Neuromodulation of the Autonomic Nervous System in Chronic Low Back Pain: A Randomized, Controlled, Crossover Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
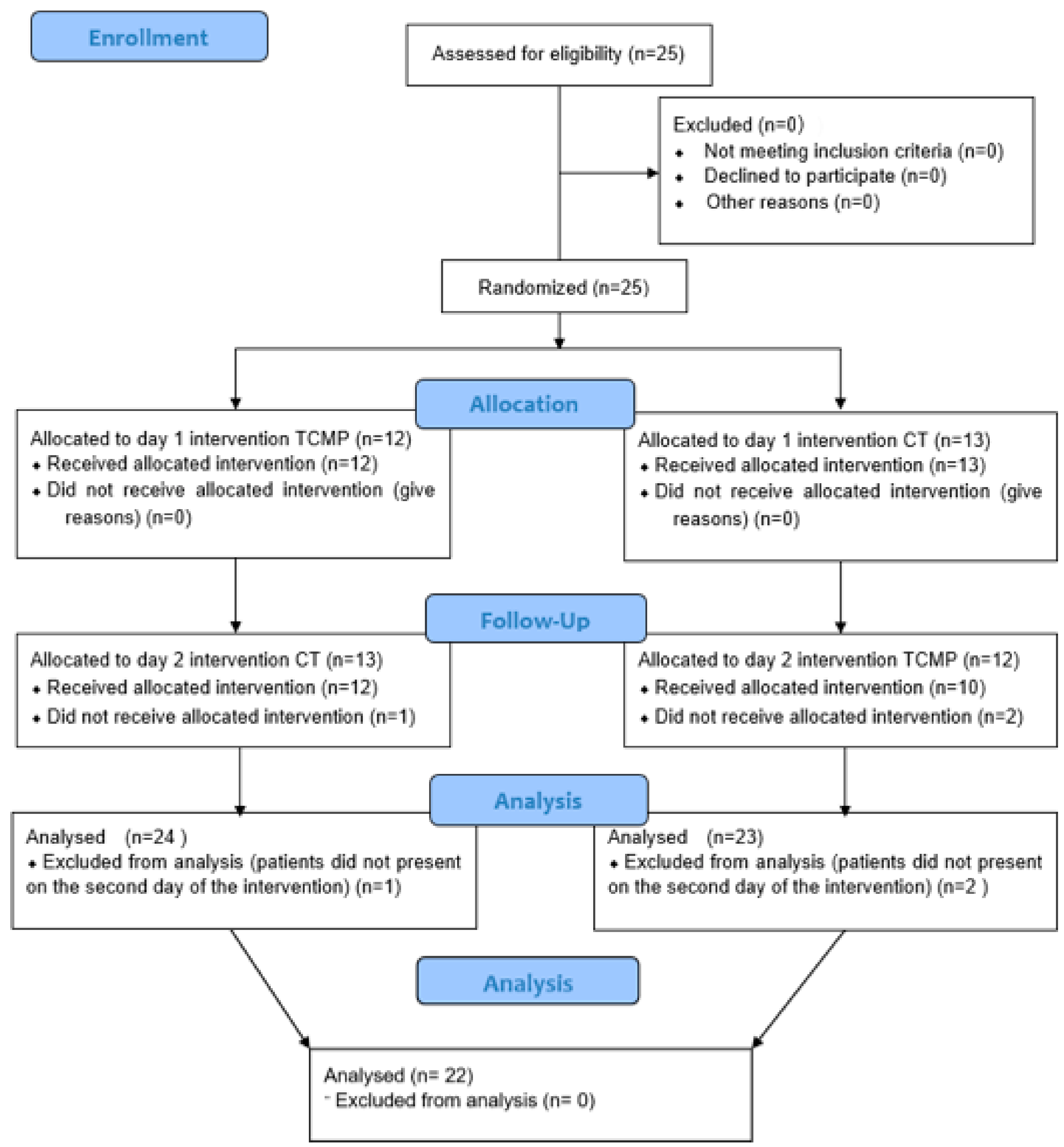
2.2. Randomization of the Sample
2.3. Automated Measures
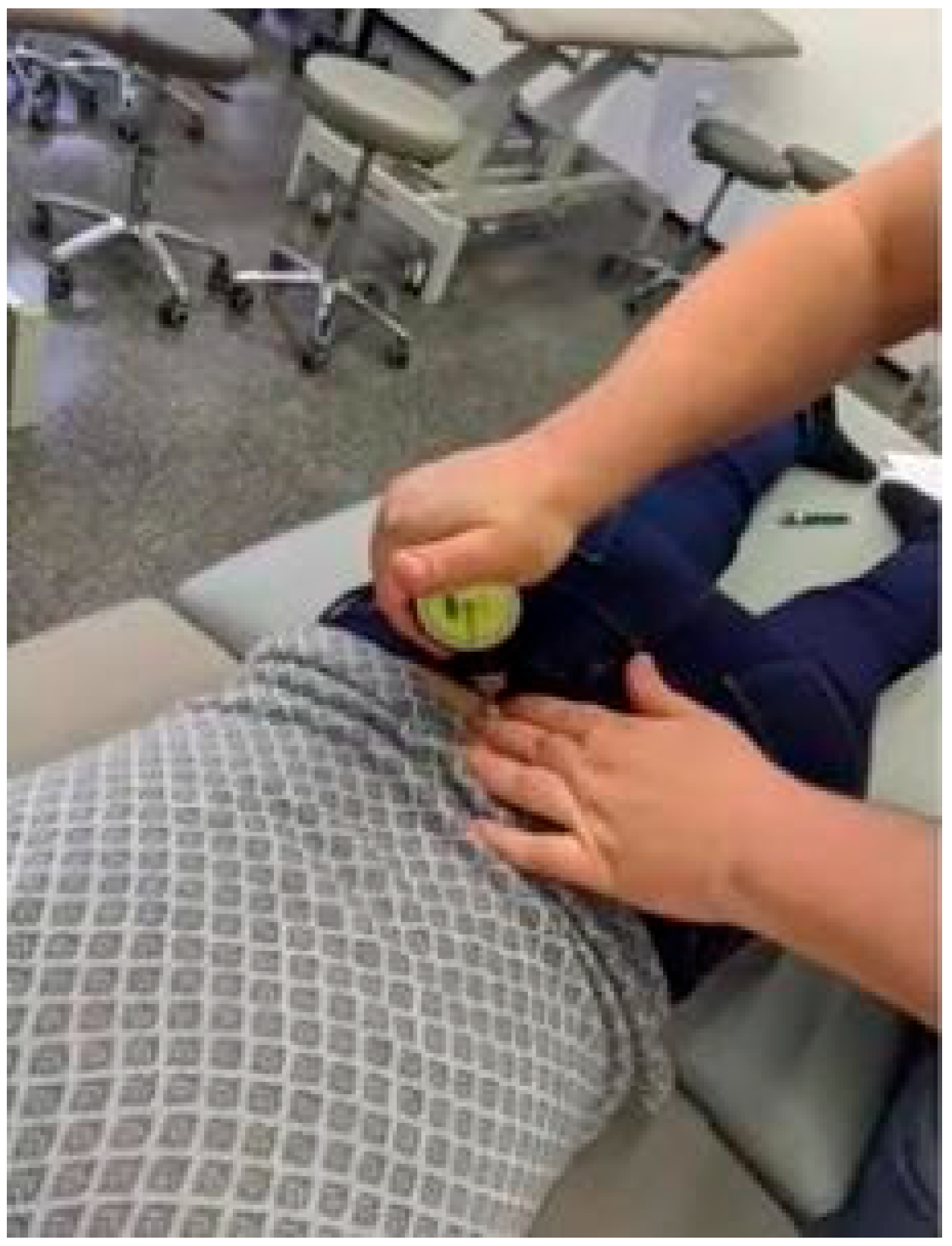
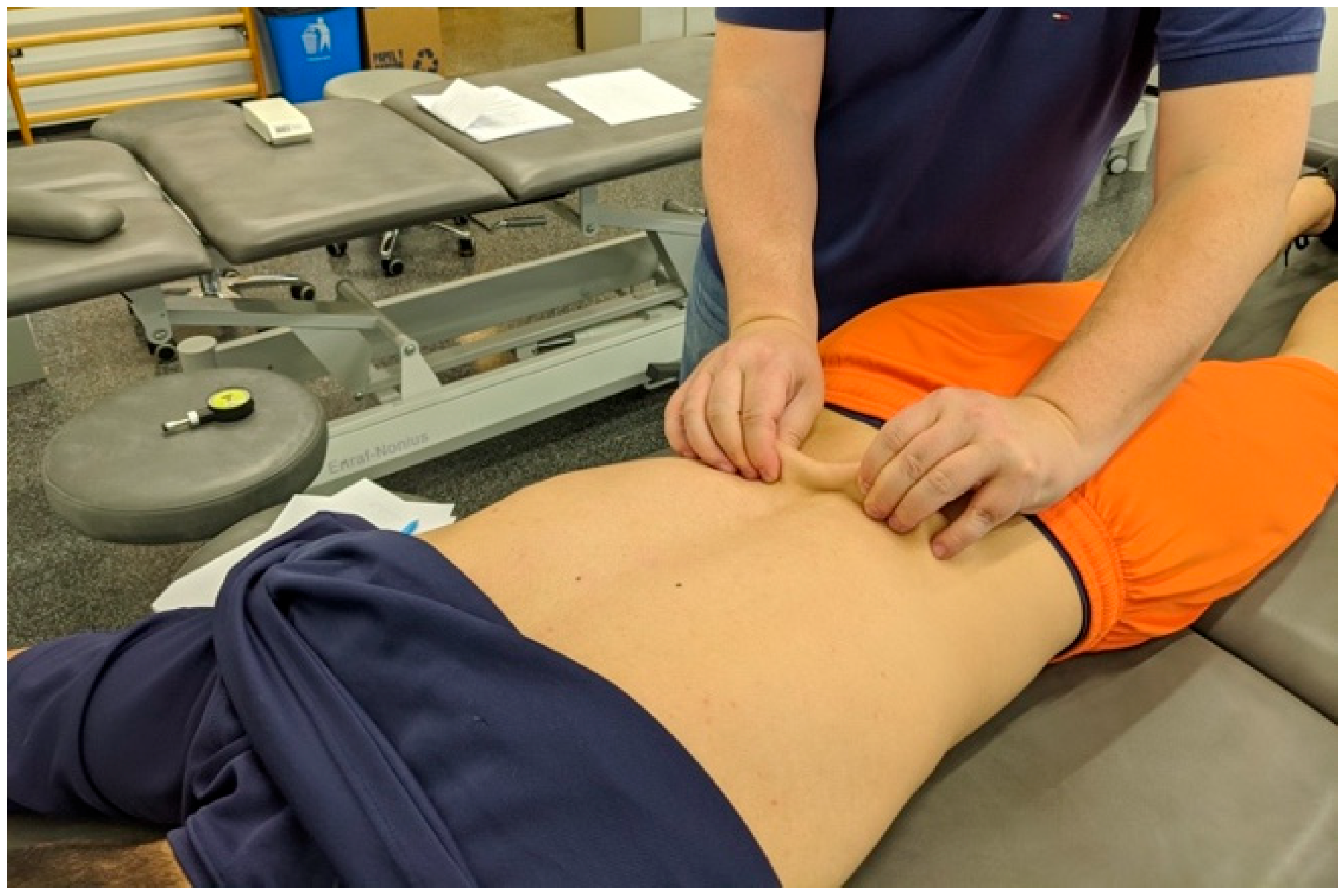
2.4. Study Protocol
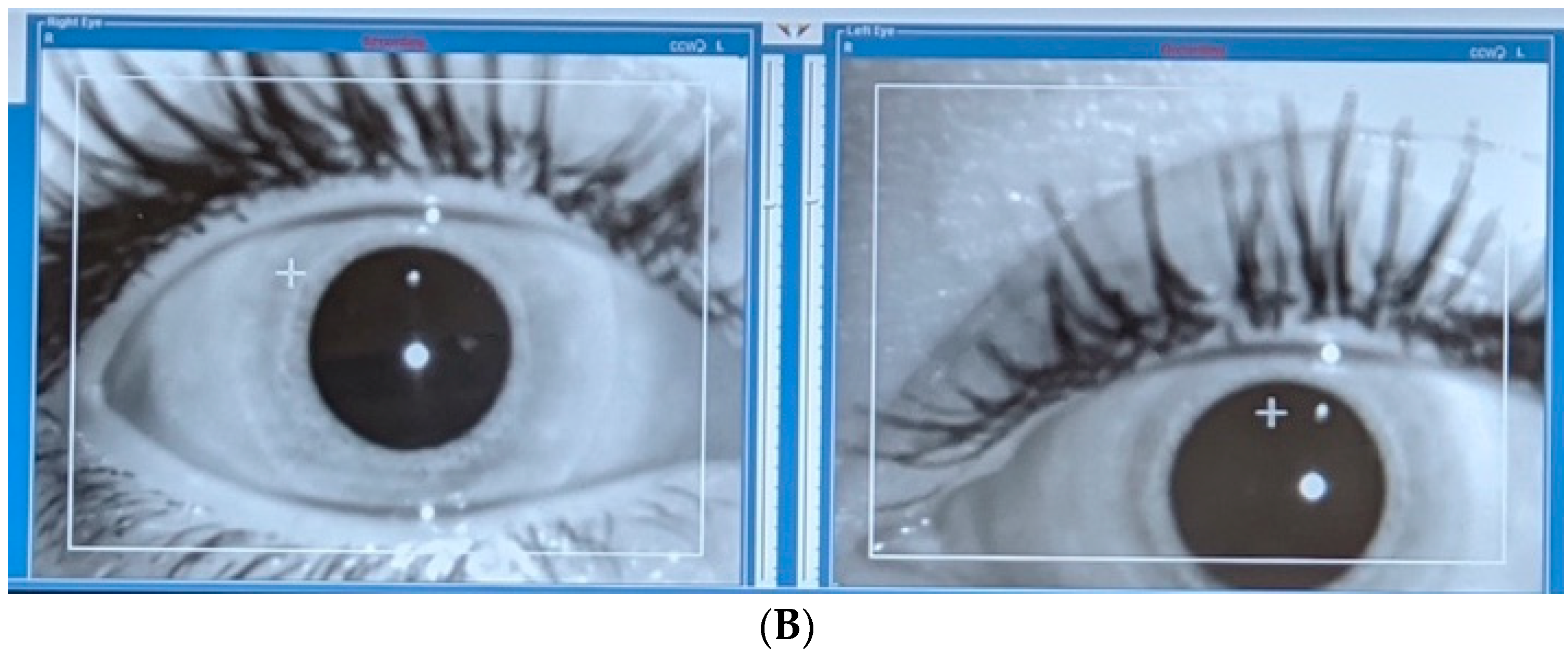
2.5. Pupillometry Measurement Protocol
2.6. Sample Size
2.7. Statistical Analysis
3. Results
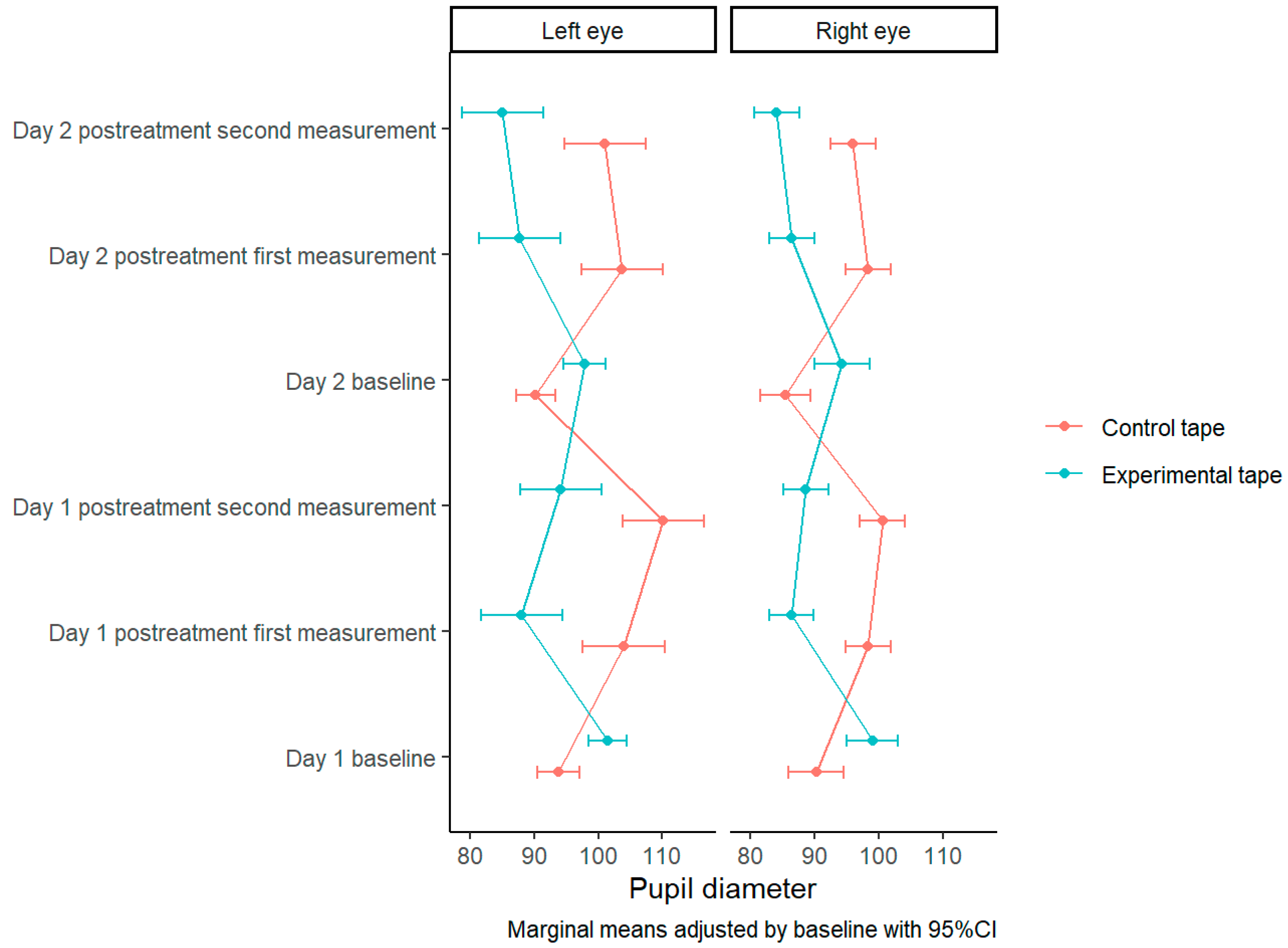
3.1. Pupillometry Outcomes
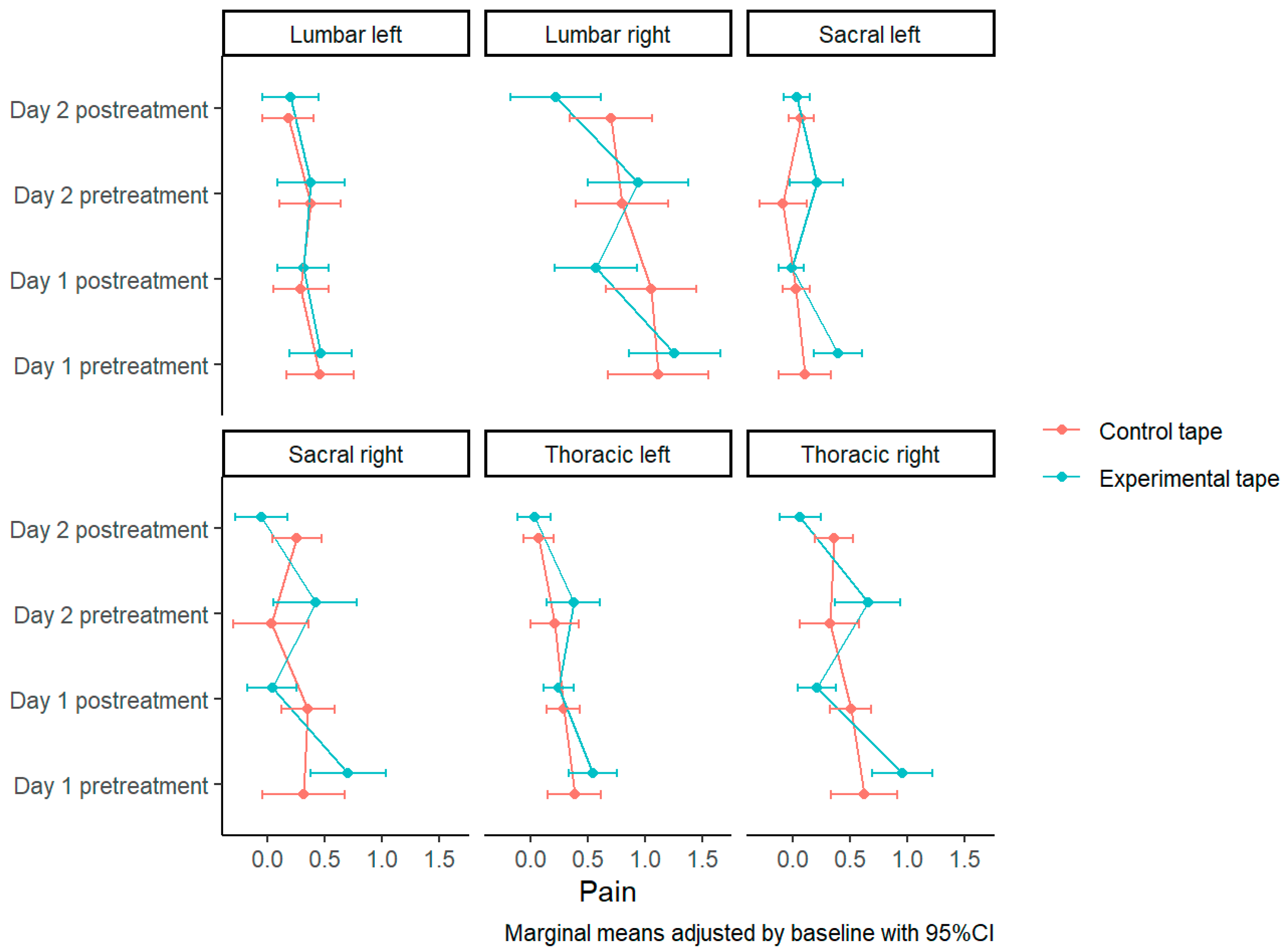
3.2. Pain with Posterior to Anterior Pressure on the Spine
3.3. Pain with Paravertebral Skin Pinch Test on the Spine
4. Discussion
Limitations and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Manchikanti, L.; Singh, V.; Datta, S.; Cohen, S.P.; Hirsch, J.A. Comprehensive review of epidemiology, scope, and impact of spinal pain. Pain Physician 2009, 12, E35–E70. [Google Scholar] [CrossRef] [PubMed]
- Sánchez Romero, E.A.; Alonso Pérez, J.L.; Muñoz Fernández, A.C.; Battaglino, A.; Castaldo, M.; Cleland, J.A.; Villafañe, J.H. Reliability of Sonography Measures of the Lumbar Multifidus and Transversus Abdominis during Static and Dynamic Activities in Subjects with Non-Specific Chronic Low Back Pain. Diagnostics 2021, 11, 632. [Google Scholar] [CrossRef] [PubMed]
- Lachance, C.C.; McCormack, S. Mindfulness Training for Chronic Non-Malignant Pain Management: A Review of the Clinical Effectiveness, Cost-Effectiveness and Guidelines; CADTH Rapid Response Reports; CADTH: Ottawa, ON Canada, 2019. [Google Scholar]
- Coutaux, A.; Adam, F.; Willer, J.; Le Bars, D. Hyperalgesia and allodynia: Peripheral mechanisms. Jt. Bone Spine 2005, 72, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Bernards, J.; Bouman, L. Fysiologie van de Mens; Bohn, Scheltema & Holkema: Utrecht, The Netherlands, 1988. [Google Scholar]
- Cyriax, J. Textbook of Orthopaedic Medicine, Volume One Diagnosis of Soft Tissue Lesions, 8th ed.; Bailliere Tindall: Paris, France, 1982. [Google Scholar]
- Staud, R.; Vierck, C.; Cannon, R.; Mauderli, A.; Price, D. Abnormal sensitization and temporal summation of second pain (wind-up) in patients with fibromyalgia syndrome. Pain 2001, 91, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.; Doubell, T. The pathophysiology of chronic pain-increased sensitivity to low threshold Abeta-fibre inputs. Curr. Opin. Neurobiol. 1994, 4, 525–534. [Google Scholar] [CrossRef]
