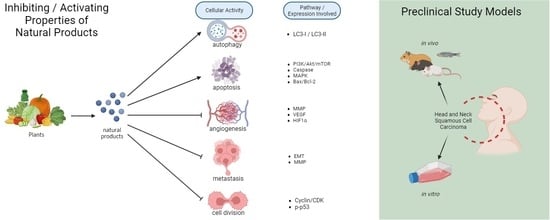
A Comprehensive Review of Natural Products as Therapeutic or Chemopreventive Agents against Head and Neck Squamous Cell Carcinoma Cells Using Preclinical Models
Abstract
:1. Introduction
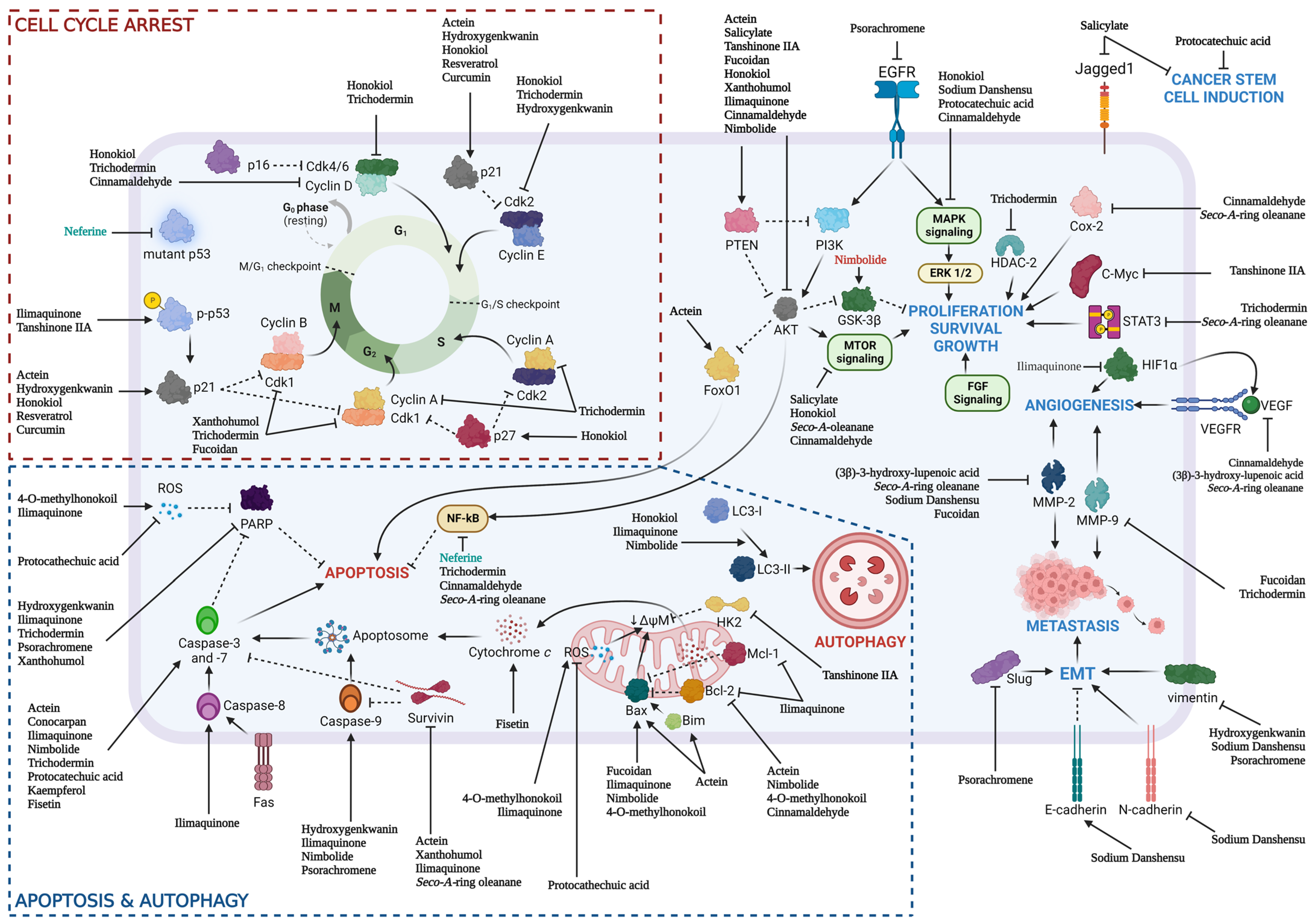
2. Chemotherapeutic Properties of Natural Products on Essential Pathways for HNSCC
2.1. PI3K/Akt/mTOR Pathway
2.2. MAPK/ERK Pathway
2.3. NF-κB and STAT3 Transcription Factors
2.4. LC3-Dependent Autophagy
2.5. Bcl-2/Bax Signaling
2.6. Cell Cycle Arrest by Cyclin and the CDK Signaling Pathway
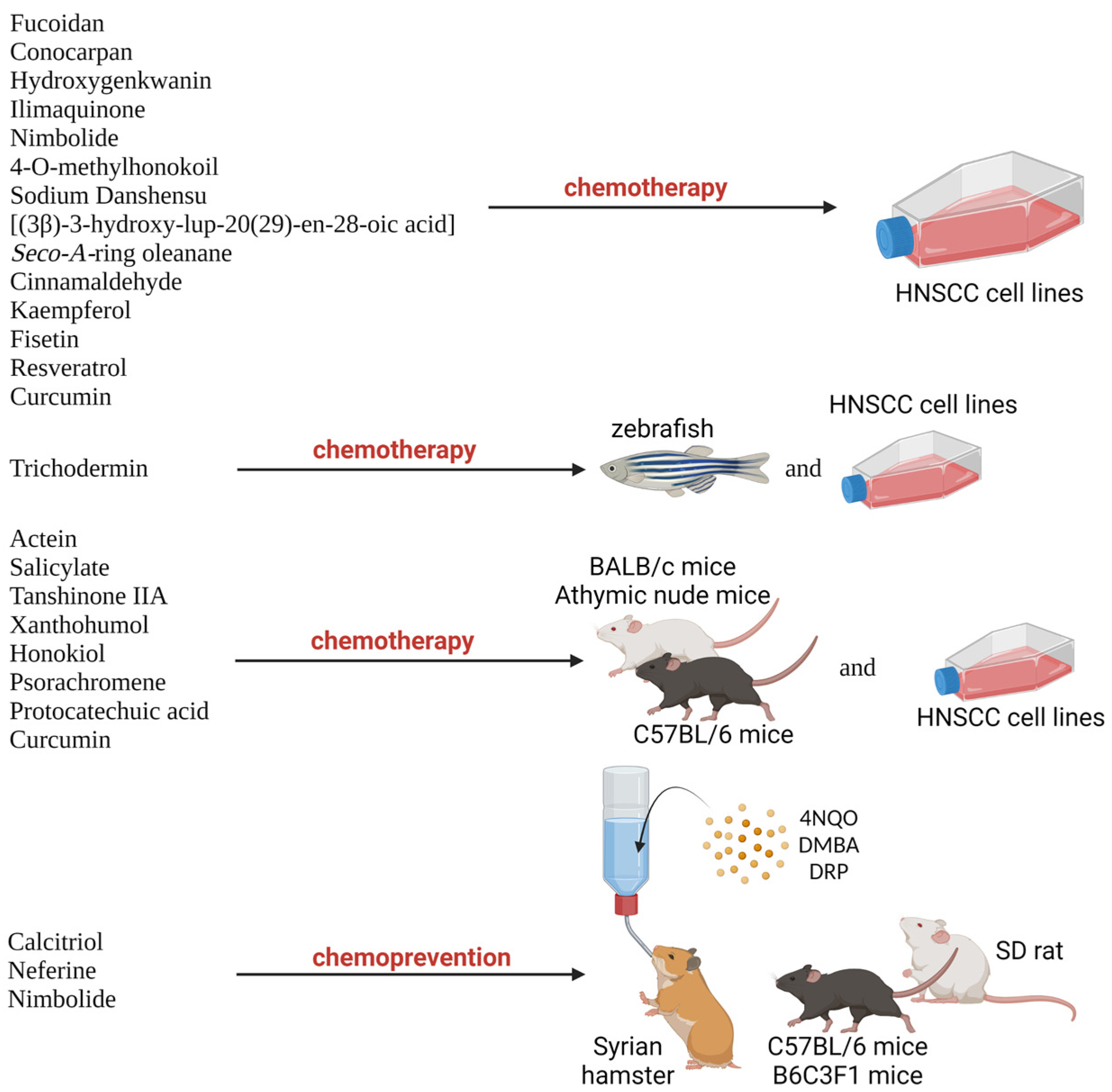
2.7. Potential Natural Products as Therapeutic Agent for HNSCC
3. Chemoprevention Properties of Natural Products against HNSCC Oral Carcinogenesis Mechanism
4. Limitation and Future Direction
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Johnson, D.E.; Burtness, B.; Leemans, C.R.; Lui, V.W.Y.; Bauman, J.E.; Grandis, J.R. Head and Neck Squamous Cell Carcinoma. Nat. Rev. Dis. Prim. 2020, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Magnes, T.; Wagner, S.M.; Melchardt, T.; Weiss, L.; Rinnerthaler, G.; Huemer, F.; Kopp, M.; Gampenrieder, S.P.; Mayrbäurl, B.; Füreder, T.; et al. Postoperative Chemoradiotherapy with Cisplatin Is Superior to Radioimmunotherapy with Cetuximab and Radiotherapy Alone: Analysis of the Austrian Head and Neck Cancer Registry of the AGMT. Wien. Klin. Wochenschr. 2021, 133, 1131–1136. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Bonner, J.A.; Harari, P.M.; Giralt, J.; Cohen, R.B.; Jones, C.U.; Sur, R.K.; Raben, D.; Baselga, J.; Spencer, S.A.; Zhu, J.; et al. Radiotherapy plus Cetuximab for Locoregionally Advanced Head and Neck Cancer: 5-Year Survival Data from a Phase 3 Randomised Trial, and Relation between Cetuximab-Induced Rash and Survival. Lancet Oncol. 2010, 11, 21–28. [Google Scholar] [CrossRef]
- Ferris, R.L.; Blumenschein, G.; Fayette, J.; Guigay, J.; Colevas, A.D.; Licitra, L.; Harrington, K.; Kasper, S.; Vokes, E.E.; Even, C.; et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N. Engl. J. Med. 2016, 375, 1856–1867. [Google Scholar] [CrossRef]
- Talamini, R.; Bosetti, C.; La Vecchia, C.; Dal Maso, L.; Levi, F.; Bidoli, E.; Negri, E.; Pasche, C.; Vaccarella, S.; Barzan, L.; et al. Combined Effect of Tobacco and Alcohol on Laryngeal Cancer Risk: A Case-Control Study. Cancer Causes Control 2002, 13, 957–964. [Google Scholar] [CrossRef]
- Lewin, F.; Norell, S.E.; Johansson, H.; Gustavsson, P.; Wennerberg, J.; Biörklund, A.; Rutqvist, L.E. Smoking Tobacco, Oral Snuff, and Alcohol in the Etiology of Squamous Cell Carcinoma of the Head and Neck A Population-Based Case-Referent Study in Sweden. Cancer 1998, 82, 1367–1375. [Google Scholar] [CrossRef]
- Hansson, B.G.; Rosenquist, K.; Antonsson, A.; Wennerberg, J.; Schildt, E.B.; Bladström, A.; Andersson, G. Strong Association between Infection with Human Papillomavirus and Oral and Oropharyngeal Squamous Cell Carcinoma: A Population-Based Case-Control Study in Southern Sweden. Acta Otolaryngol. 2005, 125, 1337–1344. [Google Scholar] [CrossRef]
