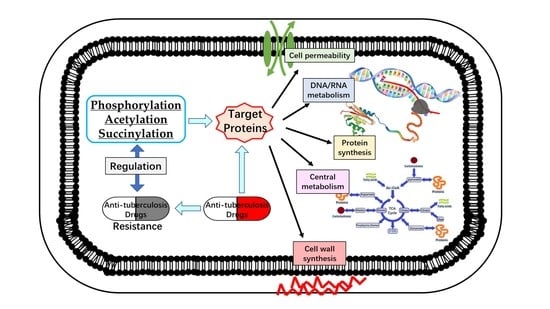
The Role of Phosphorylation and Acylation in the Regulation of Drug Resistance in Mycobacterium tuberculosis
Abstract
1. Introduction
2. Lys Acylation
2.1. Acetylation
2.2. Succinylation
3. Ser/Thr/Tyr Phosphorylation
3.1. Phosphorylation of Proteins Related to Cell Wall Synthesis
3.2. Phosphorylation of Proteins Related to DNA Metabolism and Transcription
3.3. Phosphorylation of Proteins Related to Translation
3.4. Phosphorylation of Proteins That Participate in Other Biological Processes
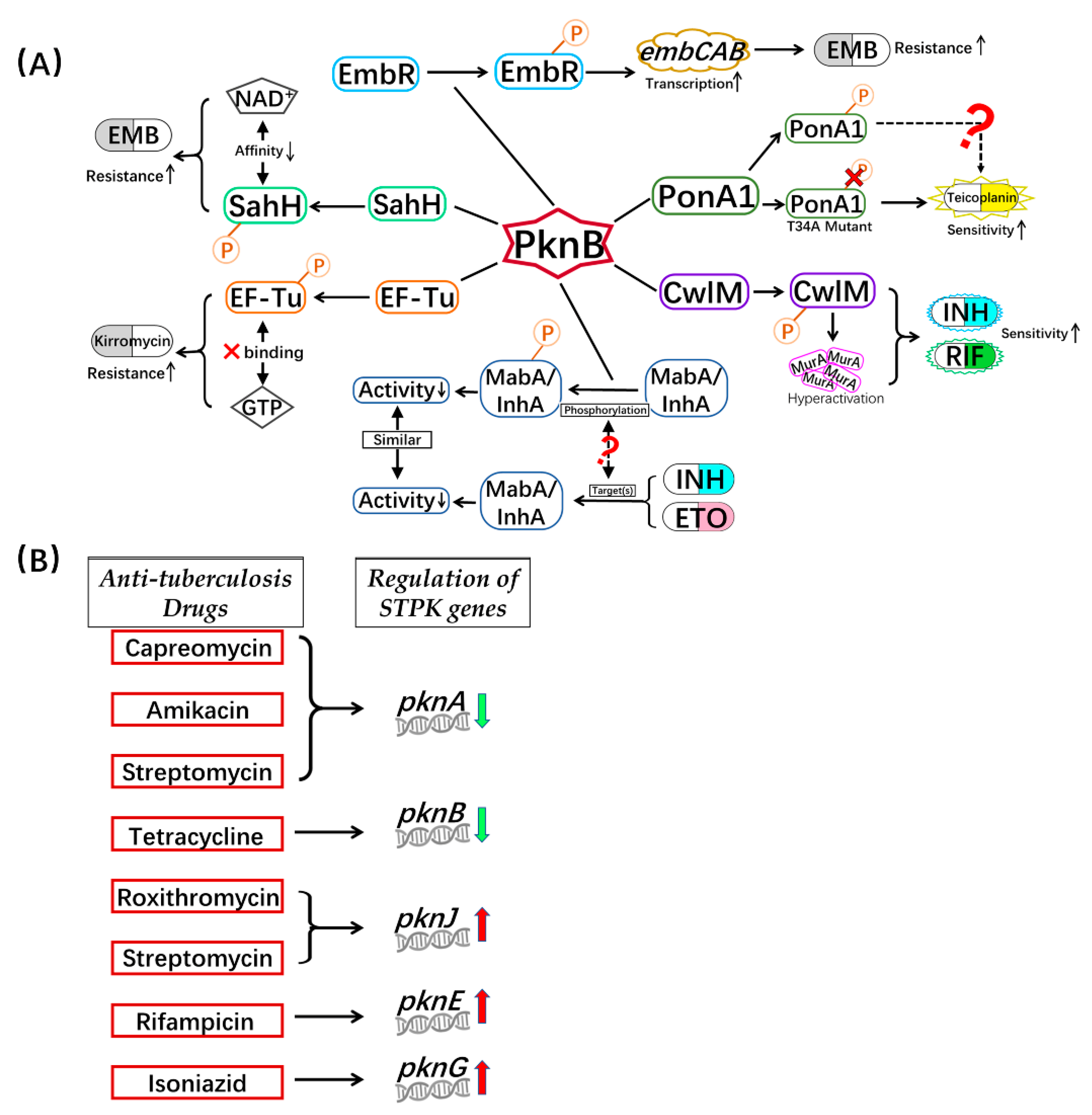
3.5. STPKs Involved in Drug Resistance
4. Summary and Prospect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Global Tuberculosis Report 2021; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Alffenaar, J.; Stocker, S.; Forsman, L.D.; Garcia-Prats, A.; Heysell, S.; Aarnoutse, R.; Akkerman, O.; Aleksa, A.; van Altena, R.; de Oñata, W.A. Clinical standards for the dosing and management of TB drugs. Int. J. Tuberc. Lung Dis. 2022, 26, 483–499. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Forsman, L.D.; Bao, Z.; Xie, Y.; Ning, Z.; Schön, T.; Bruchfeld, J.; Xu, B.; Alffenaar, J.-W.; Hu, Y. Drug exposure and susceptibility of second-line drugs correlate with treatment response in patients with multidrug-resistant tuberculosis: A multicentre prospective cohort study in China. Eur. J. Respir. Med. 2022, 59, 2101925. [Google Scholar] [CrossRef] [PubMed]
- Te Brake, L.H.; de Knegt, G.J.; de Steenwinkel, J.E.; Van Dam, T.J.; Burger, D.M.; Russel, F.G.; van Crevel, R.; Koenderink, J.B.; Aarnoutse, R.E. The role of efflux pumps in tuberculosis treatment and their promise as a target in drug development: Unraveling the black box. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 271–291. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, S.H. Evolution of drug resistance in Mycobacterium tuberculosis: Clinical and molecular perspective. Antimicrob. Agents Chemother. 2002, 46, 267–274. [Google Scholar] [CrossRef]
- Gygli, S.M.; Borrell, S.; Trauner, A.; Gagneux, S. Antimicrobial resistance in Mycobacterium tuberculosis: Mechanistic and evolutionary perspectives. FEMS Microbiol. Rev. 2017, 41, 354–373. [Google Scholar] [CrossRef]
- Buriánková, K.; Doucet-Populaire, F.; Dorson, O.; Gondran, A.; Ghnassia, J.-C.; Weiser, J.; Pernodet, J.-L. Molecular basis of intrinsic macrolide resistance in the Mycobacterium tuberculosis complex. Antimicrob. Agents Chemother. 2004, 48, 143–150. [Google Scholar] [CrossRef]
- Kandpal, M.; Aggarwal, S.; Jamwal, S.; Yadav, A.K. Emergence of drug resistance in mycobacterium and other bacterial pathogens: The posttranslational modification perspective. In Drug Resistance in Bacteria, Fungi, Malaria, and Cancer; Springer: Berlin/Heidelberg, Germany, 2017; pp. 209–231. [Google Scholar]
- Arora, G.; Bothra, A.; Prosser, G.; Arora, K.; Sajid, A. Role of post-translational modifications in the acquisition of drug resistance in Mycobacterium tuberculosis. FEBS J. 2021, 288, 3375–3393. [Google Scholar] [CrossRef]
- Nakedi, K.C.; Nel, A.J.; Garnett, S.; Blackburn, J.M.; Soares, N.C. Comparative Ser/Thr/Tyr phosphoproteomics between two mycobacterial species: The fast growing Mycobacterium smegmatis and the slow growing Mycobacterium bovis BCG. Front. Microbiol. 2015, 6, 237. [Google Scholar] [CrossRef]
