Abstract
Background: Breast cancer is the most common form of cancer in women worldwide. Advances in the early diagnosis and treatment of cancer in the last decade have progressively decreased the cancer mortality rate, and in recent years, immunotherapy has emerged as a relevant tool against cancer. HER2+ and triple-negative breast cancers (TNBCs) are considered more immunogenic and suitable for this kind of treatment due to the higher rate of tumor-infiltrating lymphocytes (TILs) and programmed death ligand 1 (PD-L1) expression. In TNBC, genetic aberrations further favor immunogenicity due to more neo-antigens in cancer cells. Methods: This review summarizes the principal ongoing conventional and investigational immunotherapies in breast cancer. Particularly, immune checkpoint inhibitors (ICIs) and their use alone or combined with DNA damage repair inhibitors (DDRis) are described. Then, the issue on immunotherapy with monoclonal antibodies against HER-2 family receptors is updated. Other investigational immunotherapies include a new schedule based on the interferon beta-interleukin-2 sequence that was given in ER+ metastatic breast cancer patients concomitant with anti-estrogen therapy, which surprisingly showed promising results. Results: Based on the scientific literature and our own findings, the current evaluation of tumor immunogenicity and the conventional model of adjuvant chemotherapy (CT) are questioned. Conclusions: A novel strategy based on additional prolonged adjuvant immunotherapy combined with hormone therapy or alternated with CT is proposed.
1. Introduction
Breast cancer is the most common cancer in women worldwide. The estimated number of new cases in 2020 was 2,260,000, while that for 2025 is predicted to be 2,500,000 [1]. Thus far, due to relevant advances in early diagnosis and treatment of the disease, a 38% decrease in breast cancer mortality has been attained [2]. However, the occurrences of therapy resistance and tumor heterogeneity remain major challenges to avoiding tumor relapse and progression and obtaining definitive success in breast cancer therapy [3]. For more than a decade, a new molecular classification has divided breast cancer into four specific subtypes with different behaviors and responsiveness to therapy. This classification includes luminal A (ER+PR+, HER2/neu negative, and Ki-67 low), luminal B (ER+PR+, HER2/neu negative, and Ki-67 high), HER2+ (ER+ PR+/−, Ki-67 high/low, and HER2+), and triple-negative (ER−PR−HER2−) subtypes. ER+ and/or PR+ endocrine-dependent breast cancer represents up to more than 70% of the molecular subtypes and the incidence increases with older age [4,5]. Endocrine therapy with aromatase inhibitors (Ais), selective ER modulators (SERMs), and selective ER down-regulators (SERDs) is the mainstay for luminal A and B treatment, with luminal A being less aggressive than all other subtypes [5,6]. HER2+ comprehends about 20% of all breast cancers; it is more aggressive than luminal but less aggressive than triple-negative breast cancer (TNBC) [7,8]. HER2 receptor overexpression is a molecular characteristic that accounts for a higher invasiveness and recurrence rate [3,9]. TNBC does not express ER, PR, or HER2 receptors [10] and roughly represents 15–20% of all breast cancers [11,12]. TNBC, which is the subtype with the worst prognosis, has the highest rate of relapses and mortality likely due to the lack of a specific molecular target, and high heterogeneity [8,13,14]. Because of this, neoadjuvant chemotherapy (NACT) is the standard treatment for TNBC [13]. Overall, breast cancer, particularly the ER+ HER2− sub-group, due to the relatively low rate of tumor-infiltrating lymphocytes (TILs) in the tumor microenvironment (TME), is considered scarcely or moderately immunogenic. However, HER2+ and TNBC, both of which are associated with higher TILs, are considered more immunogenic [15]. In particular, the median rate of TIL infiltration reported in TNBC was 20%, while those in HER2+ and HR+HER2− breast cancer were 16% and 6%, respectively [15]. Further, HER2 overexpression and, more recently, an uncovered higher programmed death ligand 1 (PD-L1) expression in HER2+ and TNBC [16], along with the occurrence of germline breast cancer (gBRCA1/2) genetic defects and homologous recombination deficiency (HRD) in the latter, suggest both are suitable for an immunotherapeutic strategy. This review examines the major ongoing conventional and investigational immune therapies in breast cancer. Particularly, the immune checkpoint inhibitors (ICIs) and their use, alone or combined with DNA damage repair inhibitors (DDRis), which is under investigation for breast cancer, are described first. Then, conventional passive immunotherapy with monoclonal antibodies against HER-2 family receptors is updated. Other investigational immune therapies, including a new immunotherapy schedule successfully tested in a clinical trial carried out in ER+ metastatic breast cancer patients, are briefly reported. Furthermore, based on the scientific literature and our findings, the current evaluation of tumor immunogenicity and the conventional model of adjuvant chemotherapy (CT) are questioned. Finally, a novel strategy based on additional prolonged adjuvant immunotherapy combined with conventional hormones or CT is proposed.
1.1. Conventional Immune Therapies in Breast Cancer
The immune checkpoint blockade (ICB) by ICIs is an emerging active immunotherapy for TNBC, while in the HER+ breast cancer subset, passive immunotherapy with monoclonal antibodies against HER-2 family receptors has been carried out for a long time.
1.1.1. ICIs and ICB in Breast Cancer Therapy
Immune checkpoints are one of the mechanisms by which the immune system promotes immune cells anergy or apoptosis to maintain the immune response homeostasis [16,17]. Immune checkpoints are inhibitory proteins/receptors usually present on the surface of effector immune cells, or T or NK cells, and their ligands are transmembrane proteins expressed on antigen-presenting cells (APCs), macrophages, or tumor cells, as well as B and T lymphocytes; the ligands are also expressed in some nonlymphoid tissues [18]. Cells can either start an immune response through signals or maintain immune tolerance; namely, the binding of the PD-L1 with its programmed death-1 (PD-1) receptor provides a suppressive signal to T lymphocytes with decreased proliferation and a decrease in the immune response [19,20]. T-cell activation is controlled by further antigen-independent co-stimulatory signals such as cluster of differentiation 28 (CD28) and cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4). CD28 and CTLA-4 compete for the same ligands: CD80 and CD86 [21]. However, CTLA-4 has a higher avidity than CD28 with regard to these ligands and induces a negative regulatory signal to the T cell [21]. Particularly and contrary to CD28 signals, which are required for T-cell activation and cytokine secretion, CTLA-4 signaling inhibits T-cell activation. Both CD28 and CTLA-4 can be stimulated by the CD80 (B7-1) and CD86 (B7-2) ligands that are expressed on activated APCs, leading either to T-cell proliferation and differentiation when the CD28:CD80/CD86 ratio is elevated [22], or to T-cell inactivation and anergy in the case of an increased CTLA-4:CD80/CD86 ratio [22]. As CTLA-4 binds to CD80/86 with very high affinity, this receptor mediates immune suppression by competing for CD28 as well as by inducing CD80/86 removal from the APCs’ surface [23]. For this reason, by blocking the interaction between CTLA-4 and CD80/86 ligands, CTLA-4 inhibitors can prevent T-cells exhaustion and boost the antitumor T-cell response [24]. PD-1, CTLA-4, and CD28 receptors are present on activated effector T cells that interact with their ligands, the B7 family members: B7-1 (CD80), B7-2 (CD-86), B7-H1 or CD274 (PD-L1), B7-H2, B7-H3, B7-DC or CD273 (PD-L2), and B7-H4 [21,25]. PD-1 interrupts the effector T-cell responses by interacting with its ligands PD-L1 and PD-L2 [26]. PD-L2 has a much higher affinity for PD-1 than PD-L1 [27], and the PD-L1 blocking antibodies do not affect PD-L2/PD-1 interaction [28]. However, PD-L1 is considered the principal PD-1 ligand. LAG3-MHC class II/Lectins, TIGIT-CD155/CD112, TIM3-Galectin 9/PtdSer/HMGB1, and VISTA/VSIG 3 are other well-investigated receptor–ligand pairs. Overexpression of PD-L1 and CTLA-4 has been reported in HER-2+ and TNBC [13,29]. ICB by ICIs is an emerging therapeutic strategy that aims to restore antitumor immune response and eradicate cancer cells by inhibiting the negative regulators of effector T-cells. Several clinical trials have been developed or are ongoing with some preliminary promising results [30].
1.1.2. PD-L1 Expression
In addition to the well-known ER positivity and HER-2-expression/amplification, PD-L1 is another biological molecule that has recently emerged as a potential biomarker in breast cancer [31,32]. Commonly, PD-L1 expression is measured semi-quantitatively by immune histochemistry (IHC) [33] in tissue microarrays or a whole section. Table 1 shows the main parameters and criteria used for PD-L1 expression assessment. Usually, PD-L1 expression is absent in normal breast tissue, while in breast cancer, PD-L1 expression is measured in tumor cells (TCs) and/or immune cells (iCs). Thus far, in a few studies, PD-L1 expression in TCs and ICs has been evaluated with a discordance rate of up to 40–50%, according to the utilized IHC assay [34,35]. IHC PD-L1 expression in breast cancer is lower (10–30%) than in other tumors, such as non-small-cell lung cancer (about 70%) [36], and different based on the molecular subtype. Particularly, PD-L1 is higher in cases of TNBC, followed by the HER2+ subtype, and decreases further in HR+ advanced breast cancer. Moreover, receptor conversion during progression from primary to metastatic disease is well known even for PD1/PD-L1 expression [37,38,39]. This highlights the need to carry out metastatic biopsies as these suggest changing management roughly in 15–20% of patients [39,40,41]. In different metastatic sites, PD-L1 positivity at biopsy, similar to HR and HER-2 expression, varied from 12% to 60% [39]. PD-L1 over-expression occurs in 9–45% in HR+ early breast cancer patients, namely around 9% in luminal A and about 42% in luminal B, while in the metastatic stage, it decreases to 0–1% in luminal A and 10–12% in luminal B [42]. In nonmetastatic HER2+ breast cancer, the PD-L1 positivity rate is 30–35% and 35–60% in early TNBC compared with 9–15% in HER2+ advanced breast cancer and 30–40% in metastatic TNBC [43]. In a recent systematic review and meta-analysis on the PD-L1 status in breast cancer [44], the positivity rate of pooled PD-L1 was increased in primary tumors compared to metastasis when evaluated in IC (51.2% vs. 37.1%, p < 0.001) and TC/IC (30.1% vs. 14.6%, p < 0.001), unlike in TC (18.7% vs. 17.8%, p = 0.65). Moreover, the lowest PD-L1 positivity was observed in bone metastases (12%) and the highest in lymph nodes (60%). Thus, more commonly, the direction of change was from PD-L1-positive primary tumor to PD-L1-negative metastasis than vice versa, independent of the evaluated cell type. These discordances suggest both spatial and temporal heterogeneities during tumor progression, which implies temporal evolution of the immune surveillance at the TME. In particular, the transition from an initial inflamed although immunosuppressed TME to an immune-desert TME [45] is supported by the decrease in TILs at the metastatic sites, as well as a higher PD-L1 positivity concordance in synchronous samples compared to metachronous samples [46].
1.1.3. PD-L1 Assay
Currently, there are multiple PD-L1 IHC assays available with various scoring algorithms that have received approval for different therapies and tumor indications [47]. However, there is no standardized method for PD-L1 assessment. SP142, SP263, 28-82, 22C3, E1L3N, 73–10, E1J2J, 5H1, 4059, and 9A11 are some assays among others [43]; of these, the PD-L1 IHC 22C3pharmDx Assay (PD-L1 22C3, DAKO) and SP142 Assay (PD-L1 SP142, VENTANA) are used most often [47,48,49,50]. Commonly, the approval of immunotherapy drugs joins with a predetermined IHC assay. For example, PD-L1 IC score (PD-L1 SP142, VENTANA) was used for the approval of atezolizumab, whereas PD-L1 combined positive score (with 22C3 assay) is predictive for pembrolizumab. This procedure defines a companion diagnostic assay, which is mandatory to recruit a candidate for treatment with the approved drug. A complementary diagnostic test, unlike a companion diagnostic assay, is not limited to a specific drug and can be used more widely with a class of agents [51]. A tumor proportion score ≥ 1% or a combined positive score ≥ 1 are the criteria most commonly used for indicating PD-L1-positive expression [47] (Table 1). In the Impassion130 trial [52,53] conducted in TNBC patients, a post hoc analysis compared SP263, 22C3, and SP142 assays and found IC ≥ 1% for SP263 and SP142 and a combined positive score of ≥1 for 22C3-defined PD-L1 positivity. Again, there was no concordance of SP263 and 22C3 vs. SP142 (75% and 81% vs. 46%, respectively). Consistent with a mathematical model, a combined positive score ≥10 for 22C3 and an IC ≥ 4% for SP263 were the optimal cut-offs for PD-L1 positivity. However, based on these optimal cutoffs, a low concordance of SP263 and 22C3 versus SP142 was found [53]. Collectively, this suggests that PD-L1 IHC assays are not interchangeable in the current clinical practice.

Table 1.
Main parameters used for the assessment of PD-L1 expression.
Table 1.
Main parameters used for the assessment of PD-L1 expression.
| Parameter | Evaluation | Ref. | |
|---|---|---|---|
| Criterion | Analysis | ||
| Immune cells (iCs) | % | Percentage of tumor area involved by any intensity of PD-L1 IHC staining | [54] |
| Score | (Area of tumor infiltrated by PD-L1+ iCs/total tumor area) × 100% | [33,54,55] | |
| Tumor cells (TCs) | % | (Number of PD-L1+ iCs/total number of viable TCs) × 100% | [33,55,56] |
| Tumor cell score | The percentage of tumor area involved by PD-L1+ TCs related to the entire tumor area | [33,56] | |
| Tumor proportion score | (Number of viable PD-L1+ TCs/total number of viable TCs) × 100% | [33,55,57] | |
| iCs and TCs | Combined positive score | * (Number of PD-L1+ TCs plus iCs/total number of viable TCs) × 100% | [33,55,56] |
IHC: immunohistochemistry; * the number of PD-L1+ TCs and iCs is summarized with reference to all viable TCs and thereafter multiplied × 100, while 100 is the highest combined positive score.
1.1.4. Pembrolizumab and Nivolumab (PD-1 Inhibitors) (Table 2)
Pembrolizumab and nivolumab are IgG4 antibodies against PD-1 receptors. The safety and antitumor activity of pembrolizumab monotherapy have been assessed in the phase Ib KEYNOTE-028 trial enrolling heavily pre-treated ER+ HER2-advanced breast cancer patients with PD-L1-positive tumors (combined positive score ≥ 1) who received pembrolizumab monotherapy for up to 2 years or until confirmed progression/intolerable toxicity; the overall response rate (ORR) was 12%, 16% of patients had stable disease (SD), the clinical benefit rate was 20%, and the median duration of response was 12 months. The authors concluded that in the studied population, pembrolizumab was well tolerated with a modest but durable overall response [58]. In the phase II Keynote-086 trial, pembrolizumab monotherapy for pretreated or nonpretreated metastatic TNBC patients was well tolerated and showed prolonged anti-tumor activity [59,60]. These preliminary promising results were not confirmed by the phase III Keynote-119 trial comparing pembrolizumab monotherapy to standard CT in metastatic TNBC patients. In this trial, pembrolizumab monotherapy did not significantly prolong overall survival (OS) [61]. Therefore, pembrolizumab combined with CT versus placebo were tested in the phase III Keynote-355 [62] and Keynote-522 [63] trials. Following the results of these trials [64], the FDA recently approved pembrolizumab in combination with CT for patients with untreated recurrent inoperable or metastatic TNBC and as NACT for patients with untreated early-stage TNBC. In the phase 1b-2 PANACEA Keynote-014 trial, pembrolizumab in combination with trastuzumab was evaluated in trastuzumab-resistant, advanced, and HER2+ breast cancer patients. Among the 52 patients recruited in phase II, 40 had PD-L1+ and 12 had PD-L1− tumors. Six (15%) of 40 PD-L1+ patients achieved an objective response (OR), while there were no objective responders among the PD-L1− patients. The authors concluded that pembrolizumab plus trastuzumab was safe and showed activity and durable clinical benefit in patients with PD-L1+, trastuzumab-resistant, advanced, and HER2+ breast cancer [65]. Some other investigational studies are being conducted with pembrolizumab in different settings and in combination with different drugs (NCT02768701, NCT03121352, NCT03025035, NCT03032107, NCT04251169, NCT03393845, and NCT02752685). In the TONIC trial conducted in metastatic TNBC, nivolumab alone with or without induction therapy showed a 20% ORR [66]. In a single-arm phase II study, the efficacy and safety of cabozantinib combined with nivolumab in metastatic TNBC patients were evaluated. Among the 17 evaluable patients, one patient confirmed partial response (PR), 14 SD, and 2 progressive disease (PD). As the study did not meet the pre-specified criteria, it was closed early. Toxicity led to cabozantinib dose reduction in 50% of patients. The responding patient had a PD-L1-negative tumor with low tumor mutational burden but high TILs and enriched immune gene expression; further, immunostaining, genomic, and proteomic studies indicated a high degree of tumor immune suppression in the studied population [12]. In a prospective, single-arm, open-label, phase 1b trial (NCT03807765), nivolumab and stereotactic radio-surgery were assessed among metastatic breast cancer patients with brain metastases. The initial dose of nivolumab was followed, 1 week later, by stereotactic radio-surgery. A total of 12 patients were treated up to 17 lesions. No dose-limiting toxicities were observed and the most common neurological adverse events (aEs) included grade 1 to 2 headaches and dizziness occurring in 5 (42%) patients. The median intracranial control was 6.2 months with 6- and 12-month control rates of 55% and 22%, respectively [67].
