Immunomodulatory Effects of High-Dose Irradiation Regimens in Renal Cell Carcinoma: Insights from an In Vitro Model with Human Peripheral Blood Mononuclear Cell
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Culture Conditions
2.2. In Vitro Irradiation Protocol
2.3. Colonogenic Assay
2.4. Cell Viability Assessment
2.5. PBMC Proliferation Assay
2.6. Flow Cytometry
2.7. Cytokine Quantification by ELISA
2.8. Real-Time Quantification Polymerase Chain Reaction Assays (RTqPCR)
2.9. Statistical Analysis
3. Results
3.1. Direct Impact of High-Dose Irradiation on Cancer Cells
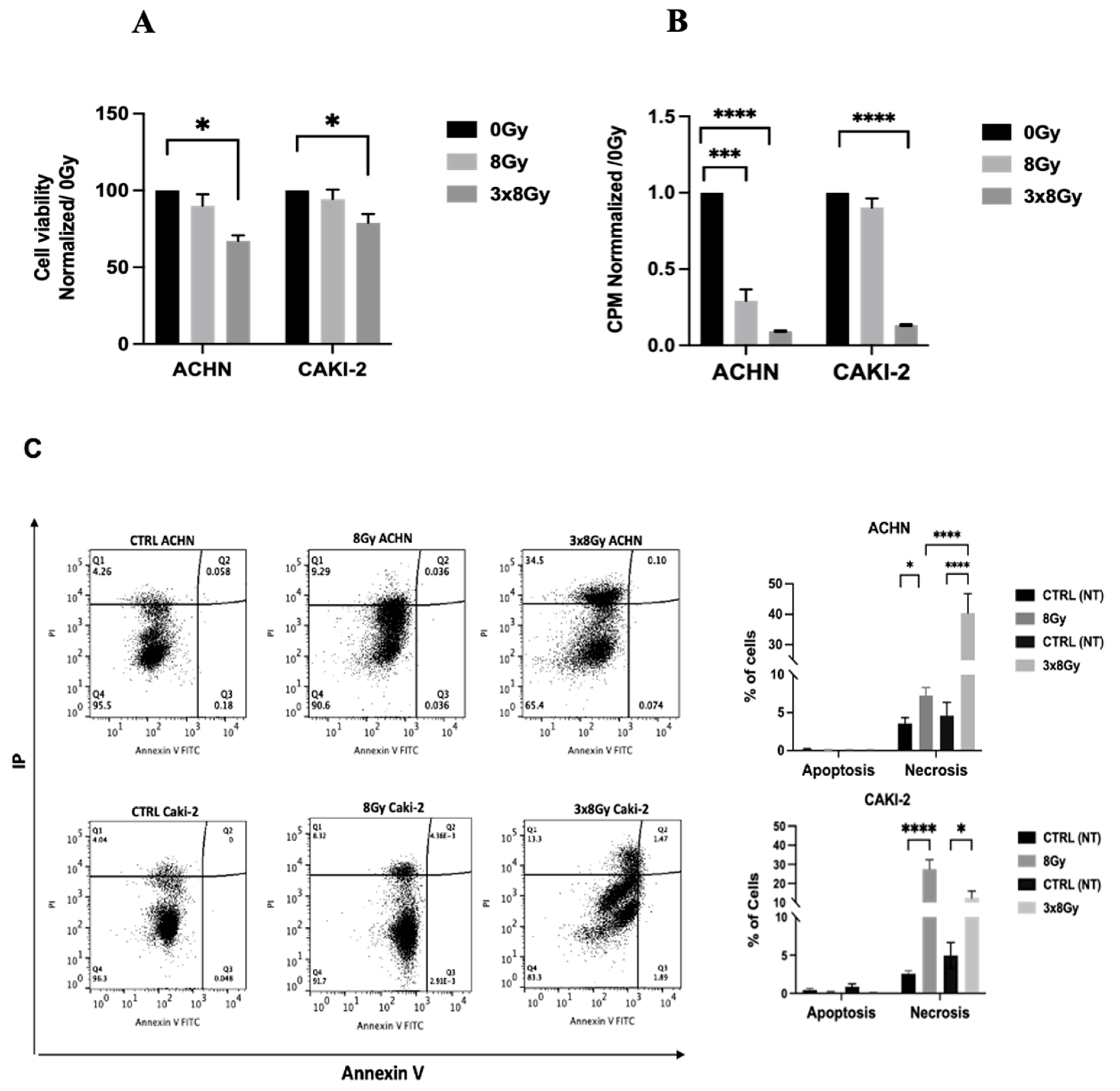
3.1.1. High-Dose Irradiation Induced Distinct Effects on Radiosensitivity in RCC Cell Lines
3.1.2. High-Dose Irradiation Reduced Cell Viability and Proliferation in Both Cell Lines
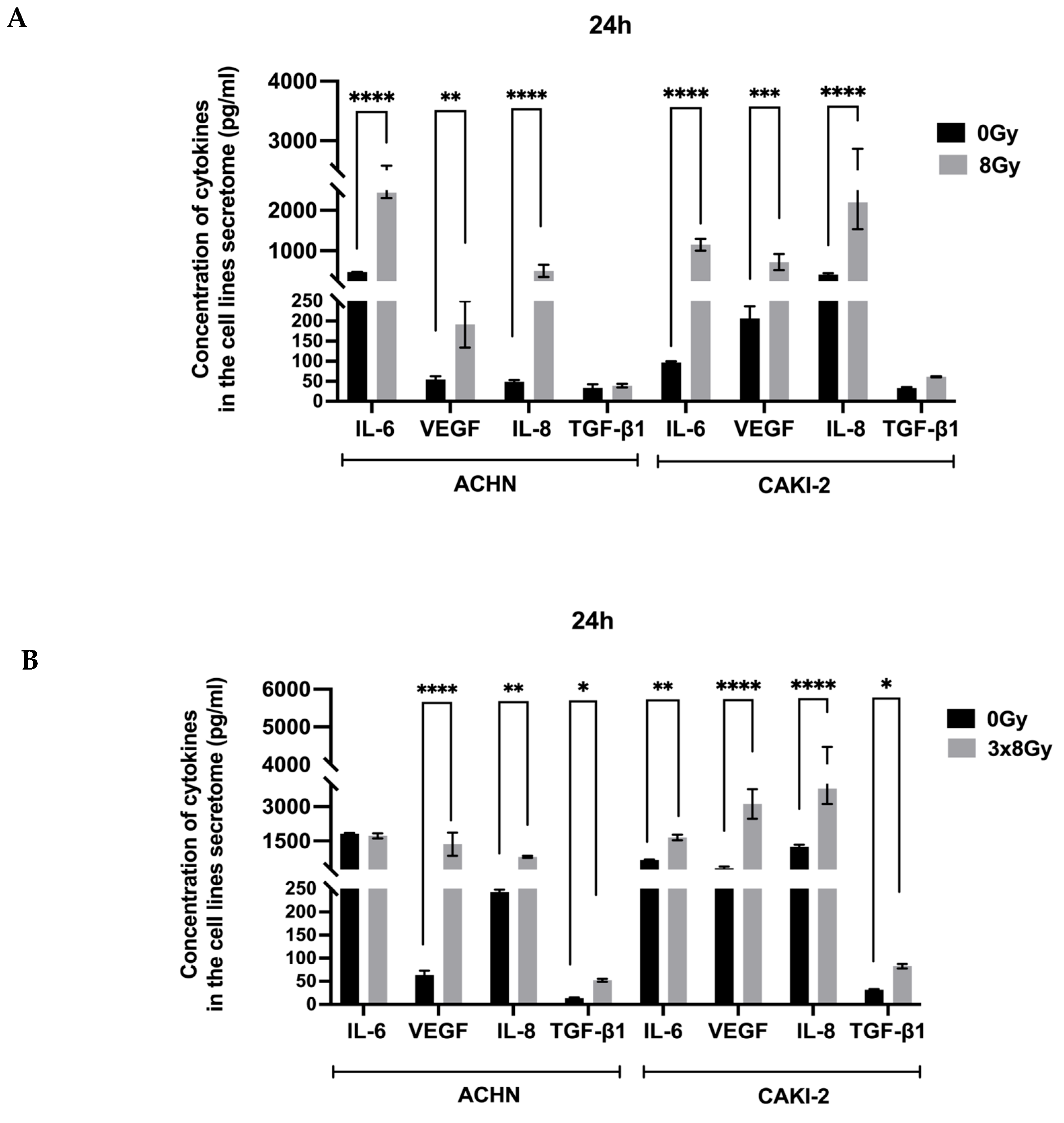
3.1.3. High-Dose Irradiation Increased the Release of VEGF and Inflammatory Cytokines
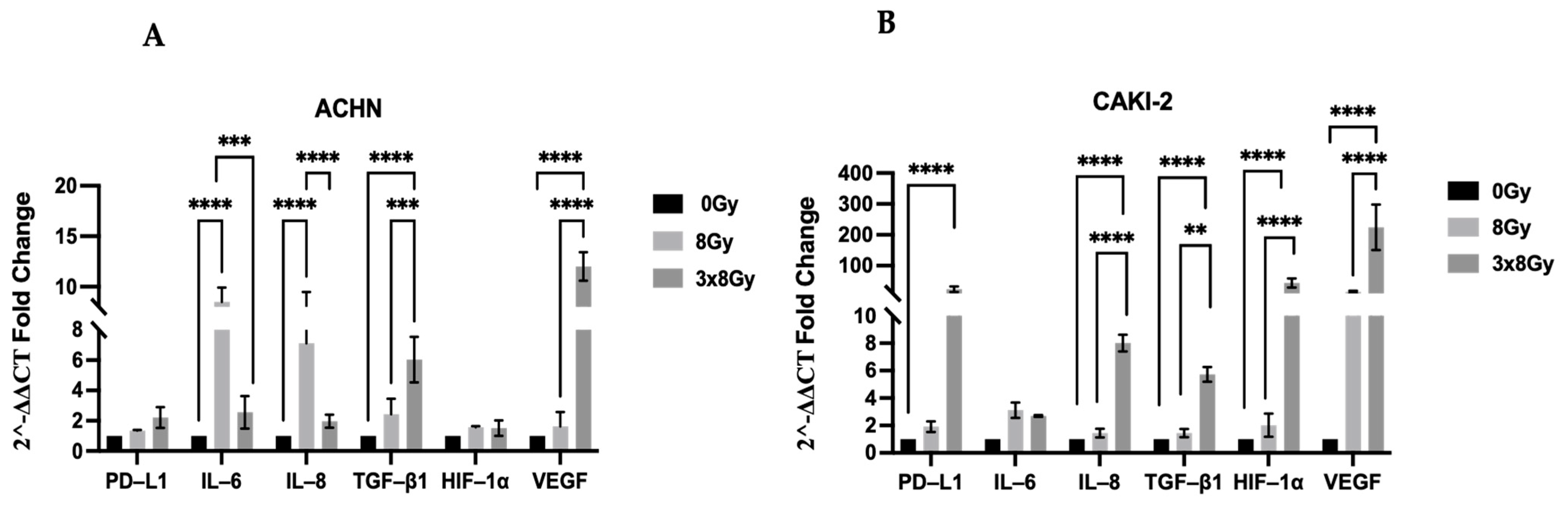
3.1.4. High-Dose Effect on Transcriptomic Expression of PD-L1 and Inflammatory Cytokines
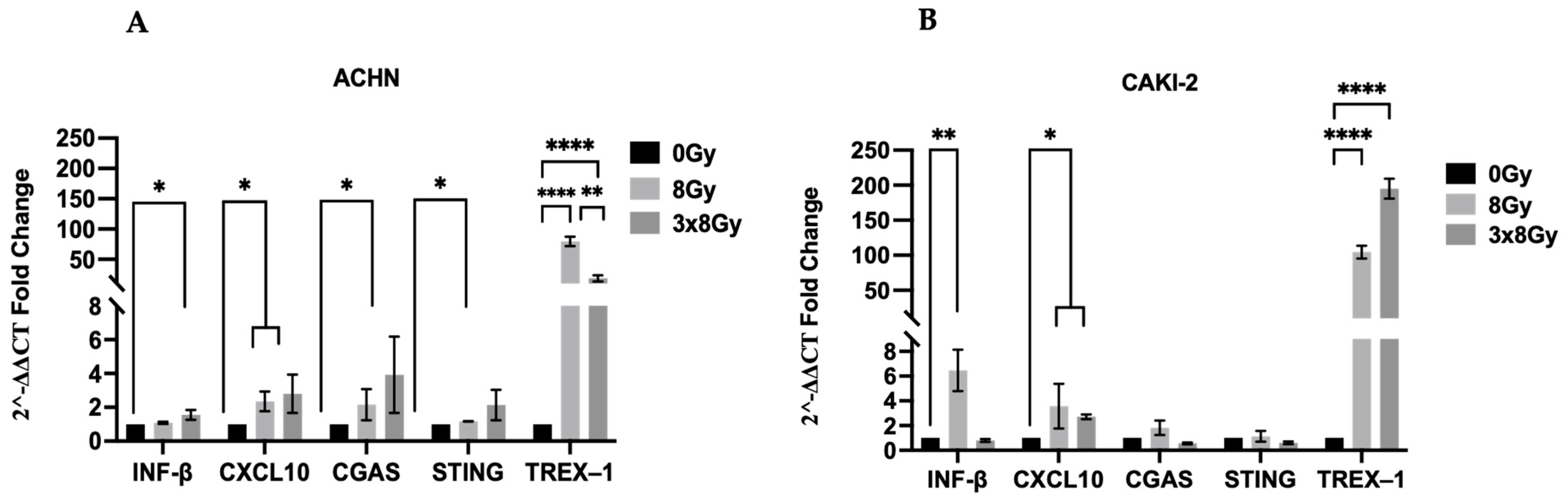
3.1.5. Influence of High Dose Irradiation on the Induction of Type I Interferon and c-Gas/Sting Pathway
3.2. Indirect Impact of High-Dose Irradiation on PBMCs
3.2.1. Effect of RCC’s Lines Irradiated Supernatants on PBMCS Viability and Proliferation
3.2.2. Impact of Irradiated Supernatants from RCC Cell Lines on Cytokine Release in Circulating Immune Cells
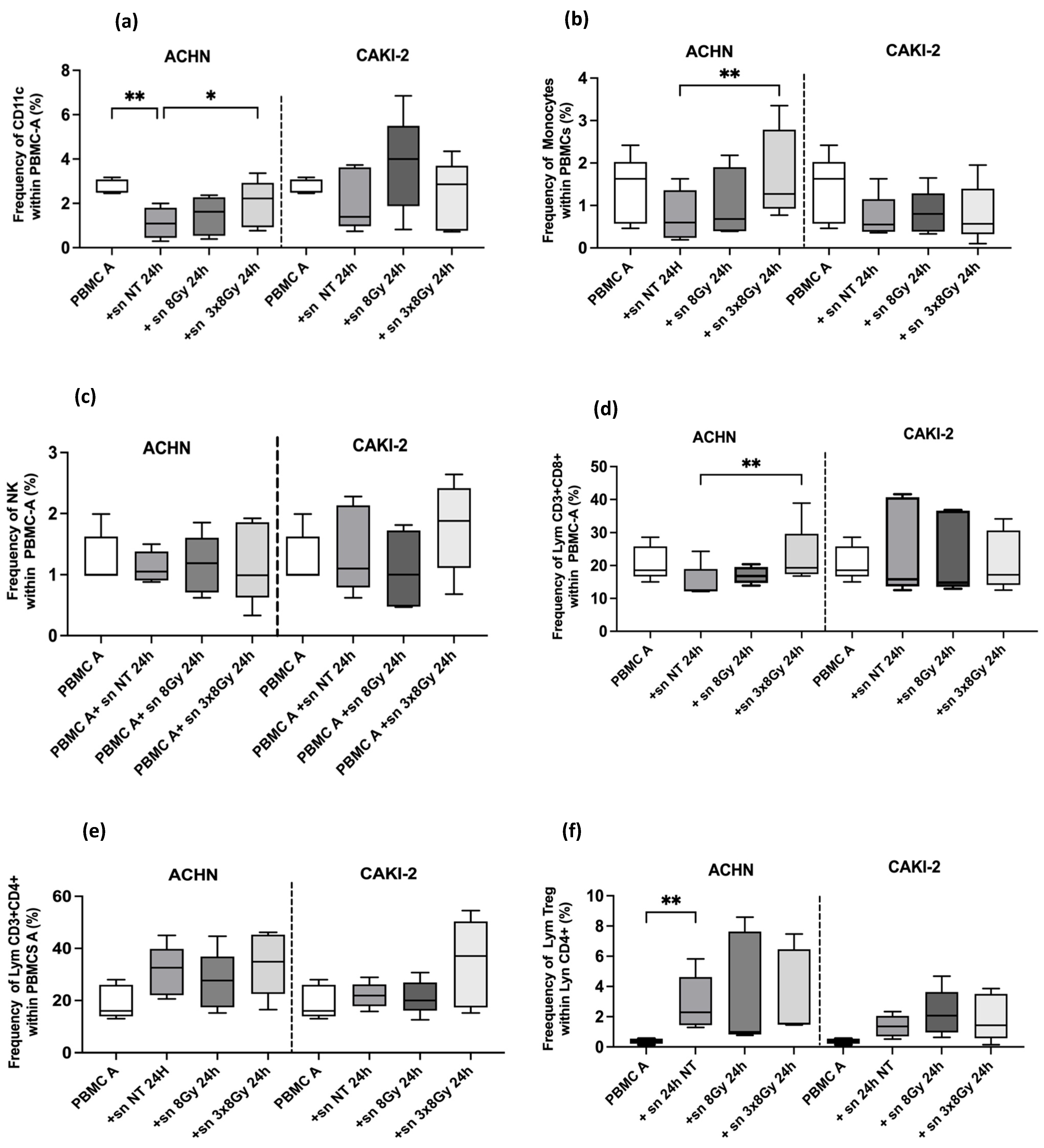
3.2.3. High Dose Irradiation Induced Immunomodulation in Circulating Immune Cells in RCC Models
3.2.4. Influence of Irradiated Supernatants from RCC Cells on Human PBMCs Genes Expression