- Martínez-Pozas, O.; Sánchez-Romero, E.A.; Beltran-Alacreu, H.; Arribas-Romano, A.; Cuenca-Martínez, F.; Villafañe, J.H.; Fernández-Carnero, J. Effects of Orthopedic Manual Therapy on Pain Sensitization in Patients with Chronic Musculoskeletal Pain: An Umbrella Review with Meta-Meta-Analysis. Am. J. Phys. Med. Rehabil. 2023. [Google Scholar] [CrossRef]
- Benarroch, E. Pain-autonomic interactions. Neurol. Sci. 2006, 27 (Suppl. S2), S130–S133. [Google Scholar] [CrossRef]
- Barman, S.; Wurster, R. Interaction of descending spinal sympathetic pathways and afferent nerves. Am. J. Physiol. 1978, 234, H223–H229. [Google Scholar] [CrossRef]
- Sato, A. Somato-Sympathetic Reflex Discharges Evoked through Supramedullary Pathways. Pflugers Arch. 1972, 332, 117–126. [Google Scholar] [CrossRef]
- Sato, A. Neural Mechanisms of Autonomic Responses Elicted by Somatic Stimulation. Neurosci. Biobehav. Physiol. 1997, 27, 610–621. [Google Scholar] [CrossRef]
- Abdelnaby, R.; Meshref, M.; Gaber, D.E.; Mohamed, K.A.; Madhav, N.; Elshabrawy, H.A. Assessing the structural and functional changes in vagus nerve in multiple sclerosis. Med. Hypotheses 2022, 164, 110863. [Google Scholar] [CrossRef]
- Vicenzino, B.; Collins, D.; Wright, T. Sudomotor Changes Induced by Neural Mobilisation Techniques in Asymptomatic Subjects. J. Man. Manip. Ther. 1994, 2, 66–74. [Google Scholar] [CrossRef]
- Bertinotti, L.; Pietrini, U.; Rosso, A.; Casale, R.; Colangelo, N.; Zoppi, M.; Matucci-Cerinic, M. The Use of Pupillometry in Joint and Connective Tissue Diseases. N. Y. Acad. Sci. 2002, 966, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Bitsios, P.; Prettyman, R.; Szabadi, E. Changes in Autonomic Function with Age: A Study of Pupillary Kinetics in Healthy Young and Old People. Age Ageing 1996, 25, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Capao Filipe, J.; Falcao-Reis, F.; Castro-Correia, J.; Barros, H. Assessment of autonomic function in high level athletes by pupillometry. Auton. Neurosci. Basic Clin. 2003, 104, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Fotiou, F.; Fountoulakis, K.; Goulas, A.; Alexopoulos, L.; Palikaras, A. Automated standardized pupillometry with optical method for purposes of clinical practice and research. Clin. Physiol. 2000, 20, 336–347. [Google Scholar] [CrossRef]
- Butler, D. The Sensitive Nervous System; Noigroup Publications: Adelaide, Australia, 2000. [Google Scholar]
- Gibbons, P.; Gosling, C.; Holmes, M. Short-Term Effects of Cervical Manipulation on Edge Light Pupil Cycle Time: A Pilot Study. J. Manip. Physiol. Ther. 2000, 23, 465–469. [Google Scholar] [CrossRef]
- Sillevis, R.; Cleland, J.; Hellman, M.; Beekhuizen, K. Immediate effects of a thoracic spine thrust manipulation on the autonomic nervous system: A randomized clinical trial. J. Man. Manip. Ther. 2010, 18, 181–190. [Google Scholar] [CrossRef]
