The Homogeneous Azorean Machado-Joseph Disease Cohort: Characterization and Contributions to Advances in Research
Abstract
1. Machado-Joseph Disease: Overview
2. The Azorean MJD Cohort: Characterization
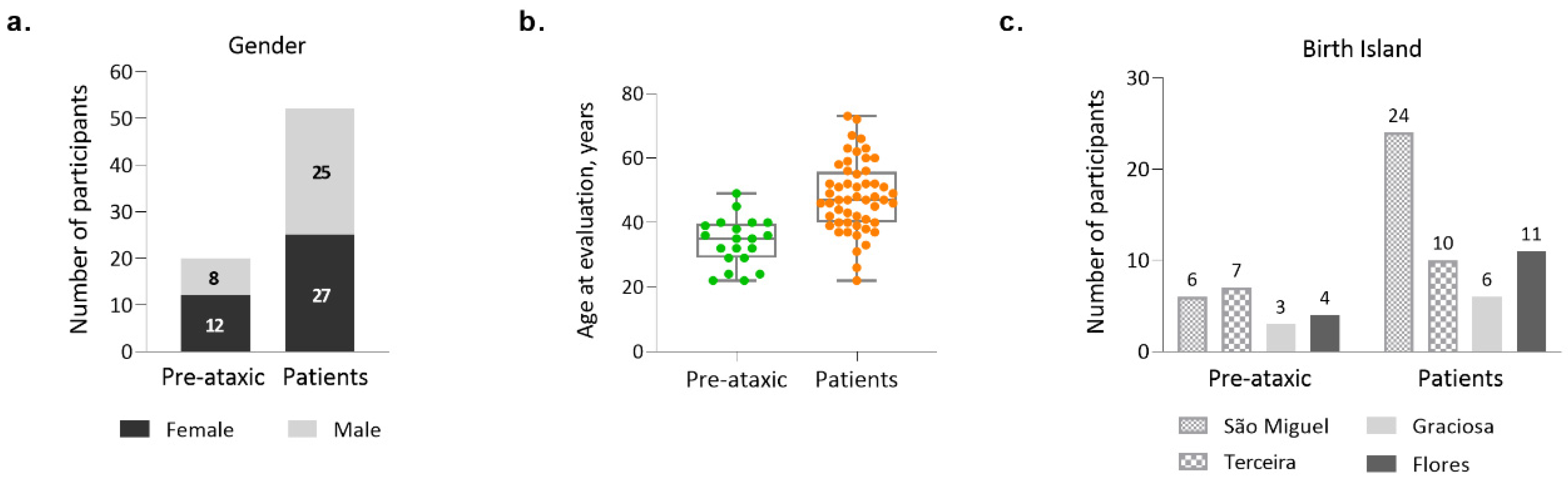
2.1. Demographic Characterization
2.2. Genetic Profile of the CAG Repeat at the ATXN3 Gene
2.3. Clinical Features
2.4. Patients’ Reported Outcome Measures (PROMs)
2.5. Relationship between Demographic, Clinical, and Patient-Reported Outcome Measures (PROMs)
3. The Azorean MJD Cohort: Contribute to Advances in MJD Research
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Klockgether, T.; Mariotti, C.; Paulson, H.L. Spinocerebellar ataxia. Nat. Rev. Dis. Prim. 2019, 5, 24. [Google Scholar] [CrossRef]
- Coutinho, P.; Ruano, L.; Loureiro, J.L.; Cruz, V.T.; Barros, J.; Tuna, A.; Barbot, C.; Guimarães, J.; Alonso, I.; Silveira, I.; et al. Hereditary ataxia and spastic paraplegia in Portugal: A population-based prevalence study. JAMA Neurol. 2013, 70, 746–755. [Google Scholar] [CrossRef] [PubMed]
- de Araujo, M.; Raposo, M.; Kazachkova, N.; Vasconcelos, J.; Kay, T.; Lima, M. Trends in the Epidemiology of Spinocerebellar Ataxia Type 3 / Machado-Joseph Disease in the Azores Islands, Portugal. JSM Brain Sci. 2016, 1, 1001. [Google Scholar]
- Paulson, H.; Shakkottai, V. Spinocerebellar Ataxia Type 3. In GeneReviews®; Adam, M.P., Everman, D.B., Mirzaa, G.M., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Gripp, K.W., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1998; pp. 1993–2022. [Google Scholar]
- Bettencourt, C.; Lima, M. Machado-Joseph Disease: From first descriptions to new perspectives. Orphanet J. Rare Dis. 2011, 6, 35. [Google Scholar] [CrossRef]
- Koeppen, A.H. The Neuropathology of Spinocerebellar Ataxia Type 3/Machado-Joseph Disease. Adv. Exp. Med. Biol. 2018, 1049, 233–241. [Google Scholar] [PubMed]
- Bettencourt, C.; Raposo, M.; Kazachkova, N.; Cymbron, T.; Santos, C.; Kay, T.; Vasconcelos, J.; Maciel, P.; Donis, K.C.; Pereira, M.L.S.; et al. The ε2 allele of APOE increases the risk of earlier age-at-onset in Machado-Joseph Disease (MJD/SCA3). Arch. Neurol. 2011, 68, 1580–1583. [Google Scholar] [CrossRef]
- de Mattos, E.P.; Leotti, V.B.; Soong, B.W.; Raposo, M.; Lima, M.; Vasconcelos, J.; Fussiger, H.; Souza, G.N.; Kersting, N.; Furtado, G.V.; et al. Age at onset prediction in spinocerebellar ataxia type 3 changes according to population of origin. Eur. J. Neurol. 2019, 26, 113–120. [Google Scholar] [CrossRef]
