Skin, Liver, and Kidney Interactions Contribute to Skin Dryness in Aging KK-Ay/Tajcl Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animal Experiments
2.2. Measurement of Dorsal Skin Transept Water Loss and Capacitance
2.3. Preparation and Staining of Dorsal Skin, Kidney, and Liver Samples
2.4. Enzyme-Linked Immunosorbent Assay (ELISA) Analysis
2.5. Western Blotting
2.6. Statistical Analysis
3. Results
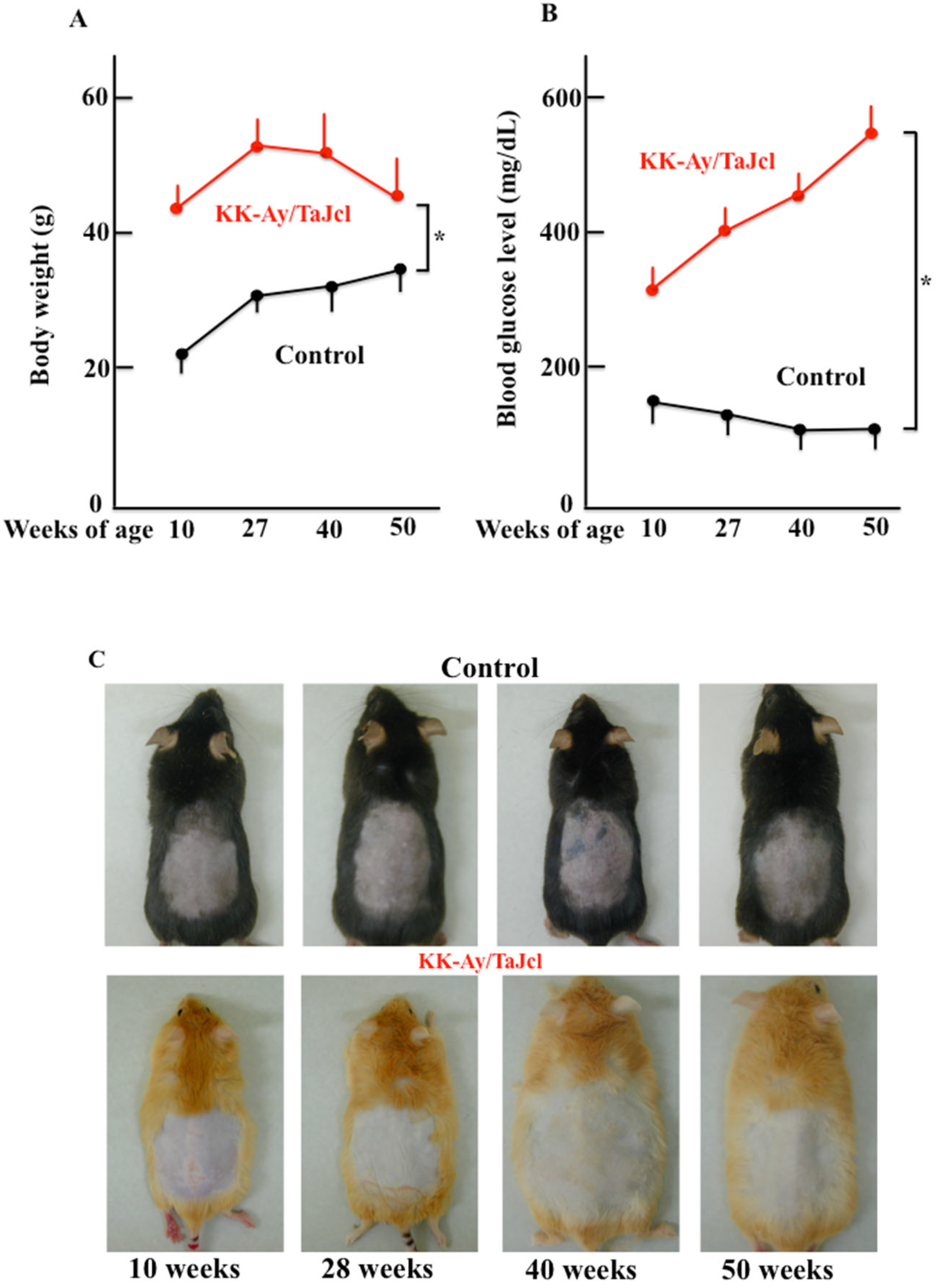
3.1. Effect of Aging on Body Weight, Blood Glucose Level, and Skin Condition in KK-Ay/Tajcl Mice
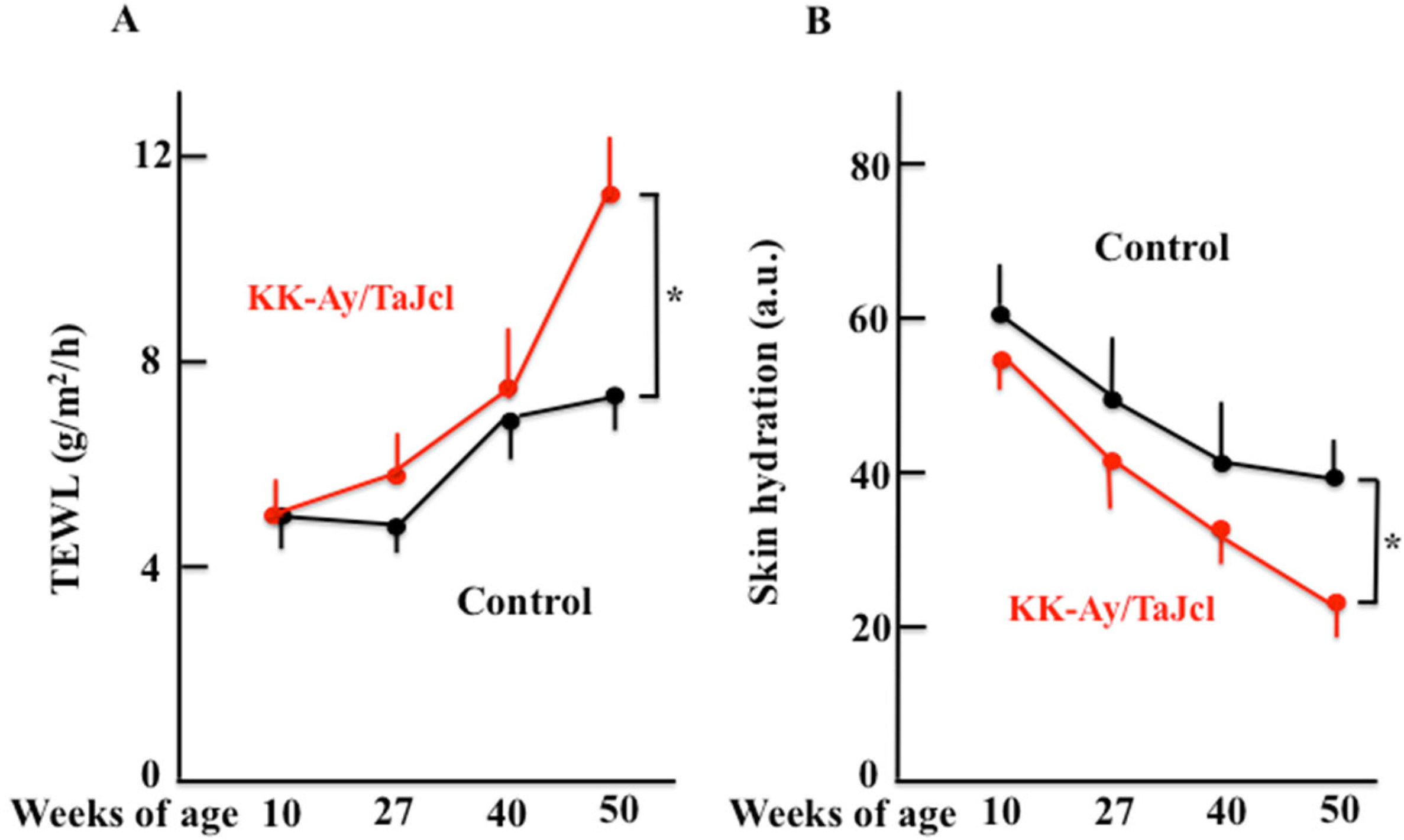
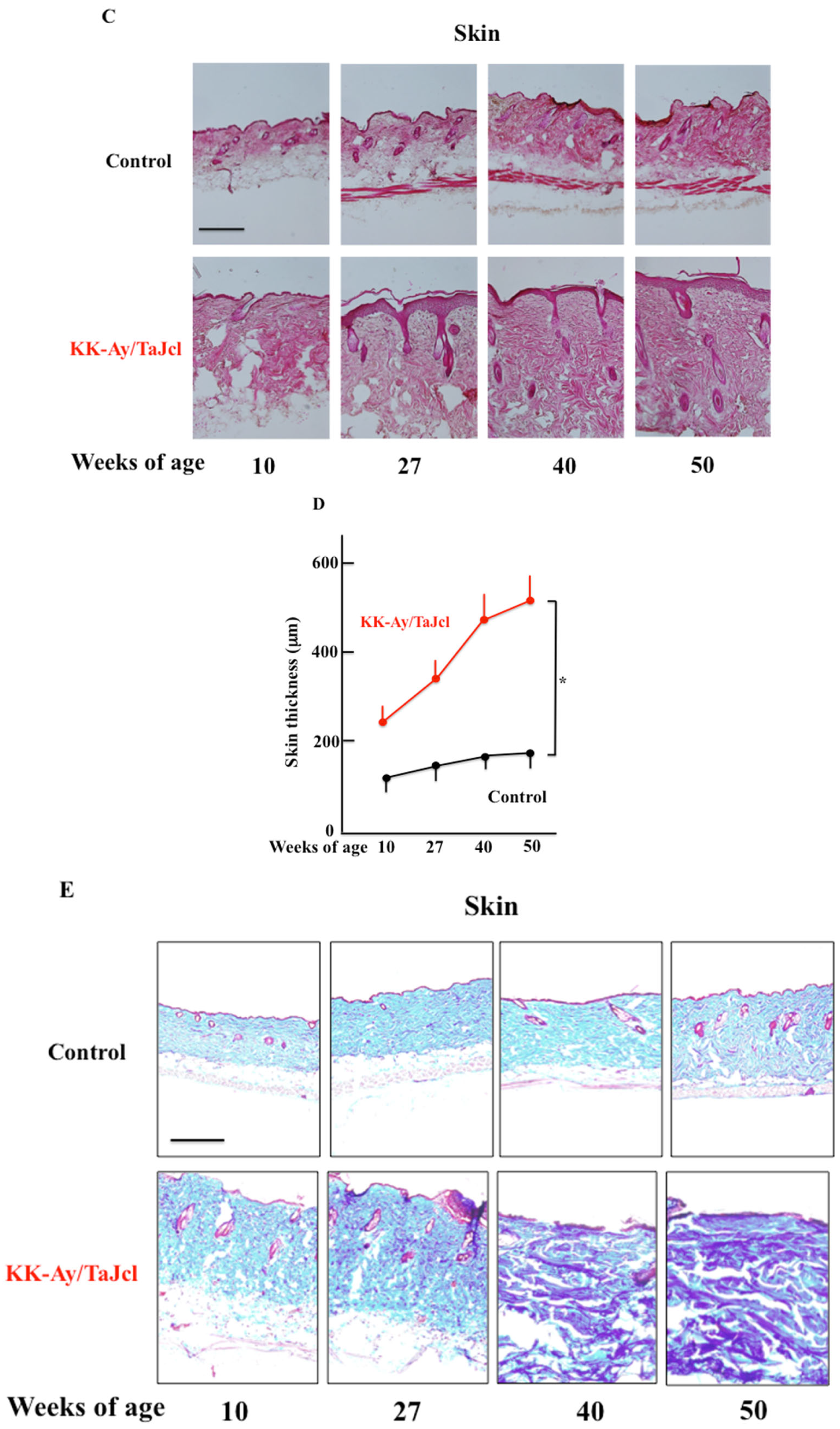
3.2. Effect of Aging on TEWL, Skin Hydration, Skin Thickness, and Mast Cell and Collagen Expression in KK-Ay/TaJcl Mice
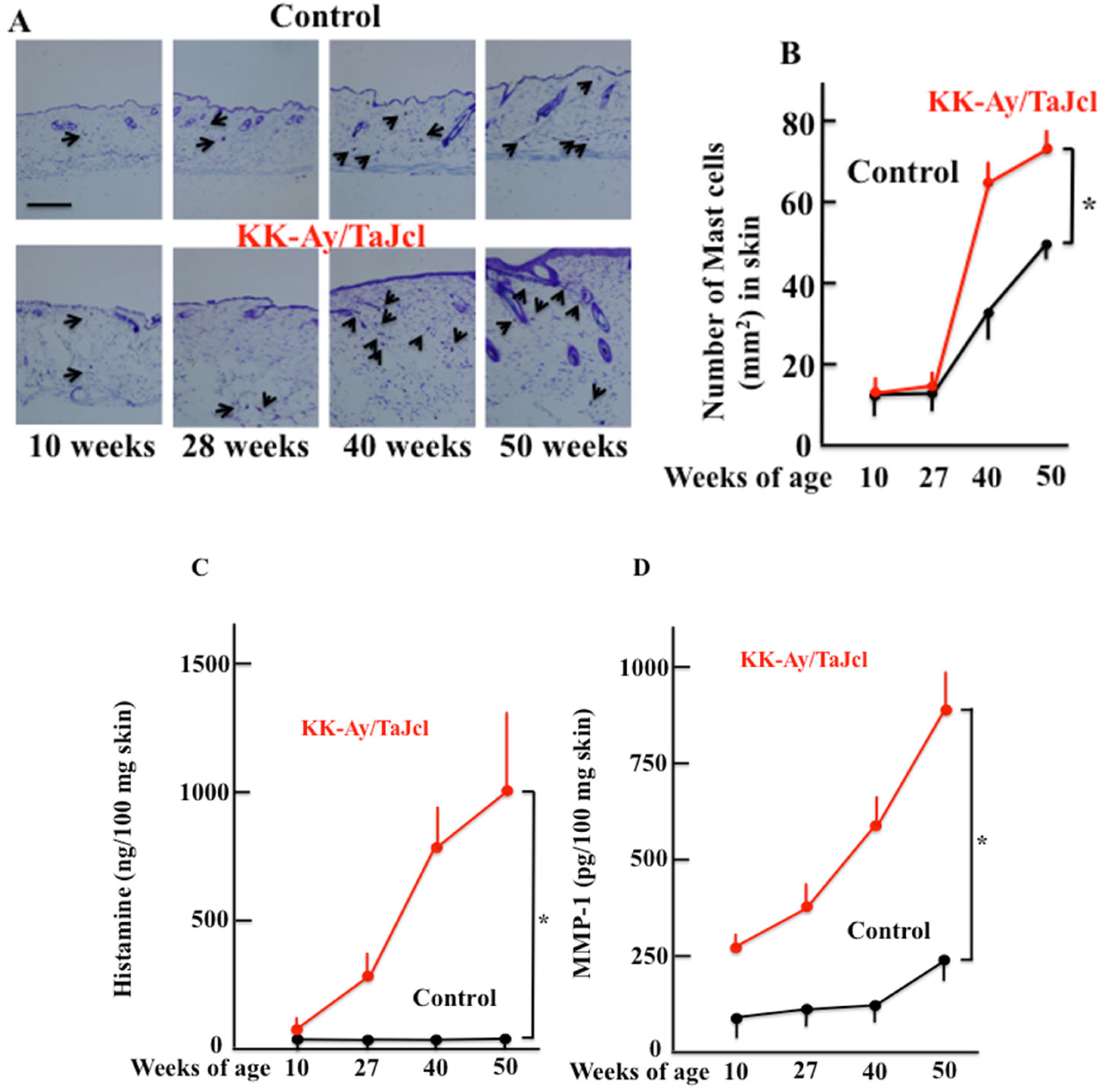
3.3. The Expression of Skin Mast Cell and Skin Histamine and Matrixmetalloproteinase-1 Expression in Aging KK-Ay/TaJcl Mice
3.4. Kidney Condition and Weight, and Blood Creatinine and Hyaluronic Acid Levels in Aging KK-Ay/Tajcl Mice
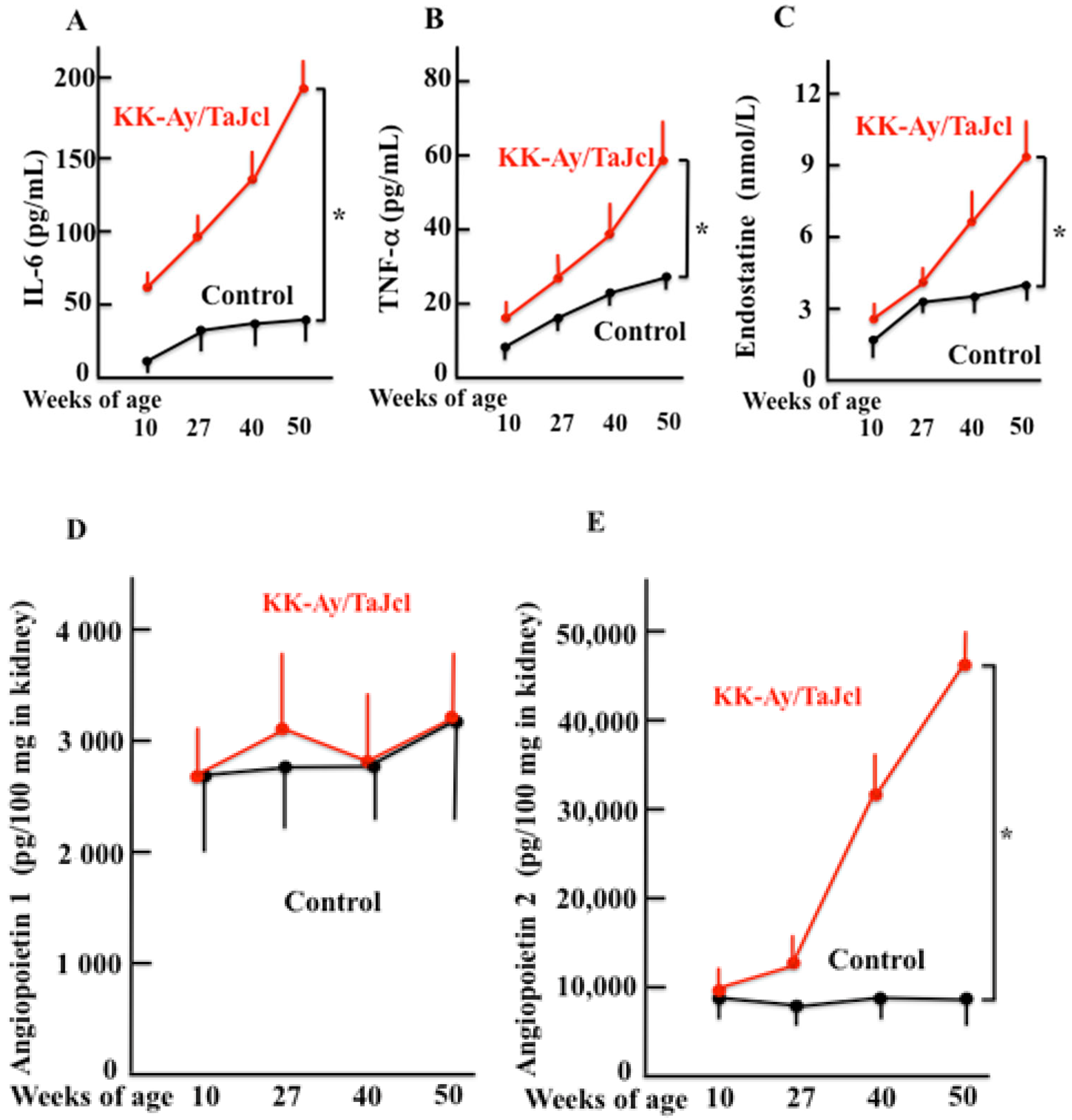
