Bioactive Cell-Derived ECM Scaffold Forms a Unique Cellular Microenvironment for Lung Tissue Engineering
Abstract
:1. Introduction
2. Materials and Methods
2.1. BETA Scaffold
2.2. Chick Chorioallantoic Membrane (CAM) Assay
2.3. Cell Sources and Maintenance
2.4. ECM-Integrated BETA Scaffold
2.5. Functional Analysis
2.6. Immunofluorescence (IF)
3. Results
4. Discussion
5. Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giwa, S.; Lewis, J.K.; Alvarez, L.; Langer, R.; Roth, A.E.; Church, G.M.; Markmann, J.F.; Sachs, D.H.; Chandraker, A.; Wertheim, J.A.; et al. The Promise of Organ and Tissue Preservation to Transform Medicine. Nat. Biotechnol. 2017, 35, 530–542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diamantopoulos, A.; Wright, E.; Vlahopoulou, K.; Cornic, L.; Schoof, N.; Maher, T.M. The Burden of Illness of Idiopathic Pulmonary Fibrosis: A Comprehensive Evidence Review. Pharmacoeconomics 2018, 36, 779–807. [Google Scholar] [CrossRef] [Green Version]
- George, P.M.; Patterson, C.M.; Reed, A.K.; Thillai, M. Lung Transplantation for Idiopathic Pulmonary Fibrosis. Lancet Respir. Med. 2019, 7, 271–282. [Google Scholar] [CrossRef]
- Weibel, E.R. What Makes a Good Lung? Swiss Med. Wkly. 2009, 139, 375–386. [Google Scholar] [CrossRef]
- Calle, E.A.; Ghaedi, M.; Sundaram, S.; Sivarapatna, A.; Tseng, M.K.; Niklason, L.E. Strategies for Whole Lung Tissue Engineering. IEEE Trans. Biomed. Eng. 2014, 61, 1482–1496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skolasinski, S.D.; Panoskaltsis-Mortari, A. Lung Tissue Bioengineering for Chronic Obstructive Pulmonary Disease: Overcoming the Need for Lung Transplantation from Human Donors. Expert Rev. Respir. Med. 2019, 13, 665–678. [Google Scholar] [CrossRef]