1.1.5. Atezolizumab, Durvalumab, and Avelumab (PD-L1 Inhibitors)
Atezolizumab, durvalumab, and avelumab are IgG1 monoclonal antibodies (mAb) and target PD-L1. Avelumab additionally has the capability to induce antibody-dependent cellular cytotoxicity, which is an indirect antitumor effect obtained by engaging mAb with Fc receptors on immune effector cells [68]. Good tolerability and efficacy of atezolizumab were found in a phase I study conducted in metastatic TNBC patients [69]. The Impassion 130 trial was conducted using atezolizumab plus nab-paclitaxel (A+nP) versus placebo plus nab-paclitaxel (P+nP) in patients with locally advanced or metastatic TNBC [70,71,72]. Immune-mediated aEs of special interest were observed in 58% and 41.6% of the patients treated with A+nP and P+nP, respectively [73]. Following the promising results of both trials, the Ventana PD-L1/SP142 IHC assay received the FDA’s approval as an atezolizumab companion diagnostic assay [29]. In the phase III Impassion 031 trial carried out in early-stage TNBC patients, atezolizumab combined with nab-paclitaxel and anthracycline-based CT compared to placebo showed a significantly higher rate of pathological complete response (pCR) with acceptable tolerability [74]. Unlike this, the Impassion 131 phase III trial of first-line paclitaxel with or without atezolizumab for unresectable locally advanced or metastatic TNBC showed that atezolizumab combined with paclitaxel did not increase progression-free survival (PFS) or OS compared to paclitaxel alone [75]. In a phase I study carried out in recurrent cancer patients including TNBC, durvalumab showed acceptable safety and preliminary antitumor activity [76]. In the phase II GeparNuevo study that recruited early TNBC patients, durvalumab was administered as NACT combined with an anthracycline-taxane-based treatment. An increased pCR occurred mainly in patients who had received durvalumab monotherapy before CT [77]. In the phase II SAFIR02-Breast Immuno trial, durvalumab monotherapy improved the outcomes of TNBC patients, compared to maintenance CT [78]. Further, avelumab was tested in a phase I study enrolling locally advanced or metastatic breast cancer patients, including those with TNBC. In this study, an acceptable toxicity and clinical anti-tumoral activity was reported mainly in tumors with PD-L1 over-expression [79].
1.1.6. Efficacy and Safety of Anti-PD1/PD-L1 Monotherapy in Metastatic Breast Cancer
Recently, interim analyses to evaluate the efficacy and safety of anti-PD-1/PD-L1 monotherapy for metastatic breast cancer have been carried out; 586 advanced breast cancer patients treated with anti-PD-1/PD-L1 monotherapy agents were included from six studies. The anti-PD-1/PD-L1 agents used for the treatment were pembrolizumab, atezolizumab, and avelumab. Overall, regarding monotherapy, the complete response (CR) rate was 1.26%, the partial response (PR) rate was 7.65%, the ORR was 9.85%, and the disease control rate was 18.33%. The one-year OS rate and 6-month PFS rate were 43.34% and 17.24%, respectively. The incidence of aEs was 64.18% in any grade and 12.94% in severe grade, while the incidence of immune-related (ir)aEs was approximately 14.75%. Moreover, comparing PD-L1-positive and PD-L1-negative groups, the correspondence between efficacy and expression of the PD-L1 biomarker was found as follows: PR was 9.93% vs. 2.69%; ORR was 10.62% vs. 3.07%; disease control rate was 17.95% vs. 4.71%, respectively [80].
1.1.7. Tremelimumab and Ipilimumab (CTLA-4 Inhibitors)
Tremelimumab and ipilimumab are IgG2 and IgG1 mAb, respectively, and are used against CTLA-4. In a phase I dose escalation study, the safety of tremelimumab, administered on the third day of palliative RT, was evaluated in patients with metastatic breast cancer. Among the six enrolled patients, five had HR+ metastatic breast cancer and one had metastatic TNBC. One dose-limiting toxicity occurred at 6 mg/kg; however, the trial closed before MTD could be determined. One patient discontinued treatment due to a pathological fracture. SD was the best response, and one patient had SD for >6 months. The median OS was 50.8 months, with one patient surviving >8 years. Peripheral blood mononuclear cell profiles showed increasing proliferating (Ki67+) Treg cells 1 week post-treatment in five patients. The authors concluded that this combination approach needed to be optimized [81]. Another investigational study with tremelimumab in addition to durvalumab in HR+/HER2− stage I to III breast cancer patients without any previous therapy is ongoing (pilot study NCT03132467). In the ICON, a phase Ib study, immunogenic CT will be evaluated with ipilimumab and nivolumab in metastatic HR+ breast cancer patients. In particular, anthracycline, which is considered “immunogenic”, and low-dose cyclophosphamide, which has been reported to counter immunosuppressive cells, are the combined CT. The trial will enroll 75 evaluable subjects randomized into two arms (A:B). Patients in Arm A will receive only CT, i.e., pegylated liposomal doxorubicin and low-dose cyclophosphamide. Patients in Arm B will receive pegylated liposomal doxorubicin + cyclophosphamide + ipilimumab + nivolumab. Patients in Arm A will be offered ipilimumab + nivolumab after PD. The rationale consists in expecting that CT synergize with ICIs and in combining PD-1 and CTLA-4 blockade, as these checkpoints are important in different phases of the immune response [82]. Table 2 shows the main clinical trials carried out with ICIs in breast cancer and their principal results.

Table 2.
Main clinical trials conducted with immune checkpoint inhibitors (ICIs) in breast cancer. (A) Clinical trials with pembrolizumab (PE) or nivolumab (NI). (B) Clinical trials with atezolizumab (ATZ), durvalumab (DRV), avelumab (AV), tremelimumab (TREMB) or ipilimumab (IPI).
Table 2.
Main clinical trials conducted with immune checkpoint inhibitors (ICIs) in breast cancer. (A) Clinical trials with pembrolizumab (PE) or nivolumab (NI). (B) Clinical trials with atezolizumab (ATZ), durvalumab (DRV), avelumab (AV), tremelimumab (TREMB) or ipilimumab (IPI).
| A | ||||||||
|---|---|---|---|---|---|---|---|---|
| Trial | Phase | Drug | Mechanism of Action | Setting | Therapeutic Regimen | Pts (n) | Outcome | Ref. |
| Keynote-028 NCT 02054806 | Ib | Pembrolizumab (PE) | PD1 inhibitor | ABC PD-L1+ | Monotherapy for up to 2 years | 25 | ORR 20% SD 16% CB 20% MDR 12 mo. | [58] |
| Keynote-086 NCT 02447003 | II | mTNBC | Monotherapy, >1 prior systemic therapy | 170 | ORR 5.7% in PDL1+ pts PFS 2 mo. OS 9 mo. | [59] | ||
| Cohort B | Monotherapy, no prior systemic therapy | 84 | ORR 20% PFS 21 mo. OS 18 mo. | [60] | ||||
| Keynote-119 NCT 02555657 | III | mTNBC | PE vs. CT, prior systemic therapy | 312 vs. 310 | OS 12.7 vs. 11.6 mo. in PDL1+ pts | [61] | ||
| Keynote-355 NCT 02819518 | III | mTNBC | PE + CT vs. PB + CT, no prior CT | 566 vs. 281 | PFS 9.7 vs. 5.6 mo. in PDL1+ pts; PFS 7.6 vs. 5.6 mo. | [62] | ||
| Keynote-522 NCT 03036488 | III | Early, stage II-III TNBC | PE + PTX + CBD vs. PB + PTX + CBD | 401 vs. 201 | pCR 64.8% vs. 51.2% | [63] | ||
| Panacea-Keynote-014 | Ib-II | HER2+, Trastuzumab resistant ABC | PE + trastuzumab | 52 | ORR 15% in PDL1+ pts No response in PDL1− pts | [65] | ||
| TONIC | II | Nivolumab (NI) | mTNBC | (1) NI without induction; (2) NI with induction *; (3) RT; (4) CY; (5) CIS; (6) DOXO 3-4-5-6: followed by NI | 67 | ORR 20% for all pts CIS 23% DOXO 35% | [66] | |
| Institutional (open-label, single arm, single-center, closed early) | II | mTNBC | NI + cabozantinib | 18 | ORR 6% (1 PR in a PDL1− pt) | [12] | ||
| NCT03807765 | IB | BC with brain metastases | NI followed by SRS | 12 | 6 mo. control rate 55% 12 mo. control rate 22% | [67] | ||
| B | ||||||||
| Trial | Phase | Drug | Mechanism of Action | Setting | Therapeutic Regimen | Pts (n) | Outcome | Ref. |
| NCT01375842 | I | Atezolizumab (ATZ) | PD-L1 inhibitor | mTNBC unselected for PDL1 | Monotherapy | 116 | ORR 10–12% in PDL1+ Median PFS 1.4 mo. Median OS 8.9 mo. | [69] |
| iMpassion 130 NCT02425891 | III | mTNBC unselected for PDL1 | ATZ + Nab-PTX vs. PB + Nab-PTX | 451 vs. 451 | ORR 56–58.9% in PDL1+ Median PFS 7.5 mo. in PDL1+ Median OS 25 mo in PDL1+ | [70,73] | ||
| iMpassion 031 NCT03197935 | III | Stage II or III TNBC, no prior systemic therapy | ATZ + CT vs. PB + CT | 165 vs. 168 | pCR 58% vs. 41% p = 0.0044 | [74] | ||
| iMpassion 131 NCT03125902 | III | Locally advanced or mTNBC, no prior systemic therapy | ATZ + PTX vs. PB + PTX | 431 vs. 220 | Median PFS 6 vs. 5.7 mo. Median OS 22.1 vs. 28.3 mo in PDL1+ pts | [75] | ||
| NCT02484404 | I | Durvalumab (DRV) | Recurrent cancers including TNBC | DRV + cediranib + olaparib | 9 (1 TNBC) | CBR 67% PR 44% | [76] | |
| GeparNuevo NCT02685059 | II | NACT in early TNBC | DRV + Nab-PTX vs. PB + Nab-PTX followed by EC ** | 174 (117 the window cohort) | pCR 53.4% vs. 44.2% pCR 61% vs. 41.4% in window cohort | [77] | ||
| SAFIR 02 Breast Immuno NCT02299999 | II | HER2− mBC with prior CT | DRV vs. maintenance CT | 32 vs. 29 assessed | HR of death: 0.37 for PDL1+ pts vs. 0.49 PDL1− pts | [78] | ||
| Javelin NCT01772004 | Ib | Avelumab (AV) | Locally advanced or mTNBC, refractory or progressing | Monotherapy | 58 TNBC | ORR 22.2% in PDL1+ pts vs. 2.6% in PDL1− pts | [79] | |
| Institutional (open-label, single-arm, single-center, closed early) | I | Tremelimumab (TREMB) | CTLA-4 inhibitor | Incurable mBC | TREMB + RT | 6 (1 TNBC) | Median OS 50.8 mo. | [81] |
| ICON | Ib | Ipilimumab (IPI) | HR+ mBC | IPI + NI + PLD + CY vs. PLD + CY | 75 | Expected | [82] | |
Pts: patients; ABC: advanced breast cancer; mBC: metastatic breast cancer; ORR: overall response rate; SD: stable disease; CB: clinical benefit; CBR: clinical benefit rate; MDR: median duration of response; PFS: progression-free survival; OS: overall survival; mTNBC: metastatic TNBC; CT: chemotherapy; PB: placebo; PTX: paclitaxel; CBD: carboplatin; pCR: pathological complete response; *: 2 weeks low dose; RT: radiotherapy; CY: cyclophosphamide; CIS: cisplatin; DOXO: doxorubicin; PR: partial response; SRS: stereotactic radiosurgery; **: in the window-phase, DRV/PB alone was given 2 weeks before the start of Nab-PTX; PLD: pegylated liposomal doxorubicin.
1.1.8. PD-L1 Testing as Prognostic and/or Predictive Biomarker in Breast Cancer
The clinical usefulness of the PD-L1 assay largely varies between cancer types and different settings of treatment [55]. The Impassion130 trial, an exploratory analysis carried out in PD-L1-iCs-positive patients, showed that the median OS was 25.4 months versus 17.9 months in patients treated with atezolizumab plus nab-paclitaxel (A + nP) vs. nab-paclitaxel plus placebo (nP + P), respectively. In the PD-L1-iCs-negative population, the median OS was 19.7 months in each treatment arm [73]. In a sub-study from the same trial [46], the PD-L1 assay, together with other immune markers, was evaluated for association with clinical benefit. PD-L1 IC > 1% of the tumor area in both primary and metastatic tumors predicted a favorable outcome and joined with PFS benefit with A + nP versus P + nP. An investigational study [83] was carried out to better understand the outcome discrepancy between the Impassion 130 and Impassion 131 clinical trials. In this study, the changes in iCs over time in 22 patients with metastatic TNBC receiving paclitaxel or paclitaxel plus atezolizumab were evaluated. It was found that high levels of baseline CD8-CXCL13 and CD4-CXCL13 could predict an efficacious response and that following treatment with paclitaxel plus atezolizumab versus paclitaxel alone, both CD8-CXCL13 and CD4-CXCL13 expanded in responsive patients after treatment. The authors concluded that the reduction in both these populations by paclitaxel may compromise the clinical outcome of the accompanying atezolizumab. In a study that examined six OS curves from three randomized clinical trials (Keynote-355, Impassion130, and iMpassion131 studies) conducted in metastatic TNBC patients, the efficacy of first-line ICIs combined with CT was analyzed. PD-L1 iCs > 1% positivity for atezolizumab and a PD-L1 combined positive score > 10% for pembrolizumab showed prognostic value. In particular, in patients with PD-L1 iCs > 1% positivity, atezolizumab combined with a significantly better OS than pembrolizumab. As for pembrolizumab, a threshold of PD-L1 combined positive score > 10% increased the median OS from 15.5 to 23 months [84]. In the study by Dirik et al. [79], where 168 heavily pretreated patients with locally advanced or MBC, including 58 patients with TNBC, received avelumab, a trend toward a higher ORR was seen in patients with PD-L1+ versus PD-L1− tumor-associated iCs in the overall population (16.7% vs. 1.6%, respectively), as well as in the TNBC subgroup (22.2% vs. 2.6%, respectively). In the meta-analysis by Qi et al. [80] considering six studies of advanced breast cancer including TNBC and HR+ HER2− patients who had received anti-PD-1/PD-L1 monotherapy, the response rate as reported was closely associated with the expression of the PD-L1 biomarker (PD-L1+ vs. PD-L1−). The PD-L1-positive expression status was defined as PD-L1 expressed in ≥1% TCs or/and tumor-associated iCs. Unlike these favorable results, in the Keynote 086 and Keynote 150 studies recruiting metastatic TNBC patients, pembrolizumab given alone or with eribulin, respectively, did not show significant clinical differences, according to PD-L1 status. Moreover, in the Keynote 086 study, 60% of patients suffered from treatment-related aEs including those that were immune-mediated [11]. Lastly, in the study where nivolumab was given along with cabozantinib for metastatic TNBC [12], a PD-L1-positive tumor occurred in one among 15 patients who were successfully tested; this patient had SD as the best response.
1.2. ICIs Combined with DDRi
ORRs for ICB monotherapy in breast cancer range from 5% to 23% [85]. To increase its efficacy, a combinatorial therapy of ICIs with DDRi is particularly promising.
1.2.1. DNA Damage Repair Defects, Tumor Mutational Burden, and Neo-Antigens
The principal task of DNA damage repair (DDR) proteins following DNA damage due to endogenous factors or exogenous insults is to protect the integrity of the genome [86]. Patients with tumors carrying a DNA mismatch repair deficiency (MMRd), HRD, BRCA1/2 genetic defects, or other defects in DDR genes [87] have a higher tumor mutation burden and more neo-antigens. The deficiency of DDR genes is reported in a wide range of malignancies including TNBC [88]. Genomic loss of heterozygosity, large-scale translocations, and telomeric allelic imbalance together with BRCA mutations [89] have been proposed as molecular hallmarks of HRD [90]. HRD occurs in more than 20% of breast cancers and 69% of TNBC [91]. Breast cancers with gBRCA1/2 mutations only account for 5–7% of breast cancer cases [92] mainly in TNBC [93], while a high mutation rate is observed in high-grade TNBC [94]. Overall, tumor mutational burden is a surrogate marker for tumor neo-antigen load and patients with higher tumor mutational burden can likely benefit from DDRi [88], which, through the increase in tumor mutational burden and neo-antigens, can enhance antitumor immunity.
1.2.2. Barriers to ICIs Response and Mechanisms by Which DDRi Synergizes with ICIs
It is commonly thought that the limited ICB efficacy is due to the presence of multi-factorial barriers in the TME, some of which involve DDR signaling pathways that could be reversed by DDRi. The mutation of genes involved in DNA replication pathways is reported to be enriched in patients showing durable clinical benefit from ICB. Considering this, the use of poly adenosine diphosphate-ribose polymerase inhibitors (PARPis) or other DDRi-targeting DDR pathways can potentially improve ICB for cancer therapy. Low tumor mutational burden, particularly low true neo-antigen burden, which is the number of alterations actually addressed by effector T-cells, is one barrier that has a strong impact on ICB response. Tumors with high microsatellite instability acquire many somatic mutations due to the failure of DNA MMRd. The following high tumor mutational burden makes tumors more immunogenic and sensitive to PD-1 ICB. Mutant neo-antigens in MMRd cancers make them sensitive to ICB therapy in many different tumor types [95,96], suggesting a pan-tumor biomarker function for ICB efficiency. However, MMRd was only observed in around 1% of breast cancers, with 1.8% in TNBC [97]. HRD has less impact than MMRd on tumor mutational burden; however, the genomic instability in HRD tumors can favor DDRi responsiveness when given together with ICB [98]. A low PD-L1 expression can be another barrier to ICB response. DNA damage enhanced by DDRi can increase PD-L1 expression by activating both IRF1 and interferons [99]. The adaptive PD-L1 up-regulation, which counteracts the PARPi-mediated immune activation, can likely be overcome by the combinatorial treatment of ICB with DDRi [98]. ICB can also be ineffective due to a dysfunctional T-cell phenotype, as it occurs with T-cell apoptosis, which is a mechanism of tumor-induced T-cell dysfunction triggered by the binding of different molecules on T-cells with the corresponding ligands [100]. Impaired DNA machinery favors T-cells apoptosis by the inhibition of telomeric repeat binding factor 2, telomerase, topoisomerase I and II alpha, and ATM kinase actions [101], while amplifying DNA damage by DDRi can reprogram TME. This reprogramming induces the recruitment of T-cells into the tumor bed [102], thus transforming “cold tumors” into “hot tumors” through the activation of the immune response. Down-regulated tumor MHC-I/II expression has been reported to contribute to ICB resistance [103]. On the other side, DNA damage favored by DDRi can increase MHC-I molecules expression on the surface of tumor cells [104], thus supporting ICB reversal. Experimental findings show that drugs targeting proteins involved in the DDR pathway, apart from PARPi, significantly synergize with anti-PD-L1 therapy, suggesting that DDRi can successfully impact immunosuppressive cells [98]. Altered genes associated with different pathways including MAPK, JAK-STAT, PI3K-AKT, WNT-beta catenin, and Hippo have been reported to affect the ICB response. Moreover, two studies found that STK11/LKB1 mutations combined with resistance to ICB. The proposed rationale was that LKB1 loss silences the stimulator of interferon genes and finally promotes the expression of type 1 interferons and other cytokines, favoring an immunosuppressive TME, which contributes to ICB resistance [98]. DDRi increasing DNA damage can reset the inflammatory microenvironment of tumors to a higher level, thereby enhancing cytokine gene expression in vivo or in vitro (CCL2, CCL3, CCL5, and CXCL10). The inflammatory cytokine increase impacts the TME by recruiting immune cells and contributes by making it more immunogenic. In summary, regarding the immunologic landscape, alterations of DNA replication amplified by DDRi favor immunological vulnerabilities in tumors by attracting effector immune cells while contemporaneously inducing immunosuppressive pathways, thus providing the rationale for the combinatorial therapy of DDRi with ICB [98].
