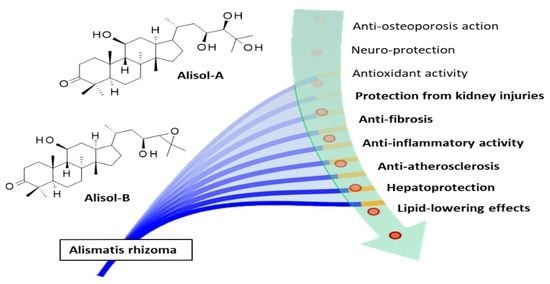
Pharmacological Properties and Molecular Targets of Alisol Triterpenoids from Alismatis Rhizoma
Abstract
1. Introduction
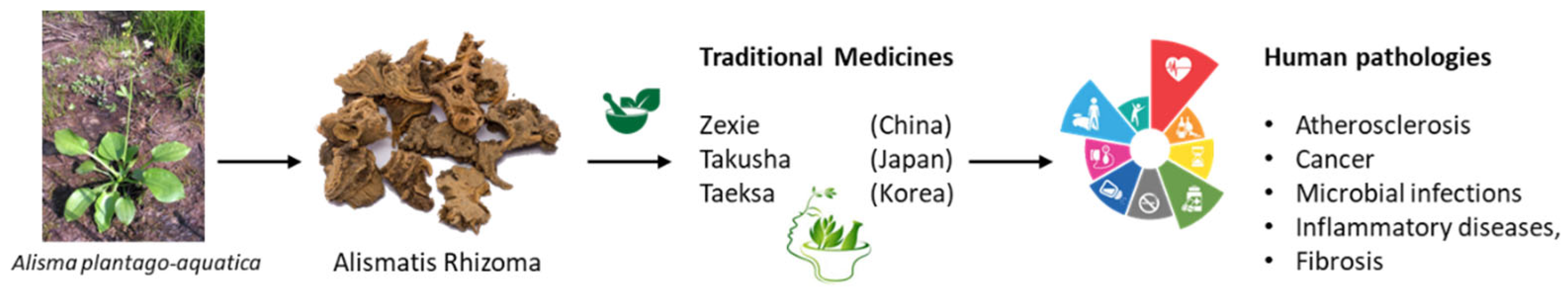
2. TCM Made from Alismatis Rhizoma
3. Alismatis Rhizoma Extracts: Phytoconstituents and Pharmacological Effects
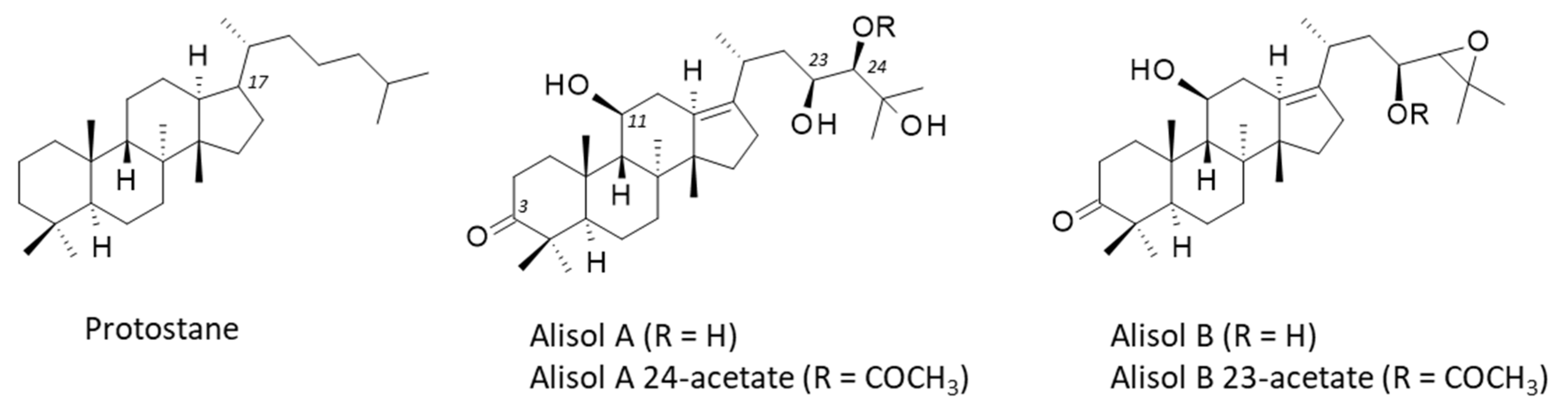
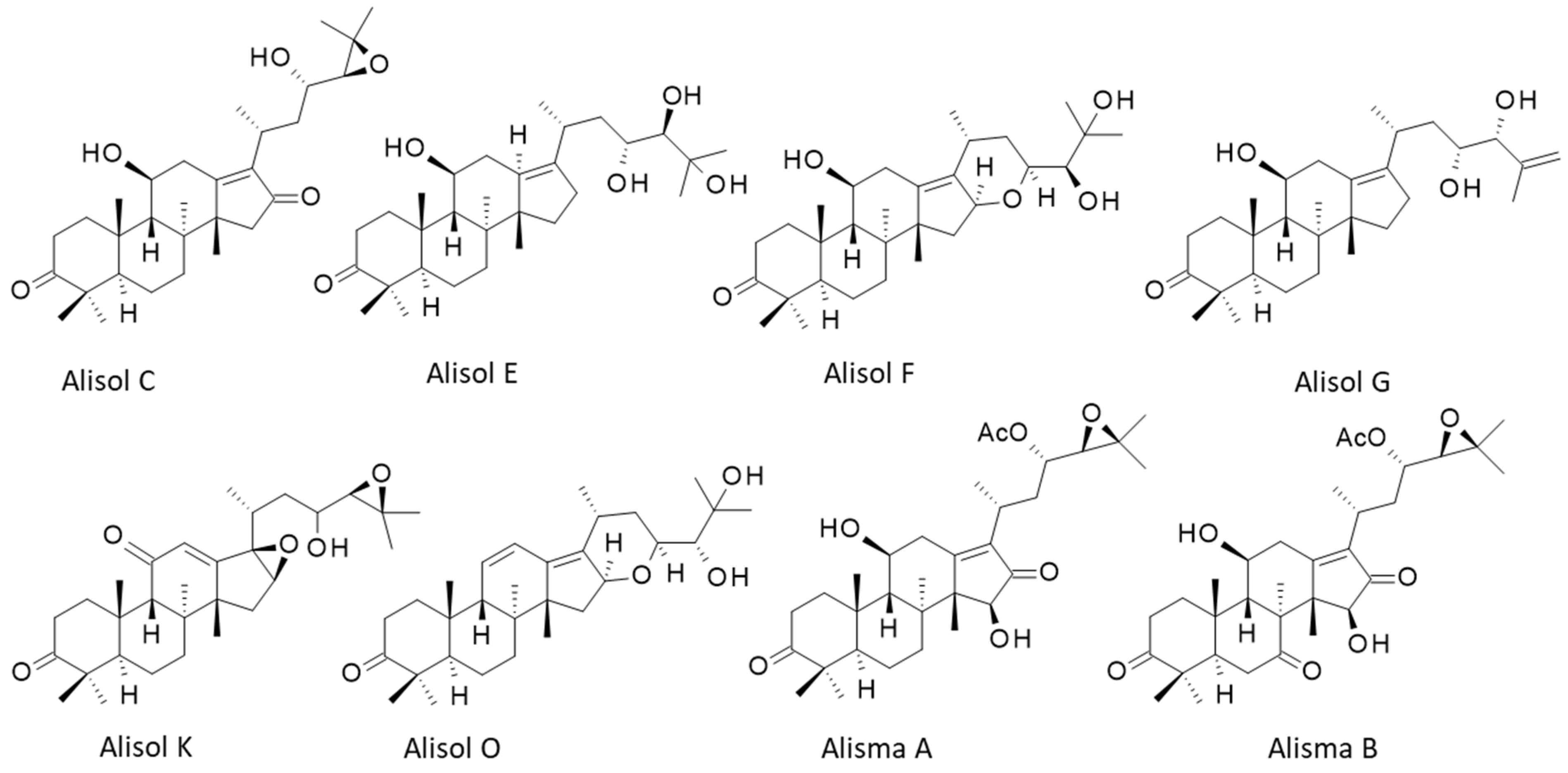
4. Alisols and Related Terpenoids
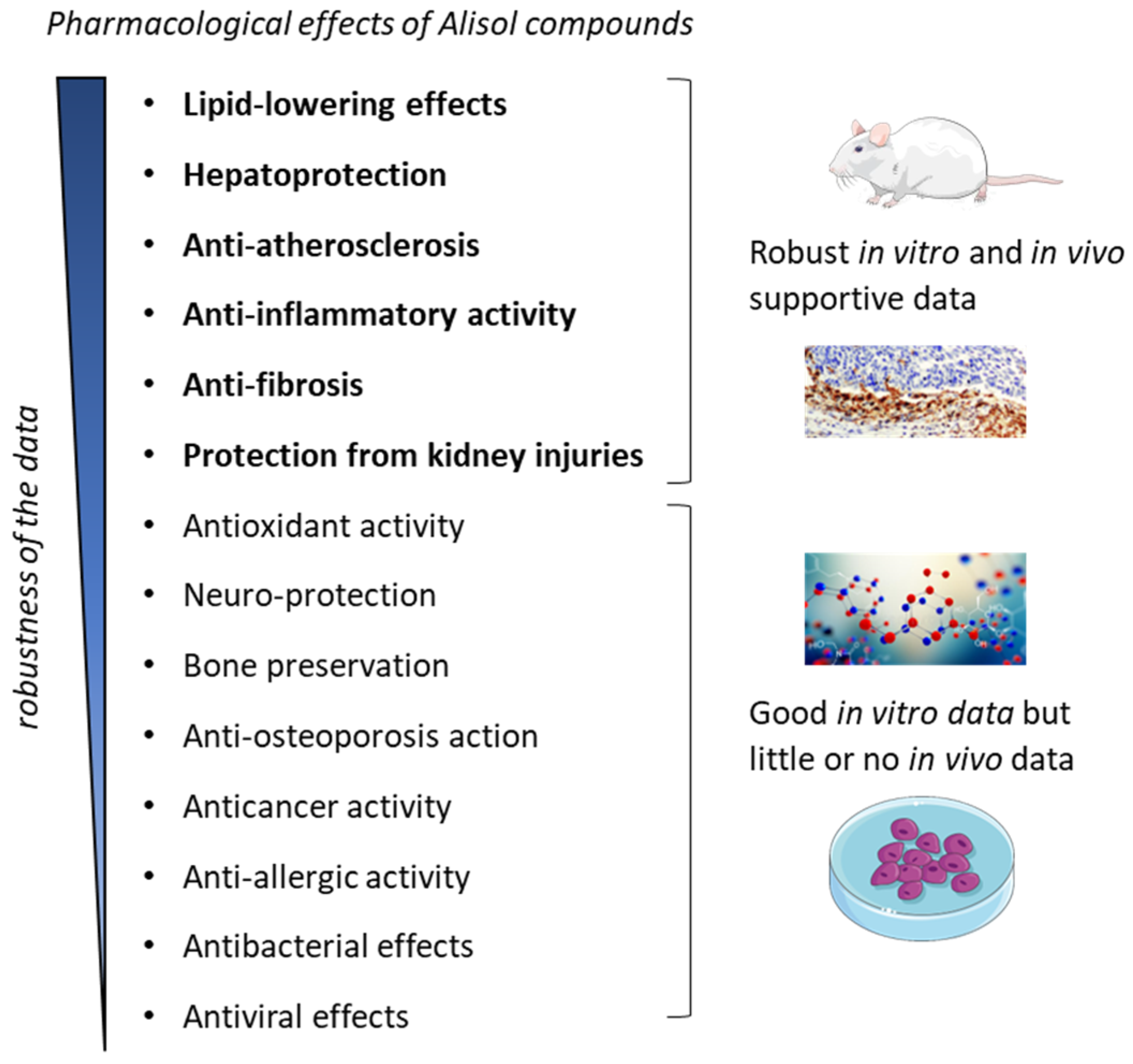
5. Pharmacological Properties of Alisols
5.1. Lipid-Lowering Effects and Hepatoprotection
5.2. Treatment of Atherosclerosis
5.3. Anti-Inflammatory Activity
5.4. Protection against Fibrosis and Kidney Injuries
5.5. Antioxidant Activity and Neuro-Protection
5.6. Bone Preservation and Prevention of Osteoporosis
5.7. Anticancer Activity
5.8. Anti-Allergic Activity
5.9. Antibacterial and Antiviral Effects
6. Discussion and Perspectives
7. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, S.S.; Wei, S.H.; Zhu, J.J.; Zhang, W.W.; Jiao, S.; Guo, J.; Yan, L.H.; Wang, Z.M. Herbal textural research, morphologic characteristics and DNA barcoding of botanical origins of Alismatis Rhizoma. Zhongguo Zhong Yao Za Zhi 2020, 45, 1536–1544. [Google Scholar] [PubMed]
- Liu, S.S.; Guo, J.; Li, Z.A.; Tian, S.S.; Zhu, J.J.; Yan, L.H.; Wang, Z.M.; Gao, L. Advances in studies on chemical compositions of Alismatis Rhizoma and their biological activities. Zhongguo Zhong Yao Za Zhi 2020, 45, 1578–1595. [Google Scholar] [PubMed]
- Feng, L.; Liu, T.T.; Huo, X.K.; Tian, X.G.; Wang, C.; Lv, X.; Ning, J.; Zhao, W.Y.; Zhang, B.J.; Sun, C.P.; et al. Alisma genus: Phytochemical constituents, biosynthesis, and biological activities. Phytother. Res. 2021, 35, 1872–1886. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jin, Q.; Wu, W.; Jin, X.; An, Y.; Liu, C.; Wei, W.; Li, Z.; Yao, C.; Yao, S.; et al. “Force iteration molecular designing” strategy for the systematic characterization and discovery of new protostane triterpenoids from Alisma Rhizoma by UHPLC/LTQ-Orbitrap-MS. Anal. Bioanal. Chem. 2021, 413, 1749–1764. [Google Scholar] [CrossRef]
- Zhao, M.; Gödecke, T.; Gunn, J.; Duan, J.A.; Che, C.T. Protostane and fusidane triterpenes: A mini-review. Molecules 2013, 18, 4054–4080. [Google Scholar] [CrossRef] [PubMed]
- Rong, T.; Chunchun, Z.; Wei, G.; Yuchen, G.; Fei, X.; Tao, L.; Yuanyuan, J.; Chenbin, W.; Wenda, X.; Wenqing, W. Proteomic insights into protostane triterpene biosynthesis regulatory mechanism after MeJA treatment in Alisma orientale (Sam.) Juz. Biochim. Biophys. Acta Proteins Proteom. 2021, 1869, 140671. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Gu, W.; Tian, R.; Xu, F.; Han, Y.; Ji, Y.; Li, T.; Zhu, Y.; Lang, P.; Wu, W. Comparative analysis of the structure and function of rhizosphere microbiome of the Chinese medicinal herb Alisma in different regions. Arch. Microbiol. 2022, 204, 448. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.M.; Tian, S.S.; Liu, S.S.; Qu, Q.; Zhao, J.Y.; Yan, L.H.; Wang, Z.M. Recommendations on quality standards of Alismatis Rhizoma in Chinese Pharmacopoeia (2020 edition). Zhongguo Zhong Yao Za Zhi 2020, 45, 1566–1577. [Google Scholar] [PubMed]
- Liu, X.Y.; Li, Y.J.; Jiang, H.W.; Li, H.P.; Xiao, L.Y.; Zhao, Y.S. Comparison between Ye Tianshi and Xue Shengbai in treatment of diarrfea with damp-heat. Zhongguo Zhong Yao Za Zhi 2018, 43, 1720–1725. [Google Scholar] [PubMed]
- Xie, Z.; Li, E.W.; Gao, G.; Du, Y.; Wang, M.; Wang, H.; Wang, P.; Qiao, Y.; Su, Y.; Xu, J.; et al. Zexie Tang targeting FKBP38/mTOR/SREBPs pathway improves hyperlipidemia. J. Ethnopharmacol. 2022, 290, 115101. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, D.; Cheng, B.; Liu, G.; Zhang, Y.X.; Zhou, W.X. Active fraction combination from Liuwei Dihuang decoction (LW-AFC) ameliorates corticosterone-induced long-term potentiation (LTP) impairment in mice in vivo. J. Ethnopharmacol. 2019, 236, 147–154. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Xiao, C.X.; Wang, C.J.; Liang, J.; Cheng, Q.Q.; Zhang, L.; Zhou, B.J.; Zhou, H. Discovery of Quality Markers in Hugan Qingzhi Formula by Integrating a Lipid-Lowering Bioassay with UHPLC-QQQ-MS/MS. Evid.-Based Complement. Alternat. Med. 2020, 2020, 1594350. [Google Scholar] [CrossRef]
- Xu, L.; Liu, Y.; Wu, H.; Wu, H.; Liu, X.; Zhou, A. Rapid identification of chemical profile in Gandou decoction by UPLC-Q-TOF-MSE coupled with novel informatics UNIFI platform. J. Pharm. Anal. 2020, 10, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Geng, H.; Wu, P.; Dong, J.; Li, H.; Xu, C.; Li, B.; Han, Y.; Cheng, N. Gandou decoction, a Chinese medicinal formula, in the treatment of hepatic injury by Wnt/β-catenin pathway regulation in models of Wilson disease. Ann. Palliat. Med. 2020, 9, 2872–2885. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, G.; Zhou, Y.; Yin, D.; Tao, C.; Han, L.; Yue, X.; Pan, Y.; Yao, Y.; Peng, D.; et al. The difference between blood-associated and water-associated herbs of Danggui-Shaoyao San in theory of TCM, based on serum pharmacochemistry. Biomed. Chromatogr. 2016, 30, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Ding, R.R.; Chen, W.; Guo, C.Y.; Liao, W.T.; Yang, X.; Liao, F.E.; Lin, J.M.; Mei, H.F.; Zeng, Y. Dangguishaoyao-San attenuates LPS-induced neuroinflammation via the TLRs/NF-κB signaling pathway. Biomed. Pharmacother. 2018, 105, 187–194. [Google Scholar] [CrossRef]