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dason, S.; Lacuna, K.; Hannan, R.; Singer, E.A.; Runcie, K. State of the Art: Multidisciplinary Management of Oligometastatic Renal Cell Carcinoma. Am. Soc. Clin. Oncol. Educ. Book 2023, 43, e390038. [Google Scholar] [CrossRef]
- Young, M.; Jackson-Spence, F.; Beltran, L.; Day, E.; Suarez, C.; Bex, A.; Powles, T.; Szabados, B. Renal cell carcinoma. Lancet 2024, 404, 476–491. [Google Scholar] [CrossRef] [PubMed]
- Deschavanne, P.J.; Fertil, B. A review of human cell radiosensitivity in vitro. Int. J. Radiat. Oncol. 1996, 34, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Wessels, B.W.; Vessella, R.L.; Palme, D.F.; Berkopec, J.M.; Smith, G.K.; Bradley, E.W. Radiobiological comparison of external beam irradiation and radioimmunotherapy in renal cell carcinoma xenografts. Int. J. Radiat. Oncol. 1989, 17, 1257–1263. [Google Scholar] [CrossRef]
- Barbour, A.B.; Kirste, S.; Grosu, A.-L.; Siva, S.; Louie, A.V.; Onishi, H.; Swaminath, A.; Teh, B.S.; Psutka, S.P.; Weg, E.S.; et al. The Judicious Use of Stereotactic Ablative Radiotherapy in the Primary Management of Localized Renal Cell Carcinoma. Cancers 2023, 15, 3672. [Google Scholar] [CrossRef]
- Christensen, M.; Hannan, R. The Emerging Role of Radiation Therapy in Renal Cell Carcinoma. Cancers 2022, 14, 4693. [Google Scholar] [CrossRef]
- Correa, R.J.M.; Louie, A.V.; Zaorsky, N.G.; Lehrer, E.J.; Ellis, R.; Ponsky, L.; Kaplan, I.; Mahadevan, A.; Chu, W.; Swaminath, A.; et al. The Emerging Role of Stereotactic Ablative Radiotherapy for Primary Renal Cell Carcinoma: A Systematic Review and Meta-Analysis. Eur. Urol. Focus 2019, 5, 958–969. [Google Scholar] [CrossRef]
- Zaorsky, N.G.; Lehrer, E.J.; Kothari, G.; Louie, A.V.; Siva, S. Stereotactic ablative radiation therapy for oligometastatic renal cell carcinoma (SABR ORCA): A meta-analysis of 28 studies. Eur. Urol. Oncol. 2019, 2, 515–523. [Google Scholar] [CrossRef]