1.2.3. Clinical Trials with ICIs and DDRi Combinatorial Therapy
In a first reported phase I clinical trial [105], 83% and 75% disease control rates were observed with the administration of durvalumab plus olaparib and durvalumab plus cediranib, respectively. However, the response to therapy was independent of PD-L1 expression, and grade 3–4 aEs occurred in a few patients. The phase Ib/II Topacio/Keynote-162 trial conducted with PARPi niraparib combined with pembrolizumab recruited 55 previously treated advanced or metastatic TNBC patients [106]. In 15 evaluable patients with tumor BRCA mutations, the ORR, disease control rate, and median PFS were 47%, 80%, and 8.3 months, respectively, while in another 27 evaluable patients with BRCA wild-type tumors, these values were 11%, 33%, and 2.1 months, respectively. The most common treatment-related AE of grade 3 or higher ranged from 7% (fatigue) to 18% (anemia); iraEs of grade 3 were observed in two patients (4%). The authors concluded that niraparib plus pembrolizumab results in a promising antitumor activity with higher response rates in patients with tumor BRCA mutations. The phase I/II MEDIOLA study that administered olaparib with durvalumab recruited four cohorts with different cancers. The breast cancer cohort included 34 patients with gBRCA1/2 or both mutations, progressive, HER2-negative, and metastatic breast cancer [107]. Patients had not received more than two previous lines of CT for metastatic breast cancer. Eleven (32%) patients experienced grade 3 or worse aEs and three (9%) patients discontinued due to aEs. A similar ORR was observed in 17 TNBC and 13 HR+ subjects (59% and 69%, respectively). The median PFS was longer in the 13 patients with HR+ disease (9.9 months and 4.9 months, respectively). Again, OS was similar in both subgroups (22.4 months and 20.5 months, respectively). Although a definitive answer to whether ICIs combined with DDRi improve the outcomes is not yet available, this combinatorial therapy is under investigation in the KEYLYNK-009 trial (NCT04191135) [108] also as maintenance treatment following induction CT independent of the mutational status.
1.3. Monoclonal Antibodies against Human Epidermal Growth Factor Receptor 2 (HER-2) Family in HER2+ Breast Cancer
HER2 overexpression identifies a breast cancer molecular subtype with worse outcome [8,109]. mAb targeting specific HER2 epitopes was the first anti-HER2 immunotherapy. They, upon binding, neutralize the target molecule expressed by cancer cells, thereby inhibiting their proliferation and survival [110]. Trastuzumab, a humanized IgG1 mAb, was the first HER2-specific mAb approved for HER2+ breast cancer in 1998 [111]. Trastuzumab, by blocking HER2 signaling, promotes G1 cell-cycle arrest and inhibits the PI3K pathway, favoring apoptosis and angiogenesis inhibition. First, trastuzumab, combined with first-line CT, obtained approval for metastatic HER2+ breast cancer [112]; then, after showing efficacy and safety in early breast cancer, it also became a conventional neo-adjuvant or adjuvant treatment in association with different anti-HER2 therapies and/or CT [9,30,113]. The rationale for combining anti-HER2 therapies with CT is their synergistic effect, which could likely be due to the depletion of DNA repair activity by the binding of antibodies to the epidermal growth factor receptor extracellular epitopes or to HER2 itself [114,115]. Following trastuzumab, specific antibodies targeting different HER2 epitopes and capable of inhibiting other cellular signaling pathways have been built. These mAbs could also elicit an indirect antitumor response on effector iCs by engaging with Fc receptors [68]. In fact, they can activate a complete (innate and adaptive) immune response inducing antibody-dependent cytotoxic cellular killing of HER2-overexpressing cells by NK cells; moreover, through the presentation of HER2 by MHC-I molecules, they can activate cytotoxic T lymphocytes and decrease intra-tumoral Tregs [116]. Pertuzumab is another IG1 mAb targeting the domain II of the HER2 receptor, thus preventing HER2 hetero-dimerization with HER3. It inhibits intracellular signaling via the PI3K/AKT pathway and can elicit antibody-dependent cellular cytotoxicity [113,117]. Pertuzumab has received approval for the treatment of metastatic and early-stages HER2+ breast cancer patients. Pertuzumab in combination with trastuzumab and CT improved OS in metastatic HER2+ breast cancer patients versus trastuzumab and CT alone [118,119,120,121]. Similarly, pertuzumab showed clinical benefit in the neo-adjuvant and adjuvant setting again in combination with trastuzumab and CT [122,123,124,125]. Overall, the use of trastuzumab and/or pertuzumab significantly increased the ORR and increased for a few months the PFS and OS [126]. However, due to intrinsic or acquired resistance, only roughly one-third of HER2+ tumors benefit from anti-HER2 antibodies [127]. To overcome the resistance to anti-HER2 antibodies, in addition to their combination with conventional CT, further strategies such as improving their functionality to enhance antibody-dependent cellular cytotoxicity have been applied. This has been attained through modification in the Fc region similar to margetuximab, a chimeric IgG1 anti-HER2 mAb [128], or with an antibody–drug conjugate (ADC) that covalently conjugates the tumor-specific mAb with a cytotoxin acting within the microtubules, thereby increasing antitumor immunity. This category includes trastuzumab-emtansine (T-DM1) [129,130], trastuzumab deruxtecan [131] and, more recently, sacituzumab-govitecan [132]. On the other hand, the higher PD-L1 expression in HER2+ breast cancer patients suggests a combination of anti-HER2 mAbs and ADCs with ICIs.
Anti-HER2 mAbs and/or ADC Combined with ICIs
Some trials have assessed ICIs in combination with trastuzumab or ADCs. The phase Ib/II PANACEA and phase Ib CCTG IND 229 trials take part in the former type of studies. In the PANACEA trial [65], PD-L1 positivity was defined as having a combined positivity score of >1, initially evaluated by QualTek and then by the Dako22C3 assay. The 58 women, who were recruited after they had progressed on the previously mentioned trastuzumab therapy, received pembrolizumab plus trastuzumab. Of these, 46 patients were PD-L1+ and had a 15% ORR and a disease control rate of 24% with no response or disease control observed in those who were PD-L1−. In the other trial, in which recruited patients had previously been treated with trastuzumab, pertuzumab, and a taxane, and were then given durvalumab and trastuzumab, no response was recorded [133]. In the phase II Kate2 trial [134], patients who were previously treated with trastuzumab and a taxane were randomized to receive atezolizumab or placebo associated with T-DM1. A trend toward a prolonged median PFS only was found in PD-L1 IC+ patients, with a PFS of 8.5 months in patients who were given the combination vs. a PFS of 6.8 months in those who received T-DM1 alone. Heavy aEs occurred more often in patients who were given the combination therapy including pyrexia and grade 3 transaminitis in 35% and 8% of patients, respectively, and one death. A similar cohort was recruited in a phase Ib trial where the 20 evaluated patients received pembrolizumab plus T-DM1 [135]. For this study, the ORR was 20% and the median PFS was 9.6 months; a trend toward higher response occurred in patients with a PD-L1 combined positivity score of <1 and TILs ≤ 10%; furthermore, 10% of the patients complained of grade 3 transaminitis. Other trials investigating NACT combined with ICIs and anti-HER2 therapies are ongoing. In the Impassion 050 trial, patients were randomized to receive atezolizumab or placebo concomitant with NACT, including doxorubiicin + cyclophosphamide, followed by paclitaxel, trastuzumab, and pertuzumab [133,136]. Atezolizumab or placebo was continued in an adjuvant setting together with trastuzumab and pertuzumab for 52 weeks overall. However, the study was stopped due to an unfavorable risk–benefit ratio.
1.4. Other Investigational Immune-therapies
Adoptive cell therapy (ACT) is an immunotherapy strategy that was started in 1987 and has since then evolved in different forms. First, it was based on the autologous or allogenic transfer of TILs following isolation from TME, ex vivo activation, and expansion through the use of interleukin-2, and infusion back to the patient. However, multiple obstacles, the most likely among them being an immunosuppressive TME, impeded this technique to exit from the investigational ground and spread out [137,138]. More recently, a similar approach using NK cells, unlike TILs, has been applied to a small sample size of breast cancer patients. This trial was based on the rationale that NK cells benefit from the ability to kill cancer cells in an MHC-independent and non-tumor antigen-restricted way. However, the outcome was unsatisfactory, possibly because of the same drawbacks that occurred with TILs [139]. Therefore, two different modalities were developed, the use of T cells that have been genetically engineered to express modified T-cell receptors or chimeric antigen receptors (CAR). In the former technique, the alpha-beta TCRs are armed with synthetic T cell receptors for targeting cancer-specific epitopes presented by MHC molecules. This procedure enhances T cell affinity toward the cancer cells presenting the targeted epitope, although the low number of potential targets is the main limitation. Clinical trials with intravenous infusion of TCR-modified T cells against HER2, MAGE-A3, or other antigens are ongoing [138]. Recently, gamma delta T cells have been considered for ACT due to their ability to recognize and kill cancer cells in a human leucocyte antigen (HLA)-independent manner, together with the ability to induce antibody-dependent cellular cytotoxicity. In addition, these T cells, such as NK cells, express activating receptors that bind their ligands present on the cancer cells. Two trials in this regard have been carried out with promising findings [140,141]. Further, CAR is a synthetic molecule that consists of an extracellular domain (scFv) combined with the intracellular signaling domain of a physiological T cell receptor (CD3 zeta chain), as well as different co-stimulatory domains (CD28, ICOS, and OX40). T or NK cells engineered to express CAR can identify multiple tumor cell surface antigens by their extracellular domain in a non-MHC-restricted way that can allow overcoming immune evasion due to MHC down-regulation. A few trials evaluating CAR-T therapy in breast cancer are ongoing [138,142]. In 1992, we recruited metastatic ER+ breast cancer patients with clinical benefit during the first-line anti-estrogens treatment. These patients additionally received an active immune stimulation using the interferon-beta interleukin-2 sequence. The promising results of the pilot study with this active immunotherapy were first reported in 2005 due to the low accrual rate in our oncological center [143,144]. Subsequently, the unexpected difficulties we encountered in launching a sponsored prospective confirmatory randomized clinical trial convinced us to resort to a more feasible 2:1 control-case retrospective observational study that included 95 controls treated with hormone therapy alone and 42 cases treated with hormone therapy plus the interferon-beta-interleukin-2 sequence. Twenty-eight controls (28.9%) unlike cases had received biological drugs including cyclin kinase inhibitors (CKIs) and most cases were given first-line SERMs or SERDs unlike controls who were treated with first-line AIs. Despite this, the median PFS and OS were significantly longer in the 42 cases who had received additional immune-stimulating immunotherapy than in the 95 controls (Table 3). No grade 3–4 AEs occurred in the 42 cases; moreover, this proposed immunotherapy is 8–18 times cheaper than CKIs [145]. Table 3 shows some major recent clinical trials with anti-HER2 mABs, ADC, anti-HER2 mAb, or ADC plus ICIs or other investigational immunotherapies.

Table 3.
Major clinical trials recently conducted with anti-HER2 monoclonal antibodies (mAbs), antibody–drug conjugates (ADCs), their combination ICIs, and other investigational immunotherapies.
1.5. The Tumor Immunogenicity: A True or an Incomplete Understanding?
In the scientific literature, the host immune system’s capability to provide a spontaneous immune response to cancer cells is a well proven and consolidated concept [146]. The intensity of this response defines tumor immunogenicity. More recent investigations support the notion that the TILs rate in the TME, tumor mutation burden, and neo-antigens, as well as other genetic alterations such as MMRd, microsatellite instability, HRD and defects in DDR genes, are tools for tumor immunogenicity assessment [147,148] and potential surrogate biomarkers that are predictive of a better immune response. Based on this, breast cancer is considered a low or moderately immunogenic tumor, unlike other types of cancers such as melanoma and lung cancer, which are reported as highly immunogenic [14,71].
1.5.1. The Immunological Balance Resulting from the Interplay between Cancer Cells and TME: Mechanisms Inhibiting or Promoting an Antitumor Immune Response
The TME contains many different cells and immune-modulatory humoral factors that, overall, contribute to the immune balance. Cellular components include stromal cells, together with cells of the innate and adaptive immune system. Cytokines, chemokines, and growth factors that are secreted by cancer, stromal, and immune cells are the immune-modulatory humoral factors, which, together with tumor-cell-derived exosomes, play a central role in the cell-to-cell communication and the crosstalk between cancer cells and their TME. The commonly prevailing immunosuppressive TME results from the balance of opposite actions that, directly or indirectly, induce or inhibit the cell-mediated immune response and tumor growth. Due to the widely documented spatial intra-tumor and inter-tumor genetic heterogeneities [149], the immunological balance differs in different tumors of the same type and different organs of the same tumor. Additionally, temporal evolution occurs after therapeutic interventions, together with genetic and metabolic alterations. This makes immunological balance a dynamic condition that changes over time [150,151].
1.5.2. Anti-Tumoral Immune Activities
TILs that can be present in breast cancer TME are made of cytotoxic (CD8+) cells, helper (CD4+) T cells, Tregs, and NK cells [152]. A higher TILs rate in TME has been reported to be associated with improved clinical outcomes in both HER2+ and TNBC [153]. Additionally, in HER2+ breast cancer, a higher TILs rate in TME directly correlates with pCR to NACT. Therefore, overall, despite the presence of immunosuppressive Tregs, TILs are commonly thought to reflect a state of immune activation. Regarding B cells, findings suggest that their principal role is to support an immunological response by producing antibodies and inducing an optimal T cell activation and cellular immunity [154]. NK cells, also defined as CD3− CD56+ cells, comprehend two subsets, CD56bright CD16low/− and CD56bright CD16+. The former can recognize cancer cells in a nonrestricted MHC class I modality and kill them by releasing cytolytic granules containing perforins and granzymes [17]. Activated dendritic cells (DCs) play a key role in the immunological response through the presentation or cross-presentation of tumor antigens to the CD4+ and CD8+ T cells, which is essential for the maturation and activation of tumor-specific cytotoxic T lymphocytes that move to the tumor’s niche to eliminate tumor cells. Among tumor-associated macrophages (TAMs), the M1-like phenotype typically performs anti-tumor functions and is capable of directly killing tumor cells through the release of reactive oxygen species and nitric oxide, and antibody-dependent cellular cytotoxicity [155]. Among tumor-associated neutrophils (TANs), the N1 phenotype induces inflammation processes through the release of reactive oxygen species as well as interleukin (IL)-1beta, TNF-alpha, IL-6, and IL-12 cytokines [156].
1.5.3. Immune Activities Promoting Immune Inhibition and Tumor Progression
In the TME, Tregs, cancer-associated fibroblasts (CAFs), TAMs, myeloid-derived suppressor cells (MDSCs), and TANs are the principal cells that, together with many different humoral factors such as cytokines (IL-4, IL6, IL-10, IL-13, TGF-beta, and TNF alpha), chemokines, their ligands (CXCL12 and CXCR4), enzymes (such as indoleamine 2,3 dioxygenase (IDO) and ARG1), exosomes, and other factors (VEGF and prostaglandin E2), inhibit the immune response. The humoral factors are largely secreted by tumor cells and the other cells within the TME [149,157]. In TME, VEGF induces the proliferation of immunosuppressive cells, decreases T cell recruitment, and increases T cell exhaustion, in addition to stimulating tumor vessel growth [158].
Tregs, TAMs, and MDSCs
Tregs move to the TME recalled by the cytokines and chemokines secreted by tumor and immunosuppressive cells. In metastatic breast cancer, they can be derived by converting resting CD4+ T cells after induction by IL-10 and TGF-beta [159]. Tregs strongly promote immune inhibition and tumor progression by impairing the cytotoxicity of effector T cells, modulating antigen-presenting cells and inducing metabolism alterations [152]. In breast cancer, an increased ratio of total FoxP3+ Tregs to CD8+ cytotoxic T lymphocytes is an unfavorable prognostic index unlike a high CD8+ rate [160]. CAFs can secrete mitogenic growth factors, pro-angiogenic factors, and TGF-beta, overall favoring tumor progression [149]. In TME, TAMs are among the most abundant cell type found [161] and the M2-like phenotype inhibits cytotoxic T lymphocytes through the depletion of metabolites, secretion of cytokines and chemokines, and the expression of receptors/ligands for ICs [138]. TAMs induce angiogenesis and tumor progression through the production of VEGF, other cytokines (IL-10, CCL2, CCL17, CCL22, and TGF-beta), and matrix-degrading enzymes [138]. TAMs attract Tregs by secreting chemokines [162] and inhibit cytotoxic T cells through an increase in interferon-gamma-induced PD-L1 expression [163]. MDSCs encompass a heterogeneous population of immature myeloid cells that favor immune suppression and tumor progression. The following mechanisms have been reported to induce immune suppression: (1) inhibition of T cell proliferation and promotion of apoptosis due to increased expression of IDO, which is an enzyme that accounts for tryptophan catabolism and kynurenine production [164]; (2) STAT3 hyper-activation correlating with a noncanonical NF-kB pathway, which leads to IDO up-regulation [165]; (3) secretion of IL-6, IL-10, and TGF beta, which are pro-inflammatory immunosuppressive cytokines [166]; (4) production of reactive oxygen and nitrogen species by attracting other immunosuppressive cells [167].