3.5. Aging Effects IL-6, TNF-α and Endostatine Blood Levels and Kidney Angiopoietin 1 and 2 Levels in KK-Ay/Tajcl Mice
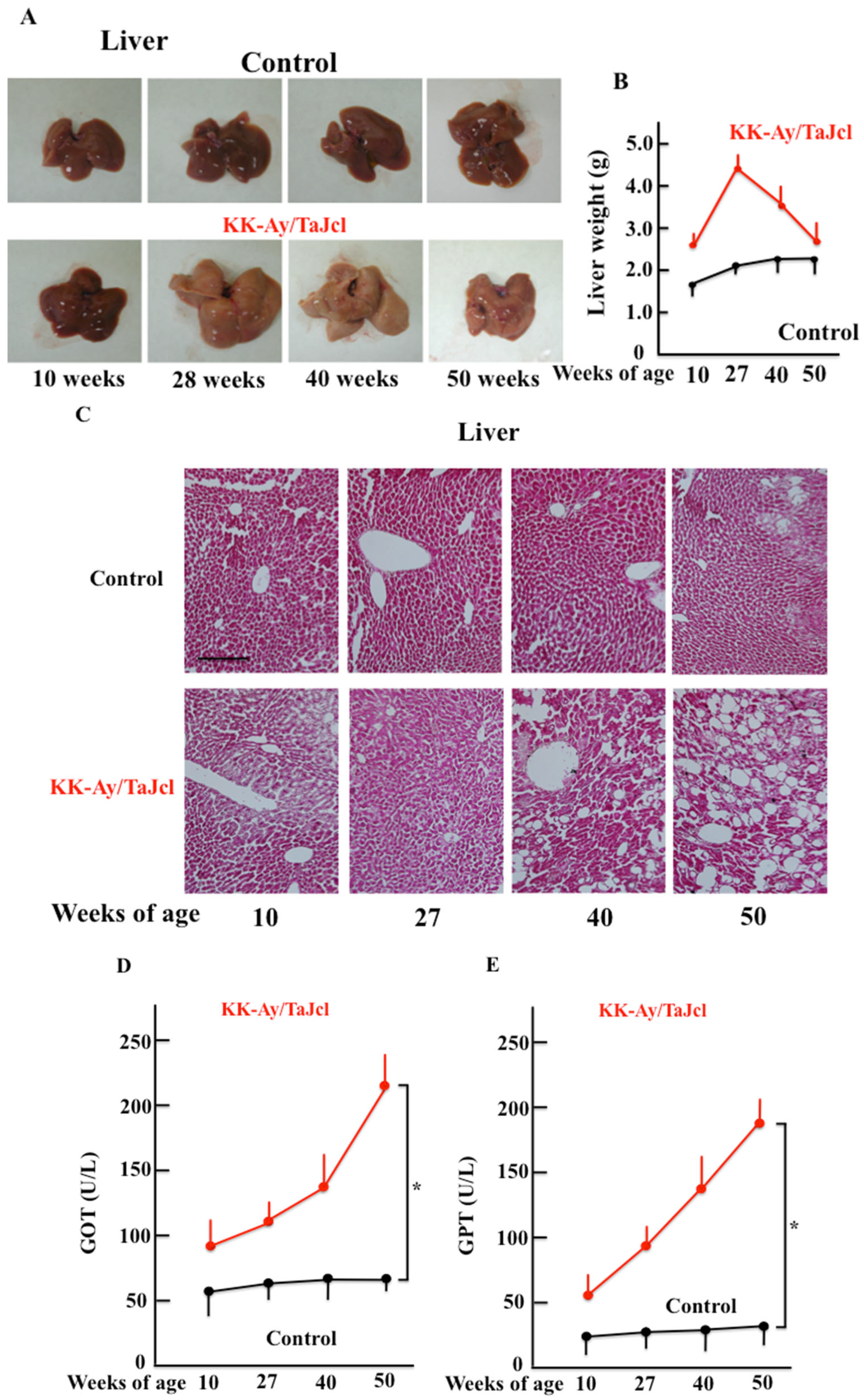
3.6. Liver Condition and Weight, and Glutamic Oxaloacetic Transaminase and Glutamic Pyruvic Transaminase Blood Levels in Response to Aging in KK-Ay/Tajcl Mice
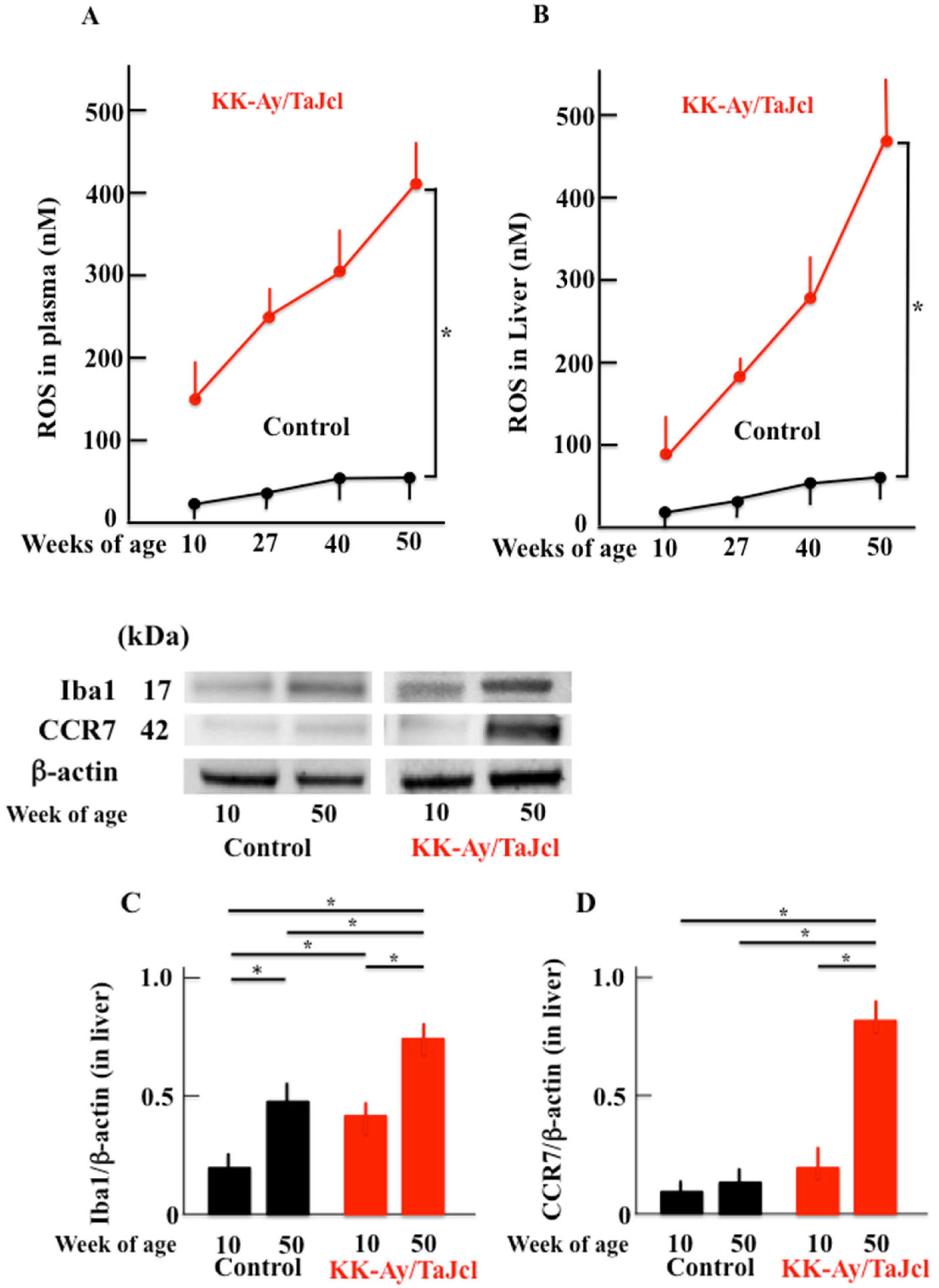
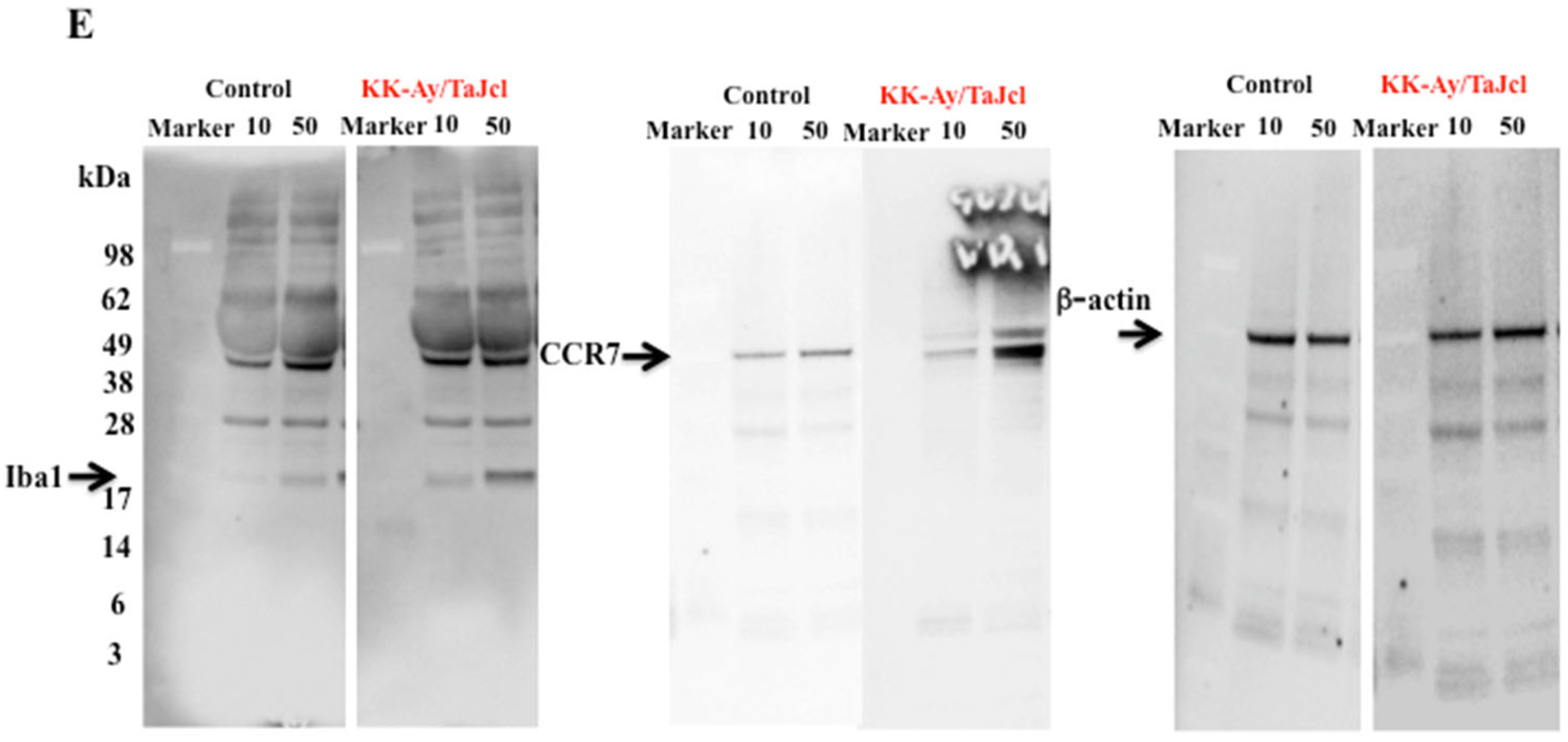
3.7. Effect of Aging on Reactive Oxygen Species and Ionized Calcium-Binding Adapter Molecule 1 and CC-Chemokine Receptor 7 Expression in KK-Ay/Tajcl Mice
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barnett, K.N.; McMurdo, M.E.T.; Ogston, S.A.; Morris, A.D.; Evans, J.M.M. Mortality in people diagnosed with type 2 diabetes at an older age: A systematic review. Age Ageing 2006, 35, 463–468. [Google Scholar] [CrossRef] [Green Version]
- Janghorbani, M.; Mansourian, M.; Hosseini, E. Systematic review and meta-analysis of age at menarche and risk of type 2 diabetes. Acta Diabetol. 2014, 51, 519–528. [Google Scholar] [CrossRef]
