On the Merits of Targeted and Individualized Physical Exercise in Persons with Diabetic Foot Disease—From Controversies to Consensus
Abstract
1. Introduction
2. Basis for the Narrative Review
3. Consequences of Diabetic Foot Disease
| Stage/Event | Pathophysiological Description | Physical Consequences | Functional Consequences |
|---|---|---|---|
| 1. Neuropathy (sensory, motor, autonomic) | loss of protective sensation, muscle weakness, dry/cracked skin | foot deformities, skin fissures | increased risk of injury, impaired proprioception, need for rehabilitation |
| 2. Angiopathy (ischemia) | impaired micro- and macrovascular circulation, tissue hypoxia | cold, pale foot, diminished or absent pulses | claudication, poor wound healing, increased risk of amputation revascularization if possible, need for rehabilitation |
| 3. Ulcer formation | due to unnoticed trauma or pressure in insensate areas | chronic wounds, often slow to heal | difficulty walking, need for footwear modification or reduced mobility, need for rehabilitation |
| 4. Infection | rapid bacterial colonization of ulcers, potentially spreading to deep tissues | swelling, redness, abscess, osteomyelitis | hospitalization, immobilization, rehabilitation * |
| 5. Tissue necrosis and gangrene | resulting from uncontrolled infection and/or ischemia | irreversible tissue damage, necrotic areas | surgical intervention or amputation often required, need for rehabilitation |
| 6. Amputation (partial or complete) | removal of necrotic tissue to prevent systemic infection or preserve life/function | loss of limb or part of the limb/foot, postural imbalance | loss of independence, need for rehabilitation, psychological impact, social isolation |
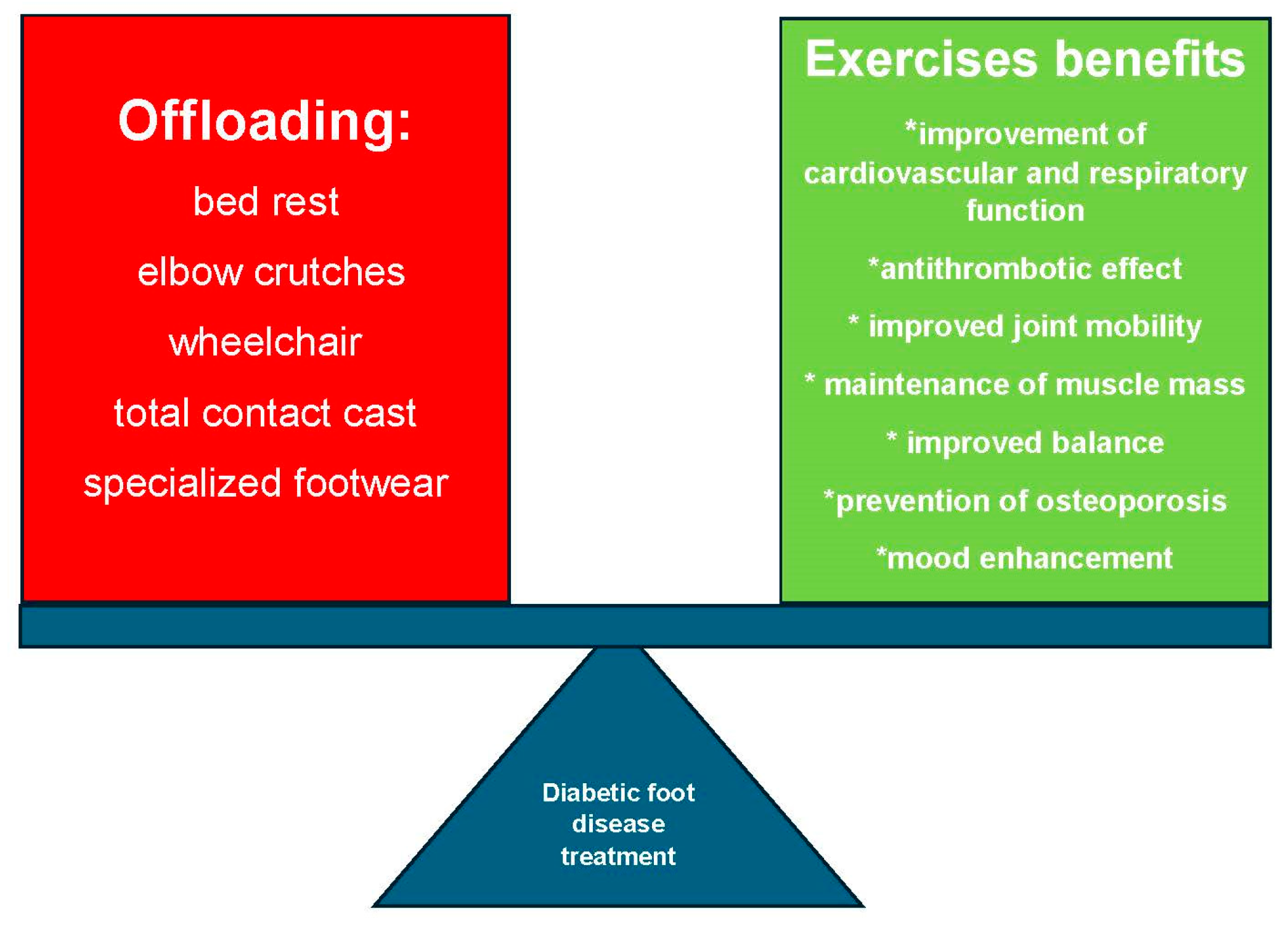
4. The Role of Exercises in the Therapeutic Process of Diabetic Foot Disease
4.1. Multimodal Treatment of DFD
4.2. The Effect of Exercise on the Local Condition in DFD
4.3. Impact of Exercises on the General Condition of a Patient with DFD
4.4. Discussion of Example Exercises for Patients with Diabetic Foot Disease and Contraindications or Precautions for Their Performance
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, L.; Pei, J.H.; Kuang, J.; Chen, H.-M.; Chen, Z.; Li, Z.-W.; Yang, H. Effect of lifestyle intervention in patients with type 2 diabetes: A meta-analysis. Metabolism 2015, 64, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.E.; Elliott, E.J.; Naughton, G.A. Exercise for type 2 diabetes mellitus. In Cochrane Database of Systematic Reviews; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar] [CrossRef]