N2 Phenotype, CD56bright CD16+ NK Subset, DCs, and Exosomes
The N2 phenotype of TANs exerts an immunosuppressive action by reducing the anti-tumor response of the CD4+ and CD8+ T lymphocytes [168]. In the TME, TGF-beta induces the formation of an N2 phenotype that inhibits T cell action through the enhanced production of inducible nitric oxide synthase and arginase [169,170]. Following the degradation of the extracellular matrix, TANs contribute to the release of VEGF, suppress NK-mediated tumor cell clearance, and increase the extravasation of cancer cells [171]. In ER+HER2− breast cancer, an increased neutrophils-to-lymphocytes ratio is directly correlated with a worse prognosis [172,173]. The CD56bright CD16+ NK subset, unlike the CD56bright CD16low/− subset, can promote tumor progression through the matrix metalloproteinase 9, VEGF, and angiogenin secretion and release [174,175]. Interestingly, to highlight the dual role of NK cells, it has also been found that in ER+ and HER2+ breast cancer patients, the infiltration of NK cells correlated with an improved outcome, unlike in TNBC, where it combined with a worse prognosis [176]. A few tumor-infiltrating DCs with immature phenotypes promote endothelial cell migration and tumor growth through the production of pro-angiogenic factors, low expression of co-stimulatory molecules, and over-expression of regulatory molecules [177]. A DCs subset, termed plasmacytoid DCs, together with a decreased antitumor immune response, Tregs increase, and a higher rate occurred in TNBC compared to the other breast cancer subtypes [178]. Breast cancer cells as well as CAFs [179] and bone-marrow-derived cells [180] secrete exosomes that carry PD-L1 [181]. In the TME, exosomes reportedly act as vehicles that transport PD-L1 to different cell types, thereby regulating immune surveillance [182] and circulating exosomes from primary breast tumors recruit MDSCs to pre-metastatic sites [183]. Finally, a less efficient or decreased surface antigen expression of MHC in tumor cells and the up-regulation of immune checkpoint receptors are two main mechanisms of tumor immune evasion that are at least in part due to genetic instability inherent in all tumor cells together with the immune selection process [184]. This means that tumor immunogenicity as a result of genomic aberrations and the TILs rate is a large restraint of the immunological balance in the TME. In fact, genomic aberrations and TILs are only some of the multiple mechanisms contributing to the immune response that are counteracted by multiple others favoring immune inhibition (Figure 1).
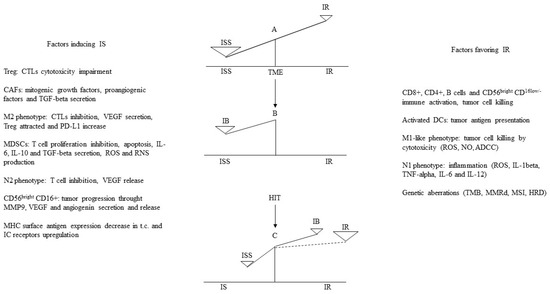
Figure 1.
A-B: In TME, factors inducing ISS commonly by far overcome those favoring IR, thus with regard IB, ISS prevails. C: In ER+ metastatic breast cancer patients in clinical benefit during hormone immunotherapy (HIT), SERMs combined with interferon-beta-interleukin-2 sequence (INF-beta-IL-2) counteract tumor proliferation and immune inhibition and stimulate significant immune response [145], therefore IR prevails in TME. ISS: immune suppression; IR: immune response; IB: immune balance; TME: tumor microenvironment; TILs: tumor infiltrating lymphocytes; DCs: dendritic cells; M: macrophage; N: neutrophil; ADCC: antibody-dependent cell-mediated cytotoxicity; Treg: regulatory T cells; CAF: cancer-associated fibroblast; TGF-beta: transforming growth factor beta; CTLs: cytotoxic T cells; VEGF: vascular endothelial growth factor; PD-L1: programmed death ligand 1; MDSC: myeloid derived suppressor cell; IL: interleukin; ROS: reactive oxygen species; NO: nitric oxide; RNS: reactive nitrogen species; MMP-9: matrix metallo proteinase protein 9; MHC: major histocompatibility complex-1; IC: immune checkpoint; TMB: tumor mutation burden; MMRd: mismatch repair deficiency; MSI: microsatellite instability; HRD: homologous recombination deficiency.
1.5.4. Conditions Favoring a Successful TME Immune Manipulation
Thus far, it is unclear if PD-L1 expression is a predictive biomarker for the response to CIs [43,44]. Similarly, findings on genomic aberrations as predictive biomarkers for the response to DDRi or DDRi + ICB are limited and still need extensive validation in large and well-designed clinical trials [98]. Further, HER2+ is a biomarker predictive of the benefit arising from anti-HER2+ mAb in only one-third of HER2+ breast cancer patients [127]. Conversely, in most cases of ER+ breast cancer, ER expression is a highly predictive biomarker for a response or clinical benefit to anti-estrogens. However, ICIs, DDRi, and anti-HER2+ mAbs therapies affect one or a few pathological molecular pathways, likely without a significant impact on the immunosuppressive TME, while the ER-mediated anti-estrogen action in ER+ breast cancer involves multiple genes and several pathological pathways [157,185,186] favoring the reversion of the immunosuppressive TME. Accordingly, due to its potential capability to revert the immunosuppressive TME, anti-estrogen therapy has been proposed in ER+ cancers, apart from breast cancer, as a treatment that synergizes with conventional therapy [187]. Moreover, ICIs, DDRi, and anti-HER2+ mAb, unlike anti-estrogens, are given in combination with conventional CT, as they often do not work when given alone. In our observational study [145], despite hormone-receptor-positive breast cancer being considered a less immunogenic molecular subtype, anti-estrogens boosted tumor immune response. We hypothesized that in our studied patients, the prolonged G0-G1 state, often concomitant with a decreased tumor burden, accounted for a down-regulation of the main hallmarks sustaining tumor growth, including immune evasion with a large reversion of the immunosuppressive TME [145,188]. A significant peripheral blood increase in T and NK effector cells occurred in all cases that received additional immune-stimulating therapy [189]. In two other pilot studies in which metastatic breast cancer patients had received maintenance-immune-modulating therapy consisting of interleukin-2 and retinoic acid or interferon-β and retinyl palmitate, the immune-modulating maintenance treatment and a concomitant low tumor burden likely favored an immune response and accounted for the significantly improved clinical outcome [190]. Consistently, it has been recently stated that “there are some possible biological explanations to justify why immunotherapy can benefit TNBC patients regardless of PD-L1 positivity in localized disease, but not in the metastatic setting. The host immune system is probably more robust in the early disease due to the limited cancer burden and the major effectiveness in triggering an antitumor immunologic response to new antigens” [43]. We have also recently reported on one breast cancer patient with minimal residual disease (MRD) at high risk of relapse following radical resection of a single lung metastasis and two others with biochemical recurrence after radical prostatectomy who received successful immune therapy [191]. In conclusion, in addition to a prolonged G0-G1 state, our findings along with others suggest a low or undetectable tumor burden for more successful tumor immune manipulation [157].
1.5.5. Metastatic Breast Cancer as an Incurable Disease
Overall, about 20% of breast cancer patients experience recurrence or metastasis within the first five years after primary radical surgery [192]. Recently, rates of distant metastases, ranging from 23.9% in luminal A (ER+ or PR+ and HER2−) to 32% in basal (ER− or PR− and HER2−) and 52.4% in HER2+ (ER− or PR− and HER2+), have been reported in a study conducted in 324 eligible patients with a median follow-up of 7.3 years [193]. In metastatic breast cancer, independent of molecular subtype, the five-year cancer-specific survival rate is approximately 29%, which reduces to 12% in the case of metastatic TNBC [194]. Although two decades have passed since precision medicine and targeted therapies usually combined with conventional chemo or hormone therapy started in oncology, the metastatic disease clinical outcome has improved in small subsets [127,138,195]. The median PFS and/or OS have increased by a few months or a few years at best, and death inevitably occurs in most patients [138,195]. Moreover, due to these targeted treatments, a significant percentage of patients suffer from additional heavy side-effects including irAEs [196,197,198]. Overall, this and the advances in the specific biology of cancer growth and progression suggest a novel therapeutic strategy aimed at obtaining a definite cure by preventing overt metastatic disease.
1.6. Locally Advanced Breast Cancer Patients: A New Paradigm for Additional Adjuvant Therapy
1.6.1. The Conventional Adjuvant CT and the Formation of the Pre-Metastatic Niches (PMNs)
In the Gompertz mathematical model first proposed by Goldie and Coldman in 1979 [199,200], the probability of the existence of nonresistant cells exponentially decreases as the size of the tumor grows, and the double time of cancer cells only begins to increase as the tumor size becomes larger. Eventually, the proliferation rate slows down as the cancer population asymptotically approaches the plateau population; then, the growth process stops due to the lack of space and oxygen. The Gompertz function realistically represents tumor growth because it limits the population growth as the tumor size attains the carrying capacity of the host organ. Since then, the Gompertz model of tumor growth by Goldie and Coldman has been elaborated upon several times [201,202]. This model remains the milestone on which adjuvant CT and radiotherapy (RT) schedules have been designed both regarding their administration timing and dosage and, in the case of CT, for the type of drugs to be combined. Consistent with this mathematical model, the first 4–6 months following the radical resection of a primary tumor is considered the optimal interval time for cyclic adjuvant CT administration. Despite this, large scientific documentation in the last two decades suggests the dissemination of cancer cells at in situ stages well before the detection of a primary tumor [190]. Furthermore, the heterogeneity of “early” metastases in breast cancer occurs due to different microenvironments [203], also termed pre-metastatic niches (PMNs) [204,205]. The formation of PMN involves three successive temporal phases: (a) first, the metastatic microenvironment is educated by the primary tumor; (b) second, the secondary sites recruit immunosuppressive cells; (c) finally, the circulating tumor cells move to PMN [206,207,208]. The process, termed “organo-tropism” or cancer cell “homing,” is driven by specific gene expression and chemokine secretion. However, an increasingly relevant role in the formation of the PMN is attributed to exosomes. Extracellular vesicles and cancer-cell-derived exosomes can interact with inflammatory cytokines (IL-6 and IL-8) to promote the formation of PMN [209,210]. Exosomes promote angiogenesis and permeability in PMN through micro-RNAs secretion [211,212,213] and by carrying many pro-angiogenic molecules including VEGF and matrix metalloproteinases [214,215]. In addition, exosomes are also involved in stromal PMN remodeling by triggering the differentiation of cells [216] or normal breast cancer fibroblasts [217] to CAFs or pro-metastatic CAFs [218].
1.6.2. The Specific Biology of Disseminated Cancer Cells (DCCs)
“Early” DCCs, also named MRD, evolve and acquire specific characteristics that allow them to generate micro-metastases resistant to therapies focused on eliminating the tumor burden residual to the surgical resection of the primary tumor. A population-level dormancy and cellular dormancy models that are not mutually exclusive and co-evolve with the maturation of the metastatic niche have been described in the study. In the former model, limited blood supply, secondary immune-editing, or apoptosis inhibit the expansion of proliferating DCCs, while in the latter model, cells enter the G0-G1 state. Thus, in the “host” metastatic microenvironment of solid cancers, DCCs may enter into a quiescent state to “awaken” and lead to clinically/radiological detectable metastases after years or even decades. During this prolonged interval time, they interact with many micro-environmental signals that can trigger the proliferation process. Therefore, the Gompertz mathematical model of tumor growth proposed by Goldie and Coldman does not fit the specific biology of DCCs that, recently, we have widely reported on [190]. Epigenetic reprogramming mechanisms triggered by micro-environmental or intra-tumor signals likely induce the long-term commitment of DCCs to quiescence while retaining growth potential [219]. We have represented this condition at the PMN as the counterbalancing of immunological and nonimmunological micro-environmental or intra-tumor signals governing an unstable virtual equilibrium line [190]. This instability over time exerts proliferative foci that progress or not depending on the environmental status of the involved niche at that specific time. Therefore, the principal aim should be to turn for a prolonged time, possibly indefinitely, the unstable to a more stable virtual equilibrium line while switching off the proliferative foci [190]. Figure 2 shows in detail this proposal for cases of breast cancer at a high risk of relapse based on additional cyclic immunotherapy combined or alternated with prolonged conventional adjuvant hormone therapy or CT, respectively. We carried out these or similar schedules for 5–6 years or until relapse with preliminary promising findings. Moreover, while the short cyclic CT administration makes it well tolerated [220], the proposed immune therapies did not show relevant AEs and irAEs [145,191,220].
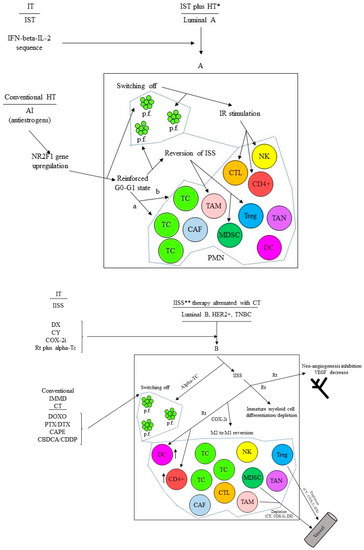
Figure 2.
(A) Additional cyclic immunotherapy (IT) concomitant with prolonged conventional hormone therapy (HT) in luminal A breast cancer at high risk of relapse. * For the schedule see references [143,145]. IST: immunostimulating therapy; IR: immune response; ISS: immune suppression; IFN: interferon; IL-2: interleukin-2; AI: aromatase inhibitor; a: population-level dormancy model; b: cellular dormancy model; PMN: pre-metastatic niche; p.f.: proliferation focus; PMN borders. (B) Additional cyclic immunotherapy (IT) alternated with prolonged conventional adjuvant chemotherapy (CT) in breast cancer other than luminal A and at high risk of relapse. IISS: inhibiting immune suppression; IMMD CT: immune modulatory CT. ** For the schedule see reference [191], this IISS therapy is alternated with four cycles (3 months overall) of conventional IMMD CT; DX: desamethasone; CY: cyclophosphamide; COX-2i: COX-2 inhibitor; Rt: retinyl palmitate; alpha-Tc: alpha tocopheryl acetate; DOXO: doxorubicin; PTX/DX: paclitaxel/docetaxel; CAPE: capecitabine; CBDCA/CDDP: carboplatin/cisplatin; PMN borders.
2. Discussion and Conclusions
Since a long time in the modern era, oncologists have been fascinated by the possibility of using the immune system of a cancer patient to fight cancer. This attractive hypothesis would have allowed cancer to be cured without relevant toxicity. The efforts to pursue this aim enormously increased recently when it was unequivocally proven that, from the beginning, the host immune system can recognize and activate a cell-mediated innate and adaptive immune response against tumor cells [221]. Breast cancer is the most common form of cancer and the second cause of death by cancer in women. In 2020, it represented 30% of female cancers with 276,480 new cases and more than 42,000 estimated deaths [222]. Although breast cancer is considered less immunogenic than others, the HER2+ and TNBC subtypes are thought to be more immunogenic due to a higher tissue TILs rate, genetic alterations, and/or PD-L1 over-expression. Nevertheless, in both these subtypes, a minority of patients benefit from the active immunotherapy given directly with anti HER2+ mAbs or indirectly by ICIs, usually combined with conventional hormone therapy or CT, with concomitant grade 3–4 AEs in about 10–20% of patients. The propensity to induce an immune response is likely inadequately evaluated by a high TILs or CD8+ rate in TME, tumor mutational burden, and other genetic aberrations in tumor cells. In fact, at the TME site, genetic aberrations and TILs or CD8+ are some of the factors contributing to the immune response among the multiple known and unknown mechanisms contributing to the immune balance. Additionally, the extent of any single contribution to the immune balance is unknown. Thus, any cancer type, independent of the currently pre-defined immunogenicity, has inducible immunogenicity proportional to the capability of the available therapeutic means to counteract tumor growth and immune inhibition. The surprisingly promising findings observed in our 2:1 control-case retrospective observational study support this notion; this, along with other findings, suggests that low tumor burden and/or tumor cell quiescence are important conditions favoring a successful immune manipulation [190,191]. This assumption and the absence of therapies to cure the overt metastatic disease suggest concentrating any effort during MRD where both conditions coexist. Our proposed strategy combines a prolonged cyclic immune manipulation with conventional hormone or short-term CT to indefinitely maintain the PMN tumor cell quiescence while switching off the occasionally proliferating foci. This therapeutic strategy could significantly decrease the rate of relapses in the first 5–6 years with a huge saving of expenses by the Health National Services and, more importantly, of lives and pains. Based on our and others’ preliminary work and pilot studies [145,191,220], specific clinical trials should be easy to design, and safe and cheap to carry out. Multinational drug companies do not have an interest to sponsor such investigational trials as the proposed schedules include repurposed drugs without patents and at low cost. Despite this, one can expect that governmental drug agencies or private institutions will start such trials that, if successful, can be extended to other solid tumors.
Author Contributions
A.N. conceived and wrote the article; P.F. and A.C. have critically revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work did not receive external funding.
Data Availability Statement
Not applicable.
Conflicts of Interest
Authors declare no conflict of interest.
References
- Globocan. Available online: https://gco.iarc.fr (accessed on 26 July 2022).