- Nanayakkara, N.; Curtis, A.J.; Heritier, S.; Gadowski, A.M.; Pavkov, M.E.; Kenealy, T.; Owens, D.R.; Thomas, R.L.; Song, S.; Wong, J.; et al. Impact of age at type 2 diabetes mellitus diagnosis on mortality and vascular complications: Systematic review and meta-analysis. Diabetologia 2021, 64, 275–287. [Google Scholar] [CrossRef]
- Neilly, J.B.; Martin, A.; Simpson, N.; MacCuish, A.C. Pruritus in diabetes mellitus: Investigation of prevalence and correlation with diabetes control. Diabetes Care. 1986, 9, 273–275. [Google Scholar] [CrossRef]
- Jabbour, S.A. Cutaneous manifestations of endocrine disorders: A guide for dermatologists. Am. J. Clin. Dermatol. 2003, 4, 315–331. [Google Scholar] [CrossRef]
- Wahid, Z.; Kanjee, A. Cutaneous manifestations of diabetes mellitus. J. Pak. Med. Assoc. 1998, 48, 304–305. [Google Scholar]
- Horikawa, T.; Hiramoto, K.; Goto, K.; Sekijima, H.; Ooi, K. Differences in the mechanism of type 1 and type 2 diabetes-induces skin dryness by using model mice. Int. J. Med. Sci. 2021, 18, 474–481. [Google Scholar] [CrossRef]
- Murota, H. Sweating in systemic abnormalities: Uremia and diabetes mellitus. Curr. Probl. Dermatol. 2016, 51, 57–61. [Google Scholar]