- Birhanu, A.G.; Yimer, S.A.; Holm-Hansen, C.; Norheim, G.; Aseffa, A.; Abebe, M.; Tønjum, T. Nε-and O-Acetylation in Mycobacterium tuberculosis lineage 7 and lineage 4 strains: Proteins involved in bioenergetics, virulence, and antimicrobial resistance are acetylated. J. Proteome. Res. 2017, 16, 4045–4059. [Google Scholar] [CrossRef]
- Manluan, S.; Hongsen, G.; Guoliang, L.; Jing, G.; Xude, W.; Xian-En, Z.; Jiaoyu, D. Lysine acetylation regulates the activity of Escherichia coli S-adenosylmethionine synthase. Acta Biochim. Biophys. Sin. 2016, 48, 723–731. [Google Scholar] [CrossRef]
- James, A.M.; Hoogewijs, K.; Logan, A.; Hall, A.R.; Ding, S.; Fearnley, I.M.; Murphy, M.P. Non-enzymatic N-acetylation of lysine residues by acetylCoA often occurs via a proximal S-acetylated thiol intermediate sensitive to glyoxalase II. Cell Rep. 2017, 18, 2105–2112. [Google Scholar] [CrossRef] [PubMed]
- Punetha, A.; Green, K.D.; Garzan, A.; Chandrika, N.T.; Willby, M.J.; Pang, A.H.; Hou, C.; Holbrook, S.Y.; Krieger, K.; Posey, J.E. Structure-based design of haloperidol analogues as inhibitors of acetyltransferase Eis from Mycobacterium tuberculosis to overcome kanamycin resistance. RSC Med. Chem. 2021, 12, 1894–1909. [Google Scholar] [CrossRef] [PubMed]
- Zaunbrecher, M.A.; Sikes, R.D.; Metchock, B.; Shinnick, T.M.; Posey, J.E. Overexpression of the chromosomally encoded aminoglycoside acetyltransferase eis confers kanamycin resistance in Mycobacterium tuberculosis. Proc. Natl. Acad. Sci. USA 2009, 106, 20004–20009. [Google Scholar] [CrossRef] [PubMed]
- Houghton, J.L.; Green, K.D.; Pricer, R.E.; Mayhoub, A.S.; Garneau-Tsodikova, S. Unexpected N-acetylation of capreomycin by mycobacterial Eis enzymes. J. Antimicrob. Chemother. 2013, 68, 800–805. [Google Scholar] [CrossRef] [PubMed]
- Bi, J.; Wang, Y.; Yu, H.; Qian, X.; Wang, H.; Liu, J.; Zhang, X. Modulation of central carbon metabolism by acetylation of isocitrate lyase in Mycobacterium tuberculosis. Sci. Rep. 2017, 7, 44826. [Google Scholar] [CrossRef]
- Ghiraldi-Lopes, L.D.; Campanerut-Sá, P.A.Z.; Evaristo, G.P.C.; Meneguello, J.E.; Fiorini, A.; Baldin, V.P.; de Souza, E.M.; de Lima Scodro, R.B.; Siqueira, V.L.; Cardoso, R.F. New insights on Ethambutol Targets in Mycobacterium tuberculosis. Infect. Disord. Drug Targets 2019, 19, 73–80. [Google Scholar] [CrossRef]
- Arun, K.; Madhavan, A.; Abraham, B.; Balaji, M.; Sivakumar, K.; Nisha, P.; Kumar, R.A. Acetylation of isoniazid is a novel mechanism of isoniazid resistance in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2020, 65, e00456-20. [Google Scholar] [CrossRef]
- Choudhury, M.; Virivinti, J.; Kandi, S.; Sritharan, V.; Sritharan, M. Th2 immune response by the iron-regulated protein HupB of Mycobacterium tuberculosis. Indian J. Tuberc. 2022, 69, 90–99. [Google Scholar] [CrossRef]
- Kalra, P.; Mishra, S.K.; Kaur, S.; Kumar, A.; Prasad, H.K.; Sharma, T.K.; Tyagi, J.S. G-quadruplex-forming DNA aptamers inhibit the DNA-binding function of HupB and Mycobacterium tuberculosis entry into host cells. Mol. Ther. Nucleic Acids 2018, 13, 99–109. [Google Scholar] [CrossRef]
- Sakatos, A.; Babunovic, G.H.; Chase, M.R.; Dills, A.; Leszyk, J.; Rosebrock, T.; Bryson, B.; Fortune, S.M. Posttranslational modification of a histone-like protein regulates phenotypic resistance to isoniazid in mycobacteria. Sci. Adv. 2018, 4, eaao1478. [Google Scholar] [CrossRef]
