Nanoparticle-Delivered Rutin Prevents Metabolic and Oxidative Imbalance in Obesity Triggered by a High-Fat Diet: In Vivo and In Silico Studies
Abstract
1. Introduction
2. Materials and Experimental Methods
2.1. Chemicals and Reagents
2.2. Preparation of Rutin-Loaded Chitosan Nanoparticles (RU-N)
2.3. RU-N Characterization
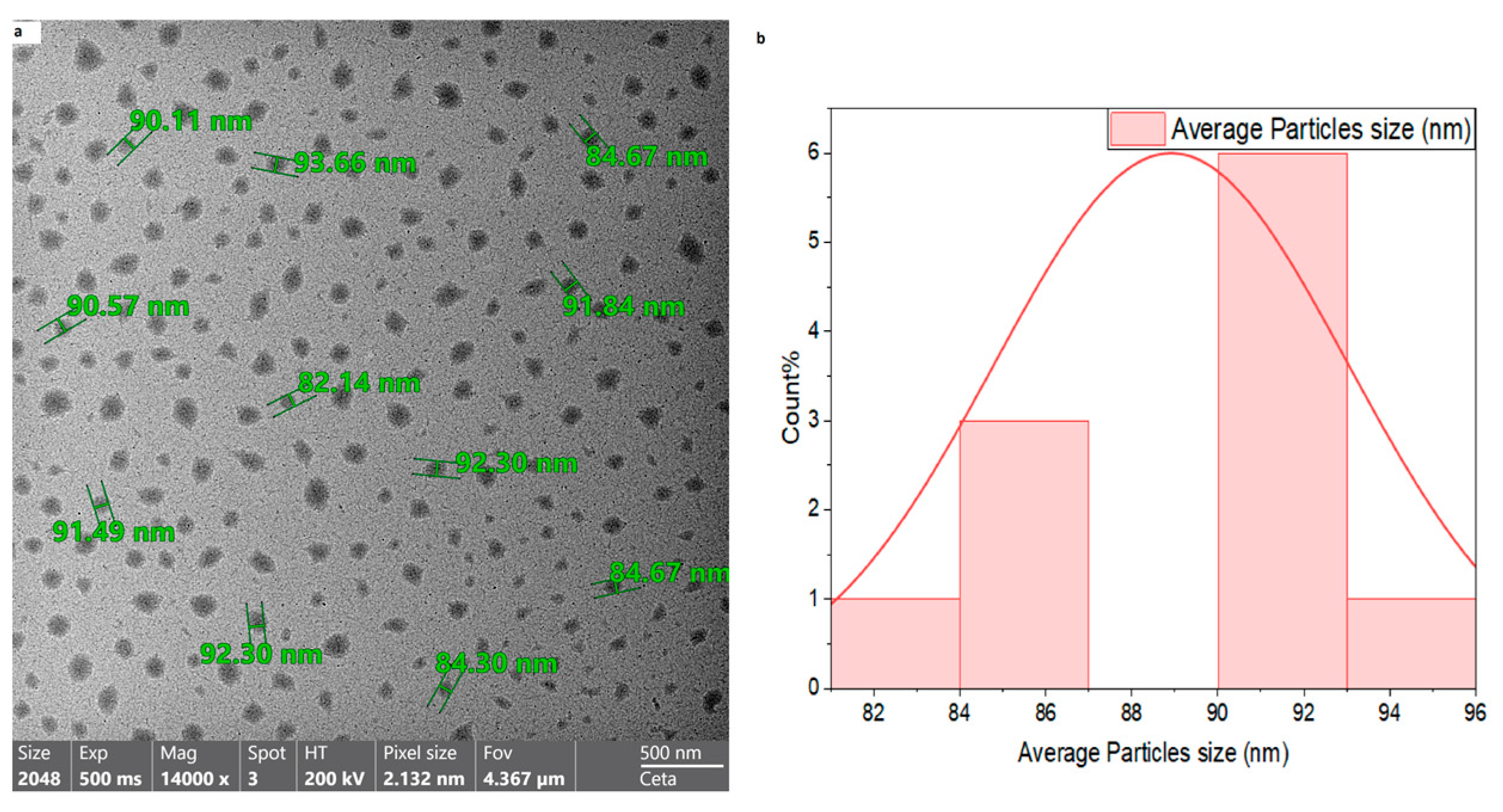
2.3.1. Transmission Electron Microscope
2.3.2. Assessment of the Entrapment Efficiency (EE) and Loading Capacity (LC)
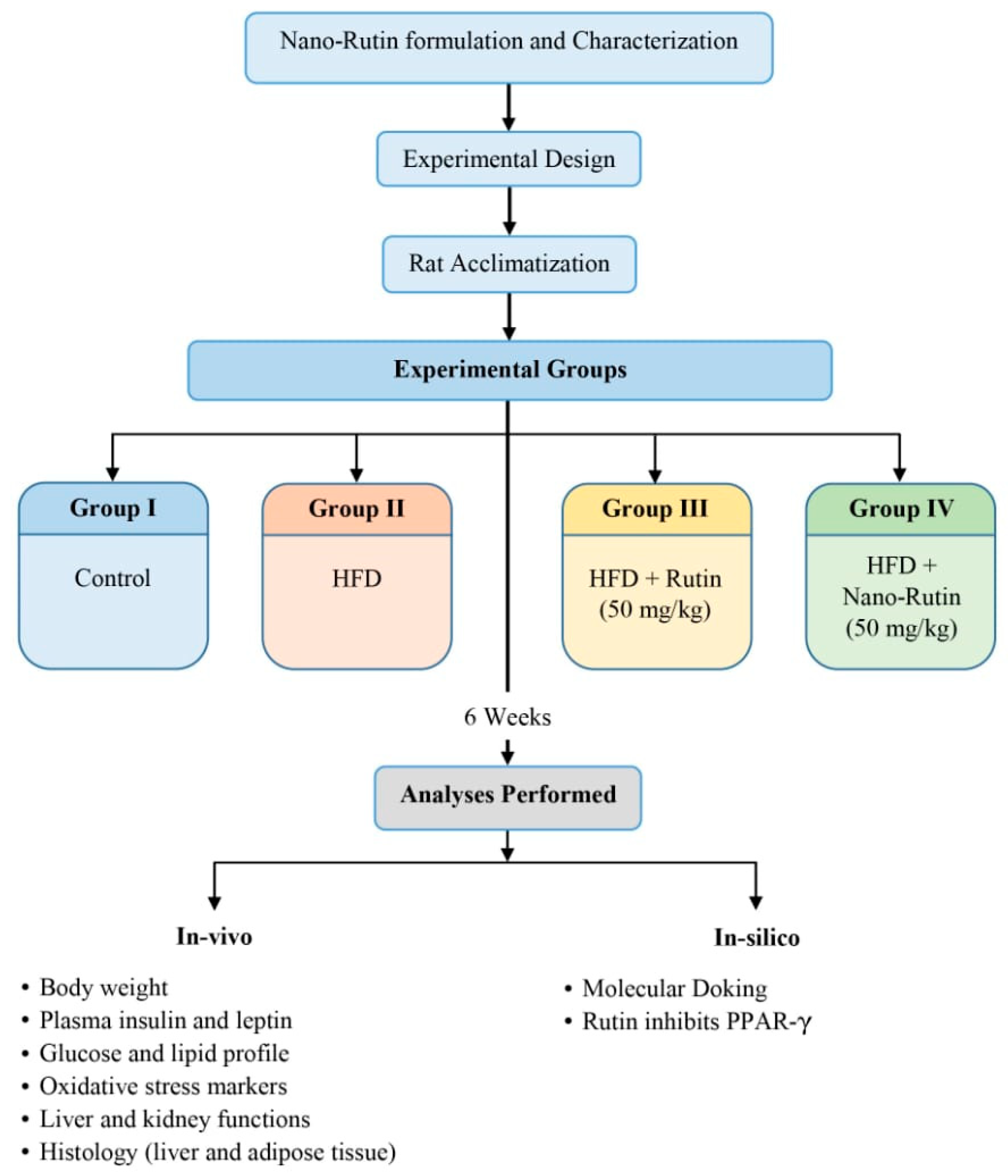
2.4. In Vivo Model
2.5. Hyperlipidemia Induction in Rats
2.6. Research Design
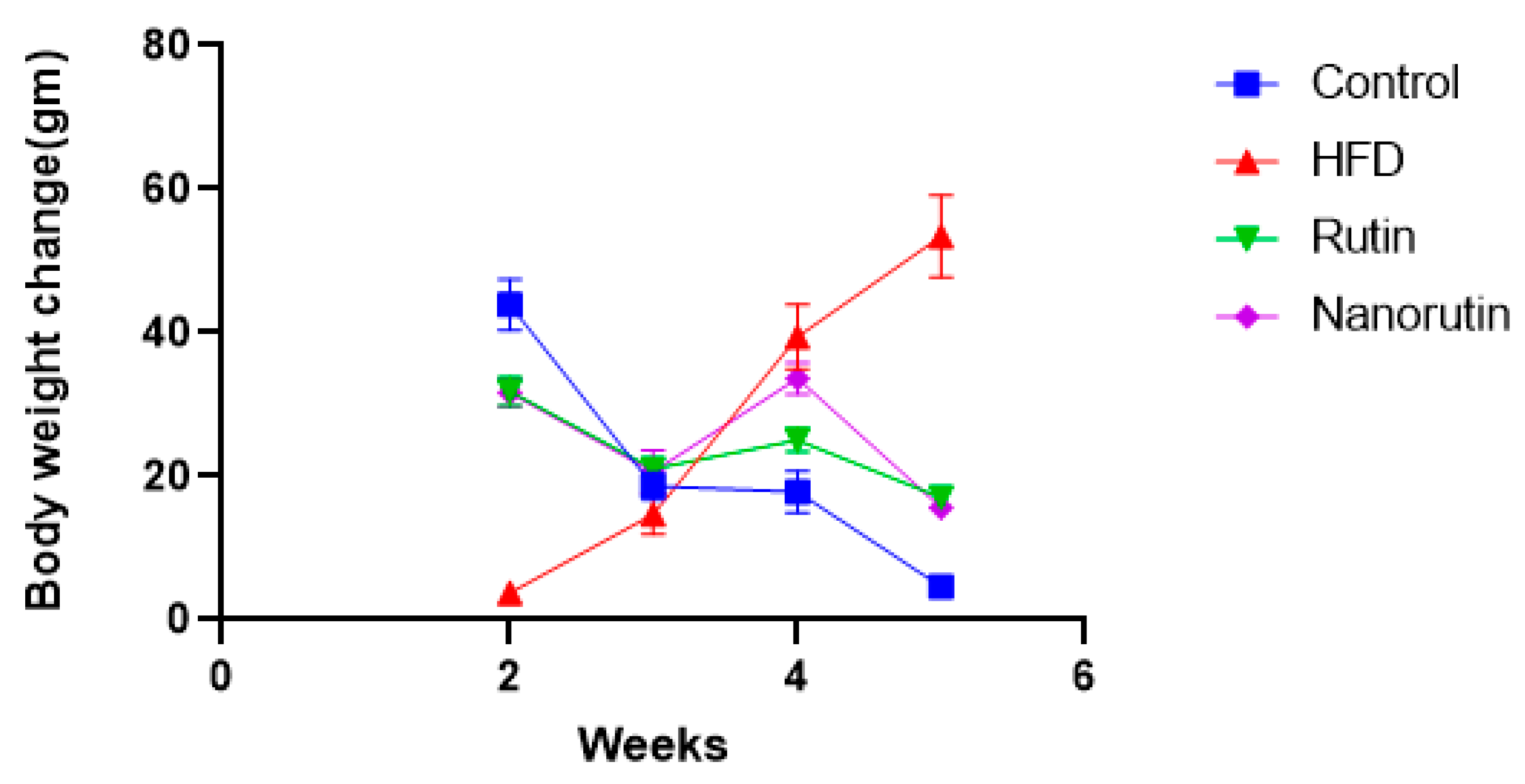
2.7. Body Weight Measurement
2.8. Animal Handling and Specimen Collection
2.9. Biochemical Analysis
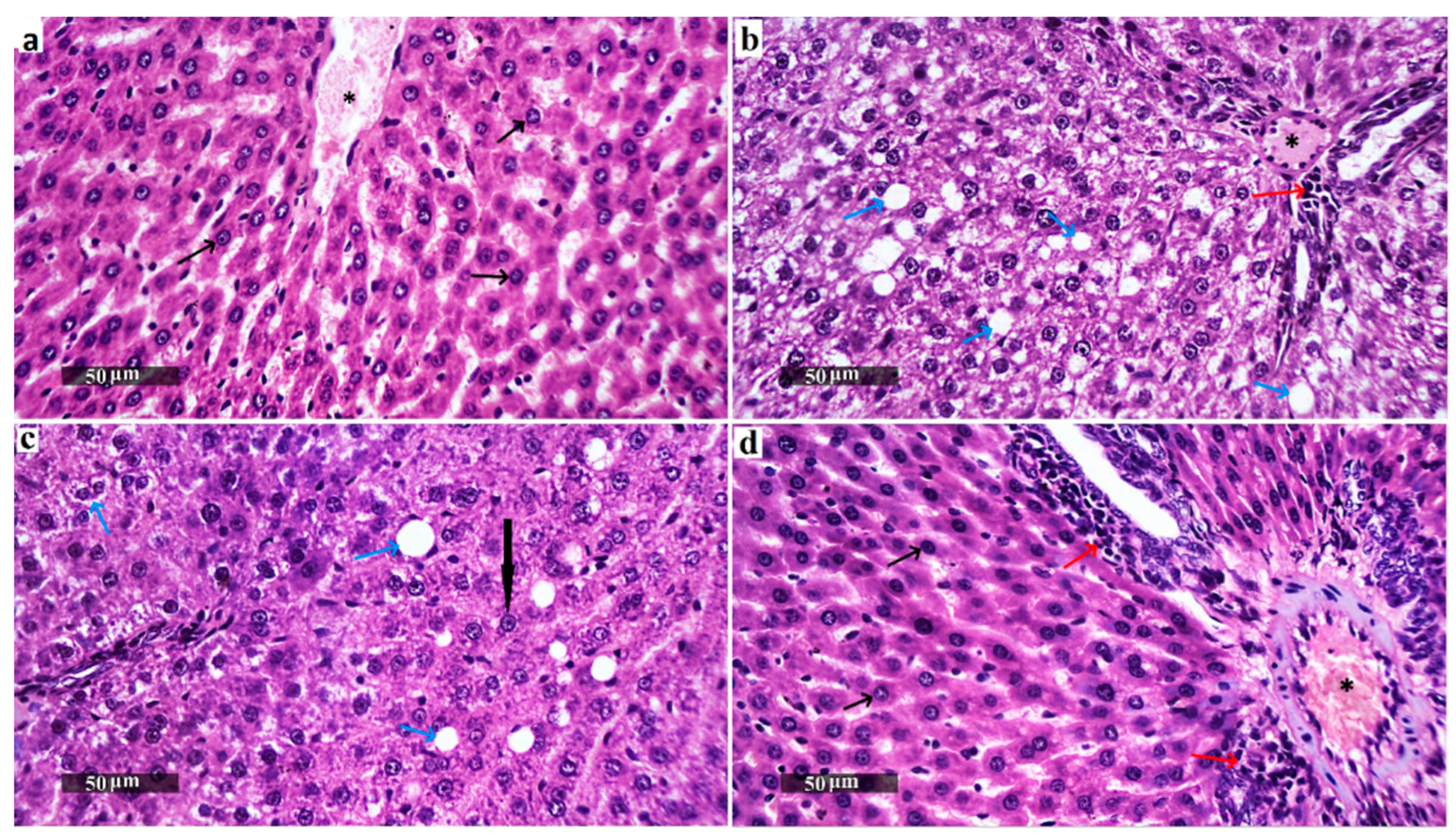
2.10. Histopathological Examination
2.11. Statistical Analysis
2.12. Molecular Docking
2.12.1. Protein and Ligand Preparation
2.12.2. Molecular Docking Performance
3. Results
3.1. Characterization of Nano-Rutin
3.2. Body Weight Gain
3.3. Serum Biomarkers
3.4. Oxidative Stress Parameters
3.5. Interaction Between Study Variables
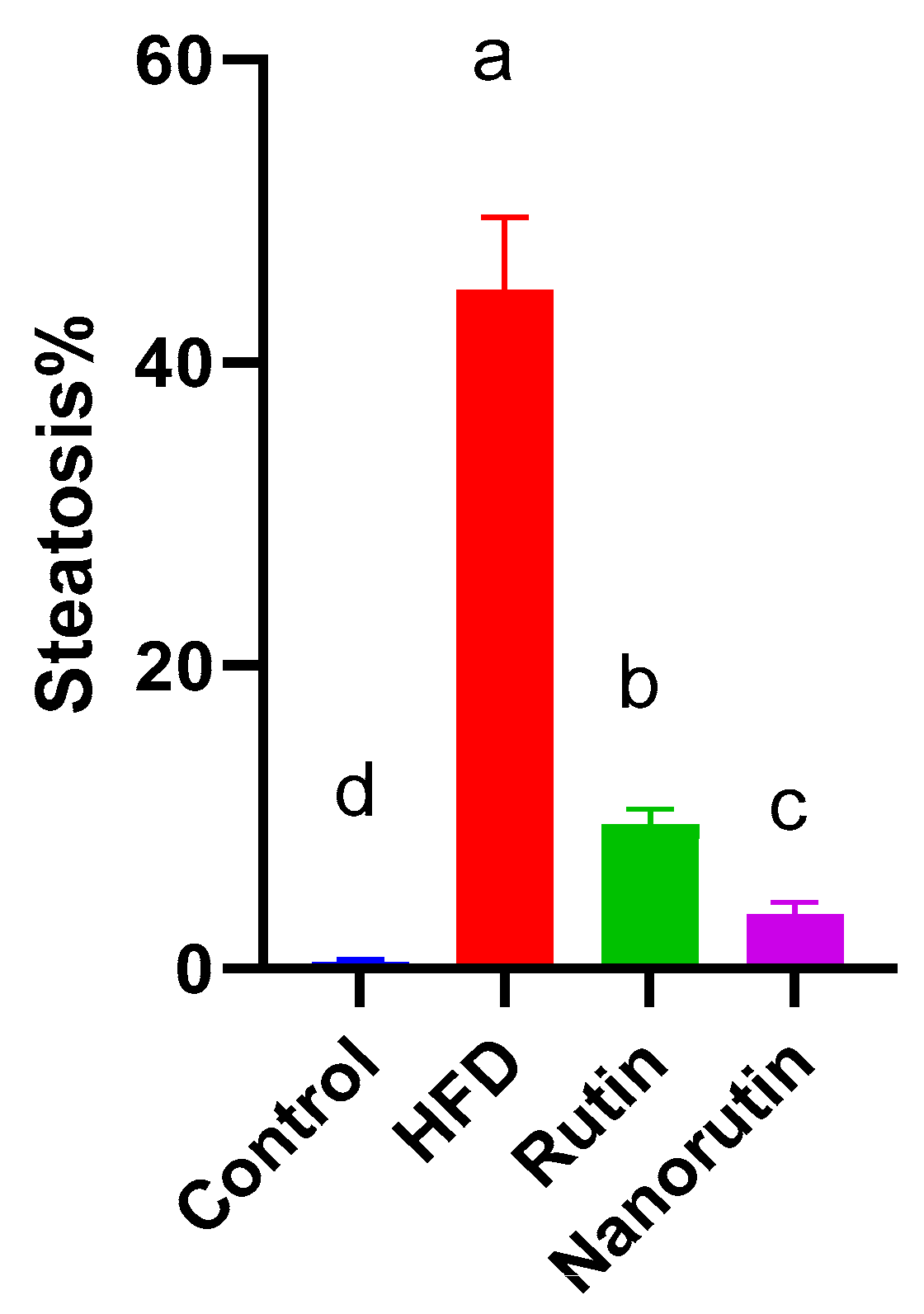
3.6. Liver Histopathological Examination
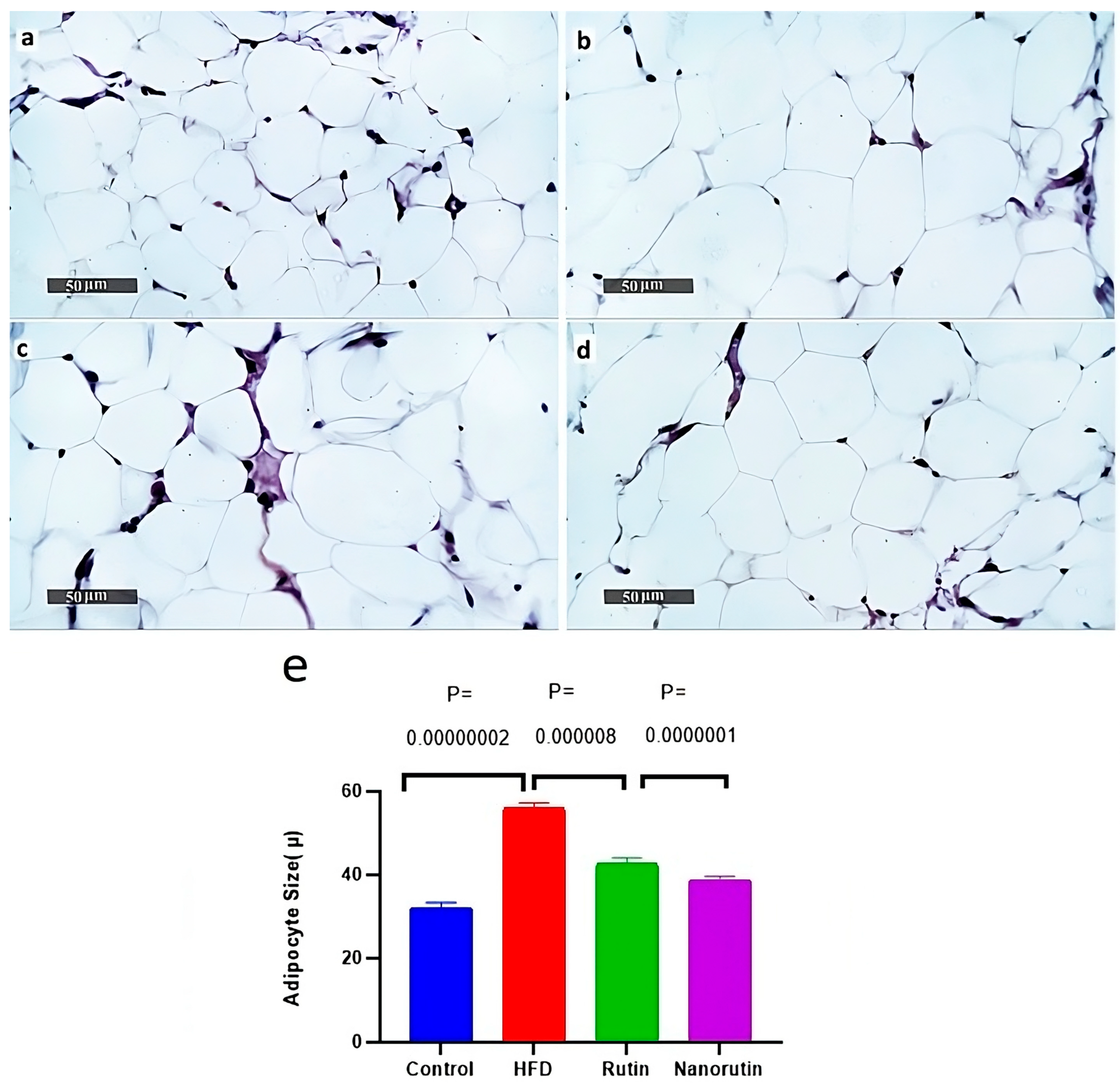
3.7. Adipose Tissue Histopathological Examination
3.8. Molecular Docking
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Manna, P.; Jain, S.K. Obesity, Oxidative Stress, Adipose Tissue Dysfunction, and the Associated Health Risks: Causes and Therapeutic Strategies. Metab. Syndr. Relat. Disord. 2015, 13, 423–444. [Google Scholar] [CrossRef]
- Sikaris, K.A. The Clinical Biochemistry of Obesity. Clin. Biochem. Rev. 2004, 25, 165. [Google Scholar]
- Baker, J.S.; Supriya, R.; Dutheil, F.; Gao, Y. Obesity: Treatments, Conceptualizations, and Future Directions for a Growing Problem. Biology 2022, 11, 160. [Google Scholar] [CrossRef]
- Hsu, C.L.; Yen, G. Phenolic compounds: Evidence for inhibitory effects against obesity and their underlying molecular signaling mechanisms. Mol. Nutr. Food Res. 2008, 52, 53–61. [Google Scholar] [CrossRef]