- Van Wyk, C.W.; Stander, I.; Padayachee, A.; Grobler-Rabie, A.F. The Areca Nut Chewing Habit and Oral Squamous Cell Carcinoma in South African Indians. A Retrospective Study. S. Afr. Med. J. 1993, 83, 425–429. [Google Scholar]
- Haddad, R.I.; Shin, D.M. Recent Advances in Head and Neck Cancer Reconstruction. N. Engl. J. Med. 2008, 359, 1143–1154. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, N.; Yadav, J.; Chhakara, S.; Janjua, D.; Tripathi, T.; Chaudhary, A.; Chhokar, A.; Thakur, K.; Singh, T.; Bharti, A.C. Phytochemicals as Potential Chemopreventive and Chemotherapeutic Agents for Emerging Human Papillomavirus–Driven Head and Neck Cancer: Current Evidence and Future Prospects. Front. Pharmacol. 2021, 12, 699044. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.R.; Hershock, D.M.; Blajman, C.R.; Mickiewicz, E.; Winquist, E.; Gorbounova, V.; Tjulandin, S.; Shin, D.M.; Cullen, K.; Ervin, T.J.; et al. Cisplatin and Fluorouracil Alone or with Docetaxel in Head and Neck Cancer Marshall. N. Engl. J. Med. 2007, 357, 1705–1715. [Google Scholar] [CrossRef] [PubMed]
- Hashim, D.; Genden, E.; Posner, M.; Hashibe, M.; Boffetta, P. Head and Neck Cancer Prevention: From Primary Prevention to Impact of Clinicians on Reducing Burden. Ann. Oncol. 2019, 30, 744–756. [Google Scholar] [CrossRef]
- Brockstein, B.; Haraf, D.J.; Rademaker, A.W.; Kies, M.S.; Stenson, K.M.; Rosen, F.; Mittal, B.B.; Pelzer, H.; Fung, B.B.; Witt, M.E.; et al. Patterns of Failure, Prognostic Factors and Survival in Locoregionally Advanced Head and Neck Cancer Treated with Concomitant Chemoradiotherapy: A 9-Year, 337-Patient, Multi-Institutional Experience. Ann. Oncol. 2004, 15, 1179–1186. [Google Scholar] [CrossRef]
- Bonner, J.A.; Harari, P.M.; Giralt, J.; Azarnia, N.; Shin, D.M.; Cohen, R.B.; Jones, C.U.; Sur, R.; Raben, D.; Jassem, J.; et al. Results of Radiotherapy plus Cetuximab for Squamous Cell Carcinoma of the Head and Neck. N. Engl. J. Med. 2006, 354, 567–578. [Google Scholar] [CrossRef]
- Naruse, T.; Yanamoto, S.; Matsushita, Y.; Sakamoto, Y.; Morishita, K.; Ohba, S.; Shiraishi, T.; Yamada, S.-I.; Asahina, I.; Umeda, M. Cetuximab for the Treatment of Locally Advanced and Recurrent/Metastatic Oral Cancer: An Investigation of Distant Metastasis. Mol. Clin. Oncol. 2016, 5, 246–252. [Google Scholar] [CrossRef]
- Sok, J.C.; Coppelli, F.M.; Thomas, S.M.; Lango, M.N.; Xi, S.; Hunt, J.L.; Freilino, M.L.; Graner, M.W.; Wikstrand, C.J.; Bigner, D.D.; et al. Mutant Epidermal Growth Factor Receptor (EGFRvIII) Contributes to Head and Neck Cancer Growth and Resistance to EGFR Targeting. Clin. Cancer Res. 2006, 12, 5064–5073. [Google Scholar] [CrossRef]
- US Food and Drug Administration. FDA Approves Pembrolizumab for First-Line Treatment of Head and Neck Squamous Cell Carcinoma. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-pembrolizumab-first-line-treatment-head-and-neck-squamous-cell-carcinoma (accessed on 1 February 2023).