- Yun, W.J.; Yao, Z.H.; Fan, C.L.; Qin, Z.F.; Tang, X.Y.; Gao, M.X.; Dai, Y.; Yao, X.S. Systematic screening and characterization of Qi-Li-Qiang-Xin capsule-related xenobiotics in rats by ultra-performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2018, 1090, 56–64. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Liu, Y.; Yang, K.; Zou, Z.; Fan, C.; Yao, Z.; Dai, Y.; Li, K.; Chen, J.; Yao, X. The discovery of Q-markers of Qiliqiangxin Capsule, a traditional Chinese medicine prescription in the treatment of chronic heart failure, based on a novel strategy of multi-dimensional “radar chart” mode evaluation. Phytomedicine 2021, 82, 153443. [Google Scholar] [CrossRef]
- Xu, F.; Hou, T.; Shen, A.; Jin, H.; Xiao, Y.; Yu, W.; Li, X.; Wang, J.; Liu, Y.; Liang, X. Mechanism deconvolution of Qing Fei Pai Du decoction for treatment of Coronavirus Disease 2019 (COVID-19) by label-free integrative pharmacology assays. J. Ethnopharmacol. 2021, 280, 114488. [Google Scholar] [CrossRef] [PubMed]
- Kubota, K.; Fukue, H.; Sato, H.; Hashimoto, K.; Fujikane, A.; Moriyama, H.; Watanabe, T.; Katsurabayashi, S.; Kainuma, M.; Iwasaki, K. The Traditional Japanese Herbal Medicine Hachimijiogan Elicits Neurite Outgrowth Effects in PC12 Cells and Improves Cognitive in AD Model Rats via Phosphorylation of CREB. Front. Pharmacol. 2017, 8, 850. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.H.; Kwon, S.; Jung, W.S.; Moon, S.K. Herbal Medicine, Oreongsan for Recurrent Chronic Subdural Hematoma: A Case Report. Explore 2017, 13, 139–141. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.; Jin, C.; Cho, K.H. Oreongsan, an herbal medicine prescription developed as a new alternative treatment in patients with chronic subdural hematoma: A narrative review. Integr. Med. Res. 2019, 8, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Seo, C.S.; Lee, M.Y. Simultaneous Determination of Fourteen Marker Compounds in the Traditional Herbal Prescription, Geumgwesingihwan, Using Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry. Molecules 2022, 27, 3890. [Google Scholar] [CrossRef]
- Hattori, T.; Nishimura, H.; Makino, B.; Shindo, S.; Kawamura, H. Sairei-to inhibits the production of endothelin-1 by nephritic glomeruli(2): Alisols, possible candidates as active compounds. Nihon Jinzo Gakkai Shi 1998, 40, 33–41. [Google Scholar] [PubMed]
- Wang, Z.; Zhu, L.; Huang, Y.; Peng, L. Successful treatment of immune-related cystitis by Chai-Ling-Tang (Sairei-To) in a gastric carcinoma patient: Case report and literature review. Explore 2022, S1550–S8307. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Zhong, L.L.; Cao, Y.; Chen, W.; Cheng, Y.; Lin, X.F.; Bian, Z.X.; Lu, A.P. Efficacy and safety of Yirui capsule in patients with hyperlipidemia: Study protocol for a multicenter, randomized, double-blind, placebo-controlled trial. Trials 2016, 17, 291. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xia, Z.; Rong, S.; Gao, H.; Yang, W.; Li, J.; Ma, C.; Deng, Q.; Huang, Q.; Xiao, L.; et al. Yirui Capsules Alleviate Atherosclerosis by Improving the Lipid Profile and Reducing Inflammation in Apolipoprotein E-Deficient Mice. Nutrients 2018, 10, 142. [Google Scholar] [CrossRef] [PubMed]
- Tai, Y.N.; Weng, Y.H.; Zhang, S.P.; Xu, W.; Li, X.Y.; Lin, Q.Q.; Chu, K.D.; Wu, S.S. Determination of seven ingredients of different grades Alismatis Rhizoma by QAMS method. Zhongguo Zhong Yao Za Zhi 2019, 44, 2292–2307. [Google Scholar] [PubMed]
- Wu, J.; Yang, W.; Pan, H.; Yao, S.; Wu, W.; Guo, D. Geographic impact evaluation of the quality of Alismatis Rhizoma by untargeted metabolomics and quantitative assay. J. Sep. Sci. 2018, 41, 839–846. [Google Scholar] [CrossRef]
- Yang, N.; Dong, Y.Q.; Wu, M.F.; Li, S.Z.; Yu, H.X.; Yang, S.S. Establishing a rapid classification and identification method for the major triterpenoids of Alisma orientale. Phytochem. Anal. 2020, 31, 384–394. [Google Scholar] [CrossRef]
- Dai, M.; Li, S.; Shi, Q.; Xiang, X.; Jin, Y.; Wei, S.; Zhang, L.; Yang, M.; Song, C.; Huang, R.; et al. Changes in Triterpenes in Alismatis rhizoma after Processing Based on Targeted Metabolomics Using UHPLC-QTOF-MS/MS. Molecules 2021, 27, 185. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.S.; Sheng, W.L.; Li, Y.; Zhang, S.S.; Zhu, J.J.; Gao, H.M.; Yan, L.H.; Wang, Z.M.; Gao, L.; Zhang, M. Chemical constituents from Alismatis Rhizoma and their anti-inflammatory activities in vitro and in vivo. Bioorg. Chem. 2019, 92, 103226. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, S.; Li, J.; Cheng, Z.; Feng, Y.; Ouyang, H.; Du, Z.; Jiang, H. Comprehensive metabolic profiling of Alismatis Rhizoma triterpenes in rats based on characteristic ions and a triterpene database. J. Pharm. Anal. 2021, 11, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Li, X.; Lin, N.; Zhang, X.; Huang, X.; Wu, T.; Tai, Y.; Chen, S.; Wu, C.H.; Huang, M.; et al. Pharmacokinetics and tissue distribution of five major triterpenoids after oral administration of Rhizoma Alismatis extract to rats using ultra high-performance liquid chromatography-tandem mass spectrometry. J. Pharm. Biomed. Anal. 2017, 146, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, M.; Yoshinaga, S.; Kashima, Y.; Nakahashi, H.; Hara, N.; Nakagawa, H.; Usami, A. Chemical Composition and Characteristic Odor Compounds in Essential Oil from Alismatis Rhizoma (Tubers of Alisma orientale). J. Oleo. Sci. 2016, 65, 91–97. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Feng, Y.L.; Chen, H.; Tian, T.; Chen, D.Q.; Zhao, Y.Y.; Lin, R.C. Diuretic and anti-diuretic activities of the ethanol and aqueous extracts of Alismatis rhizoma. J. Ethnopharmacol. 2014, 154, 386–390. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.Y.; Lin, N.; Zhao, W.L.; Huang, X.Q.; Chen, Y.; Huang, M.Q.; Xu, W.; Wu, S.S. Diuretic Activity of Compatible Triterpene Components of Alismatis rhizoma. Molecules 2017, 22, 1459. [Google Scholar] [CrossRef]