- Emens, L.A. Breast Cancer Immunotherapy: Facts and Hopes. Clin. Cancer Res. 2018, 4, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Henriques, B.; Mendes, F.; Martins, D. Immunotherapy in Breast Cancer: When, How, and What Challenges? Biomedicines 2021, 9, 1687. [Google Scholar] [CrossRef]
- Senkus, E.; Cardoso, F.; Pagani, O. Time for more optimism in metastatic breast cancer? Cancer Treat. Rev. 2014, 40, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Carey, L.A.; Perou, C.M.; Livasy, C.A.; Dressler, L.G.; Cowan, D.; Conway, K.; Karaca, G.; Troester, M.A.; Tse, C.K.; Edmiston, S.; et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 2006, 295, 2492–2502. [Google Scholar] [CrossRef]
- Tong, C.W.S.; Wu, M.; Cho, W.C.S.; To, K.K.W. Recent Advances in the Treatment of Breast Cancer. Front. Oncol. 2018, 8, 227. [Google Scholar] [CrossRef]
- Ellis, H.; Ma, C.X. PI3K Inhibitors in Breast Cancer Therapy. Curr. Oncol. Rep. 2019, 21, 110. [Google Scholar] [CrossRef]
- Barzaman, K.; Karami, J.; Zarei, Z.; Hosseinzadeh, A.; Kazemi, M.H.; Moradi-Kalbolandi, S.; Safari, E.; Farahmand, L. Breast cancer: Biology, biomarkers, and treatments. Int. Immunopharmacol. 2020, 84, 106535. [Google Scholar] [CrossRef]
- Arab, A.; Yazdian-Robati, R.; Behravan, J. HER2-Positive Breast Cancer Immunotherapy: A Focus on Vaccine Development. Arch. Immunol. Ther. Exp. 2020, 68, 2. [Google Scholar] [CrossRef]
- Sivaganesh, V.; Promi, N.; Maher, S.; Peethambaran, B. Emerging Immunotherapies against Novel Molecular Targets in Breast Cancer. Int. J. Mol. Sci. 2021, 22, 2433. [Google Scholar] [CrossRef]
- Tavares, D.F.; Ribeiro, V.C.; Andrade, M.; Cardoso-Júnior, L.M.; Teixeira, T.R.G.; Varrone, G.R.; Britto, R.L. Immunotherapy using PD-1/PDL-1 inhibitors in metastatic triple-negative breast cancer: A systematic review. Oncol. Rev. 2021, 15, 497. [Google Scholar] [CrossRef]
- Barroso-Sousa, R.; Keenan, T.E.; Li, T.; Tayob, N.; Trippa, L.; Pastorello, R.G.; Richardson Iii, E.T.; Dillon, D.; Amoozgar, Z.; Overmoyer, B.; et al. Nivolumab in combination with cabozantinib for metastatic triple-negative breast cancer: A phase II and biomarker study. NPJ Breast Cancer 2021, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Sternschuss, M.; Yerushalmi, R.; Saleh, R.R.; Amir, E.; Goldvaser, H. Efficacy and safety of neoadjuvant immune checkpoint inhibitors in early-stage triple-negative breast cancer: A systematic review and meta-analysis. J. Cancer Res. Clin. Oncol. 2021, 147, 3369–3379. [Google Scholar] [CrossRef] [PubMed]
- Galván Morales, M.A.; Barrera Rodríguez, R.; Santiago Cruz, J.R.; Teran, L.M. Overview of New Treatments with Immunotherapy for Breast Cancer and a Proposal of a Combination Therapy. Molecules 2020, 25, 5686. [Google Scholar] [CrossRef] [PubMed]
- Luen, S.J.; Savas, P.; Fox, S.B.; Salgado, R.; Loi, S. Tumour-infiltrating lymphocytes and the emerging role of immunotherapy in breast cancer. Pathology 2017, 49, 141–155. [Google Scholar] [CrossRef]
- Kim, H.M.; Lee, J.; Koo, J.S. Clinicopathological and prognostic significance of programmed death ligand-1 expression in breast cancer: A meta-analysis. BMC Cancer 2017, 17, 690. [Google Scholar] [CrossRef] [PubMed]
- Salemme, V.; Centonze, G.; Cavallo, F.; Defilippi, P.; Conti, L. The Crosstalk Between Tumor Cells and the Immune Microenvironment in Breast Cancer: Implications for Immunotherapy. Front. Oncol. 2021, 11, 610303. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Hu, L.; Zhang, X.; Jiang, S.; Li, J.; Zhang, Z.; Wang, X. The Diverse Function of PD-1/PD-L Pathway Beyond Cancer. Front. Immunol. 2019, 10, 2298. [Google Scholar] [CrossRef] [PubMed]
- Planes-Laine, G.; Rochigneux, P.; Bertucci, F.; Chrétien, A.S.; Viens, P.; Sabatier, R.; Gonçalves, A. PD-1/PD-L1 Targeting in Breast Cancer: The First Clinical Evidences Are Emerging. A Literature Review. Cancers 2019, 11, 1033. [Google Scholar] [CrossRef] [PubMed]
- Azuma, T.; Yao, S.; Zhu, G.; Flies, A.S.; Flies, S.J.; Chen, L. B7-H1 is a ubiquitous antiapoptotic receptor on cancer cells. Blood 2008, 111, 3635–3643. [Google Scholar] [CrossRef]
- Esensten, J.H.; Helou, Y.A.; Chopra, G.; Weiss, A.; Bluestone, J.A. CD28 Costimulation: From Mechanism to Therapy. Immunity 2016, 44, 973–988. [Google Scholar] [CrossRef]
- Bell, R.B.; Feng, Z.; Bifulco, C.B.; Leidner, R.; Weinberg, A.; Fox, B.A. Immunotherapy. In Oral, Head and Neck Oncology and Reconstructive Surgery; Bell, R.B., Fernandes, R.P., Andersen, P.E., Eds.; Elsevier: New York, NY, USA, 2018; pp. 314–340. [Google Scholar]
- Seidel, J.A.; Otsuka, A.; Kabashima, K. Anti-PD-1 and Anti-CTLA-4 Therapies in Cancer: Mechanisms of Action, Efficacy, and Limitations. Front. Oncol. 2018, 8, 86. [Google Scholar] [CrossRef] [PubMed]
- Fellner, C. Ipilimumab (yervoy) prolongs survival in advanced melanoma: Serious side effects and a hefty price tag may limit its use. Pharm. Ther. 2012, 37, 503–530. [Google Scholar]
- Yang, Q.; Cao, W.; Wang, Z.; Zhang, B.; Liu, J. Regulation of cancer immune escape: The roles of miRNAs in immune checkpoint proteins. Cancer Lett. 2018, 431, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M. Immune checkpoint blockade: A common denominator approach to cancer therapy. Cancer Cell 2015, 27, 450–461. [Google Scholar] [CrossRef]
- Greenwald, R.J.; Freeman, G.J.; Sharpe, A.H. The B7 family revisited. Annu. Rev. Immunol. 2005, 23, 515–548. [Google Scholar] [CrossRef]
- Nallasamy, P.; Chava, S.; Verma, S.S.; Mishra, S.; Gorantla, S.; Coulter, D.W.; Byrareddy, S.N.; Batra, S.K.; Gupta, S.C.; Challagundla, K.B. PD-L1, inflammation, non-coding RNAs, and neuroblastoma: Immuno-oncology perspective. Semin. Cancer Biol. 2018, 52 Pt 2, 53–65. [Google Scholar] [CrossRef]
- Vennapusa, B.; Baker, B.; Kowanetz, M.; Boone, J.; Menzl, I.; Bruey, J.M.; Fine, G.; Mariathasan, S.; McCaffery, I.; Mocci, S.; et al. Development of a PD-L1 Complementary Diagnostic Immunohistochemistry Assay (SP142) for Atezolizumab. Appl. Immunohistochem. Mol. Morphol. 2019, 27, 92–100. [Google Scholar] [CrossRef]
- García-Aranda, M.; Redondo, M. Immunotherapy: A Challenge of Breast Cancer Treatment. Cancers 2019, 11, 1822. [Google Scholar] [CrossRef]
- Dimitrova, N.; Saz Parkinson, Z.; Bramesfeld, A.; Uluturk Tekin, A.; Bocchi, G.; Pylkkanen, L.; Lopez Alcalde, J.; Neamtiu, L.; Ambrosio, M.; Deandrea, S.; et al. European Guidelines for Breast Cancer Screening and Diagnosis–the European Breast Guidelines; EUR 28360 EN, JRC104007; Publications Office of the European Union: Luxembourg, 2016. [Google Scholar]
- Nicolini, A.; Ferrari, P.; Duffy, M.J. Prognostic and predictive biomarkers in breast cancer: Past, present and future. Semin. Cancer Biol. 2018, 52 Pt 1, 56–73. [Google Scholar] [CrossRef]
- Erber, R.; Hartmann, A. Understanding PD-L1 Testing in Breast Cancer: A Practical Approach. Breast Care 2020, 15, 481–490. [Google Scholar] [CrossRef]
- Noske, A.; Wagner, D.C.; Schwamborn, K.; Foersch, S.; Steiger, K.; Kiechle, M.; Karapetyan, S.; Oettler, D.; Hapfelmeier, A.; Roth, W.; et al. Comparison study of different programmed death-ligand 1(PD-L1) assays, readers and scoring methods in triple-negative breast cancer (TNBC). Ann. Oncol. 2021, 32 (Suppl. 2), S26. [Google Scholar] [CrossRef]
- Vigliar, E.; Malapelle, U.; Bono, F.; Fusco, N.; Cortinovis, D.; Valtorta, E.; Spyridon, A.; Bimbatti, M.; Zocchi, M.; Piva, C.; et al. The Reproducibility of the Immunohistochemical PD-L1 Testing in Non-Small-Cell Lung Cancer: A Multicentric Italian Experience. Biomed. Res. Int. 2019, 2019, 6832909. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, C.; Abreu, D.R.; Felip, E.; Carcereny, E.; Gottfried, M.; Wehler, T.; Ahn, M.J.; Dolled-Filhart, M.; Zhang, J.; Shentu, Y.; et al. Prevalence of PD-L1 expression in patients with non-small cell lung cancer screened for enrollment in KEYNOTE-001, -010, and -024. Ann. Oncol. 2016, 27 (Suppl. S6), vi363. [Google Scholar] [CrossRef]
- Karlsson, E.; Appelgren, J.; Solterbeck, A.; Bergenheim, M.; Alvariza, V.; Bergh, J. Breast cancer during follow-up and progression—A population based cohort on new cancers and changed biology. Eur. J. Cancer 2014, 50, 2916–2924. [Google Scholar] [CrossRef]
- Lindström, L.S.; Karlsson, E.; Wilking, U.M.; Johansson, U.; Hartman, J.; Lidbrink, E.K.; Hatschek, T.; Skoog, L.; Bergh, J. Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J. Clin. Oncol. 2012, 30, 2601–2608. [Google Scholar] [CrossRef]
- Schrijver, W.A.M.E.; Suijkerbuijk, K.P.M.; van Gils, C.H.; van der Wall, E.; Moelans, C.B.; van Diest, P.J. Receptor Conversion in Distant Breast Cancer Metastases: A Systematic Review and Meta-analysis. J. Natl. Cancer Inst. 2018, 110, 568–580. [Google Scholar] [CrossRef]
- Amir, E.; Miller, N.; Geddie, W.; Freedman, O.; Kassam, F.; Simmons, C.; Oldfield, M.; Dranitsaris, G.; Tomlinson, G.; Laupacis, A.; et al. Prospective study evaluating the impact of tissue confirmation of metastatic disease in patients with breast cancer. J. Clin. Oncol. 2012, 30, 587–592. [Google Scholar] [CrossRef]
- Foukakis, T.; Astrom, G.; Lindström, L.; Hatschek, T.; Bergh, J. When to order a biopsy to characterise a metastatic relapse in breast cancer. Ann. Oncol. 2012, 23 (Suppl. 10), 349–353. [Google Scholar] [CrossRef]
- Buisseret, L.; Garaud, S.; de Wind, A.; Van den Eynden, G.; Boisson, A.; Solinas, C.; Gu-Trantien, C.; Naveaux, C.; Lodewyckx, J.N.; Duvillier, H.; et al. Tumor-infiltrating lymphocyte composition, organization and PD-1/ PD-L1 expression are linked in breast cancer. Oncoimmunology 2016, 6, e1257452. [Google Scholar] [CrossRef]
- Núñez Abad, M.; Calabuig-Fariñas, S.; Lobo de Mena, M.; Torres-Martínez, S.; García González, C.; García García, J.Á.; Iranzo González-Cruz, V.; Camps Herrero, C. Programmed Death-Ligand 1 (PD-L1) as Immunotherapy Biomarker in Breast Cancer. Cancers 2022, 14, 307. [Google Scholar] [CrossRef]
- Boman, C.; Zerdes, I.; Mårtensson, K.; Bergh, J.; Foukakis, T.; Valachis, A.; Matikas, A. Discordance of PD-L1 status between primary and metastatic breast cancer: A systematic review and meta-analysis. Cancer Treat. Rev. 2021, 99, 102257. [Google Scholar] [CrossRef] [PubMed]
- Hegde, P.S.; Chen, D.S. Top 10 Challenges in Cancer Immunotherapy. Immunity 2020, 52, 17–35. [Google Scholar] [CrossRef] [PubMed]
- Emens, L.A.; Molinero, L.; Loi, S.; Rugo, H.S.; Schneeweiss, A.; Diéras, V.; Iwata, H.; Barrios, C.H.; Nechaeva, M.; Nguyen-Duc, A.; et al. Atezolizumab and nab-Paclitaxel in Advanced Triple-Negative Breast Cancer: Biomarker Evaluation of the IMpassion130 Study. J. Natl. Cancer Inst. 2021, 113, 1005–1016. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.S.P.; Haberberger, J.; Severson, E.; Duncan, D.L.; Hemmerich, A.; Edgerly, C.; Ferguson, N.L.; Williams, E.; Elvin, J.; Vergilio, J.A.; et al. A pan-cancer analysis of PD-L1 immunohistochemistry and gene amplification, tumor mutation burden and microsatellite instability in 48,782 cases. Mod. Pathol. 2021, 34, 252–263. [Google Scholar] [CrossRef]
- Paver, E.C.; Cooper, W.A.; Colebatch, A.J.; Ferguson, P.M.; Hill, S.K.; Lum, T.; Shin, J.S.; O’Toole, S.; Anderson, L.; Scolyer, R.A.; et al. Programmed death ligand-1 (PD-L1) as a predictive marker for immunotherapy in solid tumours: A guide to immunohistochemistry implementation and interpretation. Pathology 2021, 53, 141–156. [Google Scholar] [CrossRef]
- Jeong, S.; Lee, N.; Park, M.J.; Jeon, K.; Song, W. Currently Used Laboratory Methodologies for Assays Detecting PD-1, PD-L1, PD-L2 and Soluble PD-L1 in Patients with Metastatic Breast Cancer. Cancers 2021, 13, 5225. [Google Scholar] [CrossRef]
- Parra, E.R.; Villalobos, P.; Mino, B.; Rodriguez-Canales, J. Comparison of Different Antibody Clones for Immunohistochemistry Detection of Programmed Cell Death Ligand 1 (PD-L1) on Non-Small Cell Lung Carcinoma. Appl. Immunohistochem. Mol. Morphol. 2018, 26, 83–93. [Google Scholar] [CrossRef]
- Beaver, J.A.; Tzou, A.; Blumenthal, G.M.; McKee, A.E.; Kim, G.; Pazdur, R.; Philip, R. An FDA Perspective on the Regulatory Implications of Complex Signatures to Predict Response to Targeted Therapies. Clin. Cancer Res. 2017, 23, 1368–1372. [Google Scholar] [CrossRef]
- Schmid, P.; Adams, S.; Rugo, H.S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Hegg, R.; Im, S.A.; Wright, G.S.; et al. IMpassion130 Trial Investigators Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. N. Engl. J. Med. 2018, 379, 2108–2121. [Google Scholar] [CrossRef]
- Rugo, H.S.; Loi, S.; Adams, S.; Schmid, P.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Winer, E.P.; Kockx, M.M.; et al. PD-L1 Immunohistochemistry Assay Comparison in Atezolizumab Plus nab-Paclitaxel-Treated Advanced Triple-Negative Breast Cancer. J. Natl. Cancer Inst. 2021, 113, 1733–1743. [Google Scholar] [CrossRef]
- Ma, J.; Li, J.; Qian, M.; Han, W.; Tian, M.; Li, Z.; Wan, G.Z.; He, S.; Wu, K. PD-L1 expression and the prognostic significance in gastric cancer: A retrospective comparison of three PD-L1 antibody clones (SP142, 28–8 and E1L3N). Diagn. Pathol. 2018, 13, 91. [Google Scholar] [CrossRef] [PubMed]
- Doroshow, D.B.; Bhalla, S.; Beasley, M.B.; Sholl, L.M.; Kerr, K.M.; Gnjatic, S.; Wistuba, I.I.; Rimm, D.L.; Tsao, M.S.; Hirsch, F.R. PD-L1 as a biomarker of response to immune-checkpoint inhibitors. Nat. Rev. Clin. Oncol. 2021, 18, 345–362. [Google Scholar] [CrossRef]
- Eckstein, M.; Cimadamore, A.; Hartmann, A.; Lopez-Beltran, A.; Cheng, L.; Scarpelli, M.; Montironi, R.; Gevaert, T. PD-L1 assessment in urothelial carcinoma: A practical approach. Ann. Transl. Med. 2019, 7, 690. [Google Scholar] [CrossRef] [PubMed]
- De Marchi, P.; Leal, L.F.; Duval da Silva, V.; da Silva, E.C.A.; Cordeiro de Lima, V.C.; Reis, R.M. PD-L1 expression by Tumor Proportion Score (TPS) and Combined Positive Score (CPS) are similar in non-small cell lung cancer (NSCLC). J. Clin. Pathol. 2021, 74, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Rugo, H.S.; Delord, J.P.; Im, S.A.; Ott, P.A.; Piha-Paul, S.A.; Bedard, P.L.; Sachdev, J.; Le Tourneau, C.; van Brummelen, E.; Varga, A.; et al. Safety and Antitumor Activity of Pembrolizumab in Patients with Estrogen Receptor-Positive/Human Epidermal Growth Factor Receptor 2-Negative Advanced Breast Cancer. Clin. Cancer Res. 2018, 24, 2804–2811. [Google Scholar] [CrossRef]
- Adams, S.; Schmid, P.; Rugo, H.S.; Winer, E.P.; Loirat, D.; Awada, A.; Cescon, D.W.; Iwata, H.; Campone, M.; Nanda, R.; et al. Pembrolizumab monotherapy for previously treated metastatic triple-negative breast cancer: Cohort A of the phase II KEYNOTE-086 study. Ann. Oncol. 2019, 30, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.; Loi, S.; Toppmeyer, D.; Cescon, D.W.; De Laurentiis, M.; Nanda, R.; Winer, E.P.; Mukai, H.; Tamura, K.; Armstrong, A.; et al. Pembrolizumab monotherapy for previously untreated, PD-L1-positive, metastatic triple-negative breast cancer: Cohort B of the phase II KEYNOTE-086 study. Ann. Oncol. 2019, 30, 405–411. [Google Scholar] [CrossRef]