- Duque, M.I.; Thevarajah, S.; Chan, Y.H.; Tuttle, A.B.; Freedman, B.I.; Yosipovitch, G. Uremic pruritus is associated with higher kt/V and serum calcium concentration. Clin. Nephrol. 2006, 66, 184–191. [Google Scholar] [CrossRef]
- Horio, F. Studies of genetic and nutrianal factors in disorders of glucose and lipid metabolism. Jpn. Nutr. Food Sci. 2018, 71, 267–274. [Google Scholar]
- Donath, M.Y.; Shoelson, S.E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 2011, 11, 98–107. [Google Scholar] [CrossRef]
- Barel, A.O.; Clarys, P. Study of the stratum corneum barrier function by transept water loss measurements: Comparison between two commercial instruments: Evaporimeter and tewameter. Skin Pharmacol. 1995, 8, 186–195. [Google Scholar] [CrossRef]
- Bekeredjian, R.; Walton, C.B.; MacCannell, K.A.; Ecker, J.; Kruse, F.; Outten, J.T.; Sutcliffe, D.; Gererd, R.D.; Bruick, R.K.; Shohet, R.V.; et al. Conditional HIF-1alpha expression produces a reversible cardiomyopathy. PLoS ONE 2010, 21, e11693. [Google Scholar]