- Doryab, A.; Tas, S.; Taskin, M.B.; Yang, L.; Hilgendorff, A.; Groll, J.; Wagner, D.E.; Schmid, O. Evolution of Bioengineered Lung Models: Recent Advances and Challenges in Tissue Mimicry for Studying the Role of Mechanical Forces in Cell Biology. Adv. Funct. Mater. 2019, 29, 1903114. [Google Scholar] [CrossRef]
- Petersen, T.H.; Calle, E.A.; Zhao, L.; Lee, E.J.; Gui, L.; Raredon, M.B.; Gavrilov, K.; Yi, T.; Zhuang, Z.W.; Breuer, C.; et al. Tissue-Engineered Lungs for in Vivo Implantation. Science 2010, 329, 538–541. [Google Scholar] [CrossRef] [Green Version]
- O’Brien, F.J. Biomaterials & amp; Scaffolds for Tissue Engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Hussey, G.S.; Dziki, J.L.; Badylak, S.F. Extracellular Matrix-Based Materials for Regenerative Medicine. Nat. Rev. Mater. 2018, 3, 159–173. [Google Scholar] [CrossRef]
- Nishiguchi, A.; Singh, S.; Wessling, M.; Kirkpatrick, C.J.; Möller, M. Basement Membrane Mimics of Biofunctionalized Nanofibers for a Bipolar-Cultured Human Primary Alveolar-Capillary Barrier Model. Biomacromolecules 2017, 18, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.E.; Bridge, J.C.; Brace, L.A.; Knox, A.J.; Aylott, J.W.; Brightling, C.E.; Ghaemmaghami, A.M.; Rose, F.R.A.J.A.J. A Novel Electrospun Biphasic Scaffold Provides Optimal Three-Dimensional Topography for in Vitro Co-Culture of Airway Epithelial and Fibroblast Cells. Biofabrication 2014, 6, 035014. [Google Scholar] [CrossRef] [PubMed]
- Harrington, H.; Cato, P.; Salazar, F.; Wilkinson, M.; Knox, A.; Haycock, J.W.; Rose, F.; Aylott, J.W.; Ghaemmaghami, A.M. Immunocompetent 3D Model of Human Upper Airway for Disease Modeling and in Vitro Drug Evaluation. Mol. Pharm. 2014, 11, 2082–2091. [Google Scholar] [CrossRef] [PubMed]