- Burnette, B.C.; Liang, H.; Lee, Y.; Chlewicki, L.; Khodarev, N.N.; Weichselbaum, R.R.; Fu, Y.-X.; Auh, S.L. The Efficacy of Radiotherapy Relies upon Induction of Type I Interferon–Dependent Innate and Adaptive Immunity. Cancer Res. 2011, 71, 2488–2496. [Google Scholar] [CrossRef]
- Villafuerte, C.J.Q.; Swaminath, A. Stereotactic Body Radiotherapy for Renal Cell Carcinoma—A Review of Use in the Primary, Cytoreductive and Oligometastatic Settings. Cancers 2024, 16, 3334. [Google Scholar] [CrossRef]
- Wild, A.T.; Herman, J.M.; Dholakia, A.S.; Moningi, S.; Lu, Y.; Rosati, L.M.; Hacker-Prietz, A.; Assadi, R.K.; Saeed, A.M.; Pawlik, T.M.; et al. Lymphocyte-Sparing Effect of Stereotactic Body Radiation Therapy in Patients with Unresectable Pancreatic Cancer. Int. J. Radiat. Oncol. 2016, 94, 571–579. [Google Scholar] [CrossRef]
- Siva, S.; Bressel, M.; Sidhom, M.; Sridharan, S.; Vanneste, B.G.L.; Davey, R.; Montgomery, R.; Ruben, J.; Foroudi, F.; Higgs, B.; et al. Stereotactic ablative body radiotherapy for primary kidney cancer (TROG 15.03 FASTRACK II): A non-randomised phase 2 trial. Lancet Oncol. 2024, 25, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Siva, S.; Ali, M.; Correa, R.J.M.; Muacevic, A.; Ponsky, L.; Ellis, R.J.; Lo, S.S.; Onishi, H.; Swaminath, A.; McLaughlin, M.; et al. 5-year outcomes after stereotactic ablative body radiotherapy for primary renal cell carcinoma: An individual patient data meta-analysis from IROCK (the International Radiosurgery Consortium of the Kidney). Lancet Oncol. 2022, 23, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- Vanpouille-Box, C.; Alard, A.; Aryankalayil, M.J.; Sarfraz, Y.; Diamond, J.M.; Schneider, R.J.; Inghirami, G.; Coleman, C.N.; Formenti, S.C.; Demaria, S. DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Nat. Commun. 