- Mergener, R.; Furtado, G.V.; de Mattos, E.P.; Leotti, V.B.; Jardim, L.B.; Saraiva-Pereira, M.L. Variation in DNA repair system gene as an additional modifier of age at onset in spinocerebellar ataxia type 3/Machado–Joseph disease. Neuromol. Med. 2020, 22, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Raposo, M.; Bettencourt, C.; Melo, A.R.V.; Ferreira, A.F.; Alonso, I.; Silva, P.; Vasconcelos, J.; Kay, T.; Saraiva-Pereira, M.L.; Costa, M.D.; et al. Novel Machado-Joseph disease-modifying genes and pathways identified by whole-exome sequencing. Neurobiol. Dis. 2022, 162, 105578. [Google Scholar] [CrossRef] [PubMed]
- Takiyama, Y.; Nishizawa, M.; Tanaka, H.; Kawashima, S.; Saka-Moto, H.; Karube, Y.; Shimazaki, H.; Soutome, M.; Endo, K.; Ohta, S.; et al. The gene for Machado–Joseph disease maps to human chromosome 14q. Nat. Genet. 1993, 4, 300–304. [Google Scholar] [CrossRef]
- Ichikawa, Y.; Goto, J.; Hattori, M.; Toyoda, A.; Ishii, K.; Jeong, S.Y.; Hashida, H.; Masuda, N.; Ogata, K.; Kasai, F.; et al. The genomic structure and expression of MJD, the Machado-Joseph disease gene. J. Hum. Genet. 2001, 46, 413–422. [Google Scholar] [CrossRef]
- Lima, M.; Costa, M.; Montiel, R.; Ferro, A.; Santos, C.; Silva, C.; Bettencourt, C.; Sousa, A.; Sequeiros, J.; Coutinho, P.; et al. Population Genetics of Wild-Type CAG Repeats in the Machado-Joseph Disease Gene in Portugal. Hum. Hered. 2005, 60, 156–163. [Google Scholar] [CrossRef]
- Costa, M.D.C.; Paulson, H.L. Toward understanding Machado–Joseph disease. Prog. Neurobiol. 2012, 97, 239–257. [Google Scholar] [CrossRef] [PubMed]
- Todi, V.S.; Pastore, A.; Paulson, L.H. Chapter 479-Ataxin-3. In Handbook of Proteolytic Enzymes, 3rd ed.; Rawlings, D.N., Salvesen, G., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 2123–2130. [Google Scholar]
- Matos, C.A.; de Almeida, L.P.; Nóbrega, C. Machado-Joseph disease/spinocerebellar ataxia type 3: Lessons from disease pathogenesis and clues into therapy. J. Neurochem. 2019, 148, 8–28. [Google Scholar] [CrossRef]
- Melo, A.R.V.; Ramos, A.; Kazachkova, N.; Raposo, M.; Bettencourt, B.F.; Rendeiro, A.R.; Kay, T.; Vasconcelos, J.; Bruges-Armas, J.; Lima, M. Triplet Repeat Primed PCR (TP-PCR) in Molecular Diagnostic Testing for Spinocerebellar Ataxia Type 3 (SCA3). Mol. Diagn. Ther. 2016, 20, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.; Raposo, M. Towards the Identification of Molecular Biomarkers of Spinocerebellar Ataxia Type 3 (SCA3)/Machado-Joseph Disease (MJD). Adv. Exp. Med. Biol. 2018, 1049, 309–319. [Google Scholar]
- Globas, C.; du Montcel, S.T.; Baliko, L.; Boesch, S.; Depondt, C.; DiDonato, S.; Durr, A.; Filla, A.; Klockgether, T.; Mariotti, C.; et al. Early symptoms in spinocerebellar ataxia type 1, 2, 3, and 6. Mov. Disord. 2008, 23, 2232–2238. [Google Scholar] [CrossRef]
- Schmitz-Hübsch, T.; Du Montcel, S.T.; Baliko, L.; Berciano, J.; Boesch, S.; Depondt, C.; Giunti, P.; Globas, C.; Infante, J.; Kang, J.S.; et al. Scale for the assessment and rating of ataxia: Development of a new clinical scale. Neurology 2006, 66, 1717–1720. [Google Scholar] [CrossRef]