- American Diabetes Association. Standards of medical care in diabetes—2021. Diabetes Care 2021, 44 (Suppl. S1), S15–S33. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Globalization of Diabetes. Diabetes Care 2011, 34, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Little, J.P.; Francois, M.E. High-intensity interval training for improving postprandial hyperglycemia. Res. Q. Exerc. Sport 2014, 85, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Mann, S.; Beedie, C.; Balducci, S.; Zanuso, S.; Allgrove, J.; Bertiato, F.; Jimenez, A. Changes in insulin sensitivity in response to different modalities of exercise: A review of the evidence. Diabetes Metab. Res. Rev. 2014, 30, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Shaban, N.; Kenno, K.A.; Milne, K.J. The effects of a 2 week modified high intensity interval training program on the homeostatic model of insulin resistance (HOMA-IR) in adults with type 2 diabetes. J. Sport. Med. Phys. Fit. 2014, 54, 203–209. [Google Scholar]
- Kilgour, A.H.M.; Rutherford, M.; Higson, J.; Meredith, S.J.; McNiff, J.; Mitchell, S.; Wijayendran, A.; Lim, S.E.R.; Shenkin, S.D. Barriers and motivators to undertaking physical activity in adults over 70—A systematic review of the quantitative literature. Age Ageing 2024, 53, afae080. [Google Scholar] [CrossRef] [PubMed]
- Biernat, K.; Marciniak, D.M.; Mazurek, J.; Kuciel, N.; Hap, K.; Kisiel, M.; Sutkowska, E. The Level and Limitations of Physical Activity in Elderly Patients with Diabetes. J. Clin. Med. 2024, 13, 6329. [Google Scholar] [CrossRef] [PubMed]
- Ajzen, I. The theory of planned behavior. Organ. Behav. Hum. Decis. Process. 1991, 50, 179–211. [Google Scholar] [CrossRef]
- Sutkowska, E.; Biernat, K.; Mazurek, J.; Hap, K.; Kuciel, N.; Sutkowska, M.; Marciniak, D. Level and limitations of physical activity in patients with excess body weight or diabetes. BMJ Open Sport Exerc. Med. 2024, 10, e002041. [Google Scholar] [CrossRef] [PubMed]
- van Netten, J.J.; Bus, S.A.; Apelqvist, J.; Lipsky, B.A.; Hinchliffe, R.J.; Game, F.; Jeffcoate, W.; Jirkovska, A.; Malone, M.; Rasmussen, A.; et al. Definitions and Criteria for Diabetic Foot Disease. IWGDF Guidelines. Available online: https://iwgdfguidelines.org/wp-content/uploads/2019/05/definitions-and-criteria-final.pdf (accessed on 28 May 2025).