- Yokoyama, S.; Hiramoto, K.; Koyama, M.; Ooi, K. Impaired skin barrier function in mice with colon carcinoma induced by azoxymethane and dextran sodium sulfate. Biol. Pharm. Bull. 2015, 38, 947–950. [Google Scholar] [CrossRef]
- Donate-Correa, J.; Martin-Nunez, E.; Muros-de-Fuentes, M.; Mora-Fernandez, C.; Navarro-Gonzalez, J.F. Inflammatory cytokines in diabetic nephronpathy. J. Diabetes Res. 2015, 2015, 948417. [Google Scholar] [CrossRef]
- Furuta, T.; Saito, T.; Ootaka, T.; Soma, J.; Obara, K.; Yoshinaga, K. The role of macrophages in diabetic glomerulusclerosis. Am. J. Kidney Dis. 1993, 21, 480–485. [Google Scholar] [CrossRef]
- Tsai, Y.C.; Lee, C.S.; Chiu, Y.W.; Kuo, H.T.; Lee, S.C.; Hwang, S.J.; Kuo, M.C.; Chen, H.C. Angiopoiein-2, Angiopoitin-1 and subclinical cardiovascular disease in chronic kidney disease. Sci. Rep. 2016, 19, 39400. [Google Scholar] [CrossRef] [Green Version]
- Tsai, Y.C.; Chiu, Y.W.; Kuo, H.T.; Lee, J.J.; Lee, S.C.; Chen, T.H.; Lin, M.Y.; Hwang, S.J.; Kuo, M.C.; Hsu, Y.L.; et al. The interaction between fluid status and angiopoietin-2 in adverse renal outcomes of chronic kidney disease. PLoS ONE 2017, 23, e0173906. [Google Scholar] [CrossRef] [Green Version]