- US Food and Drug Administration. Nivolumab for SCCHN. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/nivolumab-scchn (accessed on 1 February 2023).
- dos Santos, L.V.; Abrahão, C.M.; William, W.N. Overcoming Resistance to Immune Checkpoint Inhibitors in Head and Neck Squamous Cell Carcinomas. Front. Oncol. 2021, 11, 596290. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- National Cancer Institute. Dictionary of Cancer Terms. Available online: https://www.cancer.gov/publications/dictionaries/cancer-terms (accessed on 26 September 2022).
- Wang, T.H.; Leu, Y.L.; Chen, C.C.; Shieh, T.M.; Lian, J.H.; Chen, C.Y. Psorachromene Suppresses Oral Squamous Cell Carcinoma Progression by Inhibiting Long Non-Coding RNA GAS5 Mediated Epithelial-Mesenchymal Transition. Front. Oncol. 2019, 9, 1168. [Google Scholar] [CrossRef]
- Vincent-Chong, V.K.; DeJong, H.; Attwood, K.; Hershberger, P.A.; Seshadri, M. Preclinical Prevention Trial of Calcitriol: Impact of Stage of Intervention and Duration of Treatment on Oral Carcinogenesis. Neoplasia 2019, 21, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Hashem, S.; Ali, T.A.; Akhtar, S.; Nisar, S.; Sageena, G.; Ali, S.; Al-Mannai, S.; Therachiyil, L.; Mir, R.; Elfaki, I.; et al. Targeting Cancer Signaling Pathways by Natural Products: Exploring Promising Anti-Cancer Agents. Biomed. Pharmacother. 2022, 150, 113054. [Google Scholar] [CrossRef]
- Sever, R.; Brugge, J.S. Signal Transduction in Cancer. Cold Spring Harb. Perspect. Med. 2015, 5, a006098. [Google Scholar] [CrossRef]
- Sanchez-Vega, F.; Mina, M.; Armenia, J.; Chatila, W.K.; Luna, A.; La, K.C.; Dimitriadoy, S.; Liu, D.L.; Kantheti, H.S.; Saghafinia, S.; et al. Oncogenic Signaling Pathways in The Cancer Genome Atlas. Cell 2019, 173, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Crooker, K.; Aliani, R.; Ananth, M.; Arnold, L.; Anant, S.; Thomas, S.M. A Review of Promising Natural Chemopreventive Agents for Head and Neck Cancer. Cancer Prev. Res. 2018, 11, 441–450. [Google Scholar] [CrossRef]
- Rahman, M.A.; Amin, A.R.M.R.; Shin, D.M. Chemopreventive Potential of Natural Compounds in Head and Neck Cancer. Nutr. Cancer 2010, 62, 973–987. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.M.; Khuri, F.R.; Murphy, B.; Garden, A.S.; Clayman, G.; Francisco, M.; Liu, D.; Glisson, B.S.; Ginsberg, L.; Papadimitrakopoulou, V.; et al. Combined Interferon-Alfa, 13-Cis-Retinoic Acid, and Alpha-Tocopherol in Locally Advanced Head and Neck Squamous Cell Carcinoma: Novel Bioadjuvant Phase II Trial. J. Clin. Oncol. 2001, 19, 3010–3017. [Google Scholar] [CrossRef]
- Papadimitrakopoulou, V.A.; Lee, J.J.; William, W.N.; Martin, J.W.; Thomas, M.; Kim, E.S.; Khuri, F.R.; Shin, D.M.; Feng, L.; Waun, K.H.; et al. Randomized Trial of 13-Cis Retinoic Acid Compared with Retinyl Palmitate with or without Beta-Carotene in Oral Premalignancy. J. Clin. Oncol. 2009, 27, 599–604. [Google Scholar] [CrossRef]
- Tsao, A.S.; Liu, D.; Martin, J.; Tang, X.M.; Lee, J.J.; El-Naggar, A.K.; Wistuba, I.; Culotta, K.S.; Mao, L.; Gillenwater, A.; et al. Phase II Randomized, Placebo-Controlled Trial of Green Tea Extract in Patients with High-Risk Oral Premalignant Lesions. Cancer Prev. Res. 2009, 2, 931–941. [Google Scholar] [CrossRef]
- Cheng, A.L.; Hsu, C.H.; Lin, J.K.; Hse, M.W.; Ho, Y.F.; Shen, T.S.; Ko, J.Y.; Lin, J.T.; Lin, B.R.; Wu, M.S.; et al. Phase I Clinical Trial of Curcumin, a Chemopreventive Agent, in Patients with High-Risk or Pre-Malignant Lesions. Anticancer Res. 2001, 21, 2895–2900. [Google Scholar] [PubMed]
- Molinolo, A.A.; Hewitt, S.M.; Amornphimoltham, P.; Keelawat, S.; Rangdaeng, S.; García, A.M.; Raimondi, A.R.; Jufe, R.; Itoiz, M.; Gao, Y.; et al. Dissecting the Akt/Mammalian Target of Rapamycin Signaling Network: Emerging Results from the Head and Neck Cancer Tissue Array Initiative. Clin. Cancer Res. 2007, 13, 4964–4973. [Google Scholar] [CrossRef] [PubMed]
- Freudlsperger, C.; Horn, D.; Weißfuß, S.; Weichert, W.; Weber, K.J.; Saure, D.; Sharma, S.; Dyckhoff, G.; Grabe, N.; Plinkert, P.; et al. Phosphorylation of AKT(Ser473) Serves as an Independent Prognostic Marker for Radiosensitivity in Advanced Head and Neck Squamous Cell Carcinoma. Int. J. Cancer 2015, 136, 2775–2785. [Google Scholar] [CrossRef] [PubMed]
- Zumsteg, Z.S.; Morse, N.; Krigsfeld, G.; Gupta, G.; Higginson, D.S.; Lee, N.Y.; Morris, L.; Ganly, I.; Shiao, S.L.; Powell, S.N.; et al. Taselisib (GDC-0032), a Potent β-Sparing Small Molecule Inhibitor of PI3K, Radiosensitizes Head and Neck Squamous Carcinomas Containing Activating PIK3CA Alterations. Clin. Cancer Res. 2016, 22, 2009–2019. [Google Scholar] [CrossRef] [PubMed]
- Iwase, M.; Yoshiba, S.; Uchid, M.; Takaoka, S.; Kurihara, Y.; Ito, D.; Hatori, M.; Shintani, S. Enhanced Susceptibility to Apoptosis of Oral Squamous Cell Carcinoma Cells Subjected to Combined Treatment with Anticancer Drugs and Phosphatidylinositol 3-Kinase Inhibitors. Int. J. Oncol. 2007, 31, 1141–1147. [Google Scholar] [PubMed]
- Cantley, L.C. The Phosphoinositide 3-Kinase Pathway. Science 2002, 296, 1655–1657. [Google Scholar] [CrossRef]
- Sarbassov, D.D.; Guertin, D.A.; Ali, S.M.; Sabatini, D.M. Phosphorylation and Regulation of Akt/PKB by the Rictor-MTOR Complex. Science 2005, 307, 1098–1101. [Google Scholar] [CrossRef]