- Sillevis, R.; Cleland, J. Immediate effects of the audible pop from a thoracic spine thrust manipulation on the autonomic nervous system and pain: A secondary analysis of a randomized clinical trial. J. Manip. Physiol. Ther. 2011, 34, 37–45. [Google Scholar] [CrossRef]
- Sillevis, R.; Van Duijn, J.; Shamus, E.; Hard, M. Time effect for in-situ dry needling on the autonomic nervous system, a pilot study. Physiother. Theory Pract. 2019, 37, 826–834. [Google Scholar] [CrossRef]
- Rice, F.L.; Castel, D.; Ruggiero, E.; Dockum, M.; Houk, G.; Sabbag, I.; Albrecht, P.J.; Meilin, S. Human-like cutaneous neuropathologies associated with a porcine model of peripheral neuritis: A translational platform for neuropathic pain. Neurobiol. Pain 2019, 5, 100021. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.-C.; Tseng, M.-T.; Lin, Y.-H.; Hsieh, P.-C.; Lin, C.-H.J.; Huang, S.-L.; Hsieh, S.-T.; Chiang, M.-C. Brain imaging signature of neuropathic pain phenotypes in small-fiber neuropathy: Altered thalamic connectome and its associations with skin nerve degeneration. Pain 2021, 162, 1387–1399. [Google Scholar] [CrossRef] [PubMed]
- Denda, M.; Nakanishi, S. Do epidermal keratinocytes have sensory and information processing systems? Exp. Dermatol. 2022, 31, 459–474. [Google Scholar] [CrossRef]
- Kimura, S.; Hosaka, N.; Yuge, I.; Yamazaki, A.; Suda, K.; Taneichi, H.; Denda, H.; Endo, N. Cerebrospinal fluid concentrations of nitric oxide metabolites in spinal cord injury. Spine 2009, 34, E645–E652. [Google Scholar] [CrossRef]
- Boulais, N.; Misery, L. The epidermis: A sensory tissue. Eur. J. Dermatol. 2008, 18, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Kimber, I.; Cumberbatch, M. Stimulation of Langerhans cell migration by tumor necrosis factor alpha (TNF-alpha). J. Investig. Dermatol. 1992, 99, 48S–50S. [Google Scholar] [CrossRef] [PubMed]
- Talagas, M.; Lebonvallet, N.; Berthod, F.; Misery, L. Cutaneous nociception: Role of keratinocytes. Exp. Dermatol. 2019, 28, 1466–1469. [Google Scholar] [CrossRef] [PubMed]
- Talagas, M.; Lebonvallet, N.; Misery, L. Intraepidermal nerve fibres are not the exclusive tranducers of nociception. J. Neurosci. Methods 2018, 306, 92–93. [Google Scholar] [CrossRef]
- Selva-Sarzo, F.; Fernandez-Carnero, S.; Sillevis, R.; Hernandez-Garces, H.; Benitez-Martinez, J.C.; Cuenca-Zaldivar, J.N. The Direct Effect of Magnetic Tape((R)) on Pain and Lower-Extremity Blood Flow in Subjects with Low-Back Pain: A Randomized Clinical Trial. Sensors 2021, 21, 6517. [Google Scholar] [CrossRef]