- Tian, T.; Chen, H.; Zhao, Y.Y. Traditional uses, phytochemistry, pharmacology, toxicology and quality control of Alisma orientale (Sam.) Juzep: A review. J. Ethnopharmacol. 2014, 158, 373–387. [Google Scholar] [CrossRef]
- Zhang, L.L.; Xu, W.; Xu, Y.L.; Chen, X.; Huang, M.; Lu, J.J. Therapeutic potential of Rhizoma Alismatis: A review on ethnomedicinal application, phytochemistry, pharmacology, and toxicology. Ann. N. Y. Acad. Sci. 2017, 1401, 90–101. [Google Scholar] [CrossRef]
- Lee, M.J.; Jung, H.K.; Lee, K.H.; Jang, J.H.; Sim, M.O.; Seong, T.G.; Ahn, B.K.; Shon, J.H.; Ham, S.H.; Cho, H.W.; et al. A 90-Day Repeated Oral Dose Toxicity Study of Alismatis Rhizoma Aqueous Extract in Rats. Toxicol. Res. 2019, 35, 191–200. [Google Scholar] [CrossRef]
- Wang, C.F.; Ma, L.; Hou, X.F.; Chen, H.F.; Feng, L.; Jia, X.B. Study on nephrotoxicity of ethanol extract from Alismatis Rhizoma in rats and its molecular mechanism. Zhongguo Zhong Yao Za Zhi 2016, 41, 3432–3438. [Google Scholar] [PubMed]
- Wang, C.F.; Ma, L.; Feng, L.; Yin, M.R.; Gu, J.F.; Jia, X.B. Evaluation of nephrotoxicity induced by total terpenoids from Alismatis Rhizoma on HK-2 cells in vitro and its induction of apoptosis. Zhongguo Zhong Yao Za Zhi 2016, 41, 490–497. [Google Scholar] [PubMed]
- Yang, Y.; Zhang, D.M.; Liu, J.H.; Hu, L.S.; Xue, Q.C.; Ding, X.Q.; Kong, L.D. Wuling San protects kidney dysfunction by inhibiting renal TLR4/MyD88 signaling and NLRP3 inflammasome activation in high fructose-induced hyperuricemic mice. J. Ethnopharmacol. 2015, 169, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Sun, H.; Li, X.; Yan, J.; Peng, Z.; You, Y.; Zhang, L.; Chen, J. Effects of Alismatis Rhizoma and Rhizoma Smilacis Glabrae Decoction on Hyperuricemia in Rats. Evid.-Based Complement. Alternat. Med. 2019, 2019, 4541609. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Lu, L.; Zhang, Y.; Zhang, B. Study on discriminating nephrotoxic components in Zexie. Zhongguo Zhong Yao Za Zhi 2011, 36, 758–761. [Google Scholar] [PubMed]
- Tian, R.; Gu, W.; Gu, Y.; Geng, C.; Xu, F.; Wu, Q.; Chao, J.; Xue, W.; Zhou, C.; Wang, F. Methyl jasmonate promote protostane triterpenes accumulation by up-regulating the expression of squalene epoxidases in Alisma orientale. Sci. Rep. 2019, 9, 18139. [Google Scholar] [CrossRef]
- Wang, P.; Song, T.; Shi, R.; He, M.; Wang, R.; Lv, J.; Jiang, M. Triterpenoids From Alisma Species: Phytochemistry, Structure Modification, and Bioactivities. Front. Chem. 2020, 8, 363. [Google Scholar] [CrossRef]
- Murata, T.; Imai, Y.; Hirata, T.; Miyamoto, M. Biological-active trieterpenes of Alismatis rhizoma. I. Isolation of the alisols. Chem. Pharm. Bull. 1970, 18, 1347–1353. [Google Scholar] [CrossRef]
- Imai, Y.; Matsumura, H.; Aramaki, Y. Hypocholesterolemic effect of alisol A-24-monoacetate and its related compounds in rats. Jpn. J. Pharmacol. 1970, 20, 222–228. [Google Scholar] [CrossRef][Green Version]
- Zhao, Z.; Deng, Z.T.; Huang, S.; Ning, M.; Feng, Y.; Shen, Y.; Zhao, Q.S.; Leng, Y. Alisol B Alleviates Hepatocyte Lipid Accumulation and Lipotoxicity via Regulating RARα-PPARγ-CD36 Cascade and Attenuates Non-Alcoholic Steatohepatitis in Mice. Nutrients 2022, 14, 2411. [Google Scholar] [CrossRef]
- Chang, X.Y.; Wu, J.S.; Zhang, F.Q.; Li, Z.Z.; Jin, W.Y.; Wang, J.X.; Wang, W.H.; Shi, Y. A Strategy for Screening the Lipid-Lowering Components in Alismatis Rhizoma Decoction Based on Spectrum-Effect Analysis. J. Anal. Methods Chem. 2022, 2022, 2363242. [Google Scholar] [CrossRef] [PubMed]
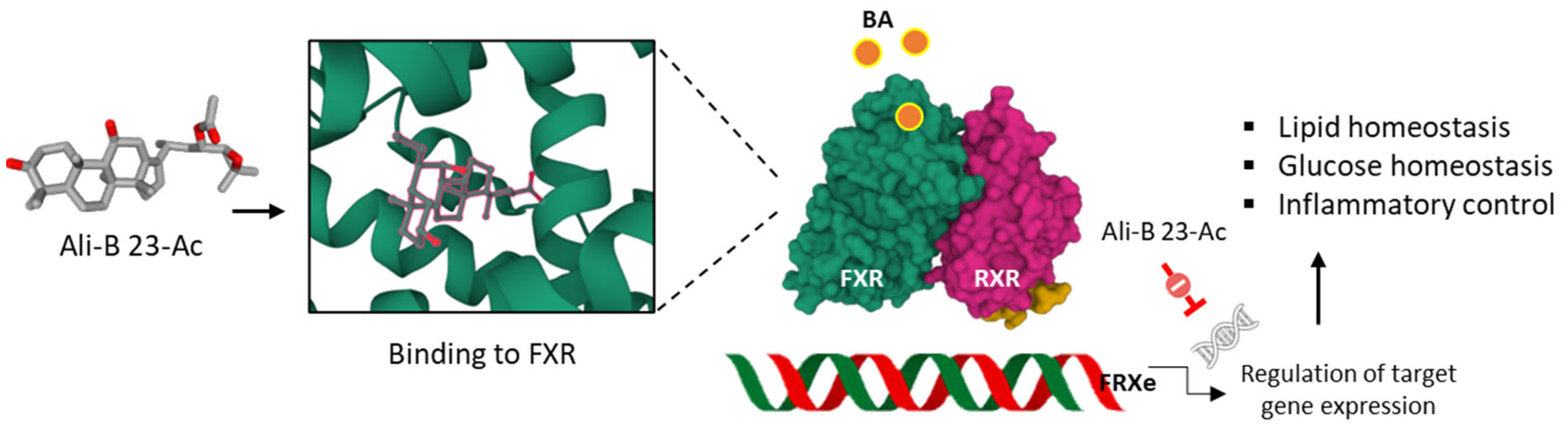
- Fu, Y.; Feng, H.; Ding, X.; Meng, Q.H.; Zhang, S.R.; Li, J.; Chao, Y.; Ji, T.T.; Bi, Y.H.; Zhang, W.W.; et al. Alisol B 23-acetate adjusts bile acid metabolisim via hepatic FXR-BSEP signaling activation to alleviate atherosclerosis. Phytomedicine 2022, 101, 154120. [Google Scholar] [CrossRef] [PubMed]
- Luan, Z.L.; Ming, W.H.; Sun, X.W.; Zhang, C.; Zhou, Y.; Zheng, F.; Yang, Y.L.; Guan, Y.F.; Zhang, X.Y. A naturally occurring FXR agonist, alisol B 23-acetate, protects against renal ischemia-reperfusion injury. Am. J. Physiol. Renal Physiol. 2021, 321, F617–F628. [Google Scholar] [CrossRef]
- Meng, Q.; Duan, X.P.; Wang, C.Y.; Liu, Z.H.; Sun, P.Y.; Huo, X.K.; Sun, H.J.; Peng, J.Y.; Liu, K.X. Alisol B 23-acetate protects against non-alcoholic steatohepatitis in mice via farnesoid X receptor activation. Acta Pharmacol. Sin. 2017, 38, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Kanno, Y.; Yatsu, T.; Yamashita, N.; Zhao, S.; Li, W.; Imai, M.; Kashima, M.; Inouye, Y.; Nemoto, K.; Koike, K. Alisol B 23-acetate from the rhizomes of Alisma orientale is a natural agonist of the human pregnane X receptor. Phytomedicine 2017, 26, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Chen, X.; Wang, C.; Liu, Q.; Sun, H.; Sun, P.; Peng, J.; Liu, K. Alisol B 23-acetate promotes liver regeneration in mice after partial hepatectomy via activating farnesoid X receptor. Biochem. Pharmacol. 2014, 92, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Chen, X.; Wang, C.; Liu, Q.; Sun, H.; Sun, P.; Huo, X.; Liu, Z.; Yao, J.; Liu, K. Protective Effects of Alisol B 23-Acetate Via Farnesoid X Receptor-Mediated Regulation of Transporters and Enzymes in Estrogen-Induced Cholestatic Liver Injury in Mice. Pharm. Res. 2015, 32, 3688–3698. [Google Scholar] [CrossRef]