- Brooker, S.M.; Edamakanti, C.R.; Akasha, S.M.; Kuo, S.; Opal, P. Spinocerebellar ataxia clinical trials: Opportunities and challenges. Ann. Clin. Transl. Neurol. 2021, 8, 1543–1556. [Google Scholar] [CrossRef]
- Maas, R.P.; Teerenstra, S.; Lima, M.; Pires, P.; Pereira de Almeida, L.; van Gaalen, J.; Timmann, D.; Infante, J.; Onyike, C.; Bushara, K.; et al. Differential Temporal Dynamics of Axial and Appendicular Ataxia in SCA3. Mov. Disord. 2022, 37, 1850–1860. [Google Scholar] [CrossRef]
- Katrin, B.; Sival, A.D. Scales for the clinical evaluation of cerebellar disorders. In Handbook of Clinical Neurology; Manto, M., Huisman, T.A.G.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 154, pp. 329–339. [Google Scholar]
- Jacobi, H.; Rakowicz, M.; Rola, R.; Fancellu, R.; Mariotti, C.; Charles, P.; Dürr, A.; Küper, M.; Timmann, D.; Linnemann, C.; et al. Inventory of Non-Ataxia Signs (INAS): Validation of a New Clinical Assessment Instrument. Cerebellum 2013, 12, 418–428. [Google Scholar] [CrossRef]
- Du Montcel, S.T.; Charles, P.; Ribai, P.; Goizet, C.; Le Bayon, A.; Labauge, P.; Guyant-Maréchal, L.; Forlani, S.; Jauffret, C.; Vandenberghe, N.; et al. Composite cerebellar functional severity score: Validation of a quantitative score of cerebellar impairment. Brain 2008, 131, 1352–1361. [Google Scholar] [CrossRef]
- Adanyeguh, I.M.; Perlbarg, V.; Henry, P.-G.; Rinaldi, D.; Petit, E.; Valabregue, R.; Brice, A.; Durr, A.; Mochel, F. Autosomal dominant cerebellar ataxias: Imaging biomarkers with high effect sizes. NeuroImage Clin. 2018, 19, 858–867. [Google Scholar] [CrossRef]
- Saute, J.A.M.; Rieder, C.R.; Castilhos, R.M.; Monte, T.L.; Schumacher-Schuh, A.F.; Donis, K.C.; D'ávila, R.; Souza, G.N.; Russo, A.D.; Furtado, G.V.; et al. Planning future clinical trials in Machado Joseph disease: Lessons from a phase 2 trial. J. Neurol. Sci. 2015, 358, 72–76. [Google Scholar] [CrossRef] [PubMed]
- D’Abreu, A.; Jr, M.F.; Conz, L.; Friedman, J.H.; Nucci, A.M.; Cendes, F.; Lopes-Cendes, I. Sleep symptoms and their clinical correlates in Machado-Joseph disease. Acta Neurol. Scand. 2009, 119, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Pedroso, J.L.; Braga-Neto, P.; Martinez, A.R.; Martins, C.R.; Filho, F.M.R.; Sobreira-Neto, M.A.; Prado, L.B.; Prado, G.; França, M.C.; Barsottini, O. Sleep disorders in Machado–Joseph disease. Curr. Opin. Psychiatry 2016, 29, 402–408. [Google Scholar] [CrossRef]