- Green, K.D.; Biswas, T.; Pang, A.H.; Willby, M.J.; Reed, M.S.; Stuchlik, O.; Pohl, J.; Posey, J.E.; Tsodikov, O.V.; Garneau-Tsodikova, S. Acetylation by Eis and deacetylation by Rv1151c of Mycobacterium tuberculosis HupB: Biochemical and structural insight. Biochemistry 2018, 57, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Bisht, D. Secretory proteome analysis of streptomycin-resistant Mycobacterium tuberculosis clinical isolates. SLAS Discov. 2017, 22, 1229–1238. [Google Scholar] [CrossRef] [PubMed]
- Boutte, C.C.; Baer, C.E.; Papavinasasundaram, K.; Liu, W.; Chase, M.R.; Meniche, X.; Fortune, S.M.; Sassetti, C.M.; Ioerger, T.R.; Rubin, E.J. A cytoplasmic peptidoglycan amidase homologue controls mycobacterial cell wall synthesis. Elife 2016, 5, e14590. [Google Scholar] [CrossRef] [PubMed]
- Weinert, B.; Scholz, C.; Wagner, S.A.; Iesmantavicius, V.; Su, D.; Daniel, J.A.; Choudhary, C. Lysine succinylation is a frequently occurring modification in prokaryotes and eukaryotes and extensively overlaps with acetylation. Cell Rep. 2013, 4, 842–851. [Google Scholar] [CrossRef]
- Xie, L.; Liu, W.; Li, Q.; Chen, S.; Xu, M.; Huang, Q.; Zeng, J.; Zhou, M.; Xie, J. First succinyl-proteome profiling of extensively drug-resistant Mycobacterium tuberculosis revealed involvement of succinylation in cellular physiology. J. Proteome Res. 2015, 14, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, X.; Wang, B.; Fu, L.; Huo, F.; Gao, T.; Pang, Y.; Lu, Y.; Li, Q. Molecular Characteristic of Both Levofloxacin and Moxifloxacin Resistance in Mycobacterium tuberculosis from Individuals Diagnosed with Preextensive Drug-Resistant Tuberculosis. Microb. Drug Resist. 2021, 12, 280–287. [Google Scholar] [CrossRef]
- Siregar, T.A.P.; Prombutara, P.; Kanjanasirirat, P.; Kunkaew, N.; Tubsuwan, A.; Boonmee, A.; Palaga, T.; Khumpanied, T.; Borwornpinyo, S.; Chaiprasert, A. The autophagy-resistant Mycobacterium tuberculosis Beijing strain upregulates KatG to evade starvation-induced autophagic restriction. Pathog. Dis. 2022, 80, ftac004. [Google Scholar] [CrossRef]
- Wei, C.-J.; Lei, B.; Musser, J.M.; Tu, S.-C. Isoniazid activation defects in recombinant Mycobacterium tuberculosis catalase-peroxidase (KatG) mutants evident in InhA inhibitor production. Antimicrob. Agents Chemother. 2003, 47, 670–675. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, J. Phosphoproteomics of Mycobacterium-host interaction and inspirations for novel measures against tuberculosis. Cell. Signal. 2022, 91, 110238. [Google Scholar] [CrossRef]
- Sherman, D.R.; Grundner, C. Agents of change–concepts in Mycobacterium tuberculosis Ser/Thr/Tyr phosphosignalling. Mol. Microbiol. 2014, 94, 231–241. [Google Scholar] [CrossRef]
- Kusebauch, U.; Ortega, C.; Ollodart, A.; Rogers, R.S.; Sherman, D.R.; Moritz, R.L.; Grundner, C. Mycobacterium tuberculosis supports protein tyrosine phosphorylation. Proc. Natl. Acad. Sci. USA 2014, 111, 9265–9270. [Google Scholar] [CrossRef] [PubMed]
- Prisic, S.; Husson, R.N. Mycobacterium tuberculosis serine/threonine protein kinases. Microbiol. Spectr. 2014, 2. [Google Scholar] [CrossRef]
- Qu, D.; Zhao, X.; Sun, Y.; Wu, F.-L.; Tao, S.-C. Mycobacterium tuberculosis Thymidylyltransferase RmlA Is Negatively Regulated by Ser/Thr Protein Kinase PknB. Front. Microbiol. 2021, 12, 643951. [Google Scholar] [CrossRef] [PubMed]
- Alsayed, S.S.; Beh, C.C.; Foster, N.R.; Payne, A.D.; Yu, Y.; Gunosewoyo, H. Kinase targets for mycolic acid biosynthesis in Mycobacterium tuberculosis. Curr. Mol. Pharmacol. 2019, 12, 27–49. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Sammartino, J.C.; Costantino, L.; Gelain, A.; Meneghetti, F.; Villa, S.; Chiarelli, L.R. An overview on the potential antimycobacterial agents targeting serine/threonine protein kinases from Mycobacterium tuberculosis. Curr. Top. Med. Chem. 2019, 19, 646–661. [Google Scholar] [CrossRef] [PubMed]