- Meng, Q.; Chen, X.L.; Wang, C.Y.; Liu, Q.; Sun, H.J.; Sun, P.Y.; Huo, X.K.; Liu, Z.H.; Yao, J.H.; Liu, K.X. Alisol B 23-acetate protects against ANIT-induced hepatotoxity and cholestasis, due to FXR-mediated regulation of transporters and enzymes involved in bile acid homeostasis. Toxicol. Appl. Pharmacol. 2015, 283, 178–186. [Google Scholar] [CrossRef]
- Meng, Q.; Chen, X.; Wang, C.; Liu, Q.; Sun, H.; Sun, P.; Huo, X.; Liu, Z.; Liu, K. Protective effects of alisol B 23-acetate from edible botanical Rhizoma alismatis against carbon tetrachloride-induced hepatotoxicity in mice. Food Funct. 2015, 6, 1241–1250. [Google Scholar] [CrossRef]
- Zhou, X.; Ren, Q.; Wang, B.; Fang, G.; Ling, Y.; Li, X. Alisol A 24-Acetate Isolated from the Alismatis Rhizoma Improves Hepatic Lipid Deposition in Hyperlipidemic Mice by ABCA1/ABCG1 Pathway. J. Nanosci. Nanotechnol. 2019, 19, 5496–5502. [Google Scholar] [CrossRef]
- Chen, J.X.; Li, H.Y.; Li, T.T.; Fu, W.C.; Du, X.; Liu, C.H.; Zhang, W. Alisol A-24-acetate promotes glucose uptake via activation of AMPK in C2C12 myotubes. BMC Complement. Med. Ther. 2020, 20, 22. [Google Scholar] [CrossRef]
- Lou, H.X.; Fu, W.C.; Chen, J.X.; Li, T.T.; Jiang, Y.Y.; Liu, C.H.; Zhang, W. Alisol A 24-acetate stimulates lipolysis in 3 T3-L1 adipocytes. BMC Complement. Med. Ther. 2021, 21, 128. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.; Jang, E.; Lee, J.H. Pharmacological Activities of Alisma orientale against Nonalcoholic Fatty Liver Disease and Metabolic Syndrome: Literature Review. Evid. Based Complement. Alternat. Med. 2019, 2019, 2943162. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Long, J.; Chen, W.; Sun, Y.; Zhou, L.; Zhang, L.; Zeng, H.; Yuan, D. Alisol B 23-acetate, a new promoter for cholesterol efflux from dendritic cells, alleviates dyslipidemia and inflammation in advanced atherosclerotic mice. Int. Immunopharmacol. 2021, 99, 107956. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.C.; Fu, Y.; Bi, Y.H.; Zhang, W.W.; Li, J.; Ji, T.; Chao, Y.; Meng, Q.H.; Chen, Q.; Ma, M.H.; et al. Alisol B 23-acetate activates ABCG5/G8 in the jejunum via the LXRα/ACAT2 pathway to relieve atherosclerosis in ovariectomized ApoE-/- mice. Aging 2020, 12, 25744–25766. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Chao, Y.; Zhang, W.; Zhang, Y.; Bi, Y.; Fu, Y.; Cai, D.; Meng, Q.; Li, Y.; Bian, H. Activation of estrogen receptor α (ERα) is required for Alisol B23-acetate to prevent post-menopausal atherosclerosis and reduced lipid accumulation. Life Sci. 2020, 258, 118030. [Google Scholar] [CrossRef]
- Lin, C.; Li, J.; Wu, C.; Bao, J. Identifying selective agonists targeting LXRβ from terpene compounds of alismatis rhizoma. J. Mol. Model. 2021, 27, 91. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, B.; Song, D.; Xi, J.; Hao, W.; Yuan, J.; Gao, C.; Cui, Z.; Cheng, Z. Alisol A Alleviates Arterial Plaque by Activating AMPK/SIRT1 Signaling Pathway in apoE-Deficient Mice. Front. Pharmacol. 2020, 11, 580073. [Google Scholar] [CrossRef]
- Zeng, L.; Tang, W.; Yin, J.; Feng, L.; Li, Y.; Yao, X.; Zhou, B. Alisol A 24-Acetate Prevents Hepatic Steatosis and Metabolic Disorders in HepG2 Cells. Cell. Physiol. Biochem. 2016, 40, 453–464. [Google Scholar] [CrossRef]
- Ho, C.; Gao, Y.; Zheng, D.; Liu, Y.; Shan, S.; Fang, B.; Zhao, Y.; Song, D.; Zhang, Y.; Li, Q. Alisol A attenuates high-fat-diet-induced obesity and metabolic disorders via the AMPK/ACC/SREBP-1c pathway. J. Cell. Mol. Med. 2019, 23, 5108–5118. [Google Scholar] [CrossRef]
- Wei, W.; Zhou, X.M.; Wang, Y.C.; Lin, Z.C.; Xue, J.H. Effect of alisol A 24-acetate on proliferation of aorta smooth muscle cells in rats induced by ox-LDL. Zhongguo Zhong Yao Za Zhi 2018, 43, 2147–2152. [Google Scholar]
- Xu, F.; Yu, H.; Lu, C.; Chen, J.; Gu, W. The Cholesterol-Lowering Effect of Alisol Acetates Based on HMG-CoA Reductase and Its Molecular Mechanism. Evid.-Based Complement. Alternat. Med. 2016, 2016, 4753852. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Jin, S.; Song, C.; Chen, C.; Zhang, Y.; Xiang, Y.; Xu, Y.; Feng, Y.; Wan, Q.; Jiang, H. The metabolic change of serum lysophosphatidylcholines involved in the lipid lowering effect of triterpenes from Alismatis rhizoma on high-fat diet induced hyperlipidemia mice. J. Ethnopharmacol. 2016, 177, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.; Wei, Y.; Wang, M.; Tao, J.; Ouyang, H.; Du, Z.; Li, S.; Jiang, H. Network pharmacology combined with metabolomics and lipidomics to reveal the hypolipidemic mechanism of Alismatis rhizoma in hyperlipidemic mice. Food Funct. 2022, 13, 4714–4733. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, L.; Du, Z.; Jin, S.; Song, C.; Jia, S.; Zhang, Y.; Jiang, H. Identification of the lipid-lowering component of triterpenes from Alismatis rhizoma based on the MRM-based characteristic chemical profiles and support vector machine model. Anal. Bioanal. Chem. 2019, 411, 3257–3268. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Lu, C.; Wu, Q.; Gu, W.; Chen, J.; Fang, F.; Zhao, B.; Du, W.; You, M. Studies on the lipid-regulating mechanism of alisol-based compounds on lipoprotein lipase. Bioorg. Chem. 2018, 80, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Chen, J.; Zhang, Y.; Wu, Q.; Shen, Y.; Gu, W.; Liu, S.; Lu, C.; Liao, H.; Bao, K. Molecular insight into the mechanism of lipid regulating effect of Alisma orientalis based on ACAT. Int. J. Biol. Macromol. 2020, 158, 1141–1162. [Google Scholar] [CrossRef]
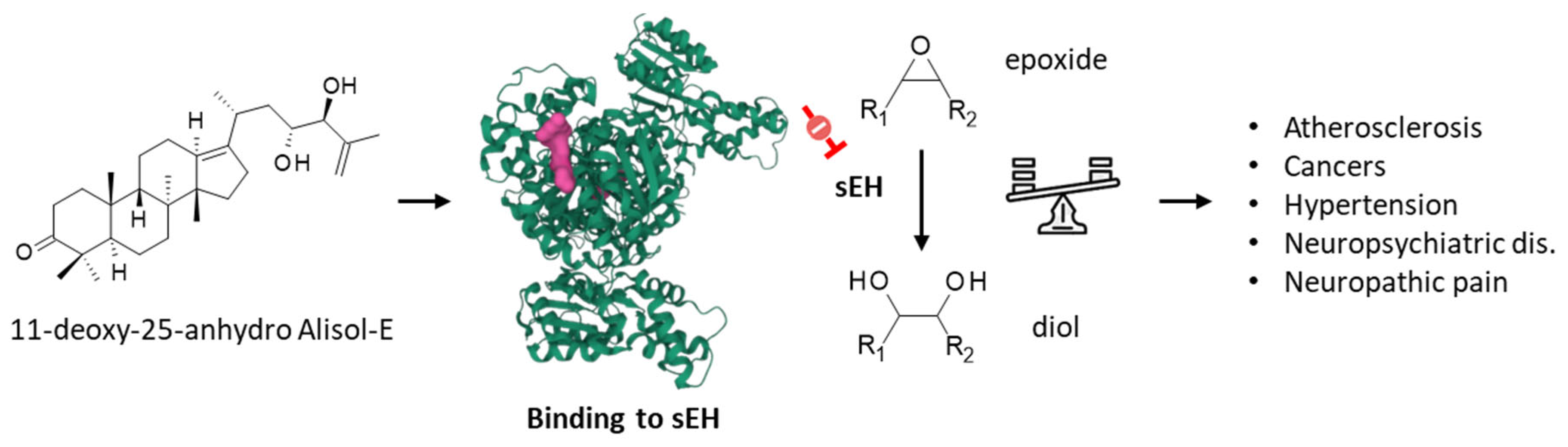