- Winer, E.P.; Lipatov, O.; Im, S.A.; Goncalves, A.; Muñoz-Couselo, E.; Lee, K.S.; Schmid, P.; Tamura, K.; Testa, L.; Witzel, I.; et al. KEYNOTE-119 investigators. Pembrolizumab versus investigator-choice chemotherapy for metastatic triple-negative breast cancer (KEYNOTE-119): A randomised, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 499–511. [Google Scholar] [CrossRef]
- Cortes, J.; Cescon, D.W.; Rugo, H.S.; Nowecki, Z.; Im, S.A.; Yusof, M.M.; Gallardo, C.; Lipatov, O.; Barrios, C.H.; Holgado, E.; et al. KEYNOTE-355 Investigators. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): A randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet 2020, 396, 1817–1828. [Google Scholar]
- Schmid, P.; Cortes, J.; Pusztai, L.; McArthur, H.; Kümmel, S.; Bergh, J.; Denkert, C.; Park, Y.H.; Hui, R.; Harbeck, N.; et al. KEYNOTE-522 Investigators. Pembrolizumab for Early Triple-Negative Breast Cancer. N. Engl. J. Med. 2020, 382, 810–821. [Google Scholar] [CrossRef]
- Adel, N.G. Current treatment landscape and emerging therapies for metastatic triple-negative breast cancer. Am. J. Manag. Care 2021, 27 (Suppl. 5), S87–S96. [Google Scholar] [PubMed]
- Loi, S.; Giobbie-Hurder, A.; Gombos, A.; Bachelot, T.; Hui, R.; Curigliano, G.; Campone, M.; Biganzoli, L.; Bonnefoi, H.; Jerusalem, G.; et al. International Breast Cancer Study Group and the Breast International Group. Pembrolizumab plus trastuzumab in trastuzumab-resistant, advanced, HER2-positive breast cancer (PANACEA): A single-arm, multicentre, phase 1b-2 trial. Lancet Oncol. 2019, 20, 371–382. [Google Scholar] [CrossRef]
- Voorwerk, L.; Slagter, M.; Horlings, H.M.; Sikorska, K.; van de Vijver, K.K.; de Maaker, M.; Nederlof, I.; Kluin, R.; Warren, S.; Ong, S.; et al. Immune induction strategies in metastatic triple-negative breast cancer to enhance the sensitivity to PD-1 blockade: The TONIC trial. Nat. Med. 2019, 25, 920–928. [Google Scholar] [CrossRef]
- Ahmed, K.A.; Kim, Y.; Arrington, J.A.; Kim, S.; DeJesus, M.; Soyano, A.E.; Armaghani, A.J.; Costa, R.; Khong, H.T.; Loftus, L.S.; et al. Nivolumab and Stereotactic Radiosurgery for Patients With Breast Cancer Brain Metastases: A Nonrandomized, Open-Label Phase 1b Study. Adv. Radiat. Oncol. 2021, 6, 100798. [Google Scholar] [CrossRef] [PubMed]
- Clynes, R.A.; Towers, T.L.; Presta, L.G.; Ravetch, J.V. Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nat. Med. 2000, 6, 443–446. [Google Scholar] [CrossRef]
- Emens, L.A.; Cruz, C.; Eder, J.P.; Braiteh, F.; Chung, C.; Tolaney, S.M.; Kuter, I.; Nanda, R.; Cassier, P.A.; Delord, J.P.; et al. Long-term Clinical Outcomes and Biomarker Analyses of Atezolizumab Therapy for Patients With Metastatic Triple-Negative Breast Cancer: A Phase 1 Study. JAMA Oncol. 2019, 5, 74–82. [Google Scholar] [CrossRef]
- Schmid, P.; Rugo, H.S.; Adams, S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Henschel, V.; Molinero, L.; Chui, S.Y.; et al. IMpassion130 Investigators. Atezolizumab plus nab-paclitaxel as first-line treatment for unresectable, locally advanced or metastatic triple-negative breast cancer (IMpassion130): Updated efficacy results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2020, 21, 44–59. [Google Scholar]
- Tokumaru, Y.; Joyce, D.; Takabe, K. Current status and limitations of immunotherapy for breast cancer. Surgery 2020, 167, 628–630. [Google Scholar] [CrossRef]
- Franzoi, M.A.; Romano, E.; Piccart, M. Immunotherapy for early breast cancer: Too soon, too superficial, or just right? Ann. Oncol. 2021, 32, 323–336. [Google Scholar] [CrossRef]
- Emens, L.A.; Adams, S.; Barrios, C.H.; Diéras, V.; Iwata, H.; Loi, S.; Rugo, H.S.; Schneeweiss, A.; Winer, E.P.; Patel, S.; et al. First-line atezolizumab plus nab-paclitaxel for unresectable, locally advanced, or metastatic triple-negative breast cancer: IMpassion130 final overall survival analysis. Ann. Oncol. 2021, 32, 983–993. [Google Scholar] [CrossRef]
- Mittendorf, E.A.; Zhang, H.; Barrios, C.H.; Saji, S.; Jung, K.H.; Hegg, R.; Koehler, A.; Sohn, J.; Iwata, H.; Telli, M.L.; et al. Neoadjuvant atezolizumab in combination with sequential nab-paclitaxel and anthracycline-based chemotherapy versus placebo and chemotherapy in patients with early-stage triple-negative breast cancer (IMpassion031): A randomised, double-blind, phase 3 trial. Lancet 2020, 396, 1090–1100. [Google Scholar] [CrossRef]
- Miles, D.; Gligorov, J.; André, F.; Cameron, D.; Schneeweiss, A.; Barrios, C.; Xu, B.; Wardley, A.; Kaen, D.; Andrade, L.; et al. IMpassion131 investigators. Primary results from IMpassion131, a double-blind, placebo-controlled, randomised phase III trial of first-line paclitaxel with or without atezolizumab for unresectable locally advanced/metastatic triple-negative breast cancer. Ann. Oncol. 2021, 32, 994–1004. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, A.S.; Nichols, E.; Cimino-Mathews, A.; Peer, C.; Cao, L.; Lee, M.J.; Kohn, E.C.; Annunziata, C.M.; Lipkowitz, S.; Trepel, J.B.; et al. A phase I study of the PD-L1 inhibitor, durvalumab, in combination with a PARP inhibitor, olaparib, and a VEGFR1-3 inhibitor, cediranib, in recurrent women’s cancers with biomarker analyses. J. Immunother. Cancer 2019, 7, 197. [Google Scholar] [CrossRef] [PubMed]
- Loibl, S.; Untch, M.; Burchardi, N.; Huober, J.; Sinn, B.V.; Blohmer, J.U.; Grischke, E.M.; Furlanetto, J.; Tesch, H.; Hanusch, C.; et al. A randomised phase II study investigating durvalumab in addition to an anthracycline taxane-based neoadjuvant therapy in early triple-negative breast cancer: Clinical results and biomarker analysis of GeparNuevo study. Ann. Oncol. 2019, 30, 1279–1288. [Google Scholar] [CrossRef]
- Bachelot, T.; Filleron, T.; Bieche, I.; Arnedos, M.; Campone, M.; Dalenc, F.; Coussy, F.; Sablin, M.P.; Debled, M.; Lefeuvre-Plesse, C.; et al. Durvalumab compared to maintenance chemotherapy in metastatic breast cancer: The randomized phase II SAFIR02-BREAST IMMUNO trial. Nat. Med. 2021, 27, 250–255. [Google Scholar] [CrossRef]
- Dirix, L.Y.; Takacs, I.; Jerusalem, G.; Nikolinakos, P.; Arkenau, H.T.; Forero-Torres, A.; Boccia, R.; Lippman, M.E.; Somer, R.; Smakal, M.; et al. Avelumab, an anti-PD-L1 antibody, in patients with locally advanced or metastatic breast cancer: A phase 1b JAVELIN Solid Tumor study. Breast Cancer Res. Treat. 2018, 167, 671–686. [Google Scholar] [CrossRef]
- Qi, Y.; Zhang, L.; Wang, Z.; Kong, X.; Zhai, J.; Fang, Y.; Wang, J. Efficacy and Safety of Anti-PD-1/ PD-L1 Monotherapy for Metastatic Breast Cancer: Clinical Evidence. Front. Pharmacol. 2021, 12, 653521. [Google Scholar] [CrossRef]
- Jiang, D.M.; Fyles, A.; Nguyen, L.T.; Neel, B.G.; Sacher, A.; Rottapel, R.; Wang, B.X.; Ohashi, P.S.; Sridhar, S.S. Phase I study of local radiation and tremelimumab in patients with inoperable locally recurrent or metastatic breast cancer. Oncotarget 2019, 10, 2947–2958. [Google Scholar] [CrossRef]
- Kyte, J.A.; Andresen, N.K.; Russnes, H.G.; Fretland, S.Ø.; Falk, R.S.; Lingjærde, O.C.; Naume, B. ICON: A randomized phase IIb study evaluating immunogenic chemotherapy combined with ipilimumab and nivolumab in patients with metastatic hormone receptor positive breast cancer. J. Transl. Med. 2020, 18, 269. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H.; Mo, H.; Hu, X.; Gao, R.; Zhao, Y.; Liu, B.; Niu, L.; Sun, X.; Yu, X.; et al. Single-cell analyses reveal key immune cell subsets associated with response to PD-L1 blockade in triple-negative breast cancer. Cancer Cell 2021, 39, 1578–1593.e8. [Google Scholar] [CrossRef]
- Di Spazio, L.; Rivano, M.; Cancanelli, L.; Chiumente, M.; Mengato, D.; Messori, A. The Degree of Programmed Death-Ligand 1 (PD-L1) Positivity as a Determinant of Outcomes in Metastatic Triple-Negative Breast Cancer Treated With First-Line Immune Checkpoint Inhibitors. Cureus 2022, 14, e21065. [Google Scholar] [CrossRef] [PubMed]
- Keenan, T.E.; Tolaney, S.M. Role of Immunotherapy in Triple-Negative Breast Cancer. J. Natl. Compr. Cancer Netw. 2020, 18, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Pilzecker, B.; Buoninfante, O.A.; Jacobs, H. DNA damage tolerance in stem cells, ageing, mutagenesis, disease and cancer therapy. Nucleic Acids Res. 2019, 47, 7163–7181. [Google Scholar] [CrossRef] [PubMed]
- Pilié, P.G.; Tang, C.; Mills, G.B.; Yap, T.A. State-of-the-art strategies for targeting the DNA damage response in cancer. Nat. Rev. Clin. Oncol. 2019, 16, 81–104. [Google Scholar] [CrossRef]
- Stewart, R.A.; Pilié, P.G.M.; Yap, T.A. Development of PARP and Immune-Checkpoint Inhibitor Combinations. Cancer Res. 2018, 78, 6717–6725. [Google Scholar] [CrossRef]
- Mirza, M.R.; Monk, B.J.; Herrstedt, J.; Oza, A.M.; Mahner, S.; Redondo, A.; Fabbro, M.; Ledermann, J.A.; Lorusso, D.; Vergote, I.; et al. ENGOT-OV16/NOVA Investigators. Niraparib Maintenance Therapy in Platinum-Sensitive, Recurrent Ovarian Cancer. N. Engl. J. Med. 2016, 375, 2154–2164. [Google Scholar] [CrossRef]
- Swisher, E.M.; Lin, K.K.; Oza, A.M.; Scott, C.L.; Giordano, H.; Sun, J.; Konecny, G.E.; Coleman, R.L.; Tinker, A.V.; O’Malley, D.M.; et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 Part 1): An international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017, 18, 75–87. [Google Scholar] [CrossRef]
- Chopra, N.; Tovey, H.; Pearson, A.; Cutts, R.; Toms, C.; Proszek, P.; Hubank, M.; Dowsett, M.; Dodson, A.; Daley, F.; et al. Homologous recombination DNA repair deficiency and PARP inhibition activity in primary triple negative breast cancer. Nat. Commun. 2020, 11, 2662. [Google Scholar] [CrossRef]
- Min, A.; Kim, K.; Jeong, K.; Choi, S.; Kim, S.; Suh, K.J.; Lee, K.H.; Kim, S.; Im, S.A. Homologous repair deficiency score for identifying breast cancers with defective DNA damage response. Sci. Rep. 2020, 10, 12506. [Google Scholar] [CrossRef]
- Timperi, E.; Vissio, E.; Marchiò, C.; Romano, E. The Immune Landscape in Women Cancers. Cancer Treat. Res. 2020, 180, 215–249. [Google Scholar]
- Denkert, C.; Liedtke, C.; Tutt, A.; von Minckwitz, G. Molecular alterations in triple-negative breast cancer-the road to new treatment strategies. Lancet 2017, 389, 2430–2442. [Google Scholar] [CrossRef]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Davies, H.; Morganella, S.; Purdie, C.A.; Jang, S.J.; Borgen, E.; Russnes, H.; Glodzik, D.; Zou, X.; Viari, A.; Richardson, A.L.; et al. Whole-Genome Sequencing Reveals Breast Cancers with Mismatch Repair Deficiency. Cancer Res. 2017, 77, 4755–4762. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, J.; Chen, J.; Zhou, Q. The developing landscape of combinatorial therapies of immune checkpoint blockade with DNA damage repair inhibitors for the treatment of breast and ovarian cancers. J. Hematol. Oncol. 2021, 14, 206. [Google Scholar] [CrossRef]
- Shevtsov, M.; Sato, H.; Multhoff, G.; Shibata, A. Novel Approaches to Improve the Efficacy of Immuno-Radiotherapy. Front. Oncol. 2019, 9, 156. [Google Scholar] [CrossRef]
- Lai, Q.; Wang, H.; Li, A.; Xu, Y.; Tang, L.; Chen, Q.; Zhang, C.; Gao, Y.; Song, J.; Du, Z. Decitibine improve the efficiency of anti-PD-1 therapy via activating the response to IFN/PD-L1 signal of lung cancer cells. Oncogene 2018, 37, 2302–2312. [Google Scholar] [CrossRef]
- Cao, D.; Zhao, J.; Nguyan, L.N.; Nguyen, L.; Khanal, S.; Dang, X.; Schank, M.; Chand Thakuri, B.K.; Wu, X.Y.; Morrison, Z.D.; et al. Disruption of Telomere Integrity and DNA Repair Machineries by KML001 Induces T Cell Senescence, Apoptosis, and Cellular Dysfunctions. Front. Immunol. 2019, 10, 1152. [Google Scholar] [CrossRef]
- Li, A.; Yi, M.; Qin, S.; Chu, Q.; Luo, S.; Wu, K. Prospects for combining immune checkpoint blockade with PARP inhibition. J. Hematol. Oncol. 2019, 12, 98. [Google Scholar] [CrossRef]
- Conway, J.R.; Kofman, E.; Mo, S.S.; Elmarakeby, H.; Van Allen, E. Genomics of response to immune checkpoint therapies for cancer: Implications for precision medicine. Genome Med. 2018, 10, 93. [Google Scholar] [CrossRef]
- Dillon, M.T.; Bergerhoff, K.F.; Pedersen, M.; Whittock, H.; Crespo-Rodriguez, E.; Patin, E.C.; Pearson, A.; Smith, H.G.; Paget, J.; Patel, R.R.; et al. ATR Inhibition Potentiates the Radiation-induced Inflammatory Tumor Microenvironment. Clin. Cancer Res. 2019, 25, 3392–3403. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Cimino-Mathews, A.; Peer, C.J.; Zimmer, A.; Lipkowitz, S.; Annunziata, C.M.; Cao, L.; Harrell, M.I.; Swisher, E.M.; Houston, N.; et al. Safety and Clinical Activity of the Programmed Death-Ligand 1 Inhibitor Durvalumab in Combination With Poly (ADP-Ribose) Polymerase Inhibitor Olaparib or Vascular Endothelial Growth Factor Receptor 1-3 Inhibitor Cediranib in Women’s Cancers: A Dose-Escalation, Phase I Study. J. Clin. Oncol. 2017, 35, 2193–2202. [Google Scholar] [PubMed]
- Vinayak, S.; Tolaney, S.M.; Schwartzberg, L.; Mita, M.; McCann, G.; Tan, A.R.; Wahner-Hendrickson, A.E.; Forero, A.; Anders, C.; Wulf, G.M.; et al. Open-label Clinical Trial of Niraparib Combined With Pembrolizumab for Treatment of Advanced or Metastatic Triple-Negative Breast Cancer. JAMA Oncol. 2019, 5, 1132–1140. [Google Scholar] [CrossRef]
- Domchek, S.M.; Postel-Vinay, S.; Im, S.A.; Park, Y.H.; Delord, J.P.; Italiano, A.; Alexandre, J.; You, B.; Bastian, S.; Krebs, M.G.; et al. Olaparib and durvalumab in patients with germline BRCA-mutated metastatic breast cancer (MEDIOLA): An open-label, multicentre, phase 1/2, basket study. Lancet Oncol. 2020, 21, 1155–1164. [Google Scholar] [CrossRef]
- Rugo, H.S.; Llombart-Cussac, A.; Andre, F.; Robson, M.E.; Saji, S.; Harbeck, N.; Schmid, P.; Cescon, D.W.; Ahn, J.S.; Nanda, R.; et al. KEYLYNK-009: A phase II/III, open-label, randomized study of pembrolizumab (pembro) plus olaparib vs. pembro plus chemotherapy after induction with first-line pembro plus chemotherapy in patients with locally recurrent inoperable or metastatic triple-negative breast cancer (TNBC). J. Clin. Oncol. 2020, 38 (Suppl. 15), TPS596. [Google Scholar]
- Press, M.F.; Pike, M.C.; Chazin, V.R.; Hung, G.; Udove, J.A.; Markowicz, M.; Danyluk, J.; Godolphin, W.; Sliwkowski, M.; Akita, R. Her-2/neu expression in node-negative breast cancer: Direct tissue quantitation by computerized image analysis and association of overexpression with increased risk of recurrent disease. Cancer Res. 1993, 53, 4960–4970. [Google Scholar]