- Cardoso, V.F.; Francesko, A.; Ribeiro, C.; Banobre-Lopez, M.; Martins, P.; Lanceros-Mendez, S. Advances in Magnetic Nanoparticles for Biomedical Applications. Adv. Healthc. Mater. 2018, 7, 1700845. [Google Scholar] [CrossRef]
- Pergolizzi, J.V., Jr.; LeQuang, J.A. Rehabilitation for Low Back Pain: A Narrative Review for Managing Pain and Improving Function in Acute and Chronic Conditions. Pain Ther. 2020, 9, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Hilliges, M.; Wang, L.; Johansson, O. Ultrastructural evidence for nerve fibers within all vital layers of the human epidermis. J. Investig. Dermatol. 1995, 104, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Alfano, A.P.; Taylor, A.G.; Foresman, P.A.; Dunkl, P.R.; McConnell, G.G.; Conaway, M.R.; Gillies, G.T. Static magnetic fields for treatment of fibromyalgia: A randomized controlled trial. J. Altern. Complement. Med. 2001, 7, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Richmond, S.J. Magnet therapy for the relief of pain and inflammation in rheumatoid arthritis (CAMBRA): A randomised placebo-controlled crossover trial. Trials 2008, 9, 53. [Google Scholar] [CrossRef]
- Burbridge, C.; Randall, J.A.; Abraham, L.; Bush, E.N. Measuring the impact of chronic low back pain on everyday functioning: Content validity of the Roland Morris disability questionnaire. J. Patient-Rep. Outcomes 2020, 4, 70. [Google Scholar] [CrossRef]
- Harle, D.; Wolffsohn, J.; Evans, B. The pupillary light reflex in migraine. Ophthal. Physiol. Opt. 2005, 25, 240–245. [Google Scholar] [CrossRef]
- Dutsch, M.; Hilz, M.; Raunhut, U.; Solomon, J.; Neundorfer, B.; Axelrod, F. Sympathetic and parasympathetic pupillary dysfunction in familial dysautonomia. J. Neurol. Sci. 2002, 195, 77–83. [Google Scholar] [CrossRef]
- Pfeifer, M.; Cook, D.; Brodsky, J.; Tice, D.; Parrish, D.; Reenan, A.; Halter, J.B.; Porte, D. Quantitative Evaluation of Sympathetic and Parasympathetic Control of Iris Function. Diabetes Care Diabetes Care 1982, 5, 518–528. [Google Scholar] [CrossRef]
- Bakes, A.; Bradshaw, M.; Szabadi, E. Attentuation of the pupillary light reflex in anxious patients. Br. J. Clin. Pharmac. 1990, 30, 377–381. [Google Scholar] [CrossRef]
- Boxer Walcher, B.; Krueger, R. Agreement and Repeatability of Infrared Pupillometry and the Comparison Method. Ophthalmology. 1999, 106, 319–323. [Google Scholar] [CrossRef]