- Tang, Y.; Huang, Y.; Zhang, B.; Luo, T.; Zhong, W. Editorial: Food rich in phenolic compounds and their potential to fight obesity. Front. Nutr. 2023, 10, 1204981. [Google Scholar] [CrossRef]
- Hsu, C.-L.; Wu, C.-H.; Huang, S.-L.; Yen, G.-C. Phenolic Compounds Rutin and o-Coumaric Acid Ameliorate Obesity Induced by High-Fat Diet in Rats. J. Agric. Food Chem. 2009, 57, 425–431. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A.K. The Pharmacological Potential of Rutin. Saudi Pharm. J. SPJ 2016, 25, 149. [Google Scholar] [CrossRef]
- Patel, K.; Patel, D.K. The Beneficial Role of Rutin, A Naturally Occurring Flavonoid in Health Promotion and Disease Prevention: A Systematic Review and Update. In Bioactive Food as Dietary Interventions for Arthritis and Related Inflammatory Diseases; Watson, R.R., Preedy, V.R., Eds.; Academic Press: Oxford, UK, 2019; pp. 457–479. [Google Scholar] [CrossRef]
- Aatif, M. Current Understanding of Polyphenols to Enhance Bioavailability for Better Therapies. Biomedicines 2023, 11, 2078. [Google Scholar] [CrossRef]
- Rahman, M.; Rahaman, S.; Islam, R.; Rahman, F.; Mithi, F.M.; Alqahtani, T.; Almikhlafi, M.A.; Alghamdi, S.Q.; Alruwaili, A.S.; Hossain, S.; et al. Role of Phenolic Compounds in Human Disease: Current Knowledge and Future Prospects. Molecules 2021, 27, 233. [Google Scholar] [CrossRef]
- Bernkop-Schnürch, A.; Dünnhaupt, S. Chitosan-Based Drug Delivery Systems. Eur. J. Pharm. Bio-Pharm. 2012, 81, 463–469. [Google Scholar] [CrossRef]
- Aminabhavi, T.M.; Dharupaneedi, S.P.; More, U.A. The Role of Nanotechnology and Chitosan-Based Biomaterials for Tissue Engineering and Therapeutic Delivery. In Chitosan Based Biomaterials; Jennings, J.A., Bumgardner, J.D., Eds.; Woodhead Publishing: London, UK, 2017; Volume 2, pp. 1–29. [Google Scholar]