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers and Their Recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Armstrong, D.G.; Lipsky, B.A. Preventing foot ulcers in patients with diabetes. JAMA 2005, 293, 217–228, International Working Group on the Diabetic Foot (IWGDF) Guidelines. Available online: https://iwgdfguidelines.org/ (accessed on 10 July 2025). [CrossRef] [PubMed]
- WHO/IDF Saint Vincent Declaration Working Group. Diabetes mellitus in Europe: A problem at all ages in all countries. A model for prevention and self care. Acta Diabetol. 1990, 27, 181–183. [Google Scholar]
- Kong, M.F.; Gregory, R. Preventing foot complications in diabetes: The St Vincent Declaration 26 years on. Pract. Diabetes 2016, 33, 154–156. [Google Scholar] [CrossRef]
- Matos, M.; Mendes, R.; Silva, A.B.; Sousa, N. Physical activity and exercise on diabetic foot related outcomes: A systematic review. Diabetes Res. Clin. Pract. 2018, 139, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.J.; Tuttle, L.J.; Lemaster, J.W.; Strube, M.J.; McGill, J.B.; Hastings, M.K.; Sinacore, D.R. Weight-bearing versus nonweight-bearing exercise for persons with diabetes and peripheral neuropathy: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2013, 94, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Sartor, C.D.; Hasue, R.H.; Cacciari, L.P.; Butugan, M.K.; Watari, R.; Pássaro, A.C.; Giacomozzi, C.; Sacco, I.C.N. Effects of strengthening, stretching and functional training on foot function in patients with diabetic neuropathy: Results of a randomized controlled trial. BMC Musculoskelet. Disord. 2014, 15, 137. [Google Scholar] [CrossRef] [PubMed]
- Lemaster, J.W.; Mueller, M.J.; Reiber, G.E.; Mehr, D.R.; Madsen, R.W.; Conn, V.S. Effect of weight-bearing activity on foot ulcer incidence in people with diabetic peripheral neuropathy: Feet first randomized controlled trial. Phys. Ther. 2008, 88, 1385–1398. [Google Scholar] [CrossRef] [PubMed]
- Balducci, S.; Iacobellis, G.; Parisi, L.; Di Biase, N.; Calandriello, E.; Leonetti, F.; Fallucca, F. Exercise training can modify the natural history of diabetic peripheral neuropathy. J. Diabetes Complicat. 2006, 20, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Dixit, S.M.; Maiya, A.G.; Shastry, B.A. Effect of aerobic exercise on peripheral nerve functions of population with diabetic peripheral neuropathy in type 2 diabetes: A single blind, parallel group randomized controlled trial. J. Diabetes Complicat. 2014, 28, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Francia, P.; Anichini, R.; De Bellis, A.; Seghieri, G.; Lazzeri, R.; Paternostro, F. Gulisano, M Diabetic foot prevention: The role of exercise therapy in the treatment of limited joint mobility, muscle weakness and reduced gait speed. Ital. J. Anat. Embryol. 2015, 120, 21–32. [Google Scholar] [PubMed]
- Tran, M.M.; Haley, M.N. Does exercise improve healing of diabetic foot ulcers? A systematic review. J. Foot Ankle Res. 2021, 14, 19. [Google Scholar] [CrossRef] [PubMed]
- Eraydin, S.; Avşar, G. The effect of foot exercises on wound healing in type 2 diabetic patients with a foot ulcer: A randomized control study. J. Wound Ostomy Cont. Nurs. 2018, 45, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Rietz, M.; Lehr, A.; Mino, E.; Lang, A.; Szczerba, E.; Schiemann, T.; Herder, C.; Saatmann, N.; Geidl, W.; Barbaresko, J.; et al. Physical Activity and Risk of Major Diabetes-Related Complications in Individuals with Diabetes: A Systematic Review and Meta-Analysis of Observational Studies. Diabetes Care 2022, 45, 3101–3111. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.; Dinh, Q.; Yu, F.; Jia, S.; Wang, X. Interventional effects of exercise on neuropathy in patients with diabetes: A systematic review with meta-analysis. BMC Sports Sci. Med. Rehabil. 2025, 17, 82. [Google Scholar] [CrossRef] [PubMed]
- Lyu, X.; Li, S.; Peng, S.; Cai, H.; Liu, G.; Ran, X. Intensive walking exercise for lower extremity peripheral arterial disease: A systematic review and meta-analysis. J. Diabetes 2016, 8, 363–377. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Young, A.; McNaught, C.E. The physiology of wound healing. Surgery 2017, 35, 473–477. [Google Scholar] [CrossRef]
- Cañedo-Dorantes, L.; Cañedo-Ayala, M. Skin acute wound healing: A comprehensive review. Int. J. Inflamm. 2019, 2019, 3706315. [Google Scholar] [CrossRef] [PubMed]