- Hengel, H.; Martus, P.; Faber, J.; Giunit, P.; Garcia-Moreno, H.; Solanky, N.; Klockgether, T.; Reetz, K.; van de Warrenburg, B.P.; Santana, M.M.; et al. The frequency of non-motor symptoms in SCA3 and their association with disease severity and lifestyle factors. J. Neurol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Sonni, A.; Kurdziel, L.B.; Baran, B.; Spencer, R.M. The effects of sleep dysfunction on cognition, affect, and quality of life in individuals with cerebellar ataxia. J. Clin. Sleep Med. 2014, 10, 535–543. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., III; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index (PSQI): A new instrument for psychiatric research and practice. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Yuan, X.; Ou, R.; Hou, Y.; Chen, X.; Cao, B.; Hu, X.; Shang, H. Extra-Cerebellar Signs and Non-motor Features in Chinese Patients With Spinocerebellar Ataxia Type 3. Front. Neurol. 2019, 10, 110. [Google Scholar] [CrossRef] [PubMed]
- Cecchin, C.; Pires, A.; Rieder, C.; Monte, T.; Silveira, I.; Carvalho, T.; Saraiva-Pereira, M.; Sequeiros, J.; Jardim, L. Depressive Symptoms in Machado-Joseph Disease (SCA3) Patients and Their Relatives. Public Health Genom. 2007, 10, 19–26. [Google Scholar] [CrossRef]
- Lo, R.Y.; Figueroa, K.P.; Pulst, S.M.; Perlman, S.; Wilmot, G.; Gomez, C.; Schmahmann, J.; Paulson, H.; Shakkottai, V.G.; Ying, S.; et al. Depression and clinical progression in spinocerebellar ataxias. Park. Relat. Disord. 2016, 22, 87–92. [Google Scholar] [CrossRef]
- Subramony, S.H.; May, W.; Lynch, D.; Gomez, C.; Fischbeck, K.; Hallett, M.; Taylor, P.; Wilson, R.; Ashizawa, T.; Cooperative Ataxia Group. Measuring Friedreich ataxia: Interrater reliability of a neurologic rating scale. Neurology 2005, 64, 1261–1262. [Google Scholar] [CrossRef] [PubMed]
- Hengel, H.; Martus, P.; Faber, J.; Garcia-Moreno, H.; Solanky, N.; Giunti, P.; Klockgether, T.; Reetz, K.; Van De Warrenburg, B.P.; de Almeida, L.P.; et al. Characterization of Lifestyle in Spinocerebellar Ataxia Type 3 and Association with Disease Severity. Mov. Disord. 2022, 37, 405–410. [Google Scholar] [CrossRef]
- Reetz, K.; the EFACTS or NICOFA Study Group; Hilgers, R.-D.; Isfort, S.; Dohmen, M.; Didszun, C.; Fedosov, K.; Kistermann, J.; Mariotti, C.; Durr, A.; et al. Protocol of a randomized, double-blind, placebo-controlled, parallel-group, multicentre study of the efficacy and safety of nicotinamide in patients with Friedreich ataxia (NICOFA). Neurol. Res. Prac. 2019, 1, 33. [Google Scholar] [CrossRef]
- Rabin, R.; Charro, F.D. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Reetz, K.; Dogan, I.; Costa, A.S.; Dafotakis, M.; Fedosov, K.; Giunti, P.; Parkinson, M.H.; Sweeney, M.G.; Mariotti, C.; Panzeri, M.; et al. Biological and clinical characteristics of the European Friedreich’s Ataxia Consortium for Translational Studies (EFACTS) cohort: A cross-sectional analysis of baseline data. Lancet Neurol. 2015, 14, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.K.; Dawson, D.M.; Spence, A. Machado disease: A hereditary ataxia in Portuguese emigrants to Massachusetts. Neurology 1972, 22, 49–55. [Google Scholar] [CrossRef]