- Charles, R.; Eaton, P. Redox Regulation of Soluble Epoxide Hydrolase-Implications for Cardiovascular Health and Disease. Cells 2022, 11, 1932. [Google Scholar] [CrossRef]
- Shan, J.; Hashimoto, K. Soluble Epoxide Hydrolase as a Therapeutic Target for Neuropsychiatric Disorders. Int. J. Mol. Sci. 2022, 23, 4951. [Google Scholar] [CrossRef]
- Hur, J.M.; Choi, J.W.; Park, J.C. Effects of methanol extract of Alisma orientale rhizome and its major component, alisol B 23-acetate, on hepatic drug metabolizing enzymes in rats treated with bromobenzene. Arch. Pharm. Res. 2007, 30, 1543–1549. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.P.; Zhang, J.; Zhao, W.Y.; Yi, J.; Yan, J.K.; Wang, Y.L.; Morisseau, C.; Liu, Z.B.; Hammock, B.D.; Ma, X.C. Protostane-type triterpenoids as natural soluble epoxide hydrolase inhibitors: Inhibition potentials and molecular dynamics. Bioorg. Chem. 2020, 96, 103637. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.Y.; Zhang, X.Y.; Zhou, M.R.; Tian, X.G.; Lv, X.; Zhang, H.L.; Deng, S.; Zhang, B.J.; Sun, C.P.; Ma, X.C. Natural soluble epoxide hydrolase inhibitors from Alisma orientale and their potential mechanism with soluble epoxide hydrolase. Int. J. Biol. Macromol. 2021, 183, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Han, L.; Bi, X.; Wang, X.; Mu, Y.; Guan, P.; Li, L.; Huang, X. Structures and biological activities of the triterpenoids and sesquiterpenoids from Alisma orientale. Phytochemistry 2016, 131, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Bi, X.; Wang, P.; Ma, Q.; Han, L.; Wang, X.; Mu, Y.; Guan, P.; Qu, X.; Wang, Z.; Huang, X. Anti-Inflammatory Activities and Liver Protection of Alisol F and 25-Anhydroalisol F through the Inhibition of MAPK, STAT3, and NF-κB Activation In Vitro and In Vivo. Molecules 2017, 22, 951. [Google Scholar] [CrossRef]
- Wang, B.; Chen, L.; Dai, L.; Fang, W.; Wang, H. Alisol B 23-Acetate Ameliorates Lipopolysaccharide-Induced Cardiac Dysfunction by Suppressing Toll-Like Receptor 4 (TLR4)/NADPH Oxidase 2 (NOX2) Signaling Pathway. Med. Sci. Monit. 2019, 25, 8472–8481. [Google Scholar] [CrossRef]
- Xia, F.; Xiang, S.; Chen, Z.; Song, L.; Li, Y.; Liao, Z.; Ge, B.; Zhou, B. The probiotic effects of AB23A on high-fat-diet-induced non-alcoholic fatty liver disease in mice may be associated with suppressing the serum levels of lipopolysaccharides and branched-chain amino acids. Arch. Biochem. Biophys. 2021, 714, 109080. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Li, Y.; Deng, L.; Ren, R.; Ge, B.; Liao, Z.; Xiang, S.; Zhou, B. Alisol B 23-Acetate Ameliorates Lipopolysaccharide-Induced Intestinal Barrier Dysfunction by Inhibiting TLR4-NOX1/ROS Signaling Pathway in Caco-2 Cells. Front. Pharmacol. 2022, 13, 911196. [Google Scholar] [CrossRef]
- Liu, Y.; Xia, X.C.; Meng, L.Y.; Wang, Y.; Li, Y.M. Alisol B 23-acetate inhibits the viability and induces apoptosis of non-small cell lung cancer cells via PI3K/AKT/mTOR signal pathway. Mol. Med. Rep. 2019, 20, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Moeller, M.J.; Kramann, R.; Lammers, T.; Hoppe, B.; Latz, E.; Ludwig-Portugall, I.; Boor, P.; Floege, J.; Kurts, C.; Weiskirchen, R.; et al. New Aspects of Kidney Fibrosis-From Mechanisms of Injury to Modulation of Disease. Front. Med. 2022, 8, 814497. [Google Scholar] [CrossRef]
- Chen, H.; Wang, M.C.; Chen, Y.Y.; Chen, L.; Wang, Y.N.; Vaziri, N.D.; Miao, H.; Zhao, Y.Y. Alisol B 23-acetate attenuates CKD progression by regulating the renin-angiotensin system and gut-kidney axis. Ther. Adv. Chronic Dis. 2020, 11, 2040622320920025. [Google Scholar] [CrossRef]
- Chen, L.; Chen, D.Q.; Wang, M.; Liu, D.; Chen, H.; Dou, F.; Vaziri, N.D.; Zhao, Y.Y. Role of RAS/Wnt/β-catenin axis activation in the pathogenesis of podocyte injury and tubulo-interstitial nephropathy. Chem. Biol. Interact. 2017, 273, 56–72. [Google Scholar] [CrossRef]
- Chen, H.; Yang, T.; Wang, M.C.; Chen, D.Q.; Yang, Y.; Zhao, Y.Y. Novel RAS inhibitor 25-O-methylalisol F attenuates epithelial-to-mesenchymal transition and tubulo-interstitial fibrosis by selectively inhibiting TGF-β-mediated Smad3 phosphorylation. Phytomedicine 2018, 42, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.H.; Zhou, X.M.; Wei, W.; Chen, T.; Su, Q.P.; Tao, J.; Chen, L.D. Alisol A 24-Acetate, a Triterpenoid Derived from Alisma orientale, Inhibits Ox-LDL-Induced Phenotypic Transformation and Migration of Rat Vascular Smooth Muscle Cells through Suppressing ERK1/2 Signaling. J. Vasc. Res. 2016, 53, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.C.; Jia, X.K.; Fan, Y.; Xu, S.H.; Li, X.Y.; Huang, M.Q.; Lan, M.L.; Xu, W.; Wu, S.S. Alisol B 23-Acetate Ameliorates Azoxymethane/Dextran Sodium Sulfate-Induced Male Murine Colitis-Associated Colorectal Cancer via Modulating the Composition of Gut Microbiota and Improving Intestinal Barrier. Front. Cell. Infect. Microbiol. 2021, 11, 640225. [Google Scholar] [CrossRef]
- Xu, X.; Li, L.; Zhang, Y.; Lu, X.; Lin, W.; Wu, S.; Qin, X.; Xu, R.; Lin, W. Hypolipidemic effect of Alisma orientale (Sam.) Juzep on gut microecology and liver transcriptome in diabetic rats. PLoS ONE 2020, 15, e0240616. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Zhai, Y.; Ji, M.; Wei, Y.; Wu, J.; Xue, W.; Tao, W.W.; Wu, H. Alisma orientalis Beverage Treats Atherosclerosis by Regulating Gut Microbiota in ApoE-/- Mice. Front. Pharmacol. 2020, 11, 570555. [Google Scholar] [CrossRef]