- Zhao, C.; Zhang, Z.; Dai, X.; Wang, J.; Liu, H.; Ma, H. Actein Antagonizes Oral Squamous Cell Carcinoma Proliferation through Activating FoxO1. Pharmacology 2021, 106, 551–563. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, F.; Zeng, Y.; Zhu, X.; Peng, L.; Zhang, L.; Gu, J.; Han, H.; Yi, X.; Shi, J. Salicylate Sensitizes Oral Squamous Cell Carcinoma to Chemotherapy through Targeting MTOR Pathway. Oral Dis. 2020, 26, 1131–1140. [Google Scholar] [CrossRef]
- Li, M.; Gao, F.; Zhao, Q.; Zuo, H.; Liu, W.; Li, W. Tanshinone IIA Inhibits Oral Squamous Cell Carcinoma via Reducing Akt-c-Myc Signaling-Mediated Aerobic Glycolysis. Cell Death Dis. 2020, 11, 381. [Google Scholar] [CrossRef]
- Li, M.; Gao, F.; Yu, X.; Zhao, Q.; Zhou, L.; Liu, W.; Li, W. Promotion of Ubiquitination-Dependent Survivin Destruction Contributes to Xanthohumol-Mediated Tumor Suppression and Overcomes Radioresistance in Human Oral Squamous Cell Carcinoma. J. Exp. Clin. Cancer Res. 2020, 39, 88. [Google Scholar] [CrossRef]
- Huang, K.J.; Kuo, C.H.; Chen, S.H.; Lin, C.Y.; Lee, Y.R. Honokiol Inhibits in Vitro and in Vivo Growth of Oral Squamous Cell Carcinoma through Induction of Apoptosis, Cell Cycle Arrest and Autophagy. J. Cell. Mol. Med. 2018, 22, 1894–1908. [Google Scholar] [CrossRef]
- Lin, C.W.; Bai, L.Y.; Su, J.H.; Chiu, C.F.; Lin, W.Y.; Huang, W.T.; Shih, M.C.; Huang, Y.T.; Hu, J.L.; Weng, J.R. Ilimaquinone Induces Apoptosis and Autophagy in Human Oral Squamous Cell Carcinoma Cells. Biomedicines 2020, 8, 296. [Google Scholar] [CrossRef]
- Sophia, J.; Kowshik, J.; Dwivedi, A.; Bhutia, S.K.; Manavathi, B.; Mishra, R.; Nagini, S. Nimbolide, a Neem Limonoid Inhibits Cytoprotective Autophagy to Activate Apoptosis via Modulation of the PI3K/Akt/GSK-3β Signalling Pathway in Oral Cancer. Cell Death Dis. 2018, 9, 1087. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Bhadana, K.; Singh, B.; Rawat, M.; Mohammad, T.; Al-Keridis, L.A.; Alshammari, N.; Hassan, M.I.; Das, S.N. Cinnamomum Zeylanicum Extract and Its Bioactive Component Cinnamaldehyde Show Anti-Tumor Effects via Inhibition of Multiple Cellular Pathways. Front. Pharmacol. 2022, 13, 918479. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Gao, L.; Ren, W.; Li, S.; Zhang, D.; Song, X.; Zhao, C.; Zhi, K. Fucoidan Affects Oral Squamous Cell Carcinoma Cell Functions in Vitro by Regulating FLNA-Derived Circular RNA. Ann. N. Y. Acad. Sci. 2020, 1462, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Aswathy, M.; Banik, K.; Parama, D.; Sasikumar, P.; Harsha, C.; Joseph, A.G.; Sherin, D.R.; Thanathu, M.K.; Kunnumakkara, A.B.; Vasu, R.K. Exploring the Cytotoxic Effects of the Extracts and Bioactive Triterpenoids from Dillenia Indica against Oral Squamous Cell Carcinoma: A Scientific Interpretation and Validation of Indigenous Knowledge. ACS Pharmacol. Transl. Sci. 2021, 4, 834–847. [Google Scholar] [CrossRef]
- Schulz, L.; Pries, R.; Lanka, A.S.; Drenckhan, M.; Rades, D.; Wollenberg, B. Inhibition of GSK3α/β Impairs the Progression of HNSCC. Oncotarget 2018, 9, 27630–27644. [Google Scholar] [CrossRef]
- Ugolkov, A.V.; Matsangou, M.; Taxter, T.J.; O’halloran, T.V.; Cryns, V.L.; Giles, F.J.; Mazar, A.P. Aberrant Expression of Glycogen Synthase Kinase-3β in Human Breast and Head and Neck Cancer. Oncol. Lett. 2018, 16, 6437–6444. [Google Scholar] [CrossRef]
- Marconi, G.D.; Della Rocca, Y.; Fonticoli, L.; Melfi, F.; Rajan, T.S.; Carradori, S.; Pizzicannella, J.; Trubiani, O.; Diomede, F. C-Myc Expression in Oral Squamous Cell Carcinoma: Molecular Mechanisms in Cell Survival and Cancer Progression. Pharmaceuticals 2022, 15, 890. [Google Scholar] [CrossRef]
- Bancroft, C.C.; Chen, Z.; Dong, G.; Sunwoo, J.B.; Yeh, N.; Park, C.; Van Waes, C. Coexpression of Proangiogenic Factors IL-8 and VEGF by Human Head and Neck Squamous Cell Carcinoma Involves Coactivation by MEK-MAPK and IKK-NF-ΚB Signal Pathways. Clin. Cancer Res. 2001, 7, 435–442. [Google Scholar] [PubMed]
- Kumar, V.B.; Lin, S.H.; Mahalakshmi, B.; Lo, Y.S.; Lin, C.C.; Chuang, Y.C.; Hsieh, M.J.; Chen, M.K. Sodium Danshensu Inhibits Oral Cancer Cell Migration and Invasion by Modulating P38 Signaling Pathway. Front. Endocrinol. 2020, 11, 568436. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Cao, L.; Yang, C.; Liu, T.; Zhao, H.; Luo, X.; Chen, Q. Protocatechuic Acid-Based Supramolecular Hydrogel Targets SerpinB9 to Achieve Local Chemotherapy for OSCC. ACS Appl. Mater. Interfaces 2022, 14, 32. [Google Scholar] [CrossRef] [PubMed]
- Gkouveris, I.; Nikitakis, N.; Karanikou, M.; Rassidakis, G.; Sklavounou, A. JNK1/2 Expression and Modulation of STAT3 Signaling in Oral Cancer. Oncol. Lett. 2016, 12, 699–706. [Google Scholar] [CrossRef]
- charoenrat, P.; Rhys-Evans, P.H.; Eccles, S.A. Expression of Matrix Metalloproteinases and Their Inhibitors Correlates with Invasion and Metastasis in Squamous Cell Carcinoma of the Head and Neck. Arch. Otolaryngol. Head Neck Surg. 2001, 127, 813–820. [Google Scholar]