- Woods, B.T.; Schaumburg, H.H. Nigro-spino-dentatal degeneration with nuclear ophthalmoplegia: A unique and partially treatable clinico-pathological entity. J. Neurol. Sci. 1972, 17, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, R.N.; Nyhan, W.L.; Bay, C.; Shore, P. Autosomal dominant striatonigral degeneration: A clinical, pathologic, and biochemical study of a new genetic disorder. Neurology 1976, 26, 703. [Google Scholar] [CrossRef] [PubMed]
- Romanul, F.C.; Fowler, H.L.; Radvany, J.; Feldman, R.G.; Feingold, M. Azorean disease of the nervous system. N. Engl. J. Med. 1977, 296, 1505–1508. [Google Scholar] [CrossRef]
- Gaspar, C.; Lopes-Cendes, I.; Hayes, S.; Goto, J.; Arvidsson, K.; Dias, A.; Silveira, I.; Maciel, P.; Coutinho, P.; Lima, M.; et al. Ancestral Origins of the Machado-Joseph Disease Mutation: A Worldwide Haplotype Study. Am. J. Hum. Genet. 2001, 68, 523–528. [Google Scholar] [CrossRef]
- Martins, S.; Calafell, F.; Gaspar, C.; Wong, V.C.N.; Silveira, I.; Nicholson, G.A.; Brunt, E.R.; Tranebjaerg, L.; Stevanin, G.; Hsieh, M.; et al. Asian Origin for the Worldwide-Spread Mutational Event in Machado-Joseph Disease. Arch. Neurol. 2007, 64, 1502–1508. [Google Scholar] [CrossRef]
- Coutinho, P.; Andrade, C. Autosomal dominant system degeneration in Portuguese families of the Azores Islands: A new genetic disorder involving cerebellar, pyramidal, extrapyramidal and spinal cord motor functions. Neurology 1978, 28, 703. [Google Scholar] [CrossRef] [PubMed]
- Giunti, P.; Sweeney, M.G.; Harding, A.E. Detection of the Machado-Joseph disease/spinocerebellar ataxia three trinucleotide repeat expansion in families with autosomal dominant motor disorders, including the Drew family of Walworth. Brain 1995, 118, 1077–1085. [Google Scholar] [CrossRef]
- Schöls, L.; Amoiridis, G.; Büttner, T.; Przuntek, H.; Epplen, J.T.; Riess, O. Autosomal dominant cerebellar ataxia: Phenotypic differences in genetically defined subtypes? Ann. Neurol. 1997, 42, 924–932. [Google Scholar] [CrossRef]
- Riess, O.; Rüb, U.; Pastore, A.; Bauer, P.; Schöls, L. SCA3: Neurological features, pathogenesis and animal models. Cerebellum 2008, 7, 125–137. [Google Scholar] [CrossRef]
- Nascimento-Ferreira, I.; Ferreira, T.; Sousa-Ferreira, L.; Auregan, G.; Onofre, I.; Alves, S.; Dufour, N.; Gould, V.F.C.; Koeppen, A.; Déglon, N.; et al. Overexpression of the autophagic beclin-1 protein clears mutant ataxin-3 and alleviates Machado–Joseph disease. Brain 2011, 134, 1400–1415. [Google Scholar] [CrossRef]