- Nagarkar, D.R.; Poposki, J.A.; Comeau, M.R.; Biyasheva, A.; Avila, P.C.; Schleimer, R.P.; Kato, A. Airway epithelial cells activate TH2 cytokine production in mast cells through IL-1 and thymic stromal lymphopoietin. J. Allergy Clin. Immunol. 2012, 130, 225–232. [Google Scholar] [CrossRef] [Green Version]
- Nakae, S.; Morita, H.; Ohno, T.; Arae, K.; Matsumoto, K.; Saito, H. Role of interleukin-33 in innate-type immune cells in allergy. Allergol. Int. 2013, 62, 13–20. [Google Scholar] [CrossRef] [Green Version]
- Goto, K.; Hiramoto, K.; Ooi, K. Th2 and Th17 induce dry skin in a mouse model of arthritis. Biol. Pharm. Bull. 2019, 42, 468–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, K.H.; Shackel, N.A.; Gorrell, M.D.; McLennan, S.V.; Twigg, S.M. Diabetes and nonalcoholic fatty liver disease: A pathogenic duo. Endocr. Rev. 2013, 34, 84–129. [Google Scholar] [CrossRef] [PubMed]
- Karkucinska-Wieckowska, A.; Simoes, I.C.M.; Kalinowski, P.; Lebiedzinska-Arciszewska, M.; Zieniewicz, K.; Milkiewicz, P.; Gorska-Ponikowska, M.; Pinton, P.; Malik, A.N.; Krawczyk, M.; et al. Mitochondria, oxidative stress and nonalcoholic fatty liver disease: A complex relationship. Eur. J. Clin. Investig. 2022, 52, e13622. [Google Scholar] [CrossRef]
- Wang, C.; Ma, C.; Gong, L.; Guo, Y.; Fu, K.; Zhang, Y.; Zhou, H.; Li, Y. Macrophage polarization and its role in liver disease. Front. Immunol. 2021, 14, 803037. [Google Scholar] [CrossRef]
- Xu, L.; Liu, W.; Bai, F.; Xu, Y.; Liang, X.; Ma, C.; Gao, L. Hepatic macrophage as a key player in fatty liver disease. Front. Immunol. 2021, 9, 708978. [Google Scholar] [CrossRef]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.N.; Saltiel, A.R. Inflammatory links between obesity and metabolic disease. J. Clin. Investig. 2011, 121, 2111–2117. [Google Scholar] [CrossRef] [Green Version]
- Ueki, K.; Kadowaki, T. The other sweet fave of XBP-1. Nat. Med. 2011, 17, 246–248. [Google Scholar] [CrossRef]