- Boxer Walcher, B.; Krueger, R. Agreement and repeatability of pupillometry using videokeratography and infrared devices. J. Cataract. Refract. Surg. 2000, 26, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Rowley, D.; Abraham, R. Portable infrared pupilloemtry using Pupilscan: Relation to somatic and autonomic nerve function in diabetes mellitus. Clin. Auton. Res. 1992, 2, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Meeker, M.; Du, R.; Bachetti, C.; Larson, M.; Holland, M.; Manley, G. Pupil Examination: Validity and Clinical Utility of an Automated Pupillometer. J. Neurosci. Nurs. 2005, 37, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Piha, S.; Halonen, J.-P. Infrared pupillometry in the assessment of autonomic function. Diabetes Res. Clin. Pract. 1994, 26, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Pop, M.; Payette, Y.; Santoriello, E. Comparison of the pupil card and pupillometer in measuring pupil size. J. Cataract. Refract. Surg. 2002, 28, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Twa, M.; Bailey, M.; Hayes, J.; Bullimore, M. Estimation of pupil size by digital photography. J. Cataract. Refract. Surg. 2004, 30, 381–389. [Google Scholar] [CrossRef]
- Taylor, W.; Chen, J.; Meltzer, H.; Gennarelli, T.A.; Kelbch, C.; Knowlton, S.; Richardson, J.; Lutch, M.J.; Farin, A.; Hults, K.N.; et al. Quantitative pupillometry, a new technology: Normative data and preliminary observations in patients with acute head injury. J. Neurosurg. 2003, 98, 205–213. [Google Scholar] [CrossRef]
- Neil, H.; Smith, S. A simple clinical test of pupillary autonomic function, Correlation with cardiac autonomic function tests in diabetes. Neuro-Ophtalmol. 1989, 9, 237–242. [Google Scholar] [CrossRef]
- Giakoumaki, S.; Hourdaki, E.; Grinakis, V.; Theou, K.; Bitsios, P. Effects of peripheral sympatehtic blockade with dapiprazole on the fear-inhibited light reflex. J. Psychopharmacol. 2005, 19, 139–148. [Google Scholar] [CrossRef]
- Miciele, G.; Tassorelli, C.; Martignoni, E.; Marcheselli, S.; Rossi, F.; Nappi, G. Further characterization of autonomic involvement in multiple system atrophy: A pupillometric study. Funct. Neurol. 1995, 10, 273–280. [Google Scholar]
- Giamberardino, M.A.; de Bigontina, P.; Martegiani, C.; Vecchiet, L. Effects of extracorporeal shock-wave lithotripsy on referred hyperalgesia from renal/ureteral calculosis. Pain 1994, 56, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Giamberardino, M.A.; Affaitati, G.; Lerza, R.; Lapenna, D.; Costantini, R.; Vecchiet, L. Relationship between pain symptoms and referred sensory and trophic changes in patients with gallbladder pathology. Pain 2005, 114, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Lawson, J. Design and Analysis of Experiments with R; CRC Press: Boca Raton, FL, USA, 2014; Volume 115. [Google Scholar]