- Carmeliet, P. VEGF as a Key Mediator of Angiogenesis in Cancer. Oncology 2005, 69, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Pepper, M.S.; Ferrara, N.; Orci, L.; Montesano, R. Potent Synergism between Vascular Endothelial Growth Factor and Basic Fibroblast Growth Factor in the Induction of Angiogenesis in Vitro. Biochem. Biophys. Res. Commun. 1992, 189, 824–831. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Wei, W.; Xu, H.; Wang, Y.Y.; Wu, W.X. Inducing Effects of Hepatocyte Growth Factor on the Expression of Vascular Endothelial Growth Factor in Human Colorectal Carcinoma Cells through MEK and PI3K Signaling Pathways. Chin. Med. J. 2007, 120, 743–748. [Google Scholar] [CrossRef]
- Lun, M.; Zhang, P.L.; Pellitteri, P.K.; Law, A.; Kennedy, T.L.; Brown, R.E. Nuclear Factor-KappaB Pathway as a Therapeutic Target in Head and Neck Squamous Cell Carcinoma: Pharmaceutical and Molecular Validation in Human Cell Lines Using Velcade and SiRNA/NF-ΚB. Ann. Clin. Lab. Sci. 2005, 35, 251–258. [Google Scholar]
- Oeckinghaus, A.; Ghosh, S. The NF-ΚB Family of Transcription Factors and Its Regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Wang, J.; Hu, Y.; Yuan, J.; Zhang, Y.; Wang, Y.; Yang, Y.; Alahmadi, T.A.; Ali Alharbi, S.; Zhuang, Z.; Wu, F. Chemomodulatory Effect of Neferine on DMBA-Induced Squamous Cell Carcinogenesis: Biochemical and Molecular Approach. Environ. Toxicol. 2021, 36, 460–471. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lo, Y.; Lin, C.; Lee, T.; Leung, W.; Wang, S.; Lin, I.; Lin, M.; Lee, C. Biomedicine & Pharmacotherapy Trichodermin Inhibits the Growth of Oral Cancer through Apoptosis-Induced Mitochondrial Dysfunction and HDAC-2-Mediated Signaling. Biomed. Pharmacother. 2022, 153, 113351. [Google Scholar] [CrossRef] [PubMed]
- Ihle, J.N. The Stat Family in Cytokine Signaling. Curr. Opin. Cell Biol. 2001, 13, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.S.; Zhou, X.; Huang, Y.Y.; Kong, L.P.; Mei, M.; Guo, W.Y.; Zhao, M.H.; Ren, Y.; Shen, Q.; Zhang, L. Targeting STAT3/MiR-21 Axis Inhibits Epithelial-Mesenchymal Transition via Regulating CDK5 in Head and Neck Squamous Cell Carcinoma. Mol. Cancer 2015, 14, 213. [Google Scholar] [CrossRef]
- Zhou, X.; Ren, Y.; Liu, A.; Han, L.; Zhang, K.; Li, S.; Li, P.; Li, P.; Kang, C.; Wang, X.; et al. STAT3 Inhibitor WP1066 Attenuates MiRNA-21 to Suppress Human Oral Squamous Cell Carcinoma Growth in Vitro and in Vivo. Oncol. Rep. 2014, 31, 2173–2180. [Google Scholar] [CrossRef]
- Riebe, C.; Pries, R.; Schroeder, K.N.; Wollenberg, B. Phosphorylation of STAT3 in Head and Neck Cancer Requires P38 MAPKinase, Whereas Phosphorylation of STAT1 Occurs via a Different Signaling Pathway. Anticancer Res. 2011, 31, 3819–3825. [Google Scholar]
- Zhao, C.; Yang, L.; Zhou, F.; Yu, Y.; Du, X.; Xiang, Y.; Li, C.; Huang, X.; Xie, C.; Liu, Z.; et al. Feedback Activation of EGFR Is the Main Cause for STAT3 Inhibition-Irresponsiveness in Pancreatic Cancer Cells. Oncogene 2020, 39, 3997–4013. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.; Cui, Y.; Li, X.; Cao, X.; Chen, A.; Xu, C.; Cao, J.; Luo, X. Co-Culture of Ovarian Cancer Stem-like Cells with Macrophages Induced SKOV3 Cells Stemness via IL-8/STAT3 Signaling. Biomed. Pharmacother. 2018, 103, 262–271. [Google Scholar] [CrossRef]
- Yang, J.; Liao, D.; Chen, C.; Liu, Y.; Chuang, T.H.; Xiang, R.; Markowitz, D.; Reisfeld, R.A.; Luo, Y. Tumor-Associated Macrophages Regulate Murine Breast Cancer Stem Cells through a Novel Paracrine Egfr/Stat3/Sox-2 Signaling Pathway. Stem Cells 2013, 31, 248–258. [Google Scholar] [CrossRef]
- Chan, C.Y.; Huang, S.Y.; Sheu, J.J.C.; Roth, M.M.; Chou, I.T.; Lien, C.H.; Lee, M.F.; Huang, C.Y. Transcription Factor HBP1 Is a Direct Anti-Cancer Target of Transcription Factor FOXO1 in Invasive Oral Cancer. Oncotarget 2017, 8, 14537–14548. [Google Scholar] [CrossRef]
- Roh, J.L.; Kang, S.K.; Minn, I.; Califano, J.A.; Sidransky, D.; Koch, W.M. P53-Reactivating Small Molecules Induce Apoptosis and Enhance Chemotherapeutic Cytotoxicity in Head and Neck Squamous Cell Carcinoma. Oral Oncol. 2011, 47, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.; Tiwari, R.P.; Mulherker, R.; Sah, N.K.; Prasad, G.B.; Shrivastava, B.R.; Bisen, P.S. Detection of Survivin and P53 in Human Oral Cancer: Correlation with Clinicopathological Findings. Head Neck 2009, 31, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Network. Comprehensive Genomic Characterization of Head and Neck Squamous Cell Carcinomas. Nature 2015, 517, 576–582. [Google Scholar] [CrossRef]
- Tanida, I.; Ueno, T.; Kominami, E. LC3 and Autophagy. Methods Mol. Biol. 2008, 445, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Kabeya, Y.; Mizushima, N.; Ueno, T.; Yamamoto, A.; Kirisako, T.; Noda, T.; Kominami, E.; Ohsumi, Y.; Yoshimori, T. LC3, a Mammalian Homologue of Yeast Apg8p, Is Localized in Autophagosome Membranes after Processing. EMBO J. 2000, 19, 5720–5728. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.W.; Lee, S.H. The Roles of Autophagy in Cancer. Int. J. Mol. Sci. 2018, 19, 3466. [Google Scholar] [CrossRef]
- Khan, T.; Relitti, N.; Brindisi, M.; Magnano, S.; Zisterer, D.; Gemma, S.; Butini, S.; Campiani, G. Autophagy Modulators for the Treatment of Oral and Esophageal Squamous Cell Carcinomas. Med. Res. Rev. 2020, 40, 1002–1060. [Google Scholar] [CrossRef]
- Lee, M.; Nam, H.Y.; Kang, H.B.; Lee, W.H.; Lee, G.H.; Sung, G.J.; Han, M.W.; Cho, K.J.; Chang, E.J.; Choi, K.C.; et al. Epigenetic Regulation of P62/SQSTM1 Overcomes the Radioresistance of Head and Neck Cancer Cells via Autophagy-Dependent Senescence Induction. Cell Death Dis. 2021, 12, 250. [Google Scholar] [CrossRef]