- Aagaard, T.V.; Moeini, S.; Skou, S.T.; Madsen, U.R.; Brorson, S. Benefits and harms of exercise therapy for patients with diabetic foot ulcers: A systematic review. Int. J. Low. Extrem. Wounds 2020, 21, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Otranto, M.; Nascimento, A.P.; Monte-Alto-Costa, A. Insulin resistance impairs cutaneous wound healing in mice. Wound Repair Regen. 2013, 21, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Winget, C.M.; Deroshia, C.W. Psychosocial and Chronophysiological Effects of Inactivity and Immobilization. In Inactivity: Physiological Effects; Sandler, H., Vernikos, J., Eds.; Academic Press: Cambridge, MA, USA, 1986; pp. 123–147. [Google Scholar]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.-M.; Nieman, D.C.; Swain, D.P. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef] [PubMed]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.-P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef] [PubMed]
- Greenleaf, J.E.; Kozlowski, S. Physiological consequences of reduced physical activity during bed rest. Exerc. Sport Sci. Rev. 1982, 10, 84–119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, X.; Zhao, X.; Li, B.; Cai, Y.; Liang, X.; Wan, Q. Effect of exercise on bone mineral density among patients with osteoporosis and osteopenia: A systematic review and network meta-analysis. J. Clin. Nurs. 2022, 31, 2100–2111. [Google Scholar] [CrossRef] [PubMed]
- Nejatian, M.M.; Sobhi, S.; Sanchez, B.N.; Linn, K.; Manning, L.; Soh, S.-C.; Hiew, J.; Ritter, J.C.; Yeap, B.B.; Hamilton, E.J.; et al. Reduction in femoral neck and total hip bone mineral density following hospitalisation for diabetes-related foot ulceration. Sci. Rep. 2021, 11, 22742. [Google Scholar] [CrossRef] [PubMed]
- Colberg, S.R.; Sigal, R.J.; Yardley, J.E.; Riddell, M.C.; Dunstan, D.W.; Dempsey, P.C.; Horton, E.S.; Castorino, K.; Tate, D.F. Physical Activity/Exercise and Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care 2016, 39, 2065–2079. [Google Scholar] [CrossRef] [PubMed]
- Wound Healing Interventions Guideline (2023 Update). Available online: https://iwgdfguidelines.org/wound-healing-2023/ (accessed on 28 May 2025).
- Wendland, D.M.; Altenburger, E.A.; Swen, S.B.; Haan, J.D. Diabetic Foot Ulcer Beyond Wound Closure: Clinical Practice Guideline. Phys. Ther. 2025, 105, pzae171. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wendland, D.M.; Kline, P.W.; Simmons, L.; Sinacore, D.R. The effect of exercise, physical activity, stepping characteristics, and loading on diabetic foot ulcer healing: A systematic review. Wounds 2023, 35, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Nayak, D.; Ray, R.; Anand, C.; Baruah, T.D.; Goyal, A. Protocolized Foot Exercises Help in Improvement of Diabetic Foot Ulcers and Quality of Life When Added to Standard Therapy–A Randomized Clinical Trial. Int. J. Low. Extrem. Wounds 2024, 15347346241262591. [Google Scholar] [CrossRef] [PubMed]
- Sekhar, M.S.; Thomas, R.R.; Unnikrishnan, M.K.; Vijayanarayana, K.; Rodrigues, G.S. Impact of diabetic foot ulcer on health-related quality of life: A cross-sectional study. Semin. Vasc. Surg. 2015, 28, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Mohseni, H.; Wrobel, J.S. Functional deficits in diabetic foot patients with and without neuropathy. J. Diabetes Res. 2021, 2021, 6636030. [Google Scholar]
- D’Silva, S.; Ziarno, N.; Kluding, P. Postural instability and diabetic peripheral neuropathy: A review. J. Diabetes Complicat. 2020, 34, 107469. [Google Scholar] [CrossRef]
- Morone, G.; Bragazzi, N.L.; Invernizzi, M.; Fusco, A.; Paolucci, T.; Paci, M. Biomechanical alterations in diabetic neuropathic foot: Impact on balance and gait. Clin. Biomech. 2022, 87, 105446. [Google Scholar] [CrossRef]
- Sinacore, D.R.; Riddle, D.L.; Mueller, M.J. Muscle and joint impairments in diabetic neuropathy and their impact on function. J. Am. Podiatr. Med. Assoc. 2018, 108, 205–213. [Google Scholar] [CrossRef]
- Wukich, D.K.; Sung, W. Bone and joint disorders in diabetic foot disease. Diabetes Metab. Res. Rev. 2021, 37, e3402. [Google Scholar] [CrossRef]
- Lavery, L.A.; Armstrong, D.G.; Murdoch, D.P.; Peters, E.J.; Lipsky, B.A. Effect of diabetic foot ulcers on patient independence and quality of life. Diabetes Metab. Res. Rev. 2020, 36, e3294. [Google Scholar] [CrossRef]
- Prompers, L.; Schaper, N.; Apelqvist, J.; Edmonds, M.; Jude, E.; Mauricio, D.; Uccioli, L.; Urbancic, V.; Bakker, K.; Holstein, P.; et al. Functional consequences of diabetic foot disease: A systematic review. Diabetologia 2023, 66, 367–375. [Google Scholar] [CrossRef]
- Turan, Y.; Ertugrul, B.M.; Lipsky, B.A.; Bayraktar, K. Does physical therapy and rehabilitation improve outcomes for diabetic foot ulcers? World J. Exp. Med. 2015, 5, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Jude, E.; Langton, K.; Martínez-De Jesus, F.R.; Harkless, L.B.; Gawish, H.; Huang, Y.Y.; Labovitz, J.; Xu, Z.; Pendsey, S.; et al. IDF Clinical Practice Recommendations on the Diabetic Foot—2017. A Guide for Healthcare Professionals. Available online: https://idf.org/media/uploads/2023/05/attachments-61.pdf (accessed on 2 August 2024).