- Higuita-Castro, N.; Nelson, M.T.; Shukla, V.; Agudelo-Garcia, P.A.; Zhang, W.; Duarte-Sanmiguel, S.M.; Englert, J.A.; Lannutti, J.J.; Hansford, D.J.; Ghadiali, S.N. Using a Novel Microfabricated Model of the Alveolar-Capillary Barrier to Investigate the Effect of Matrix Structure on Atelectrauma. Sci. Rep. 2017, 7, 11623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siddiqui, N.; Asawa, S.; Birru, B.; Baadhe, R.; Rao, S. PCL-Based Composite Scaffold Matrices for Tissue Engineering Applications. Mol. Biotechnol. 2018, 60, 506–532. [Google Scholar] [CrossRef]
- Bello, A.B.; Kim, D.; Kim, D.; Park, H.; Lee, S.-H. Engineering and Functionalization of Gelatin Biomaterials: From Cell Culture to Medical Applications. Tissue Eng. Part B Rev. 2020, 26, 164–180. [Google Scholar] [CrossRef] [Green Version]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef] [Green Version]
- Yuan, S.; Xiong, G.; Roguin, A.; Choong, C. Immobilization of Gelatin onto Poly(Glycidyl Methacrylate)-Grafted Polycaprolactone Substrates for Improved Cell–Material Interactions. Biointerphases 2012, 7, 30. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Pérez, J.; Ahearne, M. The Impact of Decellularization Methods on Extracellular Matrix Derived Hydrogels. Sci. Rep. 2019, 9, 14933. [Google Scholar] [CrossRef] [Green Version]
- Young, B.M.; Shankar, K.; Allen, B.P.; Pouliot, R.A.; Schneck, M.B.; Mikhaiel, N.S.; Heise, R.L. Electrospun Decellularized Lung Matrix Scaffold for Airway Smooth Muscle Culture. ACS Biomater. Sci. Eng. 2017, 3, 3480–3492. [Google Scholar] [CrossRef]
- Doryab, A.; Taskin, M.B.; Stahlhut, P.; Schröppel, A.; Wagner, D.E.; Groll, J.; Schmid, O. A Biomimetic, Copolymeric Membrane for Cell-Stretch Experiments with Pulmonary Epithelial Cells at the Air-Liquid Interface. Adv. Funct. Mater. 2021, 31, 2004707. [Google Scholar] [CrossRef]