- Musolino, A.; Naldi, N.; Dieci, M.V.; Zanoni, D.; Rimanti, A.; Boggiani, D.; Sgargi, P.; Generali, D.G.; Piacentini, F.; Ambroggi, M.; et al. Immunoglobulin G fragment C receptor polymorphisms and efficacy of preoperative chemotherapy plus trastuzumab and lapatinib in HER2-positive breast cancer. Pharm. J. 2016, 16, 472–477. [Google Scholar] [CrossRef]
- Albanell, J.; Baselga, J. Trastuzumab, a humanized anti-HER2 monoclonal antibody, for the treatment of breast cancer. Drugs Today 1999, 35, 9319–9346. [Google Scholar]
- Slamon, D.J.; Leyland-Jones, B.; Shak, S.; Fuchs, H.; Paton, V.; Bajamonde, A.; Fleming, T.; Eiermann, W.; Wolter, J.; Pegram, M.; et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 2001, 344, 783–792. [Google Scholar] [CrossRef]
- Tóth, G.; Szöőr, Á.; Simon, L.; Yarden, Y.; Szöllősi, J.; Vereb, G. The combination of trastuzumab and pertuzumab administered at approved doses may delay development of trastuzumab resistance by additively enhancing antibody-dependent cell-mediated cytotoxicity. MAbs 2016, 8, 1361–1370. [Google Scholar] [CrossRef]
- Arteaga, C.L.; Winnier, A.R.; Poirier, M.C.; Lopez-Larraza, D.M.; Shawver, L.K.; Hurd, S.D.; Stewart, S.J. p185c-erbB-2 signal enhances cisplatin-induced cytotoxicity in human breast carcinoma cells: Association between an oncogenic receptor tyrosine kinase and drug-induced DNA repair. Cancer Res. 1994, 54, 3758–3765. [Google Scholar] [PubMed]
- Pietras, R.J.; Pegram, M.D.; Finn, R.S.; Maneval, D.A.; Slamon, D.J. Remission of human breast cancer xenografts on therapy with humanized monoclonal antibody to HER-2 receptor and DNA-reactive drugs. Oncogene 1998, 17, 2235–2249. [Google Scholar] [CrossRef] [PubMed]
- Krasniqi, E.; Barchiesi, G.; Pizzuti, L.; Mazzotta, M.; Venuti, A.; Maugeri-Saccà, M.; Sanguineti, G.; Massimiani, G.; Sergi, D.; Carpano, S.; et al. Immunotherapy in HER2-positive breast cancer: State of the art and future perspectives. J. Hematol. Oncol. 2019, 12, 111. [Google Scholar] [CrossRef] [PubMed]
- Gaynor, N.; Crown, J.; Collins, D.M. Immune checkpoint inhibitors: Key trials and an emerging role in breast cancer. Semin. Cancer Biol. 2022, 79, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Baselga, J.; Gelmon, K.A.; Verma, S.; Wardley, A.; Conte, P.; Miles, D.; Bianchi, G.; Cortes, J.; McNally, V.A.; Ros, S.G.A.; et al. Phase II trial of pertuzumab and trastuzumab in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer that progressed during prior trastuzumab therapy. J. Clin. Oncol. 2010, 28, 1138–1144. [Google Scholar] [CrossRef]
- Baselga, J.; Cortés, J.; Kim, S.B.; Im, S.A.; Hegg, R.; Im, Y.H.; Roman, L.; Pedrini, J.L.; Pienkowski, T.; Knott, A.; et al. CLEOPATRA Study Group. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N. Engl. J. Med. 2012, 366, 109–119. [Google Scholar] [CrossRef]
- Swain, S.M.; Kim, S.B.; Cortés, J.; Ro, J.; Semiglazov, V.; Campone, M.; Ciruelos, E.; Ferrero, J.M.; Schneeweiss, A.; Knott, A.; et al. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA study): Overall survival results from a randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2013, 14, 461–471. [Google Scholar] [CrossRef]
- Swain, S.M.; Baselga, J.; Kim, S.B.; Ro, J.; Semiglazov, V.; Campone, M.; Ciruelos, E.; Ferrero, J.M.; Schneeweiss, A.; Heeson, S.; et al. CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N. Engl. J. Med. 2015, 372, 724–734. [Google Scholar] [CrossRef]
- Hurvitz, S.A.; Martin, M.; Symmans, W.F.; Jung, K.H.; Huang, C.S.; Thompson, A.M.; Harbeck, N.; Valero, V.; Stroyakovskiy, D.; Wildiers, H.; et al. Neoadjuvant trastuzumab, pertuzumab, and chemotherapy versus trastuzumab emtansine plus pertuzumab in patients with HER2-positive breast cancer (KRISTINE): A randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2018, 19, 115–126. [Google Scholar] [CrossRef]
- van Ramshorst, M.S.; van der Voort, A.; van Werkhoven, E.D.; Mandjes, I.A.; Kemper, I.; Dezentjé, V.O.; Oving, I.M.; Honkoop, A.H.; Tick, L.W.; van de Wouw, A.J.; et al. Dutch Breast Cancer Research Group (BOOG). Neoadjuvant chemotherapy with or without anthracyclines in the presence of dual HER2 blockade for HER2-positive breast cancer (TRAIN-2): A multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 1630–1640. [Google Scholar] [CrossRef]
- Swain, S.M.; Ewer, M.S.; Viale, G.; Delaloge, S.; Ferrero, J.M.; Verrill, M.; Colomer, R.; Vieira, C.; Werner, T.L.; Douthwaite, H.; et al. BERENICE Study Group. Pertuzumab, trastuzumab, and standard anthracycline- and taxane-based chemotherapy for the neoadjuvant treatment of patients with HER2-positive localized breast cancer (BERENICE): A phase II, open-label, multicenter, multinational cardiac safety study. Ann. Oncol. 2018, 29, 646–653. [Google Scholar] [PubMed]
- Schneeweiss, A.; Chia, S.; Hickish, T.; Harvey, V.; Eniu, A.; Hegg, R.; Tausch, C.; Seo, J.H.; Tsai, Y.F.; Ratnayake, J.; et al. Pertuzumab plus trastuzumab in combination with standard neoadjuvant anthracycline-containing and anthracycline-free chemotherapy regimens in patients with HER2-positive early breast cancer: A randomized phase II cardiac safety study (TRYPHAENA). Ann. Oncol. 2013, 24, 2278–2284. [Google Scholar] [CrossRef] [PubMed]
- Burguin, A.; Diorio, C.; Durocher, F. Breast Cancer Treatments: Updates and New Challenges. J. Pers. Med. 2021, 11, 808. [Google Scholar] [CrossRef]
- Rimawi, M.F.; Schiff, R.; Osborne, C.K. Targeting HER2 for the treatment of breast cancer. Annu. Rev. Med. 2015, 66, 111–128. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, P.; Morganti, S.; Uliano, J.; Giugliano, F.; Crimini, E.; Curigliano, G. Margetuximab for the treatment of HER2-positive metastatic breast cancer. Expert Opin. Biol. Ther. 2021, 21, 127–133. [Google Scholar] [CrossRef]
- Montemurro, F.; Delaloge, S.; Barrios, C.H.; Wuerstlein, R.; Anton, A.; Brain, E.; Hatschek, T.; Kelly, C.M.; Peña-Murillo, C.; Yilmaz, M.; et al. Trastuzumab emtansine (T-DM1) in patients with HER2-positive metastatic breast cancer and brain metastases: Exploratory final analysis of cohort 1 from KAMILLA, a single-arm phase IIIb clinical trial. Ann. Oncol. 2020, 31, 1350–1358. [Google Scholar] [CrossRef]
- Krop, I.E.; Kim, S.B.; Martin, A.G.; LoRusso, P.M.; Ferrero, J.M.; Badovinac-Crnjevic, T.; Hoersch, S.; Smitt, M.; Wildiers, H. Trastuzumab emtansine versus treatment of physician’s choice in patients with previously treated HER2-positive metastatic breast cancer (TH3RESA): Final overall survival results from a randomised open-label phase 3 trial. Lancet Oncol. 2017, 18, 743–754. [Google Scholar] [CrossRef]
- Modi, S.; Saura, C.; Yamashita, T.; Park, Y.H.; Kim, S.B.; Tamura, K.; Andre, F.; Iwata, H.; Ito, Y.; Tsurutani, J.; et al. DESTINY-Breast01 Investigators. Trastuzumab Deruxtecan in Previously Treated HER2-Positive Breast Cancer. N. Engl. J. Med. 2020, 382, 610–621. [Google Scholar] [CrossRef]
- Bardia, A.; Mayer, I.A.; Vahdat, L.T.; Tolaney, S.M.; Isakoff, S.J.; Diamond, J.R.; O’Shaughnessy, J.; Moroose, R.L.; Santin, A.D.; Abramson, V.G.; et al. Sacituzumab Govitecan-hziy in Refractory Metastatic Triple-Negative Breast Cancer. N. Engl. J. Med. 2019, 380, 741–751. [Google Scholar] [CrossRef]
- Howard, F.M.; Villamar, D.; He, G.; Pearson, A.T.; Nanda, R. The emerging role of immune checkpoint inhibitors for the treatment of breast cancer. Expert Opin. Investig. Drugs 2022, 31, 531–548. [Google Scholar] [CrossRef]
- Emens, L.A.; Esteva, F.J.; Beresford, M.; Saura, C.; De Laurentiis, M.; Kim, S.B.; Im, S.A.; Wang, Y.; Salgado, R.; Mani, A.; et al. Trastuzumab emtansine plus atezolizumab versus trastuzumab emtansine plus placebo in previously treated, HER2-positive advanced breast cancer (KATE2): A phase 2, multicentre, randomised, double-blind trial. Lancet Oncol. 2020, 21, 1283–1295. [Google Scholar] [CrossRef]
- Walks, A.G.; Keenan, T.; Li, T.; Tayob, N.; Wulf, G.M.; Richardson, E.T.; Mittendorf, E.A.; Overmoyer, B.; Krop, I.E.; Winer, E.P.; et al. A phase Ib study of pembrolizumab (pembro) plus trastuzumab emtansine (T-DM1) for metastatic HER2+ breast cancer (MBC). J. Clin. Oncol. 2020, 38 (Suppl. 15), 1046. [Google Scholar] [CrossRef]
- Huober, J.; Barrios, C.H.; Niikura, N.; Jarząb, M.; Chang, Y.C.; Huggins-Puhalla, S.L.; Pedrini, J.; Zhukova, L.; Graupner, V.; Eiger, D.; et al. Atezolizumab With Neoadjuvant Anti-Human Epidermal Growth Factor Receptor 2 Therapy and Chemotherapy in Human Epidermal Growth Factor Receptor 2-Positive Early Breast Cancer: Primary Results of the Randomized Phase III IMpassion050 Trial. J. Clin. Oncol. 2022, 40, 2946–2956. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Muul, L.M.; Solomon, D.; Rosenberg, S.A. Expansion of human tumor infiltrating lymphocytes for use in immunotherapy trials. J. Immunol. Methods 1987, 102, 127–141. [Google Scholar] [CrossRef]
- Retecki, K.; Seweryn, M.; Graczyk-Jarzynka, A.; Bajor, M. The Immune Landscape of Breast Cancer: Strategies for Overcoming Immunotherapy Resistance. Cancers 2021, 13, 6012. [Google Scholar] [CrossRef]
- Geller, M.A.; Cooley, S.; Judson, P.L.; Ghebre, R.; Carson, L.F.; Argenta, P.A.; Jonson, A.L.; Panoskaltsis-Mortari, A.; Curtsinger, J.; McKenna, D.; et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy 2011, 13, 98–107. [Google Scholar] [CrossRef]
- Capietto, A.H.; Martinet, L.; Fournié, J.J. Stimulated γδ T cells increase the in vivo efficacy of trastuzumab in HER-2+ breast cancer. J. Immunol. 2011, 187, 1031–1038. [Google Scholar] [CrossRef]
- Wu, Y.; Kyle-Cezar, F.; Woolf, R.T.; Naceur-Lombardelli, C.; Owen, J.; Biswas, D.; Lorenc, A.; Vantourout, P.; Gazinska, P.; Grigoriadis, A.; et al. An innate-like Vδ1+ γδ T cell compartment in the human breast is associated with remission in triple-negative breast cancer. Sci. Transl. Med. 2019, 11, eaax9364. [Google Scholar] [CrossRef]
- Rugo, H.S.; Im, S.A.; Cardoso, F.; Cortés, J.; Curigliano, G.; Musolino, A.; Pegram, M.D.; Wright, G.S.; Saura, C.; Escrivá-de-Romaní, S.; et al. SOPHIA Study Group. Efficacy of Margetuximab vs. Trastuzumab in Patients with Pretreated ERBB2-Positive Advanced Breast Cancer: A Phase 3 Randomized Clinical Trial. JAMA Oncol. 2021, 7, 573–584. [Google Scholar] [CrossRef]
- Nicolini, A.; Carpi, A. Beta-interferon and interleukin-2 prolong more than three times the survival of 26 consecutive endocrine dependent breast cancer patients with distant metastases: An exploratory trial. Biomed. Pharmacother. 2005, 59, 253–263. [Google Scholar] [CrossRef]
- Nicolini, A.; Carpi, A.; Rossi, G. An immunotherapy schedule in endocrine-dependent metastatic breast cancer: Correlation between clinical course and immunologic parameters. J. Immunother. 2005, 28, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Nicolini, A.; Rossi, G.; Ferrari, P.; Morganti, R.; Carpi, A. Final results of a 2:1 control-case observational study using interferon beta and interleukin-2, in addition to first-line hormone therapy, in estrogen receptor-positive, endocrine-responsive metastatic breast cancer patients. J. Cancer Metastasis Treat. 2022, 8, 13. [Google Scholar] [CrossRef]
- Schreiber, R.D.; Old, L.J.; Smyth, M.J. Cancer immunoediting: Integrating immunity’s roles in cancer suppression and promotion. Science 2011, 331, 1565–1570. [Google Scholar] [CrossRef] [PubMed]
- Yarmarkovich, M.; Farrel, A.; Sison, A., 3rd; di Marco, M.; Raman, P.; Parris, J.L.; Monos, D.; Lee, H.; Stevanovic, S.; Maris, J.M. Immunogenicity and Immune Silence in Human Cancer. Front. Immunol. 2020, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; Agostinis, P. Cell death and immunity in cancer: From danger signals to mimicry of pathogen defense responses. Immunol. Rev. 2017, 280, 126–148. [Google Scholar] [CrossRef] [PubMed]
- Oliver, A.J.; Lau, P.K.H.; Unsworth, A.S.; Loi, S.; Darcy, P.K.; Kershaw, M.H.; Slaney, C.Y. Tissue-Dependent Tumor Microenvironments and Their Impact on Immunotherapy Responses. Front. Immunol. 2018, 9, 70. [Google Scholar] [CrossRef]
- Dias, A.S.; Almeida, C.R.; Helguero, L.A.; Duarte, I.F. Metabolic crosstalk in the breast cancer microenvironment. Eur. J. Cancer 2019, 121, 154–171. [Google Scholar] [CrossRef]
- Deepak, K.G.K.; Vempati, R.; Nagaraju, G.P.; Dasari, V.R.; Nagini, S.; Rao, D.N.; Malla, R.R. Tumor microenvironment: Challenges and opportunities in targeting metastasis of triple negative breast cancer. Pharmacol. Res. 2020, 153, 104683. [Google Scholar] [CrossRef]
- Soysal, S.D.; Tzankov, A.; Muenst, S.E. Role of the Tumor Microenvironment in Breast Cancer. Pathobiology 2015, 82, 142–152. [Google Scholar] [CrossRef]
- Mittal, S.; Brown, N.J.; Holen, I. The breast tumor microenvironment: Role in cancer development, progression and response to therapy. Expert Rev. Mol. Diagn. 2018, 18, 227–243. [Google Scholar] [CrossRef]
- DiLillo, D.J.; Yanaba, K.; Tedder, T.F. B cells are required for optimal CD4+ and CD8+ T cell tumor immunity: Therapeutic B cell depletion enhances B16 melanoma growth in mice. J. Immunol. 2010, 184, 4006–4016. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Yu, Y.; Wang, X.; Zhang, T. Tumor-Associated Macrophages in Tumor Immunity. Front. Immunol. 2020, 11, 583084. [Google Scholar] [CrossRef] [PubMed]
- Shaul, M.E.; Fridlender, Z.G. Neutrophils as active regulators of the immune system in the tumor microenvironment. J. Leukoc. Biol. 2017, 102, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Nicolini, A.; Ferrari, P.; Rossi, G.; Carpi, A. Tumour growth and immune evasion as targets for a new strategy in advanced cancer. Endocr. Relat. Cancer 2018, 25, R577–R604. [Google Scholar] [CrossRef]
- Lapeyre-Prost, A.; Terme, M.; Pernot, S.; Pointet, A.L.; Voron, T.; Tartour, E.; Taieb, J. Immunomodulatory Activity of VEGF in Cancer. Int. Rev. Cell. Mol. Biol. 2017, 330, 295–342. [Google Scholar]
- Olkhanud, P.B.; Damdinsuren, B.; Bodogai, M.; Gress, R.E.; Sen, R.; Wejksza, K.; Malchinkhuu, E.; Wersto, R.P.; Biragyn, A. Tumor-evoked regulatory B cells promote breast cancer metastasis by converting resting CD4+ T cells to T-regulatory cells. Cancer Res. 2011, 71, 3505–3515. [Google Scholar] [CrossRef]
- Peng, G.L.; Li, L.; Guo, Y.W.; Yu, P.; Yin, X.J.; Wang, S.; Liu, C.P. CD8+ cytotoxic and FoxP3+ regulatory T lymphocytes serve as prognostic factors in breast cancer. Am. J. Transl. Res. 2019, 11, 5039–5053. [Google Scholar] [PubMed]
- Cendrowicz, E.; Sas, Z.; Bremer, E.; Rygiel, T.P. The Role of Macrophages in Cancer Development and Therapy. Cancers 2021, 13, 1946. [Google Scholar] [CrossRef]
- Curiel, T.J.; Coukos, G.; Zou, L.; Alvarez, X.; Cheng, P.; Mottram, P.; Evdemon-Hogan, M.; Conejo-Garcia, J.R.; Zhang, L.; Burow, M.; et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat. Med. 2004, 10, 942–949. [Google Scholar] [CrossRef]
- Sznol, M.; Chen, L. Antagonist antibodies to PD-1 and B7-H1 (PD-L1) in the treatment of advanced human cancer. Clin. Cancer Res. 2013, 19, 1021–1034. [Google Scholar] [CrossRef]