- Hanwarinroj, C.; Thongdee, P.; Sukchit, D.; Taveepanich, S.; Kamsri, P.; Punkvang, A.; Ketrat, S.; Saparpakorn, P.; Hannongbua, S.; Suttisintong, K. In silico design of novel quinazoline-based compounds as potential Mycobacterium tuberculosis PknB inhibitors through 2D and 3D-QSAR, molecular dynamics simulations combined with pharmacokinetic predictions. J. Mol. Graph. Model. 2022, 115, 108231. [Google Scholar] [CrossRef]
- Wlodarchak, N.; Feltenberger, J.B.; Ye, Z.; Beczkiewicz, J.; Procknow, R.; Yan, G.; King Jr, T.M.; Golden, J.E.; Striker, R. Engineering Selectivity for Reduced Toxicity of Bacterial Kinase Inhibitors Using Structure-Guided Medicinal Chemistry. ACS Med. Chem. Lett. 2021, 12, 228–235. [Google Scholar] [CrossRef]
- Shamma, F.; Rego, E.H.; Boutte, C.C. Mycobacterial Serine/Threonine phosphatase PstP is phospho-regulated and localized to mediate control of cell wall metabolism. Mol. Microbiol. 2022, 118, 47–60. [Google Scholar] [CrossRef]
- Kieser, K.J.; Boutte, C.C.; Kester, J.C.; Baer, C.E.; Barczak, A.K.; Meniche, X.; Chao, M.C.; Rego, E.H.; Sassetti, C.M.; Fortune, S.M. Phosphorylation of the peptidoglycan synthase PonA1 governs the rate of polar elongation in mycobacteria. PLoS Pathog. 2015, 11, e1005010. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, Q.; Tang, X.; An, Y.; Li, S.; Xu, H.; Li, Y.; Wang, X.; Luan, W.; Wang, Y. Effects of CwlM on autolysis and biofilm formation in Mycobacterium tuberculosis and Mycobacterium smegmatis. Int. J. Med. Microbiol. 2019, 309, 73–83. [Google Scholar] [CrossRef]
- Xu, L.; Wu, D.; Liu, L.; Zheng, Q.; Song, Y.; Ye, L.; Sha, S.; Kang, J.; Xin, Y.; Ma, Y. Characterization of mycobacterial UDP-N-acetylglucosamine enolpyruvyle transferase (MurA). Res. Microbiol. 2014, 165, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Bancroft, P.J.; Turapov, O.; Jagatia, H.; Arnvig, K.B.; Mukamolova, G.V.; Green, J. Coupling of peptidoglycan synthesis to central metabolism in mycobacteria: Post-transcriptional control of CWLM by aconitase. Cell Rep. 2020, 32, 108209. [Google Scholar] [CrossRef] [PubMed]
- Shamma, F.; Papavinasasundaram, K.; Quintanilla, S.Y.; Bandekar, A.; Sassetti, C.; Boutte, C.C. Phosphorylation on PstP regulates cell wall metabolism and antibiotic tolerance in Mycobacterium smegmatis. J. Bacteriol. 2021, 203, e00563-20. [Google Scholar] [CrossRef] [PubMed]
- Filippova, E.V.; Kieser, K.J.; Luan, C.H.; Wawrzak, Z.; Kiryukhina, O.; Rubin, E.J.; Anderson, W.F. Crystal structures of the transpeptidase domain of the Mycobacterium tuberculosis penicillin-binding protein PonA1 reveal potential mechanisms of antibiotic resistance. FEBS J. 2016, 283, 2206–2218. [Google Scholar] [CrossRef] [PubMed]
- Alsayed, S.S.; Lun, S.; Payne, A.; Bishai, W.R.; Gunosewoyo, H. Facile synthesis and antimycobacterial activity of isoniazid, pyrazinamide and ciprofloxacin derivatives. Chem. Biol. Drug Des. 2021, 97, 1137–1150. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.S.; Bhole, R.P.; Khedekar, P.B.; Chikhale, R.V. Mycobacterium enoyl acyl carrier protein reductase (InhA): A key target for antitubercular drug discovery. Bioorg. Chem. 2021, 115, 105242. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey North, E.; Jackson, M.; Lee, R.E. New approaches to target the mycolic acid biosynthesis pathway for the development of tuberculosis therapeutics. Curr. Pharm. Des. 2014, 20, 4357–4378. [Google Scholar] [CrossRef]