2017, 8, 15618. [Google Scholar] [CrossRef]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but Not Single-Dose Radiotherapy Induces an Immune-Mediated Abscopal Effect when Combined with Anti–CTLA-4 Antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar] [CrossRef]
- Theelen, W.S.M.E.; Peulen, H.M.U.; Lalezari, F.; van der Noort, V.; de Vries, J.F.; Aerts, J.G.J.V.; Dumoulin, D.W.; Bahce, I.; Niemeijer, A.-L.N.; de Langen, A.J.; et al. Effect of Pembrolizumab After Stereotactic Body Radiotherapy vs Pembrolizumab Alone on Tumor Response in Patients with Advanced Non-Small Cell Lung Cancer: Results of the PEMBRO-RT Phase 2 Randomized Clinical Trial. JAMA Oncol. 2019, 5, 1276–1282. [Google Scholar] [CrossRef]
- Storozynsky, Q.; Hitt, M.M. The Impact of Radiation-Induced DNA Damage on cGAS-STING-Mediated Immune Responses to Cancer. Int. J. Mol. Sci. 2020, 21, 8877. [Google Scholar] [CrossRef]
- Moeller, B.J.; Cao, Y.; Vujaskovic, Z.; Li, C.Y.; Haroon, Z.A.; Dewhirst, M.W. The relationship between hypoxia and angiogenesis. Semin. Radiat. Oncol. 2004, 14, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Fuks, Z.; Kolesnick, R. Engaging the vascular component of the tumor response. Cancer Cell 2005, 8, 89–91. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Joiner, M.C.; van der Kogel, A. Basic Clinical Radiobiology, 4th ed.; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar] [CrossRef]
- Raj, R.K.; Upadhyay, R.; Wang, S.-J.; Singer, E.A.; Dason, S. Incorporating Stereotactic Ablative Radiotherapy into the Multidisciplinary Management of Renal Cell Carcinoma. Curr. Oncol. 2023, 30, 10283–10298. [Google Scholar] [CrossRef]