- Pinton, L.; Solito, S.; Damuzzo, V.; Francescato, S.; Pozzuoli, A.; Berizzi, A.; Mocellin, S.; Rossi, C.R.; Bronte, V.; Mandruzzato, S. Activated T cells sustain myeloid-derived suppressor cell-mediated immune suppression. Oncotarget 2016, 7, 1168–1184. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Wang, Y.; Yan, F.; Zhang, P.; Li, H.; Zhao, H.; Yan, C.; Yan, F.; Ren, X. Noncanonical NF-κB activation mediates STAT3-stimulated IDO upregulation in myeloid-derived suppressor cells in breast cancer. J. Immunol. 2014, 193, 2574–2586. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, K.; Sakurai, M.; Yamamoto, Y.; Suzuki, E.; Tsuda, M.; Kataoka, T.R.; Hirata, M.; Nishie, M.; Nojiri, T.; Kumazoe, M.; et al. Alteration of specific cytokine expression patterns in patients with breast cancer. Sci. Rep. 2019, 9, 2924. [Google Scholar] [CrossRef] [PubMed]
- Ohl, K.; Tenbrock, K. Reactive Oxygen Species as Regulators of MDSC-Mediated Immune Suppression. Front. Immunol. 2018, 9, 2499. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, D.P.; Correia, B.F.; Salvador, R.; de Sousa, N.; Jacinto, A.; Braga, S.; Cabral, M.G. Circulating low density neutrophils of breast cancer patients are associated with their worse prognosis due to the impairment of T cell responses. Oncotarget 2021, 12, 2388–2403. [Google Scholar] [CrossRef]
- Rotondo, R.; Bertolotto, M.; Barisione, G.; Astigiano, S.; Mandruzzato, S.; Ottonello, L.; Dallegri, F.; Bronte, V.; Ferrini, S.; Barbieri, O. Exocytosis of azurophil and arginase 1-containing granules by activated polymorphonuclear neutrophils is required to inhibit T lymphocyte proliferation. J. Leukoc. Biol. 2011, 89, 721–727. [Google Scholar] [CrossRef]
- Fridlender, Z.G.; Sun, J.; Kim, S.; Kapoor, V.; Cheng, G.; Ling, L.; Worthen, G.S.; Albelda, S.M. Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN. Cancer Cell 2009, 16, 183–194. [Google Scholar] [CrossRef]
- Spiegel, A.; Brooks, M.W.; Houshyar, S.; Reinhardt, F.; Ardolino, M.; Fessler, E.; Chen, M.B.; Krall, J.A.; DeCock, J.; Zervantonakis, I.K.; et al. Neutrophils Suppress Intraluminal NK Cell-Mediated Tumor Cell Clearance and Enhance Extravasation of Disseminated Carcinoma Cells. Cancer Discov. 2016, 6, 630–649. [Google Scholar] [CrossRef]
- Ethier, J.L.; Desautels, D.; Templeton, A.; Shah, P.S.; Amir, E. Prognostic role of neutrophil-to-lymphocyte ratio in breast cancer: A systematic review and meta-analysis. Breast Cancer Res. 2017, 19, 2. [Google Scholar] [CrossRef]
- Bun, A.; Fujimoto, Y.; Higuchi, T.; Sata, A.; Fukui, R.; Ozawa, H.; Miyagawa, Y.; Imamura, M.; Watanabe, T.; Miyoshi, Y. Prognostic Significance of Neutrophil-to-lymphocyte Ratio in Luminal Breast Cancers with Low Levels of Tumour-infiltrating Lymphocytes. Anti Cancer Res. 2020, 40, 2871–2880. [Google Scholar] [CrossRef]
- Bruno, A.; Bassani, B.; D’Urso, D.G.; Pitaku, I.; Cassinotti, E.; Pelosi, G.; Boni, L.; Dominioni, L.; Noonan, D.M.; Mortara, L.; et al. Angiogenin and the MMP9-TIMP2 axis are up-regulated in proangiogenic, decidual NK-like cells from patients with colorectal cancer. FASEB J. 2018, 32, 5365–5377. [Google Scholar] [CrossRef] [PubMed]
- Levi, I.; Amsalem, H.; Nissan, A.; Darash-Yahana, M.; Peretz, T.; Mandelboim, O.; Rachmilewitz, J. Characterization of tumor infiltrating natural killer cell subset. Oncotarget 2015, 6, 13835–13843. [Google Scholar] [CrossRef] [PubMed]
- Segovia-Mendoza, M.; Morales-Montor, J. Immune Tumor Microenvironment in Breast Cancer and the Participation of Estrogen and Its Receptors in Cancer Physiopathology. Front. Immunol. 2019, 10, 348. [Google Scholar] [CrossRef] [PubMed]
- Fainaru, O.; Almog, N.; Yung, C.W.; Nakai, K.; Montoya-Zavala, M.; Abdollahi, A.; D’Amato, R.; Ingber, D.E. Tumor growth and angiogenesis are dependent on the presence of immature dendritic cells. FASEB J. 2010, 24, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Sisirak, V.; Faget, J.; Gobert, M.; Goutagny, N.; Vey, N.; Treilleux, I.; Renaudineau, S.; Poyet, G.; Labidi-Galy, S.I.; Goddard-Leon, S.; et al. Impaired IFN-α production by plasmacytoid dendritic cells favors regulatory T-cell expansion that may contribute to breast cancer progression. Cancer Res. 2012, 72, 5188–5197. [Google Scholar] [CrossRef]
- Yao, X.; Tu, Y.; Xu, Y.; Guo, Y.; Yao, F.; Zhang, X. Endoplasmic reticulum stress-induced exosomal miR-27a-3p promotes immune escape in breast cancer via regulating PD-L1 expression in macrophages. J. Cell. Mol. Med. 2020, 24, 9560–9573. [Google Scholar] [CrossRef]
- Sun, Y.; Guo, J.; Yu, L.; Guo, T.; Wang, J.; Wang, X.; Chen, Y. PD-L1+ exosomes from bone marrow-derived cells of tumor-bearing mice inhibit antitumor immunity. Cell. Mol. Immunol. 2021, 18, 2402–2409. [Google Scholar] [CrossRef]
- Chen, G.; Huang, A.C.; Zhang, W.; Zhang, G.; Wu, M.; Xu, W.; Yu, Z.; Yang, J.; Wang, B.; Sun, H.; et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 2018, 560, 382–386. [Google Scholar] [CrossRef]
- Yang, Y.; Li, C.W.; Chan, L.C.; Wei, Y.; Hsu, J.M.; Xia, W.; Cha, J.H.; Hou, J.; Hsu, J.L.; Sun, L.; et al. Exosomal PD-L1 harbors active defense function to suppress T cell killing of breast cancer cells and promote tumor growth. Cell Res. 2018, 28, 862–864. [Google Scholar] [CrossRef]
- Morrissey, S.M.; Zhang, F.; Ding, C.; Montoya-Durango, D.E.; Hu, X.; Yang, C.; Wang, Z.; Yuan, F.; Fox, M.; Zhang, H.G.; et al. Tumor-derived exosomes drive immunosuppressive macrophages in a pre-metastatic niche through glycolytic dominant metabolic reprogramming. Cell Metab. 2021, 33, 2040–2058.e10. [Google Scholar] [CrossRef]
- O’Donnell, J.S.; Teng, M.; Smyth, M.J. Cancer immunoediting and resistance to T cell-based immunotherapy. Nat. Rev. Clin. Oncol. 2019, 16, 151–167. [Google Scholar] [CrossRef] [PubMed]
- Frasor, J.; Stossi, F.; Danes, J.M.; Komm, B.; Lyttle, C.R.; Katzenellenbogen, B.S. Selective estrogen receptor modulators: Discrimination of agonistic versus antagonistic activities by gene expression profiling in breast cancer cells. Cancer Res. 2004, 64, 1522–1533. [Google Scholar] [CrossRef] [PubMed]
- Frasor, J.; Chang, E.C.; Komm, B.; Lin, C.Y.; Vega, V.B.; Liu, E.T.; Miller, L.D.; Smeds, J.; Bergh, J.; Katzenellenbogen, B.S. Gene expression preferentially regulated by tamoxifen in breast cancer cells and correlations with clinical outcome. Cancer Res. 2006, 66, 7334–7340. [Google Scholar] [CrossRef] [PubMed]
- Welte, T.; Zhang, X.H.; Rosen, J.M. Repurposing Antiestrogens for Tumor Immunotherapy. Cancer Discov. 2017, 7, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Nicolini, A.; Rossi, G.; Ferrari, P.; Morganti, R.; Carpi, A. A new immunotherapy schedule in addition to first-line hormone therapy for metastatic breast cancer patients in a state of clinical benefit during hormone therapy. J. Mol. Med. 2020, 98, 375–382. [Google Scholar] [CrossRef]
- Nicolini, A.; Rossi, G.; Ferrari, P.; Carpi, A. Clinical and laboratory patterns during immune stimulation in hormone responsive metastatic breast cancer. Biomed. Pharmacother. 2014, 68, 171–178. [Google Scholar] [CrossRef]
- Nicolini, A.; Rossi, G.; Ferrari, P.; Carpi, A. Minimal residual disease in advanced or metastatic solid cancers: The G0-G1 state and immunotherapy are key to unwinding cancer complexity. Semin. Cancer Biol. 2022, 79, 68–82. [Google Scholar] [CrossRef]
- Nicolini, A.; Ferrari, P.; Morganti, R.; Carpi, A. Treatment of Metastatic or High-Risk Solid Cancer Patients by Targeting the Immune System and/or Tumor Burden: Six Cases Reports. Int. J. Mol. Sci. 2019, 20, 5986. [Google Scholar] [CrossRef]
- Kennecke, H.; Yerushalmi, R.; Woods, R.; Cheang, M.C.; Voduc, D.; Speers, C.H.; Nielsen, T.O.; Gelmon, K. Metastatic behavior of breast cancer subtypes. J. Clin. Oncol. 2010, 28, 3271–3277. [Google Scholar] [CrossRef]
- Buonomo, O.C.; Caredda, E.; Portarena, I.; Vanni, G.; Orlandi, A.; Bagni, C.; Petrella, G.; Palombi, L.; Orsaria, P. New insights into the metastatic behavior after breast cancer surgery, according to well-established clinicopathological variables and molecular subtypes. PLoS ONE 2017, 12, e0184680. [Google Scholar] [CrossRef]
- Female Breast Cancer Subtypes-Cancer Stat Facts. Available online: https://seer.cancer.gov/statfacts/html/breast-subtypes.html (accessed on 1 September 2022).
- Ott, P.A.; Bang, Y.J.; Piha-Paul, S.A.; Razak, A.; Bennouna, J.; Soria, J.C.; Rugo, H.S.; Cohen, R.B.; O’Neil, B.H.; Mehnert, J.M.; et al. T-Cell-Inflamed Gene-Expression Profile, Programmed Death Ligand 1 Expression, and Tumor Mutational Burden Predict Efficacy in Patients Treated With Pembrolizumab Across 20 Cancers: KEYNOTE-028. J. Clin. Oncol. 2019, 37, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhang, J.; Xu, L.; Yang, H.; Liang, N.; Zhang, L.; Zhang, F.; Zhang, X. Safety and Efficacy of the Rechallenge of Immune Checkpoint Inhibitors After Immune-Related Adverse Events in Patients With Cancer: A Systemic Review and Meta-Analysis. Front. Immunol. 2021, 12, 730320. [Google Scholar] [CrossRef] [PubMed]
- von Minckwitz, G.; Procter, M.; de Azambuja, E.; Zardavas, D.; Benyunes, M.; Viale, G.; Suter, T.; Arahmani, A.; Rouchet, N.; Clark, E.; et al. APHINITY Steering Committee and Investigators. Adjuvant Pertuzumab and Trastuzumab in Early HER2-Positive Breast Cancer. N. Engl. J. Med. 2017, 377, 122–131. [Google Scholar] [CrossRef]
- Madariaga, A.; Bowering, V.; Ahrari, S.; Oza, A.M.; Lheureux, S. Manage wisely: Poly (ADP-ribose) polymerase inhibitor (PARPi) treatment and adverse events. Int. J. Gynecol. Cancer 2020, 30, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Goldie, J.H.; Coldman, A.J. A mathematic model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat. Rep. 1979, 63, 1727–1733. [Google Scholar] [PubMed]
- Goldie, J.H.; Coldman, A.J. Quantitative model for multiple levels of drug resistance in clinical tumors. Cancer Treat. Rep. 1983, 67, 923–931. [Google Scholar]
- Irurzun-Arana, I.; McDonald, T.O.; Trocóniz, I.F.; Michor, F. Pharmacokinetic Profiles Determine Optimal Combination Treatment Schedules in Computational Models of Drug Resistance. Cancer Res. 2020, 80, 3372–3382. [Google Scholar] [CrossRef]
- Kıbış, E.Y.; Büyüktahtakın, I.E. Optimizing multi-modal cancer treatment under 3D spatio-temporal tumor growth. Math. Biosci. 2019, 307, 53–69. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, H.; Song, X.; Yang, Q. Metastatic heterogeneity of breast cancer: Molecular mechanism and potential therapeutic targets. Semin. Cancer Biol. 2020, 60, 14–27. [Google Scholar] [CrossRef]
- Kaplan, R.N.; Riba, R.D.; Zacharoulis, S.; Bramley, A.H.; Vincent, L.; Costa, C.; MacDonald, D.D.; Jin, D.K.; Shido, K.; Kerns, S.A.; et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 2005, 438, 820–827. [Google Scholar] [CrossRef]
- Peinado, H.; Zhang, H.; Matei, I.R.; Costa-Silva, B.; Hoshino, A.; Rodrigues, G.; Psaila, B.; Kaplan, R.N.; Bromberg, J.F.; Kang, Y.; et al. Pre-metastatic niches: Organ-specific homes for metastases. Nat. Rev. Cancer 2017, 17, 302–317. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cao, X. Characteristics and Significance of the Pre-metastatic Niche. Cancer Cell 2016, 30, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Fertig, E.J.; Jin, K.; Sukumar, S.; Pandey, N.B.; Popel, A.S. Breast cancer cells condition lymphatic endothelial cells within pre-metastatic niches to promote metastasis. Nat. Commun. 2014, 5, 4715. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, R.; Paredes, J.; Ribeiro, A.S. Impact of breast cancer cells’ secretome on the brain metastatic niche remodeling. Semin. Cancer Biol. 2020, 60, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Che, S.P.Y.; Park, J.Y.; Stokol, T. Tissue Factor-Expressing Tumor-Derived Extracellular Vesicles Activate Quiescent Endothelial Cells via Protease-Activated Receptor-1. Front. Oncol. 2017, 7, 261. [Google Scholar] [CrossRef]
- Ham, S.; Lima, L.G.; Chai, E.P.Z.; Muller, A.; Lobb, R.J.; Krumeich, S.; Wen, S.W.; Wiegmans, A.P.; Möller, A. Breast Cancer-Derived Exosomes Alter Macrophage Polarization via gp130/STAT3 Signaling. Front. Immunol. 2018, 9, 871. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, L.; Wang, T.; Li, Y.; Xun, Q.; Zhang, R.; Liu, L.; Li, L.; Wang, W.; Tian, Y.; et al. Tumor cell-secreted exosomal miR-22-3p inhibits transgelin and induces vascular abnormalization to promote tumor budding. Mol. Ther. 2021, 29, 2151–2166. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Hagiwara, K.; Yoshioka, Y.; Takeshita, F.; Ochiya, T. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J. Biol. Chem. 2013, 288, 10849–10859. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Yoshioka, Y.; Takeshita, F.; Matsuki, Y.; Ochiya, T. Secretory mechanisms and intercellular transfer of microRNAs in living cells. J. Biol. Chem. 2010, 285, 17442–17452. [Google Scholar] [CrossRef]
- Nogués, L.; Benito-Martin, A.; Hergueta-Redondo, M.; Peinado, H. The influence of tumour-derived extracellular vesicles on local and distal metastatic dissemination. Mol. Asp. Med. 2018, 60, 15–26. [Google Scholar] [CrossRef]
- Olejarz, W.; Kubiak-Tomaszewska, G.; Chrzanowska, A.; Lorenc, T. Exosomes in Angiogenesis and Anti-angiogenic Therapy in Cancers. Int. J. Mol. Sci. 2020, 21, 5840. [Google Scholar] [CrossRef] [PubMed]
- Webber, J.; Steadman, R.; Mason, M.D.; Tabi, Z.; Clayton, A. Cancer exosomes trigger fibroblast to myofibroblast differentiation. Cancer Res. 2010, 70, 9621–9630. [Google Scholar] [CrossRef] [PubMed]
- Baroni, S.; Romero-Cordoba, S.; Plantamura, I.; Dugo, M.; D’Ippolito, E.; Cataldo, A.; Cosentino, G.; Angeloni, V.; Rossini, A.; Daidone, M.G.; et al. Exosome-mediated delivery of miR-9 induces cancer-associated fibroblast-like properties in human breast fibroblasts. Cell Death Dis. 2016, 7, e2312. [Google Scholar] [CrossRef]
- Scognamiglio, I.; Cocca, L.; Puoti, I.; Palma, F.; Ingenito, F.; Quintavalle, C.; Affinito, A.; Roscigno, G.; Nuzzo, S.; Chianese, R.V.; et al. Exosomal microRNAs synergistically trigger stromal fibroblasts in breast cancer. Mol. Ther. Nucleic Acids 2022, 28, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Sosa, M.S.; Bragado, P.; Aguirre-Ghiso, J.A. Mechanisms of disseminated cancer cell dormancy: An awakening field. Nat. Rev. Cancer 2014, 14, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Nicolini, A.; Conte, M.; Rossi, G.; Ferrari, P.; Carpi, A.; Miccoli, P. A new pharmacological approach to gastrointestinal cancer at high risk of relapse based on maintenance of the cytostatic effect. Tumour. Biol. 2010, 31, 523–532. [Google Scholar] [CrossRef]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).