- Li, M.; Huang, Q.; Zhang, W.; Cao, Y.; Wang, Z.; Zhao, Z.; Zhang, X.; Zhang, J. A Novel Acyl-AcpM-Binding Protein Confers Intrinsic Sensitivity to Fatty Acid Synthase Type II Inhibitors in Mycobacterium smegmatis. Front. Microbiol. 2022, 13, 846722. [Google Scholar] [CrossRef]
- Marrakchi, H.; Ducasse, S.; Labesse, G.; Montrozier, H.; Margeat, E.; Emorine, L.; Charpentier, X.; Daffé, M.; Quémard, A.K. MabA (FabG1), a Mycobacterium tuberculosis protein involved in the long-chain fatty acid elongation system FAS-II. Microbiology 2002, 148, 951–960. [Google Scholar] [CrossRef]
- Takayama, K.; Wang, C.; Besra, G.S. Pathway to synthesis and processing of mycolic acids in Mycobacterium tuberculosis. Clin. Microbiol. Rev. 2005, 18, 81–101. [Google Scholar] [CrossRef]
- Veyron-Churlet, R.; Zanella-Cléon, I.; Cohen-Gonsaud, M.; Molle, V.; Kremer, L. Phosphorylation of the Mycobacterium tuberculosis β-ketoacyl-acyl carrier protein reductase MabA regulates mycolic acid biosynthesis. J. Biol. Chem. 2010, 285, 12714–12725. [Google Scholar] [CrossRef] [PubMed]
- Veyron-Churlet, R.; Molle, V.; Taylor, R.C.; Brown, A.K.; Besra, G.S.; Zanella-Cléon, I.; Fütterer, K.; Kremer, L. The Mycobacterium tuberculosis β-ketoacyl-acyl carrier protein synthase III activity is inhibited by phosphorylation on a single threonine residue. J. Biol. Chem. 2009, 284, 6414–6424. [Google Scholar] [CrossRef] [PubMed]
- Molle, V.; Gulten, G.; Vilchèze, C.; Veyron-Churlet, R.; Zanella-Cléon, I.; Sacchettini, J.C.; Jacobs, W.R., Jr.; Kremer, L. Phosphorylation of InhA inhibits mycolic acid biosynthesis and growth of Mycobacterium tuberculosis. Mol. Microbiol. 2010, 78, 1591–1605. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.M.; Kohl, T.A.; Omar, S.V.; Hedge, J.; Elias, C.D.O.; Bradley, P.; Iqbal, Z.; Feuerriegel, S.; Niehaus, K.E.; Wilson, D.J. Whole-genome sequencing for prediction of Mycobacterium tuberculosis drug susceptibility and resistance: A retrospective cohort study. Lancet Infect. Dis. 2015, 15, 1193–1202. [Google Scholar] [CrossRef]
- Ando, H.; Miyoshi-Akiyama, T.; Watanabe, S.; Kirikae, T. A silent mutation in mabA confers isoniazid resistance on Mycobacterium tuberculosis. Mol. Microbiol. 2014, 91, 538–547. [Google Scholar] [CrossRef]
- Vilchèze, C.; Jacobs, W.R., Jr. Resistance to isoniazid and ethionamide in Mycobacterium tuberculosis: Genes, mutations, and causalities. Microbiol. Spectr. 2014, 2. [Google Scholar] [CrossRef]
- Abrahams, K.A.; Chung, C.-w.; Ghidelli-Disse, S.; Rullas, J.; Rebollo-López, M.J.; Gurcha, S.S.; Cox, J.A.; Mendoza, A.; Jiménez-Navarro, E.; Martínez-Martínez, M.S. Identification of KasA as the cellular target of an anti-tubercular scaffold. Nat Commun 2016, 7, 12581. [Google Scholar] [CrossRef]
- Slayden, R.; Barry, C., 3rd. The role of KasA and KasB in the biosynthesis of meromycolic acids and isoniazid resistance in Mycobacterium tuberculosis. Tuberculosis 2002, 82, 149–160. [Google Scholar] [CrossRef]
- Lata, M.; Sharma, D.; Deo, N.; Tiwari, P.K.; Bisht, D.; Venkatesan, K. Proteomic analysis of ofloxacin-mono resistant Mycobacterium tuberculosis isolates. J. Proteom. 2015, 127, 114–121. [Google Scholar] [CrossRef]
- Warner, D.F.; Rock, J.M.; Fortune, S.M.; Mizrahi, V. DNA replication fidelity in the mycobacterium tuberculosis complex. Adv. Exp. Med. Biol. 2017, 1019, 247–262. [Google Scholar] [CrossRef]