- Eisenberg-Lerner, A.; Kimchi, A. The Paradox of Autophagy and Its Implication in Cancer Etiology and Therapy. Apoptosis 2009, 14, 376–391. [Google Scholar] [CrossRef]
- Garcia-Medina, R.; Gounon, P.; Chiche, J.; Pouysse, J.; Mazure, N.M. Hypoxia-Induced Autophagy Is Mediated through Hypoxia-Inducible Factor Induction of BNIP3 and BNIP3L via Their BH3 Domains. Mol. Cell. Biol. 2009, 29, 2570–2581. [Google Scholar] [CrossRef]
- Wei, H.; Wei, S.; Gan, B.; Peng, X.; Zou, W.; Guan, J. Suppression of Autophagy by FIP200 Deletion Inhibits Mammary Tumorigenesis. Genes Dev. 2011, 37, 1510–1527. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.S.; Vats, S.; Chia, A.Y.; Zea, T.; Shuo, T.; Mei, D.; Ong, S.; Arfuso, F.; Yap, C.T.; Cher, B.; et al. Dual Role of Autophagy in Hallmarks of Cancer. Oncogene 2018, 37, 1142–1158. [Google Scholar] [CrossRef] [PubMed]
- Muz, B.; Azab, A.K. The Role of Hypoxia in Cancer Progression, Angiogenesis, Metastasis, and Resistance to Therapy. Hypoxia 2015, 3, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Oltval, Z.N.; Milliman, C.L.; Korsmeyer, S.J. Bcl-2 Heterodimerizes in Vivo with a Conserved Homolog, Bax, That Accelerates Programed Cell Death. Cell 1993, 74, 609–619. [Google Scholar] [CrossRef]
- Hockenbery, D.; Nuñez, G.; Milliman, C.; Schreiber, R.D.; Korsmeyer, S.J. Bcl-2 Is an Inner Mitochondrial Membrane Protein That Blocks Programmed Cell Death. Nature 1990, 348, 334–336. [Google Scholar] [CrossRef]
- Loro, L.L.; Vintermyr, O.K.; Liavaag, P.G.; Jonsson, R.; Johannessen, A.C. Oral Squamous Cell Carcinoma Is Associated with Decreased Bcl-2/Bax Expression Ratio and Increased Apoptosis. Hum. Pathol. 1999, 30, 1097–1105. [Google Scholar] [CrossRef]
- Zou, H.; Henzel, W.J.; Liu, X. Apaf-1, a Human Protein Homologous to C. Elegans CED-4, Participates in cytochrome c-dependent activation of caspase-3. Cell 1997, 90, 405–413. [Google Scholar] [CrossRef]
- Kubina, R.; Krzykawski, K.; Dziedzic, A. Kaempferol and Fisetin-Related Signaling Pathways Induce Apoptosis in Head and Neck Cancer Cells. Cells 2023, 12, 1568. [Google Scholar] [CrossRef]
- Xiao, S.; Chen, F.; Gao, C. Antitumor Activity of 4-O-Methylhonokiol in Human Oral Cancer Cells Is Mediated via ROS Generation, Disruption of Mitochondrial Potential, Cell Cycle Arrest and Modulation of Bcl-2/Bax Proteins. J. BUON 2017, 22, 1577–1581. [Google Scholar]
- Liu, J.; Uematsu, H.; Tsuchida, N.; Ikeda, M.A. Essential Role of Caspase-8 in P53/P73-Dependent Apoptosis Induced by Etoposide in Head and Neck Carcinoma Cells. Mol. Cancer 2011, 10, 95. [Google Scholar] [CrossRef]
- Liu, J.; Uematsu, H.; Tsuchida, N.; Ikeda, M.A. Association of Caspase-8 Mutation with Chemoresistance to Cisplatin in HOC313 Head and Neck Squamous Cell Carcinoma Cells. Biochem. Biophys. Res. Commun. 2009, 390, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Coleman, S.C.; Stewart, Z.A.; Day, T.A.; Netterville, J.L.; Burkey, B.B.; Pietenpol, J.A. Analysis of Cell-Cycle Checkpoint Pathways in Head and Neck Cancer Cell Lines: Implications for Therapeutic Strategies. Arch. Otolaryngol.-Head Neck Surg. 2002, 128, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Norbury, C.; Nurse, P. Animal Cell Cycles and Their Control. Annu. Rev. Biochem. 1992, 61, 441–470. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Connolly, T.; Futcher, B.; Beach, D. Human D-Type Cyclin. Cell 1991, 65, 691–699. [Google Scholar] [CrossRef]
- Matsushime, H.; Ewen, M.E.; Strom, D.K.; Kato, J.Y.; Hanks, S.K.; Roussel, M.F.; Sherr, C.J. Identification and Properties of an Atypical Catalytic Subunit (P34PSK-J3/Cdk4) for Mammalian D Type G1 Cyclins. Cell 1992, 71, 323–334. [Google Scholar] [CrossRef]
- Lea, N.C.; Orr, S.J.; Stoeber, K.; Williams, G.H.; Lam, E.W.-F.; Ibrahim, M.A.A.; Mufti, G.J.; Thomas, N.S.B. Commitment Point during G0→G1 That Controls Entry into the Cell Cycle. Mol. Cell. Biol. 2003, 23, 2351–2361. [Google Scholar] [CrossRef]
- Duronio, R.J.; Brook, A.; Dyson, N.; O’Farrell, P.H. E2F-Induced S Phase Requires Cyclin E. Genes Dev. 1996, 10, 2505–2513. [Google Scholar] [CrossRef]
- Oakes, V.; Wang, W.; Harrington, B.; Lee, W.J.; Beamish, H.; Chia, K.M.; Pinder, A.; Goto, H.; Inagaki, M.; Pavey, S.; et al. Cyclin A/Cdk2 Regulates Cdh1 and Claspin during Late S/G2 Phase of the Cell Cycle. Cell Cycle 2014, 13, 3302–3311. [Google Scholar] [CrossRef]
- Vigneron, S.; Sundermann, L.; Labbé, J.C.; Pintard, L.; Radulescu, O.; Castro, A.; Lorca, T. Cyclin A-Cdk1-Dependent Phosphorylation of Bora Is the Triggering Factor Promoting Mitotic Entry. Dev. Cell 2018, 45, 637–650.e7. [Google Scholar] [CrossRef]
- Gavet, O.; Pines, J. Activation of Cyclin B1-Cdk1 Synchronizes Events in the Nucleus and the Cytoplasm at Mitosis. J. Cell Biol. 2010, 189, 247–259. [Google Scholar] [CrossRef]
- Li, B.; Zhou, P.; Xu, K.; Chen, T.; Jiao, J.; Wei, H.; Yang, X.; Xu, W.; Wan, W.; Xiao, J. Metformin Induces Cell Cycle Arrest, Apoptosis and Autophagy through ROS/JNK Signaling Pathway in Human Osteosarcoma. Int. J. Biol. Sci. 2020, 16, 74–84. [Google Scholar] [CrossRef]
