Yamamoto, N.; Hayashi, K.; Kimura, H.; Miwa, S.; Bouvet, M.; Tsuchiya, H.; Hoffman, R.M. Osteosarcoma Patient-derived Orthotopic Xenograft (PDOX) Models Used to Identify Novel and Effective Therapeutics: A Review. Anticancer Res. 2021, 41, 5865–5871. [Google Scholar] [CrossRef]
- Zheng, B.; Ren, T.; Huang, Y.; Sun, K.; Wang, S.; Bao, X.; Liu, K.; Guo, W. PD-1 axis expression in musculoskeletal tumors and antitumor effect of nivolumab in osteosarcoma model of humanized mouse. J. Hematol. Oncol. 2018, 11, 16. [Google Scholar] [CrossRef]
- Johan, M.P.; Kubo, T.; Furuta, T.; Sakuda, T.; Sakaguchi, T.; Nakanishi, M.; Ochi, M.; Adachi, N. Metastatic tumor cells detection and anti-metastatic potential with vesicular stomatitis virus in immunocompetent murine model of osteosarcoma. J. Orthop. Res. 2018, 36, 2562–2569. [Google Scholar] [CrossRef] [PubMed]
- Khanna, C.; Prehn, J.; Yeung, C.; Caylor, J.; Tsokos, M.; Helman, L. An orthotopic model of murine osteosarcoma with clonally related variants differing in pulmonary metastatic potential. Clin. Exp. Metastasis 2000, 18, 261–271. [Google Scholar] [CrossRef] [PubMed]
| Lameness | Grip Capacity * | ||
|---|---|---|---|
| 0 | No lameness | 0 | Normal grip |
| 1 | Slight gait abnormality. | −1 | Mild reduction |
| 2 | Full range of motion but moderate gait abnormality (ex. Limping). Sensation in toes present. | −2 | Severe reduction |
| 3 | Impaired range of motion. Sensation in toes present. | −3 | No Grip capacity (Endpoint) |
| 4 | Not weight bearing, absence of sensation in toes (End point). | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hay, A.N.; Simon, A.; Ruger, L.N.; Gannon, J.; Coutermarsh-Ott, S.; Vickers, E.R.; Eward, W.; Neufeld, N.J.; Vlaisavljevich, E.; Tuohy, J. Establishing Human and Canine Xenograft Murine Osteosarcoma Models for Application of Focused Ultrasound Ablation. Biomedicines 2025, 13, 2122. https://doi.org/10.3390/biomedicines13092122
Hay AN, Simon A, Ruger LN, Gannon J, Coutermarsh-Ott S, Vickers ER, Eward W, Neufeld NJ, Vlaisavljevich E, Tuohy J. Establishing Human and Canine Xenograft Murine Osteosarcoma Models for Application of Focused Ultrasound Ablation. Biomedicines. 2025; 13(9):2122. https://doi.org/10.3390/biomedicines13092122
Chicago/Turabian StyleHay, Alayna N., Alex Simon, Lauren N. Ruger, Jessica Gannon, Sheryl Coutermarsh-Ott, Elliana R. Vickers, William Eward, Nathan J. Neufeld, Eli Vlaisavljevich, and Joanne Tuohy. 2025. "Establishing Human and Canine Xenograft Murine Osteosarcoma Models for Application of Focused Ultrasound Ablation" Biomedicines 13, no. 9: 2122. https://doi.org/10.3390/biomedicines13092122
APA StyleHay, A. N., Simon, A., Ruger, L. N., Gannon, J., Coutermarsh-Ott, S., Vickers, E. R., Eward, W., Neufeld, N. J., Vlaisavljevich, E., & Tuohy, J. (2025). Establishing Human and Canine Xenograft Murine Osteosarcoma Models for Application of Focused Ultrasound Ablation. Biomedicines, 13(9), 2122. https://doi.org/10.3390/biomedicines13092122