- Wipperman, M.F.; Heaton, B.E.; Nautiyal, A.; Adefisayo, O.; Evans, H.; Gupta, R.; van Ditmarsch, D.; Soni, R.; Hendrickson, R.; Johnson, J. Mycobacterial mutagenesis and drug resistance are controlled by phosphorylation-and cardiolipin-mediated inhibition of the RecA coprotease. Mol. Cell 2018, 72, 152–161.e7. [Google Scholar] [CrossRef] [PubMed]
- Lingaraju, S.; Rigouts, L.; Gupta, A.; Lee, J.; Umubyeyi, A.N.; Davidow, A.L.; German, S.; Cho, E.; Lee, J.-i.; Cho, S.-N. Geographic differences in the contribution of ubiA mutations to high-level ethambutol resistance in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2016, 60, 4101–4105. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Gupta, M.; Krupa, A.; Srinivasan, N.; Singh, Y. EmbR, a regulatory protein with ATPase activity, is a substrate of multiple serine/threonine kinases and phosphatase in Mycobacterium tuberculosis. FEBS J. 2006, 273, 2711–2721. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Gupta, M.; Pathak, M.; Gupta, N.; Koul, A.; Sarangi, S.; Baweja, R.; Singh, Y. Transcriptional control of the mycobacterial embCAB operon by PknH through a regulatory protein, EmbR, in vivo. J. Bacteriol. 2006, 188, 2936–2944. [Google Scholar] [CrossRef]
- Prieri, M.; Frita, R.; Probst, N.; Sournia-Saquet, A.; Bourotte, M.; Déprez, B.; Baulard, A.R.; Willand, N. Efficient analoging around ethionamide to explore thioamides bioactivation pathways triggered by boosters in Mycobacterium tuberculosis. Eur. J. Med. Chem. 2018, 159, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Mugumbate, G.; Mendes, V.; Blaszczyk, M.; Sabbah, M.; Papadatos, G.; Lelievre, J.; Ballell, L.; Barros, D.; Abell, C.; Blundell, T.L. Target identification of Mycobacterium tuberculosis phenotypic hits using a concerted chemogenomic, biophysical, and structural approach. Front. Pharmacol. 2017, 8, 681. [Google Scholar] [CrossRef]
- Leiba, J.; Carrère-Kremer, S.; Blondiaux, N.; Dimala, M.M.; Wohlkönig, A.; Baulard, A.; Kremer, L.; Molle, V. The Mycobacterium tuberculosis transcriptional repressor EthR is negatively regulated by Serine/Threonine phosphorylation. Biochem. Biophys. Res. Commun. 2014, 446, 1132–1138. [Google Scholar] [CrossRef]
- Sun, X.; Ge, F.; Xiao, C.-L.; Yin, X.-F.; Ge, R.; Zhang, L.-H.; He, Q.-Y. Phosphoproteomic analysis reveals the multiple roles of phosphorylation in pathogenic bacterium Streptococcus pneumoniae. J. Proteome Res. 2010, 9, 275–282. [Google Scholar] [CrossRef]
- Sajid, A.; Arora, G.; Gupta, M.; Singhal, A.; Chakraborty, K.; Nandicoori, V.K.; Singh, Y. Interaction of Mycobacterium tuberculosis elongation factor Tu with GTP is regulated by phosphorylation. J. Bacteriol. 2011, 193, 5347–5358. [Google Scholar] [CrossRef]
- Singhal, A.; Arora, G.; Sajid, A.; Maji, A.; Bhat, A.; Virmani, R.; Upadhyay, S.; Nandicoori, V.K.; Sengupta, S.; Singh, Y. Regulation of homocysteine metabolism by Mycobacterium tuberculosis S-adenosylhomocysteine hydrolase. Sci. Rep. 2013, 3, 2264. [Google Scholar] [CrossRef]
- Corrales, R.M.; Leiba, J.; Cohen-Gonsaud, M.; Molle, V.; Kremer, L. Mycobacterium tuberculosis S-adenosyl-l-homocysteine hydrolase is negatively regulated by Ser/Thr phosphorylation. Biochem. Biophys. Res. Commun. 2013, 430, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Argyrou, A.; Jin, L.; Siconilfi-Baez, L.; Angeletti, R.H.; Blanchard, J.S. Proteome-wide profiling of isoniazid targets in Mycobacterium tuberculosis. Biochemistry 2006, 45, 13947–13953. [Google Scholar] [CrossRef] [PubMed]