- Faglia, E.; Caravaggi, C.; Clerici, G.; Sganzaroli, A.; Curci, V.; Vailati, W.; Simonetti, D.; Sommalvico, F.; Manzoli, L.; Giurato, L.; et al. Effectiveness of removable walker cast versus nonremovable fiberglass off-bearing cast in the healing of diabetic plantar foot ulcer: A randomized controlled trial. Diabetes Care 2010, 33, 1419–1423. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.; Backhouse, M.R.; Bruce, J. Rehabilitation for people wearing offloading devices for diabetes-related foot ulcers: A systematic review and meta-analyses. J. Foot Ankle Res. 2023, 16, 16. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Grewal, G.S.; Bharara, M.; Menzies, R.; Talal, T.K.; Armstrong, D.G. Can’t Stand the Pressure: The Association Between Unprotected Standing, Walking, and Wound Healing in People with Diabetes. J. Diabetes Sci. Technol. 2017, 11, 657–667. [Google Scholar] [CrossRef] [PubMed]
- Gracia-Sánchez, A.; López-Pineda, A.; Lázaro-Martínez, J.L.; Pérez, A.; Pomares-Gómez, F.J.; Fernández-Seguín, L.M.; Martínez-de-Quel, Ó.; Montero, P.; Suso-Ribera, C.; Ortega-Avila, A.J.; et al. Consensus-based recommendations on physical activity and exercise in patients with diabetes at risk of foot ulcerations: A Delphi study. Braz. J. Phys. Ther. 2023, 27, 100500. [Google Scholar] [CrossRef] [PubMed]
- Pence, B.D.; Woods, J.A. Exercise, obesity, and cutaneous wound healing: Evidence from rodent and human studies. Adv. Wound Care 2014, 3, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Keylock, T.; Meserve, L.; Wolfe, A. Low-intensity Exercise Accelerates Wound Healing in Diabetic Mice. Wounds 2018, 30, 68–71. [Google Scholar] [PubMed]
- Rogers, L.C.; Frykberg, R.G.; Armstrong, D.G.; Boulton, A.J.M.; Edmonds, M.; Ha Van, G.; Hartemann, A.; Game, F.; Jeffcoate, W.; Jirkovska, A.; et al. The Charcot foot in diabetes. Diabetes Care 2011, 34, 2123–2129. [Google Scholar] [CrossRef] [PubMed]
- Aitken, E.; Hiew, J.; Hamilton, E.J.; Manning, L.; Ritter, J.C.; Raby, E.; Gittings, P.M. Exercise in adults admitted to hospital with diabetes-related foot ulcers: A pilot study of feasibility and safety. J. Foot Ankle Res. 2023, 16, 18. [Google Scholar] [CrossRef] [PubMed]
- Kanade, R.V.; van Deursen, R.W.; Harding, K.; Price, P. Walking performance in people with diabetic neuropathy: Benefits and threats. Diabetologia 2006, 49, 1747–1754. [Google Scholar] [CrossRef] [PubMed]
- Mathur, N.; Pedersen, B.K. Exercise as a means to control low-grade systemic inflammation. Mediat. Inflamm. 2008, 2008, 109502. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Xin, R.; Li, X.; Zhang, X.; Chen, Z.; Zhu, Q.; Tai, Z.; Bao, L. Physical therapy in diabetic foot ulcer: Research progress and clinical application. Int. Wound J. 2023, 20, 3417–3434. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K. Anti-inflammatory effects of exercise: Role in diabetes and cardiovascular disease. Eur. J. Clin. Investig. 2017, 47, 600–611. [Google Scholar] [CrossRef] [PubMed]
- Herder, C.; Peltonen, M.; Koenig, W.; Sütfels, K.; Lindström, J.; Martin, S.; Ilanne-Parikka, P.; Eriksson, J.G.; Aunola, S.; Keinänen-Kiukaanniemi, S.; et al. Anti-inflammatory effect of lifestyle changes in the Finnish Diabetes Prevention Study. Diabetologia 2009, 52, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Yates, T.; Davies, M.J.; Gorely, T.; Talbot, D.; Bull, F.; Sattar, N.; Khunti, K. The effect of increased ambulatory activity on markers of chronic low-grade inflammation: Evidence from the PREPARE programme randomized controlled trial. Diabet. Med. 2010, 27, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.; Qiu, P.; Xia, R.; Lin, H.; Ye, B.; Tao, J.; Chen, L.; Zhang, Y.; Wang, Y.; Liu, J.; et al. Effect of aerobic exercise on inflammatory markers in healthy middle-aged and older adults: A systematic review and meta-analysis of randomized controlled trials. Front. Aging Neurosci. 2019, 11, 98. [Google Scholar] [CrossRef] [PubMed]
- Taherkhani, S.; Suzuki, K.; Castell, L. A short overview of changes in inflammatory cytokines and oxidative stress in response to physical activity and antioxidant supplementation. Antioxidants 2020, 9, 886. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Åkerström, T.C.A.; Nielsen, A.R.; Fischer, C.P. Role of myokines in exercise and metabolism. J. Appl. Physiol. 2007, 103, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Schaper, N.; Rayman, G. Microangiopathy: Is it relevant to wound healing in diabetic foot disease? Diabetes Metab. Res. Rev. 2020, 36, e3244. [Google Scholar] [CrossRef] [PubMed]