- Hofmann, B.; Adam, A.C.; Jacobs, K.; Riemer, M.; Erbs, C.; Bushnaq, H.; Simm, A.; Silber, R.E.; Santos, A.N. Advanced glycation end product associated skin autofluorescence: A mirror of vascular function? Exp. Gerontol. 2013, 48, 38–44. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hiramoto, K.; Goto, K.; Tanaka, S.; Horikawa, T.; Ooi, K. Skin, Liver, and Kidney Interactions Contribute to Skin Dryness in Aging KK-Ay/Tajcl Mice. Biomedicines 2022, 10, 2648. https://doi.org/10.3390/biomedicines10102648
Hiramoto K, Goto K, Tanaka S, Horikawa T, Ooi K. Skin, Liver, and Kidney Interactions Contribute to Skin Dryness in Aging KK-Ay/Tajcl Mice. Biomedicines. 2022; 10(10):2648. https://doi.org/10.3390/biomedicines10102648
Chicago/Turabian StyleHiramoto, Keiichi, Kenji Goto, Shota Tanaka, Tsuneki Horikawa, and Kazuya Ooi. 2022. "Skin, Liver, and Kidney Interactions Contribute to Skin Dryness in Aging KK-Ay/Tajcl Mice" Biomedicines 10, no. 10: 2648. https://doi.org/10.3390/biomedicines10102648
APA StyleHiramoto, K., Goto, K., Tanaka, S., Horikawa, T., & Ooi, K. (2022). Skin, Liver, and Kidney Interactions Contribute to Skin Dryness in Aging KK-Ay/Tajcl Mice. Biomedicines, 10(10), 2648. https://doi.org/10.3390/biomedicines10102648