- Lu, L.; Lu, T.; Shen, J.; Lv, X.; Wei, W.; Wang, H.; Xue, X. Alisol A 24-acetate protects against brain microvascular endothelial cells injury through inhibiting miR-92a-3p/tight junctions axis. Aging 2021, 13, 15353–15365. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wei, W.; Shen, J.; Lu, L.; Lu, T.; Wang, H.; Xue, X. Alisol A 24-acetate protects oxygen-glucose deprivation-induced brain microvascular endothelial cells against apoptosis through miR-92a-3p inhibition by targeting the B-cell lymphoma-2 gene. Pharm. Biol. 2021, 59, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Li, H.; Zhou, Y.; Wei, W.; Ding, L.; Zhan, Z.; Liu, W.; Tao, J.; Xue, X. Neuroprotective effects of alisol A 24-acetate on cerebral ischaemia-reperfusion injury are mediated by regulating the PI3K/AKT pathway. J. Neuroinflammation 2022, 19, 37. [Google Scholar] [PubMed]
- Jang, E.; Lee, J.H. Promising Anticancer Activities of Alismatis rhizome and Its Triterpenes via p38 and PI3K/Akt/mTOR Signaling Pathways. Nutrients 2021, 13, 2455. [Google Scholar] [CrossRef]
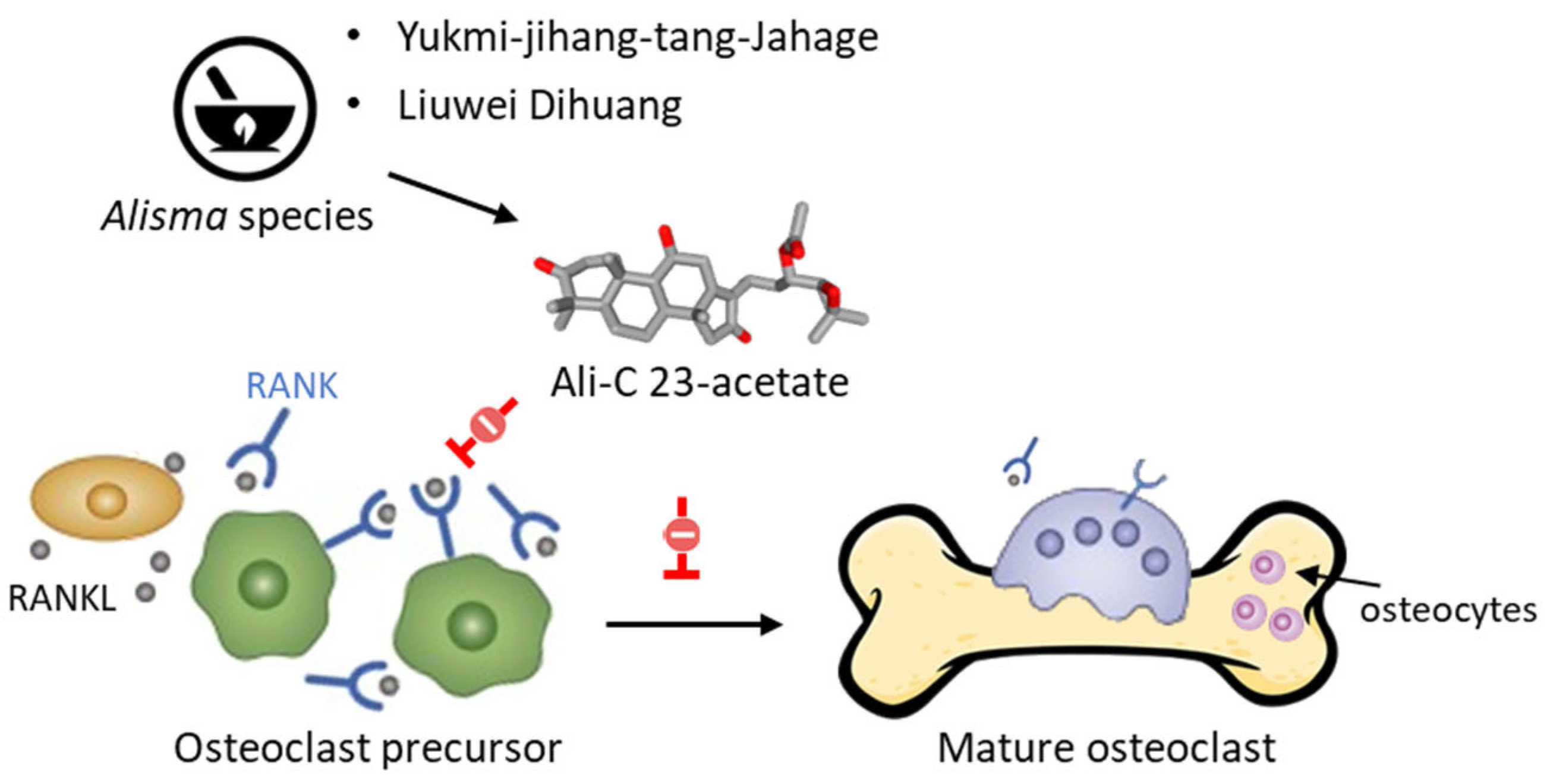
- Jin, U.H.; Kim, D.I.; Lee, T.K.; Lee, D.N.; Kim, J.K.; Lee, I.S.; Kim, C.H. Herbal formulation, Yukmi-jihang-tang-Jahage, regulates bone resorption by inhibition of phosphorylation mediated by tyrosine kinase Src and cyclooxygenase expression. J. Ethnopharmacol. 2006, 106, 333–343. [Google Scholar] [CrossRef]
- Xia, B.; Xu, B.; Sun, Y.; Xiao, L.; Pan, J.; Jin, H.; Tong, P. The effects of Liuwei Dihuang on canonical Wnt/β-catenin signaling pathway in osteoporosis. J. Ethnopharmacol. 2014, 153, 133–141. [Google Scholar] [CrossRef]
- An, J.; Hao, D.; Zhang, Q.; Chen, B.; Zhang, R.; Wang, Y.; Yang, H. Natural products for treatment of bone erosive diseases: The effects and mechanisms on inhibiting osteoclastogenesis and bone resorption. Int. Immunopharmacol. 2016, 36, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Zhu, H.; Li, G.; Lan, M.; Li, X.; Huang, M.; Xu, W.; Wu, S. Anti-osteoporotic effects of alisol C 23-acetate via osteoclastogenesis inhibition. Biomed. Pharmacother. 2021, 137, 111321. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Leutou, A.S.; Yeon, J.T.; Choi, S.W.; Kim, S.H.; Yee, S.T.; Choi, K.H.; Nam, S.J.; Son, Y.J. The Inhibitory Effect of Alisol A 24-Acetate from Alisma canaliculatum on Osteoclastogenesis. Int. J. Endocrinol. 2015, 2015, 132436. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Kang, K.Y.; Lee, S.J.; Nam, S.J.; Son, Y.J.; Yee, S.T. The Protective Effects of Alisol A 24-Acetate from Alisma canaliculatum on Ovariectomy Induced Bone Loss in Vivo. Molecules 2016, 21, 74. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Kobayashi, Y.; Nakamichi, Y.; Udagawa, N.; Takahashi, N.; Im, N.K.; Seo, H.J.; Jeon, W.B.; Yonezawa, T.; Cha, B.Y.; et al. Alisol-B, a novel phyto-steroid, suppresses the RANKL-induced osteoclast formation and prevents bone loss in mice. Biochem. Pharmacol. 2010, 80, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Li, T.; Zeng, Q.; Wang, X.; Zhu, Y. Alisol F 24 Acetate Enhances Chemosensitivity and Apoptosis of MCF-7/DOX Cells by Inhibiting P-Glycoprotein-Mediated Drug Efflux. Molecules 2016, 21, 183. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, J.X.; Shen, X.L.; Wan, C.K.; Tse, A.K.; Fong, W.F. Reversal of P-glycoprotein-mediated multidrug resistance by Alisol B 23-acetate. Biochem. Pharmacol. 2004, 68, 843–855. [Google Scholar]
- Lou, C.; Xu, X.; Chen, Y.; Zhao, H. Alisol A Suppresses Proliferation, Migration, and Invasion in Human Breast Cancer MDA-MB-231 Cells. Molecules 2019, 24, 3651. [Google Scholar]
- Shi, Y.; Wang, M.; Wang, P.; Zhang, T.; Yu, J.; Shi, L.; Li, M.; Wang, H.; Zhang, Q.; Zhao, H. Alisol A is potentially therapeutic in human breast cancer cells. Oncol. Rep. 2020, 44, 1266–1274. [Google Scholar] [CrossRef]
- Xia, J.; Luo, Q.; Huang, S.; Jiang, F.; Wang, L.; Wang, G.; Xie, J.; Liu, J.; Xu, Y. Alisol B 23-acetate-induced HepG2 hepatoma cell death through mTOR signaling-initiated G1 cell cycle arrest and apoptosis: A quantitative proteomic study. Chin. J. Cancer Res. 2019, 31, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Cheng, J.; Zhu, D.; Shi, X.; Wei, Y.; Chen, S.; Wang, Z.; Yuan, D. The effects of Alisol B 23-acetate in hepatocellular carcinoma via inducing cell apoptosis and inhibiting cell migration and invasion. Gen. Physiol. Biophys. 2020, 39, 219–228. [Google Scholar] [CrossRef]
- Han, W.; Xing, W.; Wang, K.; Wang, B.; Bai, K. Alisol A attenuates malignant phenotypes of colorectal cancer cells by inactivating PI3K/Akt signaling. Oncol. Lett. 2022, 24, 249. [Google Scholar] [CrossRef]
- Chen, X.; Liu, H. Alisol A Inhibited the Proliferation, Migration, and Invasion of Nasopharyngeal Carcinoma Cells by Inhibiting the Hippo Signaling Pathway. Yonsei Med. J. 2021, 62, 895–902. [Google Scholar] [CrossRef]
- Yoshida, I.; Ito, C.; Matsuda, S.; Tsuji, A.; Yanaka, N.; Yuasa, K. Alisol B, a triterpene from Alismatis rhizoma (dried rhizome of Alisma orientale), inhibits melanin production in murine B16 melanoma cells. Biosci. Biotechnol. Biochem. 2017, 81, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Sheng, Y.; Zou, M. Antiproliferative activity of Alisol B in MDA-MB-231 cells is mediated by apoptosis, dysregulation of mitochondrial functions, cell cycle arrest and generation of reactive oxygen species. Biomed. Pharmacother. 2017, 87, 110–117. [Google Scholar] [CrossRef]