- Aktaş, Y.; Yemisci, M.; Andrieux, K.; Gürsoy, R.N.; Alonso, M.J.; Fernandez-Megia, E.; Novoa-Carballal, R.; Quiñoá, E.; Ri-guera, R.; Sargon, M.F.; et al. Development and Brain Delivery of Chitosan−PEG Nanoparticles Functionalized with the Monoclonal Antibody OX26. Bioconjugate Chem. 2005, 16, 1503–1511. [Google Scholar] [CrossRef]
- Salem, M.A.; Aborehab, N.M.; Abdelhafez, M.M.; Ismail, S.H.; Maurice, N.W.; Azzam, M.A.; Alseekh, S.; Fernie, A.R.; Salama, M.M.; Ezzat, S.M. Anti-Obesity Effect of a Tea Mixture Nano-Formulation on Rats Occurs via the Upregulation of AMP-Activated Protein Kinase/Sirtuin-1/Glucose Transporter Type 4 and Peroxisome Proliferator-Activated Receptor Gamma Pathways. Metabolites 2023, 13, 871. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, S.P.; Bangar, J.V.; Akuskar, G.K.; Ratnaparkhi, M.P. Development and Validation of UV Spectrophotometric Method for Simultaneous Estimation of Rutin and Quercetin in Niosome Formulation. Pharm. Lett. 2014, 6, 271–276. [Google Scholar]
- Metawea, M.R.; Abdelrazek, H.M.A.; El-Hak, H.N.G.; Moghazee, M.M.; Marie, O.M. Comparative effects of curcumin versus nano-curcumin on histological, immunohistochemical expression, histomorphometric, and biochemical changes to pancreatic beta cells and lipid profile of streptozocin induced diabetes in male Sprague–Dawley rats. Environ. Sci. Pollut. Res. 2023, 30, 62067–62079. [Google Scholar] [CrossRef]
- Wang, Z.; Ge, S.; Li, S.; Lin, H.; Lin, S. Anti-obesity effect of trans-cinnamic acid on HepG2 cells and HFD-fed mice. Food Chem. Toxicol. 2020, 137, 111148. [Google Scholar] [CrossRef]
- Lu, K.; Xie, S.; Han, S.; Zhang, J.; Chang, X.; Chao, J.; Huang, Q.; Yuan, Q.; Lin, H.; Xu, L.; et al. Preparation of a nano emodin transfersome and study on its anti-obesity mechanism in adipose tissue of diet-induced obese rats. J. Transl. Med. 2014, 12, 72. [Google Scholar] [CrossRef] [PubMed]
- Radwan, R.R.; Ali, H.E. Radiation-synthesis of chitosan/poly (acrylic acid) nanogel for improving the antitumor potential of rutin in hepatocellular carcinoma. Drug Deliv. Transl. Res. 2020, 11, 261–278. [Google Scholar] [CrossRef] [PubMed]