- Wehenkel, A.; Bellinzoni, M.; Graña, M.; Duran, R.; Villarino, A.; Fernandez, P.; Andre-Leroux, G.; England, P.; Takiff, H.; Cerveñansky, C. Mycobacterial Ser/Thr protein kinases and phosphatases: Physiological roles and therapeutic potential. Biochim. Biophys. Acta Proteins Proteom. 2008, 1784, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Ge, P.; Lei, Z.; Yu, Y.; Lu, Z.; Qiang, L.; Chai, Q.; Zhang, Y.; Zhao, D.; Li, B.; Pang, Y. M. tuberculosis PknG manipulates host autophagy flux to promote pathogen intracellular survival. Autophagy 2021, 7, 576–594. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; DeJesus, M.A.; Rücker, N.; Engelhart, C.A.; Wright, M.G.; Healy, C.; Lin, K.; Wang, R.; Park, S.W.; Ioerger, T.R. Chemical genetic interaction profiling reveals determinants of intrinsic antibiotic resistance in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2017, 61, e01334-17. [Google Scholar] [CrossRef]
- Bellinzoni, M.; Wehenkel, A.M.; Durán, R.; Alzari, P.M. Novel mechanistic insights into physiological signaling pathways mediated by mycobacterial Ser/Thr protein kinases. Genes Immun. 2019, 20, 383–393. [Google Scholar] [CrossRef]
- Torfs, E.; Piller, T.; Cos, P.; Cappoen, D. Opportunities for overcoming Mycobacterium tuberculosis drug resistance: Emerging mycobacterial targets and host-directed therapy. Int. J. Mol. Sci. 2019, 20, 2868. [Google Scholar] [CrossRef]
- Nakedi, K.C.; Calder, B.; Banerjee, M.; Giddey, A.; Nel, A.J.; Garnett, S.; Blackburn, J.M.; Soares, N.C. Identification of novel physiological substrates of Mycobacterium bovis BCG protein kinase G (PknG) by label-free quantitative phosphoproteomics. Mol. Cell Proteom. 2018, 17, 1365–1377. [Google Scholar] [CrossRef]
- Iqbal, I.K.; Bajeli, S.; Akela, A.K.; Kumar, A. Bioenergetics of Mycobacterium: An emerging landscape for drug discovery. Pathogens 2018, 7, 24. [Google Scholar] [CrossRef]
- Wagner, T.; André-Leroux, G.; Hindie, V.; Barilone, N.; Lisa, M.-N.; Hoos, S.; Raynal, B.; Vulliez-Le Normand, B.; O’hare, H.M.; Bellinzoni, M. Structural insights into the functional versatility of an FHA domain protein in mycobacterial signaling. Sci. Signal. 2019, 12, 9504. [Google Scholar] [CrossRef]
- Ventura, M.; Rieck, B.; Boldrin, F.; Degiacomi, G.; Bellinzoni, M.; Barilone, N.; Alzaidi, F.; Alzari, P.M.; Manganelli, R.; O’Hare, H.M. GarA is an essential regulator of metabolism in Mycobacterium tuberculosis. Mol. Microbiol. 2013, 90, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Wolff, K.A.; de la Peña, A.H.; Nguyen, H.T.; Pham, T.H.; Amzel, L.M.; Gabelli, S.B.; Nguyen, L. A redox regulatory system critical for mycobacterial survival in macrophages and biofilm development. PLoS Pathog. 2015, 11, e1004839. [Google Scholar] [CrossRef] [PubMed]
- Kidwai, S.; Bouzeyen, R.; Chakraborti, S.; Khare, N.; Das, S.; Priya Gosain, T.; Behura, A.; Meena, C.L.; Dhiman, R.; Essafi, M. NU-6027 inhibits growth of Mycobacterium tuberculosis by targeting protein kinase D and protein kinase G. Antimicrob. Agents Chemother. 2019, 63, e00996-19. [Google Scholar] [CrossRef]
- Singhal, A.; Arora, G.; Virmani, R.; Kundu, P.; Khanna, T.; Sajid, A.; Misra, R.; Joshi, J.; Yadav, V.; Samanta, S. Systematic analysis of mycobacterial acylation reveals first example of acylation-mediated regulation of enzyme activity of a bacterial phosphatase. J. Biol. Chem. 2015, 290, 26218–26234. [Google Scholar] [CrossRef]
- Choukate, K.; Chaudhuri, B. Structural basis of self-assembly in the lipid-binding domain of mycobacterial polar growth factor Wag31. IUCrJ 2020, 7, 767–776. [Google Scholar] [CrossRef]