- Jameson, C.; Boulton, K.; Silove, N.; Nanan, R.; Guastella, A. Ectodermal origins of the skin-brain axis: A novel model for the developing brain, inflammation, and neurodevelopmental conditions. Mol. Psychiatry 2023, 28, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Rhudy, J.; Williams, A.; McCabe, K.; Thu, M.; Nguyen, V.; Rambo, P. Affective modulation of nociception at spinal and supraspinal levels. Psychophysiology 2005, 42, 579–587. [Google Scholar] [CrossRef]
- Rhudy, J.; Williams, A.; McCabe, K.; Rambo, P.; Russell, J. Emotional modulation of spinal nociception and pain: The impact of predictable noxious stimulation. Pain 2006, 126, 221–233. [Google Scholar] [CrossRef]
- Watkins, L.; Milligan, E.; Maier, S. Spinal cord glia: New players in pain. Pain 2001, 93, 201–205. [Google Scholar] [CrossRef]
- Ellermeier, W.; Westphal, W. Gender differences in pain ratings and pupil reactions to painful pressure stimuli. Pain 1995, 61, 435–439. [Google Scholar] [CrossRef]
- Oka, S.; Chapman, R.; Kim, B.; Nahajima, I.; Shimizu, O.; Oi, Y. Pupil dilation response to noxious stimulation: Effect of varying nitrous oxide concentration. Clin. Neurophysiol. 2007, 118, 2016–2024. [Google Scholar] [CrossRef]


| n | 22 | |
|---|---|---|
| Gender, n (%) | Female | 14 (63.6) |
| Male | 8 (36.4) | |
| Age | 44.59 ± 8.17 | |
| Weight (kg) | 72.41 ± 12.67 | |
| Height (cm) | 168.91 ± 8.90 | |
| Body mass index | 25.42 ± 4.41 |
| Coefficient (SE) | 95% CI | t Value | a p Value | F(df) | a p Value | Overall Model | ||
|---|---|---|---|---|---|---|---|---|
| Right eye | (Intercept) | 29.694 (SE = 7.26) | 15.19, 44.198 | 4.090 | <0.001 | 16.727(1) | <0.001 | Ra2 = 0.818 |
| Day 1 postreatment first measurement | −0.014 (SE = 1.336) | −2.682, 2.655 | −0.010 | 0.992 | 1.442(3) | 0.239 | F = 16.113, p < 0.001 | |
| Day 1 postreatment second measurement | 2.27 (SE = 1.336) | −0.399, 4.938 | 1.699 | 0.094 | NA | |||
| Day 2 postreatment first measurement | 0.034 (SE = 1.336) | −2.634, 2.702 | 0.025 | 0.98 | NA | |||
| Treatment | 5.952 (SE = 0.841) | 4.271, 7.632 | 7.077 | <0.001 | 50.078(1) | <0.001 | ||
| Left eye | (Intercept) | 30.239 (SE = 17.718) | −5.157, 65.635 | 1.707 | 0.093 | 2.913(1) | 0.093 | Ra2 = 0.659 |
| Day 1 postreatment first measurement | −0.717 (SE = 2.403) | −5.516, 4.083 | −0.298 | 0.766 | 1.9(3) | 0.138 | F = 7.527, p < 0.001 | |
| Day 1 postreatment second measurement | 5.411 (SE = 2.403) | 0.612, 10.211 | 2.252 | 0.028 | NA | |||
| Day 2 postreatment first measurement | −1.021 (SE = 2.403) | −5.82, 3.779 | −0.425 | 0.672 | NA | |||
| Treatment | 7.963 (SE = 1.551) | 4.865, 11.061 | 5.135 | <0.001 | 26.371(1) | <0.001 |
| Marginal Means Adjusted by Baseline (SE) | 95% CI | Marginal Means Difference and p Value a | |||
|---|---|---|---|---|---|
| Pupillometry | |||||
| Right eye | Control tape | 98.231 (SE = 1.134) | 95.965, 100.498 | Control tape—Experimental tape | 11.903 (SE = 1.682) |