- McConnell, B.B.; Gregory, F.J.; Stott, F.J.; Hara, E.; Peters, G. Induced Expression of P16 INK4a Inhibits Both CDK4- and CDK2-Associated Kinase Activity by Reassortment of Cyclin-CDK-Inhibitor Complexes. Mol. Cell. Biol. 1999, 19, 1981–1989. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.Q.; Livingston, D.M.; Kaelin, W.G.; Adams, P.D. Deregulated Transcription Factor E2F-1 Expression Leads to S-Phase Entry and P53-Mediated Apoptosis. Proc. Natl. Acad. Sci. USA 1994, 91, 10918–10922. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Lee, P.C.; Wang, J.J.; Hsu, Y.C. Anticancer Effect and Mechanism of Hydroxygenkwanin in Oral Squamous Cell Carcinoma. Front. Oncol. 2019, 9, 911. [Google Scholar] [CrossRef] [PubMed]
- Bostan, M.; Petrică-Matei, G.G.; Radu, N.; Hainarosie, R.; Stefanescu, C.D.; Diaconu, C.C.; Roman, V. The Effect of Resveratrol or Curcumin on Head and Neck Cancer Cells Sensitivity to the Cytotoxic Effects of Cisplatin. Nutrients 2020, 12, 2596. [Google Scholar] [CrossRef]
- Da Fonseca, A.C.C.; de Queiroz, L.N.; Sales Felisberto, J.; Jessé Ramos, Y.; Mesquita Marques, A.; Wermelinger, G.F.; Pontes, B.; de Lima Moreira, D.; Robbs, B.K. Cytotoxic Effect of Pure Compounds from Piper Rivinoides Kunth against Oral Squamous Cell Carcinoma. Nat. Prod. Res. 2021, 35, 6163–6167. [Google Scholar] [CrossRef]
- De Campos, P.S.; Matte, B.F.; Diel, L.F.; Jesus, L.H.; Bernardi, L.; Alves, A.M.; Rados, P.V.; Lamers, M.L. Low Doses of Curcuma Longa Modulates Cell Migration and Cell–Cell Adhesion. Phyther. Res. 2017, 31, 1433–1440. [Google Scholar] [CrossRef]
- Chourasia, N.R.; Borle, R.M.; Vastani, A. Concomitant Association of Oral Submucous Fibrosis and Oral Squamous Cell Carcinoma and Incidence of Malignant Transformation of Oral Submucous Fibrosis in a Population of Central India: A Retrospective Study. J. Maxillofac. Oral Surg. 2015, 14, 902–906. [Google Scholar] [CrossRef]
- Evren, I.; Brouns, E.R.; Wils, L.J.; Poell, J.B.; Peeters, C.F.W.; Brakenhoff, R.H.; Bloemena, E.; de Visscher, J.G.A.M. Annual Malignant Transformation Rate of Oral Leukoplakia Remains Consistent: A Long-Term Follow-up Study. Oral Oncol. 2020, 110, 105014. [Google Scholar] [CrossRef]
- Iocca, O.; Sollecito, T.P.; Alawi, F.; Weinstein, G.S.; Newman, J.G.; De Virgilio, A.; Di Maio, P.; Spriano, G.; Pardiñas López, S.; Shanti, R.M. Potentially Malignant Disorders of the Oral Cavity and Oral Dysplasia: A Systematic Review and Meta-Analysis of Malignant Transformation Rate by Subtype. Head Neck 2020, 42, 539–555. [Google Scholar] [CrossRef]
- Lodi, G.; Franchini, R.; Warnakulasuriya, S.; Varoni, E.M.; Sardella, A.; Kerr, A.R.; Carrassi, A.; MacDonald, L.C.; Worthington, H.V.; Mauleffinch, L.F. Interventions for Treating Oral Leukoplakia to Prevent Oral Cancer (Review). Cochrane Database Syst. Rev. 2016, 7, CD001829. [Google Scholar] [CrossRef] [PubMed]
- Papadimitrakopoulou, V.A.; William, W.N.; Dannenberg, A.J.; Lippman, S.M.; Lee, J.J.; Ondrey, F.G.; Peterson, D.E.; Feng, L.; Atwell, A.; El-Naggar, A.K.; et al. Pilot Randomized Phase II Study of Celecoxib in Oral Premalignant Lesions. Clin. Cancer Res. 2008, 14, 2095–2101. [Google Scholar] [CrossRef] [PubMed]
- William, W.N.; Papadimitrakopoulou, V.; Lee, J.J.; Mao, L.; Cohen, E.E.W.; Lin, H.Y.; Gillenwater, A.M.; Martin, J.W.; Lingen, M.W.; Boyle, J.O.; et al. Erlotinib and the Risk of Oral Cancer the Erlotinib Prevention of Oral Cancer (EPOC) Randomized Clinical Trial. JAMA Oncol. 2016, 2, 209–216. [Google Scholar] [CrossRef]
- Gutkind, J.S.; Molinolo, A.A.; Wu, X.; Wang, Z.; Nachmanson, D.; Harismendy, O.; Alexandrov, L.B.; Wuertz, B.R.; Ondrey, F.G.; Laronde, D.; et al. Inhibition of MTOR Signaling and Clinical Activity of Metformin in Oral Premalignant Lesions. JCI Insight 2021, 6, 147096. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Lyu, Y.; Yang, Y.G.; Hu, Z. Humanized Rodent Models for Cancer Research. Front. Oncol. 2020, 10, 1696. [Google Scholar] [CrossRef]
- Bouaoud, J.; De Souza, G.; Darido, C.; Tortereau, A.; Elkabets, M.; Bertolus, C.; Saintigny, P. The 4-NQO Mouse Model: An Update on a Well-Established in Vivo Model of Oral Carcinogenesis. Methods Cell Biol. 2021, 163, 197–229. [Google Scholar] [CrossRef]
- Schoop, R.A.L.; Noteborn, M.H.M.; Baatenburg De Jong, R.J. A Mouse Model for Oral Squamous Cell Carcinoma. J. Mol. Histol. 2009, 40, 177–181. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S.; Wang, Y.; Wang, L.; Xia, Q.; Tian, Z.; Guan, X. Chemopreventive Effect of Modified Zengshengping on Oral Cancer in a Hamster Model and Assessment of Its Effect on Liver. J. Ethnopharmacol. 2020, 255, 112774. [Google Scholar] [CrossRef]
- Dai, Z.; Zhu, P.F.; Liu, H.; Li, X.C.; Zhu, Y.Y.; Liu, Y.Y.; Shi, X.L.; Chen, W.D.; Liu, Y.P.; Zhao, Y.L.; et al. Discovery of Potent Immune-Modulating Molecule Taccaoside A against Cancers from Structures-Active Relationships of Natural Steroidal Saponins. Phytomedicine 2022, 104, 154335. [Google Scholar] [CrossRef]
- Cattaneo, C.M.; Dijkstra, K.K.; Fanchi, L.F.; Kelderman, S.; Kaing, S.; van Rooij, N.; van den Brink, S.; Schumacher, T.N.; Voest, E.E. Tumor Organoid–T-Cell Coculture Systems. Nat. Protoc. 2020, 15, 15–39. [Google Scholar] [CrossRef]