- Zhang, L.L.; Xu, Y.L.; Tang, Z.H.; Xu, X.H.; Chen, X.; Li, T.; Ding, C.Y.; Huang, M.Q.; Chen, X.P.; Wang, Y.T.; et al. Effects of alisol B 23-acetate on ovarian cancer cells: G1 phase cell cycle arrest, apoptosis, migration and invasion inhibition. Phytomedicine 2016, 23, 800–809. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, E.T.S.; Wang, M. Alisol B 23-acetate induces autophagic-dependent apoptosis in human colon cancer cells via ROS generation and JNK activation. Oncotarget 2017, 8, 70239–70249. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, H.; Wang, X.; Shen, T.; Wang, S.; Ren, D. Alisol B-23-acetate, a tetracyclic triterpenoid isolated from Alisma orientale, induces apoptosis in human lung cancer cells via the mitochondrial pathway. Biochem. Biophys. Res. Commun. 2018, 505, 1015–1021. [Google Scholar] [CrossRef]
- Xu, Y.H.; Zhao, L.J.; Li, Y. Alisol B acetate induces apoptosis of SGC7901 cells via mitochondrial and phosphatidylinositol 3-kinases/Akt signaling pathways. World J. Gastroenterol. 2009, 15, 2870–2877. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Kim, J.N.; Lee, M.J.; Kim, W.K.; Nam, J.H.; Kim, B.J. Apoptotic effects of alisol B 23-acetate on gastric cancer cells. Mol. Med. Rep. 2021, 23, 248. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Tang, Y.; He, J.; Cao, X.; Liu, J.; Kou, S.; Yang, Y.; Xue, J.; Li, F. Alisol B 23-Acetate Increases the Antitumor Effect of Bufalin on Liver Cancer through Inactivating Wnt/β-Catenin Axis. Comput. Math. Methods Med. 2022, 2022, 6249534. [Google Scholar] [CrossRef] [PubMed]
- El-Baba, C.; Baassiri, A.; Kiriako, G.; Dia, B.; Fadlallah, S.; Moodad, S.; Darwiche, N. Terpenoids’ anti-cancer effects: Focus on autophagy. Apoptosis 2021, 26, 491–511. [Google Scholar] [CrossRef]
- Law, B.Y.; Wang, M.; Ma, D.L.; Al-Mousa, F.; Michelangeli, F.; Cheng, S.H.; Ng, M.H.; To, K.F.; Mok, A.Y.; Ko, R.Y.; et al. Alisol B, a novel inhibitor of the sarcoplasmic/endoplasmic reticulum Ca(2+) ATPase pump, induces autophagy, endoplasmic reticulum stress, and apoptosis. Mol. Cancer Ther. 2010, 9, 718–730. [Google Scholar] [CrossRef]
- Wang, Y.L.; Zhao, J.C.; Liang, J.H.; Tian, X.G.; Huo, X.K.; Feng, L.; Ning, J.; Wang, C.; Zhang, B.J.; Chen, G.; et al. A bioactive new protostane-type triterpenoid from Alisma plantago-aquatica subsp. orientale (Sam.) Sam. Nat. Prod. Res. 2019, 33, 776–781. [Google Scholar] [CrossRef]
- Lin, H.R. Triterpenes from Alisma orientalis act as androgen receptor agonists, progesterone receptor antagonists, and glucocorticoid receptor antagonists. Bioorg. Med. Chem. Lett. 2014, 24, 3626–3632. [Google Scholar] [CrossRef]
- Xu, F.; Lu, C.; Wu, Q.; Chen, J.; Gu, W.; Du, W.; You, M. Study on antitumor molecular mechanism of Alisols based on p53DNA. Int. J. Biol. Macromol. 2018, 116, 1163–1174. [Google Scholar] [CrossRef]
- Xu, F.; Chen, J.; Wu, Q.; Gu, W.; Shen, Y.; Lu, C.; Zhang, Y.; Liu, S.; Liao, H. The antitumor molecular mechanism of Alisma orientalis with c-myc DNA: Multi-spectroscopic analysis and molecular simulation. J. Biomol. Struct. Dyn. 2020, 38, 4189–4209. [Google Scholar] [CrossRef]
- Kubo, M.; Matsuda, H.; Tomohiro, N.; Yoshikawa, M. Studies on Alismatis rhizoma. I. Anti-allergic effects of methanol extract and six terpene components from Alismatis rhizoma (dried rhizome of Alisma orientale). Biol. Pharm. Bull. 1997, 20, 511–516. [Google Scholar] [CrossRef]
- Lee, J.H.; Kwon, O.S.; Jin, H.G.; Woo, E.R.; Kim, Y.S.; Kim, H.P. The rhizomes of Alisma orientale and alisol derivatives inhibit allergic response and experimental atopic dermatitis. Biol. Pharm. Bull. 2012, 35, 1581–1587. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.; Fu, B.; Ji, N.; Pan, S.; Zhao, X.; Zhang, Z.; Qiu, Y.; Wang, R.; Jin, M.; Wen, K.; et al. Alisol B 23-Acetate Inhibits IgE/Ag-Mediated Mast Cell Activation and Allergic Reaction. Int. J. Mol. Sci. 2018, 19, 4092. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.G.; Jin, Q.; Ryun Kim, A.; Choi, H.; Lee, J.H.; Kim, Y.S.; Lee, D.G.; Woo, E.R. A new triterpenoid from Alisma orientale and their antibacterial effect. Arch. Pharm. Res. 2012, 35, 1919–1926. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jiang, Z.Y.; Luo, J.; Cheng, P.; Ma, Y.B.; Zhang, X.M.; Zhang, F.X.; Zhou, J.; Chen, J.J. Anti-HBV agents. Part 1: Synthesis of alisol A derivatives: A new class of hepatitis B virus inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 4647–4650. [Google Scholar] [CrossRef]
- Zhang, Q.; Jiang, Z.Y.; Luo, J.; Liu, J.F.; Ma, Y.B.; Guo, R.H.; Zhang, X.M.; Zhou, J.; Chen, J.J. Anti-HBV agents. Part 2: Synthesis and in vitro anti-hepatitis B virus activities of alisol A derivatives. Bioorg. Med. Chem. Lett. 2009, 19, 2148–2153. [Google Scholar] [CrossRef]
- Zhang, Q.; Jiang, Z.Y.; Luo, J.; Ma, Y.B.; Liu, J.F.; Guo, R.H.; Zhang, X.M.; Zhou, J.; Niu, W.; Du, F.F.; et al. Anti-HBV agents. Part 3: Preliminary structure-activity relationships of tetra-acylalisol A derivatives as potent hepatitis B virus inhibitors. Bioorg. Med. Chem. Lett. 2009, 19, 6659–6665. [Google Scholar] [CrossRef]
- Liao, M.; Shang, H.; Li, Y.; Li, T.; Wang, M.; Zheng, Y.; Hou, W.; Liu, C. An integrated approach to uncover quality marker underlying the effects of Alisma orientale on lipid metabolism, using chemical analysis and network pharmacology. Phytomedicine 2018, 45, 93–104. [Google Scholar] [CrossRef]
- Lin, W.; Sun, F.; Zhang, Y.; Xu, X.; Lu, X.; Li, L.; Xu, R. Comparative transcriptome and metabolite profiling of four tissues from Alisma orientale (Sam.) Juzep reveals its inflorescence developmental and medicinal characteristics. Sci. Rep. 2019, 9, 12310. [Google Scholar] [CrossRef]