- Doryab, A.; Taskin, M.B.; Stahlhut, P.; Schröppel, A.; Orak, S.; Voss, C.; Ahluwalia, A.; Rehberg, M.; Hilgendorff, A.; Stöger, T.; et al. A Bioinspired in Vitro Lung Model to Study Particokinetics of Nano-/Microparticles Under Cyclic Stretch and Air-Liquid Interface Conditions. Front. Bioeng. Biotechnol. 2021, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Staab-Weijnitz, C.A.; Fernandez, I.E.; Knüppel, L.; Maul, J.; Heinzelmann, K.; Juan-Guardela, B.M.; Hennen, E.; Preissler, G.; Winter, H.; Neurohr, C.; et al. FK506-Binding Protein 10, a Potential Novel Drug Target for Idiopathic Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2015, 192, 455–467. [Google Scholar] [CrossRef] [Green Version]
- Beacham, D.A.; Amatangelo, M.D.; Cukierman, E. Preparation of Extracellular Matrices Produced by Cultured and Primary Fibroblasts. Curr. Protoc. Cell Biol. 2006, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farag, A.; Hashimi, S.M.; Vaquette, C.; Volpato, F.Z.; Hutmacher, D.W.; Ivanovski, S. Assessment of Static and Perfusion Methods for Decellularization of PCL Membrane-Supported Periodontal Ligament Cell Sheet Constructs. Arch. Oral Biol. 2018, 88, 67–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Velden, J.L.; Wagner, D.E.; Lahue, K.G.; Abdalla, S.T.; Lam, Y.-W.; Weiss, D.J.; Janssen-Heininger, Y.M.W. TGF-Β1-Induced Deposition of Provisional Extracellular Matrix by Tracheal Basal Cells Promotes Epithelial-to-Mesenchymal Transition in a c-Jun NH2-Terminal Kinase-1-Dependent Manner. Am. J. Physiol. Cell. Mol. Physiol. 2018, 314, L984–L997. [Google Scholar] [CrossRef] [Green Version]
- Scherzer, M.T.; Waigel, S.; Donninger, H.; Arumugam, V.; Zacharias, W.; Clark, G.; Siskind, L.J.; Soucy, P.; Beverly, L. Fibroblast-Derived Extracellular Matrices: An Alternative Cell Culture System That Increases Metastatic Cellular Properties. PLoS ONE 2015, 10, e0138065. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dabaghi, M.; Saraei, N.; Carpio, M.B.; Nanduri, V.; Ungureanu, J.; Babi, M.; Chandiramohan, A.; Noble, A.; Revill, S.D.; Zhang, B.; et al. A Robust Protocol for Decellularized Human Lung Bioink Generation Amenable to 2D and 3D Lung Cell Culture. Cells 2021, 10, 1538. [Google Scholar] [CrossRef]
- Leiby, K.L.; Niklason, L.E. Lung Tissue Engineering: Toward a More Deliberate Approach. ACS Biomater. Sci. Eng. 2022. [Google Scholar] [CrossRef]
- Doryab, A.; Heydarian, M.; Amoabediny, G.; Sadroddiny, E.; Mahfouzi, S. Recellularization on Acellular Lung Tissue Scaffold Using Perfusion-Based Bioreactor: An Online Monitoring Strategy. J. Med. Biol. Eng. 2017, 37, 53–62. [Google Scholar] [CrossRef]
- Scarritt, M.E.; Pashos, N.C.; Motherwell, J.M.; Eagle, Z.R.; Burkett, B.J.; Gregory, A.N.; Mostany, R.; Weiss, D.J.; Alvarez, D.F.; Bunnell, B.A. Re-endothelialization of Rat Lung Scaffolds through Passive, Gravity-driven Seeding of Segment-specific Pulmonary Endothelial Cells. J. Tissue Eng. Regen. Med. 2018, 12, e786–e806. [Google Scholar] [CrossRef] [PubMed]
- Ott, H.C.; Clippinger, B.; Conrad, C.; Schuetz, C.; Pomerantseva, I.; Ikonomou, L.; Kotton, D.; Vacanti, J.P. Regeneration and Orthotopic Transplantation of a Bioartificial Lung. Nat. Med. 2010, 16, 927–933. [Google Scholar] [CrossRef]
- Stahl, E.C.; Bonvillain, R.W.; Skillen, C.D.; Burger, B.L.; Hara, H.; Lee, W.; Trygg, C.B.; Didier, P.J.; Grasperge, B.F.; Pashos, N.C.; et al. Evaluation of the Host Immune Response to Decellularized Lung Scaffolds Derived from α-Gal Knockout Pigs in a Non-Human Primate Model. Biomaterials 2018, 187, 93–104. [Google Scholar] [CrossRef]
- Massaro, M.S.; Pálek, R.; Rosendorf, J.; Červenková, L.; Liška, V.; Moulisová, V. Decellularized Xenogeneic Scaffolds in Transplantation and Tissue Engineering: Immunogenicity versus Positive Cell Stimulation. Mater. Sci. Eng. C 2021, 127, 112203. [Google Scholar] [CrossRef]