- Tubin, S.; Gupta, S.; Grusch, M.; Popper, H.H.; Brcic, L.; Ashdown, M.L.; Khleif, S.N.; Peter-Vörösmarty, B.; Hyden, M.; Negrini, S.; et al. Shifting the Immune-Suppressive to Predominant Immune-Stimulatory Radiation Effects by SBRT-PArtial Tumor Irradiation Targeting HYpoxic Segment (SBRT-PATHY). Cancers 2020, 13, 50. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.; Yang, Y.; Swift, M.; Zhang, Z.; Wu, S.; Sun, Y.; Yang, K. In situ vaccination caused by diverse irradiation-driven cell death programs. Theranostics 2024, 14, 1147–1167. [Google Scholar] [CrossRef]
- Marvaso, G.; Jereczek-Fossa, B.A.; Zaffaroni, M.; Vincini, M.G.; Corrao, G.; Andratschke, N.; Balagamwala, E.H.; Bedke, J.; Blanck, O.; Capitanio, U.; et al. Delphi consensus on stereotactic ablative radiotherapy for oligometastatic and oligoprogressive renal cell carcinoma—A European Society for Radiotherapy and Oncology study endorsed by the European Association of Urology. Lancet Oncol. 2024, 25, e193–e204. [Google Scholar] [CrossRef]
- Nabrinsky, E.; Macklis, J.; Bitran, J. A Review of the Abscopal Effect in the Era of Immunotherapy. Cureus 2022, 14, e29620. [Google Scholar] [CrossRef]
- Walsh, L.; Stanfield, J.L.; Cho, L.C.; Chang, C.; Forster, K.; Kabbani, W.; Cadeddu, J.A.; Hsieh, J.-T.; Choy, H.; Timmerman, R.; et al. Efficacy of Ablative High-Dose-per-Fraction Radiation for Implanted Human Renal Cell Cancer in a Nude Mouse Model. Eur. Urol. 2006, 50, 795–800. [Google Scholar] [CrossRef]
- Onal, C.; Guler, O.C.; Hurmuz, P.; Yavas, G.; Tilki, B.; Oymak, E.; Yavas, C.; Ozyigit, G. Bone-only oligometastatic renal cell carcinoma patients treated with stereotactic body radiotherapy: A multi-institutional study. Strahlenther. Onkol. 2022, 198, 940–948. [Google Scholar] [CrossRef]