- Li, R.; Li, Z.L.; Chen, Y.P.; Bu, W.Q.; Ding, W.B.; Yang, B.; Wang, C.F.; Ma, L.; Jia, X.B.; Feng, L. The structural composition of components contributes to the superiority of the geoherb Alisma orientale for “diuresis and diffusing dampness”. RSC Adv. 2020, 10, 39385–39395. [Google Scholar] [CrossRef]
- Wei, Y.C.; Yan, M.; Yang, J.; Liu, J.C.; Yin, H.M.; Wu, Y.; Sun, Y.C.; Xiao, W. Optimization of flash-type extraction technology of alisol B 23-acetate from Alismatis Rhizoma by response surface methodology. Zhongguo Zhong Yao Za Zhi 2016, 41, 438–442. [Google Scholar]
- Wang, C.F.; Cheng, X.D.; Gu, J.F.; Yuan, J.R.; Zhao, B.J.; Zhang, L.; Chen, J.; Feng, L.; Jia, X.B. Research development of the chemical material basis of Alisma orientalis and its toxicity. Zhongguo Zhong Yao Za Zhi 2015, 40, 840–846. [Google Scholar] [PubMed]
- Yu, Y.; Ma, C.; Bi, K.; Yang, G.; Xie, P.; Wang, J.; Chen, X.H. A metabonomic analysis of urine from rats treated with rhizoma alismatis using ultra-performance liquid chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2011, 25, 2633–2640. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.Q.; Feng, Y.L.; Tian, T.; Chen, H.; Yin, L.; Zhao, Y.Y.; Lin, R.C. Diuretic and anti-diuretic activities of fractions of Alismatis rhizoma. J. Ethnopharmacol. 2014, 157, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Feng, L.; Ma, L.; Chen, H.; Tan, X.; Hou, X.; Song, J.; Cui, L.; Liu, D.; Chen, J.; et al. Alisol A 24-Acetate and Alisol B 23-Acetate Induced Autophagy Mediates Apoptosis and Nephrotoxicity in Human Renal Proximal Tubular Cells. Front. Pharmacol. 2017, 8, 172. [Google Scholar] [CrossRef]
- Xu, F.; Wu, Q.N.; Chen, J.; Gu, W.; Fang, F.; Zhang, L.Q.; Zhao, B. The binding mechanisms of plasma protein to active compounds in Alismaorientale rhizomes (Alismatis Rhizoma). Bioorg. Med. Chem. Lett. 2014, 24, 4099–4105. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Huang, S.; Yan, J.; Cai, B. Pharmacokinetic study of six triterpenoids of raw and processed Alisma plantago-aquatica in rat plasma by using ultra performance liquid chromatography-tandem mass spectrometry approach. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2019, 1124, 323–330. [Google Scholar] [CrossRef]
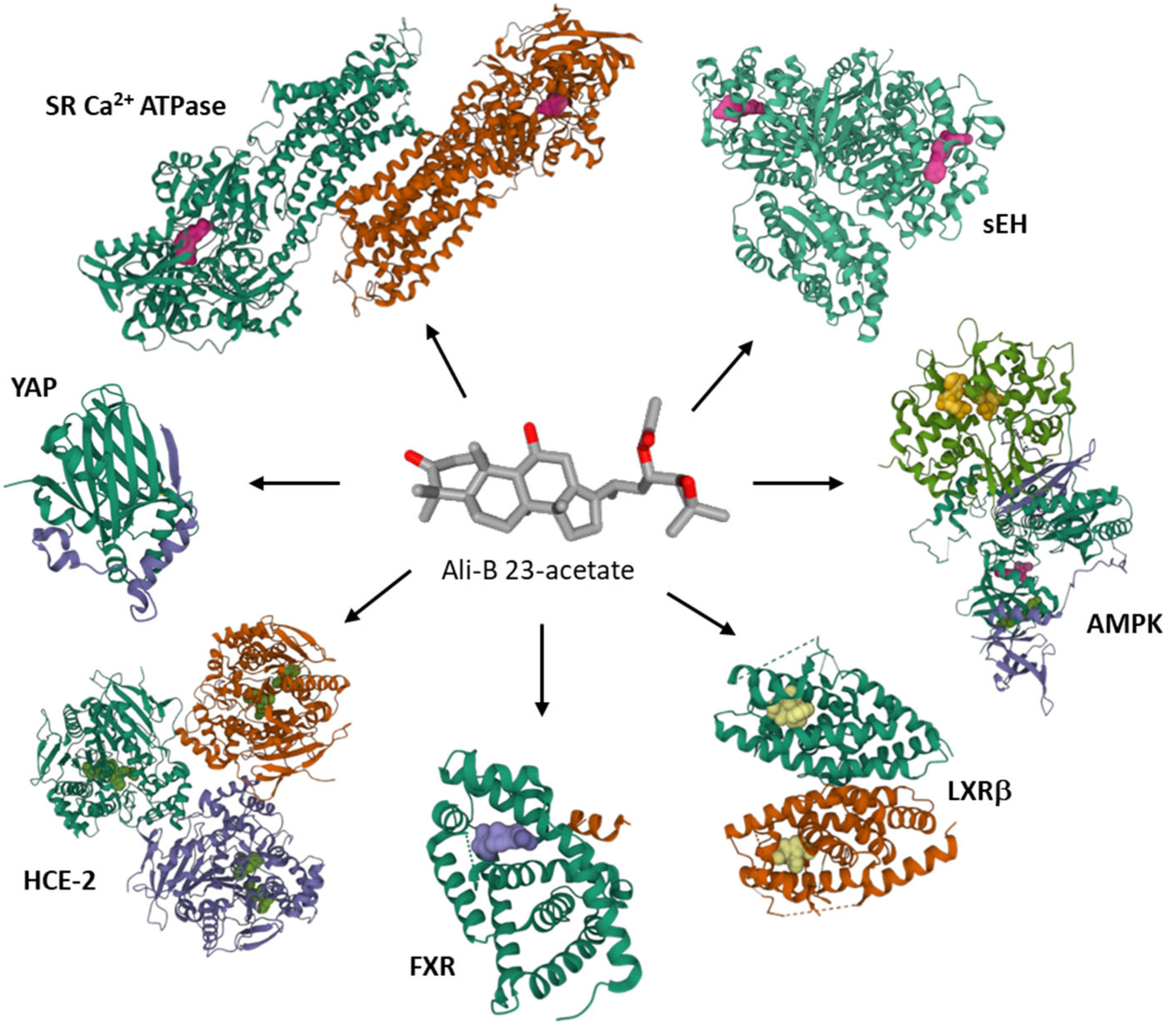
| Compounds | Cancer Cell Types (Cell Lines) | Observed Effects | Reference |
|---|---|---|---|
| Ali-A | Colorectal cancer cells (HCT-116, HT-29) | Cell growth inhibition. Induction caspase-dependent apoptosis and pyroptosis. Repression of cell migration, through down-regulation of N-cadherin, up-regulation of E-cadherin. | [114] |
| Ali-A | Nasopharyngeal carcinoma cells (C666-1, HK1) | Cell growth inhibition, G0/G1 cell cycle arrest with down-regulation of cyclins D1/E1 and CDK2/4. Inhibition of cell migration, with down-regulation of MMP2/9. Binding to and phosphorylation of the transcriptional coactivator YAP. | [115] |
| Ali-A | Breast cancer cells (MDA-MB-231) | Inhibition of cell proliferation, G1 cell cycle arrest, induction of apoptosis and autophagy. Induction of ROS and DNA damage. Blockade of NFκB and PI3K/Akt/mTOR pathways. Suppression of cell migration and invasion, via inhibition of MMP2/9. | [110,111] |
| Ali-B | Melanoma cells (B16) | Cell growth inhibition. Downregulation of MITF, via suppression of CREB and activation of ERK. | [116] |
| Ali-B | Breast cancer cells (MDA-MB-231) | Inhibition of cell growth; caspase-dependent mitochondrial apoptosis; accumulation of ROS; downregulation of p-AKT, p-p65, and p-mTOR. | [117] |
| Ali-B 23-acetate | Ovarian cancer cells (A2780, HEY) | G1 cell cycle arrest, down-regulation of CDK4/6, cyclin D1. Up-regulation of Bax/Bcl-2 ratio and induction of endoplasmic reticulum stress through IRE1 signaling. Suppression of cells migration and invasion, with inhibition of MMP-2/9. | [118] |
| Ali-B 23-acetate | Colon cancer cells (SW620, HCT116) | Cell growth inhibition, induction of apoptosis and autophagy, dependent on the production of ROS and phosphorylation of JNK. | [119] |
| Ali-B 23-acetate | Lung cancer cells (A549, NCI-H292) | Cell growth inhibition, induction of mitochondrial apoptosis, generation of ROS. Reduced phosphorylation of AKT, PI3K, and mTor. Inhibition of cell migration/invasion. | [88,120] |
| Ali-B 23-acetate | Gastric cancer cells (AGS, SGC7901) | Inhibition of cell proliferation, induction of mitochondrial apoptosis, generation of ROS. Regulation of MAPK activation. | [121,122] |
| Ali-B 23-acetate | Hepatocellular carcinoma cells(SK-HEP-1, HepG2, SMMC-7721, MHCC97) | Cell growth inhibition, G1 cell cycle arrest, induction of apoptosis. Inhibition of cell migration. Reduction in tumorigenesis (in vivo) with pretreatment of the cells in vitro (before grafting). Repression of mTOR pathway-related proteins. Inhibition of the Wnt/β-catenin pathway. | [112,113,123] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bailly, C. Pharmacological Properties and Molecular Targets of Alisol Triterpenoids from Alismatis Rhizoma. Biomedicines 2022, 10, 1945. https://doi.org/10.3390/biomedicines10081945
Bailly C. Pharmacological Properties and Molecular Targets of Alisol Triterpenoids from Alismatis Rhizoma. Biomedicines. 2022; 10(8):1945. https://doi.org/10.3390/biomedicines10081945
Chicago/Turabian StyleBailly, Christian. 2022. "Pharmacological Properties and Molecular Targets of Alisol Triterpenoids from Alismatis Rhizoma" Biomedicines 10, no. 8: 1945. https://doi.org/10.3390/biomedicines10081945
APA StyleBailly, C. (2022). Pharmacological Properties and Molecular Targets of Alisol Triterpenoids from Alismatis Rhizoma. Biomedicines, 10(8), 1945. https://doi.org/10.3390/biomedicines10081945