- Nishikimi, M.; Appaji, N.; Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Culling, C.F.A. Handbook of Histopathological and Histochemical Techniques: Including Museum Techniques; Elsevier: Amsterdam, The Netherlands, 2014; p. 727. [Google Scholar]
- Yustisia, I.; Tandiari, D.; Cangara, M.H.; Hamid, F.; Daud, N.A. A high-fat, high-fructose diet induced hepatic steatosis, renal lesions, dyslipidemia, and hyperuricemia in non-obese rats. Heliyon 2022, 8, e10896. [Google Scholar] [CrossRef]
- Al-Rawashde, F.A.; Al-Omari, L.; Saad, H.K.M.; Alqaraleh, M.; Zahran, N.H.; Al-Jamal, H.A.N. Establishing the Role of Thymoquinone as Potential Inhibitor of Mutant Oncoproteins in FLT3/RAS/RAF/MEK/ERK Pathway against Acute Myeloid Leukemia. J. Med. Chem. 2024, 7, 1511–1525. [Google Scholar] [CrossRef]
- Bolivar Avila, S. Validation of Docking Methodology (Redocking). Available online: https://www.researchgate.net/publication/384293232_Validation_of_Docking_Methodology_Redocking#fullTextFileContent (accessed on 12 July 2025).
- Yang, J.; Lee, J.; Kim, Y. Effect of Deglycosylated Rutin by Acid Hydrolysis on Obesity and Hyperlipidemia in High-Fat Diet-Induced Obese Mice. Nutrients 2020, 12, 1539. [Google Scholar] [CrossRef]
- Lin, X.; Li, H. Obesity: Epidemiology, Pathophysiology, and Therapeutics. Front. Endocrinol. 2021, 12, 706978. [Google Scholar] [CrossRef]
- Angelidi, A.M.; Belanger, M.J.; Kokkinos, A.; Koliaki, C.C.; Mantzoros, C.S. Novel Noninvasive Approaches to the Treatment of Obesity: From Pharmacotherapy to Gene Therapy. Endocr. Rev. 2021, 43, 507–557. [Google Scholar] [CrossRef]
- Sayed, U.F.S.M.; Moshawih, S.; Goh, H.P.; Kifli, N.; Gupta, G.; Singh, S.K.; Chellappan, D.K.; Dua, K.; Hermansyah, A.; Ser, H.L.; et al. Natural products as novel anti-obesity agents: Insights into mechanisms of action and potential for therapeutic management. Front. Pharmacol. 2023, 14, 1182937. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.Z.; Zhu, Y.; Yu, J.N.; Xu, X.M. Advanced in solubilization methods of water-insoluble natural drugs. Zhongguo Zhong Yao Za Zhi 2014, 39, 3226–3231. [Google Scholar] [CrossRef] [PubMed]