- Franken, N.A.P.; Rodermond, H.M.; Stap, J.; Haveman, J.; Van Bree, C. Clonogenic assay of cells in vitro. Nat. Protoc. 2006, 1, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Cohen–Jonathan, E.; Bernhard, E.J.; McKenna, W.G. How does radiation kill cells? Curr. Opin. Chem. Biol. 1999, 3, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Fiers, W.; Beyaert, R.; Declercq, W.; Vandenabeele, P. More than one way to die: Apoptosis, necrosis and reactive oxygen damage. Oncogene 1999, 18, 7719–7730. [Google Scholar] [CrossRef]
- Krysko, O.; Løve Aaes, T.; Bachert, C.; Vandenabeele, P.; Krysko, D.V. Many faces of DAMPs in cancer therapy. Cell Death Dis. 2013, 4, e631. [Google Scholar] [CrossRef]
- Rosa, R.; Damiano, V.; Nappi, L.; Formisano, L.; Massari, F.; Scarpa, A.; Martignoni, G.; Bianco, R.; Tortora, G. Angiogenic and signalling proteins correlate with sensitivity to sequential treatment in renal cell cancer. Br. J. Cancer 2013, 109, 686–693. [Google Scholar] [CrossRef]
- Wang, N.-H.; Lei, Z.; Yang, H.-N.; Tang, Z.; Yang, M.-Q.; Wang, Y.; Sui, J.-D.; Wu, Y.-Z. Radiation-induced PD-L1 expression in tumor and its microenvironment facilitates cancer-immune escape: A narrative review. Ann. Transl. Med. 2022, 10, 1406. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Varnier, L.; Pezzicoli, G.; Pirovano, M.; Cosmai, L.; Porta, C. IL-8 and its role as a potential biomarker of resistance to anti-angiogenic agents and immune checkpoint inhibitors in metastatic renal cell carcinoma. Front. Oncol. 2022, 12, 990568. [Google Scholar] [CrossRef] [PubMed]
- Schwanhäusser, B.; Busse, D.; Li, N.; Dittmar, G.; Schuchhardt, J.; Wolf, J.; Chen, W.; Selbach, M. Global quantification of mammalian gene expression control. Nature 2011, 473, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Annes, J.P. Making sense of latent TGFβ activation. J. Cell Sci. 2003, 116, 217–224. [Google Scholar] [CrossRef]