- Le, A.V.; Hatachi, G.; Beloiartsev, A.; Ghaedi, M.; Engler, A.J.; Baevova, P.; Niklason, L.E.; Calle, E.A. Efficient and Functional Endothelial Repopulation of Whole Lung Organ Scaffolds. ACS Biomater. Sci. Eng. 2017, 3, 2000–2010. [Google Scholar] [CrossRef] [PubMed]
- Aldana, A.A.; Abraham, G.A. Current Advances in Electrospun Gelatin-Based Scaffolds for Tissue Engineering Applications. Int. J. Pharm. 2017, 523, 441–453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Assunção, M.; Dehghan-Baniani, D.; Yiu, C.H.K.; Später, T.; Beyer, S.; Blocki, A. Cell-Derived Extracellular Matrix for Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Burgstaller, G.; Oehrle, B.; Gerckens, M.; White, E.S.; Schiller, H.B.; Eickelberg, O. The Instructive Extracellular Matrix of the Lung: Basic Composition and Alterations in Chronic Lung Disease. Eur. Respir. J. 2017, 50, 1601805. [Google Scholar] [CrossRef] [Green Version]
- Ehrhardt, C.; Kneuer, C.; Fiegel, J.; Hanes, J.; Schaefer, U.; Kim, K.-J.; Lehr, C.-M. Influence of Apical Fluid Volume on the Development of Functional Intercellular Junctions in the Human Epithelial Cell Line 16HBE14o-: Implications for the Use of This Cell Line as an in Vitro Model for Bronchial Drug Absorption Studies. Cell Tissue Res. 2002, 308, 391–400. [Google Scholar] [CrossRef]
- Bosquillon, C.; Madlova, M.; Patel, N.; Clear, N.; Forbes, B. A Comparison of Drug Transport in Pulmonary Absorption Models: Isolated Perfused Rat Lungs, Respiratory Epithelial Cell Lines and Primary Cell Culture. Pharm. Res. 2017, 34, 2532–2540. [Google Scholar] [CrossRef]