- Maas, R.P.; van Gaalen, J.; Klockgether, T.; van de Warrenburg, B.P. The preclinical stage of spinocerebellar ataxias. Neurology 2015, 85, 96–103. [Google Scholar] [CrossRef]
- Tezenas du Montcel, S.; Durr, A.; Bauer, P.; Figueroa, K.P.; Ichikawa, Y.; Brussino, A.; Forlani, S.; Rakowicz, M.; Schöls, L.; Mariotti, C.; et al. Modulation of the age at onset in spinocerebellar ataxia by CAG tracts in various genes. Brain 2014, 137, 2444–2455. [Google Scholar] [CrossRef]
- van de Warrenburg, B.P.; Hendriks, H.; Dürr, A.; van Zuijlen, M.C.; Stevanin, G.; Camuzat, A.; Sinke, R.J.; Brice, A.; Kremer, B.P. Age at onset variance analysis in spinocerebellar ataxias: A study in a Dutch-French cohort. Ann. Neurol. 2005, 57, 505–512. [Google Scholar] [CrossRef]
- Raposo, M.; Ramos, A.; Bettencourt, C.; Lima, M. Replicating studies of genetic modifiers in Spinocerebellar ataxia type 3: Can homogeneous cohorts aid? Brain 2015, 138, e398. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, P. Doença de Machado-Joseph: Tentativa de Definição. Ph.D. Thesis, Universidade do Porto, Porto, Portugal, 1992. 247p. [Google Scholar]
- Klockgether, T.; Lüdtke, R.; Kramer, B.; Abele, M.; Bürk, K.; Schöls, L.; Riess, O.; Laccone, F.; Boesch, S.; Lopes-Cendes, I.; et al. The natural history of degenerative ataxia: A retrospective study in 466 patients. Brain 1998, 121, 589–600. [Google Scholar] [CrossRef]
- Schmahmann, J.D.; Pierce, S.; MacMore, J.; L'Italien, G.J. Development and Validation of a Patient-Reported Outcome Measure of Ataxia. Mov. Disord. 2021, 36, 2367–2377. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.; Lima, M.; Montiel, R.; Angles, N.; Pires, L.M.L.M.; Abade, A.; Aluja, M.P. Genetic Structure and Origin of Peopling in The Azores Islands (Portugal): The View from mtDNA. Ann. Hum. Genet. 2003, 67, 433–456. [Google Scholar] [CrossRef] [PubMed]
- Montiel, R.; Bettencourt, C.; Silva, C.; Santos, C.; Prata, M.J.; Lima, M. Analysis of Y-chromosome variability and its comparison with mtDNA variability reveals different demographic histories between islands in the Azores Archipelago (Portugal). Ann. Hum. Genet. 2005, 69, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.; Mayer, F.; Coutinho, P.; Abade, A. Prevalence, geographical distribution, and genealogical investigation of Machado-Joseph disease in the islands of the Azores (Portugal). Hum. Biol. 1997, 69, 383–391. [Google Scholar] [PubMed]
- Lima, M.; Mayer, F.M.; Coutinho, P.; Abade, A. Origins of a mutation: Population genetics of Machado-Joseph disease in the Azores (Portugal). Hum. Biol. 1998, 70, 1011–1023. [Google Scholar] [PubMed]
- O’Brien, E.; Jorde, L.B.; Rönnlöf, B.; Fellman, J.O.; Eriksson, A.W. Founder effect and genetic disease in Sottunga, Finland. Am. J. Phys. Anthr. 1988, 77, 335–346. [Google Scholar] [CrossRef]