- Ganjayi, M.S.; Karunakaran, R.S.; Gandham, S.; Meriga, B. Quercetin-3-O-rutinoside from Moringa oleifera Downregulates Adipogenesis and Lipid Accumulation and Improves Glucose Uptake by Activation of AMPK/Glut-4 in 3T3-L1 Cells. Rev. Bras. de Farm. 2023, 33, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Oh, S.; Shin, Y.; Jung, S.; Kim, Y. Reduction of body weight by rutin is associated with an increase of brown adipose tissue mitochondrial biogenesis in high-fat diet induced obese rat (LB430). FASEB J. 2014, 28, LB430. [Google Scholar] [CrossRef]
- Jalili, V.; Poorahmadi, Z.; Ardekanizadeh, N.H.; Gholamalizadeh, M.; Ajami, M.; Houshiarrad, A.; Hajipour, A.; Shafie, F.; Alizadeh, A.; Mokhtari, Z.; et al. The association between obesity with serum levels of liver enzymes, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase and gamma-glutamyl transferase in adult women. Endocrinol. Diabetes Metab. 2022, 5, e367. [Google Scholar] [CrossRef]
- Demori, I.; Grasselli, E. The Role of the Stress Response in Metabolic Dysfunction-Associated Fatty Liver Disease: A Psychoneuroendocrineimmunology-Based Perspective. Nutrients 2023, 15, 795. [Google Scholar] [CrossRef]
- Kathak, R.R.; Sumon, A.H.; Molla, N.H.; Hasan, M.; Miah, R.; Tuba, H.R.; Habib, A.; Ali, N. The association between elevated lipid profile and liver enzymes: A study on Bangladeshi adults. Sci. Rep. 2022, 12, 1711. [Google Scholar] [CrossRef]
- Guo, Y.; Jiang, S.; Li, H.; Xie, G.; Pavel, V.; Zhang, Q.; Li, Y.; Huang, C. Obesity induces osteoimmunology imbalance: Molecular mechanisms and clinical implications. Biomed. Pharmacother. 2024, 177, 117139. [Google Scholar] [CrossRef] [PubMed]
- High Bone Resorption|International Osteoporosis Foundation. Available online: https://www.osteoporosis.foundation/health-professionals/skeletal-rare-disorders/altered-osteoclast-osteoblast-or-osteocyte-activity--high-bone-resorption (accessed on 25 March 2025).