- Han, Z.; Dong, Y.; Lu, J.; Yang, F.; Zheng, Y.; Yang, H. Role of hypoxia in inhibiting dendritic cells by VEGF signaling in tumor microenvironments: Mechanism and application. Am. J. Cancer Res. 2021, 11, 3777–3793. [Google Scholar]
- Liston, A.; Piccirillo, C.A. Developmental Plasticity of Murine and Human Foxp3+ Regulatory T Cells. In Advances in Immunology; Elsevier: Amsterdam, The Netherlands, 2013; Volume 119, pp. 85–106. ISBN 978-0-12-407707-2. [Google Scholar]
- Yoshida, N.; Ikemoto, S.; Narita, K.; Sugimura, K.; Wada, S.; Yasumoto, R.; Kishimoto, T.; Nakatani, T. Interleukin-6, tumour necrosis factor a and interleukin-1b in patients with renal cell carcinoma. Br. J. Cancer 2002, 86, 1396–1400. [Google Scholar] [CrossRef]
- Busse, A.; Asemissen, A.; Nonnenmacher, A.; Ochsenreither, S.; Fusi, A.; Braun, F.; Stather, D.; Schmittel, A.; Miller, K.; Thiel, E.; et al. Systemic Immune Tuning in Renal Cell Carcinoma: Favorable Prognostic Impact of TGF-b1 mRNA-Expression in Peripheral Blood Mononuclear Cells. J. Immunother. 2011, 34, 7. [Google Scholar] [CrossRef]
- Formenti, S.C.; Demaria, S. Systemic effects of local radiotherapy. Lancet Oncol. 2009, 10, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Auh, S.L.; Wang, Y.; Burnette, B.; Wang, Y.; Meng, Y.; Beckett, M.; Sharma, R.; Chin, R.; Tu, T.; et al. Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: Changing strategies for cancer treatment. Blood 2009, 114, 589–595. [Google Scholar] [CrossRef]