- Versey, Z.; da Cruz Nizer, W.S.; Russell, E.; Zigic, S.; DeZeeuw, K.G.; Marek, J.E.; Overhage, J.; Cassol, E. Biofilm-innate immune Interface: Contribution to chronic wound formation. Front. Immunol. 2021, 12, 648554. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.S.; Chang, C.C.; Su, C.L.; Li, J.-R.; Chen, M.-L.; Chen, M.-Y.; Huang, Y.-K. The assessment of Buerger’s exercise on dorsal foot skin circulation in patients with vasculopathic diabetic foot ulcer by using wireless near-infrared spectroscope: A cohort prospective study. Lasers Med. Sci. 2018, 33, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Chen, M.Y.; Shen, J.H.; Lin, Y.B.; Hsu, W.W.; Lin, B.S. A quantitative real-time assessment of Buerger exercise on dorsal foot peripheral skin circulation in patients with diabetes foot. Medicine 2016, 95, e5334. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.F.; Chang, C.C.; Hwang, S.L.; Chen, M.Y. Effects of Buerger Exercise Combined Health-Promoting Program on Peripheral Neurovasculopathy Among Community Residents at High Risk for Diabetic Foot Ulceration. Worldviews Evid.-Based Nurs. 2015, 12, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.Y.L.; Zheng, Y.-P.; Cheing, G.L.Y. Epidermal thickness and biomechanical properties of plantar tissues in diabetic foot. Ultrasound Med. Amp. Biol. 2011, 37, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, J.R.; Lidtke, R.H.; Shott, S. The effects of range-of-motion therapy on the plantar pressures of patients with diabetes mellitus. J. Am. Podiatr. Med. Assoc. 2002, 92, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Jarl, G.; van Netten, J.J.; Lazzarini, P.A.; Crews, R.T.; Najafi, B.; Mueller, M.J. Should weight-bearing activity be reduced during healing of plantar diabetic foot ulcers, even when using appropriate offloading devices? Diabetes Res. Clin. Pract. 2021, 175, 108733. [Google Scholar] [CrossRef] [PubMed]
- Coombes, B.K.; Sierra-Silvestre, E.; Bisset, L.M.; Mielke, G.I.; Ware, R.S.; Coombes, J.S.; Burton, N.W. Pain exacerbation following physical activity in adults with diabetic neuropathy: Ecological momentary assessment of foot symptoms. Aust. J. Gen. Pract. 2024, 53, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A.; Eijsvogels, T.M.H.; Pandey, A.; Quindry, J.; Toth, P.P. Physical activity, cardiorespiratory fitness, and cardiovascular health: A clinical practice statement of the ASPC Part I: Bioenergetics, contemporary physical activity recommendations, benefits, risks, extreme exercise regimens, potential maladaptations. Am. J. Prev. Cardiol. 2022, 12, 100424. [Google Scholar] [CrossRef] [PubMed]
- Fiuza-Luces, C.; Garatachea, N.; Berger, N.A.; Lucia, A. Exercise is the real polypill. Physiology 2013, 28, 330–358. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.M.; Shiroma, E.J.; Lobelo, F.; Puska, P.; Blair, S.N.; Katzmarzyk, P.T.; Alkandari, J.R.; Andersen, L.B.; Bauman, A.E.; Brownson, R.C.; et al. Effect of physical inactivity on major non-communicable diseases worldwide: An analysis of burden of disease and life expectancy. Lancet 2012, 380, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Saltin, B. Exercise as medicine—Evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 2015, 25, 1–72. [Google Scholar] [CrossRef]
- Peng, C.J.; Chen, S.; Yan, S.J.; Zhao, J.N.; Luo, Z.W.; Qian, Y.; Zhao, G.L. Mechanism underlying the effects of exercise against type 2 diabetes: A review on research progress. World J. Diabetes 2024, 15, 1704–1720. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Zhang, X.; Guo, J.; Roberts, C.K.; McKenzie, S.; Wu, W.-C.; Liu, S.; Song, Y. Effects of exercise training on cardiorespiratory fitness and biomarkers of cardiometabolic health: A systematic review and meta-analysis of randomized controlled trials. J. Am. Heart Assoc. 2015, 4, e002014. [Google Scholar] [CrossRef] [PubMed]
- Hafstad, A.D.; Lund, J.; Handler-Olsen, E.; Höper, A.C.; Larsen, T.S.; Aasum, E. High- and moderate-intensity training normalizes ventricular function and mechanoenergetics in mice with diet-induced obesity. Diabetes 2013, 62, 2287–2294. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, M.H. Physical activity-induced remodeling of vasculature in skeletal muscle: Role in treatment of type 2 diabetes. J. Appl. Physiol. 2016, 120, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Olver, T.D.; Laughlin, M.H. Endurance, interval sprint, and resistance exercise training: Impact on microvascular dysfunction in type 2 diabetes. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H337–H350. [Google Scholar] [CrossRef] [PubMed]