- Cetin, E.G. The Relationship between Insulin Resistance and Liver Damage in non-alcoholic Fatty Liver Patients. Med. Bull. Sisli Etfal Hosp. 2020, 54, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Sanyal, D.; Mukherjee, P.; Raychaudhuri, M.; Ghosh, S.; Mukherjee, S.; Chowdhury, S. Profile of liver enzymes in non-alcoholic fatty liver disease in patients with impaired glucose tolerance and newly detected untreated type 2 diabetes. Indian J. Endocrinol. Metab. 2015, 19, 597–601. [Google Scholar] [CrossRef]
- Del Campo, J.A.; Gallego, P.; Grande, L. Role of inflammatory response in liver diseases: Therapeutic strategies. World J. Hepatol. 2018, 10, 1–7. [Google Scholar] [CrossRef]
- Čolak, E.; Pap, D. The role of oxidative stress in the development of obesity and obesity-related metabolic disorders. J. Med Biochem. 2021, 40, 1–9. [Google Scholar] [CrossRef]
- Naderi, M.; Seyedabadi, M.; Amiri, F.T.; Akbari, S.; Shaki, F. Rutin mitigates perfluorooctanoic acid-induced liver injury via modulation of oxidative stress, apoptosis, and inflammation. Iran. J. Basic Med. Sci. 2023, 26, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, S.; Naraki, K.; Roohbakhsh, A.; Hayes, A.W.; Karimi, G. The protective effects of rutin on the liver, kidneys, and heart by counteracting organ toxicity caused by synthetic and natural compounds. Food Sci. Nutr. 2022, 11, 39–56. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, Y.; Lu, R.; Zhao, Q.; Gao, Y. The multiple mechanisms and therapeutic significance of rutin in metabolic dysfunction-associated fatty liver disease (MAFLD). Fitoterapia 2024, 178, 106178. [Google Scholar] [CrossRef]
- Shen, Y.; Zheng, X.; Hu, Y.; Deng, Y.; Xiong, Q. Editorial: Bile acids in obesity-related diseases. Front. Endocrinol. 2024, 15, 1518604. [Google Scholar] [CrossRef]
- A Shipovskaya, A.; Dudanova, O.P. Intrahepatic cholestasis in nonalcoholic fatty liver disease. Ter. Arkhiv 2018, 90, 69–74. [Google Scholar] [CrossRef]
- Shah, R.; Grant, L.M.; John, S. Cholestatic Jaundice. StatPearls. January 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482279/ (accessed on 25 March 2025).
- Ahmed, O.M.; Elkomy, M.H.; Fahim, H.I.; Ashour, M.B.; Naguib, I.A.; Alghamdi, B.S.; Mahmoud, H.U.R.; Ahmed, N.A. Rutin and Quercetin Counter Doxorubicin-Induced Liver Toxicity in Wistar Rats via Their Modulatory Effects on Inflammation, Oxidative Stress, Apoptosis, and Nrf2. Oxidative Med. Cell. Longev. 2022, 2022, 2710607. [Google Scholar] [CrossRef]
- Yau, K.; Kuah, R.; Cherney, D.Z.I.; Lam, T.K.T. Obesity and the kidney: Mechanistic links and therapeutic advances. Nat. Rev. Endocrinol. 2024, 20, 321–335. [Google Scholar] [CrossRef]
- Kovesdy, C.P.; Furth, S.L.; Zoccali, C. Obesity and Kidney Disease: Hidden Consequences of the Epidemic. Can. J. Kidney Health Dis. 2017, 4, 1–10. [Google Scholar] [CrossRef]
- Basolo, A.; Salvetti, G.; Giannese, D.; Genzano, S.B.; Ceccarini, G.; Giannini, R.; Sotgia, G.; Fierabracci, P.; Piaggi, P.; Santini, F. Obesity, Hyperfiltration, and Early Kidney Damage: A New Formula for the Estimation of Creatinine Clearance. J. Clin. Endocrinol. Metab. 2023, 108, 3280–3286. [Google Scholar] [CrossRef]
- García-Carro, C.; Vergara, A.; Bermejo, S.; Azancot, M.A.; Sellarés, J.; Soler, M.J. A Nephrologist Perspective on Obesity: From Kidney Injury to Clinical Management. Front. Med. 2021, 8, 655871. [Google Scholar] [CrossRef]
- Ávila, M.; Sánchez, M.G.M.; Amador, A.S.B.; Paniagua, R. The Metabolism of Creatinine and Its Usefulness to Evaluate Kidney Function and Body Composition in Clinical Practice. Biomolecules 2025, 15, 41. [Google Scholar] [CrossRef]
- Colombo, G.; Altomare, A.; Astori, E.; Landoni, L.; Garavaglia, M.L.; Rossi, R.; Giustarini, D.; Lionetti, M.C.; Gagliano, N.; Milzani, A.; et al. Effects of Physiological and Pathological Urea Concentrations on Human Microvascular Endothelial Cells. Int. J. Mol. Sci. 2022, 24, 691. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, A.M.; Wagdi, M.A.; Salama, A. Rutin Loaded Bilosomes for Enhancing the Oral Activity and Nephroprotective Effects of Rutin in Potassium Dichromate Induced Acute Nephrotoxicity in Rats. Sci. Rep. 2024, 14, 23799. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Ren, X.; Zhang, X.; Wang, G.; Liu, H.; Wang, L. Rutin ameliorate PFOA induced renal damage by reducing oxidative stress and improving lipid metabolism. J. Nutr. Biochem. 2023, 123, 109501. [Google Scholar] [CrossRef]
- Ahmad, A.; Dempsey, S.K.; Daneva, Z.; Azam, M.; Li, N.; Li, P.-L.; Ritter, J.K. Role of Nitric Oxide in the Cardiovascular and Renal Systems. Int. J. Mol. Sci. 2018, 19, 2605. [Google Scholar] [CrossRef] [PubMed]
- Franssen, R.; Monajemi, H.; Stroes, E.S.; Kastelein, J.J. Obesity and Dyslipidemia. Med Clin. N. Am. 2011, 95, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Rutin: An Antioxidant and Vascular—Protecting Agent from Natural Plants. Available online: https://www.greenskybio.com/blog5/rutin-an-antioxidant-and-vascular-protecting-agent-from-natural-plants.html (accessed on 18 July 2025).