| Experimental tape | 86.328 (SE = 1.134) | 84.062, 88.595 | t(64) = 7.077, p ≤ 0.001 | ||
| Left eye | Control tape | 104.698 (SE = 2.069) | 100.565, 108.832 | Control tape—Experimental tape | 15.926 (SE = 3.101) |
| Experimental tape | 88.772 (SE = 2.069) | 84.638, 92.906 | t(64) = 5.135, p ≤ 0.001 | ||
| Posterior-anterior directed pressure | |||||
| Control tape | 0.163 (SE = 0.051) | 0.062, 0.264 | Control tape—Experimental tape | 0.109 (SE = 0.073) | |
| Experimental tape | 0.054 (SE = 0.051) | −0.046, 0.155 | Control tape—Experimental tape | t(250) = 1.493, p = 0.137 | |
| Paravertebral skin pinch test | |||||
| Thoracic right | Control tape | 0.433 (SE = 0.073) | 0.289, 0.576 | Control tape—Experimental tape | 0.293 (SE = 0.104) |
| Experimental tape | 0.14 (SE = 0.073) | −0.003, 0.283 | t(526) = 0.293, p = 0.005 | ||
| Lumbar right | Control tape | 0.88 (SE = 0.155) | 0.574, 1.186 | Control tape—Experimental tape | 0.482 (SE = 0.22) |
| Experimental tape | 0.398 (SE = 0.155) | 0.092, 0.704 | t(204) = 0.482, p = 0.03 | ||
| Sacral right | Control tape | 0.302 (SE = 0.092) | 0.12, 0.483 | Control tape—Experimental tape | 0.31 (SE = 0.131) |
| Experimental tape | −0.008 (SE = 0.092) | −0.19, 0.173 | t(158) = 0.31, p = 0.019 | ||
| Thoracic left | Control tape | 0.183 (SE = 0.057) | 0.072, 0.295 | Control tape—Experimental tape | 0.041 (SE = 0.081) |
| Experimental tape | 0.143 (SE = 0.057) | 0.031, 0.254 | t(526) = 0.041, p = 0.614 | ||
| Lumbar left | Control tape | 0.234 (SE = 0.096) | 0.044, 0.424 | Control tape—Experimental tape | −0.019 (SE = 0.137) |
| Experimental tape | 0.253 (SE = 0.096) | 0.063, 0.443 | t(204) = −0.019, p = 0.891 | ||
| Sacral left | Control tape | 0.052 (SE = 0.047) | −0.04, 0.144 | Control tape—Experimental tape | 0.039 (SE = 0.067) |
| Experimental tape | 0.013 (SE = 0.047) | −0.079, 0.105 | t(158) = 0.039, p = 0.557 | ||
| Coefficient (SE) | 95% CI | F(df) | a p Value | Overall Model | ||
|---|---|---|---|---|---|---|
| Posterior-anterior directed pressure | ||||||
| (Intercept) | 0.051 (SE = 0.036) | −0.021, 0.122 | 1.921(1) | 0.167 | Ra2 = 0.445 | |
| Period | 0.107 (SE = 0.036) | 0.036, 0.179 | 8.768(1) | 0.003 | F = 9.805, p ≤ 0.001 | |
| Treatment | 0.054 (SE = 0.036) | −0.017, 0.126 | 2.229(1) | 0.137 | ||
| Paravertebral skin pinch test | ||||||
| Thoracic right | (Intercept) | 0.153 (SE = 0.054) | 0.046, 0.26 | 7.956(1) | 0.005 | Ra2 = 0.139 |
| Period | 0.074 (SE = 0.052) | −0.028, 0.176 | 2.025(1) | 0.155 | F = 4.546, p ≤ 0.001 | |
| Treatment | 0.146 (SE = 0.052) | 0.044, 0.248 | 7.933(1) | 0.005 | ||