- Obeid, M.; Tesniere, A.; Ghiringhelli, F.; Fimia, G.M.; Apetoh, L.; Perfettini, J.-L.; Castedo, M.; Mignot, G.; Panaretakis, T.; Casares, N.; et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat. Med. 2007, 13, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Yang, M.; Zhang, J.; Yin, Y.; Fan, X.; Zhang, Y.; Qin, S.; Zhang, H.; Yu, F. Immunogenic Cell Death Induction by Ionizing Radiation. Front. Immunol. 2021, 12, 705361. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meniai Merzouki, F.; Grolez, G.P.; Bouchez, C.; Leroux, B.; Benoit, J.; Morales, O.; Delhem, N. Immunomodulatory Effects of High-Dose Irradiation Regimens in Renal Cell Carcinoma: Insights from an In Vitro Model with Human Peripheral Blood Mononuclear Cell. Biomedicines 2025, 13, 2107. https://doi.org/10.3390/biomedicines13092107
Meniai Merzouki F, Grolez GP, Bouchez C, Leroux B, Benoit J, Morales O, Delhem N. Immunomodulatory Effects of High-Dose Irradiation Regimens in Renal Cell Carcinoma: Insights from an In Vitro Model with Human Peripheral Blood Mononuclear Cell. Biomedicines. 2025; 13(9):2107. https://doi.org/10.3390/biomedicines13092107
Chicago/Turabian StyleMeniai Merzouki, Fatima, Guillaume Paul Grolez, Clément Bouchez, Bertrand Leroux, Jérôme Benoit, Olivier Morales, and Nadira Delhem. 2025. "Immunomodulatory Effects of High-Dose Irradiation Regimens in Renal Cell Carcinoma: Insights from an In Vitro Model with Human Peripheral Blood Mononuclear Cell" Biomedicines 13, no. 9: 2107. https://doi.org/10.3390/biomedicines13092107
APA StyleMeniai Merzouki, F., Grolez, G. P., Bouchez, C., Leroux, B., Benoit, J., Morales, O., & Delhem, N. (2025). Immunomodulatory Effects of High-Dose Irradiation Regimens in Renal Cell Carcinoma: Insights from an In Vitro Model with Human Peripheral Blood Mononuclear Cell. Biomedicines, 13(9), 2107. https://doi.org/10.3390/biomedicines13092107