- Singh, V.; Dhar, N.; Pató, J.; Kolly, G.S.; Korduláková, J.; Forbak, M.; Evans, J.C.; Székely, R.; Rybniker, J.; Palčeková, Z. Identification of aminopyrimidine-sulfonamides as potent modulators of Wag31-mediated cell elongation in mycobacteria. Mol. Microbiol. 2017, 103, 13–25. [Google Scholar] [CrossRef]
- Birhanu, A.G.; Gómez-Munñoz, M.; Kalayou, S.; Riaz, T.; Lutter, T.; Yimer, S.A.; Abebe, M.; Tønjum, T. Proteome Profiling of Mycobacterium tuberculosis Cells Exposed to Nitrosative Stress. ACS Omega. 2022, 7, 3470–3482. [Google Scholar] [CrossRef]


| Protein | Functions | Post-Translational Modifications | Related Antibiotics-Resistance |
|---|---|---|---|
| KatG | catalase peroxidase, INH-resistance | Phosphorylation, acetylation, succinylation | INH |
| MetK | S-adenosylmethionine synthase, decreased expression after INH treatment | Phosphorylation, acetylation | INH |
| InhA | Enoyl Acyl carrier protein reductase, inhA transcription change after INH treatment | Phosphorylation, acetylation | INH |
| MabA | Beta-ketoacyl-acyl carrier protein reductase, participate in INH tolerance together with InhA | Phosphorylation, acetylation | INH |
| EF-Tu | elongation factor Tu, decreased expression after INH treatment | Phosphorylation, acetylation | INH, kirromycin |
| KasA and KasB | beta-ketoacyl carrier protein synthases | Phosphorylation | INH, RIF, ETO |
| HupB | essential regulatory protein, Lys acetylation results in INH resistance | Acetylation, lysine methylation | INH |
| SahH | S-adenosylhomocysteine hydrolase, INH-NADP complex will be affected after its phosphorylation | Phosphorylation, acetylation | INH |
| EmbR | transcription factor for embCAB, tolerance to EMB will be changed by its phosphorylation | Phosphorylation, acetylation | EMB |
| RecA and LexA | phosphorylation of RecA inhibits its binding with LexA repressor | Phosphorylation | RIF |
| GyrA and GyrB | DNA gyrase | Succinylation, phosphorylation | FQs |
| GroEL2 | molecular chaperone protein, increased expression after LZD, streptomycin, ofloxacin treatment | Phosphorylation, acetylation | LZD, streptomycin, ofloxacin |
| Eis | enhanced intracellular survival protein | Acetylation | kanamycin, capreomycin |
| EthA | monooxygenase | Acetylation | ETO |
| EthR | EthA transcriptional regulator | Phosphorylation | ETO |
| PonA1 | penicillin-binding protein, transglycoside activity of PonA1 will be regulated by phosphorylation | Phosphorylation | penicillin V, meropenem |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, M.; Ge, S.; Li, Z. The Role of Phosphorylation and Acylation in the Regulation of Drug Resistance in Mycobacterium tuberculosis. Biomedicines 2022, 10, 2592. https://doi.org/10.3390/biomedicines10102592
Sun M, Ge S, Li Z. The Role of Phosphorylation and Acylation in the Regulation of Drug Resistance in Mycobacterium tuberculosis. Biomedicines. 2022; 10(10):2592. https://doi.org/10.3390/biomedicines10102592
Chicago/Turabian StyleSun, Manluan, Sai Ge, and Zhaoyang Li. 2022. "The Role of Phosphorylation and Acylation in the Regulation of Drug Resistance in Mycobacterium tuberculosis" Biomedicines 10, no. 10: 2592. https://doi.org/10.3390/biomedicines10102592
APA StyleSun, M., Ge, S., & Li, Z. (2022). The Role of Phosphorylation and Acylation in the Regulation of Drug Resistance in Mycobacterium tuberculosis. Biomedicines, 10(10), 2592. https://doi.org/10.3390/biomedicines10102592