- Li, L.; Guo, Z.; Zhao, Y.; Liang, C.; Zheng, W.; Tian, W.; Chen, Y.; Cheng, Y.; Zhu, F.; Xiang, X. The impact of oxidative stress on abnormal lipid metabolism-mediated disease development. Arch. Biochem. Biophys. 2025, 766, 110348. [Google Scholar] [CrossRef]
- Klop, B.; Elte, J.W.F.; Cabezas, M.C. Dyslipidemia in Obesity: Mechanisms and Potential Targets. Nutrients 2013, 5, 1218–1240. [Google Scholar] [CrossRef]
- Manzoni, A.G.; Passos, D.F.; da Silva, J.L.; Bernardes, V.M.; Bremm, J.M.; Jantsch, M.H.; de Oliveira, J.S.; Mann, T.R.; de Andrade, C.M.; Leal, D.B. Rutin and Curcumin Reduce Inflammation, Triglyceride Levels and ADA Activity in Serum and Immune Cells in a Model of Hyperlipidemia. Blood Cells Mol. Dis. 2019, 76, 13–21. [Google Scholar] [CrossRef]
- Kotob, S.E.; Ahmed, H.H.; Mohawed, O.A.; Mahmoud, K.F.; Aglan, H.A. Sodium alginate-based nanoformulation: Optimistic approach for enhancing the anti-obesogenic effect of rutin. Egypt. Pharm. J. 2025, 24, 216–230. [Google Scholar] [CrossRef]
- Klok, M.D.; Jakobsdottir, S.; Drent, M.L. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: A review. Obes. Rev. 2006, 8, 21–34. [Google Scholar] [CrossRef]
- Sahu, A. Leptin signaling in the hypothalamus: Emphasis on energy homeostasis and leptin resistance. Front. Neuroendocrinol. 2003, 24, 225–253. [Google Scholar] [CrossRef]
- Frederich, R.C.; Löllmann, B.; Hamann, A.; Napolitano-Rosen, A.; Kahn, B.B.; Lowell, B.B.; Flier, J.S. Expression of ob mRNA and its encoded protein in rodents. Impact of nutrition and obesity. J. Clin. Investig. 1995, 96, 1658–1663. [Google Scholar] [CrossRef]
- Fantuzzi, G.; Faggioni, R. Leptin in the regulation of immunity, inflammation, and hematopoiesis. J. Leukoc. Biol. 2000, 68, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Gurarie, M. Leptin Resistance: Role in Diet, Obesity, and Hunger. Available online: https://www.verywellhealth.com/leptin-resistance-8684009 (accessed on 18 July 2025).
- Lee, S.-J.; Cho, K.H.; Kim, J.C.; Choo, H.J.; Hwang, J.-Y.; Cho, H.C.; Hah, Y.-S. Comparative analysis of anti-obesity effects of green, fermented, and γ-aminobutyric acid teas in a high-fat diet-induced mouse model. Appl. Biol. Chem. 2024, 67, 36. [Google Scholar] [CrossRef]
- Bano, G. Glucose homeostasis, obesity and diabetes. Best Pract. Res. Clin. Obstet. Gynaecol. 2013, 27, 715–726. [Google Scholar] [CrossRef]
- Wondmkun, Y.T. Obesity, Insulin Resistance, and Type 2 Diabetes: Associations and Therapeutic Implications. Diabetes Metab. Syndr. Obes. 2020, 13, 3611–3616. [Google Scholar] [CrossRef]
- Eshima, H.; Tamura, Y.; Kakehi, S.; Kurebayashi, N.; Murayama, T.; Nakamura, K.; Kakigi, R.; Okada, T.; Sakurai, T.; Kawamori, R.; et al. Long-term, but not short-term high-fat diet induces fiber composition changes and impaired contractile force in mouse fast-twitch skeletal muscle. Physiol. Rep. 2017, 5, e13250. [Google Scholar] [CrossRef] [PubMed]
- Badin, P.-M.; Louche, K.; Mairal, A.; Liebisch, G.; Schmitz, G.; Rustan, A.C.; Smith, S.R.; Langin, D.; Moro, C. Altered skeletal muscle lipase expression and activity contribute to insulin resistance in humans. Diabetes 2011, 60, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, A. Mechanisms of antidiabetic effects of flavonoid rutin. Biomed. Pharmacother. 2017, 96, 305–312. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Abotaleb, M.; Kubatka, P.; Kajo, K.; Büsselberg, D. Flavonoids and Their Anti-Diabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules 2019, 9, 430. [Google Scholar] [CrossRef]
- Adnan, T.; Amin, M.N.; Uddin, G.; Hussain, S.; Sarwar, S.; Hossain, K.; Uddin, S.N.; Islam, M.S. Increased concentration of serum MDA, decreased antioxidants and altered trace elements and macro-minerals are linked to obesity among Bangladeshi population. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 933–938. [Google Scholar] [CrossRef]
- Forman, H.J.; Zhang, H.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Asp. Med. 2009, 30, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Fukai, T.; Ushio-Fukai, M. Superoxide Dismutases: Role in Redox Signaling, Vascular Function, and Diseases. Antioxid. Redox Signal. 2011, 15, 1583–1606. [Google Scholar] [CrossRef]
- Koval’skii, I.V.; Krasnyuk, I.I.; Nikulina, O.I.; Belyatskaya, A.V.; Kharitonov, Y.Y.; Feldman, N.B.; Lutsenko, S.V. Mechanisms of Rutin Pharmacological Action (Review). Pharm. Chem. J. 2014, 48, 73–76. [Google Scholar] [CrossRef]
- Goodman, Z.D. The impact of obesity on liver histology. Clin. Liver Dis. 2014, 18, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, A.; Birk, R. Adipose Tissue Hyperplasia and Hypertrophy in Common and Syndromic Obesity—The Case of BBS Obesity. Nutrients 2023, 15, 3445. [Google Scholar] [CrossRef] [PubMed]
- Almahmoud, S.; Zhong, H.A. Molecular Modeling of Allosteric Site of Isoform-Specific Inhibition of the Peroxisome Proliferator-Activated Receptor PPARγ. Biomolecules 2022, 12, 1614. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-S.; Jung, J.-E.; Marciano, D.P.; Jo, A.; Koo, J.Y.; Choi, S.Y.; Yang, Y.R.; Jang, H.-J.; Kim, E.-K.; Park, J.; et al. PPARγ Antagonist Gleevec Improves Insulin Sensitivity and Promotes the Browning of White Adipose Tissue. Diabetes 2016, 65, 829–839. [Google Scholar] [CrossRef]