- Zhu, M.; Li, W.; Dong, X.; Yuan, X.; Midgley, A.C.; Chang, H.; Wang, Y.; Wang, H.; Wang, K.; Ma, P.X.; et al. In Vivo Engineered Extracellular Matrix Scaffolds with Instructive Niches for Oriented Tissue Regeneration. Nat. Commun. 2019, 10, 4620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingber, D.E. Is It Time for Reviewer 3 to Request Human Organ Chip Experiments Instead of Animal Validation Studies? Adv. Sci. 2020, 7, 2002030. [Google Scholar] [CrossRef] [PubMed]


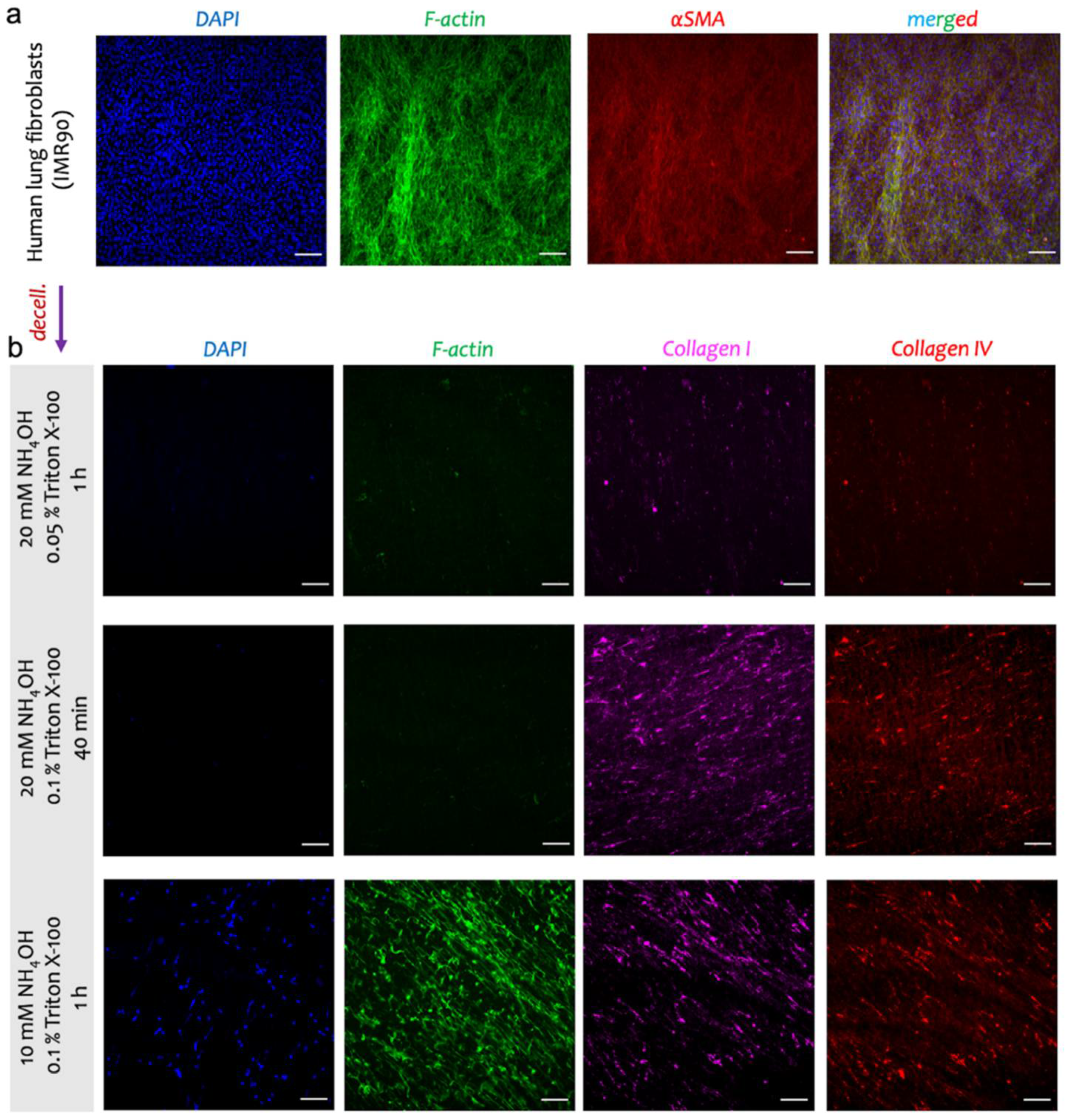
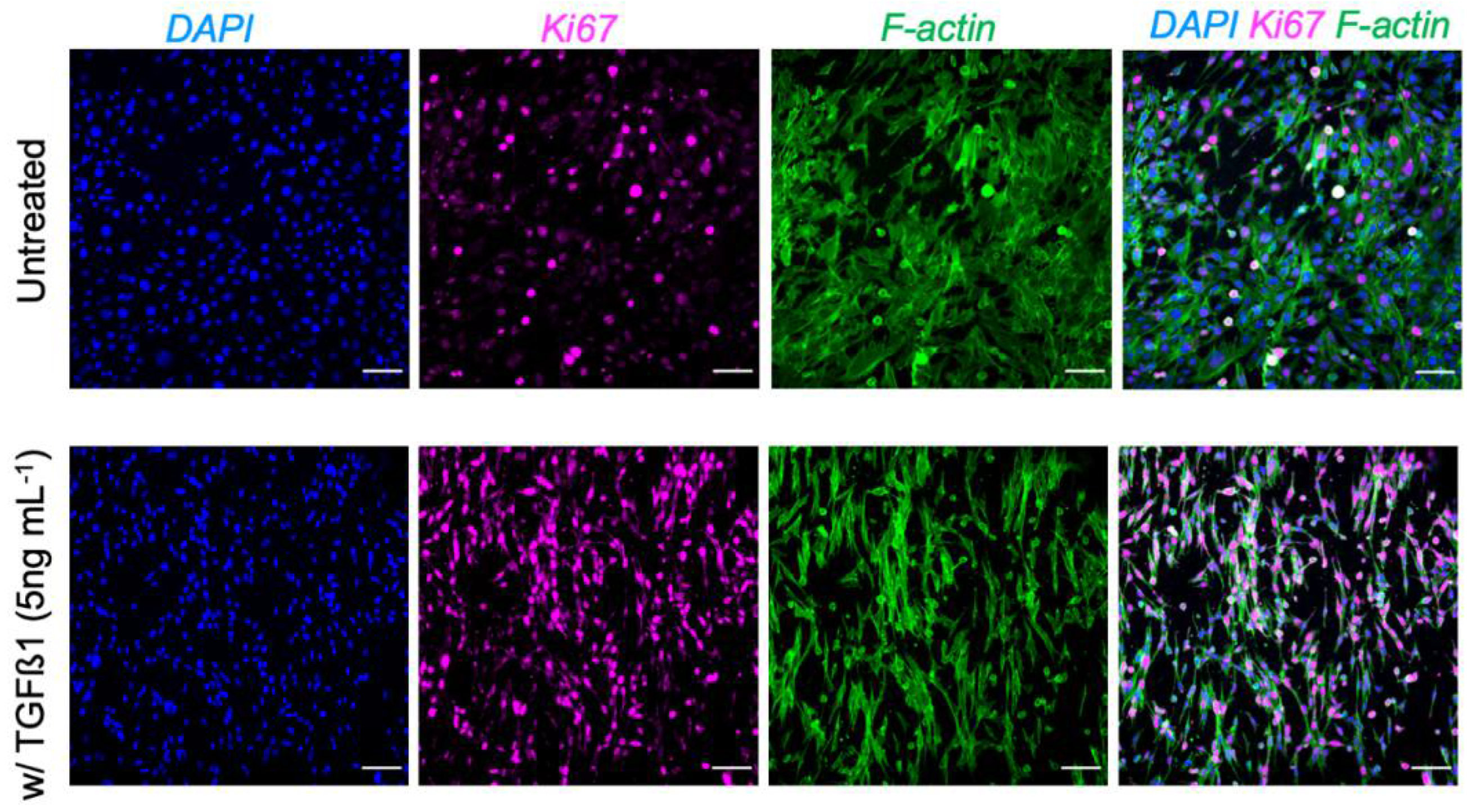
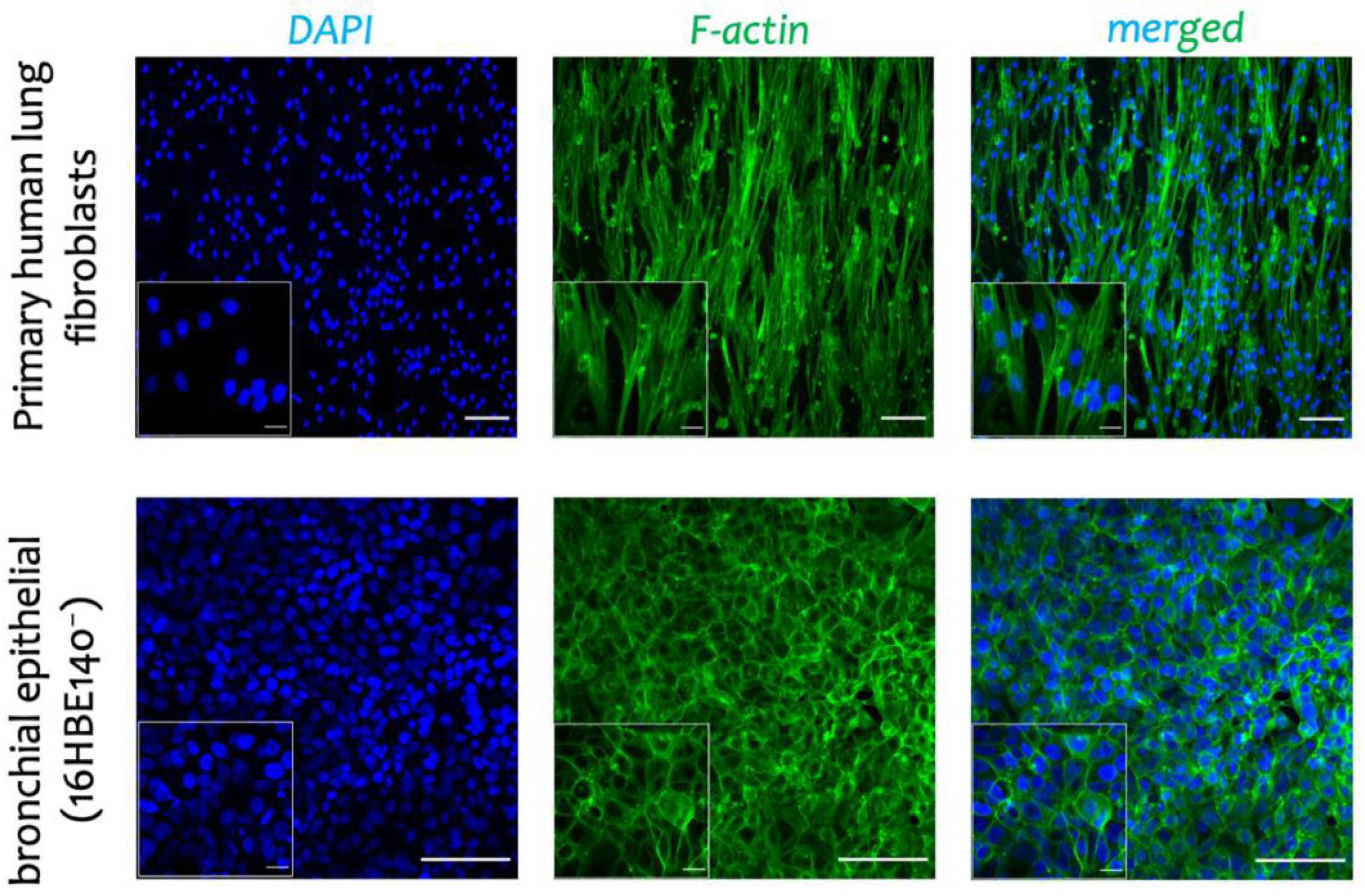
| Protocol # | Description | ||
|---|---|---|---|
| Decellularization | Duration | Post-Treatment | |
| 1 | 20 mM NH4OH, 0.05% Triton X-100 | 1 h | 100 U/mL DNAse I in PBS with Ca2+ and Mg2+ for 1 h |
| 2 | 20 mM NH4OH, 0.1% Triton X-100 | 40 min | |
| 3 | 10 mM NH4OH, 0.1% Triton X-100 | 1 h | |
| 4 | 1 mM CHAPS and 0.1% Triton X-100 | 1 h | |
| 5 | 0.1% SDS and 0.1% Triton X-100 | 1 h | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doryab, A.; Schmid, O. Bioactive Cell-Derived ECM Scaffold Forms a Unique Cellular Microenvironment for Lung Tissue Engineering. Biomedicines 2022, 10, 1791. https://doi.org/10.3390/biomedicines10081791
Doryab A, Schmid O. Bioactive Cell-Derived ECM Scaffold Forms a Unique Cellular Microenvironment for Lung Tissue Engineering. Biomedicines. 2022; 10(8):1791. https://doi.org/10.3390/biomedicines10081791
Chicago/Turabian StyleDoryab, Ali, and Otmar Schmid. 2022. "Bioactive Cell-Derived ECM Scaffold Forms a Unique Cellular Microenvironment for Lung Tissue Engineering" Biomedicines 10, no. 8: 1791. https://doi.org/10.3390/biomedicines10081791
APA StyleDoryab, A., & Schmid, O. (2022). Bioactive Cell-Derived ECM Scaffold Forms a Unique Cellular Microenvironment for Lung Tissue Engineering. Biomedicines, 10(8), 1791. https://doi.org/10.3390/biomedicines10081791