| Lumbar right | (Intercept) | 0.486 (SE = 0.127) | 0.236, 0.735 | 14.71(1) | <0.001 | Ra2 = 0.171 |
| Period | 0.176 (SE = 0.111) | −0.042, 0.394 | 2.527(1) | 0.113 | F = 2.891, p ≤ 0.001 | |
| Treatment | 0.241 (SE = 0.11) | 0.024, 0.458 | 4.78(1) | 0.03 | ||
| Sacral right | (Intercept) | 0.031 (SE = 0.067) | −0.101, 0.164 | 0.221(1) | 0.639 | Ra2 = 0.23 |
| Period | 0.048 (SE = 0.065) | −0.081, 0.177 | 0.549(1) | 0.46 | F = 3.191, p ≤ 0.001 | |
| Treatment | 0.155 (SE = 0.066) | 0.025, 0.285 | 5.576(1) | 0.019 | ||
| Thoracic left | (Intercept) | 0.077 (SE = 0.041) | −0.004, 0.158 | 3.451(1) | 0.064 | Ra2 = 0.153 |
| Period | 0.108 (SE = 0.04) | 0.028, 0.187 | 7.111(1) | 0.008 | F = 4.994, p ≤ 0.001 | |
| Treatment | 0.02 (SE = 0.04) | −0.059, 0.1 | 0.255(1) | 0.614 | ||
| Lumbar left | (Intercept) | 0.21 (SE = 0.072) | 0.068, 0.352 | 8.516(1) | 0.004 | Ra2 = 0.146 |
| Period | 0.056 (SE = 0.069) | −0.079, 0.191 | 0.672(1) | 0.413 | F = 2.568, p ≤ 0.001 | |
| Treatment | −0.009 (SE = 0.068) | −0.144, 0.126 | 0.019(1) | 0.891 | ||
| Sacral left | (Intercept) | 0.043 (SE = 0.033) | −0.023, 0.108 | 1.66(1) | 0.2 | Ra2 = 0.008 |
| Period | −0.023 (SE = 0.033) | −0.088, 0.042 | 0.477(1) | 0.491 | F = 1.057, p = 0.398 | |
| Treatment | 0.02 (SE = 0.033) | −0.046, 0.085 | 0.347(1) | 0.557 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sillevis, R.; Cuenca-Zaldívar, J.N.; Fernández-Carnero, S.; García-Haba, B.; Sánchez Romero, E.A.; Selva-Sarzo, F. Neuromodulation of the Autonomic Nervous System in Chronic Low Back Pain: A Randomized, Controlled, Crossover Clinical Trial. Biomedicines 2023, 11, 1551. https://doi.org/10.3390/biomedicines11061551
Sillevis R, Cuenca-Zaldívar JN, Fernández-Carnero S, García-Haba B, Sánchez Romero EA, Selva-Sarzo F. Neuromodulation of the Autonomic Nervous System in Chronic Low Back Pain: A Randomized, Controlled, Crossover Clinical Trial. Biomedicines. 2023; 11(6):1551. https://doi.org/10.3390/biomedicines11061551
Chicago/Turabian StyleSillevis, Rob, Juan Nicolás Cuenca-Zaldívar, Samuel Fernández-Carnero, Beatriz García-Haba, Eleuterio A. Sánchez Romero, and Francisco Selva-Sarzo. 2023. "Neuromodulation of the Autonomic Nervous System in Chronic Low Back Pain: A Randomized, Controlled, Crossover Clinical Trial" Biomedicines 11, no. 6: 1551. https://doi.org/10.3390/biomedicines11061551
APA StyleSillevis, R., Cuenca-Zaldívar, J. N., Fernández-Carnero, S., García-Haba, B., Sánchez Romero, E. A., & Selva-Sarzo, F. (2023). Neuromodulation of the Autonomic Nervous System in Chronic Low Back Pain: A Randomized, Controlled, Crossover Clinical Trial. Biomedicines, 11(6), 1551. https://doi.org/10.3390/biomedicines11061551