- Nataraj, M.; Maiya, G.A.; Nagaraju, S.P.; Shastry, B.A.; Shivashankara, K.N.; Shetty, S.; Mayya, S.S. Effect of exercise-based rehabilitation on functional capacity and renal function in type 2 diabetes mellitus with nephropathy: A randomized controlled trial. Int. Urol. Nephrol. 2024, 56, 2671–2682. [Google Scholar] [CrossRef] [PubMed]
- Amaravadi, S.K.; Maiya, G.A.; Vaishali, K.; Shastry, B.A. Effectiveness of structured exercise program on insulin resistance and quality of life in type 2 diabetes mellitus—A randomized controlled trial. PLoS ONE 2024, 19, e0302831. [Google Scholar] [CrossRef] [PubMed]
- Keylock, K.T.; Vieira, V.J.; Wallig, M.A.; DiPietro, L.; Schrementi, M.; Woods, J.A. Exercise accelerates cutaneous wound healing and decreases wound inflammation in aged mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Zogaib, F.G.; Monte-Alto-Costa, A. Moderate intensity physical training accelerates healing of full-thickness wounds in mice. Braz. J. Med. Biol. Res. 2011, 44, 1025–1035. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Liu, G.H.; Yang, S.H.; Mi, B.B.; Ye, S.N. Low-intensity treadmill exercise promotes rat dorsal wound healing. J. Huazhong Univ. Sci. Technol. Med. Sci. 2016, 36, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yuan, T.; Wang, J.H. Moderate treadmill running exercise prior to tendon injury enhances wound healing in aging rats. Oncotarget 2016, 7, 8498–8512. [Google Scholar] [CrossRef] [PubMed]
- Emery, C.F.; Kiecolt-Glaser, J.K.; Glaser, R.; Malarkey, W.B.; Frid, D.J. Exercise accelerates wound healing among healthy older adults: A preliminary investigation. J. Gerontol. Ser. A 2005, 60, 1432–1436. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.D.; Sheldahl, L.M.; Buley, K.J.; Sandford, P.R. Physiological comparison of walking among bilateral above-knee amputee and able-bodied subjects, and a model to account for the differences in metabolic cost. Arch. Phys. Med. Rehabil. 1997, 78, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Amaro-Gahete, F.J.; Sanchez-Delgado, G.; Alcantara, J.M.A.; Martinez-Tellez, B.; Acosta, F.M.; Merchan-Ramirez, E.; Löf, M.; Labayen, I.; Ruiz, J. Energy expenditure differences across lying, sitting, and standing positions in young healthy adults. PLoS ONE 2019, 14, e0217029. [Google Scholar] [CrossRef]
- Keyser, R.E.; Rasch, E.K.; Finley, M.; Rodgers, M.M. Improved upper-body endurance following a 12-week home exercise program for manual wheelchair users. J. Rehabil. Res. Dev. 2003, 40, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Vokac, Z.; Bell, H.; Bautz-Holter, E.; Rodahl, K. Oxygen uptake/heart rate relationship in leg and arm exercise, sitting and standing. J. Appl. Physiol. 1975, 39, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Olivier, N.; Legrand, R.; Rogez, J.; Berthoin, S.; Prieur, F.; Weissland, T. One-leg cycling versus arm cranking: Which is most appropriate for physical conditioning after knee surgery? Arch. Phys. Med. Rehabil. 2008, 89, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.; Puddiford, M.; Talbot, C.; Price, M. The validity and reproducibility of perceptually regulated exercise responses during combined arm + leg cycling. Eur. J. Appl. Physiol. 2020, 120, 2203–2212. [Google Scholar] [CrossRef] [PubMed]
- Moran, J.; Murphy, A.; Murphy, D.; Austin, A.; Moran, D.; Cronin, C.; Guinan, E.; Hussey, J. The energy expenditure of non-weight bearing crutch walking on the level and ascending stairs. Gait Posture 2015, 42, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Carter, S.E.; Jones, M.; Gladwell, V.F. Energy expenditure and heart rate response to breaking up sedentary time with three different physical activity interventions. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Volkers, K.M.; van Dijk, T.C.W.; Eggermont, L.H.; Hollander, A.P.; Scherder, E.J.A. The intensity of chair-assisted exercises in cognitively healthy older adults. J. Aging Phys. Act. 2013, 21, 260–271. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mollà-Casanova, S.; Muñoz-Gómez, E.; Sempere-Rubio, N.; Inglés, M.; Aguilar-Rodríguez, M.; Page, A.; López-Pascual, J.; Serra-Añó, P. Effect of virtual running with exercise on functionality in pre-frail and frail elderly people: Randomized clinical trial. Aging Clin. Exp. Res. 2023, 35, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.C.; Lee, H.M. EEG-based evidence of mirror neuron activity from app-mediated stroke patient observation. Medicina 2021, 57, 979. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, D.; Sterba, A.; Khatter, H.; Pandian, J. Mirror therapy in stroke rehabilitation: Current perspectives. Ther. Clin. Risk Manag. 2020, 16, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Maggio, M.G.; Torrisi, M.; Buda, A.; De Luca, R.; Piazzitta, D.; Cannavò, A.; Leo, A.; Milardi, D.; Manuli, A.; Calabrò, R.S.; et al. Effects of robotic neurorehabilitation through Lokomat plus virtual reality on cognitive function in patients with traumatic brain injury: A retrospective case-control study. Int. J. Neurosci. 2020, 130, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Abbruzzese, G.; Marchese, R.; Avanzino, L.; Pelosin, E. Rehabilitation for Parkinson’s disease: Current outlook and future challenges. Park. Relat. Disord. 2016, 22, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Caligiore, D.; Mustile, M.; Spalletta, G.; Baldassarre, G. Action observation and motor imagery for rehabilitation in Parkinson’s disease: A systematic review and an integrative hypothesis. Neurosci. Biobehav. Rev. 2017, 72, 210–222. [Google Scholar] [CrossRef] [PubMed]
| Type of Exercise | Benefits | Example of Exercise |
|---|---|---|
| Active range of motion exercises | ↓ local inflammation ↑ local circulation ↓ swelling ↑ muscle pump antithrombotic effect ↑ joints mobility improvement of plantar load ↓ callus formation ↑ flexibility | -plantar/dorsal flexion -pronation/inversion of the foot -foot circles -alternating flexion/extension of the knee joint -alternating flexion/extension of the hip joint |
| Stretching | ↑ local circulation ↓ swelling ↑ flexibility | -attempt to touch the toes of an extended leg with your hand (Hamstring Stretch) -attempt to touch the toes of an extended leg with your hand while sitting with legs spread apart (Inner Thigh Stretch) -briefly sitting on the heels (Shin Stretch) |
| Aerobic | improve respiratory endurance improve circulatory endurance ↓ glucose ↑ energy expenditure | -exercises in standing or walking position after fitting an orthotic if not contraindicated -non-weight bearing combined arm + leg cycling -non-weight bearing arm ergometer |
| Strength | ↓ risk of osteoporosis ↑ energy expenditure ↑ muscle tone ↑ muscle strength ↓ glucose (in the long-term context) ↑ blood flow ↑ flexibility improvement of coordination | -weight bearing combined arm and leg (if not contraindicated) cycling -upper and lower limbs exercises with weights -stretching rubber bands (exercise for the upper and lower extremity) |
| Isometric | ↑ muscle strength ↑ blood flow ↓ swelling antithrombotic effect | -alternating tensing and relaxing the muscle of the calf, thigh, buttock |
| Other specific exercise | ↑ muscle strength | -calisthenics (using natural body movements) |
| ↑ energy expenditure | -calisthenics, Buerger exercises | |
| ↑ local circulation | -Buerger exercises | |
| ↑ blood flow improve respiratory endurance | -Respiratory exercises: -diaphragmatic breathing -pursed-lip breathing -resisted breathing |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sutkowska, E.; Korzon-Burakowska, A.; Biernat, K. On the Merits of Targeted and Individualized Physical Exercise in Persons with Diabetic Foot Disease—From Controversies to Consensus. Biomedicines 2025, 13, 1752. https://doi.org/10.3390/biomedicines13071752
Sutkowska E, Korzon-Burakowska A, Biernat K. On the Merits of Targeted and Individualized Physical Exercise in Persons with Diabetic Foot Disease—From Controversies to Consensus. Biomedicines. 2025; 13(7):1752. https://doi.org/10.3390/biomedicines13071752
Chicago/Turabian StyleSutkowska, Edyta, Anna Korzon-Burakowska, and Karolina Biernat. 2025. "On the Merits of Targeted and Individualized Physical Exercise in Persons with Diabetic Foot Disease—From Controversies to Consensus" Biomedicines 13, no. 7: 1752. https://doi.org/10.3390/biomedicines13071752
APA StyleSutkowska, E., Korzon-Burakowska, A., & Biernat, K. (2025). On the Merits of Targeted and Individualized Physical Exercise in Persons with Diabetic Foot Disease—From Controversies to Consensus. Biomedicines, 13(7), 1752. https://doi.org/10.3390/biomedicines13071752