- Lima, M.; Smith, M.; Silva, C.; Abade, A.; Mayer, F.; Coutinho, P. Natural selection at the MJD locus: Phenotypic diversity, survival and fertility among Machado-Joseph disease patients from the Azores (Portugal). J. Biosoc. Sci. 2001, 33, 361–373. [Google Scholar] [CrossRef]
- Bettencourt, C.; Fialho, R.N.; Santos, C.; Montiel, R.; Armas, J.; Maciel, P.; Lima, M. Segregation distortion of wild-type alleles at the Machado-Joseph disease locus: A study in normal families from the Azores islands (Portugal). J. Hum. Genet. 2008, 53, 333–339. [Google Scholar] [CrossRef]
- Bettencourt, C.; Santos, C.; Kay, T.; Vasconcelos, J.; Lima, M. Analysis of segregation patterns in Machado–Joseph disease pedigrees. J. Hum. Genet. 2008, 53, 920–923. [Google Scholar] [CrossRef]
- Sequeiros, J. Protocolo Geral do Programa Nacional de Teste Preditivo e Aconselhamento Genético na Doença de Machado-Joseph. In O Teste Preditivo Da Doença de Machado-Joseph UnIGENe; Sequeiros, J., Ed.; IBMC: Porto, Portugal, 1996; pp. 123–149. [Google Scholar]
- Lima, M.; Kay, T.; Vasconcelos, J.; Mota-Vieira, L.; Gonzalez, C.; Peixoto, A.; Abade, A.; MacLeod, P.; Graça, R.; Santos, J. Disease Knowledge and Attitudes toward Predictive Testing and Prenatal Diagnosis in Families with Machado-Joseph Disease from the Azores Islands (Portugal). Public Health Genom. 2001, 4, 36–42. [Google Scholar] [CrossRef]
- Gonzalez, C.; Lima, M.; Kay, T.; Silva, C.; Santos, C.; Santos, J. Short-Term Psychological Impact of Predictive Testing for the Machado-Joseph Disease: Depression and Anxiety Levels in Individuals at Risk from the Azores (Portugal). Public Health Genom. 2004, 7, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, C.; Gomes, E.; Kazachkova, N.; Bettencourt, C.; Raposo, M.; Kay, T.T.; MacLeod, P.; Vasconcelos, J.; Lima, M. Psychological Well-Being and Family Satisfaction Levels Five Years After Being Confirmed as a Carrier of the Machado-Joseph Disease Mutation. Genet. Test. Mol. Biomark. 2012, 16, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- Bettencourt, C.; Santos, C.; Montiel, R.; da Costa, M.D.C.P.; Cruz-Morales, P.; Santos, L.R.; Simões, N.; Kay, T.; Vasconcelos, J.; Maciel, P.; et al. Increased transcript diversity: Novel splicing variants of Machado–Joseph Disease gene (ATXN3). Neurogenetics 2010, 11, 193–202. [Google Scholar] [CrossRef]
- Bettencourt, C.; Raposo, M.; Kazachkova, N.; Santos, C.; Kay, T.; Vasconcelos, J.; Maciel, P.; Donis, K.C.; Saraiva-Pereira, M.L.; Jardim, L.B.; et al. Sequence analysis of 5′ regulatory regions of Machado-Joseph disease gene (ATXN3). Cerebellum. 2012, 11, 1045–1050. [Google Scholar] [CrossRef]