- Wong, A.R.; Yang, A.W.H.; Gill, H.; Lenon, G.B.; Hung, A. Mechanisms of Nelumbinis folium targeting PPARγ for weight management: A molecular docking and molecular dynamics simulations study. Comput. Biol. Med. 2023, 166, 107495. [Google Scholar] [CrossRef]


| Biomarker | Groups | ANOVA p-Value | |||
|---|---|---|---|---|---|
| Control | HFD | Rutin | Nano-Rutin | ||
| Glucose (mg/dL) | 92.17 ± 3.53 c | 161.75 ± 8.37 a | 142 ± 1.55 b | 133.5 ± 3.55 b | <0.001 *** |
| Insulin (mIU/L) | 13.31 ± 0.32 c | 19.97 ± 0.88 a | 16.12 ± 0.58 b | 12.94 ± 0.24 c | <0.001 *** |
| Leptin (ng/mL) | 10.43 ± 0.8 d | 28.95 ± 1.06 a | 19.18 ± 0.62 b | 15.58 ± 0.65 c | <0.001 *** |
| SGPT (U/L) | 37.1 ± 2.48 c | 52.29 ± 1.13 a | 45.03 ± 1.06 b | 37.06 ± 2.31 c | <0.001 *** |
| SGOT (U/L) | 130.71 ± 2.93 c | 148.8 ± 1.44 a | 136.92 ± 1.81 b | 133.33 ± 1.21 b,c | <0.001 *** |
| ALP (mg/dL) | 275 ± 5.48 b | 340.55 ± 8.31 a | 245.75 ± 10.08 c | 237.3 ± 4.36 c | <0.001 *** |
| DBIL (μg/dL) | 13.48 ± 0.51 b | 16.83 ± 1.16 a | 13.36 ± 0.29 b | 13.22 ± 0.36 b | <0.001 *** |
| Creatinine (mg/dL) | 0.37 ± 0.02 c | 0.57 ± 0.01 a | 0.52 ± 0.02 b | 0.5 ± 0.01 b | <0.001 *** |
| Urea (mg/dL) | 19.52 ± 0.32 b | 21.76 ± 0.37 a | 20.85 ± 0.06 a | 17.71 ± 0.52 c | <0.001 *** |
| TC (mg/dL) | 102 ± 1.93 d | 140.85 ± 1.6 a | 134.68 ± 2.04 b | 128.07 ± 1.79 c | <0.001 *** |
| TG (mg/dL) | 86.58 ± 2.24 c | 160.75 ± 3.31 a | 136.68 ± 2.87 b | 127.9 ± 11.12 b | <0.001 *** |
| LDL (mg/dL) | 23.77 ± 0.38 c | 61.36 ± 2.18 a | 49.07 ± 1.96 b | 48.11 ± 1.41 b | <0.001 *** |
| HDL (mg/dL) | 35.33 ± 1.94 a | 17.53 ± 2.81 c | 23.13 ± 1.50 b,c | 28.81 ± 1.56 b | <0.001 *** |
| Biomarker | Group | ANOVA p-Value | |||
|---|---|---|---|---|---|
| Control | HFD | Rutin | Nano-Rutin | ||
| MDA (U/µL) | 1.29 ± 0.13 d | 8.43 ± 0.2 a | 7.04 ± 0.18 b | 6.57 ± 0.08 c | <0.001 *** |
| GSH (µmol/L) | 4.08 ± 0.22 a | 1.72 ± 0.18 c | 3.16 ± 0.13 b | 4.05 ± 0.08 a | <0.001 *** |
| SOD (nmol/mL) | 0.89 ± 0.05 a | 0.42 ± 0.03 b | 0.37 ± 0 b | 0.40 ± 0 b | <0.001 *** |
| Parameters | Corrected Model | HFD | Rutin | Nano-Rutin (RuN) | ||||
|---|---|---|---|---|---|---|---|---|
| F | p-Value | F | p-Value | F | p-Value | F | p-Value | |
| Wilk’s Lambda | 223.7 | <0.001 *** | 36.3 | <0.001 *** | 67.6 | <0.001 *** | ||
| SGPT | 15.4 | <0.001 *** | 33.2 | <0.001 *** | 7.6 | 0.012 * | 33.4 | <0.001 *** |
| SGOT | 16.6 | <0.001 *** | 42.6 | <0.001 *** | 18.4 | <0.001 *** | 31.1 | <0.001 *** |
| D.BIL | 6.6 | 0.003 ** | 12.3 | 0.002 ** | 13.1 | 0.002 ** | 14.3 | 0.001 *** |
| ALK.PH | 39.9 | <0.001 *** | 39.1 | <0.001 *** | 81.8 | <0.001 *** | 97.1 | <0.001 *** |
| UREA | 24.1 | <0.001 *** | 19.6 | <0.001 *** | 3.2 | 0.088 ns | 63.7 | <0.001 *** |
| CREAT | 27.9 | <0.001 *** | 77.6 | <0.001 *** | 5.5 | 0.029 * | 10.4 | 0.004 ** |
| SOD | 72.1 | <0.001 *** | 108.9 | <0.001 *** | 5.9 | 0.025 * | 3.1 | 0.093 ns |
| MDA | 405.0 | <0.001 *** | 1053.8 | <0.001 *** | 40.0 | <0.001 *** | 71.2 | <0.001 *** |
| GSH | 45.2 | <0.001 *** | 102.9 | <0.001 *** | 38.0 | <0.001 *** | 100.0 | <0.001 *** |
| Glucose | 35.2 | <0.001 *** | 99.3 | <0.001 *** | 8.0 | 0.010 ** | 16.4 | 0.001 *** |
| Insulin | 33.4 | <0.001 *** | 70.1 | <0.001 *** | 23.4 | <0.001 *** | 78.1 | <0.001 *** |
| Leptin | 95.3 | <0.001 *** | 267.6 | <0.001 *** | 74.4 | <0.001 *** | 139.4 | <0.001 *** |
| Cholesterol | 85.6 | <0.001 *** | 221.4 | <0.001 *** | 5.6 | 0.028 * | 24.0 | <0.001 *** |
| TG | 25.8 | <0.001 *** | 74.5 | <0.001 *** | 7.8 | 0.011 * | 14.6 | 0.001 *** |
| HDL | 14.2 | <0.001 *** | 37.5 | <0.001 *** | 22.8 | <0.001 *** | 20.4 | <0.001 *** |
| LDL | 92.6 | <0.001 *** | 264.1 | <0.001 *** | 28.3 | <0.001 *** | 32.8 | <0.001 *** |
| Target | Interaction Type | Residues/Regions Involved | Number of Interactions |
|---|---|---|---|
| PPARγ + SR1664 | Conventional Hydrogen bond | TYR327, PHE282, GLN286 | 3 |
| Pi-Alkyl | CYS285, ARG288, ILE341, LEU330 | 7 | |
| Pi-Sigma | LEU330 | 1 | |
| Pi-Sulfur | MET364 | 1 | |
| Carbon-Hydrogen bond | LYS367 | 1 | |
| PPARγ + Rutin | Conventional Hydrogen bond | ILE326, GLU295, SER342, ARG288, SER289 | 5 |
| Pi-Sulfur | CYS285 | 2 | |
| Pi-Alkyl | LEU330, CYS285, ARG288, ILE341 | 6 | |
| Amide-Pi stacked | GLY284 | 2 | |
| Unfavorable Acceptor-Acceptor | GLU343 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zahran, N.H.; Abu-Elsaoud, A.M.; Mohamed, A.S.; Marie, O.M. Nanoparticle-Delivered Rutin Prevents Metabolic and Oxidative Imbalance in Obesity Triggered by a High-Fat Diet: In Vivo and In Silico Studies. Biomedicines 2025, 13, 2106. https://doi.org/10.3390/biomedicines13092106
Zahran NH, Abu-Elsaoud AM, Mohamed AS, Marie OM. Nanoparticle-Delivered Rutin Prevents Metabolic and Oxidative Imbalance in Obesity Triggered by a High-Fat Diet: In Vivo and In Silico Studies. Biomedicines. 2025; 13(9):2106. https://doi.org/10.3390/biomedicines13092106
Chicago/Turabian StyleZahran, Nourhan H., Abdelghafar M. Abu-Elsaoud, Ayman Saber Mohamed, and Ohoud M. Marie. 2025. "Nanoparticle-Delivered Rutin Prevents Metabolic and Oxidative Imbalance in Obesity Triggered by a High-Fat Diet: In Vivo and In Silico Studies" Biomedicines 13, no. 9: 2106. https://doi.org/10.3390/biomedicines13092106
APA StyleZahran, N. H., Abu-Elsaoud, A. M., Mohamed, A. S., & Marie, O. M. (2025). Nanoparticle-Delivered Rutin Prevents Metabolic and Oxidative Imbalance in Obesity Triggered by a High-Fat Diet: In Vivo and In Silico Studies. Biomedicines, 13(9), 2106. https://doi.org/10.3390/biomedicines13092106