- Neal, J.T.; Li, X.; Zhu, J.; Giangarra, V.; Grzeskowiak, C.L.; Ju, J.; Liu, I.H.; Chiou, S.H.; Salahudeen, A.A.; Smith, A.R.; et al. Organoid Modeling of the Tumor Immune Microenvironment. Cell 2018, 175, 1972–1988.e16. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Van der Jeught, K.; Fang, Y.; Yu, T.; Li, Y.; Ao, Z.; Liu, S.; Zhang, L.; Yang, Y.; Eyvani, H.; et al. An Organoid-Based Screen for Epigenetic Inhibitors That Stimulate Antigen Presentation and Potentiate T-Cell-Mediated Cytotoxicity. Nat. Biomed. Eng. 2021, 5, 1320–1335. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Vincent-chong, V.K.; Dejong, H.; Hershberger, P.A.; Seshadri, M. Impact of Dietary Vitamin D on Initiation and Progression of Oral Cancer. J. Steroid Biochem. Mol. Biol. 2020, 199, 105603. [Google Scholar] [CrossRef] [PubMed]
| No | Author | PMID | Natural Product | Preclinical Model (Cell Lines) | Test & Dosage | Findings | Pathway Related |
|---|---|---|---|---|---|---|---|
| 1 | Zhang et al. [49] | 31495936 | Fucoidan | In vitro (SCC15, SCC25) |
|
| Akt Bax/Bcl-2 |
| 2 | Fonseca et al. [109] | 33078660 | Conocarpan | In vitro (SCC9, SCC4, SCC25) |
|
| Caspase |
| 3 | Huang et al. [107] | 31620368 | Hydroxygenkwanin | In vitro (SAS & OECM1) |
|
| Caspase |
| 4 | Lin et al. [46] | 32825464 | Ilimaquinone | In vitro (SCC4, SCC2095) |
|
| Akt Caspase Bax/Bcl-2 |
| 5 | Sophia et al. [47] | 30352996 | Nimbolide | In vitro (SCC131, SCC4) |
|
| PI3K/Akt/GSK-3β Caspase Bax/Bcl-2 |
| 6 | Xiao et al. [92] | 29332355 | 4-O-methylhonokoil | In vitro (PE/CA-PJ41) |
|
| Bax/Bcl-2 |
| 7 | Kumar et al. [55] | 33101201 | Sodium Danshensu | In vitro (FaDu, CA9-22) |
|
| MAPK/ERK |
| 8 | Aswathy et al. [50] | 33860206 | [(3β)-3-hydroxy-lup-20(29)-en-28-oic acid] | In vitro (SAS) |
|
| Akt/mTOR JAK/STAT3 VEGF NF-κB |
| Seco-A-ring oleanane | In vitro (SAS) |
|
| ||||
| 9 | Aggarwal et al. [48] | 35774603 | Cinnamaldehyde | In vitro (SCC9, SCC25) |
|
| PI3K/Akt/mTOR NF-κB MAPK |
| 10 | Kubina et al. [91] | 37371038 | Kaempferol & Fisetin | In vitro (SCC9, SCC25) |
|
| Bax/Bcl-2 |
| 11 | Bostan et al. [108] | 32859062 | Resveratrol & Curcumin | In vitro (PE/CA-PJ49) |
|
| Caspase |
| No | Author | PMID | Natural Product | Preclinical Model | Test & Dosage | Findings | Pathway Related |
|---|---|---|---|---|---|---|---|
| 1 | Zhao et al. [41] | 34175854 | Actein | In vitro (CAL27, SCC9) |
|
| Akt/FoxO1 Bax/Bcl-2 |
| In vivo (CAL27) C57BL/6 mice |
|
| |||||
| 2 | Zhang et al. [42] | 32267053 | Salicylate | In vitro (SAS) |
|
| Akt/mTOR |
| In vivo (SAS) Nude mice |
|
| |||||
| 3 | Li et al. [43] | 32424132 | Tanshinone IIA | In vitro (CAL27, SCC9, SCC15, SCC25) |
|
| Akt/c-Myc |
| In vivo (CAL27, SCC15) Athymic nude mice |
|
| |||||
| 4 | Li et al. [44] | 32410646 | Xanthohumol | In vitro (CAL27, SCC9, SCC15, SCC25) |
|
| Akt/Wee1/Cdk1 Bax/Bcl-2 Caspase |
| In vivo (CAL27, SCC25) Athymic nude mice |
|
| |||||
| 5 | Huang et al. [45] | 29363886 | Honokiol | In vitro (OC2, OCSL) |
|
| Akt/mTOR MAPK |
| In vivo (SAS) BALB/c nude mice, AnN.Cg-Foxn1nu/CrlNarl |
|
| |||||
| 6 | Chen et al. [65] | 35785707 | Trichodermin | In vitro (Ca922, HSC3) |
|
| HDAC-2 Caspase |
| In vivo (HSC3) Zebrafish |
|
| |||||
| 7 | Wang et al. [24] | 31750253 | Psorachromene | In vitro (SAS, OECM1) |
|
| EGFR EMT-related Caspase SLUG |
| In vivo (SAS) BALB/c nude mice |
|
| |||||
| 8 | Li et al. [56] | 35904511 | Protocatechuic acid | In vitro (HSC3, CAL27) |
|
| JNK/p38 Caspase |
| In vivo (CAL27) Nude mice |
|
| |||||
| 9 | de Compos et al. [110] | 28782139 | Curcumin | In vitro (CAL27, SCC25, HACAT, NIH-3T3) |
|
| |
| In vivo (HNSCC Biopsy) BALB/c nude mice |
|
|
| No | Author | PMID | Natural Product | Preclinical Model | Test & Dosage | Findings | Pathway Related |
|---|---|---|---|---|---|---|---|
| 1 | Vincent-Chong et al. [25] | 30875566 | Calcitriol | In vivo (4NQO-induced carcinogenesis) C57BL/6NCr mice |
|
| |
| 2 | Wang et al. [64] | 33156559 | Neferine | In vivo (DMBA-induced carcinogenesis) Syrian hamster |
|
| NF-κB |
| 3 | Sophia et al. [47] | 30352996 | Nimbolide | In vivo (DMBA-induced carcinogenesis) Syrian hamster |
|
| PI3K/Akt/GSK-3β |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liew, Y.X.; Karen-Ng, L.P.; Vincent-Chong, V.K. A Comprehensive Review of Natural Products as Therapeutic or Chemopreventive Agents against Head and Neck Squamous Cell Carcinoma Cells Using Preclinical Models. Biomedicines 2023, 11, 2359. https://doi.org/10.3390/biomedicines11092359
Liew YX, Karen-Ng LP, Vincent-Chong VK. A Comprehensive Review of Natural Products as Therapeutic or Chemopreventive Agents against Head and Neck Squamous Cell Carcinoma Cells Using Preclinical Models. Biomedicines. 2023; 11(9):2359. https://doi.org/10.3390/biomedicines11092359
Chicago/Turabian StyleLiew, Yoon Xuan, Lee Peng Karen-Ng, and Vui King Vincent-Chong. 2023. "A Comprehensive Review of Natural Products as Therapeutic or Chemopreventive Agents against Head and Neck Squamous Cell Carcinoma Cells Using Preclinical Models" Biomedicines 11, no. 9: 2359. https://doi.org/10.3390/biomedicines11092359
APA StyleLiew, Y. X., Karen-Ng, L. P., & Vincent-Chong, V. K. (2023). A Comprehensive Review of Natural Products as Therapeutic or Chemopreventive Agents against Head and Neck Squamous Cell Carcinoma Cells Using Preclinical Models. Biomedicines, 11(9), 2359. https://doi.org/10.3390/biomedicines11092359