- Melo, A.R.V.; Raposo, M.; Ventura, M.; Martins, S.; Pavão, S.; Alonso, I.; Bettencourt, C.; Lima, M. Genetic Variation in ATXN3 (Ataxin-3) 3′UTR: Insights into the Downstream Regulatory Elements of the Causative Gene of Machado-Joseph Disease/Spinocerebellar Ataxia Type 3. Cerebellum 2022. [Google Scholar] [CrossRef] [PubMed]
- Kazachkova, N.; Raposo, M.; Ramos, A.; Montiel, R.; Lima, M. Promoter Variant Alters Expression of the Autophagic BECN1 Gene: Implications for Clinical Manifestations of Machado-Joseph Disease. Cerebellum 2017, 16, 957–963. [Google Scholar] [CrossRef]
- Raposo, M.; Bettencourt, C.; Ramos, A.; Kazachkova, N.; Vasconcelos, J.; Kay, T.; Bruges-Armas, J.; Lima, M. Promoter Variation and Expression Levels of Inflammatory Genes IL1A, IL1B, IL6 and TNF in Blood of Spinocerebellar Ataxia Type 3 (SCA3) Patients. Neuromol. Med. 2017, 19, 41–45. [Google Scholar] [CrossRef]
- Raposo, M.; Bettencourt, C.; Maciel, P.; Gao, F.; Ramos, A.; Kazachkova, N.; Vasconcelos, J.; Kay, T.; Rodrigues, A.J.; Bettencourt, B.; et al. Novel candidate blood-based transcriptional biomarkers of machado-joseph disease. Mov. Disord. 2015, 30, 968–975. [Google Scholar] [CrossRef]
- Raposo, M.; Ramos, A.; Santos, C.; Kazachkova, N.; Teixeira, B.; Bettencourt, C.; Lima, M. Accumulation of Mitochondrial DNA Common Deletion Since The Preataxic Stage of Machado-Joseph Disease. Mol. Neurobiol. 2019, 56, 119–124. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, M.; Raposo, M.; Ferreira, A.; Melo, A.R.V.; Pavão, S.; Medeiros, F.; Teves, L.; Gonzalez, C.; Lemos, J.; Pires, P.; et al. The Homogeneous Azorean Machado-Joseph Disease Cohort: Characterization and Contributions to Advances in Research. Biomedicines 2023, 11, 247. https://doi.org/10.3390/biomedicines11020247
Lima M, Raposo M, Ferreira A, Melo ARV, Pavão S, Medeiros F, Teves L, Gonzalez C, Lemos J, Pires P, et al. The Homogeneous Azorean Machado-Joseph Disease Cohort: Characterization and Contributions to Advances in Research. Biomedicines. 2023; 11(2):247. https://doi.org/10.3390/biomedicines11020247
Chicago/Turabian StyleLima, Manuela, Mafalda Raposo, Ana Ferreira, Ana Rosa Vieira Melo, Sara Pavão, Filipa Medeiros, Luís Teves, Carlos Gonzalez, João Lemos, Paula Pires, and et al. 2023. "The Homogeneous Azorean Machado-Joseph Disease Cohort: Characterization and Contributions to Advances in Research" Biomedicines 11, no. 2: 247. https://doi.org/10.3390/biomedicines11020247
APA StyleLima, M., Raposo, M., Ferreira, A., Melo, A. R. V., Pavão, S., Medeiros, F., Teves, L., Gonzalez, C., Lemos, J., Pires, P., Lopes, P., Valverde, D., Gonzalez, J., Kay, T., & Vasconcelos, J. (2023). The Homogeneous Azorean Machado-Joseph Disease Cohort: Characterization and Contributions to Advances in Research. Biomedicines, 11(2), 247. https://doi.org/10.3390/biomedicines11020247





